Physiologically Based Pharmacokinetic Simulation of Tofacitinib in Humans Using Extrapolation from Single-Species Renal Failure Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Data
2.2. Dedrick Plot
2.3. Extrapolation from Rats to Humans Using Single-Species Method
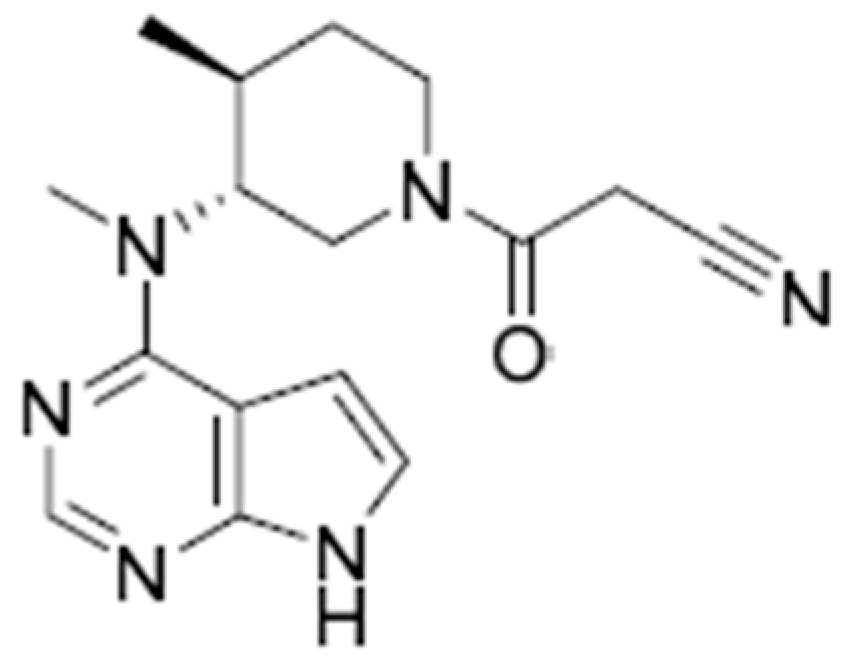
2.4. PBPK Model Development for Tofacitinib
2.5. PBPK Model Structure for Healthy Subjects and Patients with Renal Failure
| PK-Sim | Simcyp | |||
|---|---|---|---|---|
| Value | Reference | Value | Reference | |
| Absorption | ||||
| Intestinal permeability (cm/min) | 6.3 × 10−6 | Predicted based on MDCK cell [34] | ||
| Peff,man (cm/s) | 22.1 × 10−6 | Predicted based on Caco-2 cell [34] | ||
