A BLE-Connected Piezoresistive and Inertial Chest Band for Remote Monitoring of the Respiratory Activity by an Android Application: Hardware Design and Software Optimization
Abstract
1. Introduction
- Hardware and firmware development of a piezoresistive chest band to detect the breathing parameters using a custom textile-based strain sensor;
- Design of a custom mobile application for displaying the breathing parameters wirelessly transmitted by the chest band.
- Comprehensive characterization of the developed device in measuring the RR in different operative conditions of the user (seated, standing, walking).
Overview of Wearable Sensing Systems for Breathing Monitoring Based on Piezoresistive and Piezoelectric Sensors
2. Materials and Methods
2.1. Architecture of Piezoresistive Chest Band Based on Strain-Sensitive Textile
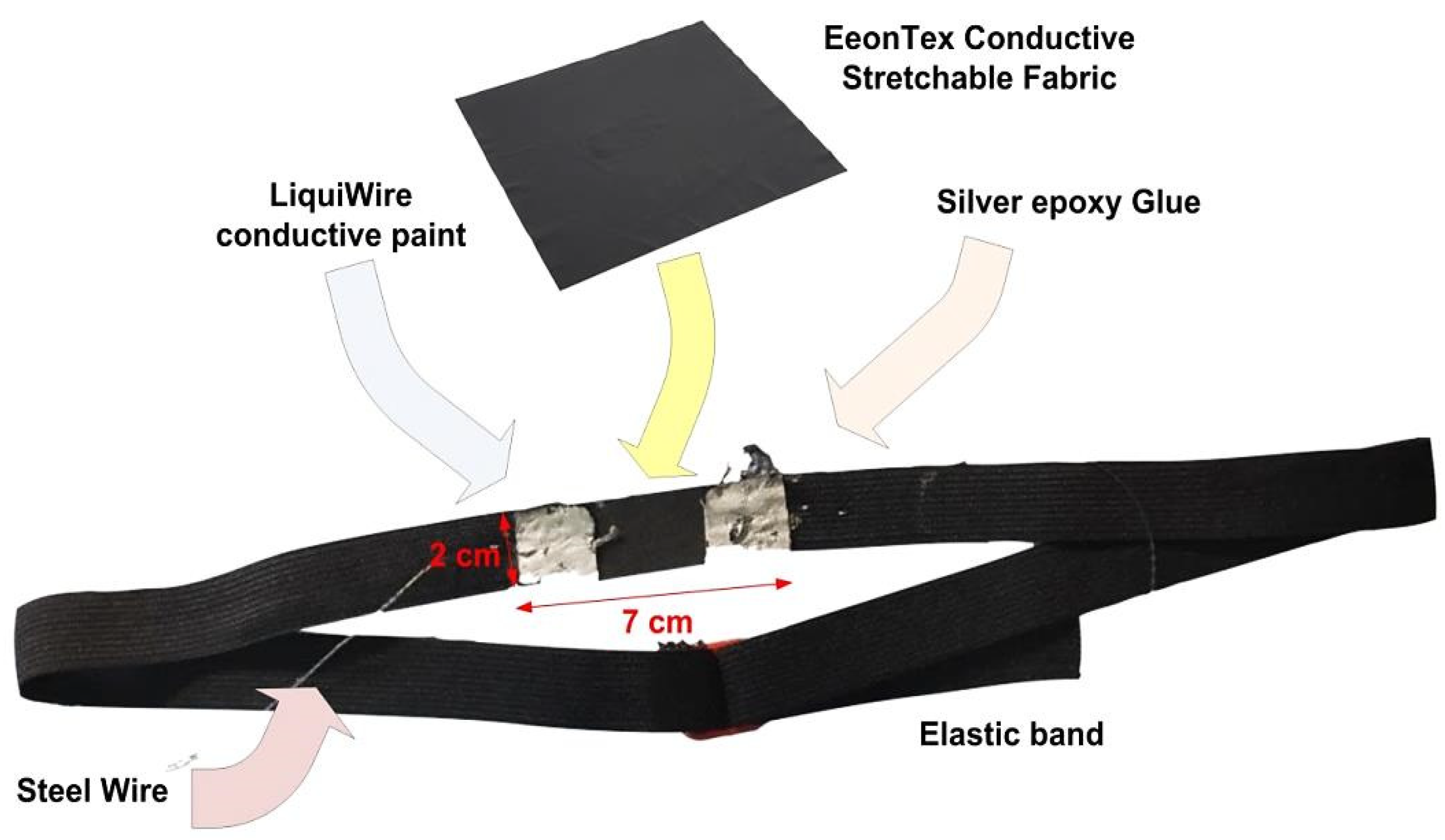
2.1.1. Development of the Piezoresistive Sensor Band
- Composition: nylon 72%/spandex 28%;
- Mass per unit area: 4.8 ounces/square yard;
- Surface resistivity: 20 KΩ/ square;
- Thickness: ~15 mils (0.38 mm);
- Elongation: warp 198–248 yarns/inch, warp recovery 85%;
- Dynamic range: 5 g–150 kg.
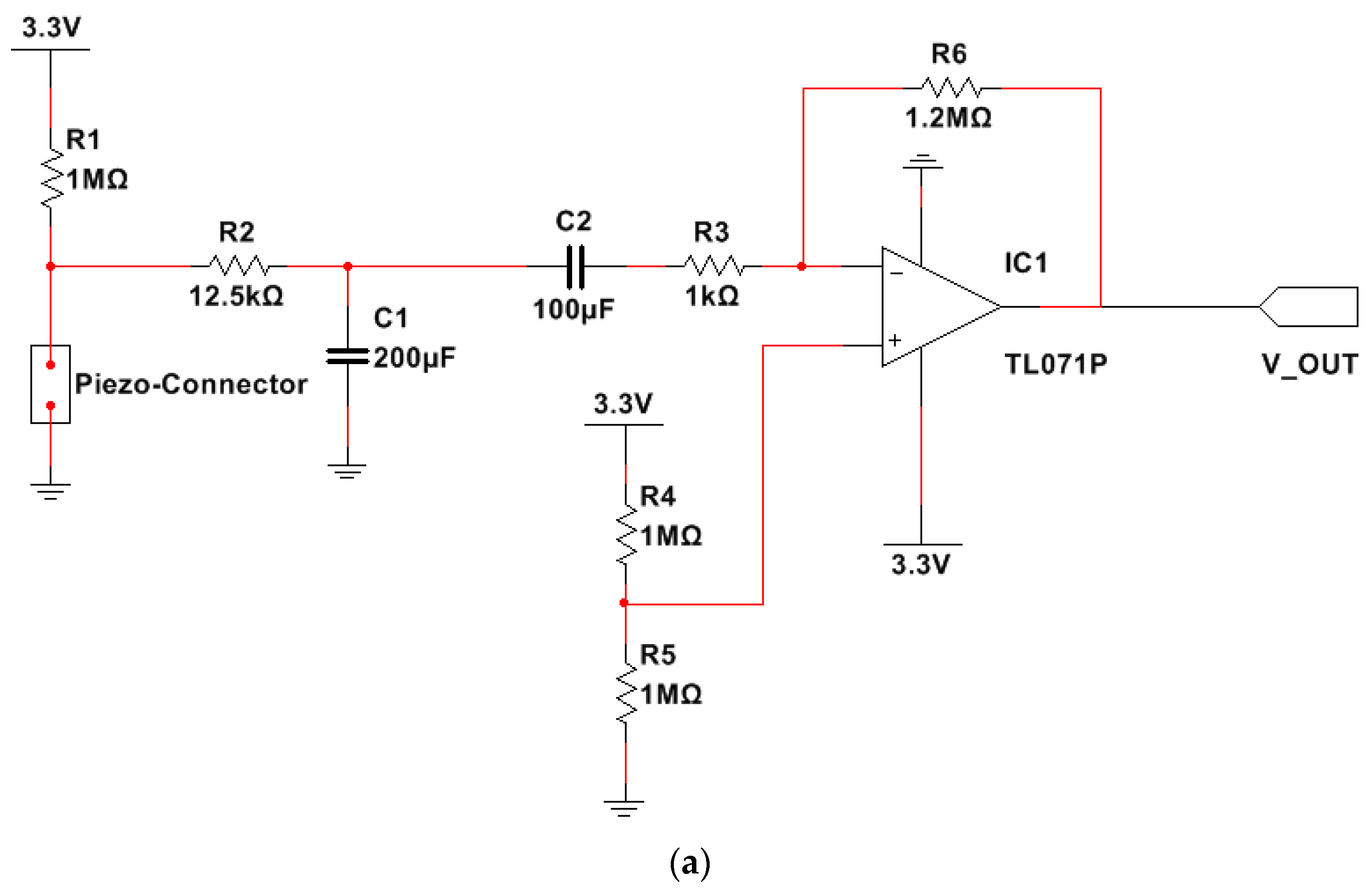
2.1.2. Electronic Conditioning and Processing Section of the Developed Chest Band
- Cut-off frequencies equal to about 41 mHz () and 2.5 Hz, respectively;
- Band gain ( equal to 30 dB (31.62 in linear scale).
