Preformulation Study of Controlled-Release Galantamine Matrix Tablets Containing Polyethylene Oxide, Hydroxypropyl Methylcellulose, and Ethylcellulose
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Powder Mixtures
2.3. Evaluation of Excipient Compatibility Through Thermal and Spectroscopic Analysis
2.4. Physical Analysis of Powder Excipients and Formulations
2.4.1. Particle Size and Size Distribution
2.4.2. Granulometric Characterization Based on Flowability Indices and Porosity
2.5. Compressibility, Compactability, and Tabletability Analysis
2.5.1. Powder Compression
2.5.2. Kawakita Model
2.5.3. Heckel Model
2.5.4. Leuenberger Model
2.6. Preparation and Characterization of Controlled-Release Matrix Tablets
2.6.1. Tablet Physical Characterization
2.6.2. In Vitro Dissolution Tests
2.7. Data Analysis
3. Results and Discussion
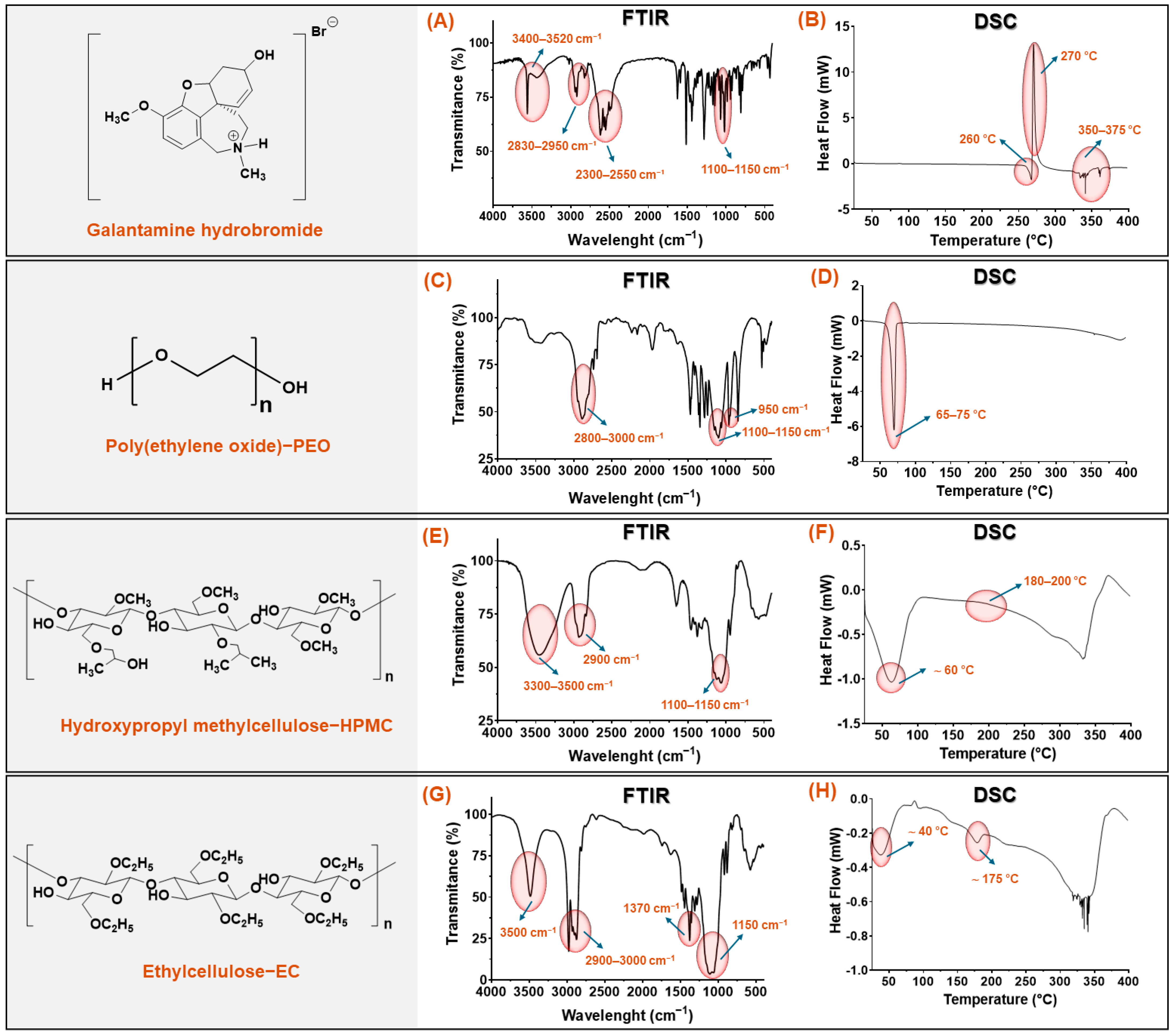
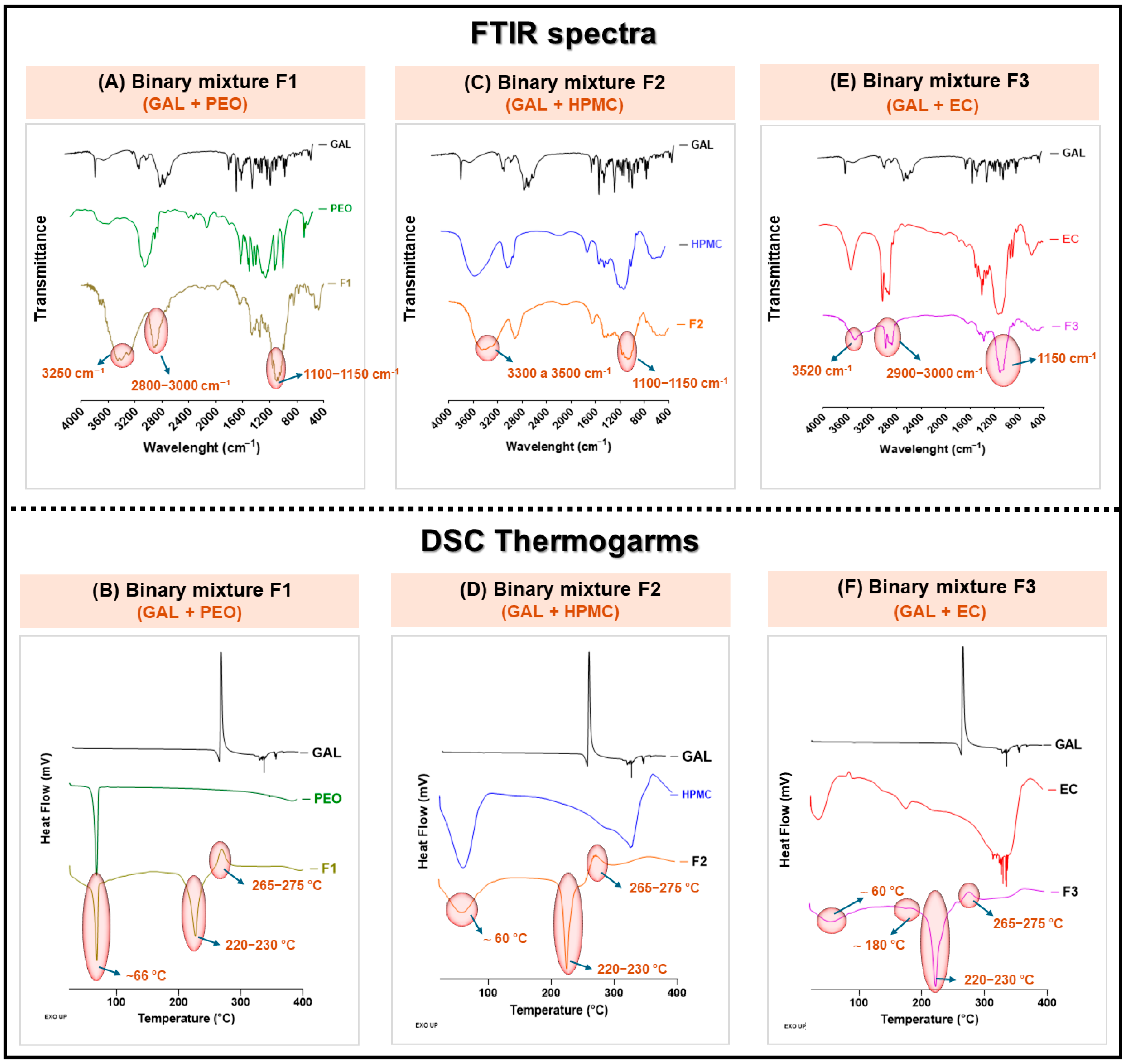
3.1. Evaluation of Excipient Compatibility Through Thermal and Spectroscopic Analysis
3.1.1. Pure Formulation Ingredients
3.1.2. Binary Mixtures of GAL Hydrobromide and Controlled-Release Polymers
3.2. Physical Analysis of Powder Excipients and Formulations
3.2.1. Particle Size and Size Distribution
3.2.2. Granulometric Characterization Based on Flowability Indices and Porosity
3.2.3. Compressibility, Compactability, and Tabletability Analysis
3.3. Galantamine Modified-Release Tablets Characterization
3.3.1. Tablet Physical Characterization
3.3.2. In Vitro Dissolution Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| API | Active Pharmaceutical Ingredient |
| BCS | Biopharmaceutics Classification System |
| CI | Carr Index |
| DE% | Dissolution Efficiency Percentage |
| DSC | Differential Scanning Calorimetry |
| EC | Ethylcellulose |
| FTIR | Fourier Transform Infrared Spectroscopy |
| GAL | Galantamine |
| HPLC | High-Performance Liquid Chromatography |
| HPMC | Hydroxypropyl Methylcellulose |
| HR | Hausner Ratio |
| Kp | Kilopond (Tablet Hardness Unit) |
| PBS | Phosphate Buffered Saline |
| PEO | Polyethylene Oxide |
| Py | Yield Pressure |
| R2 | Coefficient of Determination (Regression Analysis) |
| SLS | Static Light Scattering |
| Tmax | Maximum Tensile Strength |
| USP | United States Pharmacopeia |
References
- Lim, A.W.Y.; Schneider, L.; Loy, C. Galantamine for Dementia Due to Alzheimer’s Disease and Mild Cognitive Impairment. Cochrane Database Syst. Rev. 2024, 2024, CD001747. [Google Scholar] [CrossRef]
- Janssen Research Foundation. Galantamine: Clinical Pharmacology and Biopharmaceutics Review; Janssen Research Foundation: Raritan, NJ, USA, 2000. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/21-224_REMINYL_biopharmr.pdf (accessed on 19 May 2025).
- Zhao, Q.; Janssens, L.; Verhaeghe, T.; Brashear, H.R.; Truyen, L. Pharmacokinetics of Extended-Release and Immediate-Release Formulations of Galantamine at Steady State in Healthy Volunteers. Curr. Med. Res. Opin. 2005, 21, 1547–1554. [Google Scholar] [CrossRef]
- Reddi Sree, R.; Kalyan, M.; Anand, N.; Mani, S.; Gorantla, V.R.; Sakharkar, M.K.; Song, B.J.; Chidambaram, S.B. Newer Therapeutic Approaches in Treating Alzheimer’s Disease: A Comprehensive Review. ACS Omega 2025, 10, 5148–5171. [Google Scholar] [CrossRef]
- Xing, B.; Yang, J.; Hua, H.; Jiang, R. Rapid Health Technology Assessment of Galantamine for the Treatment of Alzheimer’s Disease: A Review. Medicine 2025, 104, e42744. [Google Scholar] [CrossRef] [PubMed]
- Jaqua, E.E.; Tran, M.-L.N.; Hanna, M. Alzheimer Disease: Treatment of Cognitive and Functional Symptoms. Am. Fam. Physician 2024, 110, 281–293. [Google Scholar] [PubMed]
- Varadharajan, A.; Davis, A.D.; Ghosh, A.; Jagtap, T.; Xavier, A.; Menon, A.J.; Roy, D.; Gandhi, S.; Gregor, T. Guidelines for Pharmacotherapy in Alzheimer’s Disease—A Primer on FDA-Approved Drugs. J. Neurosci. Rural. Pract. 2023, 14, 566–573. [Google Scholar] [CrossRef]
- Ono, A.; Tomono, T.; Ogihara, T.; Terada, K.; Sugano, K. Investigation of Biopharmaceutical and Physicochemical Drug Properties Suitable for Orally Disintegrating Tablets. ADMET DMPK 2016, 4, 335–360. [Google Scholar] [CrossRef][Green Version]
- Huang, F.; Fu, Y. A Review of Clinical Pharmacokinetics and Pharmacodynamics of Galantamine, a Reversible Acetylcholinesterase Inhibitor for the Treatment of Alzheimer s Disease, in Healthy Subjects and Patients. Curr. Clin. Pharmacol. 2010, 5, 115–124. [Google Scholar] [CrossRef]
