Hypertrophic Cardiomyopathy and Chronic Kidney Disease: An Updated Review
Abstract
1. Introduction
2. What Is Hypertrophic Cardiomyopathy?
2.1. Classification of Hypertrophic Cardiomyopathy
2.2. Genetics of Hypertrophic Cardiomyopathy
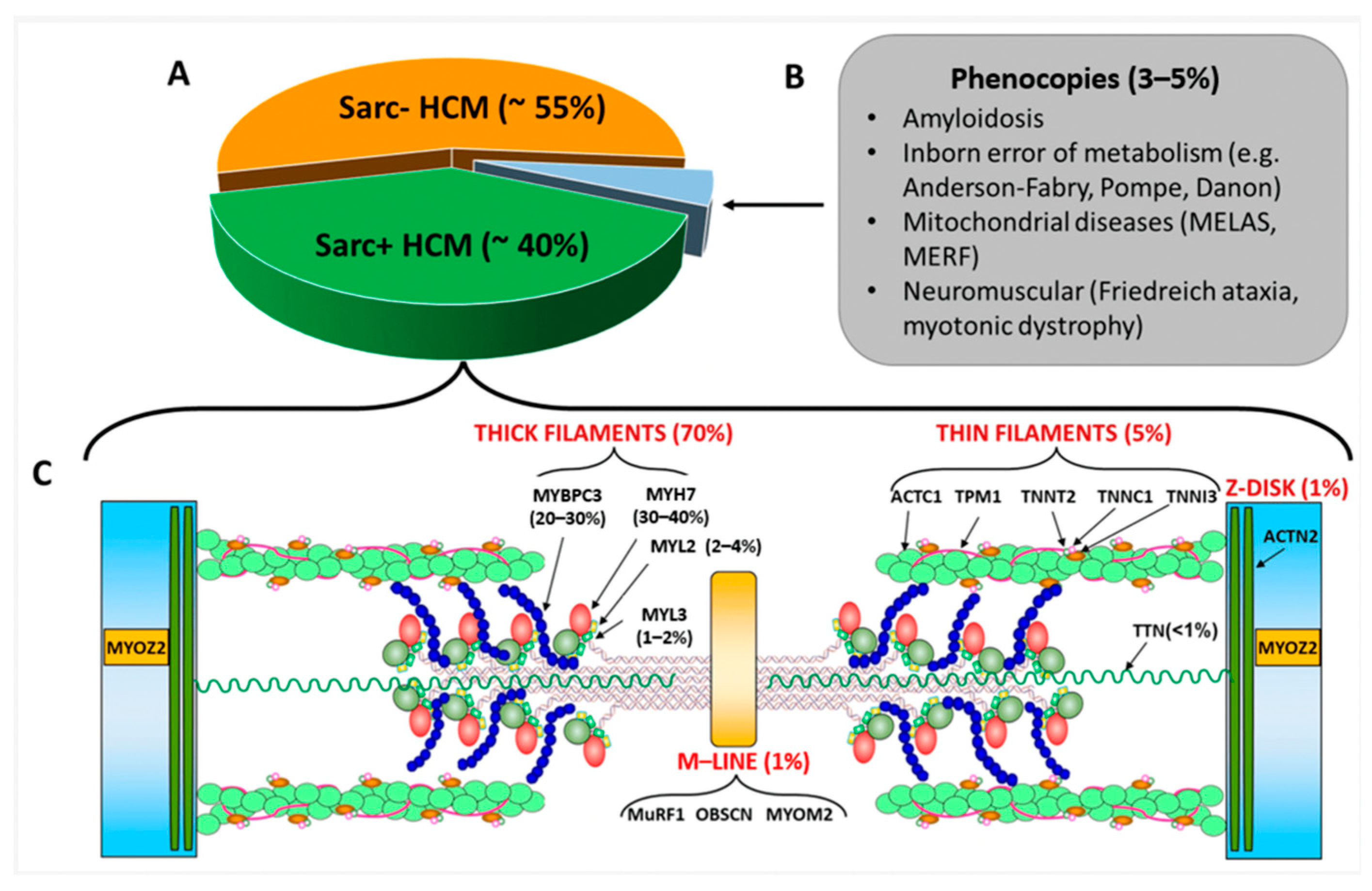
2.3. Mimics of Hypertrophic Cardiomyopathy
2.4. Diagnosing Hypertrophic Cardiomyopathy
3. Association between Hypertrophic Cardiomyopathy and Chronic Kidney Disease
4. Renal Dysfunction as a Poor Prognostic Indicator in Hypertrophic Cardiomyopathy
5. Management of Hypertrophic Cardiomyopathy in Chronic Kidney Disease
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, J.; Floege, J.; Fliser, D.; Bohm, M.; Marx, N. Cardiovascular Disease in Chronic Kidney Disease. Circulation 2021, 143, 1157–1172. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Epidemiology of chronic kidney disease: An update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Van Biesen, W.; De Bacquer, D.; Verbeke, F.; Delanghe, J.; Lameire, N.; Vanholder, R. The glomerular filtration rate in an apparently healthy population and its relation with cardiovascular mortality during 10 years. Eur. Heart J. 2007, 28, 478–483. [Google Scholar] [CrossRef] [PubMed]
- McKenna, W.J.; Judge, D.P. Epidemiology of the inherited cardiomyopathies. Nat. Rev. Cardiol. 2021, 18, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Richard, P.; Charron, P.; Carrier, L.; Ledeuil, C.; Cheav, T.; Pichereau, C.; Benaiche, A.; Isnard, R.; Dubourg, O.; Burban, M.; et al. Hypertrophic cardiomyopathy: Distribution of disease genes, spectrum of mutations, and implications for a molecular diagnosis strategy. Circulation 2003, 107, 2227–2232. [Google Scholar] [CrossRef] [PubMed]