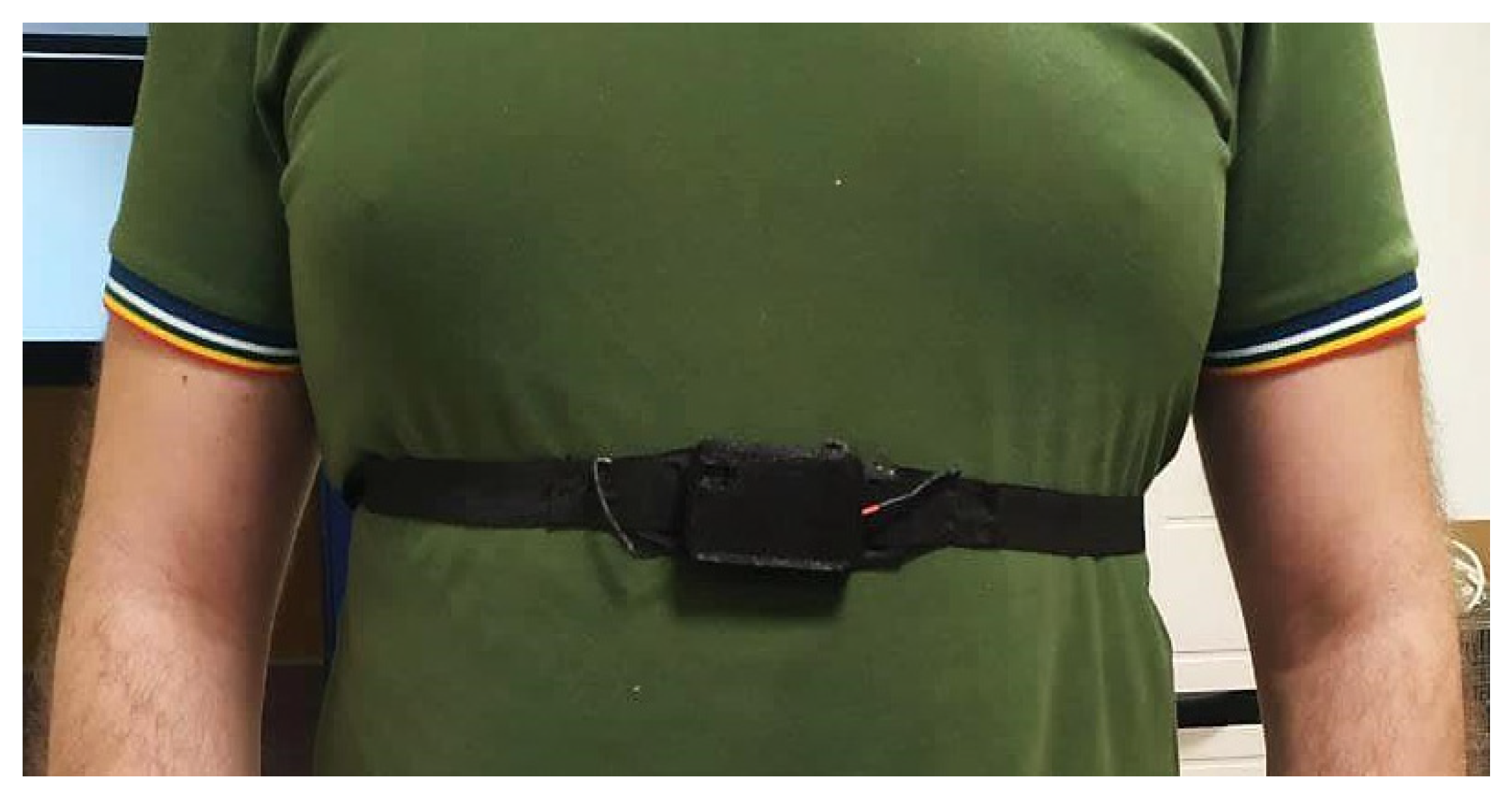
2.1.3. Assembly of the Chest Band Prototype
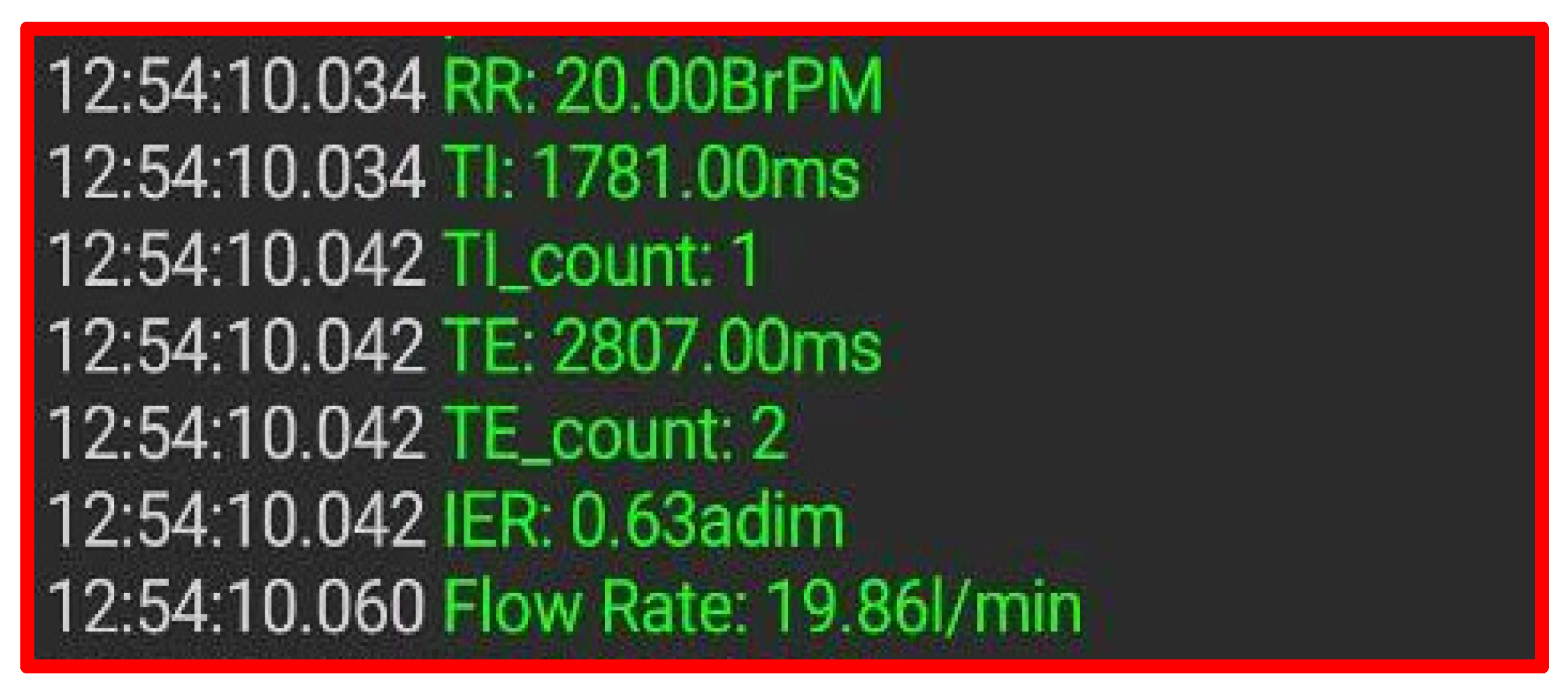
2.2. Chest Band’s Firmware for Measuring the Breathing Parameters
2.3. Mobile Application to Monitor the Data Transmitted by the Chest Band
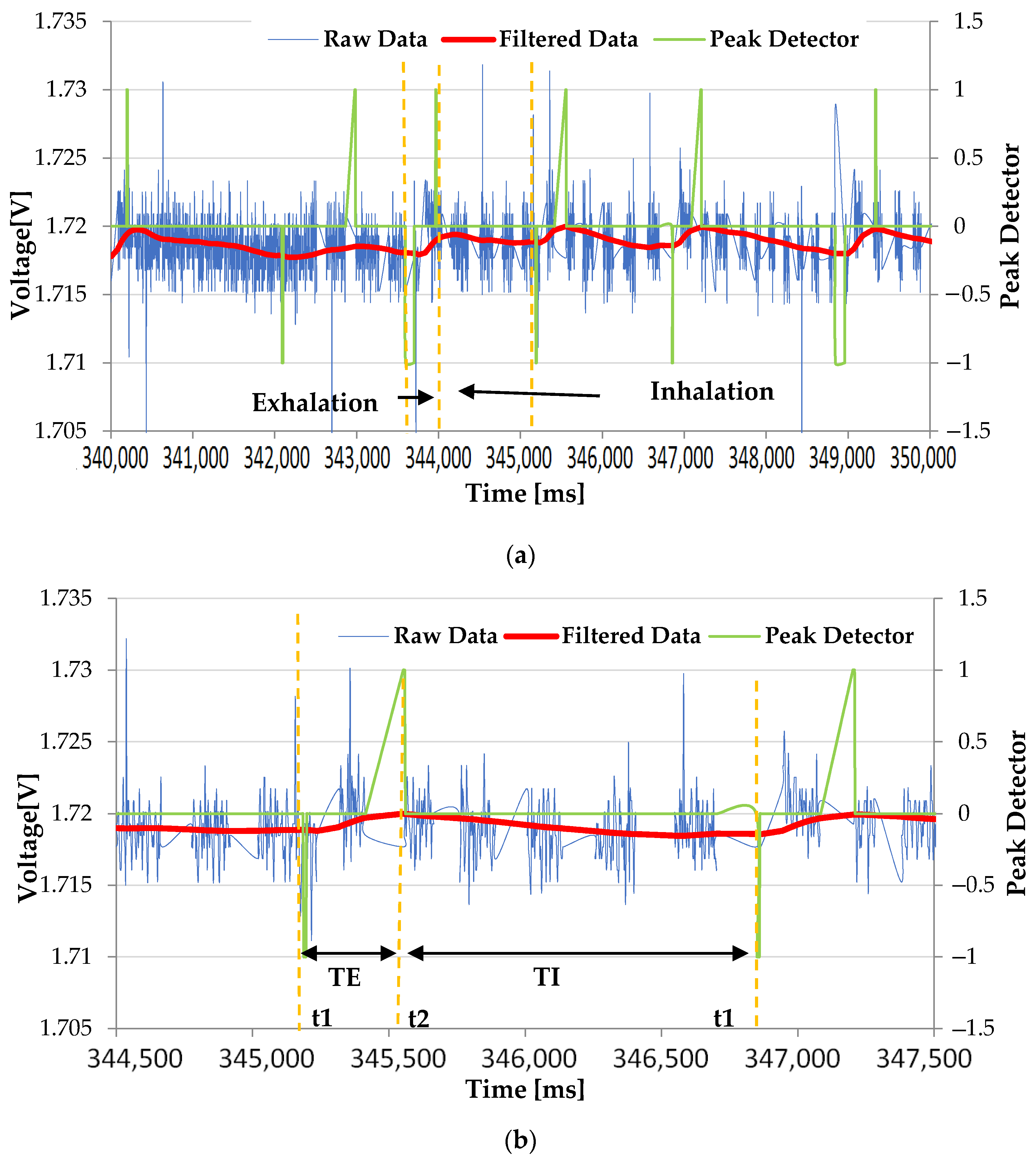
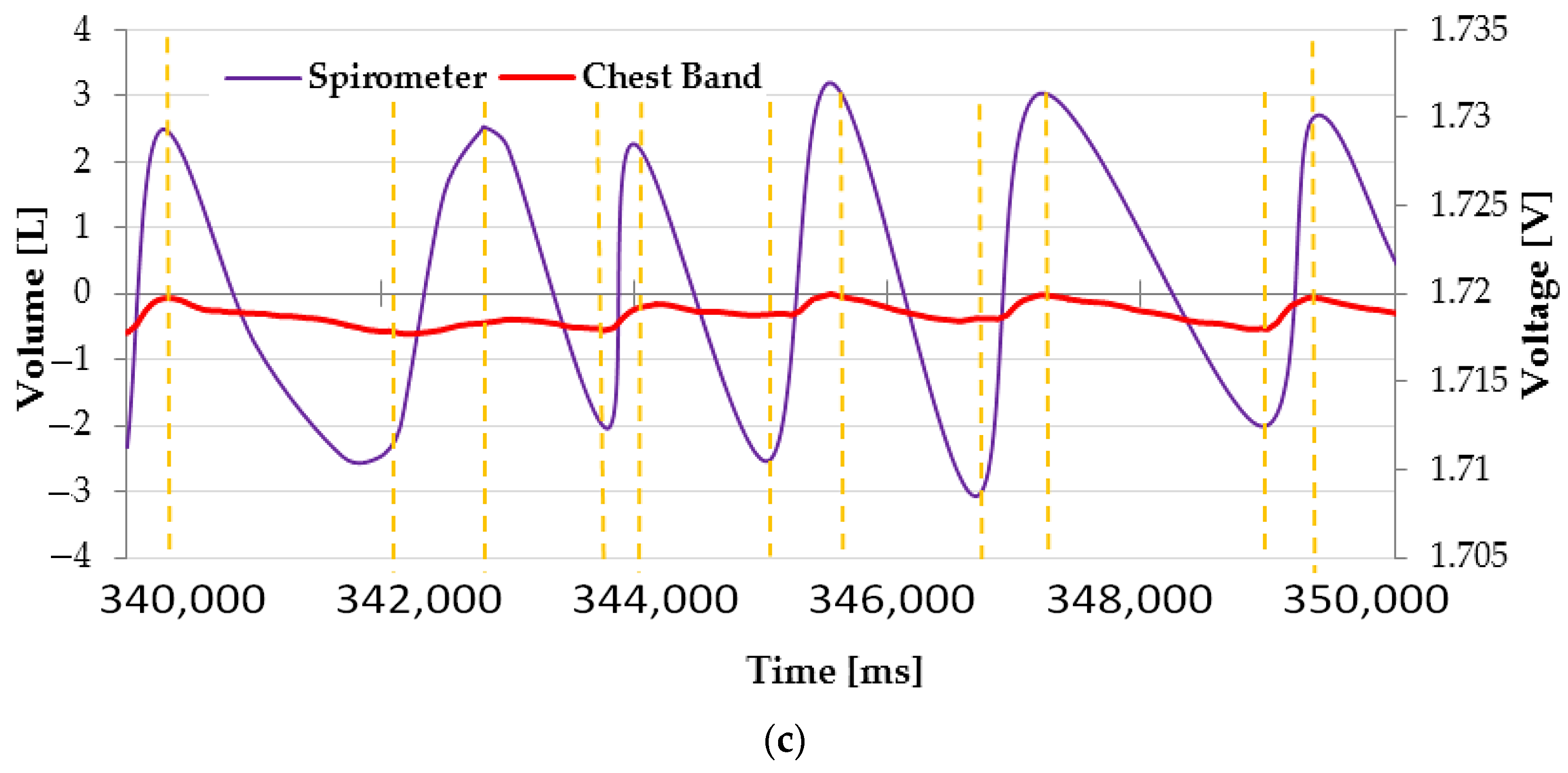
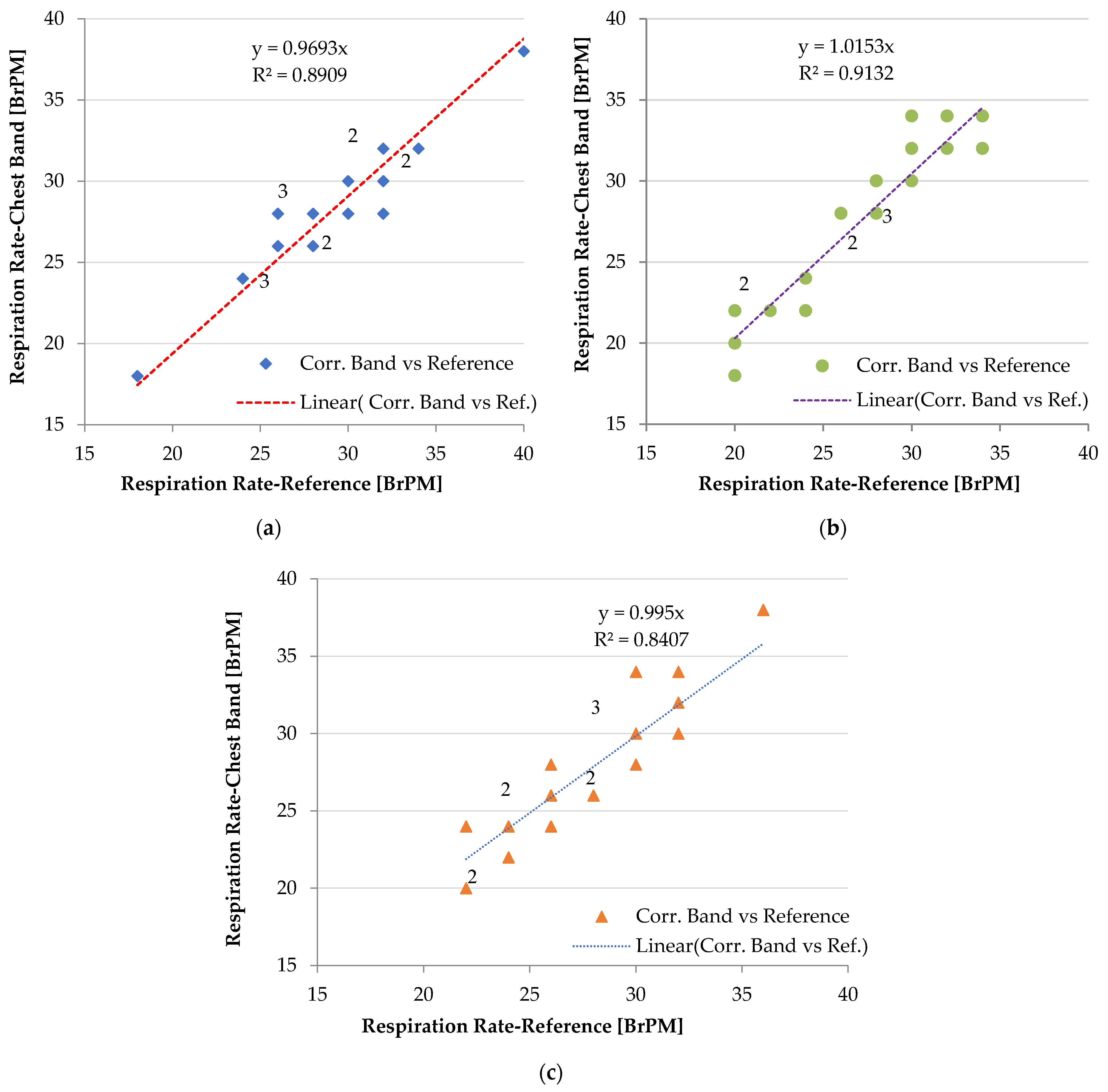
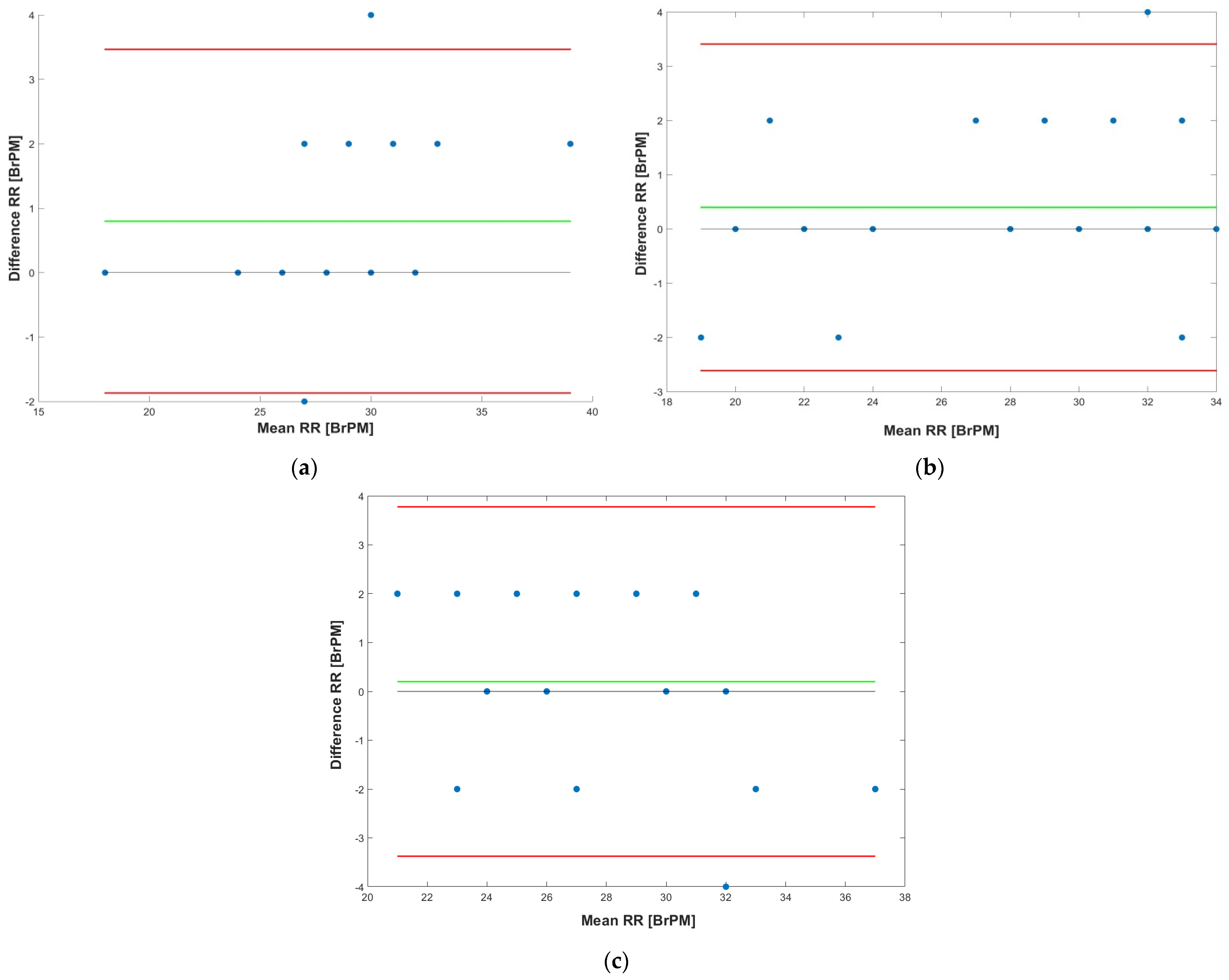
3. Results
4. Discussion
4.1. Performance of the Chest Band on Heterogeneous Patients
4.2. Comparative Analysis of the Proposed Piezoresistive Chest Band with Similar Devices
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aliverti, A. Wearable Technology: Role in Respiratory Health and Disease. Breathe 2017, 13, e27–e36. [Google Scholar] [CrossRef] [PubMed]
- Tipparaju, V.V.; Xian, X.; Bridgeman, D.; Wang, D.; Tsow, F.; Forzani, E.; Tao, N. Reliable Breathing Tracking with Wearable Mask Device. IEEE Sens. J. 2020, 20, 5510–5518. [Google Scholar] [CrossRef] [PubMed]
- Perez, A.J.; Zeadally, S. Recent Advances in Wearable Sensing Technologies. Sensors 2021, 21, 6828. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.P.; Howe, T.A.; Malarvili, M.B. Real-Time Human Respiration Carbon Dioxide Measurement Device for Cardiorespiratory Assessment. J. Breath Res. 2018, 12, 026003. [Google Scholar] [CrossRef] [PubMed]
- Kano, S.; Yamamoto, A.; Ishikawa, A.; Fujii, M. Respiratory Rate on Exercise Measured by Nanoparticle-Based Humidity Sensor. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2019, 2019, 3567–3570. [Google Scholar] [CrossRef] [PubMed]
- Longmore, S.K.; Lui, G.Y.; Naik, G.; Breen, P.P.; Jalaludin, B.; Gargiulo, G.D. A Comparison of Reflective Photoplethysmography for Detection of Heart Rate, Blood Oxygen Saturation, and Respiration Rate at Various Anatomical Locations. Sensors 2019, 19, 1874. [Google Scholar] [CrossRef]
- De Fazio, R.; Sponziello, A.; Cafagna, D.; Velazquez, R.; Visconti, P. An Overview on Technologies and Devices against COVID-19 Pandemic Diffusion: Virus Detection and Monitoring Solutions. Int. J. Smart Sens. Intell. Syst. 2021, 14, 1–28. [Google Scholar] [CrossRef]
