Emerging Triglyceride-Rich Lipoprotein Targeted Therapies: An Unmet Need in Cardiometabolic Disease
Abstract
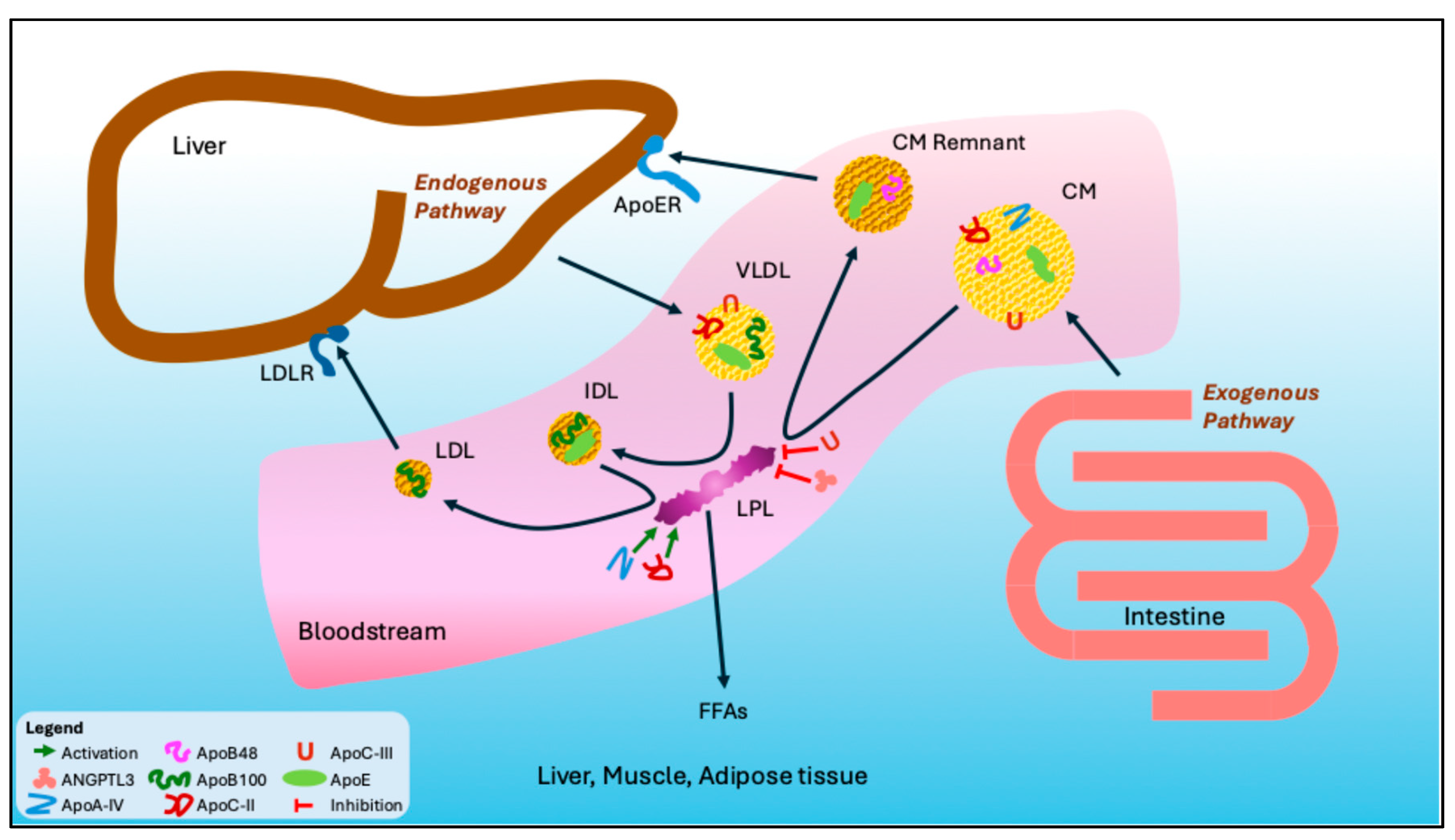
1. Synthesis and Metabolism of Triglycerides
2. Causes and Clinical Relevance of Hypertriglyceridemia
2.1. Causes of Hypertriglyceridemia
2.2. Clinical Relevance of Hypertriglyceridemia
3. Triglycerides and Risk of Atherosclerotic Cardiovascular Disease
3.1. Epidemiological Data
3.2. Genetic Data
3.3. Biological Data
4. Management of Hypertriglyceridemia
4.1. Identifying Secondary Causes
4.2. Therapeutic Goals by Triglyceride Level
4.3. Lifestyle and Dietary Intervention
4.4. Pharmacologic Therapy
4.4.1. Fibrates
4.4.2. Omega-3 Fatty Acids
4.4.3. Statins
4.5. Guideline Recommendations
4.6. Clinical Uncertainty
5. Emerging Therapies
5.1. Apolipoprotein C-III Inhibitors
| Clinical Trial | Population | Design | Active Treatment | Control | Duration (Months) | TG Reduction (%) | RC Reduction (%) | LDL-C Reduction (%) | ApoC-III Reduction (%) |
|---|---|---|---|---|---|---|---|---|---|
| Volanesorsen | |||||||||
| NCT01647308 [70] Phase 2 | N = 15 TG > 200 and <500 mg/dL and type 2 diabetes | Double-blind 2:1 | Volanesorsen 300 mg q1week | Placebo | 12 weeks | −69.1 at 3 months | −75.5 at 3 months | −3.2 at 3 months | −87.5 at 3 months |
| NCT01529424 [71] Phase 2 | N = 85 HTG of 350 to 2000 mg/dL (or 225–2000 mg/dL if on fibrate therapy) | Double-blind, dose ranging (1 of 3 doses or placebo) | Volanesorsen 100 mg q1week 200 mg q1week 300 mg q1week | Placebo | 13 weeks | −31.3 to −70.9 at 3 months | −54.1 to −86.7 at 3 months | +48.0 to +118.3 at 3 months | −40.0 to −79.6 at 3 months |
| COMPASS [72] NCT02300233 Phase 3 | N = 114; MH or FCS and TG ≥ 500 mg/dL | Double-blind 2:1 | Volanesorsen 300 mg q1week | Placebo | 26 weeks | −71.2 at 3 months | −72.4 at 3 months | +95.5 at 3 months | −76.1 at 3 months |
| APPROACH [73] NCT02211209 Phase 3 | N = 66; FCS and TG ≥ 750 mg/dL | Double-blind 1:1 | Volanesorsen 300 mg q1week | Placebo | 52 weeks | −76.5 at 3 months | −71.7 at 3 months | +135.6 at 3 months | −84.2 at 3 months |
| BROADEN [74] NCT02527343 Phase 3 | N = 40; FPLD and TG ≥ 500 mg/dL (≥200 mg/dL if genetic diagnosis/family history), type 2 diabetes and fatty liver | Double-blind 1:1 | Volanesorsen 300 mg q1week | Placebo | 52 weeks | −88.0 at 3 months | - | - | −61.0 at 3 months |
| Olezarsen | |||||||||
| NCT03385239 [78] Phase 2 | N = 114 TG 200–499 mg/dL and established ASCVD or high ASCVD risk | Double-blind dose ranging (1 of 4 doses or placebo) | Olezarsen 10 mg q4week 15 mg q2week 10 mg q1week 50 mg q4week | Placebo | 52 weeks | −23.0 to −60.0 at 6 months | −22.0 −67.1 at 6 months | −6.0 to +16.0 at 6 months | −29.0 to −74.0 at 6 months |
| Bridge-TIMI 73a [79] NCT05355402 Phase 2b | N = 154 TG 150–499 mg/dL and elevated ASCVD risk or severe HTG (TG > 500 mg/dL) | Double-blind 3:1 | Olezarsen 50 mg q4week 80 mg q4week | Placebo | 49 weeks | −60.5 and −67.6 at 6 months | −60.0 and −68.0 at 6 months | +5.9 and +13.0 at 6 months | −65.6 and −76.4 at 6 months |
| BALANCE [80] NCT04568434 Phase 3 | N = 66 FCS and TG ≥ 880 mg/dL | Double-blind 3:1 | Olezarsen 50 mg q4week 80 mg q4week | Placebo | 49 weeks | −22.4 and −43.5 at 6 months | −22.4 and −43.5 at 6 months | +34.7 and 64.9 at 6 months | −57.9 and −66.1 at 6 months |
| Plozasiran | |||||||||
| MUIR [82] NCT04998201 Phase 2b | N = 353 TG 150–499 mg/dL with LDL ≥ 70 mg/dL or non-HDL ≥ 100 mg/dL | Double-blind dose ranging (1 of 4 doses or placebo) | Plozasiran 10 mg q12week; 25 mg q12week; 50 mg q12week; 50 mg q24week | Placebo | 48 weeks | −44.2 to −62.4 at 24 weeks | −44.2 to −62.4 at 24 weeks | −41.7 to −54.2 at 24 weeks | −10.4 to +6.0 at 24 weeks |
| SHASTA-2 [83] NCT04720534 Phase 2b | N= 229; Severe HTG (≥500 mg/dL) | Double blind, dose ranging (1 of 3 doses or placebo) | Plozasiran 10 mg q12week 25 mg q12week 50 mg q12week | Placebo | 48 weeks | −48.8 to −57 at 24 weeks | −58.8 to −63.6 at 24 weeks | +25.8 to +60.3 at 24 weeks | −67.7 to −77.4 at 24 weeks |
