Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
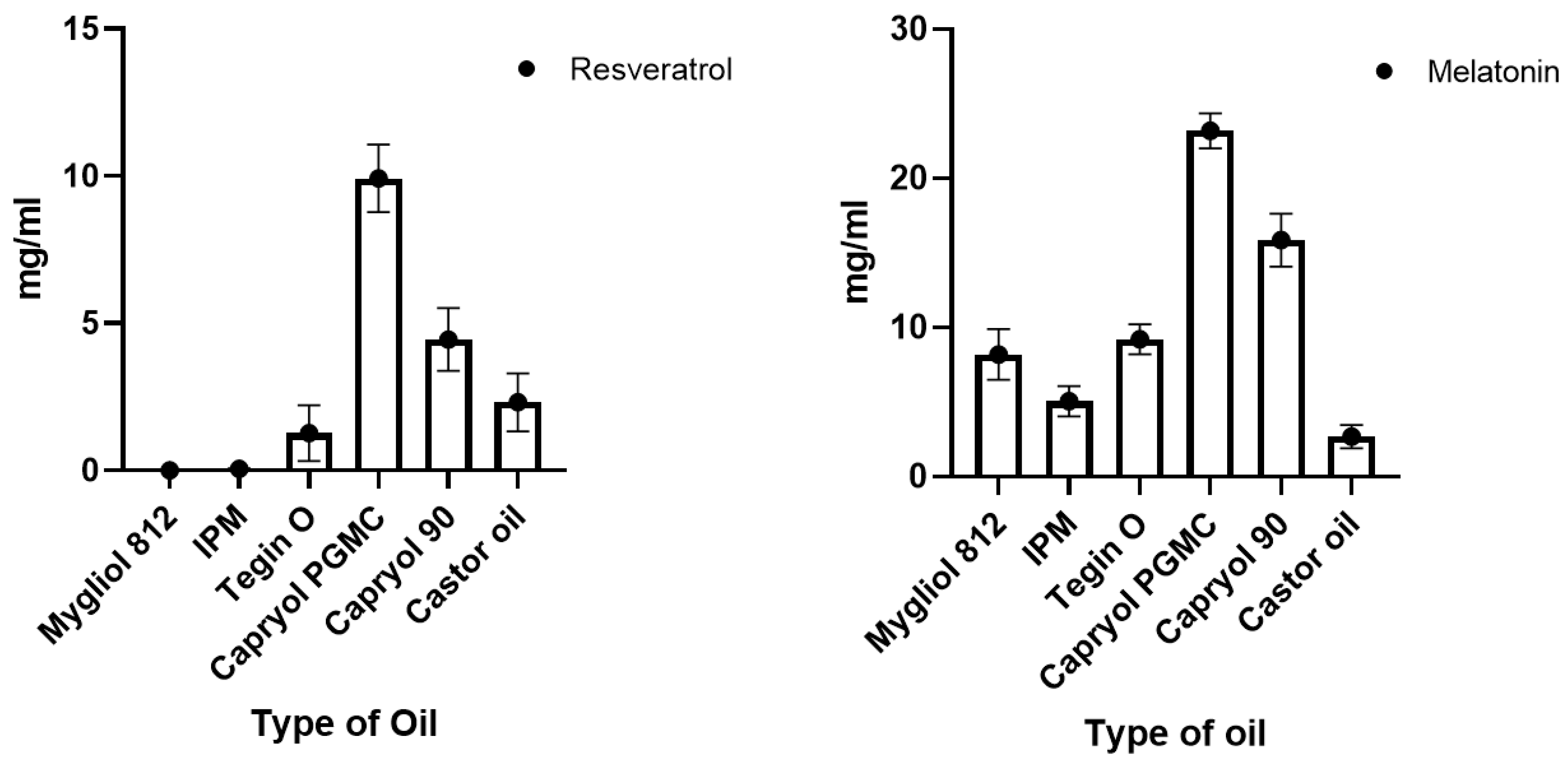
2.2. Solubility in Different Oil
2.3. Development of SNEDDSs Employing the QbD Approach
