Yeast-Derived Glucan Particles: Biocompatibility, Efficacy, and Immunomodulatory Potential as Adjuvants and Delivery Systems
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. GPs’ Purification
2.2.2. Protein and CL097 Encapsulation
2.2.3. Size and Zeta Potential
2.2.4. Protein Electrophoresis
2.2.5. Transmission Electron Microscopy (TEM)
2.2.6. Raw 264.7 Cell Line
Uptake Studies by Flow Cytometry and TEM
Cell Viability Assay
Reactive Oxygen Species (ROS) Production Assay
LPS-Spiked ROS Production Assay
Nitric Oxide (NO) Production Assay
LPS-Spiked NO Production Assay
2.2.7. Hemolysis Assay
2.2.8. Human PBMCs’ Isolation, Monocyte Purification, and moDCs’ Differentiation
2.2.9. Proliferation Assay in Human PBMCs
2.2.10. Cytokine Quantification in Human PBMCs
2.2.11. Uptake Studies in Human Monocytes Through Confocal Laser Scanning Microscopy (CLSM)
2.2.12. Cytokine and Chemokine Quantification in Human moDCs
2.2.13. In Vivo Immunization Study
Blood Collection
Quantification of Serum Total IgG
Spleen Cell Isolation
Cytokine Quantification
Flow Cytometry Analysis
2.2.14. Statistical Analysis
3. Results and Discussion
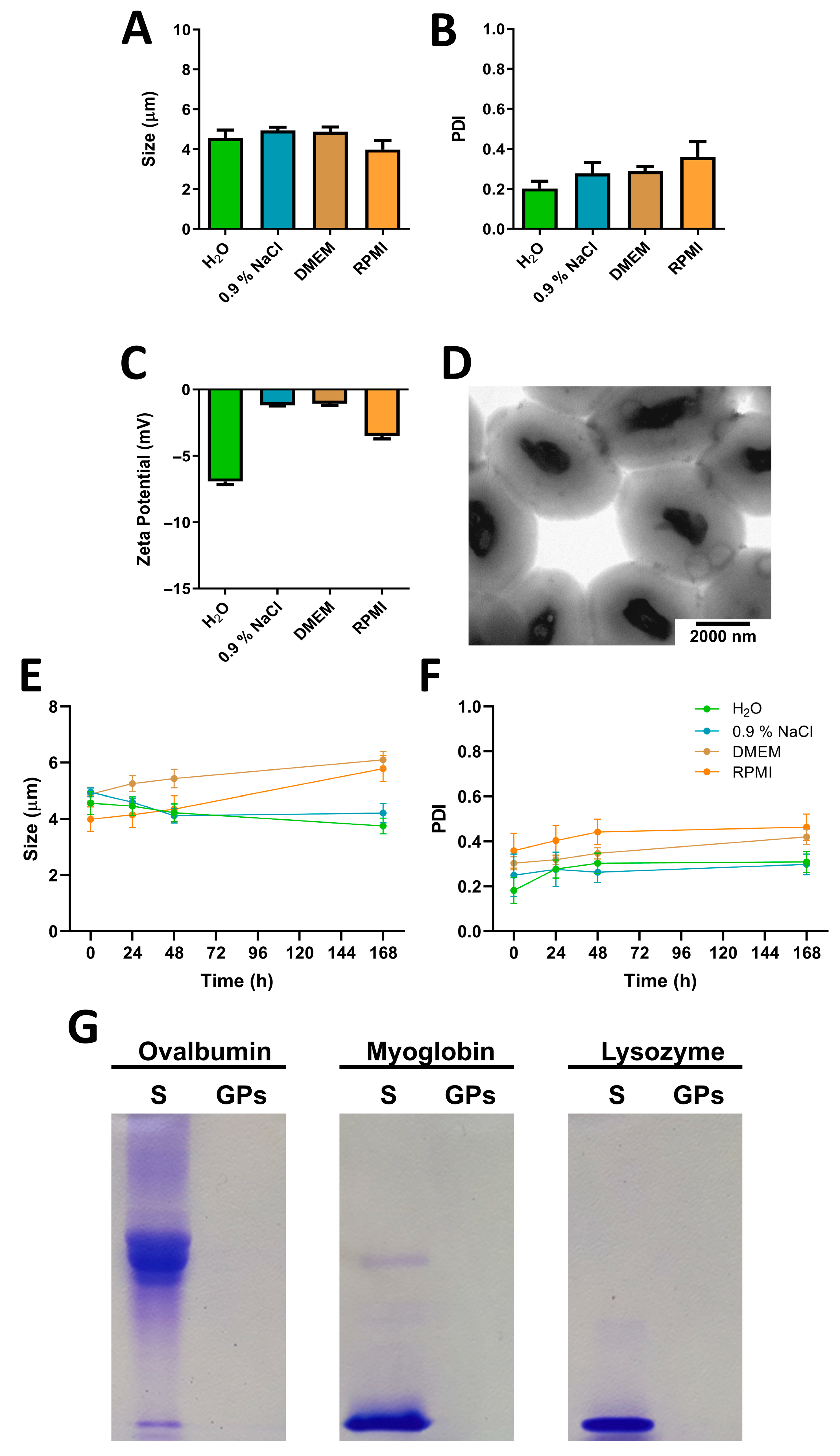
3.1. Physicochemical Characterization of GPs: Insights into Size, Surface Charge and Protein Encapsulation
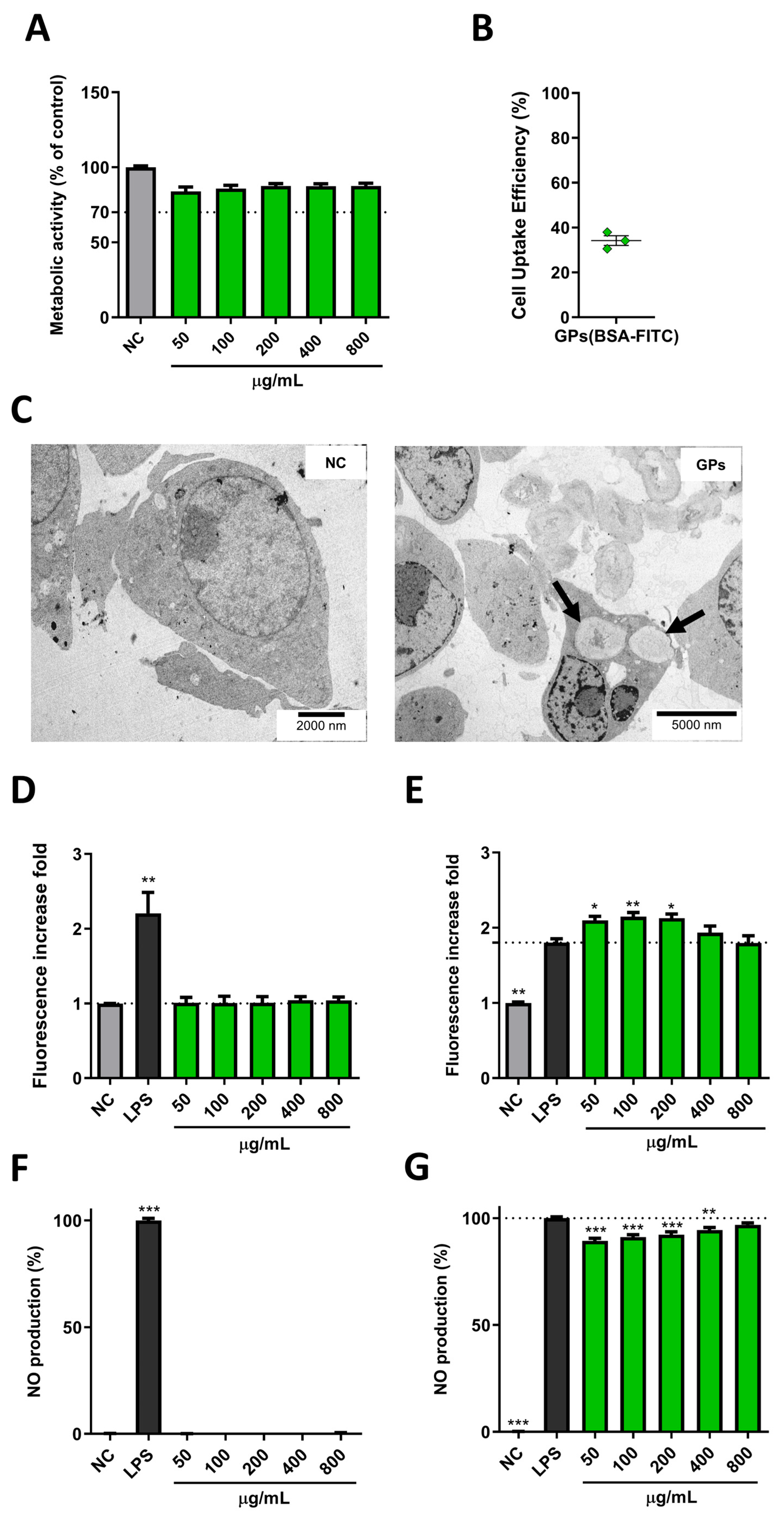
3.2. Immunotoxicity Profile of GPs in RAW 264.7 Macrophages
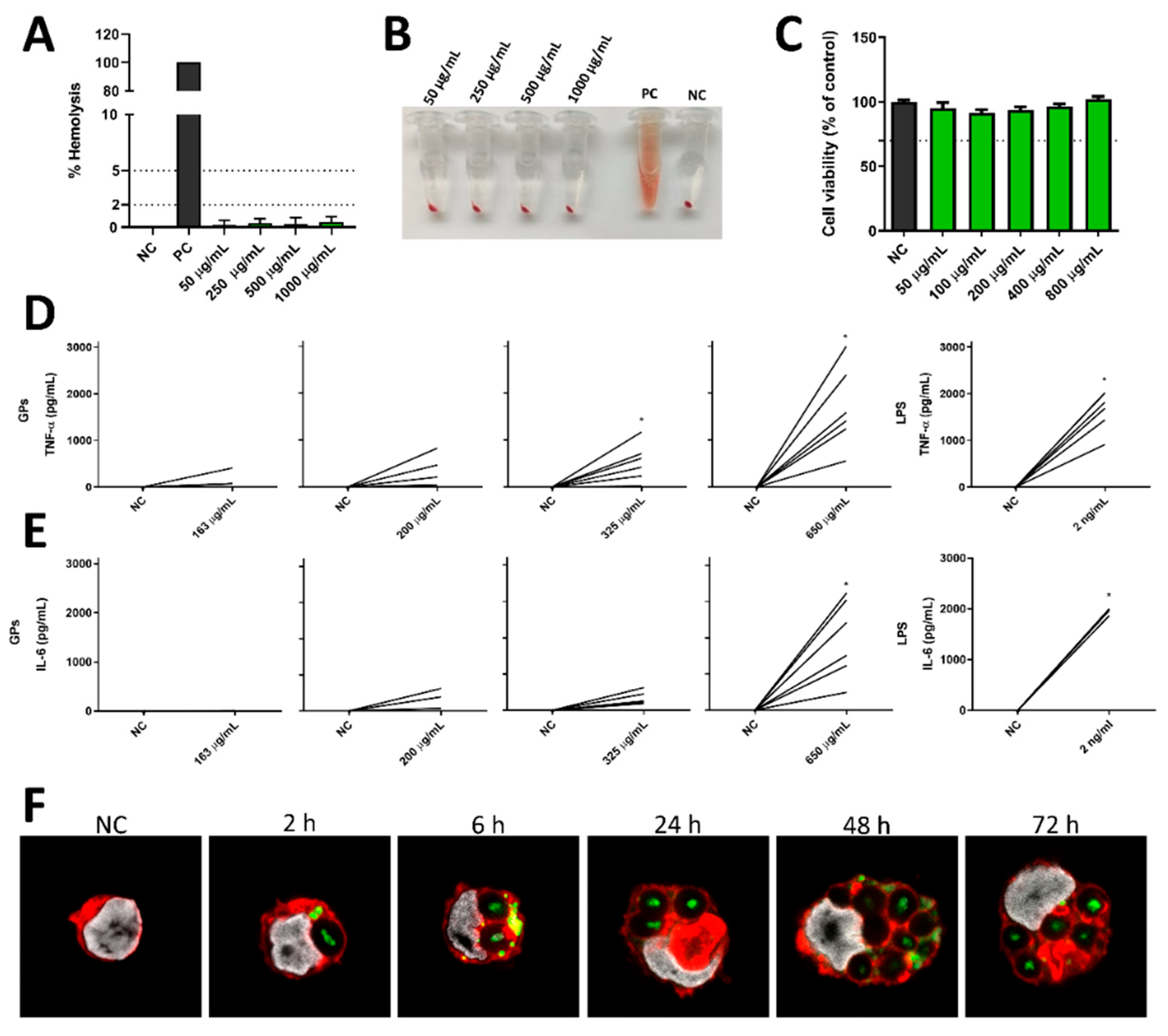
3.3. GPs’ Effects on Hemolysis and Immunomodulation in Human PBMCs
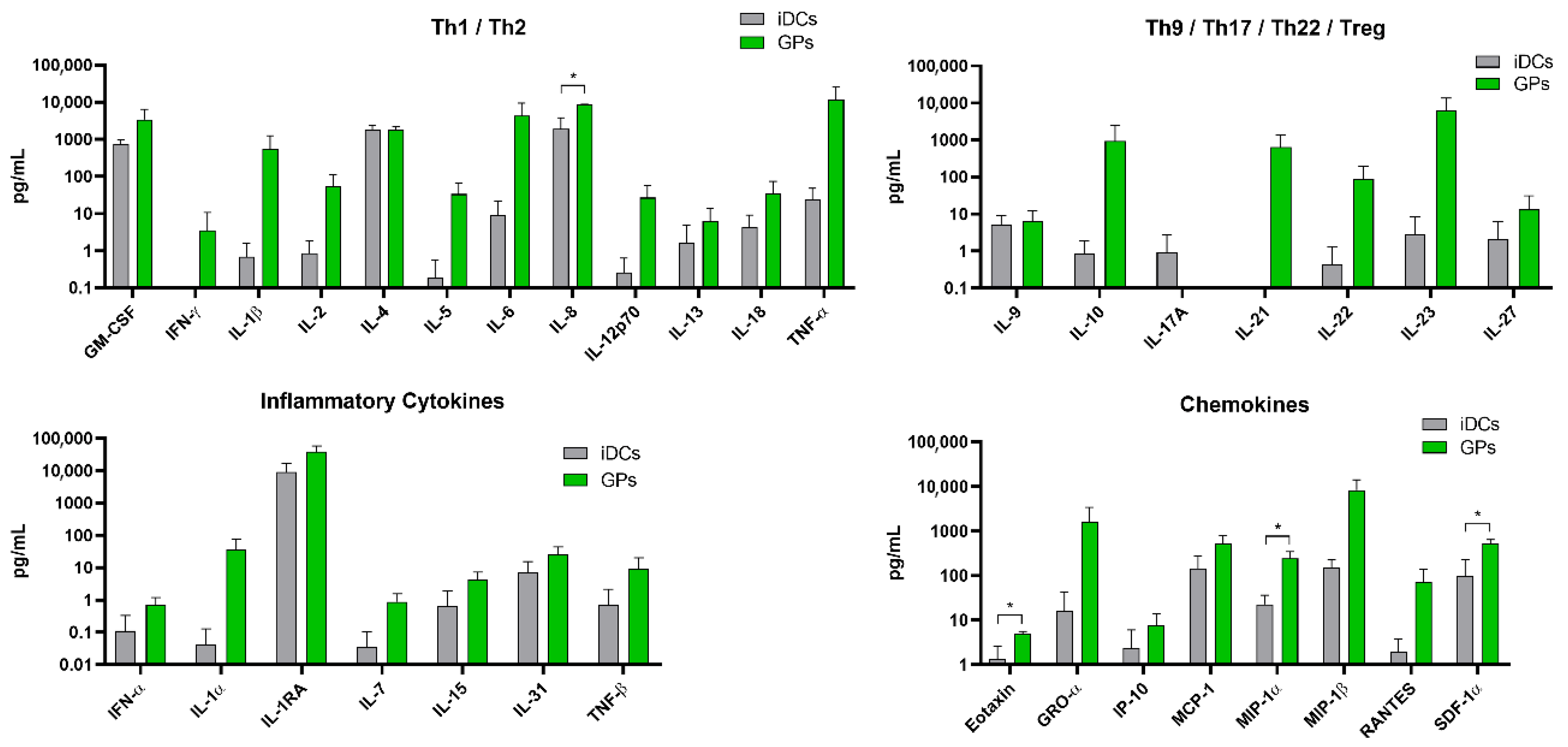
3.4. GPs’ Influence on Human moDCs’ Cytokine Release
3.5. GPs (HHC) Vaccination’s Influence on Humoral Immune Response
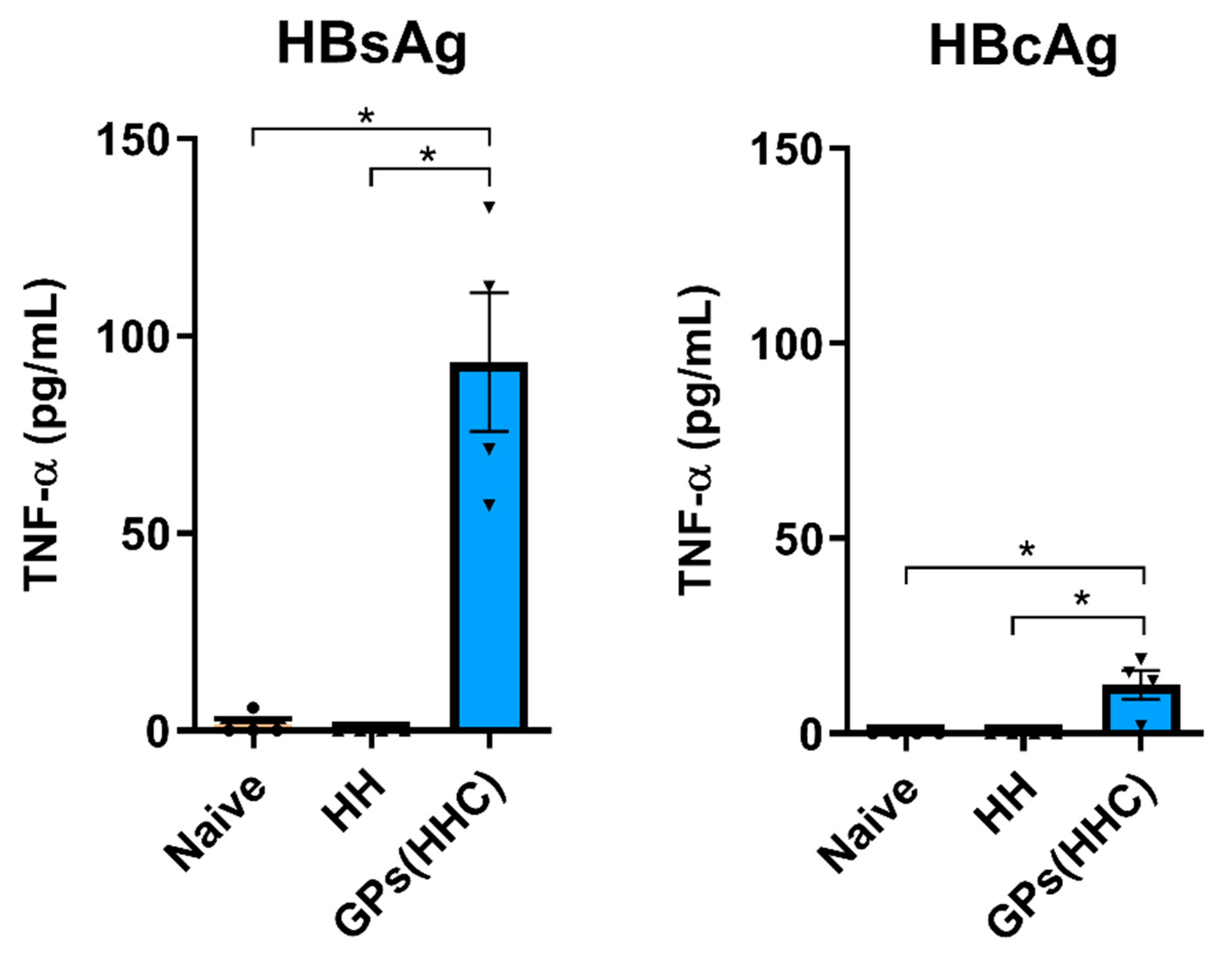
3.6. Spleen Cell TNF-α Production upon Antigen Restimulation
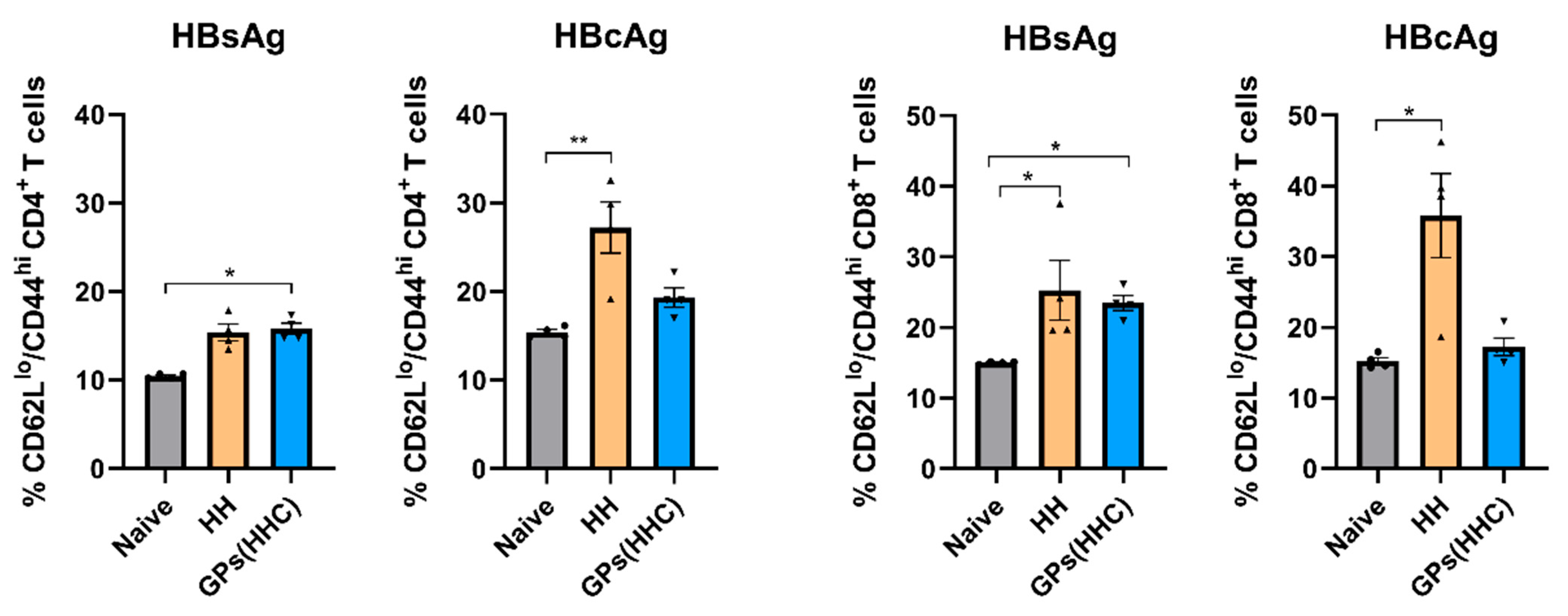
3.7. Effector Memory T Cell Phenotype After Spleen Cell Restimulation
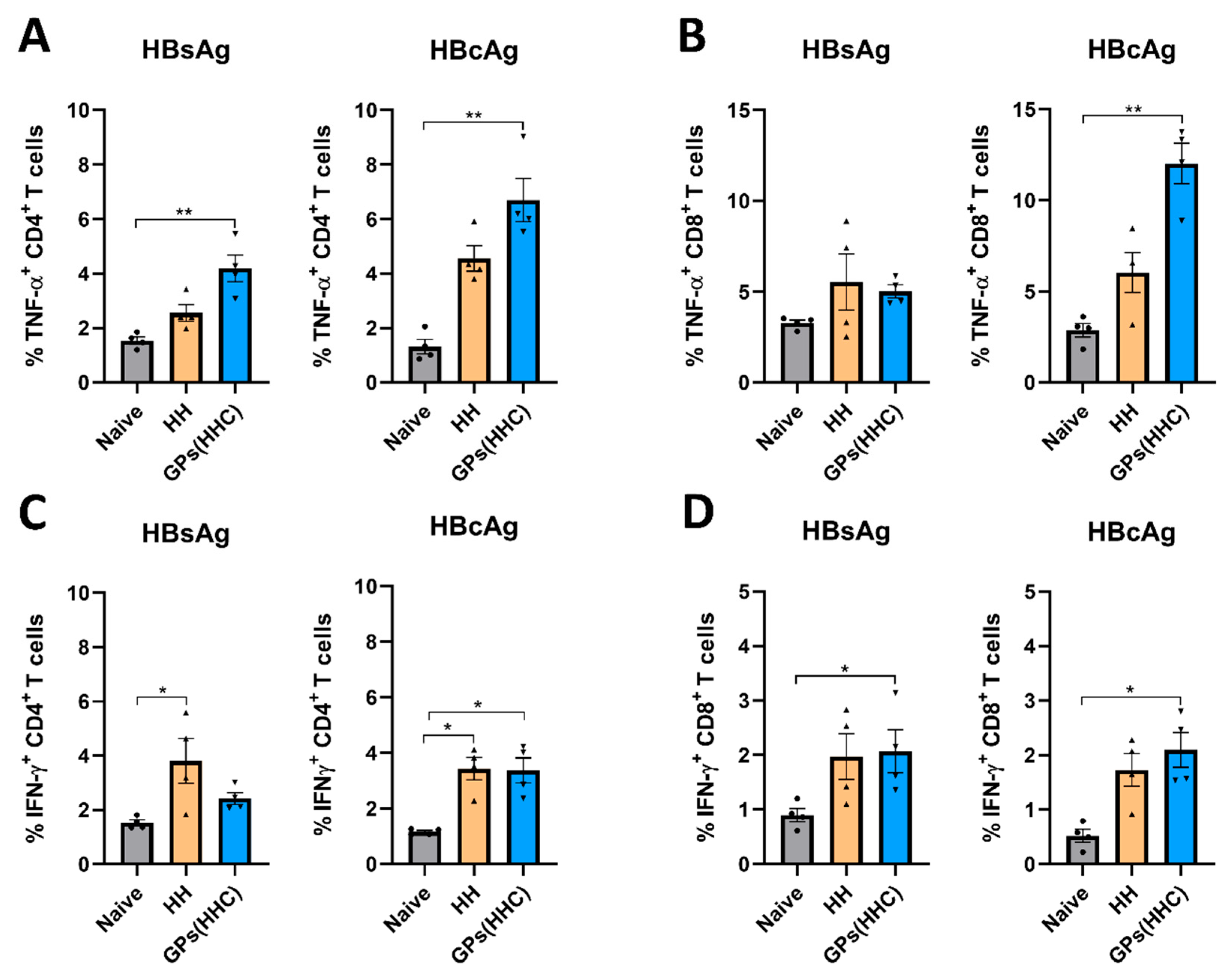
3.8. Specific T Cell Cytokine Production Elicited by GPs (HHC) in C57BL/6 Mice
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Zhu, M.; Wang, R.; Nie, G. Applications of nanomaterials as vaccine adjuvants. Hum. Vaccines Immunother. 2014, 10, 2761–2774. [Google Scholar] [CrossRef]