| PALISADE [84] NCT05089084 Phase 3 | N = 75 FCS with TG ≥ 880 mg/dL, low LPL activity or a history of acute pancreatitis | Double-blind 2:1 | Plozasiran 25 mg q3month 50 mg q3month | Placebo | 12 months | −80.0 and −78.0 at 10 months | −60.1 and −49.6 at 10 months | +105.9 and +83.0 at 10 months | −93.0 and −96.0 at 10 months |
5.2. Angiopoietin-like 3 Inhibitors
| Clinical Trial | Population | Design | Active Treatment | Control | Duration (Months) | TG Reduction (%) | RC Reduction (%) | LDL-C Reduction (%) | ANGPTL3 Reduction (%) |
|---|---|---|---|---|---|---|---|---|---|
| Vupanorsen | |||||||||
| NCT03360747 [87] Phase 2 | N = 105 TG > 150 mg/dL, type 2 diabetes and hepatic steatosis | Double-blind | Vupanorsen 40 mg q4week 80 mg q4week 20 mg q1week | Placebo | 6 months | −36 to −53 at 6 months | −35 to −47 at 6 months | +6 to −12 at 6 months | −41 to −59 at 6 months |
| TRANSLATE-TIMI 70 [88] NCT04516291 Phase 2b | N = 286 Statin-treated with non–HDL-C ≥ 100 mg/dL and TG 150–500 mg/dL | Double-blind, dose ranging (1 of 7 dose regimens or placebo) | Vupanorsen 80, 120, or 160 mg q4 weeks or 60, 80, 120, or 160 mg q2weeks | Placebo | 24 weeks | −41.3 to −56.8 at 24 weeks | - | −9.1 to −17.3 at 24 weeks | −56.6 to −81.9 at 24 weeks |
| Zodasiran | |||||||||
| ARCHES-2 [89] NCT04832971 Phase 2 | N = 204 Mixed hyperlipidemia with TG 150–499 mg/dL, LDL-C ≥ 70 mg/dL or non-HDL-C ≥ 100 mg/dL | Double-blind, dose ranging (1 of 3 doses or placebo) | Zodasiran 50 mg q12week; 100 mg q12week; 200 mg q12week | Placebo | 6 months | −51.2 to −63.1 at 6 months | −53.0 to −62.0 at 6 months | −12.0 to −18.0 at 6 months | −54.3 to −73.7 at 6 months |
| Solbinsiran | |||||||||
| PROLONG-ANG3 [90] NCT05256654 Phase 2b | N = 205 Mixed dyslipidemia: TG 150–499 mg/dL), LDL > 70 mg/dL, non-HDL > 130 mg/dL; on statin for >2 months | Double-blind, dose ranging (1 of 3 doses or placebo) | Solbinsiran 100 mg D0/D90 400 mg D0/D90 800 mg D0/D90 | Placebo | 270 days | −36.3 to −52.5 at day 180 | - | −1.3 to −16.8 at day 180 | −54.3 to −76.6 at day 180 |
| Evinacumab | |||||||||
| NCT03175367 [92] Phase 2 | N = 272 Hypercholesterolemia with or without ASCVD, refractory to maximum OMT (statin/PCSK9i) | Double-blind, dose ranging (SC or IV) | Evinacumab (SC) 300 mg q2week 300 mg q1week 450 mg q1week (IV) 15mg/Kg q4weeks 5mg/Kg q4weeks | Placebo | 16 weeks | −38 to −53.4 at 16 weeks | - | −29.7 to −49.9 at 16 weeks | - |
| ELIPSE HoFH [93] NCT03399786 Phase 3 | N = 65 HoFH under LLT (LDL > 70 mg/dL) | Double-blind, 2:1 | Evinacumab (IV) 15mg/Kg q4weeks | Placebo | 24 weeks | −55.0 at 24 weeks | −79.6 at 24 weeks | −47.1 at 24 weeks | - |
| NCT03452228 [94] Phase 2 | N = 51 Severe HTG (≥500 mg/dL); FCS or MH with or without LPL mutations | Double-blind, 2:1 | Evinacumab (IV) 15mg/Kg q4weeks | Placebo | 12 weeks | FCS: −27.7; MH: −64.8 to −81.7 at 12 weeks | FCS: −37.5; MH: −62.8 to −79 at 12 weeks | FCS: +25; MH: +26.5 to +32 at 12 weeks | - |
5.3. Other Therapeutic Strategies
6. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Alves-Bezerra, M.; Cohen, D.E. Triglyceride metabolism in the liver. Compr. Physiol. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- Laufs, U.; Parhofer, K.G.; Ginsberg, H.N.; Hegele, R.A. Clinical review on triglycerides. Eur. Heart J. 2020, 41, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Boren, J.; Taskinen, M.R. Metabolism of Triglyceride-Rich Lipoproteins. Prevention and Treatment of Atherosclerosis. In Handbook of Experimental Pharmacology; Von Eckardstein, A., Binder, C.J., Eds.; Springer: Cham, Switzerland, 2021; Volume 270. [Google Scholar]
- Visser, A.; Hussain, M.M.; Kuivenhoven, J.A. The intracellular chylomicron highway: Novel insights into chylomicron biosynthesis, trafficking, and secretion. Curr. Opin. Lipidol. 2025, 36, 145–152. [Google Scholar] [CrossRef]
- Borén, J.; Taskinen, M.R.; Packard, C.J. Biosynthesis and metabolism of ApoB-containing lipoproteins. Annu. Rev. Nutr. 2024, 44, 179–204. [Google Scholar] [CrossRef] [PubMed]
- Dominiczak, M.H.; Caslake, M.J. Apolipoproteins: Metabolic role and clinical biochemistry applications. Ann. Clin. Biochem. 2011, 48, 498–515. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Zheng, Z. Walking the VLDL tightrope in cardiometabolic diseases. Trends Endocrinol. Metab. 2025, 36, 278–291. [Google Scholar] [CrossRef]