- De Fazio, R.; De Vittorio, M.; Visconti, P. Innovative IoT Solutions and Wearable Sensing Systems for Monitoring Human Biophysical Parameters: A Review. Electronics 2021, 10, 1660. [Google Scholar] [CrossRef]
- Soon, S.; Svavarsdottir, H.; Downey, C.; Jayne, D.G. Wearable Devices for Remote Vital Signs Monitoring in the Outpatient Setting: An Overview of the Field. BMJ Innov. 2020, 6, 55–71. [Google Scholar] [CrossRef]
- Hahanov, V.; Miz, V. Big Data Driven Healthcare Services and Wearables. In Proceedings of the Experience of Designing and Application of CAD Systems in Microelectronics, Lviv, Ukraine, 24–27 February 2015; IEEE: Lviv, Ukraine, 2015; pp. 310–312. [Google Scholar]
- Blanco-Almazán, D.; Groenendaal, W.; Catthoor, F.; Jané, R. Chest Movement and Respiratory Volume Both Contribute to Thoracic Bioimpedance during Loaded Breathing. Sci. Rep. 2019, 9, 20232. [Google Scholar] [CrossRef]
- Zhong, J.; Li, C.; Zhu, W.; Zhou, H.; Liu, Y.; Han, X. Wearable Respiratory Strain Monitoring System Based on Textile-Based Capacitive Strain Sensor. J. Phys. Conf. Ser. 2020, 1570, 012033. [Google Scholar] [CrossRef]
- De Fazio, R.; Stabile, M.; De Vittorio, M.; Velázquez, R.; Visconti, P. An Overview of Wearable Piezoresistive and Inertial Sensors for Respiration Rate Monitoring. Electronics 2021, 10, 2178. [Google Scholar] [CrossRef]
- Rahmani, M.H.; Berkvens, R.; Weyn, M. Chest-Worn Inertial Sensors: A Survey of Applications and Methods. Sensors 2021, 21, 2875. [Google Scholar] [CrossRef] [PubMed]
- De Fazio, R.; Perrone, E.; Velázquez, R.; De Vittorio, M.; Visconti, P. Development of a Self-Powered Piezo-Resistive Smart Insole Equipped with Low-Power BLE Connectivity for Remote Gait Monitoring. Sensors 2021, 21, 4539. [Google Scholar] [CrossRef]
- Quy, V.N.; Duy, D.T.X.; Kien, D.T.; Tu, V.H.; Sun, Q.; Roy, V.A.L.; Pham, V.; Dao, T.T. Wearable Device for Monitoring Heart Rate Based on Low-Cost Piezoresistive Sensor. In Proceedings of the 2019 8th International Conference on Modern Circuits and Systems Technologies (MOCAST), Thessaloniki, Greece, 13–15 May 2019; pp. 1–4. [Google Scholar]