- Hubbell, J.A.; Thomas, S.N.; Swartz, M.A. Materials engineering for immunomodulation. Nature 2009, 462, 449–460. [Google Scholar] [CrossRef]
- Jesus, S.; Marques, A.P.; Duarte, A.; Soares, E.; Costa, J.P.; Colaço, M.; Schmutz, M.; Som, C.; Borchard, G.; Wick, P.; et al. Chitosan Nanoparticles: Shedding Light on Immunotoxicity and Hemocompatibility. Front. Bioeng. Biotechnol. 2020, 8, 100. [Google Scholar] [CrossRef]
- Engin, A.B.; Hayes, A.W. The impact of immunotoxicity in evaluation of the nanomaterials safety. Toxicol. Res. Appl. 2018, 2, 239784731875557. [Google Scholar] [CrossRef]
- Da Silva, J.; Jesus, S.; Bernardi, N.; Colaço, M.; Borges, O. Poly(D,L-Lactic Acid) Nanoparticle Size Reduction Increases Its Immunotoxicity. Front. Bioeng. Biotechnol. 2019, 7, 137. [Google Scholar] [CrossRef]
- Lanone, S.; Boczkowski, J. Biomedical Applications and Potential Health Risks of Nanomaterials: Molecular Mechanisms. Curr. Mol. Med. 2006, 6, 651–663. [Google Scholar] [CrossRef]
- Abraham, A.; Ostroff, G.; Levitz, S.M.; Oyston, P.C.F. A novel vaccine platform using glucan particles for induction of protective responses against Francisella tularensis and other pathogens. Clin. Exp. Immunol. 2019, 198, 143–152. [Google Scholar] [CrossRef]
- Bohn, J.A.; BeMiller, J.N. (1→3)-β-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995, 28, 3–14. [Google Scholar] [CrossRef]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. β-glucan recognition by the innate immune system. Immunol. Rev. 2009, 230, 38–50. [Google Scholar] [CrossRef]
- Soares, E.; Groothuismink, Z.M.A.; Boonstra, A.; Borges, O. Glucan Particles Are a Powerful Adjuvant for the HBsAg, Favoring Antiviral Immunity. Mol. Pharm. 2019, 16, 1971–1981. [Google Scholar] [CrossRef]
- Geller, A.; Shrestha, R.; Yan, J. Yeast-Derived β-Glucan in Cancer: Novel Uses of a Traditional Therapeutic. Int. J. Mol. Sci. 2019, 20, 3618. [Google Scholar] [CrossRef] [PubMed]