- Elliott, P.; Andersson, B.; Arbustini, E.; Bilinska, Z.; Cecchi, F.; Charron, P.; Dubourg, O.; Kühl, U.; Maisch, B.; McKenna, W.J.; et al. Classification of the cardiomyopathies: A position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2008, 29, 270–276. [Google Scholar] [CrossRef]
- Sankaranarayanan, R.; Fleming, E.J.; Garratt, C.J. Mimics of Hypertrophic Cardiomyopathy—Diagnostic Clues to Aid Early Identification of Phenocopies. Arrhythmia Electrophysiol. Rev. 2013, 2, 36–40. [Google Scholar] [CrossRef]
- Raj, M.A.; Ranka, S.; Goyal, A. Hypertrophic Obstructive Cardiomyopathy; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Prinz, C.; Farr, M.; Hering, D.; Horstkotte, D.; Faber, L. The diagnosis and treatment of hypertrophic cardiomyopathy. Dtsch. Arztebl. Int. 2011, 108, 209–215. [Google Scholar] [CrossRef]
- Albakri, A. Hypertrophic cardiomyopathy: A review of literature on clinical status and meta-analysis of diagnosis and clinical management methods. Clin. Med. Investig. 2018, 3, 3–16. [Google Scholar] [CrossRef]
- Wigle, E.D.; Rakowski, H.; Kimball, B.P.; Williams, W.G. Hypertrophic cardiomyopathy. Clinical spectrum and treatment. Circulation 1995, 92, 1680–1692. [Google Scholar] [CrossRef]
- Todde, G.; Canciello, G.; Borrelli, F.; Perillo, E.F.; Esposito, G.; Lombardi, R.; Losi, M.A. Diagnosis and Treatment of Obstructive Hypertrophic Cardiomyopathy. Cardiogenetics 2023, 13, 75–91. [Google Scholar] [CrossRef]
- European Society of Cardiology. Available online: https://www.escardio.org/Councils/Council-on-Cardiovascular-Genomics/Cardiovascular-Genomics-Insight/Volume-1/how-to-measure-intraventricular-obstruction-in-hypertrophic-cardiomyopathy (accessed on 10 December 2023).