- Marucci, G.; Buccioni, M.; Ben, D.D.; Lambertucci, C.; Volpini, R.; Amenta, F. Efficacy of Acetylcholinesterase Inhibitors in Alzheimer’s Disease. Neuropharmacology 2021, 190, 108352. [Google Scholar] [CrossRef]
- Chbib, C.; Rashid, M.A.; Shah, S.M.; Kazi, M.; Uddin, M.N. Evaluating the Release of Different Commercial Orally Modified Niacin Formulations In Vitro. Polymers 2023, 15, 3046. [Google Scholar] [CrossRef]
- Wen, X.; Deng, Z.; Xu, Y.; Yan, G.; Deng, X.; Wu, L.; Liang, Q.; Fang, F.; Feng, X.; Yu, M.; et al. Preparation and in Vitro/in Vivo Evaluation of Orally Disintegrating/Modified-Release Praziquantel Tablets. Pharmaceutics 2021, 13, 1567. [Google Scholar] [CrossRef]
- Mubeen, I.; Zaman, M.; Farooq, M.; Mehmood, A.; Azeez, F.K.; Rehman, W.; Akhtar, S.; Chaudhry, M.A.; Butt, M.H.; Shamim, Q.U.A.; et al. Formulation of Modified-Release Bilayer Tablets of Atorvastatin and Ezetimibe: An In-Vitro and In-Vivo Analysis. Polymers 2022, 14, 3770. [Google Scholar] [CrossRef]
- Hollenbeck, R.G.; Fahmy, R.; Martinez, M.N.; Ibrahim, A.; Hoag, S.W. Design and Process Considerations for Preparation of Modified Release Ivermectin and Praziquantel Tablets by Wet Granulation. AAPS PharmSciTech 2025, 26, 43. [Google Scholar] [CrossRef]
- Vlachou, M.; Siamidi, A.; Anagnostopoulou, D.; Christodoulou, E.; Bikiaris, N.N. Modified Release of the Pineal Hormone Melatonin from Matrix Tablets Containing Poly (L-Lactic Acid) and Its PLA-Co-PEAd and PLA-Co-PBAd Copolymers. Polymers 2022, 14, 1504. [Google Scholar] [CrossRef] [PubMed]
- Khan, B.; Choi, H.I.; Ryu, J.S.; Noh, H.Y.; Shah, F.A.; Khan, N.; Ansari, M.M.; Zeb, A.; Kim, J.K. Core-Shell Tablets Designed for Modified and Sequential Release of Ibuprofen and Rabeprazole. Int. J. Pharm. 2024, 666, 124839. [Google Scholar] [CrossRef]
- Hussain, A.; Misbah, M.; Abbas, N.; Irfan, M.; Arshad, M.S.; Shamim, R.; Bukhari, N.I.; Mahmood, F. Design and in Vitro Characterization of Orally Disintegrating Modified Release Tablets of Naproxen Sodium. Turk. J. Pharm. Sci. 2020, 17, 486–491. [Google Scholar] [CrossRef]
- Kostewicz, E.S.; Vertzoni, M.; Benson, H.A.E.; Roberts, M.S. Oral Drug Delivery for Modified Release Formulations; John Wiley and Sons Inc.: Hoboken, NJ, USA, 2021; ISBN 9781119772729. [Google Scholar]
- Bertocchi, P.; Antoniella, E.; Valvo, L.; Alimonti, S.; Memoli, A. Diclofenac Sodium Multisource Prolonged Release Tablets—A Comparative Study on the Dissolution Profiles. J. Pharm. Biomed. Anal. 2005, 37, 679–685. [Google Scholar] [CrossRef]
- Harrower, A.D.B. Efficacy of Gliclazide in Comparison with Other Sulphonylureas in the Treatment of NIDDM. Diabetes Res. Clin. Pract. 1991, 14, S65–S67. [Google Scholar] [CrossRef]
- Palmer, K.J.; Brogden, R.N. Gliclazide: An Update of Its Pharmacological Properties and Therapeutic Efficacy in Non-Insulin-Dependent Diabetes Mellitus. Drugs 1993, 46, 92–125. [Google Scholar] [CrossRef] [PubMed]
- Mandal, U.; Ray, K.K.; Gowda, V.; Ghosh, A.; Pal, T.K. In-Vitro and in-Vivo Correlation for Two Gliclazide Extended-Release Tablets. J. Pharm. Pharmacol. 2010, 59, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Ervasti, T.; Simonaho, S.P.; Ketolainen, J.; Forsberg, P.; Fransson, M.; Wikström, H.; Folestad, S.; Lakio, S.; Tajarobi, P.; Abrahmsén-Alami, S. Continuous Manufacturing of Extended Release Tablets via Powder Mixing and Direct Compression. Int. J. Pharm. 2015, 495, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Velasco, M.V.; Muñoz, A.; Jiménez-Castellanos, M.R.; Castellano, I.; Goñi, I.; Gurruchaga, M. In Vitro Evaluation of Sustained-Release Matrix Tablets Prepared with New Modified Polymeric Carbohydrates. Int. J. Pharm. 1996, 136, 107–115. [Google Scholar] [CrossRef]
- Samy, W.; Elnoby, A.; El-Gowelli, H.M.; Elgindy, N. Hybrid Polymeric Matrices for Oral Modified Release of Desvenlafaxine Succinate Tablets. Saudi Pharm. J. 2017, 25, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, L.; Li, J.; Jiang, S.; Wang, Y.; Zhang, X.; Ding, J.; Yu, T.; Mao, S. Insights into the Mechanisms of Chitosan–Anionic Polymers-Based Matrix Tablets for Extended Drug Release. Int. J. Pharm. 2014, 476, 253–265. [Google Scholar] [CrossRef]
- Samaro, A.; Vergaelen, M.; Purino, M.; Tigrine, A.; de la Rosa, V.R.; Goudarzi, N.M.; Boone, M.N.; Vanhoorne, V.; Hoogenboom, R.; Vervaet, C. Poly (2-Alkyl-2-Oxazoline)s: A Polymer Platform to Sustain the Release from Tablets with a High Drug Loading. Mater. Today Bio 2022, 16, 100414. [Google Scholar] [CrossRef]