- Soto, E.R.; Ostroff, G.R. Characterization of Multilayered Nanoparticles Encapsulated in Yeast Cell Wall Particles for DNA Delivery. Bioconjugate Chem. 2008, 19, 840–848. [Google Scholar] [CrossRef]
- Hong, F.; Yan, J.; Baran, J.T.; Allendorf, D.J.; Hansen, R.D.; Ostroff, G.R.; Xing, P.X.; Cheung, N.-K.V.; Ross, G.D. Mechanism by Which Orally Administered β-1,3-Glucans Enhance the Tumoricidal Activity of Antitumor Monoclonal Antibodies in Murine Tumor Models. J. Immunol. 2004, 173, 797–806. [Google Scholar] [CrossRef]
- Soto, E.R.; Caras, A.C.; Kut, L.C.; Castle, M.K.; Ostroff, G.R. Glucan Particles for Macrophage Targeted Delivery of Nanoparticles. J. Drug Deliv. 2012, 2012, 143524. [Google Scholar] [CrossRef]
- Huang, H.; Ostroff, G.R.; Lee, C.K.; Specht, C.A.; Levitz, S.M.; Dromer, F. Robust Stimulation of Humoral and Cellular Immune Responses following Vaccination with Antigen-Loaded β-Glucan Particles. mBio 2010, 1, e00164-10. [Google Scholar] [CrossRef]
- Aouadi, M.; Tesz, G.J.; Nicoloro, S.M.; Wang, M.; Chouinard, M.; Soto, E.; Ostroff, G.R.; Czech, M.P. Orally delivered siRNA targeting macrophage Map4k4 suppresses systemic inflammation. Nature 2009, 458, 1180–1184. [Google Scholar] [CrossRef]
- Soto, E.R.; O’Connell, O.; Dikengil, F.; Peters, P.J.; Clapham, P.R.; Ostroff, G.R. Targeted Delivery of Glucan Particle Encapsulated Gallium Nanoparticles Inhibits HIV Growth in Human Macrophages. J. Drug Deliv. 2016, 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Ostroff, G.R.; Lee, C.K.; Specht, C.A.; Levitz, S.M. Characterization and Optimization of the Glucan Particle-Based Vaccine Platform. Clin. Vaccine Immunol. 2013, 20, 1585–1591. [Google Scholar] [CrossRef]
- Meng, Z.; Chen, Y.; Lu, M. Advances in Targeting the Innate and Adaptive Immune Systems to Cure Chronic Hepatitis B Virus Infection. Front. Immunol. 2019, 10, 3127. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ge, J.; Ren, S.; Zhou, T.; Sun, Y.; Sun, H.; Gu, Y.; Huang, H.; Xu, Z.; Chen, X.; et al. Hepatitis B surface antigen (HBsAg) and core antigen (HBcAg) combine CpG oligodeoxynucletides as a novel therapeutic vaccine for chronic hepatitis B infection. Vaccine 2015, 33, 4247–4254. [Google Scholar] [CrossRef]