| Distribution | ||||
| Partition coefficients | Rodgers and Rowland | |||
| Vss (L/kg) | [35] | Predicted | ||
| Metabolism (CL) | Type: Plasma CL | Type: In vivo CL | ||
| Normal | 5.93 | Calculated using single species method [36] (mL/min/kg) | 26.9 | Calculated using single-species method [37] (L/h) |
| Moderate renal failure | 3.69 | 17.3 | ||
| Severe renal failure | 2.23 | 11.7 | ||
| Excretion (CLR) | ||||
| Normal | 1.95 | Calculated using single-species method (mL/min/kg) | 8.86 | Calculated using single-species method for healthy volunteer (L/h) |
| Moderate renal failure | 0.202 | |||
| Severe renal failure | 0.0164 | |||
2.6. Statistical Analysis
3. Results
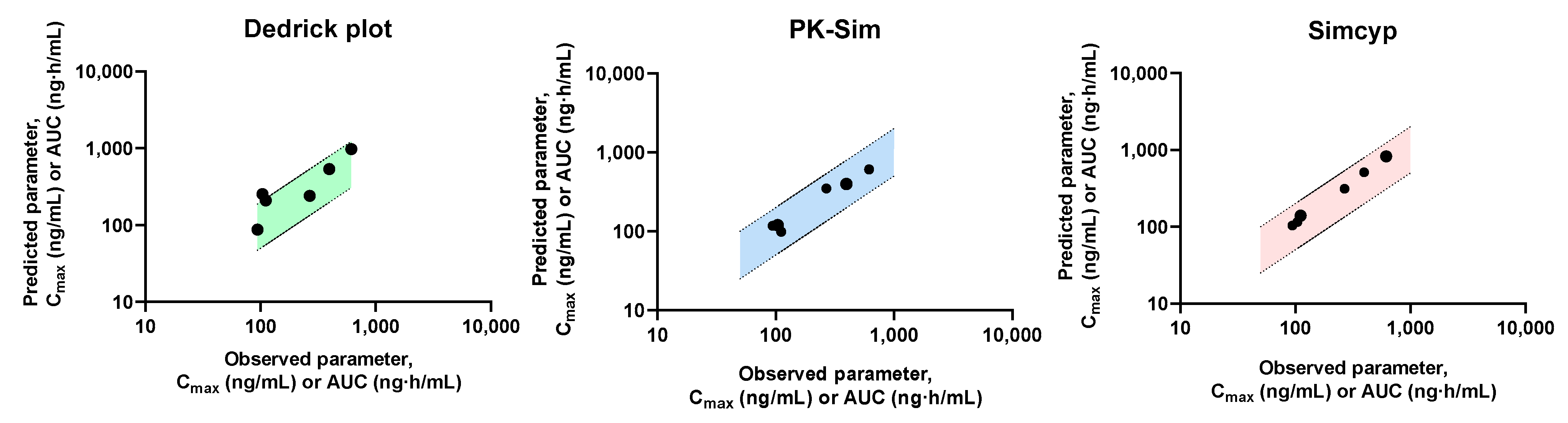