- Gupta, C.R.; Kishore, G.K.; Ratna, J.V. Development and Evaluation of Aceclofenac Matrix Tablets Using Polyethylene Oxides as Sustained Release Polymers. J. Pharm. Res. 2013, 6, 249–254. [Google Scholar] [CrossRef]
- Grund, J.; Körber, M.; Bodmeier, R. Predictability of Drug Release from Water-Insoluble Polymeric Matrix Tablets. Eur. J. Pharm. Biopharm. 2013, 85, 650–655. [Google Scholar] [CrossRef]
- Knöös, P.; Schulz, C.; Piculell, L.; Ludwig, R.; Gorton, L.; Wahlgren, M. Quantifying the Release of Lactose from Polymer Matrix Tablets with an Amperometric Biosensor Utilizing Cellobiose Dehydrogenase. Int. J. Pharm. 2014, 468, 121–132. [Google Scholar] [CrossRef]
- Grund, J.; Koerber, M.; Walther, M.; Bodmeier, R. The Effect of Polymer Properties on Direct Compression and Drug Release from Water-Insoluble Controlled Release Matrix Tablets. Int. J. Pharm. 2014, 469, 94–101. [Google Scholar] [CrossRef]
- Agrawal, S.; Fernandes, J.; Shaikh, F.; Patel, V. Quality Aspects in the Development of Pelletized Dosage Forms. Heliyon 2022, 8, e08956. [Google Scholar] [CrossRef]
- Issa, M.G.; de Souza, N.V.; Duque, M.D.; Ferraz, H.G. Physical Characterization of Multiparticulate Systems. Braz. J. Pharm. Sci. 2018, 53, 216. [Google Scholar] [CrossRef][Green Version]
- Alaimo, A.; Zhao, X.; Mi, X.; Atre, P.; Rizvi, S.A.A. Advances in Oral Solid Drug Delivery Systems: Quality by Design Approach in Development of Controlled Release Tablets. BioChem 2025, 5, 9. [Google Scholar] [CrossRef]
- Singh, K.; Nainwal, N.; Chitme, H.R. A Review on Recent Advancements in Pharmaceutical Technology Transfer of Tablets from an Indian Perspective. Ann. Pharm. Françaises 2025, 83, 211–227. [Google Scholar] [CrossRef]
- Prudhvi, C.; Sivaneswari, S.; Preethi, N.; Mounika, B.; Naveen Kumar, B.; Vasudeva Murthy, S.; Karthikeyan, E. Development of Controlled-Release Matrix Tablets of Anti-Diabetic Agent Using Natural and Synthetic Polymers. Intell. Pharm. 2023, 1, 145–151. [Google Scholar] [CrossRef]
- Viridén, A.; Wittgren, B.; Larsson, A. Investigation of Critical Polymer Properties for Polymer Release and Swelling of HPMC Matrix Tablets. Eur. J. Pharm. Sci. 2009, 36, 297–309. [Google Scholar] [CrossRef]
- Bagde, S.; Rohera, B.D. Modification of the Swelling Behavior of a Hydrophilic Polymer as an Approach to Maintaining a Constant Gel Layer Thickness. J. Drug Deliv. Sci. Technol. 2024, 92, 105294. [Google Scholar] [CrossRef]
- Viridén, A.; Larsson, A.; Wittgren, B. The Effect of Substitution Pattern of HPMC on Polymer Release from Matrix Tablets. Int. J. Pharm. 2010, 389, 147–156. [Google Scholar] [CrossRef]
- Viridén, A.; Wittgren, B.; Andersson, T.; Larsson, A. The Effect of Chemical Heterogeneity of HPMC on Polymer Release from Matrix Tablets. Eur. J. Pharm. Sci. 2009, 36, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Caviglioli, G.; Baldassari, S.; Cirrincione, P.; Russo, E.; Parodi, B.; Gatti, P.; Drava, G. An Innovative Matrix Controlling Drug Delivery Produced by Thermal Treatment of DC Tablets Containing Polycarbophil and Ethylcellulose. Int. J. Pharm. 2013, 458, 74–82. [Google Scholar] [CrossRef]
- Patil, H.; Tiwari, R.V.; Upadhye, S.B.; Vladyka, R.S.; Repka, M.A. Formulation and Development of PH-Independent/Dependent Sustained Release Matrix Tablets of Ondansetron HCl by a Continuous Twin-Screw Melt Granulation Process. Int. J. Pharm. 2015, 496, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, E.; De Beer, T.R.M.; Schacht, E.; Van den Mooter, G.; Remon, J.P.; Vervaet, C. Influence of Polyethylene Glycol/Polyethylene Oxide on the Release Characteristics of Sustained-Release Ethylcellulose Mini-Matrices Produced by Hot-Melt Extrusion: In Vitro and in Vivo Evaluations. Eur. J. Pharm. Biopharm. 2009, 72, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Saurí, J.; Zachariah, M.; Macovez, R.; Tamarit, J.L.; Millán, D.; Suñé-Pou, M.; García-Montoya, E.; Pérez-Lozano, P.; Miñarro, M.; Ticó, J.R.; et al. Formulation and Characterization of Mucoadhesive Controlled Release Matrix Tablets of Captopril. J. Drug Deliv. Sci. Technol. 2017, 42, 215–226. [Google Scholar] [CrossRef]