- Jordà, P.; García-Álvarez, A. Hypertrophic cardiomyopathy: Sudden cardiac death risk stratification in adults. Glob. Cardiol. Sci. Pract. 2018, 2018, 25. [Google Scholar] [CrossRef] [PubMed]
- Pelliccia, F.; Pasceri, V.; Limongelli, G.; Autore, C.; Basso, C.; Corrado, D.; Imazio, M.; Rapezzi, C.; Sinagra, G.; Mercuro, G.; et al. Long-term outcome of nonobstructive versus obstructive hypertrophic cardiomyopathy: A systematic review and meta-analysis. Int. J. Cardiol. 2017, 243, 379–384. [Google Scholar] [CrossRef]
- Pozios, I.; Corona-Villalobos, C.; Sorensen, L.L.; Bravo, P.E.; Canepa, M.; Pisanello, C.; Pinheiro, A.; Dimaano, V.L.; Luo, H.; Dardari, Z.; et al. Comparison of Outcomes in Patients With Nonobstructive, Labile-Obstructive, and Chronically Obstructive Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2015, 116, 938–944. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, O. Direct Sarcomere Modulators Are Promising New Treatments for Cardiomyopathies. Int. J. Mol. Sci. 2020, 21, 226. [Google Scholar] [CrossRef]
- Geisterfer-Lowrance, A.A.; Kass, S.; Tanigawa, G.; Vosberg, H.P.; McKenna, W.; Seidman, C.E.; Seidman, J.G. A molecular basis for familial hypertrophic cardiomyopathy: A beta cardiac myosin heavy chain gene missense mutation. Cell 1990, 62, 999–1006. [Google Scholar] [CrossRef]
- Marian, A.J. Molecular Genetic Basis of Hypertrophic Cardiomyopathy. Circulation 2021, 128, 1533–1553. [Google Scholar] [CrossRef]
- Alfares, A.; Kelly, M.; McDermott, G.; Funke, B.H.; Lebo, M.S.; Baxter, S.B.; Shen, J.; McLaughlin, H.M.; Clark, E.H.; Babb, L.J.; et al. Results of clinical genetic testing of 2912 probands with hypertrophic cardiomyopathy: Expanded panels offer limited additional sensitivity. Genet. Med. 2015, 17, 880–888. [Google Scholar] [CrossRef]
- Ho, C.Y.; Day, S.M.; Ashley, E.A.; Michels, M.; Pereira, A.C.; Jacoby, D.; Cirino, A.L.; Fox, J.C.; Lakdawala, N.K.; Ware, J.S.; et al. Genotype and Lifetime Burden of Disease in Hypertrophic Cardiomyopathy: Insights from the Sarcomeric Human Cardiomyopathy Registry (SHaRe). Circulation 2018, 138, 1387–1398. [Google Scholar] [CrossRef]
- Borrelli, F.; Losi, M.A.; Canciello, G.; Todde, G.; Perillo, E.F.; Ordine, L.; Frisso, G.; Esposito, G.; Lombardi, R. Sarcomeric versus Non-Sarcomeric HCM. Cardiogenetics 2023, 13, 92–105. [Google Scholar] [CrossRef]
- Rowin, E.J.; Maron, M.S.; Chan, R.H.; Hausvater, A.; Wang, W.; Rastegar, H.; Maron, B.J. Interaction of Adverse Disease Related Pathways in Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2017, 120, 2256–2264. [Google Scholar] [CrossRef]
- Rowin, E.; Maron, B.; Carrick, R.; Patel, P.P.; Koethe, B.; Wells, S.; Maron, M.S. Outcomes in Patients With Hypertrophic Cardiomyopathy and Left Ventricular Systolic Dysfunction. J. Am. Coll. Cardiol. 2020, 75, 3033–3043. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.R. Left ventricular hypertrophy: Its Prevalence, Etiology, and Significance. Clin. Cardiol. 1991, 14, 13–17. [Google Scholar] [CrossRef]
- Schirmer, H.; Lunde, P.; Rasmussen, K. Prevalence of left ventricular hypertrophy in a general population; The Tromsø Study. Eur. Heart J. 1999, 20, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, A.B.; Rao, S.S.; Marwaha, K. Left Ventricular Hypertrophy; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Ruilope, L.M.; Schmieder, R.E. Left Ventricular Hypertrophy and Clinical Outcomes in Hypertensive Patients. Am. J. Hypertens. 2008, 21, 500–508. [Google Scholar] [CrossRef]