- Sandesara, P.B.; Virani, S.S.; Fazio, S.; Shapiro, M.D. The forgotten lipids: Triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk. Endocr. Rev. 2019, 40, 537–557. [Google Scholar] [CrossRef]
- Wen, Y.; Chen, Y.Q.; Konrad, R.J. The Regulation of triacylglycerol metabolism and lipoprotein lipase activity. Adv. Biol. 2022, 6, e2200093. [Google Scholar] [CrossRef]
- Boren, J.; Chapman, M.J.; Krauss, R.M.; Packard, C.J.; Bentzon, J.F.; Binder, C.J.; Daemen, M.J.; Demer, L.L.; Hegele, R.A.; Nicholls, S.J.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: Pathophysiological, genetic, and therapeutic insights: A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2020, 41, 2313–2330. [Google Scholar] [CrossRef]
- Xue, H.; Zhang, M.; Liu, J.; Wang, J.; Ren, G. Structure-based mechanism and inhibition of cholesteryl ester transfer protein. Curr. Atheroscler. Rep. 2023, 25, 155–166. [Google Scholar] [CrossRef]
- Brown, E.M.; Clardy, J.; Xavier, R.J. Gut microbiome lipid metabolism and its impact on host physiology. Cell Host Microbe 2023, 31, 173–186. [Google Scholar] [CrossRef]
- Miller, M.; Stone, N.J.; Ballantyne, C.; Bittner, V.; Criqui, M.H.; Ginsberg, H.N.; Goldberg, A.C.; Howard, W.J.; Jacobson, M.S.; Kris-Etherton, P.M.; et al. Triglycerides and cardiovascular disease: A scientific statement from the American Heart Association. Circulation 2011, 123, 2292–2333. [Google Scholar] [CrossRef]
- Berglund, L.; Brunzell, J.D.; Goldberg, A.C.; Goldberg, I.J.; Sacks, F.; Murad, M.H.; Stalenhoef, A.F.H. Evaluation and Treatment of Hypertriglyceridemia: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2012, 97, 2969–2989. [Google Scholar] [CrossRef] [PubMed]
- Christian, J.B.; Bourgeois, N.; Snipes, R.; Lowe, K.A. Prevalence of severe (500 to 2000 mg/dL) hypertriglyceridemia in United States adults. Am. J. Cardiol. 2011, 107, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Philip, S.; Granowitz, C.; Toth, P.P.; Wong, N.D. Hypertriglyceridemia in statin-treated US adults: The National Health and Nutrition Examination Survey. J. Clin. Lipidol. 2019, 13, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Hegele, R.A.; Ginsberg, H.N.; Chapman, M.J.; Nordestgaard, B.G.; Kuivenhoven, J.A.; Averna, M.; Borén, J.; Bruckert, E.; Catapano, A.L.; Descamps, O.S.; et al. The polygenic nature of hypertriglyceridaemia: Implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol. 2014, 2, 655–666. [Google Scholar] [CrossRef]
- Hegele, R.A.; Borén, J.; Ginsberg, H.N.; Arca, M.; Averna, M.; Binder, C.J.; Calabresi, L.; Chapman, M.J.; Cuchel, M.; von Eckardstein, A.; et al. Rare dyslipidaemias, from phenotype to genotype to management: A European Atherosclerosis Society task force consensus statement. Lancet Diabetes Endocrinol. 2020, 8, 50–67. [Google Scholar] [CrossRef]
- Pedersen, S.B.; Langsted, A.; Nordestgaard, B.G. Nonfasting Mild-to-Moderate Hypertriglyceridemia and Risk of Acute Pancreatitis. JAMA Intern. Med. 2016, 176, 1834–1842. [Google Scholar] [CrossRef]
- Yang, A.L.; McNabb-Baltar, J. Hypertriglyceridemia and acute pancreatitis. Pancreatology 2020, 20, 795–800. [Google Scholar] [CrossRef]
- Rinella, M.E.; Lazarus, J.V.; Ratziu, V.; Francque, S.M.; Sanyal, A.J.; Kanwal, F.; Romero, D.; Abdelmalek, M.F.; Anstee, Q.M.; NAFLD Nomenclature consensus group; et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 2023, 78, 1966–1986. [Google Scholar] [CrossRef]
- White, D.L.; Kanwal, F.; El-Serag, H.B. Association between nonalcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin. Gastroenterol. Hepatol. 2012, 10, 1342–1359. [Google Scholar] [CrossRef]
- Martínez, D.P.; Díaz, J.O.; Bobes, C.M. Eruptive xanthomas and acute pancreatitis in a patient with hypertriglyceridemia. Int. Arch. Med. 2008, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Wierzbicki, A.S. Images in clinical medicine. Lipemia retinalis. N. Engl. J. Med. 2005, 353, 823. [Google Scholar] [CrossRef] [PubMed]