- Abdelbary, G.A.; Tadros, M.I. Design and in Vitro/in Vivo Evaluation of Novel Nicorandil Extended Release Matrix Tablets Based on Hydrophilic Interpolymer Complexes and a Hydrophobic Waxy Polymer. Eur. J. Pharm. Biopharm. 2008, 69, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, E.; Vervaet, C.; Remon, J.P. Xanthan Gum to Tailor Drug Release of Sustained-Release Ethylcellulose Mini-Matrices Prepared via Hot-Melt Extrusion: In Vitro and in Vivo Evaluation. Eur. J. Pharm. Biopharm. 2006, 63, 320–330. [Google Scholar] [CrossRef]
- Muhamad, H.; Bashir, A.B.; Charlton-Harrison, J.; Abdulhussain, R.; Mawla, N.; Patel, K.; Williamson, J.; Blunt, L.; Walton, K.; Conway, B.; et al. Hot-Melt Extruded-FDM 3D-Printed Polyethylene Oxide Tablets: Dissolution Imaging Analysis of Swelling and Drug Release. Eur. J. Pharm. Biopharm. 2025, 208, 114636. [Google Scholar] [CrossRef]
- Muhamad, H.; Ward, A.; Patel, K.; Williamson, J.; Blunt, L.; Conway, B.; Østergaard, J.; Asare-Addo, K. Investigation into the Swelling and Dissolution Behaviour of Polymer-Excipient Blends of PEO Utilising Dissolution Imaging. Int. J. Pharm. 2024, 666, 124850. [Google Scholar] [CrossRef]
- Petrović, J.; Ibrić, S.; Betz, G.; Parojčić, J.; Durić, Z. Application of Dynamic Neural Networks in the Modeling of Drug Release from Polyethylene Oxide Matrix Tablets. Eur. J. Pharm. Sci. 2009, 38, 172–180. [Google Scholar] [CrossRef]
- Muhamad, H.; Mawla, N.; Dereiah, S.; Ward, A.; Williamson, J.; Asare-Addo, K. Comparative Analysis of Drug Release Kinetics in Polyethylene Oxide and Xanthan Gum Matrices with Various Excipients. RSC Pharm. 2024, 2, 303–317. [Google Scholar] [CrossRef]
- Heng, P.W.S.; Chan, L.W.; Easterbrook, M.G.; Li, X. Investigation of the Influence of Mean HPMC Particle Size and Number of Polymer Particles on the Release of Aspirin from Swellable Hydrophilic Matrix Tablets. J. Control. Release 2001, 76, 39–49. [Google Scholar] [CrossRef]
- Sung, K.C.; Nixon, P.R.; Skoug, J.W.; Ju, T.R.; Gao, P.; Topp, E.M.; Patel, M.V. Effect of Formulation Variables on Drug and Polymer Release from HPMC-Based Matrix Tablets. Int. J. Pharm. 1996, 142, 53–60. [Google Scholar] [CrossRef]
- Barra, J.; Falson-Rieg, F.; Doelker, E. Modified Drug Release from Inert Matrix Tablets Prepared from Formulations of Identical Composition but Different Organisations. J. Control. Release 2000, 65, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, O.A. Premium Ethylcellulose Polymer Based Architectures at Work in Drug Delivery. Int. J. Pharm. X 2019, 1, 100023. [Google Scholar] [CrossRef] [PubMed]
- Siepmann, J.; Kranz, H.; Bodmeier, R.; Peppas, N.A. HPMC-Matrices for Controlled Drug Delivery: A New Model Combining Diffusion, Swelling, and Dissolution Mechanisms and Predicting the Release Kinetics. Pharm. Res. 1999, 16, 1748–1756. [Google Scholar] [CrossRef] [PubMed]
- Saša, B.; Odon, P.; Stane, S.; Julijana, K. Analysis of Surface Properties of Cellulose Ethers and Drug Release from Their Matrix Tablets. Eur. J. Pharm. Sci. 2006, 27, 375–383. [Google Scholar] [CrossRef]
- Katakam, P.; Padala, N.R.; Chandu, B.R.; Elfituri, A.; Adiki, S.K.; Kommu, R. Design of Lamivudine XR Matrix Tablets: Influence of HPMC and PEO on in Vitro Drug Release and Bioavailability in Rabbits. J. Pharm. Res. 2013, 6, 845–852. [Google Scholar] [CrossRef]
- Saker, A.; Cares-Pacheco, M.G.; Marchal, P.; Falk, V. Powders Flowability Assessment in Granular Compaction: What about the Consistency of Hausner Ratio? Powder Technol. 2019, 354, 52–63. [Google Scholar] [CrossRef]
- Kaleem, M.A.; Alam, M.Z.; Khan, M.; Jaffery, S.H.I.; Rashid, B. An Experimental Investigation on Accuracy of Hausner Ratio and Carr Index of Powders in Additive Manufacturing Processes. Metal. Powder Report. 2021, 76, S50–S54. [Google Scholar] [CrossRef]
- Kawakita, K.; Lüdde, K.H. Some Considerations on Powder Compression Equations. Powder Technol. 1971, 4, 61–68. [Google Scholar] [CrossRef]
- Ilić, I.; Kása, P.; Dreu, R.; Pintye-Hódi, K.; Srčič, S. The Compressibility and Compactibility of Different Types of Lactose. Drug Dev. Ind. Pharm. 2009, 35, 1271–1280. [Google Scholar] [CrossRef]
- Alderborn, G.; Nyström, C. Pharmaceutical Powder Compaction Technology, 1st ed.; CRC Press: Boca Raton, FL, USA, 1996; ISBN 9780824793760. [Google Scholar]
- Fell, J.T.; Newton, J.M. The Tensile Strength of Lactose Tablets. J. Pharm. Pharmacol. 2011, 20, 657–659. [Google Scholar] [CrossRef]
- Farmacopea de los Estados Unidos de América USP Monographs. Galantamina, Cápsulas de Liberación Prolongada. Available online: https://doi.usp.org/USPNF/USPNF_M5070_07_02.html (accessed on 27 August 2025).
