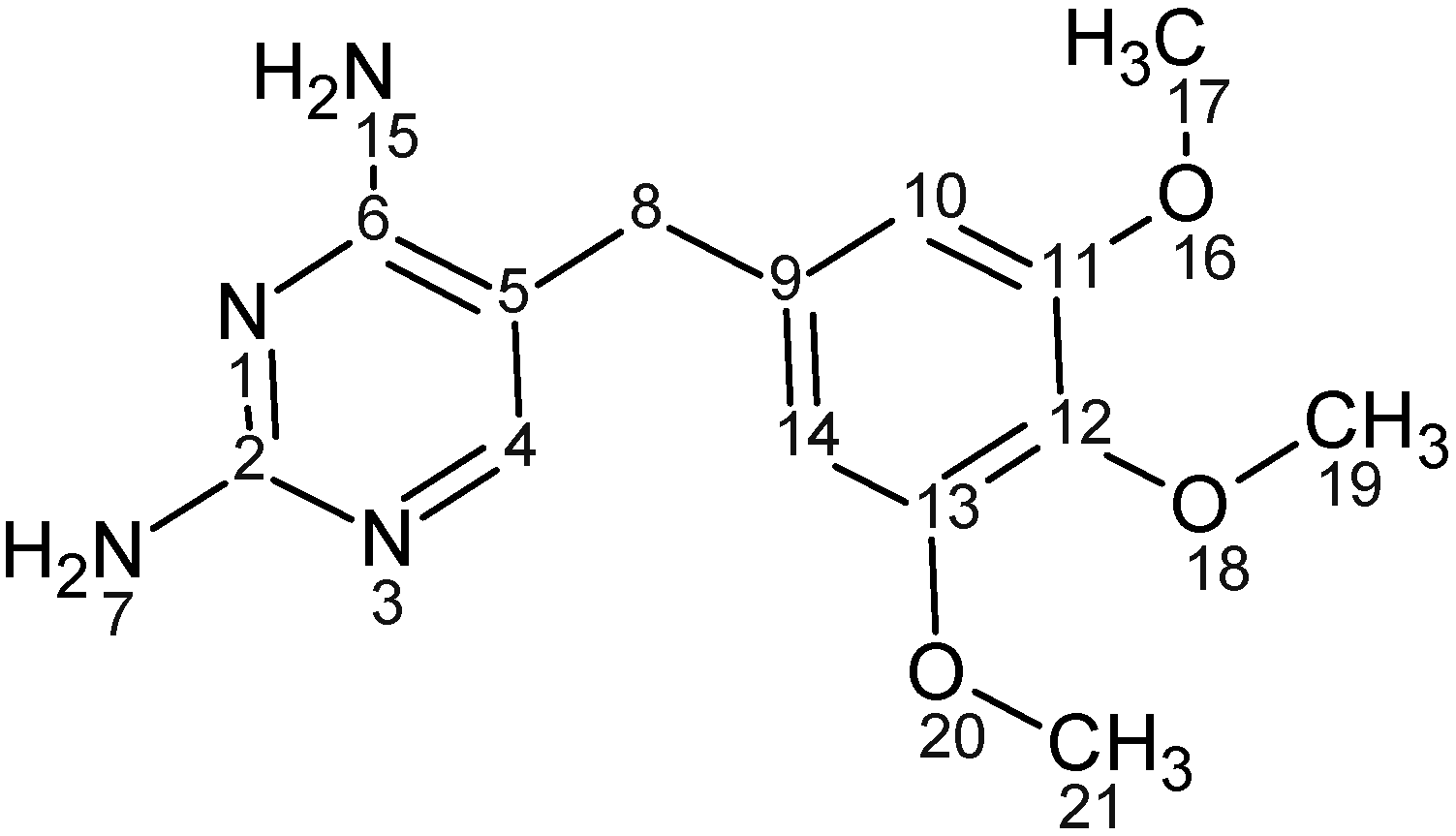
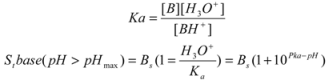In order to evaluate the effect of isomer form on the salt characteristics, D and L isomers of glutamic and aspartic acid were used to prepare TMP salts. The prepared salts were further characterized by Fourier transform infra-red, differential scanning calorimetry, thermo-gravimetric analysis and 1H nuclear magnetic resonance.
3.2.2. Differential Scanning Calorimetry and Thermogravimetric Analysis
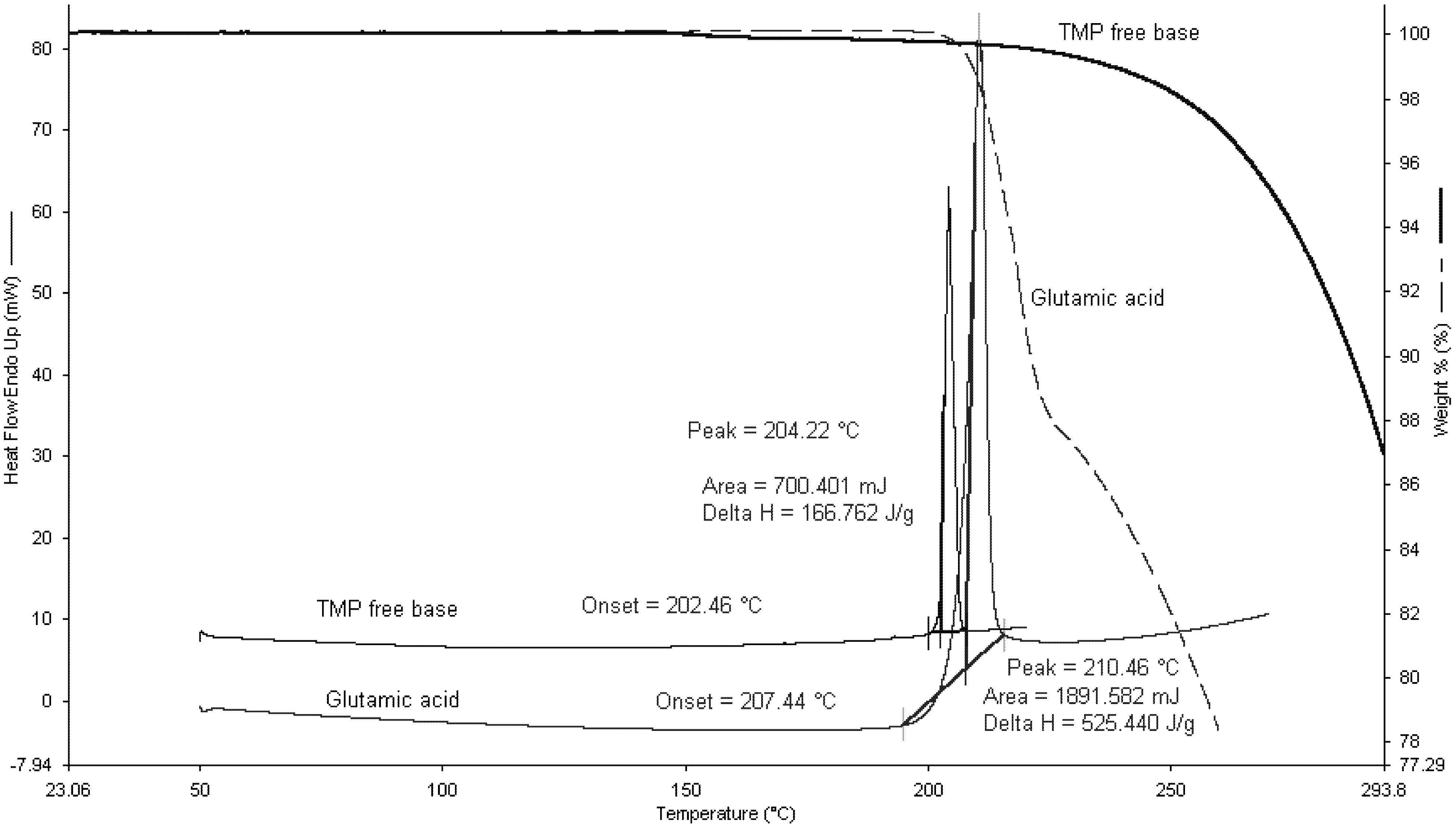
Figure 5 shows the TGA and DSC scans for TMP and L-glutamic acid. TMP had one single endothermic peak at 204.22 °C with an enthalpy of fusion of 166.7 J/g. TGA data revealed that TMP starts to degrade after around 30 °C of its melting.
