Could the BGN Gene Be Pathogenic with Spontaneous Coronary Artery Dissection (SCAD) and Fibromuscular Dysplasia (FMD)?
Abstract
1. Introduction
2. Materials and Methods
| ACTA2 |
| BGN |
| CBS |
| COL3A1 |
| COL5A1 |
| COL5A2 |
| EFEMP2 |
| FBN1 |
| FBN2 |
| FLNA |
| FOXE3 |
| LOX |
| MED12 |
| MFAP5 |
| MYH11 |
| MYLK |
| NOTCH1 |
| PLOD1 |
| PRKG1 |
| SKI NM_ |
| SLC2A10 |
| SMAD2 |
| SMAD3 |
| SMAD4 |
| TGFB2 |
| TGFB3 |
| TGFBR1 |
| TGFBR2 NM_003242.5 |
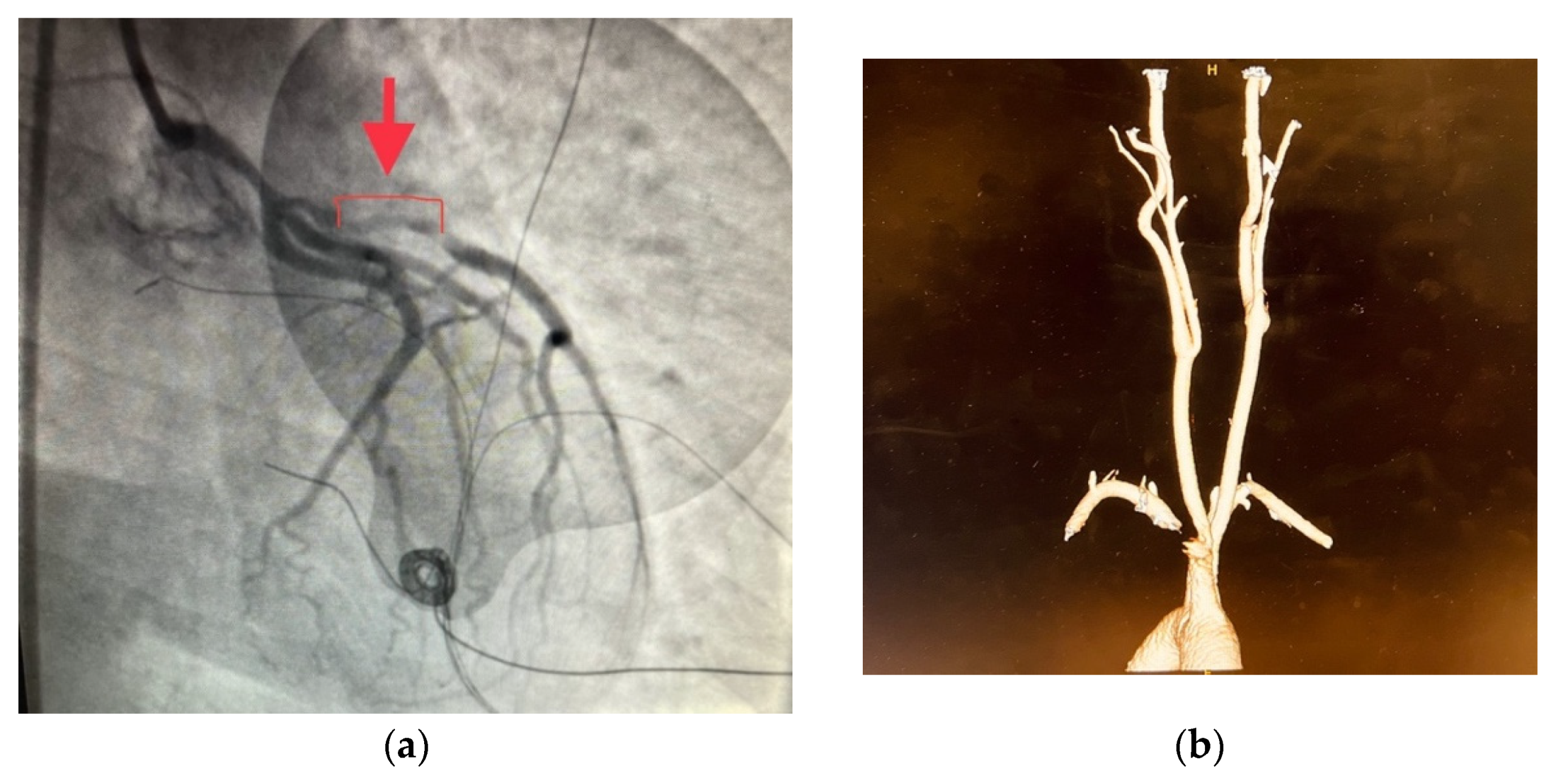
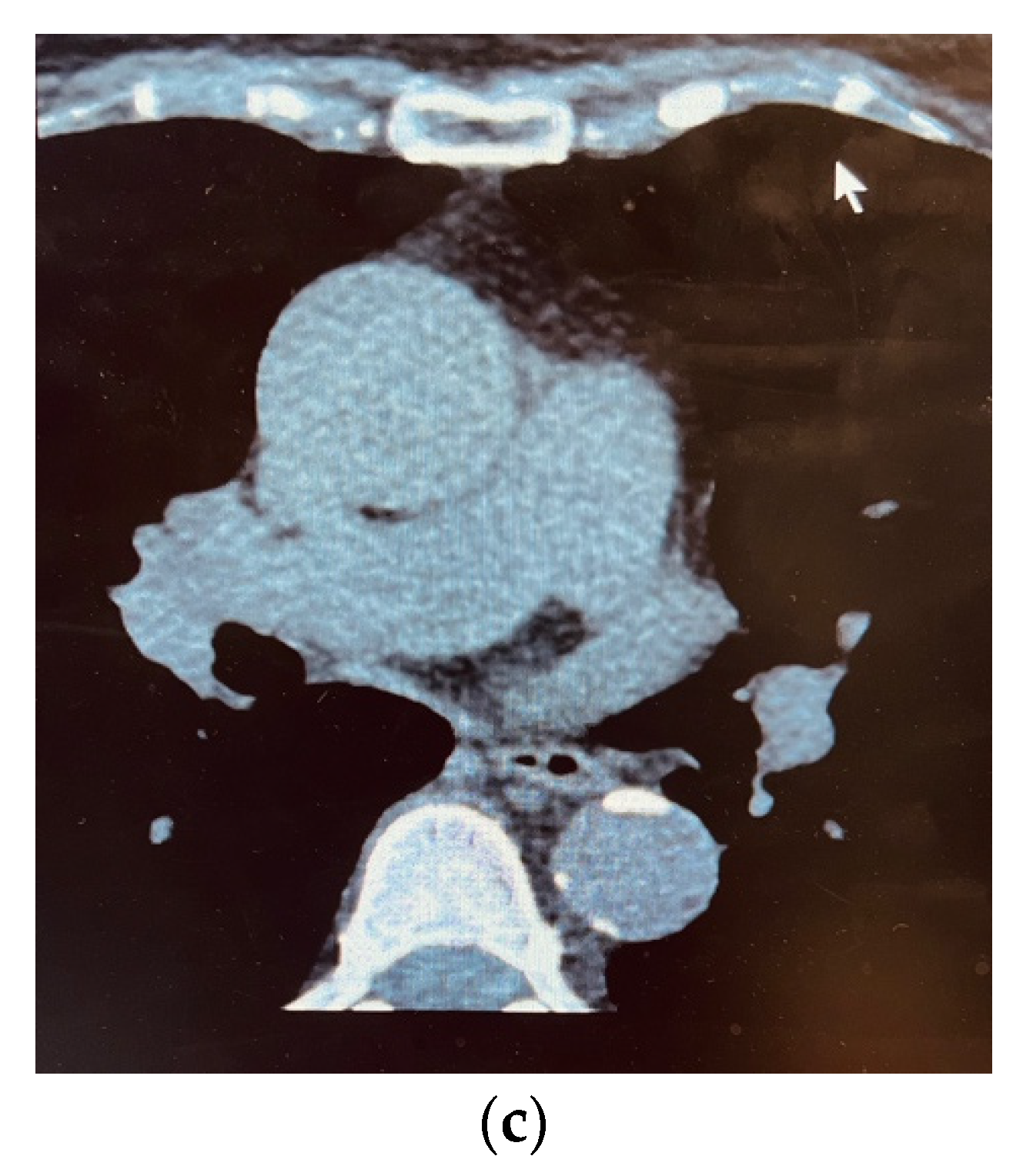
3. Results
4. Discussions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hayes, S.N.; Kim, E.S.H.; Saw, J.; Adlam, D.; Arslanian-Engoren, C.; Economy, K.E.; Ganesh, S.K.; Gulati, R.; Lindsay, M.E.; Mieres, J.H.; et al. Spontaneous Coronary Artery Dissection: Current State of the Science: A Scientific Statement From the American Heart Association. Circulation 2018, 137, e523–e557. [Google Scholar] [CrossRef] [PubMed]
- Tanis, W.; Stella, P.R.; Pijlman, A.H.; Kirkels, J.H.; Peters, R.H.J.; de Man, F.H. Spontaneous coronary artery dissection: Current insights and therapy. Neth. Hear. J. 2008, 16, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.N.; Tweet, M.S.; Adlam, D.; Kim, E.S.H.; Gulati, R.; Price, J.E.; Rose, C.H. Spontaneous Coronary Artery Dissection. J. Am. Coll. Cardiol. 