- Braunwald, E. Triglycerides: The past, the present, and the future. Eur. Heart J. 2024, 45, 3780–3781. [Google Scholar] [CrossRef]
- Nordestgaard, B.G. Triglyceride-rich lipoproteins and atherosclerotic cardiovascular disease: New insights from epidemiology, genetics, and biology. Circ. Res. 2016, 118, 547–563. [Google Scholar] [CrossRef]
- Austin, M.A.; Hokanson, J.E.; Edwards, K.L. Hypertriglyceridemia as a cardiovascular risk factor. Am. J. Cardiol. 1998, 81, 7B–12B. [Google Scholar] [CrossRef]
- Sarwar, N.; Danesh, J.; Eiriksdottir, G.; Sigurdsson, G.; Wareham, N.; Bingham, S.; Boekholdt, M.; Khaw, K.-T.; Gudnason, V. Triglycerides and the risk of coronary heart disease: 10,158 incident cases among 262,525 participants in 29 Western prospective studies. Circulation 2007, 115, 450–458. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Benn, M.; Schnohr, P.; Tybjaerg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 2007, 298, 299–308. [Google Scholar] [CrossRef]
- Varbo, A.; Nordestgaard, B.G. Remnant cholesterol and risk of ischemic stroke in 112,512 individuals from the general population. Ann. Neurol. 2019, 85, 550–559. [Google Scholar] [CrossRef]
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA 2007, 298, 309–316. [Google Scholar] [CrossRef]
- Di, A.E.; Sarwar, N.; Perry, P.; Kaptoge, S.; Ray, K.K.; Thompson, A.; Wood, A.M.; Lewington, S.; Sattar, N.; Packard, C.J.; et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 2009, 302, 1993–2000. [Google Scholar] [CrossRef] [PubMed]
- Varbo, A.; Benn, M.; Tybjaerg-Hansen, A.; Jorgensen, A.B.; Frikke- Schmidt, R.; Nordestgaard, B.G. Remnant cholesterol as a causal risk factor for ischemic heart disease. J. Am. Coll. Cardiol. 2013, 61, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Dron, J.S.; Hegele, R.A. Genetics of triglycerides and the risk of atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 31. [Google Scholar] [CrossRef]
- Uffelmann, E.; Huang, Q.Q.; Munung, N.S.; De Vries, J.; Okada, Y.; Martin, A.R.; Martin, H.C.; Lappalainen, T.; Posthuma, D. Genome-wide association studies. Nat. Rev. Methods Primers 2021, 1, 59. [Google Scholar] [CrossRef]
- Do, R.; Willer, C.J.; Schmidt, E.M.; Sengupta, S.; Gao, C.; Peloso, G.M.; Gustafsson, S.; Kanoni, S.; Ganna, A.; Chen, J.; et al. Common variants associated with plasma triglycerides and risk for coronary artery disease. Nat. Genet. 2013, 45, 1345–1352. [Google Scholar] [CrossRef]
- Holmes, M.V.; Asselbergs, F.W.; Palmer, T.M.; Drenos, F.; Lanktree, M.B.; Nelson, C.P.; Dale, C.E.; Padmanabhan, S.; Finan, C.; Swerdlow, D.I.; et al. Mendelian randomization of blood lipids for coronary heart disease. Eur. Heart J. 2015, 36, 539–550. [Google Scholar] [CrossRef]
- Khera, A.V.; Won, H.-H.; Peloso, G.M.; O’dushlaine, C.; Liu, D.; Stitziel, N.O.; Natarajan, P.; Nomura, A.; Emdin, C.A.; Gupta, N.; et al. Association of rare and common variation in the lipoprotein lipase gene with coronary artery disease. JAMA 2017, 317, 937–946. [Google Scholar] [CrossRef]
- Pollin, T.I.; Damcott, C.M.; Shen, H.; Ott, S.H.; Shelton, J.; Horenstein, R.B.; Post, W.; McLenithan, J.C.; Bielak, L.F.; Peyser, P.A.; et al. A null mutation in human APOC3 confers a favorable plasma lipid profile and apparent cardioprotection. Science 2008, 322, 1702–1705. [Google Scholar] [CrossRef]
- Jørgensen, A.B.; Frikke-Schmidt, R.; Nordestgaard, B.G.; Tybjærg-Hansen, A. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N. Engl. J. Med. 2014, 371, 32–41. [Google Scholar] [CrossRef]
- Crosby, J.; Peloso, G.M.; Auer, P.L.; Crosslin, D.R.; Stitziel, N.O.; Lange, L.A.; Lu, Y.; Tang, Z.; Zhang, H.; Hindy, G.; et al. Loss-of-function mutations in APOC3, triglycerides, and coronary disease. N. Engl. J. Med. 2014, 371, 22–31. [Google Scholar]
- Saleheen, D.; Natarajan, P.; Armean, I.M.; Zhao, W.; Rasheed, A.; Khetarpal, S.A.; Won, H.-H.; Karczewski, K.J.; O’Donnell-Luria, A.H.; Samochaet, K.E.; et al. Human knockouts and phenotypic analysis in a cohort with a high rate of consanguinity. Nature 2017, 544, 235–239. [Google Scholar] [CrossRef]