- Maron, B.J.; Pelliccia, A.; Spirito, P. Cardiac disease in young trained athletes. Insights into methods for distinguishing athlete’s heart from structural heart disease, with particular emphasis on hypertrophic cardiomyopathy. Circulation 1995, 91, 1596–1601. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.C.; Rohan, S.; Dastidar, A.G.; Burchell, A.E.; Ratcliffe, L.E.; Hart, E.C.; Paton, J.F.; Hamilton, M.; Nightingale, A.K.; Manghat, N.E. Hypertensive heart disease versus hypertrophic cardiomyopathy: Multi-parametric CMR predictors beyond end-diastolic wall thickness ≥15 mm. J. Cardiovasc. Magn. Reason. 2016, 18, 264. [Google Scholar] [CrossRef]
- Rapezzi, C.; Arbustini, E.; Caforio, A.L.; Charron, P.; Gimeno-Blanes, J.; Heliö, T.; Linhart, A.; Mogensen, J.; Pinto, Y.; Ristic, A.; et al. Diagnostic work-up in cardiomyopathies: Bridging the gap between clinical phenotypes and final diagnosis. A position statement from the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013, 34, 1448–1458. [Google Scholar] [CrossRef]
- Arbelo, E.; Protonotarios, A.; Gimeno, J.R.; Arbustini, E.; Barriales-Villa, R.; Basso, C.; Bezzina, C.R.; Biagini, E.; Blom, N.A.; de Boer, R.A.; et al. 2023 ESC Guidelines for the management of cardiomyopathies. Eur. Heart J. 2023, 44, 3503–3626. [Google Scholar] [CrossRef]
- Mayala, H.A.; Bakari, K.H.; Wang, Z. The role of cardiac magnetic resonance (CMR) in the diagnosis of cardiomyopathy: A systematic review. Malawi Med. J. 2018, 30, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Mitevksa, I.P. Focus on echocardiography in hypertrophic cardiomyopathy—Fourth in series. E-J. Counc. Cardiol Pract. 2015, 13, 20. [Google Scholar]
- Brenes, J.C.; Doltra, A.; Prat, S. Cardiac magnetic resonance imaging in the evaluation of patients with hypertrophic cardiomyopathy. Glob. Cardiol. Sci. Pract. 2018, 2018, 22. [Google Scholar] [CrossRef]
- Sivalokanathan, S. The Role of Cardiovascular Magnetic Resonance Imaging in the Evaluation of Hypertrophic Cardiomyopathy. Diagnostics 2022, 12, 314. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Han, K.; Park, J.B.; Hwang, I.C.; Yoon, Y.E.; Park, H.E.; Choi, S.Y.; Kim, Y.J.; Cho, G.Y.; Kim, H.K.; et al. Risk of end-stage renal disease in patients with hypertrophic cardiomyopathy: A nationwide population-based cohort study. Sci. Rep. 2019, 9, 14565. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.Y.; Zhang, J.L.; Huang, B.T.; Peng, Y.; Chen, S.J.; Chen, M. Renal function as a predictor of outcomes in patients with hypertrophic cardiomyopathy: A cohort study of a hospitalized population. Clin. Chim. Acta Int. J. Clin. Chem. 2021, 512, 92–99. [Google Scholar] [CrossRef]
- Higuchi, S.; Minami, Y.; Shoda, M.; Shirotani, S.; Kanai, M.; Kataoka, S.; Yazaki, K.; Saito, C.; Haruki, S.; Yagishita, D.; et al. Effect of Renal Dysfunction on Risk of Sudden Cardiac Death in Patients With Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2021, 144, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, S.; Kagoshima, T.; Hashimoto, T.; Nakajima, T.; Dohi, K. Left ventricular diastolic function in patients on maintenance hemodialysis: Comparison with hypertensive heart disease and hypertrophic cardiomyopathy. Clin. Nephrol. 1994, 42, 109–116. [Google Scholar]
- Parfrey, P.S.; Griffiths, S.M.; Harnett, J.D.; Taylor, R.; King, A.; Hand, J.; Barre, P.E. Outcome of congestive heart failure, dilated cardiomyopathy, hypertrophic hyperkinetic disease, and ischemic heart disease in dialysis patients. Am. J. Nephrol. 1990, 10, 213–221. [Google Scholar] [CrossRef]