- Georg, P.; Sander, L.E. Innate sensors that regulate vaccine responses. Curr. Opin. Immunol. 2019, 59, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, K.; Wu, Z.; Liu, Y.; Liu, S.; Zou, Z.; Chen, S.H.; Qu, C. Immunizations with hepatitis B viral antigens and a TLR7/8 agonist adjuvant induce antigen-specific immune responses in HBV-transgenic mice. Int. J. Infect. Dis. 2014, 29, 31–36. [Google Scholar] [CrossRef]
- De Smet, R.; Demoor, T.; Verschuere, S.; Dullaers, M.; Ostroff, G.R.; Leclercq, G.; Allais, L.; Pilette, C.; Dierendonck, M.; De Geest, B.G.; et al. beta-Glucan microparticles are good candidates for mucosal antigen delivery in oral vaccination. J. Control. Release 2013, 172, 671–678. [Google Scholar] [CrossRef]
- Jesus, S.; Soares, E.; Costa, J.; Borchard, G.; Borges, O. Immune response elicited by an intranasally delivered HBsAg low-dose adsorbed to poly-epsilon-caprolactone based nanoparticles. Int. J. Pharm. 2016, 504, 59–69. [Google Scholar] [CrossRef]
- Volpato, H.; Scariot, D.B.; Soares, E.F.P.; Jacomini, A.P.; Rosa, F.A.; Sarragiotto, M.H.; Ueda-Nakamura, T.; Rubira, A.F.; Pereira, G.M.; Manadas, R.; et al. In Vitro anti-Leishmania activity of T6 synthetic compound encapsulated in yeast-derived β-(1,3)-d-glucan particles. Int. J. Biol. Macromol. 2018, 119, 1264–1275. [Google Scholar] [CrossRef]
- Soares, E.; Jesus, S.; Borges, O. Oral hepatitis B vaccine: Chitosan or glucan based delivery systems for efficient HBsAg immunization following subcutaneous priming. Int. J. Pharm. 2018, 535, 261–271. [Google Scholar] [CrossRef]
- Félix, P.; Melo, A.A.; Costa, J.P.; Silva, V.; Borges, O. Exploring TLR agonists as adjuvants for COVID-19 oral vaccines. Vaccine 2025, 53, 127078. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Gou, J.; Sun, W.; Tao, X.; Tan, X.; Wang, P.; Zhang, Y.; He, H.; Yin, T.; Tang, X. Entrapping of Nanoparticles in Yeast Cell Wall Microparticles for Macrophage-Targeted Oral Delivery of Cabazitaxel. Mol. Pharm. 2018, 15, 2870–2882. [Google Scholar] [CrossRef]