- Yarce, C.J.; Pineda, D.; Correa, C.E.; Salamanca, C.H. Relationship between Surface Properties and In Vitro Drug Release from a Compressed Matrix Containing an Amphiphilic Polymer Material. Pharmaceuticals 2016, 9, 34. [Google Scholar] [CrossRef]
- Švecová, M.; Palounek, D.; Volochanskyi, O.; Prokopec, V. Vibrational Spectroscopic Study of Selected Alkaloids with Therapeutic Effects. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 229, 117969. [Google Scholar] [CrossRef]
- Valor, A.; Reguera, E.; Torres-García, E.; Mendoza, S.; Sanchez-Sinencio, F. Thermal Decomposition of the Calcium Salts of Several Carboxylic Acids. Thermochim. Acta 2002, 389, 133–139. [Google Scholar] [CrossRef]
- Georgieva, D.; Nikolova, D.; Vassileva, E.; Kostova, B. Chitosan-Based Nanoparticles for Targeted Nasal Galantamine Delivery as a Promising Tool in Alzheimer’s Disease Therapy. Pharmaceutics 2023, 15, 829. [Google Scholar] [CrossRef]
- Nagajothi, A.J.; Kannan, R.; Rajashabala, S. Preparation and Characterization of PEO-Based Composite Gel-Polymer Electrolytes Complexed with Lithium Trifluoro Methane Sulfonate. Mater. Sci.-Pol. 2018, 36, 185–192. [Google Scholar] [CrossRef]
- Babanejad, N.; Kandalam, U.; Omidi, Y.; Omidian, H. Functional Properties of Thermally Tampered Poly (Ethylene Oxide). Bioimpacts 2022, 12, 471. [Google Scholar] [CrossRef] [PubMed]
- Zupanc, A.; Petkovšek, M.; Zdovc, B.; Žagar, E.; Zupanc, M. Degradation of Hydroxypropyl Methylcellulose (HPMC) by Acoustic and Hydrodynamic Cavitation. Ultrason. Sonochem. 2024, 109, 107020. [Google Scholar] [CrossRef] [PubMed]
- Jani, R.; Patel, D. Hot Melt Extrusion: An Industrially Feasible Approach for Casting Orodispersible Film. Asian J. Pharm. Sci. 2015, 10, 292–305. [Google Scholar] [CrossRef]
- Gieroba, B.; Kalisz, G.; Krysa, M.; Khalavka, M.; Przekora, A. Application of Vibrational Spectroscopic Techniques in the Study of the Natural Polysaccharides and Their Cross-Linking Process. Int. J. Mol. Sci. 2023, 24, 2630. [Google Scholar] [CrossRef]
- The Dow Chemical Company. Ethylcellulose Polymers Tecnical Handbook. Available online: https://cms.chempoint.com/ic/getattachment/18c67e2f-5ae1-4989-97a4-23faf2cfd6ec/attachment.aspx (accessed on 27 August 2025).
- Dragar, Č.; Roškar, R.; Kocbek, P. The Incorporated Drug Affects the Properties of Hydrophilic Nanofibers. Nanomaterials 2024, 14, 949. [Google Scholar] [CrossRef]
- Wong, R.S.H.; Dodou, K. Effect of Drug Loading Method and Drug Physicochemical Properties on the Material and Drug Release Properties of Poly (Ethylene Oxide) Hydrogels for Transdermal Delivery. Polymers 2017, 9, 286. [Google Scholar] [CrossRef] [PubMed]
- Latreche, M.; Willart, J.F. Analysis of the Dissolution Mechanism of Drugs into Polymers: The Case of the PVP/Sulindac System. Pharmaceutics 2023, 15, 1505. [Google Scholar] [CrossRef] [PubMed]
- Fule, R.; Amin, P. Hot Melt Extruded Amorphous Solid Dispersion of Posaconazole with Improved Bioavailability: Investigating Drug-Polymer Miscibility with Advanced Characterisation. Biomed. Res. Int. 2014, 2014, 146781. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.A.; Lamichhane, S.; Vierhout, T.; Sherman, A.; Engebretson, D.; Pohlson, K.; Remund, T.; Kelly, P. In Vitro Particulate and in Vivo Drug Retention Study of a Novel Polyethylene Oxide Formulation for Drug-Coated Balloons. J. Vasc. Surg. 2018, 67, 1537–1545.e7. [Google Scholar] [CrossRef]
- Crowley, M.M.; Schroeder, B.; Fredersdorf, A.; Obara, S.; Talarico, M.; Kucera, S.; McGinity, J.W. Physicochemical Properties and Mechanism of Drug Release from Ethyl Cellulose Matrix Tablets Prepared by Direct Compression and Hot-Melt Extrusion. Int. J. Pharm. 2004, 269, 509–522. [Google Scholar] [CrossRef]
- Bashir, S.; Zafar, N.; Lebaz, N.; Mahmood, A.; Elaissari, A. Hydroxypropyl Methylcellulose-Based Hydrogel Copolymeric for Controlled Delivery of Galantamine Hydrobromide in Dementia. Processes 2020, 8, 1350. [Google Scholar] [CrossRef]
- Li, J.; Zhao, J.; Tao, L.; Wang, J.; Waknis, V.; Pan, D.; Hubert, M.; Raghavan, K.; Patel, J. The Effect of Polymeric Excipients on the Physical Properties and Performance of Amorphous Dispersions: Part I, Free Volume and Glass Transition. Pharm. Res. 2015, 32, 500–515. [Google Scholar] [CrossRef]
- Ganjoo, R.; Soni, S.; Ram, V.; Verma, A. Medium Molecular Weight Chitosan as a Carrier for Delivery of Lincomycin Hydrochloride from Intra-Pocket Dental Film: Design, Development, in Vitro and Ex Vivo Characterization. J. Appl. Pharm. Sci. 2016, 6, 008–019. [Google Scholar] [CrossRef]