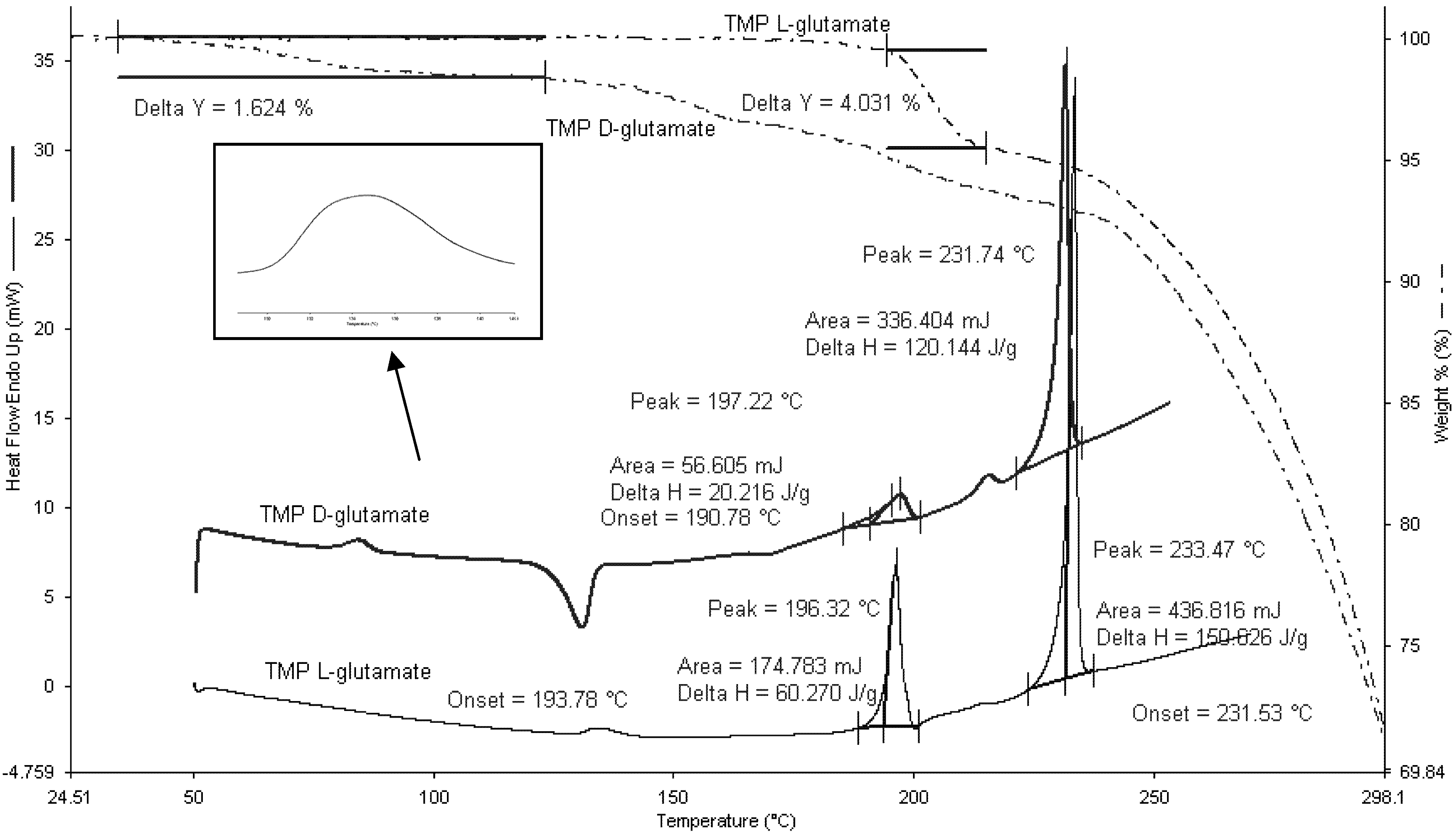
TMP L-glutamate salt was found to be in the crystalline form as no crystallization exotherm was observed in the DSC scans (
Figure 6). At around 130 °C, TMP L-glutamate had an endothermic peak which could possibly correspond to moisture loss. Two endothermic peaks appeared at 197.22 °C (ΔH = 20.22 J/g) and 233.47 °C (ΔH = 150.02 J/g). This data suggests the presence of two enantiotrophs or monotrophs. A second scan was carried out after cooling and the results showed that the first thermal event was irreversible which suggests the presence of monotrophs rather than enantiotrophs. Interestingly, coupling the DSC scans with TGA showed a weight loss (4.03%) at 196 °C which indicates that the thermal event is not a phase transition as polymorphic transitions are not associated with weight loss. In addition,
1HNMR data (discussed in details in Section 3.3.3) shows the absence of any impurities which provides further evidence that the weight loss is due to associated water molecules within the crystalline structure of the salt [
17].” TGA was carried out to further validate DSC results as mass loss (which is routinely studied with TGA) indicates the presence of moisture [
17].
Figure 5.
Thermogravimetric analysis (TGA) and Differential Scanning Calorimetry (DSC) scans for L-glutamic acid and TMP free base.
Figure 5.
Thermogravimetric analysis (TGA) and Differential Scanning Calorimetry (DSC) scans for L-glutamic acid and TMP free base.
Figure 6.
DSC and TGA scans for TMP D-glutamate and TMP L-glutamate salts. 2–5 mg of the sample was heated to 300 °C at rate of 10 °C/min (n = 3).
Figure 6.
DSC and TGA scans for TMP D-glutamate and TMP L-glutamate salts. 2–5 mg of the sample was heated to 300 °C at rate of 10 °C/min (n = 3).
On the other hand, TMP D-glutamate salt is partially crystalline and the amorphous portion of the salt starts to crystallize at 140 °C. Moisture loss appeared as endothermic hump at around 80 °C corresponding to 1.62%. Similar to TMP L-glutamate, TMP D-glutamate showed hydrate loss at 197.22 °C (ΔH = 18.16 J/g) appeared as small peak reflecting the small amount of the hydrate crystals formed during the freeze drying while the rest of the salt was in the amorphous state and crystallizes at 140°C as discussed before. After dehydration, the salt started to melt at 231.74 °C (
Figure 6) similar results were suggested by [
18].