- Klein, J.; McLeish, K.; Hodsden, J.; Lordon, R. Hypertrophic cardiomyopathy: An acquired disorder of end–stage renal disease. Trans. Am. Soc. Artif. Intern. Organs 1983, 29, 120–123. [Google Scholar]
- Drukker, A.; Urbach, J.; Glaser, J. Hypertrophic cardiomyopathy in children with end-stage renal disease and hypertension. Proc. Eur. Dial. Transpl. Assoc. 1981, 18, 542–547. [Google Scholar]
- Whelton, P.K.; Watson, A.J.; Kone, B.; Fortuin, N.J. Calcium channel blockade in dialysis patients with left ventricular hypertrophy and well-preserved systolic function. J. Cardiovasc. Pharmacol. 1987, 10, 185–186. [Google Scholar] [CrossRef]
- Timio, M.; Martini, F.; Venanzi, S.; Ronconi, M.; Lippi, G.; Pippi, C. La funzione ventricolare sinistra nei pazienti in trattamento dialitico peritoneale [Left ventricular function in patients under peritoneal dialysis treatment]. G. Ital. Di Cardiol. 1984, 14, 570–576. [Google Scholar]
- Verma, A.; Anavekar, N.S.; Meris, A.; Thune, J.J.; Arnold, J.M.; Ghali, J.K.; Velazquez, E.J.; McMurray, J.J.; Pfeffer, M.A.; Solomon, S.D. The relationship between renal function and cardiac structure, function, and prognosis after myocardial infarction: The VALIANT Echo Study. J. Am. Coll. Cardiol. 2007, 50, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Nampoothiri, S.; Yesodharan, D.; Bhattacherjee, A.; Ahamed, H.; Puri, R.D.; Gupta, N.; Kabra, M.; Ranganath, P.; Bhat, M.; Phadke, S.; et al. Fabry disease in India: A multicenter study of the clinical and mutation spectrum in 54 patients. JIMD Rep. 2020, 56, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Jahan, S.; Sarathchandran, S.; Akhter, S.; Goldblatt, J.; Stark, S.; Crawford, D.; Mallett, A.; Thomas, M. Prevalence of Fabry disease in dialysis patients: Western Australia Fabry disease screening study—The FoRWARD study. Orphanet J. Rare Dis. 2020, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.Y.; You, Z.H.; Tsai, S.F.; Wu, M.J.; Yu, T.M.; Chuang, Y.W.; Chen, C.H. Diagnosis and Management of Fabry Disease in High-Risk Renal Disease Patients in Taiwan: A Single Center Study. Transplant. Proc. 2023, 55, 788–791. [Google Scholar] [CrossRef]
- Tonelli, M.; Wiebe, N.; Culleton, B.; House, A.; Rabbat, C.; Fok, M.; McAlister, F.; Garg, A. Chronic Kidney Disease and Mortality Risk: A Systematic Review. J. Am. Soc. Nephrol. 2006, 17, 2034–2047. [Google Scholar] [CrossRef]
- Izumaru, K.; Hata, J.; Nakano, T.; Nakashima, Y.; Nagata, M.; Fukuhara, M.; Oda, Y.; Kitazono, T.; Ninomiya, T. Reduced Estimated GFR and Cardiac Remodeling: A Population-Based Autopsy Study. Am. J. Kidney Dis. 2019, 74, 373–381. [Google Scholar] [CrossRef]
- Moran, A.; Katz, R.; Jenny, N.S.; Astor, B.; Bluemke, D.A.; Lima, J.A.; Siscovick, D.; Bertoni, A.G.; Shlipak, M.G. Left ventricular hypertrophy in mild and moderate reduction in kidney function determined using cardiac magnetic resonance imaging and cystatin C: The multi-ethnic study of atherosclerosis (MESA). Am. J. Kidney Dis. 2008, 52, 839–848. [Google Scholar] [CrossRef]
- Mace, H.; Rizwan, A.; Lutz, W.; Hamid, A.; Campbell, W.F.; McMullan, M.R.; Hall, M.E. Hypertension in patients with hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 2023, 81, 1605. [Google Scholar] [CrossRef]
- UpToDate. Available online: https://www.uptodate.com/contents/hypertrophic-cardiomyopathy-management-of-patients-with-outflow-tract-obstruction?search=hcm%20management (accessed on 11 December 2023).