- Musunuru, K.; Pirruccello, J.P.; Do, R.; Peloso, G.M.; Guiducci, C.; Sougnez, C.; Garimella, K.V.; Fisher, S.; Abreu, J.; Barry, A.J.; et al. Exome sequencing, ANGPTL3 mutations, and familial combined hypolipidemia. N. Engl. J. Med. 2010, 363, 2220–2227. [Google Scholar] [CrossRef]
- Dewey, F.E.; Gusarova, V.; Dunbar, R.L.; O’dUshlaine, C.; Schurmann, C.; Gottesman, O.; McCarthy, S.; Van Hout, C.V.; Bruse, S.; Dansky, H.M.; et al. Genetic and pharmacologic inactivation of ANGPTL3 and cardiovascular disease. N. Engl. J. Med. 2017, 377, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Stitziel, N.; Khera, A.; Wang, X.; Bierhals, A.; Vourakis, A.; Sperry, A.; Natarajan, P.; Klarin, D.; Emdin, C.; Zekavat, S.; et al. ANGPTL3 deficiency and protection against coronary artery disease. J. Am. Coll. Cardiol. 2017, 69, 2054–2063. [Google Scholar] [CrossRef] [PubMed]
- Dewey, F.E.; Gusarova, V.; O’Dushlaine, C.; Gottesman, O.; Trejos, J.; Hunt, C.; Van Hout, C.V.; Habegger, L.; Buckler, D.; Lai, K.-M.V.; et al. Inactivating variants in ANGPTL4 and risk of coronary artery disease. N. Engl. J. Med. 2016, 374, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Barros, D.R.; Hegele, R.A. Fibroblast growth factor 21, update on genetics and molecular biology. Curr. Opin. Lipidol. 2025, 36, 88–95. [Google Scholar] [CrossRef]
- Larsson, S.C.; Gill, D. Genetic evidence supporting fibroblast growth factor 21 signalling as a pharmacological target for cardiometabolic outcomes and Alzheimer’s Disease. Nutrients 2021, 13, 1504. [Google Scholar] [CrossRef]
- Larsson, S.C.; Michaëlsson, K.; Mola-Caminal, M.; Höijer, J.; Mantzoros, C.S. Genome-wide association and Mendelian randomization study of fibroblast growth factor 21 reveals causal associations with hyperlipidemia and possibly NASH. Metabolism 2022, 137, 155329. [Google Scholar] [CrossRef]
- Ginsberg, H.N.; Packard, C.J.; Chapman, M.J.; Borén, J.; Aguilar-Salinas, C.A.; Averna, M.; Ference, B.A.; Gaudet, D.; Hegele, R.A.; Kersten, S.; et al. Triglyceride-rich lipoproteins and their remnants: Metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies—A consensus statement from the European Atherosclerosis Society. Eur. Heart J. 2021, 42, 4791–4806. [Google Scholar] [CrossRef]
- Moreno-Vedia, J.; Llop, D.; Rodríguez-Calvo, R.; Plana, N.; Amigó, N.; Rosales, R.; Esteban, Y.; Masana, L.; Ibarretxe, D.; Girona, J. Lipidomics of triglyceride-rich lipoproteins derived from hyperlipidemic patients on inflammation. Eur. J. Clin. Investig. 2024, 54, e14132. [Google Scholar] [CrossRef]
- Netea, M.G.; Balkwill, F.; Chonchol, M.; Cominelli, F.; Donath, M.Y.; Giamarellos-Bourboulis, E.J.; Golenbock, D.; Gresnigt, M.S.; Heneka, M.T.; Hoffman, H.M.; et al. A guiding map for inflammation. Nat. Immunol. 2017, 18, 826–831. [Google Scholar] [CrossRef]
- Stark, K.; Massberg, S. Interplay between inflammation and thrombosis in cardiovascular pathology. Nat. Rev. Cardiol. 2021, 18, 666–682. [Google Scholar] [CrossRef]
- Gubensek, J. The role of apheresis and insulin therapy in hypertriglyceridemic acute pancreatitis—A concise review. BMC Gastroenterol. 2023, 23, 341. [Google Scholar] [CrossRef] [PubMed]
- Akivis, Y.; Alkaissi, H.; McFarlane, S.I.; Bukharovich, I. The Role of Triglycerides in Atherosclerosis: Recent Pathophysiologic Insights and Therapeutic Implications. Curr. Cardiol. Rev. 2024, 20, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, C.F.; Sikand, G.; Petersen, K.S.; Anderson, C.A.M.; Aspry, K.E.; Bolick, J.P.; Kris-Etherton, P.M.; Maki, K.C. Nutrition interventions for adults with dyslipidemia: A Clinical Perspective from the National Lipid Association. J. Clin. Lipidol. 2023, 17, 428–451. [Google Scholar] [CrossRef] [PubMed]
- Keech, A.; Simes, R.J.; Barter, P.; Best, J.; Scott, R.; Taskinen, M.R.; Forder, P.; Pillai, A.; Davis, T.; Glasziou, P.; et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (FIELD): A randomised controlled trial. Lancet 2005, 366, 1849–1861. [Google Scholar] [CrossRef]
- ACCORD Study Group; Ginsberg, H.N.; Elam, M.B.; Lovato, L.C.; Crouse, J.R., 3rd; Leiter, L.A.; Linz, P.; Friedewald, W.T.; Buse, J.B.; Gerstein, H.C.; et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N. Engl. J. Med. 2010, 362, 1563–1574. [Google Scholar]