- Yasin, H.; Al-Taani, B.; Salem, M. Preparation and Characterization of Ethylcellulose Microspheres for Sustained-Release of Pregabalin. Res. Pharm. Sci. 2021, 16, 1–15. [Google Scholar] [CrossRef]
- Shah, R.B.; Tawakkul, M.A.; Khan, M.A. Comparative Evaluation of Flow for Pharmaceutical Powders and Granules. AAPS PharmSciTech 2008, 9, 250–258. [Google Scholar] [CrossRef]
- Shojaee, S.; Emami, P.; Mahmood, A.; Rowaiye, Y.; Dukulay, A.; Kaialy, W.; Cumming, I.; Nokhodchi, A. An Investigation on the Effect of Polyethylene Oxide Concentration and Particle Size in Modulating Theophylline Release from Tablet Matrices. AAPS PharmSciTech 2015, 16, 1281. [Google Scholar] [CrossRef]
- Jeong, G.Y.; Bak, J.H.; Yoo, B. Physical and Rheological Properties of Xanthan Gum Agglomerated in Fluidized Bed: Effect of HPMC as a Binder. Int. J. Biol. Macromol. 2019, 121, 424–428. [Google Scholar] [CrossRef]
- Agrawal, A.M.; Manek, R.V.; Kolling, W.M.; Neau, S.H. Studies on the Interaction of Water with Ethylcellulose: Effect of Polymer Particle Size. AAPS PharmSciTech 2003, 4, 469–479. [Google Scholar] [CrossRef]
- Huang, W.; Shi, Y.; Wang, C.; Yu, K.; Sun, F.; Li, Y. Using Spray-Dried Lactose Monohydrate in Wet Granulation Method for a Low-Dose Oral Formulation of a Paliperidone Derivative. Powder Technol. 2013, 246, 379–394. [Google Scholar] [CrossRef]
- Hu, X.; Xu, T.; Bu, J.; Cheng, L.; Wang, T. Impact of Particle Size Distribution and Morphology of Starch on Microstructural–Functional Changes of Dough. LWT 2025, 215, 117251. [Google Scholar] [CrossRef]
- Alyami, H.; Dahmash, E.; Bowen, J.; Mohammed, A.R. An Investigation into the Effects of Excipient Particle Size, Blending Techniques and Processing Parameters on the Homogeneity and Content Uniformity of a Blend Containing Low-Dose Model Drug. PLoS ONE 2017, 12, e0178772. [Google Scholar] [CrossRef] [PubMed]
- Denduyver, P.; Vervaet, C.; Vanhoorne, V. The Effect of Filler Particle Size on API Homogeneity of Controlled Release Formulations via Continuous Twin-Screw Wet Granulation. Int. J. Pharm. 2025, 669, 124990. [Google Scholar] [CrossRef]
- Jakubowska, E.; Ciepluch, N. Blend Segregation in Tablets Manufacturing and Its Effect on Drug Content Uniformity—A Review. Pharmaceutics 2021, 13, 1909. [Google Scholar] [CrossRef]
- United States Pharmacopeia 〈1174〉. Powder Flow; United States Pharmacopeia: Rockville, MD, USA, 2023; Available online: https://doi.usp.org/USPNF/USPNF_M99885_02_01.html (accessed on 27 June 2025).
- Rojas, J.; Uribe, Y.; Zuluaga, A.; Rojas, J. Powder and Compaction Characteristics of Pregelatinized Starches. Pharmazie 2012, 67, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Castañeda Hernández, O.; Domínguez-Robles, J.; Caraballo, I.; Bernad, M.J.; Melgoza Contreras, L.M. Comparison between Polymeric Excipients Using SeDeM Expert System in Combination with Mathematical Modeling and Quality Control Tools. J. Drug Deliv. Sci. Technol. 2023, 86, 104750. [Google Scholar] [CrossRef]
- Kar, A.K.; Mahanti, B.; Kar, B.; Jana, A.; Chakrabarty, S.; Singh, S.; Majumdar, S. Development, Optimization, and in-Vivo Bioavailability Study of Erlotinib Hydrochloride Loaded Microsponge for Colon Targeting. Intell. Pharm. 2025, 3, 1–10. [Google Scholar] [CrossRef]
- Ren, M.; Xu, H.; Zhang, X.; Guan, J.; Mao, S. Evaluation Methods and Strategies to Improve Compression Characteristics of Pharmaceutical Powders. J. Drug Deliv. Sci. Technol. 2024, 100, 106140. [Google Scholar] [CrossRef]
- Rashid, I.; Haddadin, R.R.; Alkafaween, A.A.; Alkaraki, R.N.; Alkasasbeh, R.M. Understanding the Implication of Kawakita Model Parameters Using In-Die Force-Displacement Curve Analysis for Compacted and Non-Compacted API Powders. AAPS Open 2022, 8, 6. [Google Scholar] [CrossRef]
- Mishra, S.M.; Rohera, B.D. Mechanics of tablet formation: A comparative evaluation of percolation theory with classical concepts. Pharm. Dev. Technol. 2019, 24, 954–966. [Google Scholar] [CrossRef] [PubMed]