3.1. Human Extrapolation Using Dedrick Plot
3.2. PBPK Model Development Using PK-SIM and Simcyp
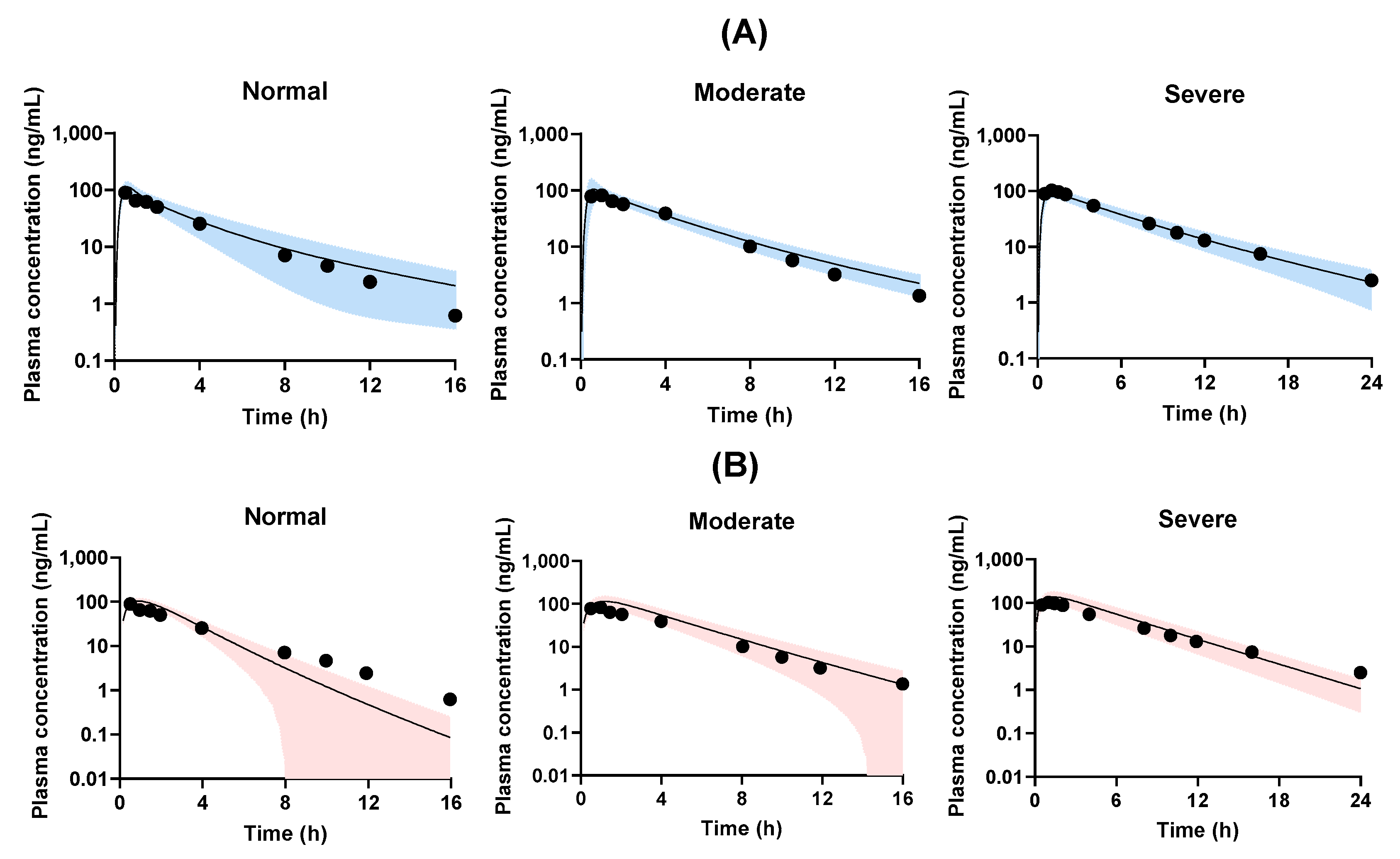
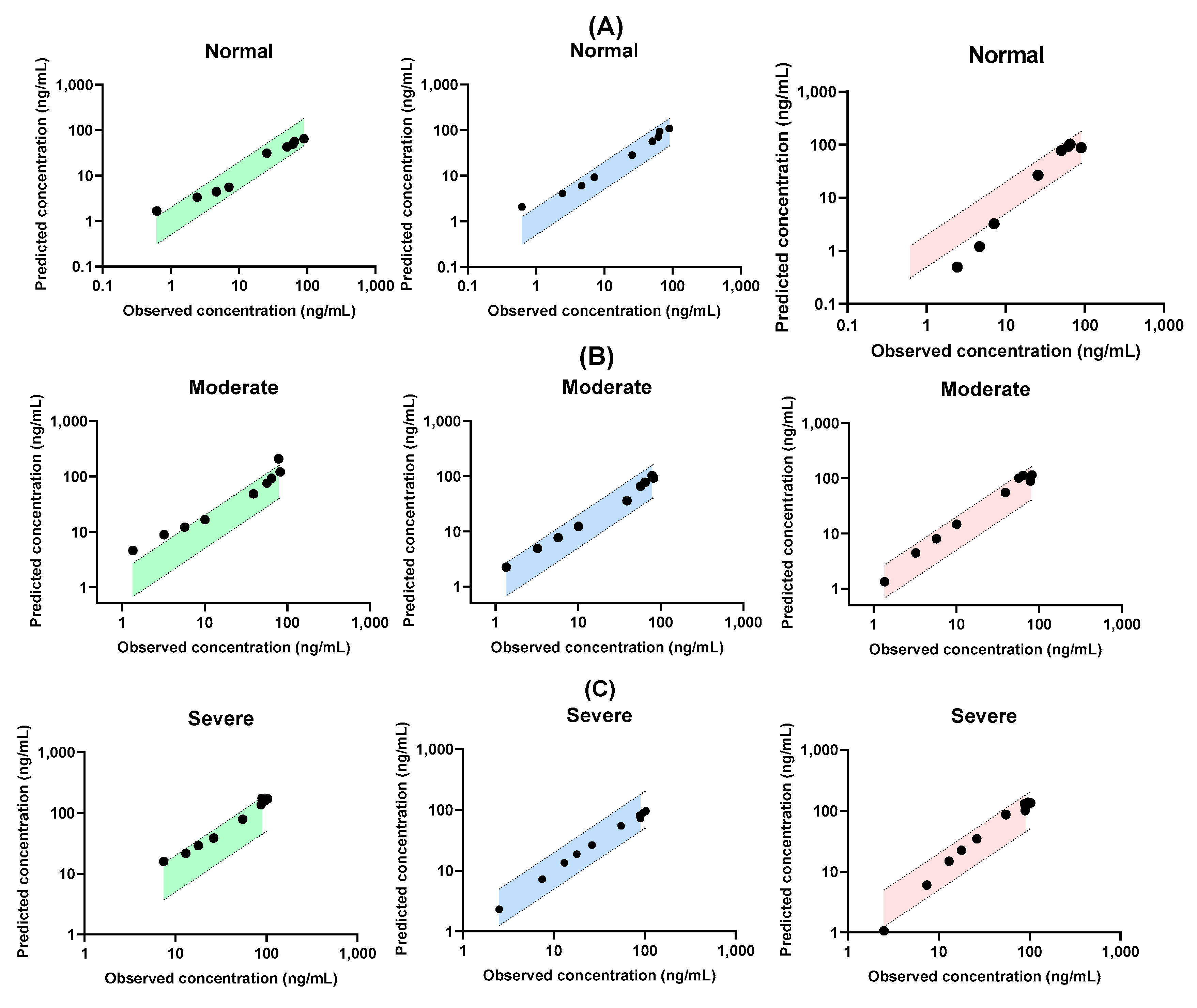
3.3. Predicted Model Validation for Renal Failure Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhuang, X.; Lu, C. PBPK modeling and simulation in drug research and development. Acta Pharm. Sin. B 2016, 6, 430–440. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. European Public Assessment Report for Xeljanz, International Non-Proprietary Name: Tofacitinib. Available online: https://www.ema.europa.eu/en/documents/assessment-report/xeljanz-epar-public-assessment-report_en.pdf (accessed on 4 March 2024).