- Yu, Z.; Li, Q.; Wang, J.; Yu, Y.; Wang, Y.; Zhou, Q.; Li, P. Reactive Oxygen Species-Related Nanoparticle Toxicity in the Biomedical Field. Nanoscale Res. Lett. 2020, 15, 115. [Google Scholar] [CrossRef]
- Colaco, M.; Marques, A.P.; Jesus, S.; Duarte, A.; Borges, O. Safe-by-Design of Glucan Nanoparticles: Size Matters When Assessing the Immunotoxicity. Chem. Res. Toxicol. 2020, 33, 915–932. [Google Scholar] [CrossRef] [PubMed]
- Panão Costa, J.; Jesus, S.; Colaco, M.; Duarte, A.; Soares, E.; Borges, O. Endotoxin contamination of nanoparticle formulations: A concern in vaccine adjuvant mechanistic studies. Vaccine 2023, 41, 3481–3485. [Google Scholar] [CrossRef] [PubMed]
- Colaço, M.; Roquito, T.; Panão Costa, J.; Cruz, M.T.; Borges, O. The Effect of Curcumin-Loaded Glucan Nanoparticles on Immune Cells: Size as a Critical Quality Attribute. Pharmaceutics 2023, 15, 623. [Google Scholar] [CrossRef]
- Li, P.; Zhang, X.; Cheng, Y.; Li, J.; Xiao, Y.; Zhang, Q.; Zong, A.; Zhong, C.; Wang, F. Preparation and in vitro immunomodulatory effect of curdlan sulfate. Carbohydr. Polym. 2014, 102, 852–861. [Google Scholar] [CrossRef]
- Sun, Y.; Shi, X.; Zheng, X.; Nie, S.; Xu, X. Inhibition of dextran sodium sulfate-induced colitis in mice by baker’s yeast polysaccharides. Carbohydr. Polym. 2019, 207, 371–381. [Google Scholar] [CrossRef]
- Xu, X.; Yasuda, M.; Mizuno, M.; Ashida, H. β-Glucan from Saccharomyces cerevisiae reduces lipopolysaccharide-induced inflammatory responses in RAW264.7 macrophages. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2012, 1820, 1656–1663. [Google Scholar] [CrossRef]
- Cai, Y.; Xu, X.; Zhang, Y.; Liu, Q.; Wang, Z.; Liu, Z. Immunomodulatory melanin@Pt nanoparticle-reinforced adhesive hydrogels for healing diabetic oral ulcers. Chem. Eng. J. 2024, 475, 150372. [Google Scholar] [CrossRef]
- Qi, X.; Li, Y.; Xiang, Y.; Chen, Y.; Shi, Y.; Ge, X.; Zeng, B.; Shen, J. Hyperthermia-enhanced immunoregulation hydrogel for oxygenation and ROS neutralization in diabetic foot ulcers. Cell Biomater. 2025, 1, 100020. [Google Scholar] [CrossRef]