- Kuentz, M.; Leuenberger, H. A New Theoretical Approach to Tablet Strength of a Binary Mixture Consisting of a Well and a Poorly Compactable Substance. Eur. J. Pharm. Biopharm. 2000, 49, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Mallick, S.; Kumar Pradhan, S.; Chandran, M.; Acharya, M.; Digdarsini, T.; Mohapatra, R. Study of particle rearrangement, compression behavior and dissolution properties after melt dispersion of ibuprofen, Avicel and Aerosil. Results Pharma Sci. 2011, 1, 1–10. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Grumann, H.D.; Kleinebudde, P. Effect of Tableting Temperature on Tablet Properties and Dissolution Behavior of Heat Sensitive Formulations. Int. J. Pharm. 2023, 648, 123603. [Google Scholar] [CrossRef]
- Goldoozian, S.; Mohylyuk, V.; Dashevskiy, A.; Bodmeier, R. Gel Strength of Hydrophilic Matrix Tablets in Terms of In Vitro Robustness. Pharm. Res. 2021, 38, 1297–1306. [Google Scholar] [CrossRef]
- Adetunji, O.A.; Odeniyi, M.A.; Itiola, O.A. Effect of Formulation and Process Variables on the Release, Mechanical and Mucoadhesive Properties of Ibuprofen Tablet Formulations. Acta Pol. Pharm. ñ Drug Res. 2015, 72, 357–365. [Google Scholar]
- Chandran, S.; Asghar, L.; Mantha, N. Design and Evaluation of Ethyl Cellulose Based Matrix Tablets of Ibuprofen with PH Modulated Release Kinetics. Indian. J. Pharm. Sci. 2008, 70, 596. [Google Scholar] [CrossRef]
- Verma, R.K.; Krishna, D.M.; Garg, S. Formulation Aspects in the Development of Osmotically Controlled Oral Drug Delivery Systems. J. Control. Release 2002, 79, 7–27. [Google Scholar] [CrossRef]
- Huang, X.; Brazel, C.S. On the Importance and Mechanisms of Burst Release in Matrix-Controlled Drug Delivery Systems. J. Control. Release 2001, 73, 121–136. [Google Scholar] [CrossRef]
- Zhang, F.; McGinity, J.W. Properties of Hot-Melt Extruded Theophylline Tablets Containing Poly (Vinyl Acetate). Drug Dev. Ind. Pharm. 2000, 26, 931–942. [Google Scholar] [CrossRef]
- Shaikh, N.A.; Abidi, S.E.; Block, L.H. Evaluation of Ethylcellulose as a Matrix for Prolonged Release Formulations. I. Water Soluble Drugs: Acetaminophen and Theophylline. Drug Dev. Ind. Pharm. 1987, 13, 1345–1369. [Google Scholar] [CrossRef]
- Hu, A.; Chen, C.; Mantle, M.D.; Wolf, B.; Gladden, L.F.; Rajabi-Siahboomi, A.; Missaghi, S.; Mason, L.; Melia, C.D. The Properties of HPMC: PEO Extended Release Hydrophilic Matrices and Their Response to Ionic Environments. Pharm. Res. 2017, 34, 941–956. [Google Scholar] [CrossRef] [PubMed]
- Seltzer, B. Galantamine-ER for the Treatment of Mild-to-Moderate Alzheimer’s Disease. Clin. Interv. Aging 2010, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
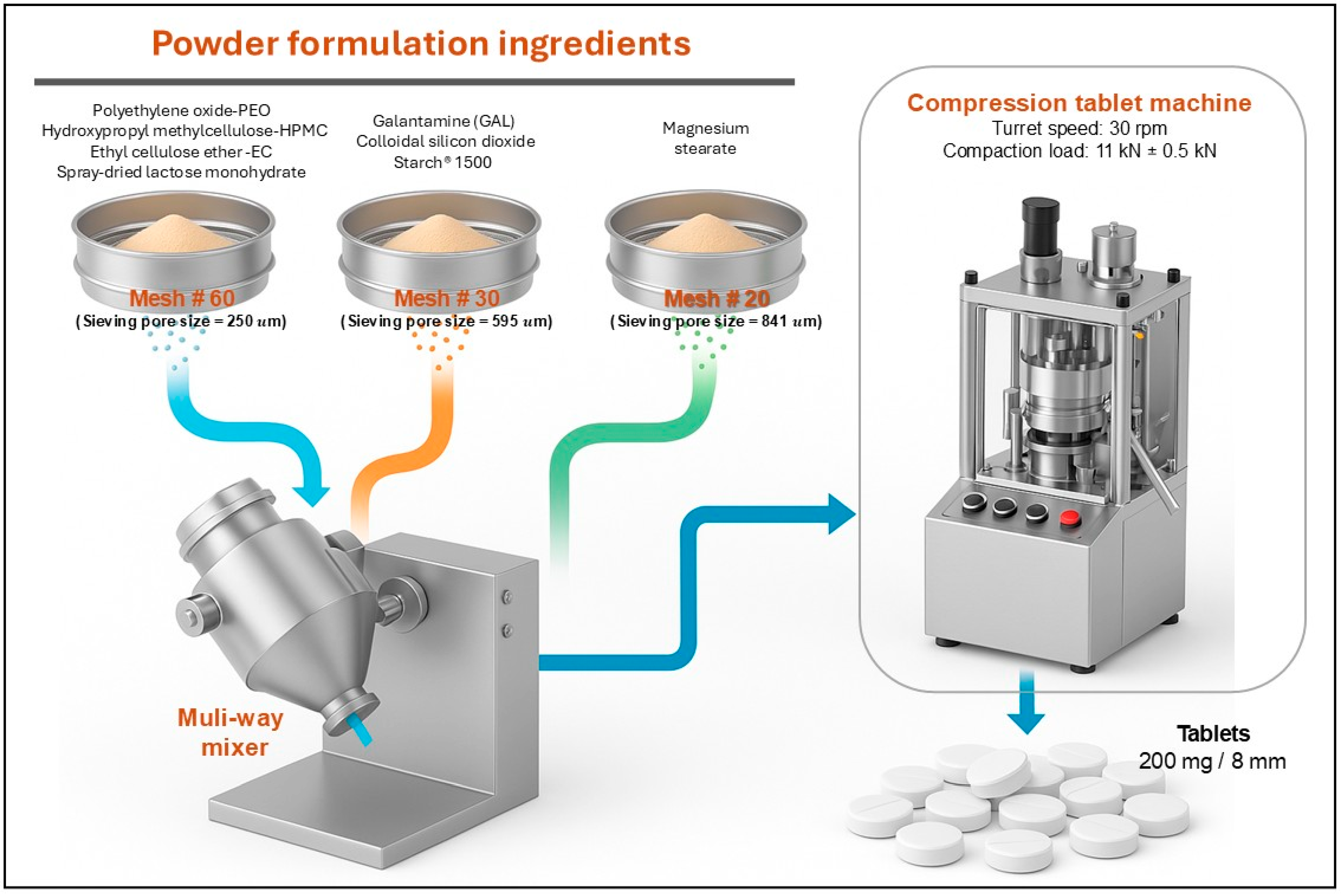


| Formulation Ingredient | Formulation Function | % w/w | ||
|---|---|---|---|---|
| Tablet Formulation 1 | Tablet Formulation 2 | Tablet Formulation 3 | ||
| Galantamine HBr (GAL) | API | 4.44 | 4.44 | 4.44 |
| Polyethylene oxide (PEO) | Controlled-release polymer | 31.52 | -- | -- |
| Hydroxypropyl methylcellulose (HPMC) | Controlled-release polymer | -- | 31.52 | -- |
| Ethyl cellulose (EC) | Controlled-release polymer | -- | -- | 31.52 |
| Starch® 1500 | Diluent 1 | 31.52 | 31.52 | 31.52 |
| Spray-dried lactose monohydrate | Diluent 2 | 31.52 | 31.52 | 31.52 |
| Colloidal silicon dioxide | Lubricant | 0.5 | 0.5 | 0.5 |
| Magnesium stearate | Glidant | 0.5 | 0.5 | 0.5 |
| Formulation Ingredients and Tablet Formulations | Dv10 (µm) | Dv50 (µm) | Dv90 (µm) | Span |
|---|---|---|---|---|
| GAL | 74.4 | 166.9 | 654.0 | 3.5 |
| Polyethylene oxide (PEO) | 375.1 ± 16.2 | 634.7 ± 5.4 | 1028.3 ± 39.3 | 1.0 ± 0.1 |
| Hydroxypropyl methylcellulose (HPMC) | 65.4 ± 4.4 | 263.7 ± 4.2 | 516.5 ± 11.0 | 1.7 ± 0.1 |
| Ethyl cellulose ether (EC) | 1.9 ± 0.0 | 6.2 ± 0.1 | 13.0 ± 0.4 | 1.8 ± 0.0 |
| Starch® 1500 | 14.8 ± 0.2 | 73.1 ± 2.8 | 229.6 ± 7.2 | 2.9 ± 0.0 |
| Spray-dried lactose monohydrate | 40.8 ± 8.0 | 110.9 ± 14.1 | 282.3 ± 73.6 | 2.2 ± 0.3 |
| Formulation 1 | 13.9 ± 0.7 | 81.5 ± 10.6 | 316.4 ± 54.8 | 3.7 ± 0.2 |
| Formulation 2 | 27.1 ± 5.9 | 146.2 ± 29.3 | 358.6 ± 27.8 | 2.3 ± 0.3 |
| Formulation 3 | 8.6 ± 0.3 | 79.7 ± 3.2 | 241.0 ± 8.1 | 2.9 ± 0.0 |
| Formulation Ingredients and Tablet Formulations | Carr Index (%) | Hausner Ratio | Porosity-ε (%) |
|---|---|---|---|
| Polyethylene oxide (PEO) | 12 ± 0.25 | 1.14 ± 0.00 | 62.27 ± 0.70 |
| Hydroxypropyl methylcellulose (HPMC) | 21 ± 0.33 | 1.26 ± 0.01 | 78.41 ± 0.34 |
| Ethyl cellulose ether (EC) | 15 ± 0.24 | 1.18 ± 0.00 | 73.16 ± 0.41 |
| Starch® 1500 | 21 ± 0.66 | 1.26 ± 0.01 | 50.94 ± 1.56 |
| Spray-dried lactose monohydrate | 3 ± 0.10 | 1.03 ± 0.00 | 56.63 ± 1.40 |
| Formulation 1 (GAL + PEO + Excipients) | 46 ± 1.66 | 1.87 ± 0.06 | 70.86 ± 1.04 |
| Formulation 2 (GAL + HPMC + Excipients) | 12 ± 0.69 | 1.13 ± 0.01 | 55.15 ± 2.64 |