- UpToDate. Available online: https://www.uptodate.com/contents/hypertrophic-cardiomyopathy-management-of-patients-without-outflow-tract-obstruction?search=hcm%20management&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1 (accessed on 11 December 2023).
- UpToDate. Available online: https://www.uptodate.com/contents/renal-effects-of-ace-inhibitors-in-heart-failure (accessed on 11 December 2023).
- Singhania, N.; Bansal, S.; Mohandas, S.; Nimmatoori, D.P.; Ejaz, A.A.; Singhania, G. Role of renin-angiotensin-aldosterone system inhibitors in heart failure and chronic kidney disease. Drugs Context 2020, 9. [Google Scholar] [CrossRef]
- Cho, I.J.; Kang, S.M. Angiotensin receptor-neprilysin inhibitor in patients with heart failure and chronic kidney disease. Kidney Res. Clin. Pract. 2021, 40, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.R.; Lakkis, J.I.; Jaar, B.; Rocco, M.V.; Choi, M.J.; Kramer, H.J.; Ku, E. Use of Renin-Angiotensin System Blockade in Advanced CKD: An NKF-KDOQI Controversies Report. Am. J. Kidney Dis. 2018, 72, 873–884. [Google Scholar] [CrossRef]
- Walther, C.P.; Winkelmayer, W.C.; Richardson, P.A.; Virani, S.S.; Navaneethan, S.D. Renin-angiotensin system blocker discontinuation and adverse outcomes in chronic kidney disease. Nephrol. Dial. Transplant. 2021, 36, 1893–1899. [Google Scholar] [CrossRef] [PubMed]
- Mukoyama, M.; Kuwabara, T. Role of renin-angiotensin system blockade in advanced CKD: To use or not to use? Hypertens. Res. 2022, 45, 1072–1075. [Google Scholar] [CrossRef]
- Van der Aart-van der Beek, A.B.; de Boer, R.A.; Heerspink, H.J.L. Kidney and heart failure outcomes associated with SGLT2 inhibitor use. Nat. Rev. Nephrol. 2022, 18, 294–306. [Google Scholar] [CrossRef]
- McCullough, P.A.; Chan, C.T.; Weinhandl, E.D.; Burkart, J.M.; Bakris, G.L. Intensive Hemodialysis, Left Ventricular Hypertrophy, and Cardiovascular Disease. Am. J. Kidney Dis. 2016, 68, 5–14. [Google Scholar] [CrossRef]
| Study | Population | Findings | Comments |
|---|---|---|---|
| Risk of ESKD in patients with hypertrophic cardiomyopathy: A nationwide population-based cohort study [38]. | A total of 10,300 adult patients with HCM and 51,500 age- and sex-matched controls | (1) The incidence of ESKD was higher in the HCM (1.08%) vs. non-HCM group (0.17%) despite a longer follow up of the non-HCM group (2) When adjusted for generally accepted prognosticators, HCM was found to be an independent predictor of ESKD (3) The risk of developing ESKD was higher in younger and healthier men | Generally accepted prognosticators included age, hypertension, DM, pre-existing renal disease, and prior use of renin–angiotensin system blockers. HCM was defined using the International Classification of Disease, 10th Revision codes, which was validated in a previous study by reviewing medical records, echocardiography, or cardiac magnetic resonance imaging. |
| Renal function as a predictor of outcomes in patients with hypertrophic cardiomyopathy: A cohort study of a hospitalized population [39]. | A total of 581 patients with hypertrophic cardiomyopathy | (1) 15% of patients with HCM had moderate to severe renal dysfunction (2) eGFR was strongly associated with prognosis in patients with HCM. All-cause mortality risk was higher in patients with moderate to severe renal dysfunction (3) The correlation between renal dysfunction and all-cause mortality was higher in patients below the age of 55 | Renal dysfunction was defined as eGFR < 60 mL/min/1.73 m2. HCM was determined using echocardiography findings of left ventricular wall thickness ≥15 mm in adults in the absence of any cardiac or systemic etiology that could explain hypertrophy. |
| Effect of Renal Dysfunction on Risk of Sudden Cardiac Death in Patients with Hypertrophic Cardiomyopathy [40]. | A total of 450 patients with HCM | Patients with renal dysfunction were at a higher risk of sudden death than patients without renal dysfunction | Renal dysfunction was defined as an eGFR < 60 mL/min/1.73 m2. |