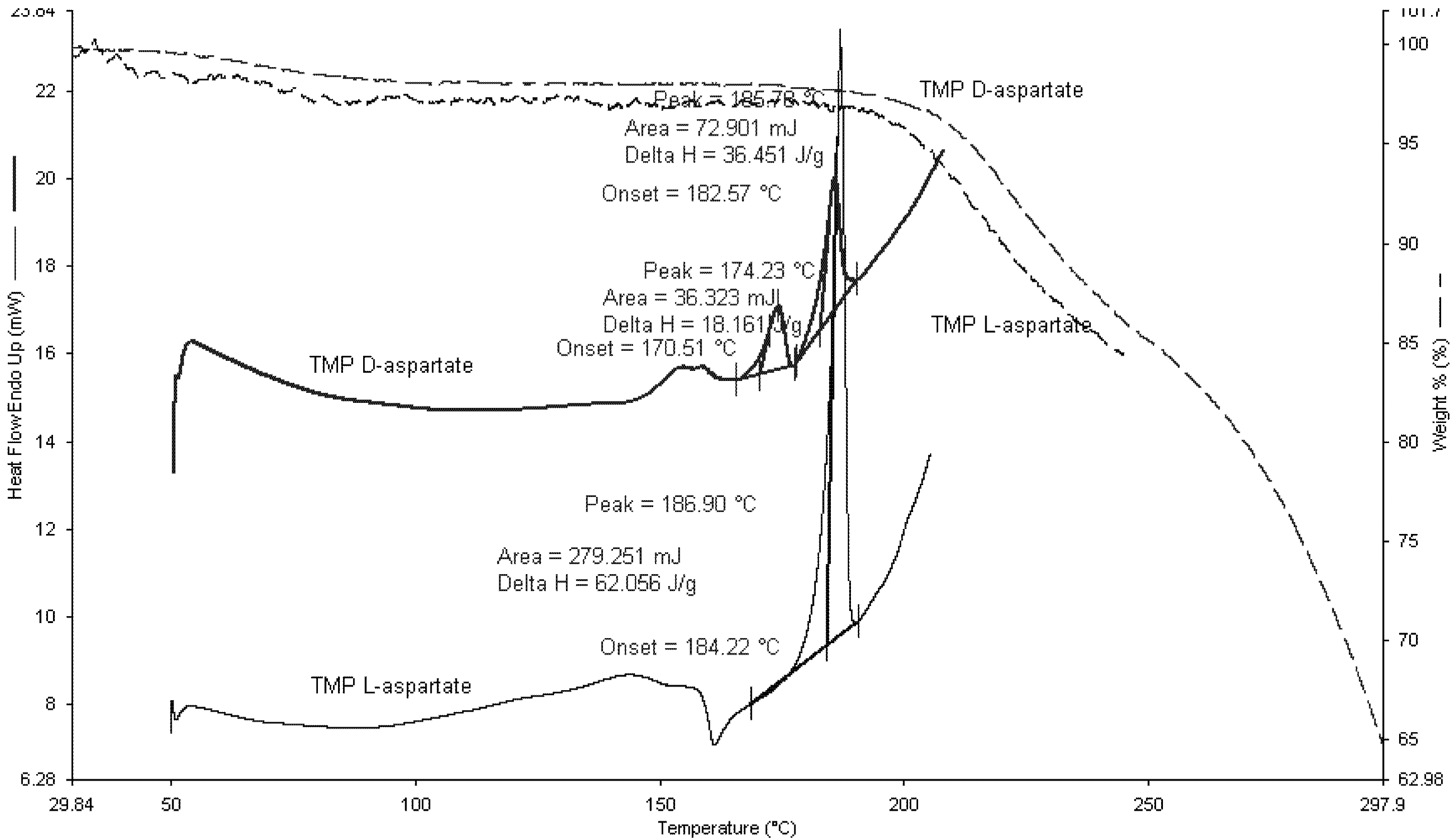
The thermal events of TMP L-aspartate and TMP D-aspartate were studied using TGA and DSC (
Figure 7). TMP L-aspartate showed an up drafted base line between 100–150 °C which corresponds to moisture loss and the salt converted from the amorphous form to the crystalline form at 160 °C followed by crystal melt at 184.22 °C.
Figure 7.
DSC and TGA scans for TMP D-aspartate and TMP L-aspartate. 2–5 mg of the sample was heated to 300 °C at rate of 10 °C/min (n=3).
Figure 7.
DSC and TGA scans for TMP D-aspartate and TMP L-aspartate. 2–5 mg of the sample was heated to 300 °C at rate of 10 °C/min (n=3).
3.2.3. 1HNMR Studies
The electronic and chemical environment of protons are affected during salt formation which could be reflected by the changes in δ. 1HNMR is considered a powerful tool in studying salt formation as it provides insight in to the ratio of molar interaction between the two moieties.