- Wagner, C.; Pan, Y.; Hsu, V.; Grillo, J.A.; Zhang, L.; Reynolds, K.S.; Sinha, V.; Zhao, P. Predicting the effect of cytochrome P450 inhibitors on substrate drugs: Analysis of physiologically based pharmacokinetic modeling submissions to the US Food and Drug Administration. Clin. Pharmacokinet. 2015, 54, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Luzon, E.; Blake, K.; Cole, S.; Nordmark, A.; Versantvoort, C.; Berglund, E.G. Physiologically based pharmacokinetic modeling in regulatory decision-making at the European Medicines Agency. Clin. Pharmacol. Ther. 2017, 102, 98–105. [Google Scholar] [CrossRef]
- Claxton, L.; Taylor, M.; Soonasra, A.; Bourret, J.A.; Gerber, R.A. An economic evaluation of tofacitinib treatment in rheumatoid arthritis after methotrexate or after 1 or 2 TNF inhibitors from a U.S. payer perspective. J. Manag. Care Spec. Pharm. 2018, 24, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Peyrin-Biroulet, L.; Sharara, A.I.; Su, C.; Modesto, I.; Mundayat, R.; Gunay, L.M.; Salese, L.; Sands, B.E. Efficacy and safety of tofacitinib in ulcerative colitis based on prior tumor necrosis factor inhibitor failure status. Clin. Gastroenterol. Hepatol. 2022, 20, 591–601. [Google Scholar] [CrossRef]
- Fukuda, T.; Naganuma, M.; Kanai, T. Current new challenges in the management of ulcerative colitis. Intest. Res. 2019, 17, 36–44. [Google Scholar] [CrossRef]
- Dowty, M.E.; Lin, J.; Ryder, T.F.; Wang, W.; Walker, G.S.; Vaz, A.; Chan, G.L.; Krishnaswami, S.; Prakash, C. The pharmacokinetics, metabolism, and clearance mechanisms of tofacitinib, a Janus kinase inhibitor, in humans. Drug Metab. Dispos. 2014, 42, 759–773. [Google Scholar] [CrossRef]
- Cada, D.J.; Demaris, K.; Levien, T.L.; Baker, D.E. Tofacitinib. Hosp. Pharm. 2013, 48, 413–424. [Google Scholar] [CrossRef]
- Scott, L.J. Tofacitinib: A review of its use in adult patients with rheumatoid arthritis. Drugs 2013, 73, 857–874. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, S.H. Dose-dependent pharmacokinetics of tofacitinib in rats: Influence of hepatic and intestinal first-pass metabolism. Pharmaceutics 2019, 11, 318. [Google Scholar] [CrossRef]
- Feng, B.; LaPerle, J.L.; Chang, G.; Varma, M.V. Renal clearance in drug discovery and development: Molecular descriptors, drug transporters and disease state. Expert Opin. Drug Metab. Toxicol. 2010, 6, 939–952. [Google Scholar] [CrossRef]
- Saran, R.; Robinson, B.; Abbott, K.C.; Agodoa, L.Y.C.; Bhave, N.; Bragg-Gresham, J.; Balkrishnan, R.; Dietrich, X.; Eckard, A.; Eggers, P.W.; et al. US renal data system 2017 annual data report: Epidemiology of kidney disease in the united States. Am. J. Kidney Dis. 2018, 71 (Suppl. 1), A7. [Google Scholar] [CrossRef]
- Bae, S.H.; Kim, H.S.; Choi, H.; Chang, S.Y.; Kim, S.H. Effects of dextran sulfate sodium-induced ulcerative colitis on the disposition of tofacitinib in rats. Biomol. Ther. 2022, 6, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Jairam, R.K.; Mallurwar, S.R.; Sulochana, S.P.; Chandrasekhar, D.V.; Todmal, U.; Bhamidipati, R.K.; Richter, W.; Srinivas, N.R.; Mullangi, R. Prediction of human pharmacokinetics of fomepizole from preclinical species pharmacokinetics based on normalizing time course profiles. AAPS PharmSciTech 2019, 20, 221. [Google Scholar] [CrossRef] [PubMed]
- Gibaldi, M.; Perrier, D. Pharmacokinetics, 2nd ed.; Marcel-Dekker: New York, NY, USA, 1982. [Google Scholar]
- Chiou, W.L. Critical evaluation of the potential error in pharmacokinetic studies of using the linear trapezoidal rule method for the calculation of the area under the plasma level-time curve. J. Pharmacokinet. Biopharm. 1978, 6, 539–546. [Google Scholar] [CrossRef]