- Lanatà, A.; Scilingo, E.P.; Nardini, E.; Loriga, G.; Paradiso, R.; De-Rossi, D. Comparative Evaluation of Susceptibility to Motion Artifact in Different Wearable Systems for Monitoring Respiratory Rate. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 378–386. [Google Scholar] [CrossRef]
- Yang, W.; Li, N.-W.; Zhao, S.; Yuan, Z.; Wang, J.; Du, X.; Wang, B.; Cao, R.; Li, X.; Xu, W.; et al. A Breathable and Screen-Printed Pressure Sensor Based on Nanofiber Membranes for Electronic Skins. Adv. Mater. Technol. 2018, 3, 1700241. [Google Scholar] [CrossRef]
- Massaroni, C.; Nicolò, A.; Lo Presti, D.; Sacchetti, M.; Silvestri, S.; Schena, E. Contact-Based Methods for Measuring Respiratory Rate. Sensors 2019, 19, 908. [Google Scholar] [CrossRef]
- Reinvuo, T.; Hannula, M.; Sorvoja, H.; Alasaarela, E.; Myllyla, R. Measurement of Respiratory Rate with High-Resolution Accelerometer and Emfit Pressure Sensor. In Proceedings of the 2006 IEEE Sensors Applications Symposium, Houston, TX, USA, 7–9 February 2006; pp. 192–195. [Google Scholar]
- Vanegas, E.; Igual, R.; Plaza, I. Piezoresistive Breathing Sensing System with 3D Printed Wearable Casing. J. Sens. 2019, 2019, 2431731. [Google Scholar] [CrossRef]
- Francés-Morcillo, L.; Morer-Camo, P.; Rodríguez-Ferradas, M.I.; Cazón-Martín, A. Wearable Design Requirements Identification and Evaluation. Sensors 2020, 20, 2599. [Google Scholar] [CrossRef]
- Zheng, Q.; Liu, X.; Xu, H.; Cheung, M.-S.; Choi, Y.-W.; Huang, H.-C.; Lei, H.-Y.; Shen, X.; Wang, Z.; Wu, Y.; et al. Sliced Graphene Foam Films for Dual-Functional Wearable Strain Sensors and Switches. Nanoscale Horiz. 2018, 3, 35–44. [Google Scholar] [CrossRef]
- Raji, R.K.; Miao, X.; Wan, A.; Niu, L.; Li, Y.; Boakye, A. Knitted Piezoresistive Smart Chest Band and Its Application for Respiration Patterns Assessment. J. Eng. Fibers Fabr. 2019, 14, 1558925019868474. [Google Scholar] [CrossRef]
- Massaroni, C.; Di Tocco, J.; Lo Presti, D.; Longo, U.G.; Miccinilli, S.; Sterzi, S.; Formica, D.; Saccomandi, P.; Schena, E. Smart Textile Based on Piezoresistive Sensing Elements for Respiratory Monitoring. IEEE Sens. J. 2019, 19, 7718–7725. [Google Scholar] [CrossRef]