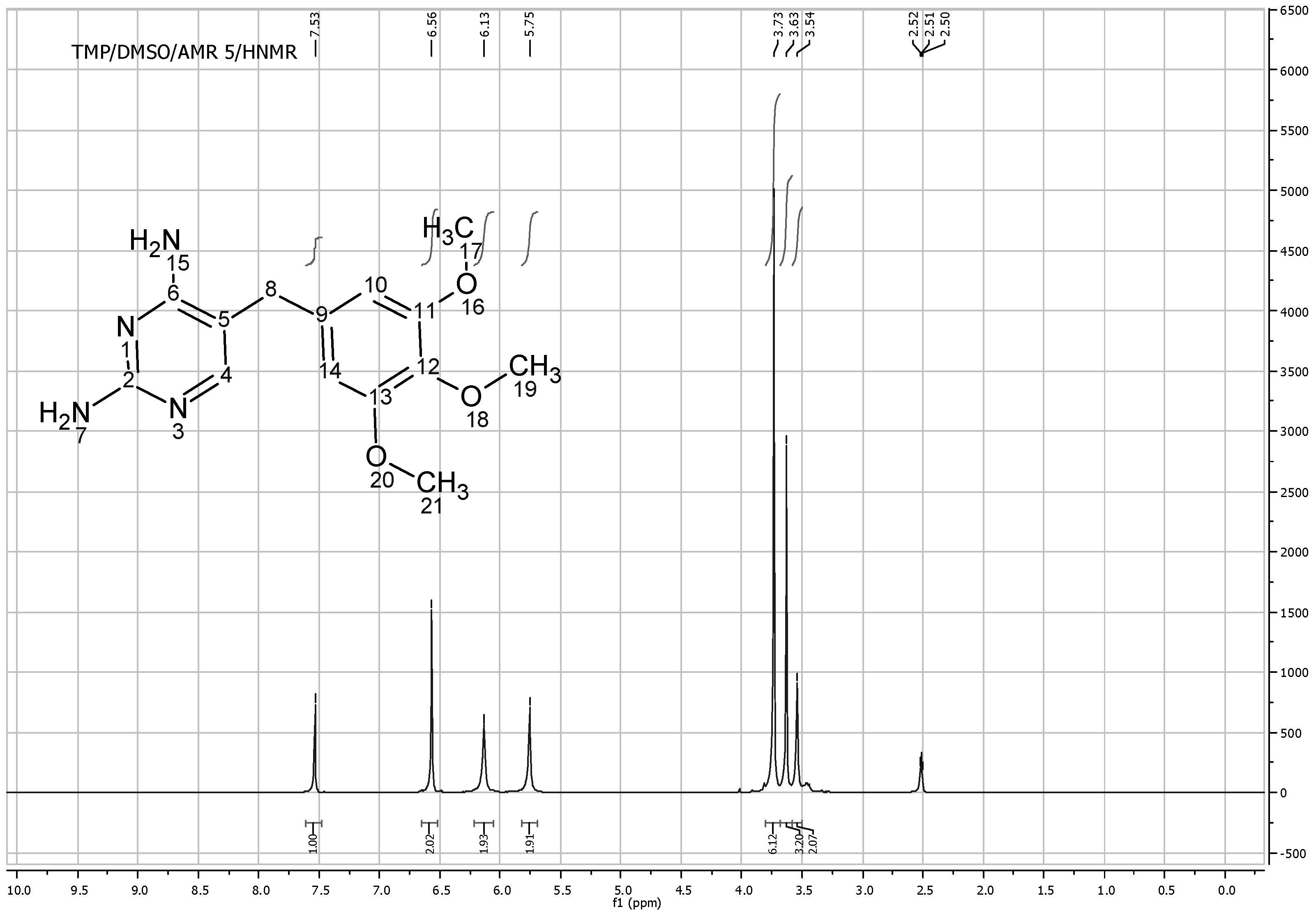
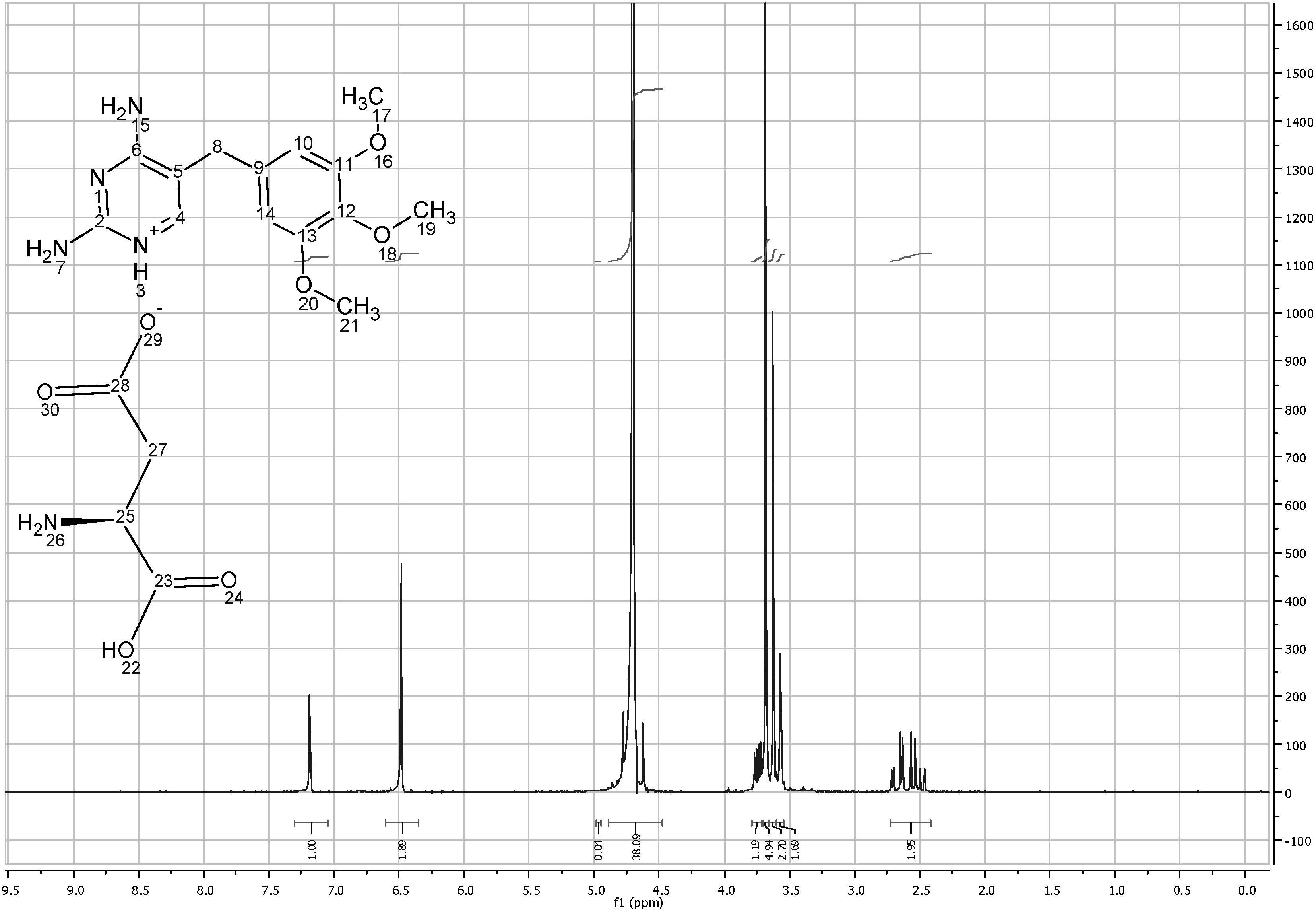
Figure 8 shows the
1HNMR of TMP free base (solubilized in DMSO). At 7.53 ppm frequency one proton of TMP heterocyclic ring C
4H appeared as singlet and the two protons on the aromatic ring C
10H and C
14H appeared at 6.56 ppm. The nine hydrogens of OCH
3 groups appeared as two different peaks; (6H) appeared as a singlet at 3.73 ppm as they have the same surrounding environment, while the other (3H) appeared as a singlet at 3.63 ppm. The two hydrogens of aliphatic (C
8H
2) appeared as singlet at 3.54 ppm while two singlet bands appeared at 5.7 and 6.2 which correspond to N
5H
2 and N
7H
2 respectively. The total integrated H count for TMP was 18 (
Figure 8).