- ASTM-E2524–08; Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles. ASTM International: West Conshohocken, PA, USA, 2013.
- Soares, E.; Cordeiro, R.; Faneca, H.; Borges, O. Polym. Polymeric nanoengineered HBsAg DNA vaccine designed in combination with beta-glucan. Int. J. Biol. Macromol. 2019, 122, 930–939. [Google Scholar] [CrossRef]
- Soto, E.; Ostroff, G. Glucan Part. Carr. Nanoparticles Macrophage-Target. Delivery 2012, 1119, 57–79. [Google Scholar]
- Davenport, R.D. An introduction to chemokines and their roles in transfusion medicine. Vox Sang. 2009, 96, 183–198. [Google Scholar] [CrossRef]
- Feijoo, E.; Alfaro, C.; Mazzolini, G.; Serra, P.; Penuelas, I.; Arina, A.; Huarte, E.; Tirapu, I.; Palencia, B.; Murillo, O.; et al. Dendritic cells delivered inside human carcinomas are sequestered by interleukin-8. Int. J. Cancer 2005, 116, 275–281. [Google Scholar] [CrossRef]
- Chen, H.-C.; Kung, M.-L.; Huang, W.-X.; Fu, R.-H.; Yu, A.Y.-H.; Yang, Y.-T.; Hung, H.-S. Delivery of stromal-derived factor-1α via biocompatible gold nanoparticles promotes dendritic cells viability and migration. Colloids. Surf. A Physicochem. Eng. Asp. 2021, 628, 127298. [Google Scholar] [CrossRef]
- Lukacs-Kornek, V.; Engel, D.; Tacke, F.; Kurts, C. The role of chemokines and their receptors in dendritic cell biology. Front. Biosci. 2008, 13, 2238–2252. [Google Scholar] [CrossRef] [PubMed]
- Kara, E.E.; Comerford, I.; Fenix, K.A.; Bastow, C.R.; Gregor, C.E.; McKenzie, D.R.; McColl, S.R. Tailored immune responses: Novel effector helper T cell subsets in protective immunity. PLoS Pathog. 2014, 10, e1003905. [Google Scholar] [CrossRef]
- Sallusto, F.; Geginat, J.; Lanzavecchia, A. Central memory and effector memory T cell subsets: Function, generation, and maintenance. Annu. Rev. Immunol. 2004, 22, 745–763. [Google Scholar] [CrossRef] [PubMed]
- Baaten, B.J.; Li, C.R.; Deiro, M.F.; Lin, M.M.; Linton, P.J.; Bradley, L.M. CD44 regulates survival and memory development in Th1 cells. Immunity 2010, 32, 104–115. [Google Scholar] [CrossRef]