- Das Pradhan, A.; Glynn, R.J.; Fruchart, J.C.; MacFadyen, J.G.; Zaharris, E.S.; Everett, B.M.; Campbell, S.E.; Oshima, R.; Amarenco, P.; Blom, D.J.; et al. Triglyceride lowering with pemafibrate to reduce cardiovascular risk. N. Engl. J. Med. 2022, 387, 1923–1934. [Google Scholar] [CrossRef]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- ASCEND Study Collaborative Group; Bowman, L.; Mafham, M.; Wallendszus, K.; Stevens, W.; Buck, G.; Barton, J.; Murphy, K.; Aung, T.; Haynes, R.; et al. Effects of n–3 fatty acid supplements in diabetes mellitus. N. Engl. J. Med. 2018, 379, 1540–1550. [Google Scholar]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, S.J.; Lincoff, A.M.; Garcia, M.; Bash, D.; Ballantyne, C.M.; Barter, P.J.; Davidson, M.H.; Kastelein, J.J.P.; Koenig, W.; McGuire, D.K.; et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: The STRENGTH trial. JAMA 2020, 324, 2268–2280. [Google Scholar] [CrossRef] [PubMed]
- Cholesterol Treatment Trialists Collaboration; Fulcher, J.; O’Connell, R.; Voysey, M.; Emberson, J.; Blackwell, L.; Mihaylova, B.; Simes, J.; Collins, R.; Kirby, A.; et al. Efficacy and safety of LDL-lowering therapy among men and women: Meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet 2015, 385, 1397–1405. [Google Scholar] [PubMed]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2019, 73, 3168–3209. [Google Scholar] [CrossRef]
- Writing Committee; Lloyd-Jones, D.M.; Morris, P.B.; Ballantyne, C.M.; Birtcher, K.K.; Covington, A.M.; DePalma, S.M.; Minissian, M.B.; Orringer, C.E.; Smith, S.C., Jr.; et al. 2022 ACC Expert Consensus Decision Pathway on the Role of Nonstatin Therapies for LDL-Cholesterol Lowering in the Management of Atherosclerotic Cardiovascular Disease Risk: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2022, 80, 1366–1418. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; A Ference, B.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Giammanco, A.; Spina, R.; Cefalù, A.B.; Averna, M. APOC-III: A Gatekeeper in Controlling Triglyceride Metabolism. Curr. Atheroscler. Rep. 2023, 25, 67–76. [Google Scholar] [CrossRef]
- Gaudet, D.; Brisson, D.; Tremblay, K.; Alexander, V.J.; Singleton, W.; Hughes, S.G.; Geary, R.S.; Baker, B.F.; Graham, M.J.; Crooke, R.M.; et al. Targeting APOC3 in the familial chylomicronemia syndrome. N. Engl. J. Med. 2014, 371, 2200–2206. [Google Scholar] [CrossRef]
- Gouni-Berthold, I.; Alexander, V.J.; Yang, Q.; Hurh, E.; Steinhagen-Thiessen, E.; Moriarty, P.M.; Hughes, S.G.; Gaudet, D.; A Hegele, R.; O’DEa, L.S.L.; et al. Efficacy and safety of volanesorsen in patients with multifactorial chylomicronaemia (COMPASS): A multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2021, 9, 264–275. [Google Scholar] [CrossRef]
- Digenio, A.; Dunbar, R.L.; Alexander, V.J.; Hompesch, M.; Morrow, L.; Lee, R.G.; Graham, M.J.; Hughes, S.G.; Yu, R.; Singleton, W.; et al. Antisense-mediated lowering of plasma apolipoprotein C-III by volanesorsen improves dyslipidemia and insulin sensitivity in type 2 diabetes. Diabetes Care 2016, 39, 1408–1415. [Google Scholar] [CrossRef]
- Gaudet, D.; Alexander, V.J.; Baker, B.F.; Brisson, D.; Tremblay, K.; Singleton, W.; Geary, R.S.; Hughes, S.G.; Viney, N.J.; Graham, M.J.; et al. Antisense inhibition of apolipoprotein C-III in patients with hypertriglyceridemia. N. Engl. J. Med. 2015, 373, 438–447. [Google Scholar] [CrossRef]
- Witztum, J.L.; Gaudet, D.; Freedman, S.D.; Alexander, V.J.; Digenio, A.; Williams, K.R.; Yang, Q.; Hughes, S.G.; Geary, R.S.; Arca, M.; et al. Volanesorsen and triglyceride levels in Familial Chylomicronemia Syndrome. N. Engl. J. Med. 2019, 381, 531–542. [Google Scholar] [CrossRef]
- Oral, E.A.; Garg, A.; Tami, J.; Huang, E.A.; O’Dea, L.S.L.; Schmidt, H.; Tiulpakov, A.; Mertens, A.; Alexander, V.J.; Watts, L.; et al. Assessment of efficacy and safety of volanesorsen for treatment of metabolic complications in patients with familial partial lipodystrophy: Results of the BROADEN study: Volanesorsen in FPLD.; The BROADEN Study. J. Clin. Lipidol. 2022, 16, 833–849. [Google Scholar] [CrossRef] [PubMed]