- Gargiulo, G.D.; O’Loughlin, A.; Breen, P.P. Electro-Resistive Bands for Non-Invasive Cardiac and Respiration Monitoring, a Feasibility Study. Physiol. Meas. 2015, 36, N35–N49. [Google Scholar] [CrossRef] [PubMed]
- Jayarathna, T.; Gargiulo, G.D.; Breen, P.P. Polymer Sensor Embedded, IOT Enabled t-Shirt for Long-Term Monitoring of Sleep Disordered Breathing. In Proceedings of the 2019 IEEE 5th World Forum on Internet of Things (WF-IoT), Limerick, Ireland, 15–18 April 2019; pp. 139–143. [Google Scholar]
- Molinaro, N.; Massaroni, C.; Lo Presti, D.; Saccomandi, P.; Di Tomaso, G.; Zollo, L.; Perego, P.; Andreoni, G.; Schena, E. Wearable Textile Based on Silver Plated Knitted Sensor for Respiratory Rate Monitoring. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 2865–2868. [Google Scholar]
- Chu, M.; Nguyen, T.; Pandey, V.; Zhou, Y.; Pham, H.N.; Bar-Yoseph, R.; Radom-Aizik, S.; Jain, R.; Cooper, D.M.; Khine, M. Respiration Rate and Volume Measurements Using Wearable Strain Sensors. npj Digit. Med. 2019, 2, 8. [Google Scholar] [CrossRef]
- Cay, G.; Ravichandran, V.; Saikia, M.J.; Hoffman, L.; Laptook, A.; Padbury, J.; Salisbury, A.L.; Gitelson-Kahn, A.; Venkatasubramanian, K.; Shahriari, Y.; et al. An E-Textile Respiration Sensing System for NICU Monitoring: Design and Validation. J. Sign. Process. Syst. 2021, 1–15. [Google Scholar] [CrossRef]
- Rasheed, A.; Iranmanesh, E.; Li, W.; Xu, Y.; Zhou, Q.; Ou, H.; Wang, K. An Active Self-Driven Piezoelectric Sensor Enabling Real-Time Respiration Monitoring. Sensors 2019, 19, 3241. [Google Scholar] [CrossRef]
- Zanetti, J.M.; Tavakolian, K. Seismocardiography: Past, Present and Future. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 7004–7007. [Google Scholar]
- Andreozzi, E.; Fratini, A.; Esposito, D.; Naik, G.; Polley, C.; Gargiulo, G.D.; Bifulco, P. Forcecardiography: A Novel Technique to Measure Heart Mechanical Vibrations onto the Chest Wall. Sensors 2020, 20, 3885. [Google Scholar] [CrossRef]
- Andreozzi, E.; Centracchio, J.; Punzo, V.; Esposito, D.; Polley, C.; Gargiulo, G.D.; Bifulco, P. Respiration Monitoring via Forcecardiography Sensors. Sensors 2021, 21, 3996. [Google Scholar] [CrossRef]