- Bae, S.H.; Chang, S.Y.; Kim, S.H. Slower elimination of tofacitinib in acute renal failure rat Models: Contribution of hepatic metabolism and renal excretion. Pharmaceutics 2020, 12, 714. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Hussain, A.; Leal, M.; Mayersohn, M.; Fluhler, E. Interspecies prediction of human drug clearance based on scaling data from one or two animal species. Drug Metab. Dispos. 2007, 35, 1886–1893. [Google Scholar] [CrossRef]
- Paine, S.W.; Ménochet, K.; Denton, R.; McGinnity, D.F.; Riley, R.J. Prediction of human renal clearance from preclinical species for a diverse set of drugs that exhibit both active secretion and net reabsorption. Drug Metab. Dispos. 2011, 39, 1008–1013. [Google Scholar] [CrossRef]
- Edginton, A.N.; Schmitt, W.; Willmann, S. Development and evaluation of a generic physiologically based pharmacokinetic model for children. Clin. Pharmacokinet. 2006, 45, 1013–1034. [Google Scholar] [CrossRef]
- Malik, P.R.V.; Yeung, C.H.T.; Ismaeil, S.; Advani, U.; Djie, S.; Edginton, A.N. A physiological approach to pharmacokinetics in chronic kidney disease. J. Clin. Pharmacol. 2020, 60 (Suppl. 1), S52–S62. [Google Scholar] [CrossRef]
- Klotman, P.E.; Yarger, W.E. Reduction of renal blood flow and proximal bicarbonate reabsorption in rats by gentamicin. Kidney Int. 1983, 24, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, H.; Yonemura, K.; Ohishi, K.; Hishida, A. The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J. Lab. Clin. Med. 1998, 131, 518–526. [Google Scholar] [CrossRef]
- Hiraoka, H.; Yamamoto, K.; Miyoshi, S.; Morita, T.; Nakamura, K.; Kadoi, Y.; Kunimoto, F.; Horiuchi, R. Kidneys contribute to the extrahepatic clearance of propofol in humans, but not lungs and brain. Br. J. Clin. Pharmacol. 2005, 60, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.; Lueshen, E.; Mošat’, A.; Linninger, A.A. Interspecies scaling in pharmacokinetics: A novel whole-body physiologically based modeling framework to discover drug biodistribution mechanisms in vivo. J. Pharm. Sci. 2012, 101, 1221–1241. [Google Scholar] [CrossRef] [PubMed]
- Tse, S.; Dowty, M.E.; Menon, S.; Gupta, P.; Krishnaswami, S. Application of physiologically based pharmacokinetic modeling to predict drug exposure and support dosing recommendations for potential drug-drug interactions or in special populations: An example using tofacitinib. J. Clin. Pharmacol. 2020, 60, 1617–1628. [Google Scholar] [CrossRef]
- Kim, J.E.; Park, M.Y.; Kim, S.H. Simple determination and quantification of tofacitinib, a JAK inhibitor, in rat plasma, urine and tissue homogenates by HPLC and its application to a pharmacokinetic study. J. Pharm. Investig. 2020, 50, 603–612. [Google Scholar] [CrossRef]
- Eichner, A.; Wohlrab, J. Pharmacology of inhibitors of Janus kinases—Part 1: Pharmacokinetics. J. Dtsch. Dermatol. Ges. 2022, 20, 1485–1499. [Google Scholar] [CrossRef]
- Kathuria, H.; Handral, H.K.; Cha, S.; Nguyen, D.; Cai, J.; Cao, T.; Wu, C.; Kang, L. Enhancement of skin delivery of drugs using proposome depends on drug lipophilicity. Pharmaceutics 2021, 13, 1457. [Google Scholar] [CrossRef]
- Krishnaswami, S.; Chow, V.; Boy, M.; Wang, C.; Chan, G. Pharmacokinetics of tofacitinib, a janus kinase inhibitor, in patients with impaired renal function and end-stage renal disease. J. Clin. Pharmacol. 2014, 54, 46–52. [Google Scholar] [CrossRef]
- Australian Product Information for XELJANZ Tofacitinib (As Citrate). Available online: https://www.tga.gov.au/sites/default/files/auspar-tofacitinib-as-citrate-190910-pi.pdf (accessed on 4 March 2024).
- National Kidney Foundation. Available online: https://www.kidney.org/kidney-topics/estimated-glomerular-filtration-rate-egfr (accessed on 13 July 2022).