- Alexander, V.J.; Karwatowska-Prokopczuk, E.; Prohaska, T.A.; Li, L.; Geary, R.S.; Gouni-Berthold, I.; Oral, E.A.; Hegele, R.A.; Stroes, E.S.; Witztum, J.L.; et al. Volanesorsen to prevent acute pancreatitis in hypertriglyceridemia. N. Engl. J. Med. 2024, 390, 476–477. [Google Scholar] [CrossRef] [PubMed]
- Brinton, E.A.; Eckel, R.H.; Gaudet, D.; Ballantyne, C.M.; Baker, B.F.; Ginsberg, H.N.; Witztum, J.L. Familial chylomicronemia syndrome and treatments to target hepatic APOC3 mRNA. Atherosclerosis 2025, 403, 119114. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, R.Z.; Henry, S.; Geary, R.S. Pharmacokinetics and clinical pharmacology considerations of GalNAc3-conjugated antisense oligonucleotides. Expert Opin. Drug Metab. Toxicol. 2019, 15, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.-C.; Karwatowska-Prokopczuk, E.; Amour, E.S.; Ballantyne, C.M.; Shapiro, M.D.; Moriarty, P.M.; Baum, S.J.; Hurh, E.; Bartlett, V.J.; Kingsbury, J.; et al. Apolipoprotein C-III reduction in subjects with moderate hypertriglyceridaemia and at high cardiovascular risk. Eur. Heart J. 2022, 43, 1401–1412. [Google Scholar] [CrossRef]
- Bergmark, B.A.; Marston, N.A.; Prohaska, T.A.; Alexander, V.J.; Zimerman, A.; Moura, F.A.; Murphy, S.A.; Goodrich, E.L.; Zhang, S.; Gaudet, D.; et al. Olezarsen for hypertriglyceridemia in patients at high cardiovascular risk. N. Engl. J. Med. 2024, 390, 1770–1780. [Google Scholar] [CrossRef]
- Stroes, E.S.G.; Alexander, V.J.; Karwatowska-Prokopczuk, E.; Hegele, R.A.; Arca, M.; Ballantyne, C.M.; Soran, H.; Prohaska, T.A.; Xia, S.; Ginsberg, H.N.; et al. Olezarsen, acute pancreatitis, and familial chylomicronemia syndrome. N. Engl. J. Med. 2024, 390, 1781–1792. [Google Scholar] [CrossRef]
- Bergmark, B.A.; Marston, N.A.; Prohaska, T.A.; Alexander, V.J.; Zimerman, A.; Moura, F.A.; Kang, Y.M.; Murphy, S.A.; Zhang, S.; Lu, M.T.; et al. Olezarsen in patients with hypertriglyceridemia at high cardiovascular risk: Rationale and design of the Essence-TIMI 73b trial. Am. Heart J. 2025, 286, 116–124. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Vasas, S.; Azizad, M.; Clifton, P.; Rosenson, R.S.; Chang, T.; Melquist, S.; Zhou, R.; Mushin, M.; Leeper, N.J.; et al. Plozasiran, an RNA interference agent targeting APOC3, for mixed hyperlipidemia. N. Engl. J. Med. 2024, 391, 899–912. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, D.; Pall, D.; Watts, G.F.; Nicholls, S.J.; Rosenson, R.S.; Modesto, K.; Martin, J.S.; Hellawell, J.; Ballantyne, C.M. Plozasiran (ARO-APOC3) for Severe Hypertriglyceridemia: The SHASTA-2 Randomized Clinical Trial. JAMA Cardiol. 2024, 9, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Watts, G.F.; Rosenson, R.S.; Hegele, R.A.; Goldberg, I.J.; Gallo, A.; Mertens, A.; Baass, A.; Zhou, R.; Muhsin, M.; Hellawell, J.; et al. Plozasiran for Managing Persistent Chylomicronemia and Pancreatitis Risk. N. Engl. J. Med. 2025, 392, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Dijk, W.; Kersten, S. Regulation of lipid metabolism by angiopoietin-like proteins. Curr. Opin. Lipidol. 2016, 27, 249–256. [Google Scholar] [CrossRef]
- Kraaijenhof, J.M.; Tromp, T.R.; Nurmohamed, N.S.; Reeskamp, L.F.; Langenkamp, M.; Levels, J.H.M.; Boekholdt, S.M.; Wareham, N.J.; Hoekstra, M.; Stroes, E.S.G.; et al. ANGPTL3 (Angiopoietin-Like 3) preferentially resides on high-density lipoprotein in the human circulation, affecting its activity. J. Am. Heart Assoc. 2023, 12, e030476. [Google Scholar] [CrossRef]
- Gaudet, D.; Karwatowska-Prokopczuk, E.; Baum, S.J.; Hurh, E.; Kingsbury, J.; Bartlett, V.J.; Figueroa, A.L.; Piscitelli, P.; Singleton, W.; Witztum, J.L.; et al. Vupanorsen, an N-acetyl galactosamine-conjugated antisense drug to ANGPTL3 mRNA, lowers triglycerides and atherogenic lipoproteins in patients with diabetes, hepatic steatosis, and hypertriglyceridaemia. Eur. Heart J. 2020, 41, 3936–3945. [Google Scholar] [CrossRef]