2020, 76, 961–984. [Google Scholar] [CrossRef] [PubMed]
- Verstraeten, A.; Perik, M.H.A.M.; Baranowska, A.A.; Meester, J.A.N.; Van Den Heuvel, L.; Bastianen, J.; Kempers, M.; Krapels, I.P.C.; Maas, A.; Rideout, A.; et al. Enrichment of Rare Variants in Loeys–Dietz Syndrome Genes in Spontaneous Coronary Artery Dissection but not in Severe Fibromuscular Dysplasia. Circulation 2020, 142, 1021–1024. [Google Scholar] [CrossRef]
- Georges, A.; Yang, M.-L.; Berrandou, T.-E.; Bakker, M.K.; Dikilitas, O.; Kiando, S.R.; Ma, L.; Satterfield, B.A.; Sengupta, S.; Yu, M.; et al. Genetic Investigation of Fibromuscular Dysplasia Identifies Risk Loci and Shared Genetics with Common Cardiovascular Diseases. Nat. Commun. 2021, 12, 6031. [Google Scholar] [CrossRef] [PubMed]
- Meester, J.A.N.; Vandeweyer, G.; Pintelon, I.; Lammens, M.; Van Hoorick, L.; De Belder, S.; Waitzman, K.; Young, L.; Markham, L.W.; Vogt, J.; et al. Loss-of-Function Mutations in the X-Linked Biglycan Gene Cause a Severe Syndromic Form of Thoracic Aortic Aneurysms and Dissections. Genet. Med. 2017, 19, 386–395. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolton, L.; Robin, C.; Xu, J.; Wei, J.; Patil, R.; Robin, J. Could the BGN Gene Be Pathogenic with Spontaneous Coronary Artery Dissection (SCAD) and Fibromuscular Dysplasia (FMD)? Cardiogenetics 2022, 12, 270-275. https://doi.org/10.3390/cardiogenetics12040026
Kolton L, Robin C, Xu J, Wei J, Patil R, Robin J. Could the BGN Gene Be Pathogenic with Spontaneous Coronary Artery Dissection (SCAD) and Fibromuscular Dysplasia (FMD)? Cardiogenetics. 2022; 12(4):270-275. https://doi.org/10.3390/cardiogenetics12040026
Chicago/Turabian StyleKolton, Lexie, Charlie Robin, Jianfeng Xu, Jun Wei, Rupa Patil, and Jason Robin. 2022. "Could the BGN Gene Be Pathogenic with Spontaneous Coronary Artery Dissection (SCAD) and Fibromuscular Dysplasia (FMD)?" Cardiogenetics 12, no. 4: 270-275. https://doi.org/10.3390/cardiogenetics12040026
APA StyleKolton, L., Robin, C., Xu, J., Wei, J., Patil, R., & Robin, J. (2022). Could the BGN Gene Be Pathogenic with Spontaneous Coronary Artery Dissection (SCAD) and Fibromuscular Dysplasia (FMD)? Cardiogenetics, 12(4), 270-275. https://doi.org/10.3390/cardiogenetics12040026