| Left ventricular diastolic function in patients on maintenance haemodialysis: comparison with hypertensive heart disease and hypertrophic cardiomyopathy [41]. | A total of 20 patients on maintenance hemodialysis, 11 patients with hypertensive heart disease, 11 with hypertrophic cardiomyopathy, and 11 age-matched healthy individuals | Patients on hemodialysis were found to have a left ventricular diastolic dysfunction similar to that observed in patients with hypertensive heart disease but which was less severe than that found in patients with HCM | Small sample size Comparative study |
| Outcome of Congestive Heart Failure, Dilated Cardiomyopathy, Hypertrophic Hyperkinetic Disease, and ischemic heart disease in Dialysis Patients [42]. | A total of 150 dialysis patients | Survival was worse in patients with an echocardiographic diagnosis of dilated cardiomyopathy (67% 2-year survival rate) compared to patients with normal echocardiogram findings (2-year survival rate, 90%). In hypertrophic hyperkinetic cardiomyopathy, the 2-year survival rate was 30% after entry into the study, and 43% after the first congestive heart failure episode | Cohort study |
| Hypertrophic cardiomyopathy: an acquired disorder of ESKD [43]. | A total of 52 patients on hemodialysis | (1) The incidence of asymmetric septal hypertrophy (ASH) of the heart was 9.6% (2) Patients with ASH were found to have a higher number of hypotensive episodes on dialysis (3) In one patient, ASH was shown to develop over time | HCM is defined by the presence of ASH. Small sample size Comparative study |
| Hypertrophic cardiomyopathy in children with ESKD and hypertension [44]. | A total of 10 pediatric patients on maintenance dialysis | Four out of ten patients showed asymmetric septal hypertrophy typical of hypertrophic cardiomyopathy (HCM) findings on echocardiogram. Two patients also had signs suggestive of left ventricular outflow obstruction | Patients had moderate to severe hypertension despite intensive medical therapy. Primary cardiomyopathy was excluded by negative family history. Case series |
| Calcium Channel Blockade in Dialysis Patients with Left Ventricular Hypertrophy and Well-Preserved Systolic Function [45]. | Four patients on hemodialysis with persistent heart failure and recurrent unexplained hemodialysis-related hypotensive episodes | (1) A subset of patients with refractory heart failure and hypotension complications on dialysis have HCM with preserved systolic function (2) Verapamil may benefit these patients | Small sample size LVH and HCM are used interchangeably. |
| Left ventricular function in patients under peritoneal dialysis treatment [46]. | A total of 24 patients with ESKD and LVH before and after peritoneal dialysis | (1) Before dialysis, 9 patients showed features of dilated cardiomyopathy on echocardiogram (group A), 7 patients showed features of asymmetric septal hypertrophy (group B), and 8 patients had non-specific signs of myocardium involvement (group C) (2) After initiation of peritoneal dialysis, patients in group B showed a reduction in asymmetric septal hypertrophy | Small sample size ASH is referring to HCM. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhuny, S.; Wu, H.H.L.; David, M.; Chinnadurai, R. Hypertrophic Cardiomyopathy and Chronic Kidney Disease: An Updated Review. Cardiogenetics 2024, 14, 26-37. https://doi.org/10.3390/cardiogenetics14010002
Dhuny S, Wu HHL, David M, Chinnadurai R. Hypertrophic Cardiomyopathy and Chronic Kidney Disease: An Updated Review. Cardiogenetics. 2024; 14(1):26-37. https://doi.org/10.3390/cardiogenetics14010002
Chicago/Turabian StyleDhuny, Sheefah, Henry H. L. Wu, Manova David, and Rajkumar Chinnadurai. 2024. "Hypertrophic Cardiomyopathy and Chronic Kidney Disease: An Updated Review" Cardiogenetics 14, no. 1: 26-37. https://doi.org/10.3390/cardiogenetics14010002
APA StyleDhuny, S., Wu, H. H. L., David, M., & Chinnadurai, R. (2024). Hypertrophic Cardiomyopathy and Chronic Kidney Disease: An Updated Review. Cardiogenetics, 14(1), 26-37. https://doi.org/10.3390/cardiogenetics14010002