- Bergmark, B.A.; Marston, N.A.; Bramson, C.R.; Curto, M.; Ramos, V.; Jevne, A.; Kuder, J.F.; Park, J.-G.; Murphy, S.A.; Verma, S.; et al. Effect of Vupanorsen on Non-High-Density Lipoprotein Cholesterol Levels in Statin-Treated Patients with Elevated Cholesterol: TRANSLATE-TIMI 70. Circulation 2022, 145, 1377–1386. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Gaudet, D.; Hegele, R.A.; Ballantyne, C.M.; Nicholls, S.J.; Lucas, K.J.; Martin, J.S.; Zhou, R.; Muhsin, M.; Chang, T.; et al. Zodasiran, an RNAi Therapeutic Targeting ANGPTL3, for Mixed Hyperlipidemia. N. Engl. J. Med. 2024, 391, 913–925. [Google Scholar] [CrossRef]
- Ray, K.K.; Oru, E.; Rosenson, R.S.; Jones, J.; Ma, X.; Walgren, J.; Haupt, A.; Verma, S.; Gaudet, D.; Nicholls, S.J.; et al. Durability and efficacy of solbinsiran, a GalNAc-conjugated siRNA targeting ANGPTL3, in adults with mixed dyslipidaemia (PROLONG-ANG3): A double-blind, randomised, placebo-controlled, phase 2 trial. Lancet 2025, 405, 1594–1607. [Google Scholar] [CrossRef]
- Gusarova, V.; Alexa, C.A.; Wang, Y.; Rafique, A.; Kim, J.H.; Buckler, D.; Mintah, I.J.; Shihanian, L.M.; Cohen, J.C.; Hobbs, H.H.; et al. ANGPTL3 blockade with a human monoclonal antibody reduces plasma lipids in dyslipidemic mice and monkeys. J. Lipid Res. 2015, 56, 1308–1317. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Burgess, L.J.; Ebenbichler, C.F.; Baum, S.J.; Stroes, E.S.; Ali, S.; Khilla, N.; Hamlin, R.; Pordy, R.; Dong, Y.; et al. Evinacumab in patients with refractory hypercholesterolemia. N. Engl. J. Med. 2020, 383, 2307–2319. [Google Scholar] [CrossRef]
- Raal, F.J.; Rosenson, R.S.; Reeskamp, L.F.; Hovingh, G.K.; Kastelein, J.J.; Rubba, P.; Ali, S.; Banerjee, P.; Chan, K.-C.; Gipe, D.A.; et al. Evinacumab for Homozygous Familial Hypercholesterolemia. N. Engl. J. Med. 2020, 383, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R.S.; Gaudet, D.; Ballantyne, C.M.; Baum, S.J.; Bergeron, J.; Kershaw, E.E.; Moriarty, P.M.; Rubba, P.; Whitcomb, D.C.; Banerjee, P.; et al. Evinacumab in severe hypertriglyceridemia with or without lipoprotein lipase pathway mutations: A phase 2 randomized trial. Nat. Med. 2023, 29, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Cummings, B.B.; Joing, M.P.; Bouchard, P.R.; Milton, M.N.; Moesta, P.F.; Ramanan, V.; Lane, A.; Hirman, J.; Trauger, J.W.; Maratos-Flier, E.; et al. Safety and efficacy of a novel ANGPTL4 inhibitory antibody for lipid lowering: Results from phase 1 and phase 1b/2a clinical studies. Lancet 2025, 405, 1923–1934. [Google Scholar] [CrossRef] [PubMed]
- The Swedish Medical Products Agency Approves Lipigon’s Phase II Study with Lipisense®. Available online: https://www.lipigon.se/en/investors/press-releases/?slug=the-swedish-medical-products-agency-approves-lipigon-s-phase-96913 (accessed on 7 February 2024).
- Malick, W.A.; Waksman, O.; Do, R.; Koenig, W.; Pradhan, A.D.; Stroes, E.S.; Rosenson, R.S. Clinical Trial Design for Triglyceride- Rich Lipoprotein-Lowering Therapies: JACC Focus Seminar 3/3. J. Am. Coll. Cardiol. 2023, 81, 1646–1658. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Bays, H.E.; Miller, M.; Cain, J.E., 3rd; Wasilewska, K.; Andrawis, N.S.; Parli, T.; Feng, S.; Sterling, L.; Tseng, L.; et al. The FGF21 analog pegozafermin in severe hypertriglyceridemia: A randomized phase 2 trial. Nat. Med. 2023, 29, 1782–1792. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, J.; Domingues, M.; Ferreira, A. Emerging Triglyceride-Rich Lipoprotein Targeted Therapies: An Unmet Need in Cardiometabolic Disease. Pharmaceutics 2025, 17, 1107. https://doi.org/10.3390/pharmaceutics17091107
Ferreira J, Domingues M, Ferreira A. Emerging Triglyceride-Rich Lipoprotein Targeted Therapies: An Unmet Need in Cardiometabolic Disease. Pharmaceutics. 2025; 17(9):1107. https://doi.org/10.3390/pharmaceutics17091107
Chicago/Turabian StyleFerreira, Jorge, Miguel Domingues, and António Ferreira. 2025. "Emerging Triglyceride-Rich Lipoprotein Targeted Therapies: An Unmet Need in Cardiometabolic Disease" Pharmaceutics 17, no. 9: 1107. https://doi.org/10.3390/pharmaceutics17091107
APA StyleFerreira, J., Domingues, M., & Ferreira, A. (2025). Emerging Triglyceride-Rich Lipoprotein Targeted Therapies: An Unmet Need in Cardiometabolic Disease. Pharmaceutics, 17(9), 1107. https://doi.org/10.3390/pharmaceutics17091107







