Development of Drug-Loaded Gelatin-Based Hydrogel Films for Impaired Wound Healing in Simulated Chronic Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Gelatin-Based Hydrogel Films
2.2.2. Physicochemical Characterization of the Gelatin-Based Hydrogel Films
- Surface characterization of the films
- Optical properties of the films
- Water vapor transmission rate (WVTR)
- Water contact angle and work of adhesion
- Swelling and dissolution behavior
- Kinetics of 5-FU release
2.2.3. Biological Characterization of the Gelatin-Based Hydrogel Films
- Hemocompatibility studies
- Obtention and extraction of the erythrocytes
- 2.
- Hemolysis assay
- Cytotoxicity studies
- Cell cultures
- 2.
- Cell viability assays
- 3.
- Selectivity toward cancer cells
2.2.4. Statistical Analysis
3. Results
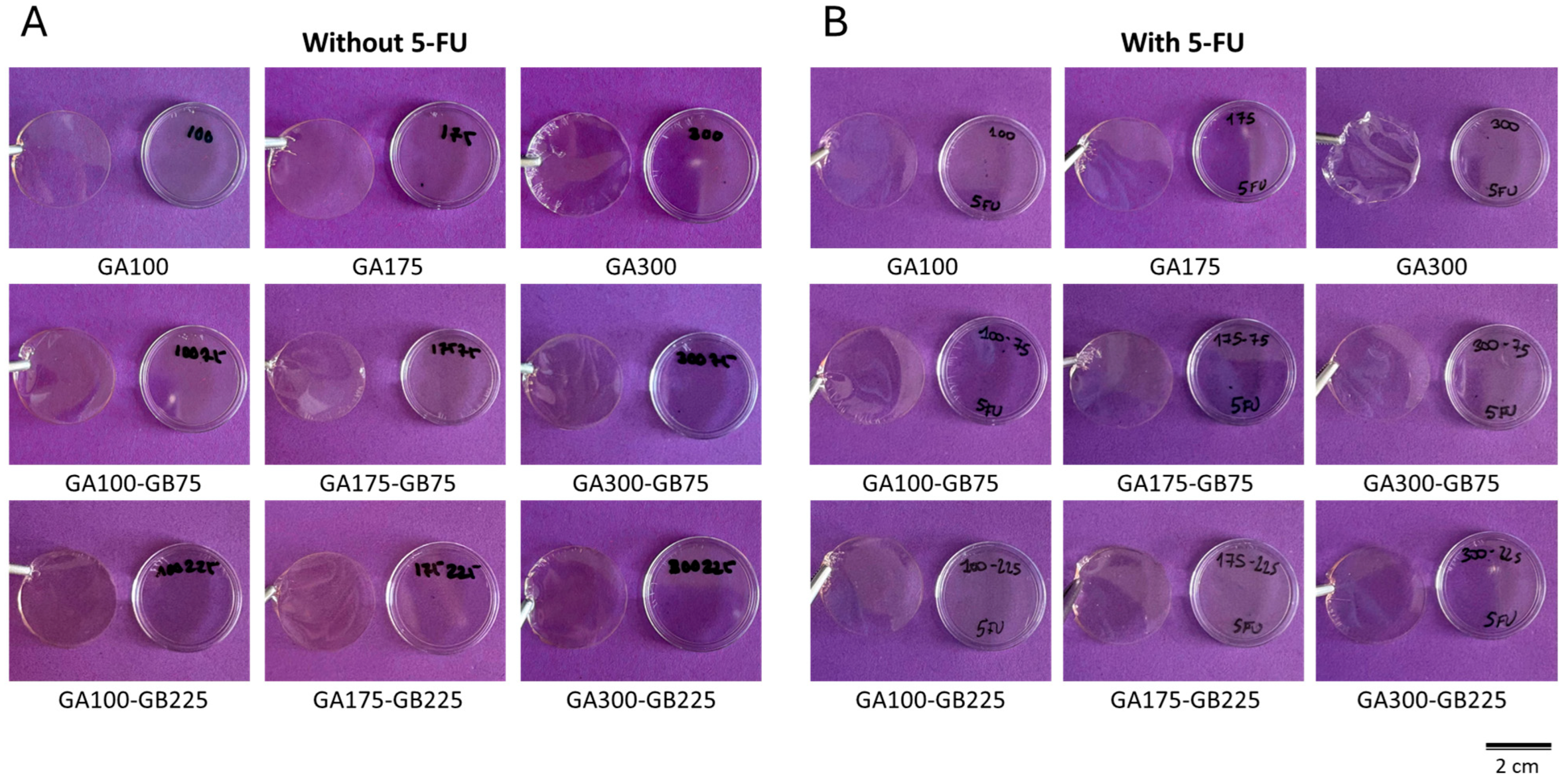
3.1. Preparation of Gelatin-Based Hydrogel Films
3.2. Physical and Physicochemical Characterization of the Gelatin-Based Hydrogel Films
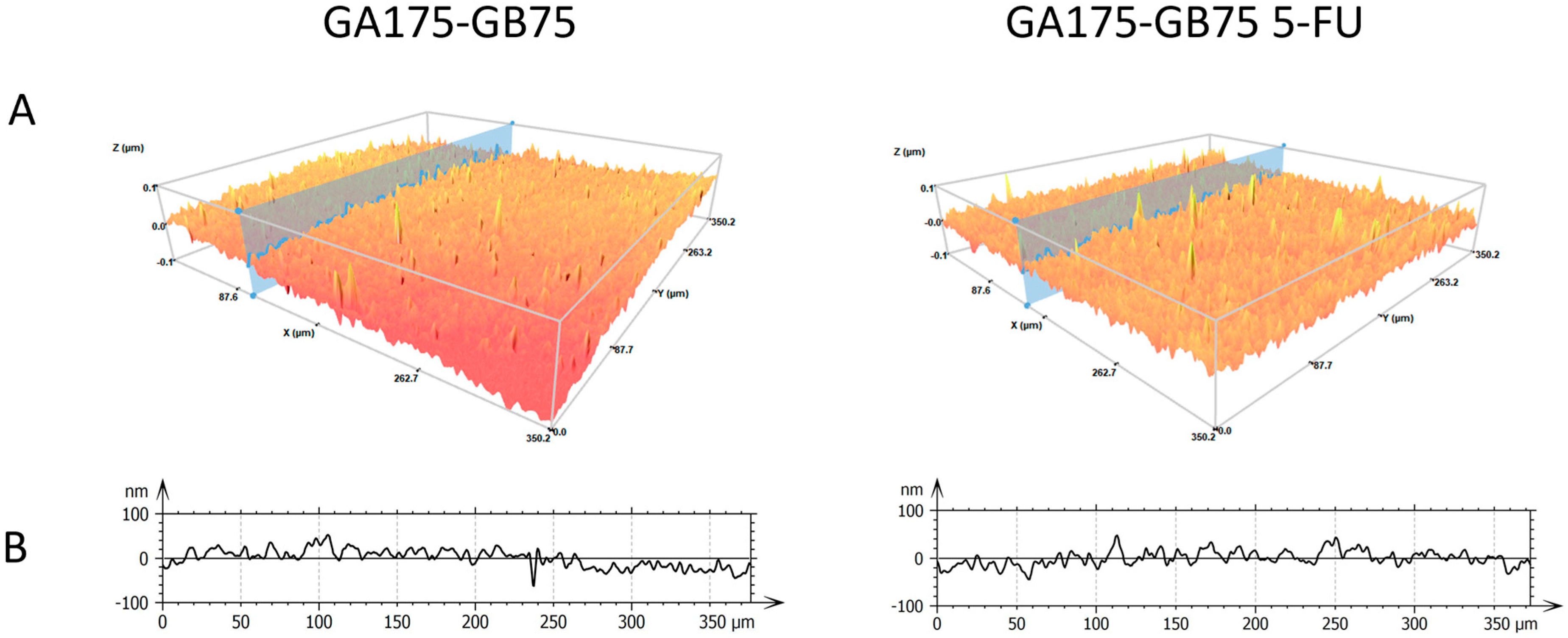
3.2.1. Surface Characterization of the Films
3.2.2. Optical Properties of the Films
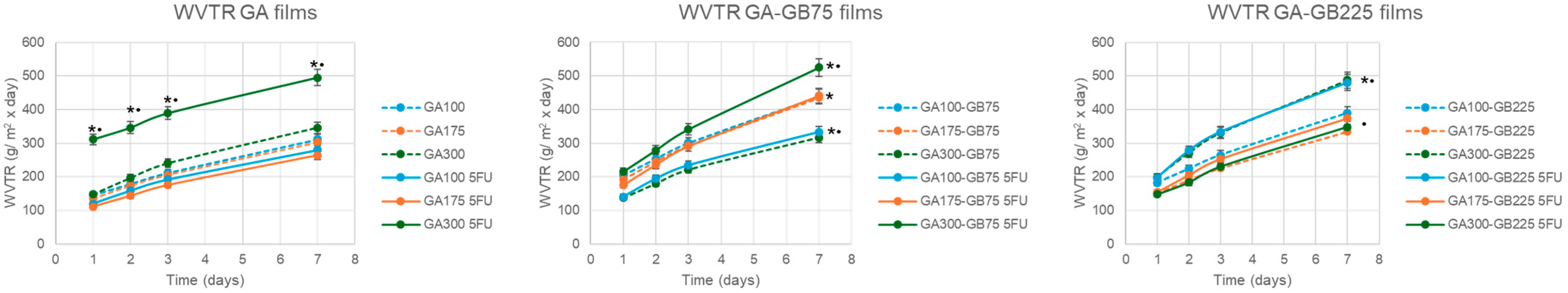
3.2.3. Water Vapor Transmission Rate (WVTR)
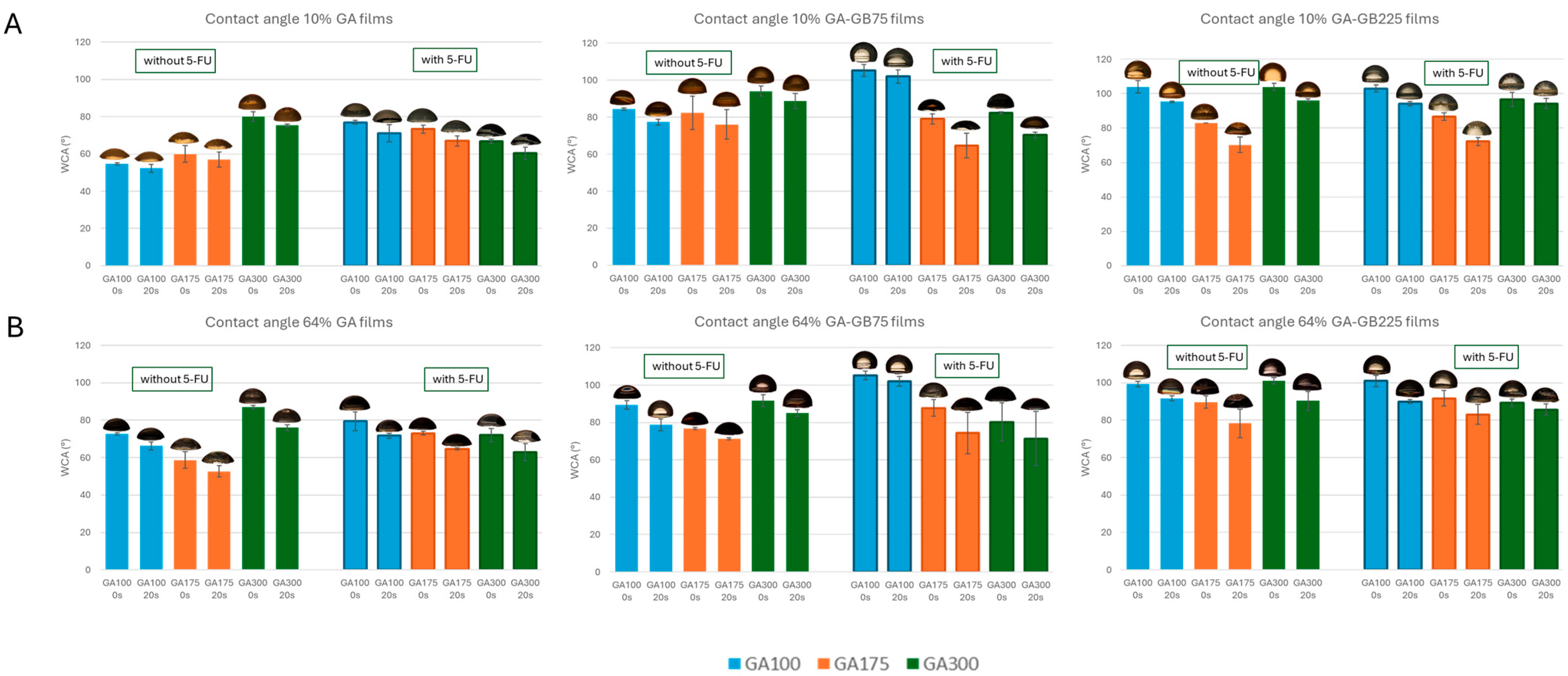
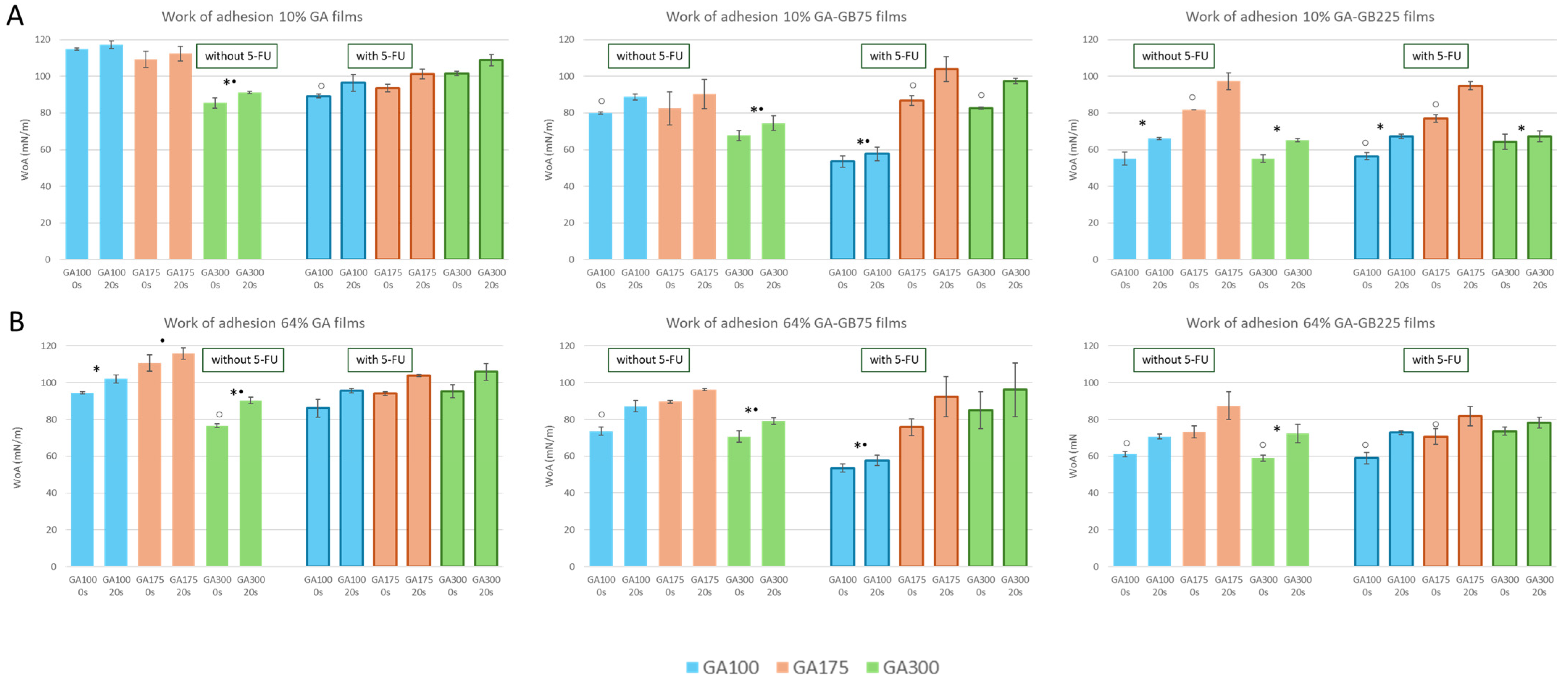
3.2.4. Water Contact Angle (WCA) and Work of Adhesion (WoA)
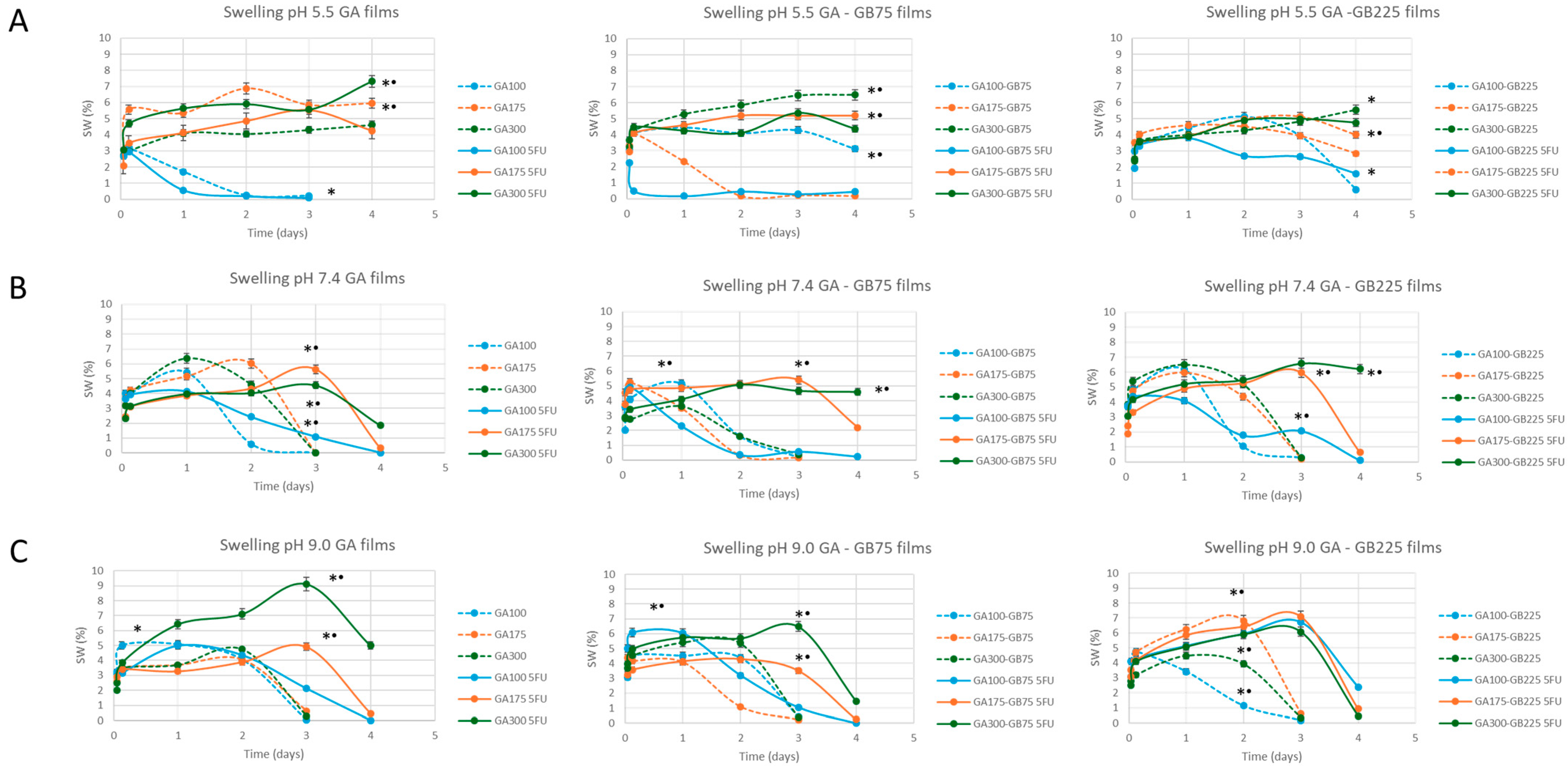
3.2.5. Swelling/Dissolution Behavior
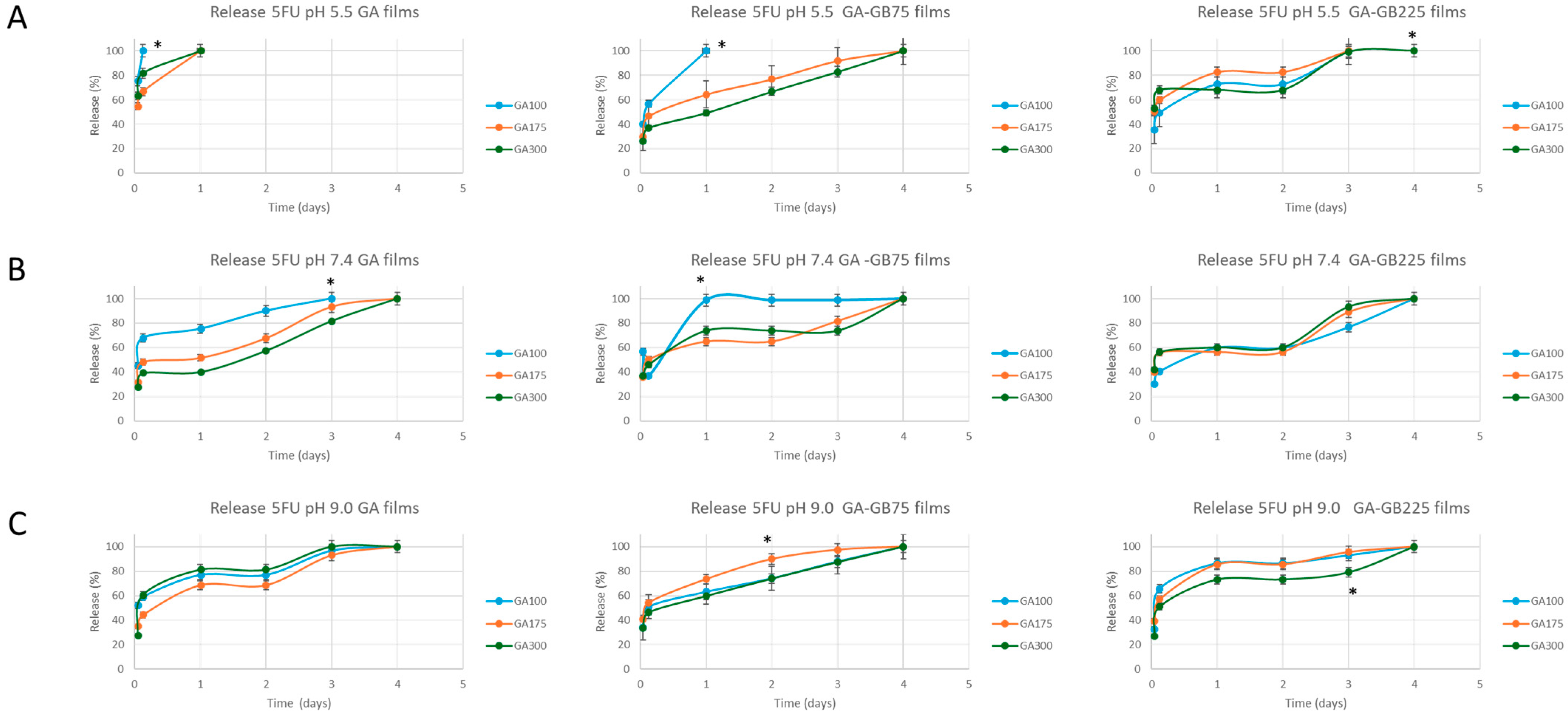
3.2.6. Kinetics of 5-FU Release