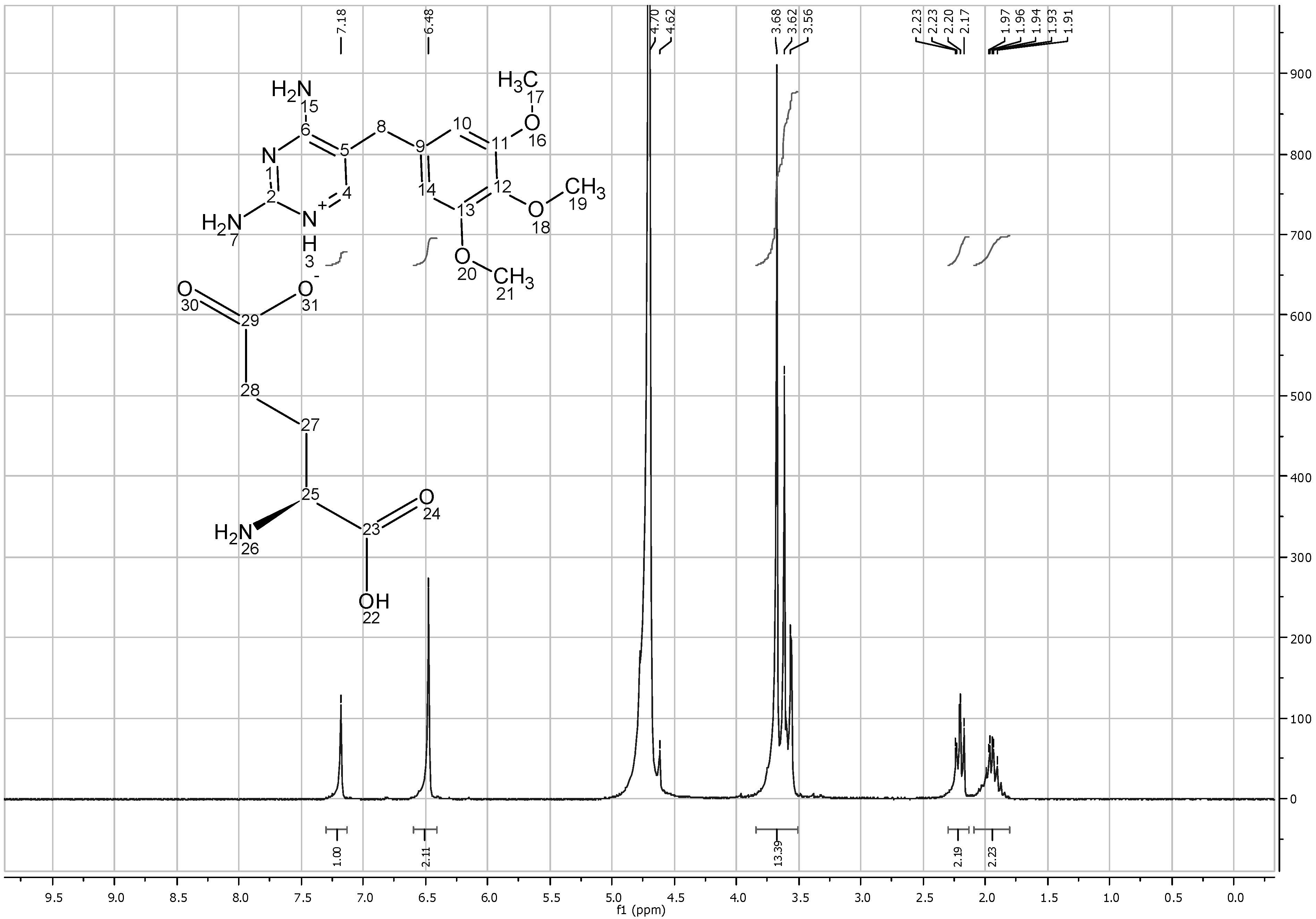
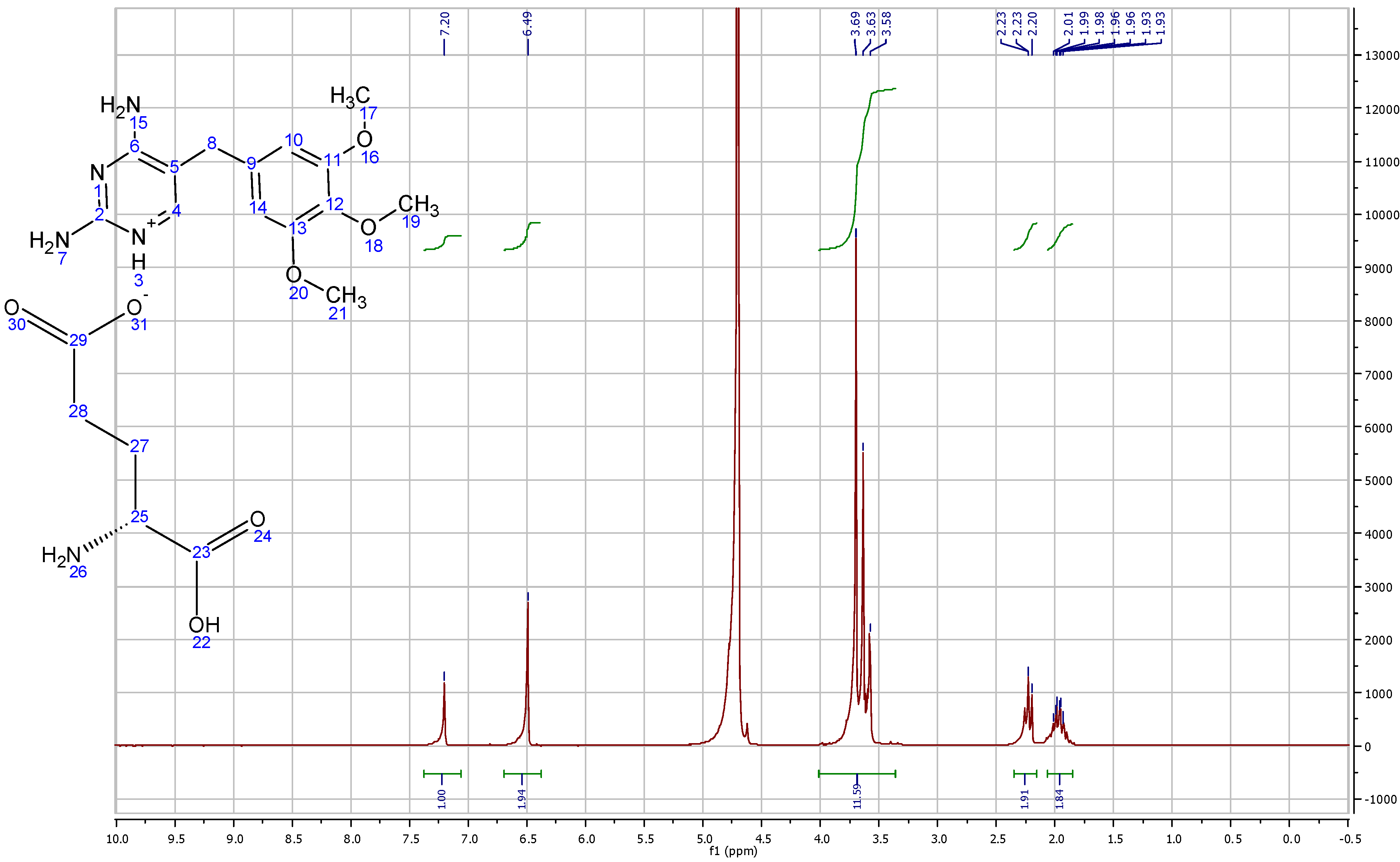
TMP L-aspartate salt (solubilized in D
2O) showed new (3H) assignment at 2.6 corresponding to (C
27H
2) and a highly deshielded C
25H group at 3.75 ppm, which represents the aliphatic chain of L-aspartic acid. As D
2O was used as a solvent for TMP salts, NH
2 groups were masked and the total integration of TMP aspartate salt was 17 instead of 21. These results confirm that one mole of L-aspartic acid interacts with 1 mole of TMP to form the salt (
i.e, 1:1 ratio) as shown in
Figure 9.