- Andreozzi, E.; Gargiulo, G.D.; Esposito, D.; Bifulco, P. A Novel Broadband Forcecardiography Sensor for Simultaneous Monitoring of Respiration, Infrasonic Cardiac Vibrations and Heart Sounds. Front. Physiol. 2021, 12, 725716. [Google Scholar] [CrossRef]
- Lee, J.; Kim, M.; Park, H.-K.; Kim, I.Y. Motion Artifact Reduction in Wearable Photoplethysmography Based on Multi-Channel Sensors with Multiple Wavelengths. Sensors 2020, 20, 1493. [Google Scholar] [CrossRef]
- Piuzzi, E.; Capuano, A.; Pisa, S.; Cappa, P.; Patané, F.; Rossi, S.; Giaquinto, N.; D’Aucelli, G.M. Impedance Plethysmography System with Inertial Measurement Units for Motion Artefact Reduction: Application to Continuous Breath Activity Monitoring. In Proceedings of the 2015 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Turin, Italy, 7–9 July 2015. [Google Scholar] [CrossRef]
- Whitlock, J.; Sill, J.; Jain, S. A-Spiro: Towards Continuous Respiration Monitoring. Smart Health 2020, 15, 100105. [Google Scholar] [CrossRef]
- Munz, M.; Wolf, N. Simulation of Breathing Patterns and Classification of Sensor Data for the Early Detection of Impending Sudden Infant Death. Curr. Dir. Biomed. Eng. 2019, 5, 401–403. [Google Scholar] [CrossRef][Green Version]
- MG Chemicals Silver Conductive Epoxy Adhesive 8221 Technical Datasheet. Available online: https://www.alliedelec.com/m/d/7989ddf713cf916cf8c8b6db80c7c854.pdf (accessed on 13 July 2021).
- LiquiwireTM Elettricamente Conducendo Vernice per PCB Circuito Elettrico & Riparazione. Available online: https://www.ebay.it/itm/223704549098 (accessed on 12 July 2021).
- EeonTex Conductive Stretchable Fabric—COM-14112—SparkFun Electronics. Available online: https://www.sparkfun.com/products/retired/14112 (accessed on 12 July 2021).
- Liang, A.; Stewart, R.; Bryan-Kinns, N. Analysis of Sensitivity, Linearity, Hysteresis, Responsiveness, and Fatigue of Textile Knit Stretch Sensors. Sensors 2019, 19, 3618. [Google Scholar] [CrossRef] [PubMed]
- Texas Instruments TL071 Datasheet. Available online: https://www.ti.com/product/TL071 (accessed on 13 July 2021).