3.3. Biological Characterization of the Gelatin-Based Hydrogel Films
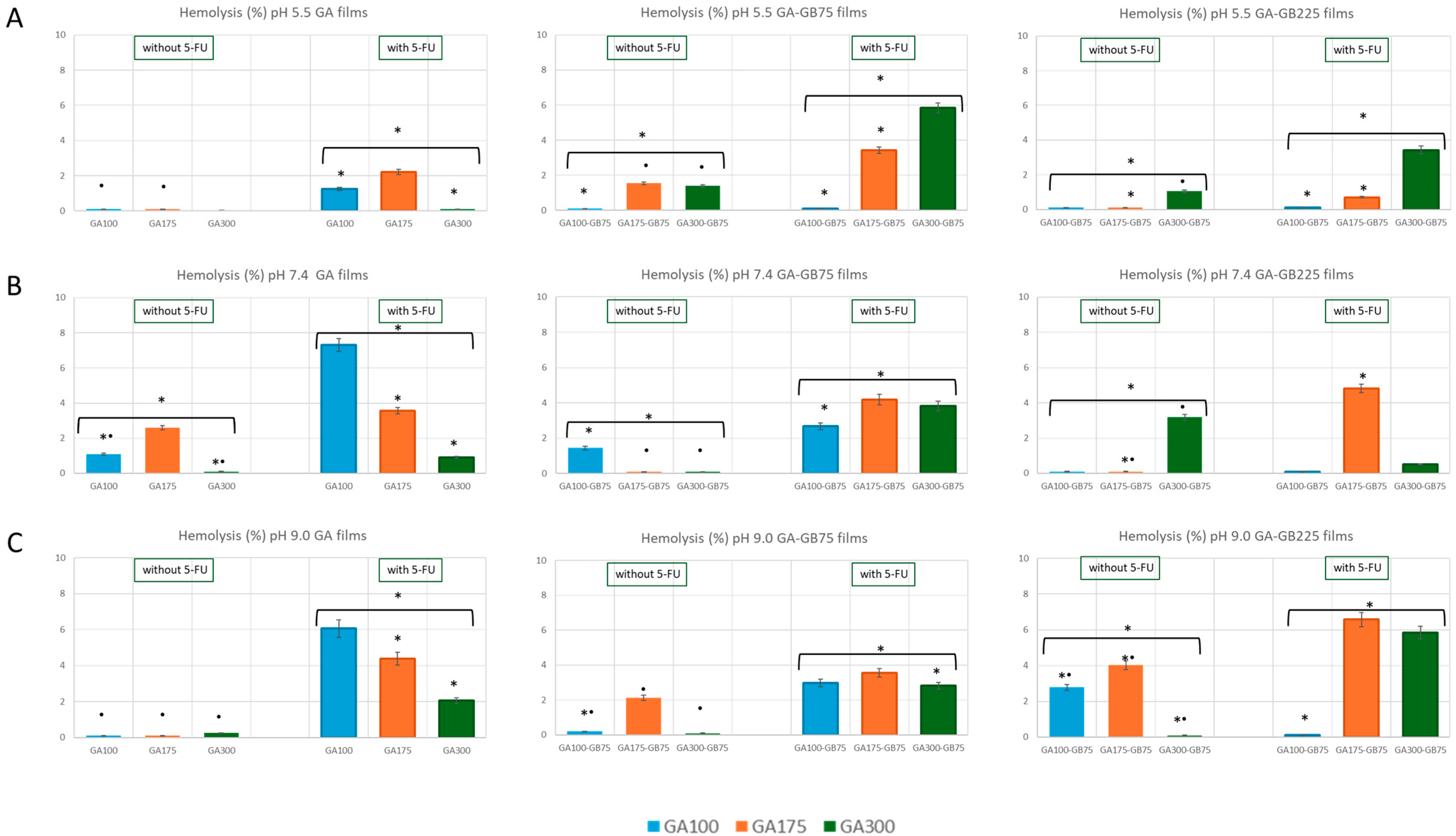
3.3.1. Hemocompatibility Studies
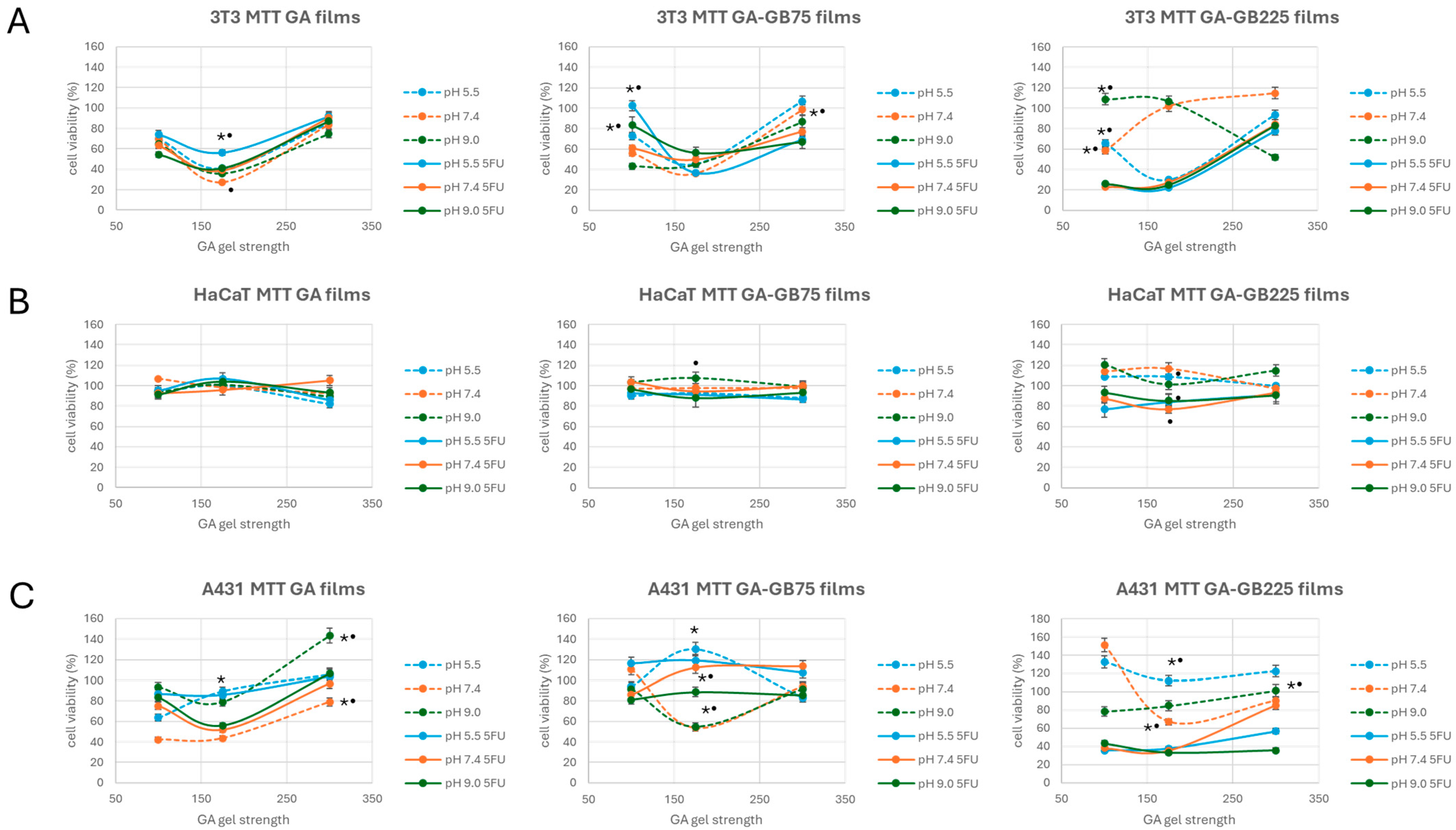
3.3.2. Cytotoxicity Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Falanga, V.; Isseroff, R.R.; Soulika, A.M.; Romanelli, M.; Margolis, D.; Kapp, S.; Granick, M.; Harding, K. Chronic Wounds. Nat. Rev. Dis. Primers 2022, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706. [Google Scholar] [CrossRef]
- Schneider, L.A.; Korber, A.; Grabbe, S.; Dissemond, J. Influence of PH on Wound-Healing: A New Perspective for Wound-Therapy? Arch. Dermatol. Res. 2007, 298, 413–420. [Google Scholar] [CrossRef]