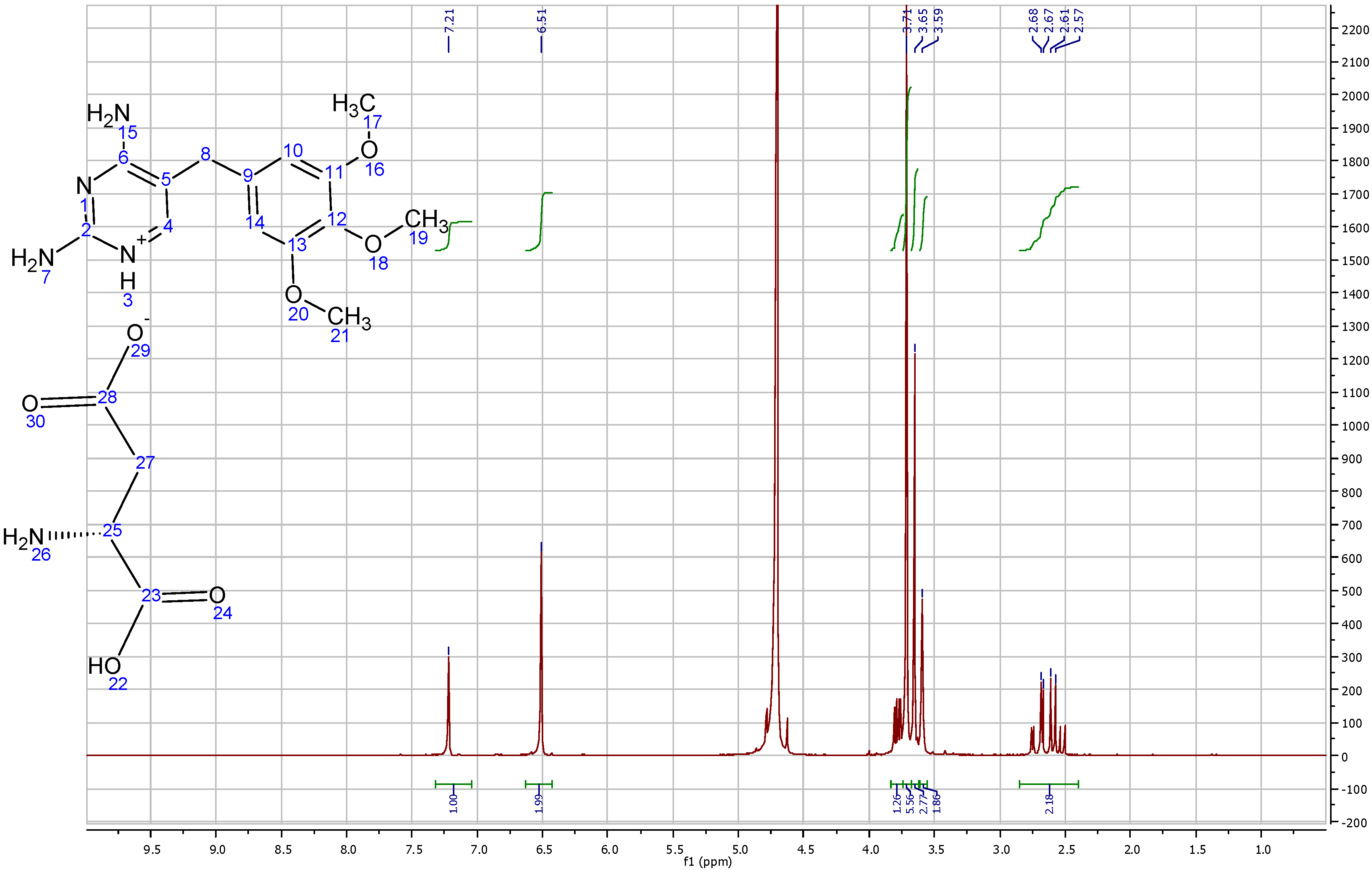
On the other hand, TMP L-glutamate had 5 new protons representing the aliphatic chain of the amino acid. (C
28H
2, C
27H
2) appeared at 1.96 and 2.2 ppm, while the third C
25H group appeared at around 3.6 ppm due to the deshielding by NH
2 group. The total integration also shows that 1 mole of L-glutamic acid interacts with 1 mole of TMP (
Figure 10).
Figure 8.
1HNMR spectra of trimethoprim free base solubilized in DMSO.
Figure 8.
1HNMR spectra of trimethoprim free base solubilized in DMSO.
Figure 9.
1HNMR spectra of trimethoprim L-aspartate solubilized in D2O.
Figure 9.
1HNMR spectra of trimethoprim L-aspartate solubilized in D2O.
Figure 10.
1HNMR spectra of trimethoprim L-glutamate solubilized in D2O.
Figure 10.
1HNMR spectra of trimethoprim L-glutamate solubilized in D2O.
1HNMR studies of TMP D-aspartate and TMP D-glutamate salts did not reveal any difference in the spectra between the L and D isomers (
Figure A &
Figure B Appendix 1). The high similarity of the salts formed despite using different isomers suggest that water used to solubilize the amino acids during the preparation step might provide free space for the rotation of the molecules and potentially catalyze racemisation as suggested by [
19].
3.2.4. Aqueous Solubility and Dissolution Studies
TMP is usually used in combination with sulfonamides in various formulations such as suspension, tablets and solution dosage forms. Therefore TMP solubility and dissolution are important determinants for its bioavailability.
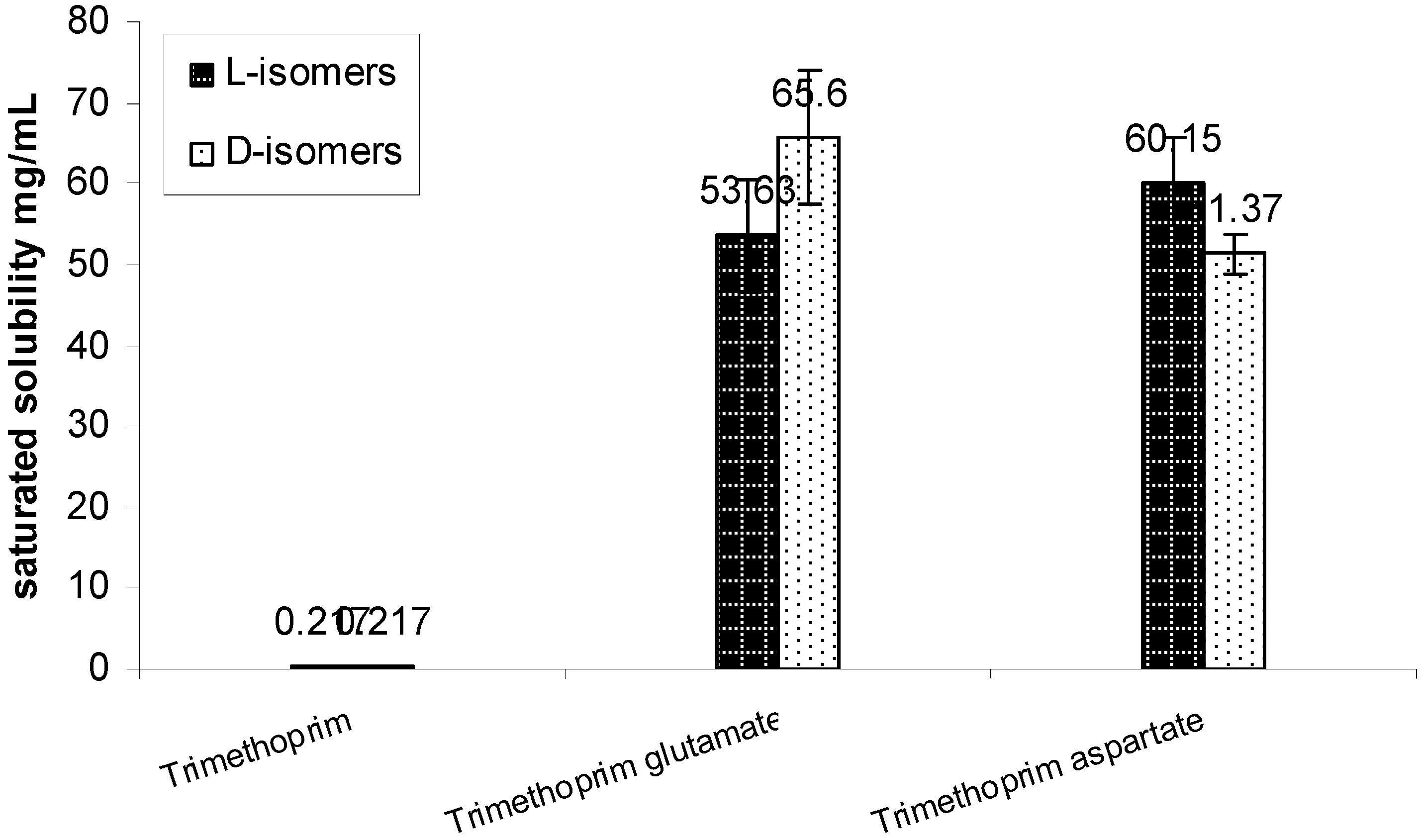
Solubility studies in our laboratory have found that the saturation solubility of TMP free base was 0.217 mg/mL in water. Using acidic amino acids counter ions improved the solubility of the drug by around 280 fold when compared against the free drug. The solubility of the prepared salts was around 55 mg/mL with no significant difference between the L and D isomers for both the counter ions (
Figure 11).