- Lucifora, J.; Xia, Y.; Reisinger, F.; Zhang, K.; Stadler, D.; Cheng, X.; Protzer, U. Specific and Nonhepatotoxic Degradation of Nuclear Hepatitis B Virus cccDNA. Science 2014, 343, 1221–1228. [Google Scholar] [CrossRef]
- Allweiss, L.; Dandri, M. The Role of cccDNA in HBV Maintenance. Viruses 2017, 9, 156. [Google Scholar] [CrossRef]
- Hsu, H.-C.; Su, I.-J.; Lai, M.-Y.; Chen, D.-S.; Chang, M.-H.; Chuang, S.-M.; Sung, J.-L. Biologic and prognostic significance of hepatocyte hepatitis B core antigen expressions in the natural course of chronic hepatitis B virus infection. J. Hepatol. 1987, 5, 45–50. [Google Scholar] [CrossRef]
- Yang, L.; Shi, L.P.; Chen, H.J.; Tong, X.K.; Wang, G.F.; Zhang, Y.M.; Wang, W.L.; Feng, C.L.; He, P.L.; Zhu, F.H.; et al. Isothiafludine, a novel non-nucleoside compound, inhibits hepatitis B virus replication through blocking pregenomic RNA encapsidation. Acta. Pharmacol. Sin. 2014, 35, 410–418. [Google Scholar] [CrossRef]
- Liu, Y.; Meng, F.; Feng, W.; Chen, Z.; Xing, H.; Zheng, A. Oral DNA vaccine utilizing the yeast cell wall for Dectin-1 receptor-mediated enhancement of mucosal immunity. Mol. Pharmaceutics. 2025, 22, 1241–1252. [Google Scholar] [CrossRef]
- Azevedo-Silva, J.; Amorim, M.; Tavares-Valente, D.; Sousa, P.; Mohamath, R.; Voigt, E.A.; Guderian, J.A.; Kinsey, R.; Viana, S.; Reis, F.; et al. Exploring yeast glucans for vaccine enhancement: Sustainable strategies for overcoming adjuvant challenges in a SARS-CoV-2 model. Eur. J. Pharm. Biopharm. 2024, 205, 114538. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Zhu, Z.; Qi, C. β-Glucan—A promising immunocyte-targeting drug delivery vehicle: Superiority, applications and future prospects. Carbohydr. Polym. 2024, 339, 122252. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.; Chen, X.; Chen, Y.; Li, M.; Zhang, L.; Luo, Q.; Wei, C.; Huang, G.; Zhao, P.; Sun, B.; et al. Self-cascade of ROS/glucose-scavenging immunomodulatory hydrogels for programmed therapeutics of infected diabetic ulcers via Nrf2/NF-κB pathway. Small 2025, 21, 2411189. [Google Scholar] [CrossRef] [PubMed]