- Morton, L.M.; Phillips, T.J. Wound Healing and Treating Wounds: Differential Diagnosis and Evaluation of Chronic Wounds. J. Am. Acad. Dermatol. 2016, 74, 589–605. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.E.; Eccleston, G.M. Wound Healing Dressings and Drug Delivery Systems: A Review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef]
- Echave, M.C.; Saenz del Burgo, L.; Pedraz, J.L.; Orive, G. Gelatin as Biomaterial for Tissue Engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef] [PubMed]
- Goudie, K.J.; McCreath, S.J.; Parkinson, J.A.; Davidson, C.M.; Liggat, J.J. Investigation of the Influence of PH on the Properties and Morphology of Gelatin Hydrogels. J. Polym. Sci. 2023, 61, 2316–2332. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J. Current Status and Prospects of Gelatin and Its Derivatives in Oncological Applications: Review. Int. J. Biol. Macromol. 2024, 274, 133590. [Google Scholar] [CrossRef]
- Yasmin, R.; Shah, M.; Khan, S.A.; Ali, R. Gelatin Nanoparticles: A Potential Candidate for Medical Applications. Nanotechnol. Rev. 2017, 6, 191–207. [Google Scholar] [CrossRef]
- Ferriol, A.; Morán, M.d.C. Enhanced Performance of Gelatin 5-Fluorouracil-Containing Nanoparticles against Squamous Cell Carcinoma in Simulated Chronic Wounds Conditions. Mater. Sci. Eng. C 2021, 124, 112073. [Google Scholar] [CrossRef] [PubMed]
- Morán, M.d.C.; Porredon, C.; Gibert, C. Insight into the Antioxidant Activity of Ascorbic Acid-Containing Gelatin Nanoparticles in Simulated Chronic Wound Conditions. Antioxidants 2024, 13, 299. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Mohanty, M.; Umashankar, P.R.; Jayakrishnan, A. Evaluation of an in Situ Forming Hydrogel Wound Dressing Based on Oxidized Alginate and Gelatin. Biomaterials 2005, 26, 6335–6342. [Google Scholar] [CrossRef]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Francis, F.J. Wiley Encyclopedia of Food Science and Technology; John Wiley and Sons: Hoboken, NJ, USA, 1999; p. 3130. [Google Scholar]
- Trent, J.T.; Kirsner, R.S. Wounds and Malignancy. Adv. Skin. Wound Care 2003, 16, 31–34. [Google Scholar] [CrossRef]
- ISO 25178-2:2021; Geometrical Product Specifications (GPS)—Surface Texture: Area—Part 2: Terms, Definitions and Surface Texture Parameters. International Organisation for Standardization: Geneva, Switzerland, 2021.