- Wirth, A.; Gates, C.; Smith, J. Medical Device Cybersecurity for Engineers and Manufacturers; Artech House: Norwood, MA, USA, 2020; ISBN 978-1-63081-816-6. [Google Scholar]
- McCarron, M.M.; Devine, B.J. Clinical Pharmacy: Case Studies: Case Number 25 Gentamicin Therapy. Drug Intell. Clin. Pharm. 1974, 8, 650–655. [Google Scholar] [CrossRef]
- Scott, J.B.; Kaur, R. Monitoring Breathing Frequency, Pattern, and Effort. Respir. Care 2020, 65, 793–806. [Google Scholar] [CrossRef]
- Fieselmann, J.F.; Hendryx, M.S.; Helms, C.M.; Wakefield, D.S. Respiratory Rate Predicts Cardiopulmonary Arrest for Internal Medicine Inpatients. J. Gen. Intern. Med. 1993, 8, 354–360. [Google Scholar] [CrossRef]
- Van Diest, I.; Verstappen, K.; Aubert, A.E.; Widjaja, D.; Vansteenwegen, D.; Vlemincx, E. Inhalation/Exhalation Ratio Modulates the Effect of Slow Breathing on Heart Rate Variability and Relaxation. Appl. Psychophysiol. Biofeedback 2014, 39, 171–180. [Google Scholar] [CrossRef]
- Laborde, S.; Iskra, M.; Zammit, N.; Borges, U.; You, M.; Sevoz-Couche, C.; Dosseville, F. Slow-Paced Breathing: Influence of Inhalation/Exhalation Ratio and of Respiratory Pauses on Cardiac Vagal Activity. Sustainability 2021, 13, 7775. [Google Scholar] [CrossRef]
- Pleil, J.D.; Wallace, M.A.G.; Davis, M.D.; Matty, C.M. The Physics of Human Breathing: Flow, Timing, Volume, and Pressure Parameters for Normal, on-Demand, and Ventilator Respiration. J. Breath Res. 2021, 15, 042002. [Google Scholar] [CrossRef]
- Samarghandi, A.; Ioachimescu, O.C.; Qayyum, R. Association between Peak Inspiratory Flow Rate and Hand Grip Muscle Strength in Hospitalized Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease. PLoS ONE 2020, 15, e0227737. [Google Scholar] [CrossRef]
- Langone, M.; Setola, R.; Lopez, J. Cybersecurity of Wearable Devices: An Experimental Analysis and a Vulnerability Assessment Method. In Proceedings of the 2017 IEEE 41st Annual Computer Software and Applications Conference (COMPSAC), Turin, Italy, 4–8 July 2017; Volume 2, pp. 304–309. [Google Scholar]
- Beattie, Z.T.; Jacobs, P.G.; Riley, T.C.; Hagen, C.C. A Time-Frequency Respiration Tracking System Using Non-Contact Bed Sensors with Harmonic Artifact Rejection. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; Volume 2015, pp. 8111–8114. [Google Scholar] [CrossRef]
- Giavarina, D. Understanding Bland Altman Analysis. Biochem. Med. 2015, 25, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Saha, U.; Kamat, A.; Sengupta, D.; Jayawardhana, B.; Kottapalli, A.G.P. A Low-Cost Lung Monitoring Point-of-Care Device Based on a Flexible Piezoresistive Flow Sensor. In Proceedings of the 2020 IEEE Sensors, Rotterdam, The Netherlands, 25 October 2020; pp. 1–4. [Google Scholar]
- Nguyen, T.-V.; Ichiki, M. MEMS-Based Sensor for Simultaneous Measurement of Pulse Wave and Respiration Rate. Sensors 2019, 19, 4942. [Google Scholar] [CrossRef] [PubMed]
- Abbasnejad, B.; Thorby, W.; Razmjou, A.; Jin, D.; Asadnia, M.; Ebrahimi Warkiani, M. MEMS Piezoresistive Flow Sensors for Sleep Apnea Therapy. Sens. Actuators A Phys. 2018, 279, 577–585. [Google Scholar] [CrossRef]
- Capineri, L. Resistive Sensors with Smart Textiles for Wearable Technology: From Fabrication Processes to Integration with Electronics. Procedia Eng. 2014, 87, 724–727. [Google Scholar] [CrossRef]
- Liu, G.-Z.; Guo, Y.-W.; Zhu, Q.-S.; Huang, B.-Y.; Wang, L. Estimation of Respiration Rate from Three-Dimensional Acceleration Data Based on Body Sensor Network. Telemed. e-Health 2011, 17, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Siam, A.I.; El-Bahnasawy, N.A.; Banby, G.M.E.; Elazm, A.A.; El-Samie, F.E.A.; El-Samie, F.E.A. Efficient Video-Based Breathing Pattern and Respiration Rate Monitoring for Remote Health Monitoring. J. Opt. Soc. Am. A JOSAA 2020, 37, 118–124. [Google Scholar] [CrossRef]
- Ali, M.; Elsayed, A.; Mendez, A.; Savaria, Y.; Sawan, M. Contact and Remote Breathing Rate Monitoring Techniques: A Review. IEEE Sens. J. 2021, 21, 14569–14586. [Google Scholar] [CrossRef]
- Tachiquin, R.; Velázquez, R.; Del-Valle-Soto, C.; Gutiérrez, C.A.; Carrasco, M.; De Fazio, R.; Trujillo-León, A.; Visconti, P.; Vidal-Verdú, F. Wearable Urban Mobility Assistive Device for Visually Impaired Pedestrians using a Smartphone and a Tactile-Foot Interface. Sensors 2021, 21, 5274. [Google Scholar] [CrossRef]
- Jeong, J.-W.; Jang, Y.; Lee, I.; Shin, S.; Kim, S.-B. Wearable Respiratory Rate Monitoring Using Piezo-Resistive Fabric Sensor. In Proceedings of the 2009 World Congress on Medical Physics and Biomedical Engineering, Munich, Germany, 7–12 September 2009; Springer: Munich, Germany, 2009; pp. 282–284. [Google Scholar] [CrossRef]
- Huang, C.-T.; Shen, C.-L.; Tang, C.-F.; Chang, S.-H. A Wearable Yarn-Based Piezo-Resistive Sensor. Sens. Actuators A. Phys. 2008, 2, 396–403. [Google Scholar] [CrossRef]
- Di Tocco, J.; Raiano, L.; Sabbadini, R.; Massaroni, C.; Formica, D.; Schena, E. A Wearable System with Embedded Conductive Textiles and an IMU for Unobtrusive Cardio-Respiratory Monitoring. Sensors 2021, 21, 3018. [Google Scholar] [CrossRef]






| Work | Transduction Mechanism | Advantages | Disadvantages |
|---|---|---|---|
| E. Vanegas et al. [21] | Piezoresistive | Lightness, Small dimension, and Flexibility | Moving mechanical parts No compensation for motion artefacts |
| R.K. Raji et al. [24] | Piezoresistive | Flexibility, Easy to wear, Lightness | Cumbersome electronic section No compensation for motion artefacts |
| C. Massironi et al. [25] | Piezoresistive | Flexibility, Lightness, High Accuracy | Obstructive (bulky connections) No compensation for motion artefacts |