| Formulation 3 (GAL + EC + Excipients) | 29 ± 0.64 | 1.41 ± 0.01 | 67.17 ± 0.73 |
| Ingredients and Formulations | Parameters of the Kawakita Linear Function | |||
| R2 | ||||
| (Obtained from Intercept) | (Obtained from Slop) | |||
| Compressibility constant | Pressure term | Kawakita pressure | ||
| PEO | 0.14 ± 0.00 | 0.28 ± 0.15 | 4.21 ± 2.02 | 1.0000 |
| HPMC | 0.23 ± 0.00 | 0.02 ± 0.00 | 43.46 ± 2.80 | 0.9995 |
| EC | 0.18 ± 0.01 | 0.02 ± 0.00 | 56.78 ± 7.10 | 0.9961 |
| Formulation 1 | 0.47 ± 0.00 | 1.20 ± 0.66 | 1.00 ± 0.49 | 0.9999 |
| Formulation 2 | 0.13 ± 0.01 | 0.04 ± 0.02 | 26.86 ± 11.86 | 0.9837 |
| Formulation 3 | 0.39 ± 0.01 | 0.01 ± 0.00 | 138.69 ± 3.11 | 0.9955 |
| Ingredients and formulations | Parameters of the Heckel linear function | |||
| A | K | R2 | ||
| (Intercept) | (Slop) | |||
| Initial packing behavior | Compressibility coefficient | Yield Pressure | ||
| PEO | 1.94788 ± 0.03269 | 0.05500 ± 0.0018 | 18.195 ± 0.486 | 0.99786 |
| HPMC | 1.09569 ± 0.04689 | 0.05589 ± 0.00342 | 17.937 ± 0.898 | 0.99255 |
| EC | 1.69532 ± 0.07564 | 0.05754 ± 0.00434 | 17.446 ± 1.077 | 0.98324 |
| Formulation 1 | 1.3325 ± 0.02682 | 0.03874 ± 0.00167 | 25.845 ± 0.911 | 0.99446 |
| Formulation 2 | 1.15816 ± 0.06378 | 0.06726 ± 0.00473 | 14.917 ± 0.859 | 0.99022 |
| Formulation 3 | 1.21027 ± 0.02513 | 0.07138 ± 0.00318 | 14.028 ± 0.511 | 0.99407 |
| Ingredients and formulations | Leuenberger parameters | |||
| R2 | ||||
| (Intercept) | (Slop) | |||
| Maximum Tensile Strength | Critical Relative Density | |||
| PEO | 6.33247 ± 0.09791 | 0.04141 ± 0.0055 | 1.0000 | |
| HPMC | 7.01417 ± 0.41633 | 0.00622 ± 0.00102 | 1.0000 | |
| EC | 0.94655 ± 0.0111 | 0.04205 ± 0.00527 | 1.0000 | |
| Formulation 1 | 4.00407 ± 0.31389 | 0.00373 ± 6.59635 × 10−4 | 1.0000 | |
| Formulation 2 | 4.60899 ± 0.7821 | 0.00455 ± 0.00199 | 0.9999 | |
| Formulation 3 | 6.2971 ± 0.41643 | 0.00432 ± 7.27968 × 10−4 | 1.0000 | |
| Tablet Formulation | Weight (mg) | Hardness (kN) | Friability (%) | Disintegration Time |
|---|---|---|---|---|
| p-Value | <0.0001 | <0.0001 | 0.072 | - |
| Formulation 1 | 200.62 ± 1.85 | 0.0576 ± 0.0035 | 0.1 ± 0.1 | >4 h |
| Formulation 2 | 201.27 ± 2.72 | 0.1058 ± 0.0028 | 0.4 ± 0.3 | Does not happen |
| Formulation 3 | 176.03 ± 4.50 | 0.1143 ± 0.0043 | 0.1 ± 0.0 | 30 min |
| Tablet Formulation | Time (h) | GAL Amount (%) | Meets the Acceptance Criteria |
|---|---|---|---|
| Control | 1 | 20–40 | |
| Formulation 1 | 27.4 | Yes | |
| Formulation 2 | 27.6 | Yes | |
| Formulation 3 | 76.6 | No | |
| Control | 4 | 40–65 | |
| Formulation 1 | 61.3 | Yes | |
| Formulation 2 | 53.2 | Yes | |
| Formulation 3 | 76.1 | No | |
| Control | 12 | No less than 75 | |
| Formulation 1 | 88.7 | Yes | |
| Formulation 2 | 85.4 | Yes | |
| Formulation 3 | 78.2 | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arana-Linares, A.C.; Caicedo, P.A.; Villegas-Torres, M.F.; González-Barrios, A.F.; Cortes, N.; Osorio, E.H.; Salamanca, C.H.; Barrera-Ocampo, A. Preformulation Study of Controlled-Release Galantamine Matrix Tablets Containing Polyethylene Oxide, Hydroxypropyl Methylcellulose, and Ethylcellulose. Pharmaceutics 2025, 17, 1139. https://doi.org/10.3390/pharmaceutics17091139
Arana-Linares AC, Caicedo PA, Villegas-Torres MF, González-Barrios AF, Cortes N, Osorio EH, Salamanca CH, Barrera-Ocampo A. Preformulation Study of Controlled-Release Galantamine Matrix Tablets Containing Polyethylene Oxide, Hydroxypropyl Methylcellulose, and Ethylcellulose. Pharmaceutics. 2025; 17(9):1139. https://doi.org/10.3390/pharmaceutics17091139
Chicago/Turabian StyleArana-Linares, Andres C., Paola A. Caicedo, María Francisca Villegas-Torres, Andrés F. González-Barrios, Natalie Cortes, Edison H. Osorio, Constain H. Salamanca, and Alvaro Barrera-Ocampo. 2025. "Preformulation Study of Controlled-Release Galantamine Matrix Tablets Containing Polyethylene Oxide, Hydroxypropyl Methylcellulose, and Ethylcellulose" Pharmaceutics 17, no. 9: 1139. https://doi.org/10.3390/pharmaceutics17091139
APA StyleArana-Linares, A. C., Caicedo, P. A., Villegas-Torres, M. F., González-Barrios, A. F., Cortes, N., Osorio, E. H., Salamanca, C. H., & Barrera-Ocampo, A. (2025). Preformulation Study of Controlled-Release Galantamine Matrix Tablets Containing Polyethylene Oxide, Hydroxypropyl Methylcellulose, and Ethylcellulose. Pharmaceutics, 17(9), 1139. https://doi.org/10.3390/pharmaceutics17091139