- Han, J.H.; Floros, J.D. Casting Antimicrobial Packaging Films and Measuring Their Physical Properties and Antimicrobial Activity. J. Plast. Film Sheeting 1997, 13, 287–298. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, Y.; Liu, C. Film Transparency and Opacity Measurements. Food Anal. Methods 2022, 15, 2840–2846. [Google Scholar] [CrossRef]
- Ahn, H.H.; Lee, I.W.; Lee, H.B.; Kim, M.S. Cellular Behavior of Human Adipose-Derived Stem Cells on Wettable Gradient Polyethylene Surfaces. Int. J. Mol. Sci. 2014, 15, 2075–2086. [Google Scholar] [CrossRef]
- Han, B.; Wang, P.; Jin, H.; Hou, Z.; Bai, X. Wettability and Surface Energy of Parylene F Deposited on PDMS. Phys. Lett. A 2020, 384, 126628. [Google Scholar] [CrossRef]
- Dowling, D.P.; Miller, I.S.; Ardhaoui, M.; Gallagher, W.M. Effect of Surface Wettability and Topography on the Adhesion of Osteosarcoma Cells on Plasma-Modified Polystyrene. J. Biomater. Appl. 2011, 26, 327–347. [Google Scholar] [CrossRef]
- ASTM F756-17; Standard Practice for Assessment of Hemolytic Properties of Materials. ASTM International: West Conshohocken, PA, USA, 2017.
- Focaccetti, C.; Bruno, A.; Magnani, E.; Bartolini, D.; Principi, E.; Dallaglio, K.; Bucci, E.O.; Finzi, G.; Sessa, F.; Noonan, D.M.; et al. Effects of 5-Fluorouracil on Morphology, Cell Cycle, Proliferation, Apoptosis, Autophagy and ROS Production in Endothelial Cells and Cardiomyocytes. PLoS ONE 2015, 10, e0115686. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, Characterization, and Applications: A Review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Cao, H.; Wang, J.; Hao, Z.; Zhao, D. Gelatin-Based Biomaterials and Gelatin as an Additive for Chronic Wound Repair. Front. Pharmacol. 2024, 15, 1398939. [Google Scholar] [CrossRef]
- Maikovych, O.; Pasetto, P.; Nosova, N.; Kudina, O.; Ostapiv, D.; Samaryk, V.; Varvarenko, S. Functional Properties of Gelatin–Alginate Hydrogels for Use in Chronic Wound Healing Applications. Gels 2025, 11, 174. [Google Scholar] [CrossRef]
- Lou, X.; Chirila, T.V. Swelling Behavior and Mechanical Properties of Chemically Cross-Linked Gelatin Gels for Biomedical Use. J. Biomater. Appl. 1999, 14, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Chen, Q.; Wang, Q.; Zhang, S.; Liu, J.; Yang, L.; Ma, W.; Li, W.; Tan, P.; Yang, G. Gelatin-Based Adaptive Injectable Nanocomposite Hydrogel for Closure of Irregular Wounds and Immunoregulation in Diabetic Wound Healing. Int. J. Biol. Macromol. 2025, 315, 144313. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Yao, Y.; Yim, E.K.F. The Effects of Surface Topography Modification on Hydrogel Properties. APL Bioeng. 2021, 5, 31509. [Google Scholar] [CrossRef] [PubMed]
- Rusu, A.G.; Nita, L.E.; Simionescu, N.; Ghilan, A.; Chiriac, A.P.; Mititelu-Tartau, L. Enzymatically-Crosslinked Gelatin Hydrogels with Nanostructured Architecture and Self-Healing Performance for Potential Use as Wound Dressings. Polymers 2023, 15, 780. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in Pharmaceutical Formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Feng, W.; Wang, Z. Tailoring the Swelling-Shrinkable Behavior of Hydrogels for Biomedical Applications. Adv. Sci. 2023, 10, 2303326. [Google Scholar] [CrossRef]
- Rezvanian, M.; Ahmad, N.; Mohd Amin, M.C.I.; Ng, S.F. Optimization, Characterization, and in Vitro Assessment of Alginate-Pectin Ionic Cross-Linked Hydrogel Film for Wound Dressing Applications. Int. J. Biol. Macromol. 2017, 97, 131–140. [Google Scholar] [CrossRef]
- Atiyeh, B.S.; Hayek, S.N.; Gunn, S.W. New Technologies for Burn Wound Closure and Healing—Review of the Literature. Burns 2005, 31, 944–956. [Google Scholar] [CrossRef] [PubMed]