- Malnoë, D.; Fardel, O.; Le Corre, P. Involvement of transporters in intestinal drug-drug interactions of oral targeted anticancer drugs assessed by changes in drug absorption time. Pharmaceutics 2022, 14, 2493. [Google Scholar] [CrossRef]
- Rowland Yeo, K.; Aarabi, M.; Jamei, M.; Rostami-Hodjegan, A. Modeling and predicting drug pharmacokinetics in patients with renal impairment. Expert Rev. Clin. Pharmacol. 2011, 4, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Jacobsson, L. A method for the calculation of renal clearance based on a single plasma sample. Clin. Physiol. 1983, 3, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Poulin, P.; Kenny, J.R.; Hop, C.E.; Haddad, S. In vitro-in vivo extrapolation of clearance: Modeling hepatic metabolic clearance of highly bound drugs and comparative assessment with existing calculation methods. J. Pharm. Sci. 2012, 101, 838–851. [Google Scholar] [CrossRef]
- Wagner, C.; Zhao, P.; Pan, Y.; Hsu, V.; Grillo, J.; Huang, S.; Sinha, V. Application of physiologically based pharmacokinetic (PBPK) modeling to support dose selection: Report of an FDA public workshop on PBPK. CPT Pharmacomet. Syst. Pharmacol. 2015, 4, 226–230. [Google Scholar] [CrossRef]
- Liang, N.; Zhou, S.; Li, T.; Zhang, Z.; Zhao, T.; Li, R.; Li, M.; Shao, F.; Wang, G.; Sun, J. Physiologically based pharmacokinetic modeling to assess the drug-drug interactions of anaprazole with clarithromycin and amoxicillin in patients undergoing eradication therapy of H. pylori infection. Eur. J. Pharm. Sci. 2023, 189, 106534. [Google Scholar] [CrossRef] [PubMed]
- Marston, S.A.; Polli, J.E. Evaluation of direct curve comparison metrics applied to pharmacokinetic profiles and relative bioavailability and bioequivalence. Pharm. Res. 1997, 1410, 1363–1369. [Google Scholar] [CrossRef]
- Prieto Garcia, L.; Janzén, D.; Kanebratt, K.P.; Ericsson, H.; Lennernäs, H.; Lundahl, A. Physiologically based pharmacokinetic model of itraconazole and two of its metabolites to improve the predictions and the mechanistic understanding of CYP3A4 drug-drug interactions. Drug Metab. Dispos. 2018, 46, 1420–1433. [Google Scholar] [CrossRef]
- Li, X.; Chen, C.; Ding, N.; Zhang, T.; Zheng, P.; Yang, M. Physiologically based pharmacokinetic modelling and simulation to predict the plasma concentration profile of schaftoside after oral administration of total flavonoids of Desmodium styracifolium. Front. Pharmacol. 2022, 13, 1073535. [Google Scholar] [CrossRef]
- Edginton, A.N.; Willmann, S. Physiology-based simulations of a pathological condition: Prediction of pharmacokinetics in patients with liver cirrhosis. Clin. Pharmacokinet. 2008, 47, 743–752. [Google Scholar] [CrossRef]
- Lee, B.I.; Park, M.H.; Shin, S.H.; Byeon, J.J.; Park, Y.; Kim, N.; Choi, J.; Shin, Y.G. Quantitative analysis of tozadenant using liquid chromatography-mass spectrometric method in rat plasma and its human pharmacokinetics prediction using physiologically based pharmacokinetic modeling. Molecules 2019, 24, 1295. [Google Scholar] [CrossRef]
- Gao, Z.W.; Zhu, Y.T.; Yu, M.M.; Zan, B.; Liu, J.; Zhang, Y.F.; Chen, X.Y.; Li, X.N.; Zhong, D.F. Preclinical pharmacokinetics of TPN729MA, a novel PDE5 inhibitor, and prediction of its human pharmacokinetics using a PBPK model. Acta Pharmacol. Sin. 2015, 36, 1528–1536. [Google Scholar] [CrossRef]
- Jamei, M. Recent advances in development and application of physiologically based pharmacokinetic (PBPK) Models: A transition from academic curiosity to regulatory acceptance. Curr. Pharmacol. Rep. 2016, 2, 161–169. [Google Scholar] [CrossRef]
- Fleischmann, R.; Mease, P.J.; Schwartzman, S.; Hwang, L.J.; Soma, K.; Connell, C.A.; Takiya, L.; Bananis, E. Efficacy of tofacitinib in patients with rheumatoid arthritis stratified by background methotrexate dose group. Clin. Rheumatol. 2017, 36, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Craig, E.; Cappelli, L.C. Gastrointestinal and hepatic disease in rheumatoid arthritis. Rheum. Dis. Clin. N. Am. 2018, 44, 89–111. [Google Scholar] [CrossRef] [PubMed]