| G.D. Gargiulo et al. [26] | Piezoresistive | Flexibility, Lightness, Easy to wear | Obstructive (covers chest and abdomen) No compensation for motion artefacts |
| N. Molinaro et al. [28] | Piezoresistive | Flexibility, Lightness | High hysteresis |
| M. Chu et al. [29] | Piezoresistive | High Accuracy, Lightness, Flexibility | Obstructive (application on the skin) |
| A. Rasheed et al. [31] | Piezoelectric | High sensitivity and reliability Lightness, Flexibility | No compensation for motion artefacts |
| E. Andreozzi et al. [35] | Piezoelectric | High Accuracy, Lightness, | Obstructive (application on the skin) |
| J. Whitlock et al. [38] | Capacitive | Lightness, Small dimension, and Flexibility | Cumbersome electronic section Processing unit not on board |
| Chest Band | Spirometer | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TI [ms] | TE [ms] | IER [adim] | V [l\min] | TI [ms] | TE [ms] | IER [adim] | V [l\min] | ETI [%] | ETE [%] | EIER [%] | EV [%] | |
| 1 | 895 | 750 | 1.2 | 39.5 | 850 | 760 | 1.1 | 41.6 | −5.3 | 1.3 | −6.7 | 5.0 |
| 2 | 1121 | 860 | 1.3 | 31.6 | 1180 | 880 | 1.3 | 30.0 | 5.0 | 2.3 | 2.8 | −5.3 |
| 3 | 1210 | 862 | 1.4 | 29.2 | 1120 | 810 | 1.4 | 31.6 | −8.0 | −6.4 | −1.5 | 7.4 |
| 4 | 1220 | 751 | 1.6 | 29.0 | 1180 | 750 | 1.6 | 30.0 | −3.4 | −0.1 | −3.3 | 3.3 |
| 5 | 1317 | 874 | 1.5 | 26.9 | 1360 | 850 | 1.6 | 26.0 | 3.2 | −2.8 | 5.8 | −3.3 |
| 6 | 1423 | 963 | 1.5 | 24.9 | 1450 | 950 | 1.5 | 24.4 | 1.9 | −1.4 | 3.2 | −1.9 |
| 7 | 1452 | 979 | 1.5 | 24.4 | 1510 | 980 | 1.5 | 23.4 | 3.8 | 0.1 | 3.7 | −4.0 |
| 8 | 1510 | 965 | 1.6 | 23.4 | 1580 | 950 | 1.7 | 22.4 | 4.4 | −1.6 | 5.9 | −4.6 |
| 9 | 1528 | 982 | 1.6 | 23.1 | 1560 | 980 | 1.6 | 22.7 | 2.1 | −0.2 | 2.3 | −2.1 |
| 10 | 1653 | 1024 | 1.6 | 21.4 | 1620 | 1050 | 1.5 | 21.8 | −2.0 | 2.5 | −4.6 | 2.0 |
| Patient | Gender | Age | Weight [kg] |
|---|---|---|---|
| 1 | Male | 30 | 81 |
| 2 | Male | 24 | 78 |
| 3 | Male | 22 | 80 |
| 4 | Female | 25 | 62 |
| 5 | Female | 24 | 65 |
| 6 | Femal | 28 | 72 |
| Seated | Standing | Walking | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient | r [adim] | MD [BrPM] | LoA [BrPM] | ME [%] | r [adim] | MD [BrPM] | LoA [BrPM] | ME [%] | r [adim] | MD [BrPM] | LoA [BrPM] | ME [%] |
| 1 | 0.958 | 0.8 | +3.47/−1.87 | +3.39 | 0.956 | 0.4 | +3.40/−2.61 | +3.76 | 0.923 | 0.2 | +3.77/−3.37 | +5.10 |
| 2 | 0.961 | 0.7 | +3.21/−1.64 | +3.12 | 0.963 | 0.5 | +3.25/−2.34 | +3.42 | 0.931 | 0.3 | +3.62/−2.56 | +4.12 |
| 3 | 0.962 | 0.6 | +2.83/−1.89 | +2.98 | 0.952 | 0.3 | +3.01/−2.56 | +3.12 | 0.927 | 0.2 | +3.54/−2.89 | +4.32 |
| 4 | 0.951 | 0.7 | +2.99/−1.98 | +3.24 | 0.949 | 0.4 | +3.42/−2.52 | +3.67 | 0.934 | 0.1 | +3.62/−3.67 | +4.76 |
| 5 | 0.957 | 0.6 | +3.25/−1.67 | +2.82 | 0.947 | 0.3 | +3.39/−2.56 | +3.78 | 0.921 | 0.3 | +3.56/−3.54 | +4.65 |
| 6 | 0.968 | 0.7 | +3.45/−1.45 | +3.25 | 0.956 | 0.5 | +3.56/−2.78 | +3.62 | 0.932 | 0.3 | +3.78/−3.65 | +4.78 |
| Work | Sensing Device | Processing Unit | Sampling Frequency [Hz] | Availability of Wireless Communication | Wearability |
|---|---|---|---|---|---|
| E. Venegas et al. [21] | A201 FlexiForce sensor | Arduino Pro Mini (ATMega32U) | 50 | Bluetooth (HC-05) | Medium |
| U. Saha et al. [56] | PDMS/Graphene sensor | Arduino Uno (ATMega328p) | 1000 | WiFi (ESP8266) | Low |
| T.V. Nguyen et al. [57] | MEMS pressure sensor | DL850 Acquisition board/PC | 1000 | No | High |
| R. K. Raji et al. [24] | piezoresistive fabric | STM32F401RCT6 | 1000 | BLE (HC-08) | Medium |
| B. Abbasnejad et al. [58] | MEMS LCP pressure sensor | NI-USB6003 board/PC | 2000 | No | Low |
| M. Chu et al. [29] | Strain sensor | NI-USB6003 board/PC | 1000 | Bluetooth | High |
| C. Massironi et al. [25] | Silver conductive yarns | PIC18F46J50 | 60 | Bluetooth (SPBT2632C2A) | Medium |
| Proposed Chest Band | LTT-SLPA-20K Piezoresistive fabric | SAMD21G18 | 1000 | BLE (JDY-23) | High |
| Work | Number of Sensors | Mean Error |
|---|---|---|
| Proposed Chest Band | 1 | 3.13% a |
| E. Venegas et al. [21] | 1 | 3.53%/3.99% |
| J. Jeong et al. [64] | 1 | 2.99% |
| M Chu et al. [29] | 2 | 2% |
| C. T. Huang et al. [65] | 1 | 0.5% |
| G. Cay et al. [30] | 2 | 2.06% |
| C. Massironi et al. [66] | 2 | 1.45% b −3.49 a −9.51% c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Fazio, R.; De Vittorio, M.; Visconti, P. A BLE-Connected Piezoresistive and Inertial Chest Band for Remote Monitoring of the Respiratory Activity by an Android Application: Hardware Design and Software Optimization. Future Internet 2022, 14, 183. https://doi.org/10.3390/fi14060183
De Fazio R, De Vittorio M, Visconti P. A BLE-Connected Piezoresistive and Inertial Chest Band for Remote Monitoring of the Respiratory Activity by an Android Application: Hardware Design and Software Optimization. Future Internet. 2022; 14(6):183. https://doi.org/10.3390/fi14060183
Chicago/Turabian StyleDe Fazio, Roberto, Massimo De Vittorio, and Paolo Visconti. 2022. "A BLE-Connected Piezoresistive and Inertial Chest Band for Remote Monitoring of the Respiratory Activity by an Android Application: Hardware Design and Software Optimization" Future Internet 14, no. 6: 183. https://doi.org/10.3390/fi14060183
APA StyleDe Fazio, R., De Vittorio, M., & Visconti, P. (2022). A BLE-Connected Piezoresistive and Inertial Chest Band for Remote Monitoring of the Respiratory Activity by an Android Application: Hardware Design and Software Optimization. Future Internet, 14(6), 183. https://doi.org/10.3390/fi14060183