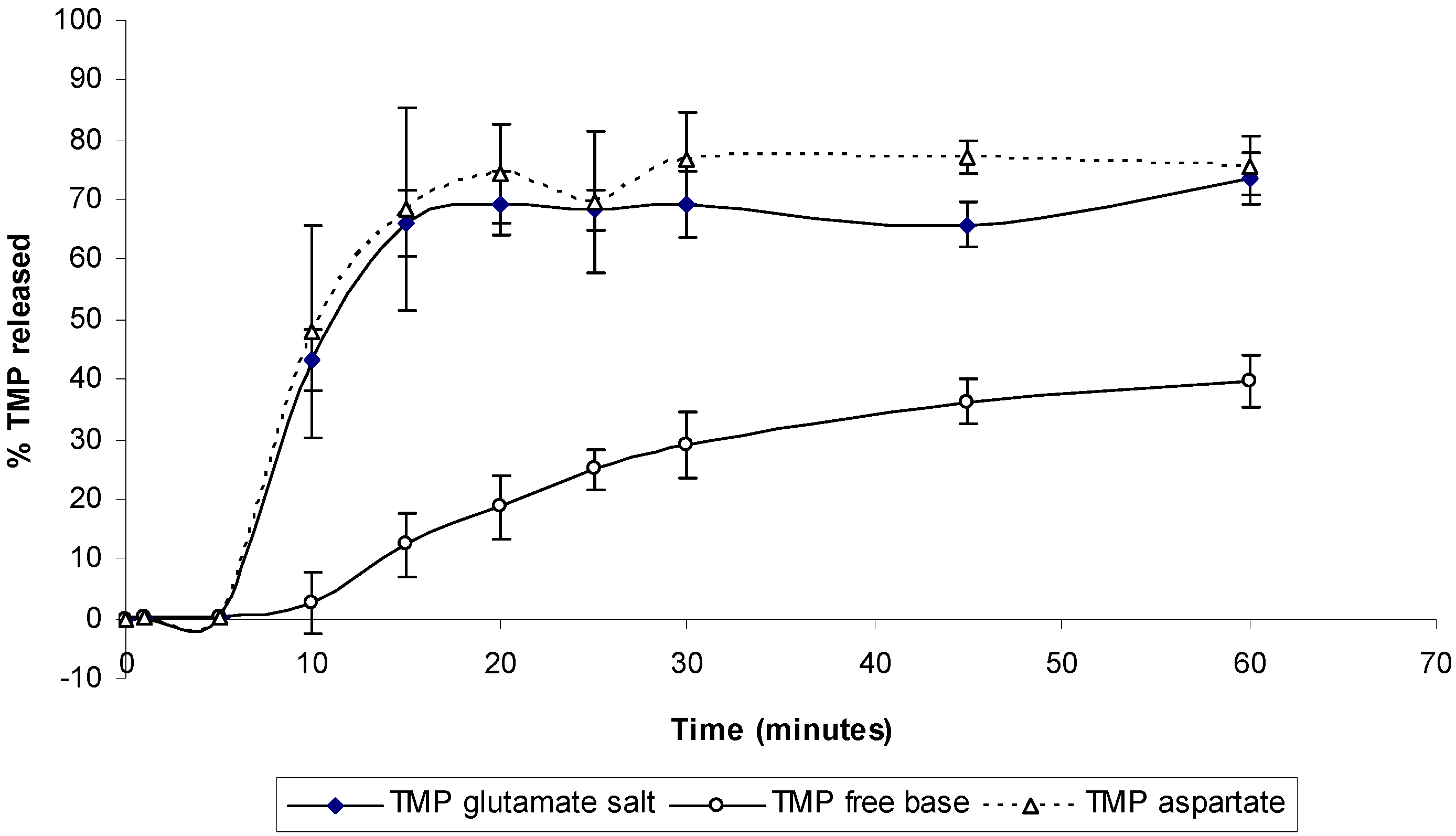
The dissolution profile of TMP and its different salts was also studied (
Figure 12). Dissolution studies show a significant improvement of the salt form when compared to the free base. Around 70% of the TMP was released from aspartate and glutamate salt form after only 10 minutes of the dissolution experiment while only 10% release was seen from TMP free form (
Figure 12). Such changes in the dissolution profile could be attributed to the ability of TMP salts to exert a self-buffering action, which yield different pH values at the dissolving surface in the diffusion layer [
20]. No significant difference was observed between the two salts, which suggest that both salts might have similar buffering capacity and hence similar pH at the diffusion layer.
Figure 11.
Solubility of trimethoprim and its prepared L & D aspartate and glutamate salts mean ± SD (n = 3).
Figure 11.
Solubility of trimethoprim and its prepared L & D aspartate and glutamate salts mean ± SD (n = 3).
Figure 12.
Dissolution profile for TMP free base and its salts in deionized water. Data are mean ± SD (n = 3).
Figure 12.
Dissolution profile for TMP free base and its salts in deionized water. Data are mean ± SD (n = 3).
Table 1.
pH measurements during solubility and dissolution studies.
Table 1.
pH measurements during solubility and dissolution studies.