- Suh, S.H.; Jung, J.H.; Oh, T.R.; Yang, E.M.; Choi, H.S.; Kim, C.S.; Bae, E.H.; Ma, S.K.; Han, K.D.; Kim, S.W. Rheumatoid arthritis and the risk of end-stage renal disease: A nationwide, population-based study. Front. Med. 2023, 10, 1116489. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Yang, J.; Zhang, Y.; Wang, Y.; Zhang, J.; Zhao, Y.; Dong, W. Prediction of pharmacokinetics and penetration of moxifloxacin in uman with Intra-Abdominal Infection Based on Extrapolated PBPK Model. Korean J. Physiol. Pharmacol. 2015, 19, 99–104. [Google Scholar] [CrossRef][Green Version]
- Romero, F.; Pérez, M.; Chávez, M.; Parra, G.; Durante, P. Effect of uric acid on gentamicin-induced nephrotoxicity in rats—Role of matrix metalloproteinases 2 and 9. Basic Clin. Pharmacol. Toxicol. 2009, 105, 416–424. [Google Scholar] [CrossRef]
- Abd El-Rhman, R.H.; El-Naga, R.N.; Gad, A.M.; Tadros, M.G.; Hassaneen, S.K. Dibenzazepine attenuates against cisplatin-induced nephrotoxicity in rats: Involvement of NOTCH pathway. Front. Pharmacol. 2020, 11, 567852. [Google Scholar] [CrossRef]
- Lee, Y.K.; Chin, Y.W.; Choi, Y.H. Effects of Korean red ginseng extract on acute renal failure induced by gentamicin and pharmacokinetic changes by metformin in rats. Food Chem. Toxicol. 2013, 59, 153–159. [Google Scholar] [CrossRef]
- Ngo, L.T.; Yun, H.Y.; Chae, J.W. Application of the population pharmacokinetics model-based approach to the prediction of drug-drug interaction between rivaroxaban and carbamazepine in humans. Pharmaceuticals 2023, 16, 684. [Google Scholar] [CrossRef]
- Sharma, V.; McNeill, J.H. To scale or not to scale: The principles of dose extrapolation. Br. J. Pharmacol. 2009, 157, 907–921. [Google Scholar] [CrossRef]
- Wajima, T.; Yano, Y.; Fukumura, K.; Oguma, T. Prediction of human pharmacokinetic profile in animal scale up based on normalizing time course profiles. J. Pharm. Sci. 2004, 93, 1890–1900. [Google Scholar] [CrossRef]
- Naritomi, Y.; Sanoh, S.; Ohta, S. Utility of chimeric mice with humanized liver for predicting human pharmacokinetics in drug discovery: Comparison with in vitro-in vivo extrapolation and allometric scaling. Biol. Pharm. Bull. 2019, 42, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Fairman, K.; Li, M.; Kabadi, S.V.; Lumen, A. Physiologically based pharmacokinetic modeling: A promising tool for translational research and regulatory toxicology. Curr. Opin. Toxicol. 2020, 23, 17–22. [Google Scholar] [CrossRef]
- Mahmood, I.; Balian, J.D. Interspecies scaling: A comparative study for the prediction of clearance and volume using two or more than two species. Life Sci. 1996, 59, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.W.; Lee, Y.B.; Cho, H.Y. Interpretation of non-clinical data for prediction of human pharmacokinetic parameters: In vitro-in vivo extrapolation and allometric scaling. Pharmaceutics 2019, 11, 168. [Google Scholar] [CrossRef]
- De Buck, S.S.; Sinha, V.K.; Fenu, L.A.; Nijsen, M.J.; Mackie, C.E.; Gilissen, R.A. Prediction of human pharmacokinetics using physiologically based modeling: A retrospective analysis of 26 clinically tested drugs. Drug Metab. Dispos. 2007, 35, 1766–1780. [Google Scholar] [CrossRef]
- Dreisbach, A.W. The influence of chronic renal failure on drug metabolism and transport. Clin. Pharmacol. Ther. 2009, 86, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Déri, M.T.; Kiss, Á.F.; Tóth, K.; Paulik, J.; Sárváry, E.; Kóbori, L.; Monostory, K. End-stage renal disease reduces the expression of drug-metabolizing cytochrome P450s. Pharmacol. Rep. 2020, 72, 1695–1705. [Google Scholar] [CrossRef]