- Colaco, M.; Costa, J.P.; Borges, O. Glucan Particles: Choosing the Appropriate Size to Use as a Vaccine Adjuvant. In Vaccine Design: Methods and Protocols. Vol. 3: Resources for Vaccine Development; Thomas, S., Ed.; Springer: New York, NY, USA, 2022; Volume 2412, pp. 269–288. [Google Scholar]

| S.C. Route | HBsAg (µg/Dose) | HBcAg (µg/Dose) | GPs (µg/Dose) | CL097 (µg/Dose) | Priming, Boost, Euthanasia (Days) |
|---|---|---|---|---|---|
| Naive (n = 4) | --- | --- | --- | --- | 21 |
| HH (n = 4) | 1.5 | 1.5 | 300 | --- | 0, 14, 21 |
| GPs (HHC) (n = 4) | 1.5 | 1.5 | 300 | 5 | 0, 14, 21 |
| H2O | 0.9% NaCl | DMEM | RPMI | |
|---|---|---|---|---|
| Size (µm) | 4565 ± 692 | 4945 ± 274 | 4875 ± 419 | 3987 ± 772 |
| PDI | 0.203 ± 0.120 | 0.278 ± 0.163 | 0.288 ± 0.074 | 0.358 ± 0.135 |
| Zeta Potential (mV) | −6.94 ± 0.81 | −1.19 ± 0.19 | −1.07 ± 0.43 | −3.49 ± 0.42 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panão-Costa, J.; Colaço, M.; Jesus, S.; Lebre, F.; Cruz, M.T.; Alfaro-Moreno, E.; Borges, O. Yeast-Derived Glucan Particles: Biocompatibility, Efficacy, and Immunomodulatory Potential as Adjuvants and Delivery Systems. Pharmaceutics 2025, 17, 1032. https://doi.org/10.3390/pharmaceutics17081032
Panão-Costa J, Colaço M, Jesus S, Lebre F, Cruz MT, Alfaro-Moreno E, Borges O. Yeast-Derived Glucan Particles: Biocompatibility, Efficacy, and Immunomodulatory Potential as Adjuvants and Delivery Systems. Pharmaceutics. 2025; 17(8):1032. https://doi.org/10.3390/pharmaceutics17081032
Chicago/Turabian StylePanão-Costa, João, Mariana Colaço, Sandra Jesus, Filipa Lebre, Maria T. Cruz, Ernesto Alfaro-Moreno, and Olga Borges. 2025. "Yeast-Derived Glucan Particles: Biocompatibility, Efficacy, and Immunomodulatory Potential as Adjuvants and Delivery Systems" Pharmaceutics 17, no. 8: 1032. https://doi.org/10.3390/pharmaceutics17081032
APA StylePanão-Costa, J., Colaço, M., Jesus, S., Lebre, F., Cruz, M. T., Alfaro-Moreno, E., & Borges, O. (2025). Yeast-Derived Glucan Particles: Biocompatibility, Efficacy, and Immunomodulatory Potential as Adjuvants and Delivery Systems. Pharmaceutics, 17(8), 1032. https://doi.org/10.3390/pharmaceutics17081032