- Eaglstein, W.H. Moist Wound Healing with Occlusive Dressings: A Clinical Focus. Dermatol. Surg. 2001, 27, 175–182. [Google Scholar] [CrossRef]
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound Dressings-a Review. Biomedicine 2015, 5, 24–28. [Google Scholar] [CrossRef]
- Arbeiter, D.; Reske, T.; Teske, M.; Bajer, D.; Senz, V.; Schmitz, K.P.; Grabow, N.; Oschatz, S. Influence of Drug Incorporation on the Physico-Chemical Properties of Poly(l-Lactide) Implant Coating Matrices—A Systematic Study. Polymers 2021, 13, 292. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Nair, S.V.; Tamura, H. Biomaterials Based on Chitin and Chitosan in Wound Dressing Applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Ngoc Le, T.T.; Nguyen, A.T.; Thien Le, H.N.; Pham, T.T. Biomedical Materials for Wound Dressing: Recent Advances and Applications. RSC Adv. 2023, 13, 5509. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int. J. Mol. Sci. 2020, 21, 9656. [Google Scholar] [CrossRef]
- Hydrogel Dressing Market. Report Code: SNS/HC/7354. Available online: https://www.snsinsider.com/reports/hydrogel-dressing-market-7354 (accessed on 15 November 2025).
| Films | GA | Thickness (μm) | T600 | TUVB | |
|---|---|---|---|---|---|
| 280 nm | 320 nm | ||||
| GA | 100 | 181.56 (177.68) | 96.36 (98.53) | 19.54 (0) | 69.55 (67.37) |
| 175 | 144.91 (175.15) | 93.16 (92.77) | 18.13 (0) | 65.87 (66.48) | |
| 300 | 151.46 (147.01) | 99.16 (94.83) | 34.37 (0) | 84.93 (87.94) | |
| GA-GB75 | 100 | 228.90 (134.81) | 100.24 (96.71) | 17.47 (0) | 65.14 (67.16) |
| 175 | 181.38 (216.80) | 99.77 (98.24) | 17.91 (0) | 71.00 (61.72) | |
| 300 | 153.02 (229.35) | 103.63 (92.34) | 21.36 (0) | 71.20 (62.05) | |
| GA-GB225 | 100 | 227.70 (208.11) | 95.00 (100.8) | 17.75 (0) | 56.30 (60.83) |
| 175 | 197.58 (138.96) | 70.1 (88.73) | 7.39 (0) | 41.97 (58.52) | |
| 300 | 207.3 (179.43) | 100.64 (87.64) | 17.76 (0) | 68.42 (57.23) | |
| Film | GA | 3T3/A431 pH Value | HaCaT/A431 pH Value | ||||
|---|---|---|---|---|---|---|---|
| 5.5 | 7.4 | 9.0 | 5.5 | 7.4 | 9.0 | ||
| GA | 100 | 0.86 | 0.84 | 0.65 | 1.10 | 1.23 | 2.15 |
| 175 | 0.66 | 0.75 | 0.73 | 1.25 | 1.84 | 1.85 | |
| 300 | 0.89 | 0.93 | 0.82 | 0.83 | 1.08 | 0.88 | |
| GA-GB75 | 100 | 0.88 | 0.71 | 1.03 | 0.79 | 1.20 | 1.19 |
| 175 | 0.31 | 0.44 | 0.64 | 0.76 | 0.84 | 0.99 | |
| 300 | 0.64 | 0.68 | 0.79 | 0.81 | 0.88 | 1.09 | |
| GA-GB225 | 100 | 0.72 | 0.60 | 0.60 | 2.15 | 2.30 | 2.15 |
| 175 | 0.58 | 0.78 | 0.76 | 2.22 | 2.15 | 2.58 | |
| 300 | 1.37 | 0.98 | 2.33 | 1.61 | 1.09 | 2.54 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Morán, M.d.C.; Cocci Grifoni, A.; Cirisano, F.; Ferrari, M. Development of Drug-Loaded Gelatin-Based Hydrogel Films for Impaired Wound Healing in Simulated Chronic Conditions. Pharmaceutics 2026, 18, 43. https://doi.org/10.3390/pharmaceutics18010043
Morán MdC, Cocci Grifoni A, Cirisano F, Ferrari M. Development of Drug-Loaded Gelatin-Based Hydrogel Films for Impaired Wound Healing in Simulated Chronic Conditions. Pharmaceutics. 2026; 18(1):43. https://doi.org/10.3390/pharmaceutics18010043
Chicago/Turabian StyleMorán, María del Carmen, Alessia Cocci Grifoni, Francesca Cirisano, and Michele Ferrari. 2026. "Development of Drug-Loaded Gelatin-Based Hydrogel Films for Impaired Wound Healing in Simulated Chronic Conditions" Pharmaceutics 18, no. 1: 43. https://doi.org/10.3390/pharmaceutics18010043
APA StyleMorán, M. d. C., Cocci Grifoni, A., Cirisano, F., & Ferrari, M. (2026). Development of Drug-Loaded Gelatin-Based Hydrogel Films for Impaired Wound Healing in Simulated Chronic Conditions. Pharmaceutics, 18(1), 43. https://doi.org/10.3390/pharmaceutics18010043