- Nolin, T.D.; Frye, R.F.; Matzke, G.R. Hepatic drug metabolism and transport in patients with kidney disease. Am. J. Kidney Dis. 2003, 42, 906–925. [Google Scholar] [CrossRef]
- Shin, W.; Yang, A.Y.; Yun, H.; Cho, D.Y.; Park, K.H.; Shin, H.; Kim, A. Bioequivalence of the pharmacokinetics between tofacitinib aspartate and tofacitinib citrate in healthy subjects. Transl. Clin. Pharmacol. 2020, 28, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Wong, H. Predicting oral drug absorption: Mini review on physiologically-based pharmacokinetic models. Pharmaceutics 2017, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, S.; Takai, K. Deep-sea vent chemoautotrophs: Diversity, biochemistry and ecological significance. FEMS Microbiol. Ecol. 2008, 65, 1–14. [Google Scholar] [CrossRef]
- Riedmaier, A.E.; DeMent, K.; Huckle, J.; Bransford, P.; Stillhart, C.; Lloyd, R.; Alluri, R.; Basu, S.; Chen, Y.; Dhamankar, V.; et al. Use of physiologically based pharmacokinetic (PBPK) modeling for predicting drug-food interactions: An industry perspective. AAPS J. 2020, 22, 123. [Google Scholar] [CrossRef] [PubMed]

| Normal (n = 6) | Moderate (n = 8) | Severe (n = 7) | |
|---|---|---|---|
| Body weight (g) | 280 ± 19.0 | 251 ± 21.3 | 188 ± 10.2 |
| AUC (μg·min/mL) | 264 ± 45.4 | 433 ± 90.0 | 693 ± 105 |
| CL (mL/min/kg) | 39.0 ± 7.97 | 24.3 ± 6.95 | 14.7 ± 2.29 |
| CLR (mL/min/kg) | 4.75 ± 1.28 | 1.45 ± 1.54 | 00679 ± 0.0917 |
| Physicochemical Properties | Value | Reference |
|---|---|---|
| Molecular weight (g/mol) | 312.4 | [29] |
| Logp * | 1.15 | [30] |
| pKa | 5.07 | [32] |
| fu,p | 0.61 | [2] |
| Normal | Moderate | Severe | |
|---|---|---|---|
| Age (years) | 37–65 | 37–63 | 31–72 |
| Height (cm) | 165–193 | 160–175 | 155–175 |
| Weight (kg) | 65–87 | 65–116 | 74–109 |
| BMI (kg/m2) | 21–29 | 23–41 | 27–40 |
| Parameters | Observed | Dedrick Plot | PK-Sim | Simcyp | |
|---|---|---|---|---|---|
| Normal | Cmax (ng/mL) | 94.2 ± 25.3 | 87.3 ± 30.4 | 117 ± 25.4 | 104 ± 18.9 |
| AUC (ng∙h/mL) | 268 ± 71.5 | 240 ± 26.5 | 347 ± 141 | 312 ± 78.8 | |
| Tmax (h) | 0.75 (0.50–1.50) | 1.39 (0.112–4.08) | 0.600 (0.50–0.95) | 0.937 (0.866–1.16) | |
| Moderate | Cmax (ng/mL) | 104 ± 47.5 | 254 ± 136 * | 119 ± 53.4 | 115 ± 39.3 |
| AUC (ng∙h/mL) | 396 ± 154 | 535 ± 269 | 397 ± 97.9 | 512 ± 203 | |
| Tmax (h) | 0.75 (0.50–2.00) | 0.372 (0.121–1.13) | 0.800 (0.450–1.15) | 1.16 (1.04–1.39) | |
| Severe | Cmax (ng/mL) | 111 ± 28.6 | 210 ± 152 | 98.6 ± 23.1 | 139 ± 48.0 |
| AUC (ng∙h/mL) | 615 ± 214 | 975 ± 551 | 608 ± 161 | 826 ± 344 | |
| Tmax (h) | 0.75 (0.50–1.50) | 0.777 (0.378–3.12) | 0.600 (1.08–1.30) | 1.33 (1.20–1.44) |
| f1 Value (%) | Normal | Moderate | Severe |
|---|---|---|---|
| Dedrick plot | 37.3 | 101 | 68.5 |
| PK-Sim | 51.9 | 29.3 | 6.02 |
| Simcyp | 51.5 | 41.1 | 34.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bae, S.H.; Park, S.Y.; Choi, H.G.; Kim, S.H. Physiologically Based Pharmacokinetic Simulation of Tofacitinib in Humans Using Extrapolation from Single-Species Renal Failure Model. Pharmaceutics 2025, 17, 914. https://doi.org/10.3390/pharmaceutics17070914
Bae SH, Park SY, Choi HG, Kim SH. Physiologically Based Pharmacokinetic Simulation of Tofacitinib in Humans Using Extrapolation from Single-Species Renal Failure Model. Pharmaceutics. 2025; 17(7):914. https://doi.org/10.3390/pharmaceutics17070914
Chicago/Turabian StyleBae, Sung Hun, So Yeon Park, Hyeon Gyeom Choi, and So Hee Kim. 2025. "Physiologically Based Pharmacokinetic Simulation of Tofacitinib in Humans Using Extrapolation from Single-Species Renal Failure Model" Pharmaceutics 17, no. 7: 914. https://doi.org/10.3390/pharmaceutics17070914
APA StyleBae, S. H., Park, S. Y., Choi, H. G., & Kim, S. H. (2025). Physiologically Based Pharmacokinetic Simulation of Tofacitinib in Humans Using Extrapolation from Single-Species Renal Failure Model. Pharmaceutics, 17(7), 914. https://doi.org/10.3390/pharmaceutics17070914