| | | Formulation |
|---|
| pH measurement | | TMP free base | TMP Aspartate | TMP glutamate |
| Solubility study (Equilibrium) | 7.7 ± 0.05 | 5.075 ± 0.06 | 5.2 ± 0.11 |
| End of dissolution study | 8.6 ± 0.07 | 5.37 ± 0.08 | 5.88 ± 0.05 |
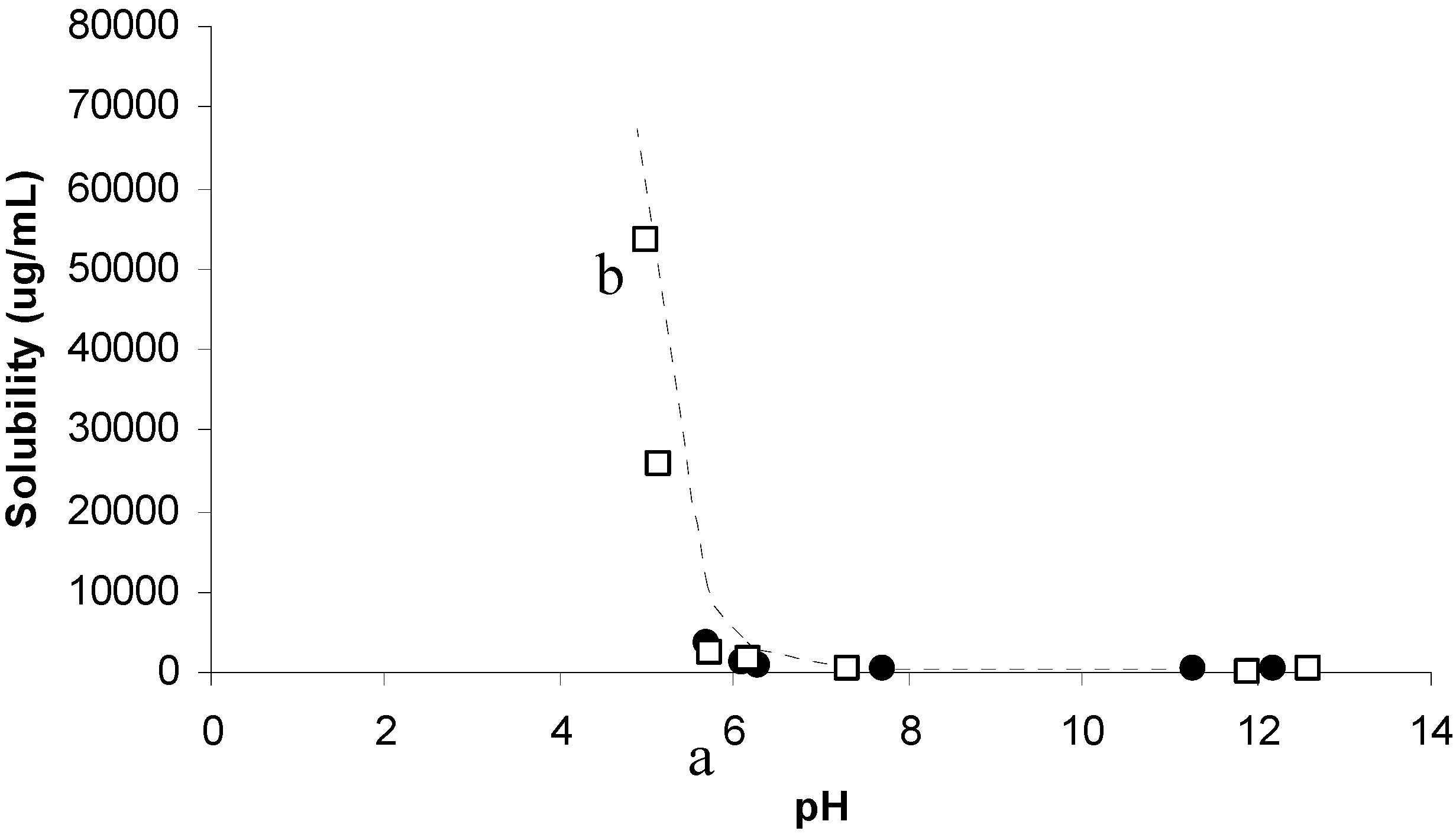
3.2.5. pH-Solubility Profile
TMP pka and intrinsic solubility are 7.3 and 0.19 mg/mL respectively.
Figure 13 shows the pH solubility profile of TMP when TMP glutamate salt and TMP free base were used as starting materials for determination of solubilities. The intrinsic solubility (Bs) was first determined by increasing the pH of TMP solution to 11.28 using NaOH and Bs was found to be 0.266 mg/mL. When TMP was used as a starting material the solubility was found to increase exponentially upon titrating with glutamic acid (
Figure 13). TMP solubility increased from 0.37 mg/mL at pH 7.7 (point a) to 13.8 mg/mL at pH 4.23.
Figure 13.
pH solubility profile of TMP and TMP-glutamate salt at ambient conditions using free acid (circles) and TMP-glutamate salt (squares) as starting materials. Points b and a represent the saturation solubility of the salt and TMP free base respectively. [Bs] = 266.35µg/mL and pKa = 7.3.
Figure 13.
pH solubility profile of TMP and TMP-glutamate salt at ambient conditions using free acid (circles) and TMP-glutamate salt (squares) as starting materials. Points b and a represent the saturation solubility of the salt and TMP free base respectively. [Bs] = 266.35µg/mL and pKa = 7.3.
On the other hand, when TMP glutamate salt was used as a starting point, the solubility started to decrease upon titrating with NaOH. TMP glutamate solubility was 53.6 mg/mL at pH 5 and decreased to 0.194 mg/mL at pH 11.9. A similar trend was observed upon titrating TMP with aspartic acid (data not shown). Generally, a good agreement was observed between the experimental values (squares and circles) and the theoretical predicted values (dotted line) see Table (2). Theoretical calculations were carried out using Kramer and Flynn [
21] (Equation 1).
Where St is the total solubility at given pH, [Bs] is the intrinsic solubility of the free base and Ka is the acid dissociation constant.
Table 2.
Theoretical calculations of pH solubility profile using (Equation 1). [Bs] = 266.35µg/mL and pKa =7.3.
Table 2.
Theoretical calculations of pH solubility profile using (Equation 1). [Bs] = 266.35µg/mL and pKa =7.3.
| pH | Solubility (µg/mL) |
|---|
| 11.28 | 266.35 |
| 7.7 | 372.35 |
| 6.3 | 2929.63 |
| 6.1 | 4487.37 |
| 5.7 | 10869.11 |
| 4.9 | 67165.4 |
3.2.6. Microbiology Studies
After having significantly improved both the solubility as well as the dissolution characteristics using the salt forms of TMP, the next stage of the work was to evaluate microbiological properties to study if the therapeutic effect of the drug was still retained upon preparation of the different salts.
Interestingly, zone of inhibition studies showed that use of amino acid salts not only retained the anti-bacterial activity against TMP sensitive species;
Escherichia coli but also had a significant increase in the total area of inhibition (
Table 3). To rule out the possible effects of amino acids alone on bacterial growth inhibition, control experiments with the amino acids were performed which resulted in no inhibition of bacterial growth on the plates. Therefore, the higher zone of inhibition can be attributed to the significant improvement in solubility which potentially results in higher permeability across the agar plates resulting in larger zone of inhibition. This could possibly translate into a faster therapeutic effect along with reduced therapeutic dose in a clinical setting.
On the other hand, neither the drug nor the salts showed any activity against TMP resistance species Pseudomonas aeruginosa. This is probably due to the complex structure of Pseudomonas aeruginosa membrane as it is intrinsically resistant to TMP and prevents the entry of both the drug and amino acid salts.
Table 3.
Zone of inhibition studies of TMP and its prepared salts against Escherichia coli and Pseudomonas aeruginosa.
Table 3.
Zone of inhibition studies of TMP and its prepared salts against Escherichia coli and Pseudomonas aeruginosa.
| | Escherichia coli | Pseudomonas aeruginosa |
|---|
| Area (cm2) | % compare to the total plate area | |
|---|
| TMP | 4.03 ± 0.2 | 18.7 | **** |
| TMP aspartate | 4.6 ± 0.2 | 21.4 | **** |
| TMP glutamate | 4.65 ± 0.2 | 21.6 | **** |
Bacterial minimum inhibitory concentration was carried out as well in order to accurately determine the MIC of the TMP and its salts. Two species were used in this study;
Escherichia coli and the intrinsically resistant organism,
Pseudomonas aeruginosa. The MIC of TMP on
E. coli was 0.00048 mM and TMP aspartate and glutamate salt had similar MIC values which confirm our previous finding that salt preparation did not impair the pharmacological function of TMP (
Table 4).
Table 4.
MICs of TMP and its prepared salts against Escherichia coli and Pseudomonas aeruginosa.
Table 4.
MICs of TMP and its prepared salts against Escherichia coli and Pseudomonas aeruginosa.
| | Escherichia coli | Pseudomonas aeruginosa |
|---|
| TMP | 0.00048 mM. | >0.5 mM |
| TMP aspartate | 0.00048 mM | >0.5 mM |
| TMP glutamate | 0.00048 mM | >0.5 mM |