2.3.1. Ternary Phase Construction: Choice of Oil
2.3.2. Construction of the Design Space
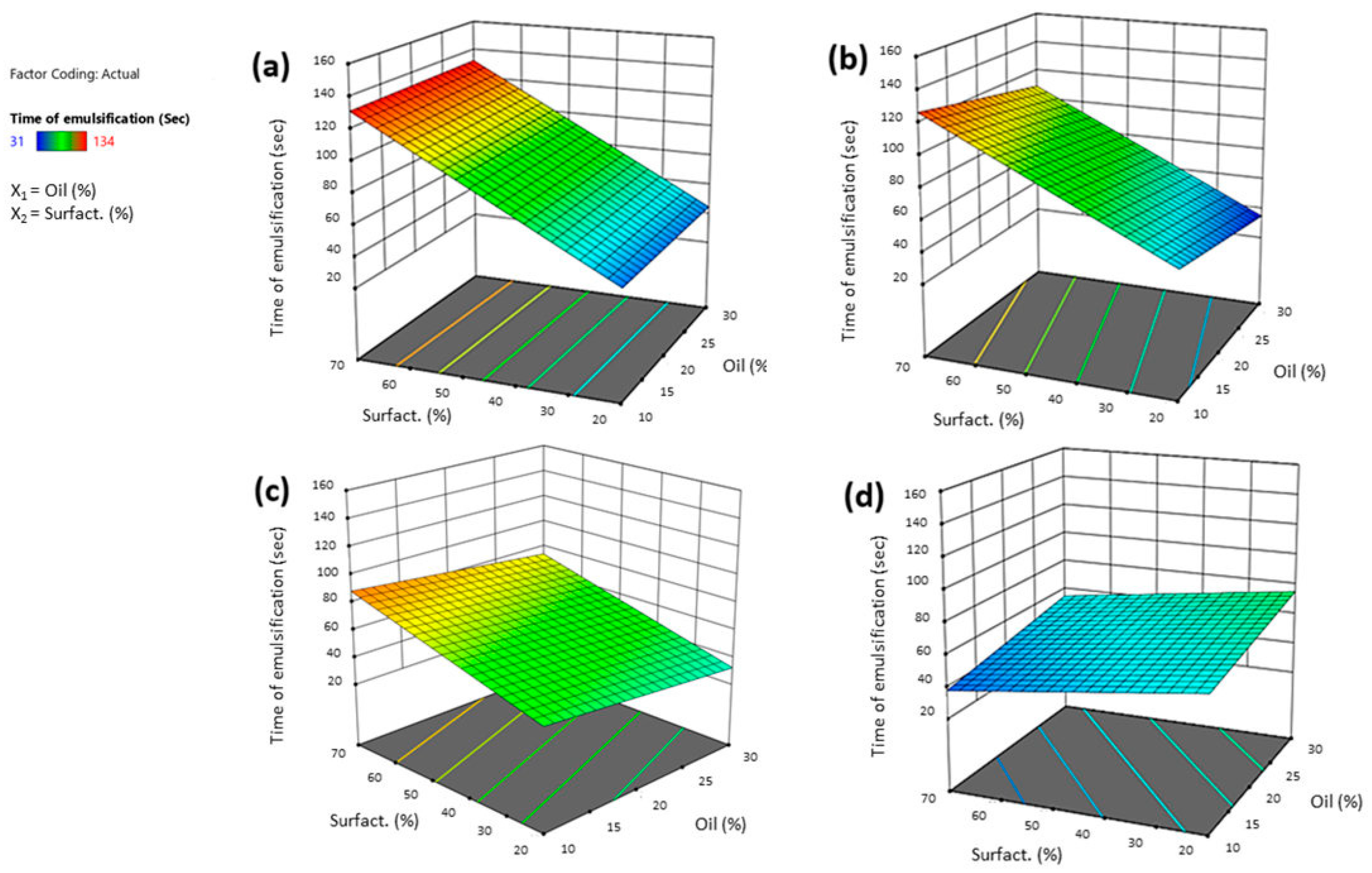
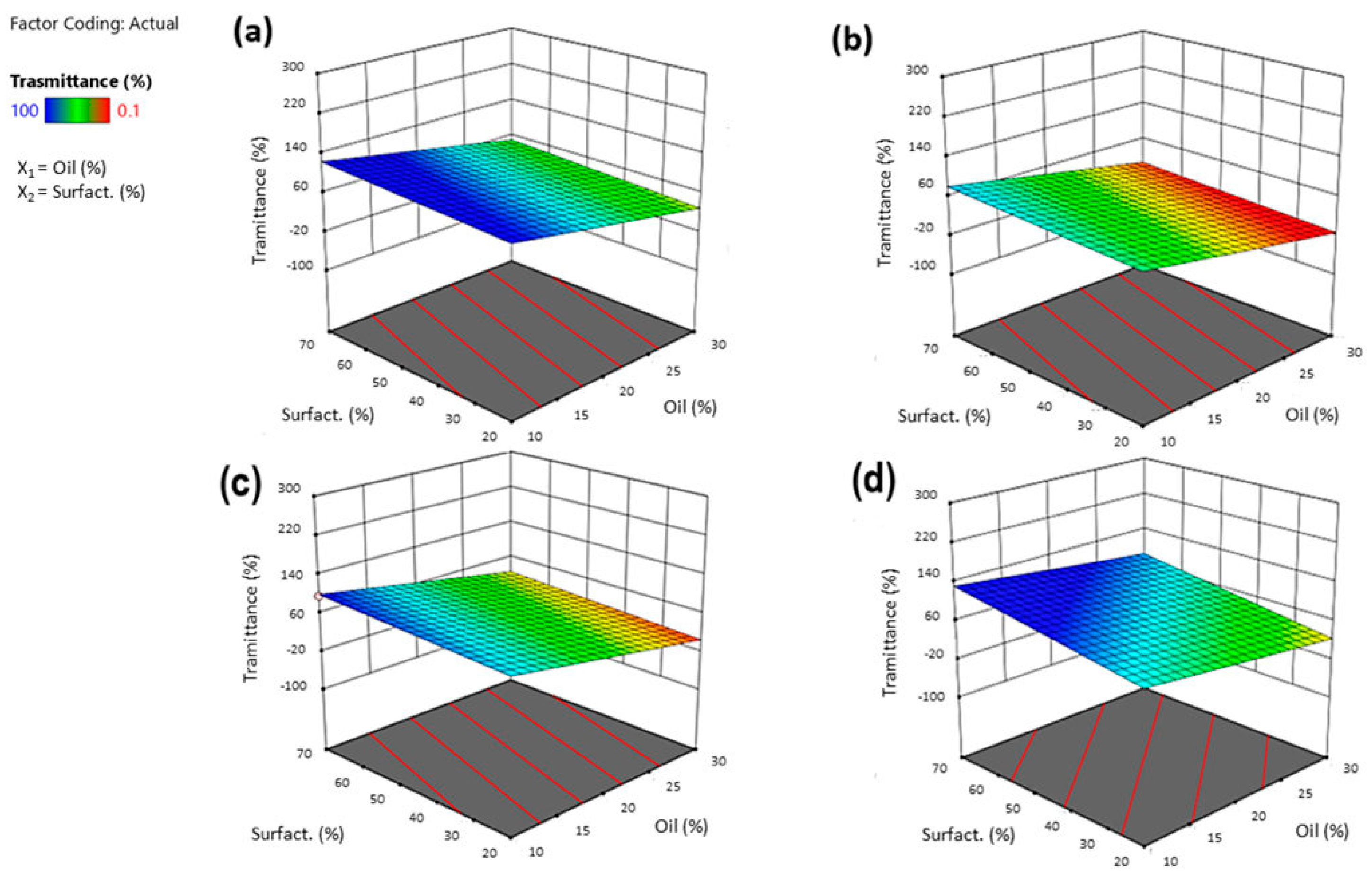
2.3.3. Characterization of SNEDDS (Particle Size, Time of Emulsification and % Transmittance)
2.3.4. Optimization of SNEDDS
2.4. Preparation of RSV-SNEDDS and MEL-SNEDDS
2.5. Characterization of Optimized SNEDDS
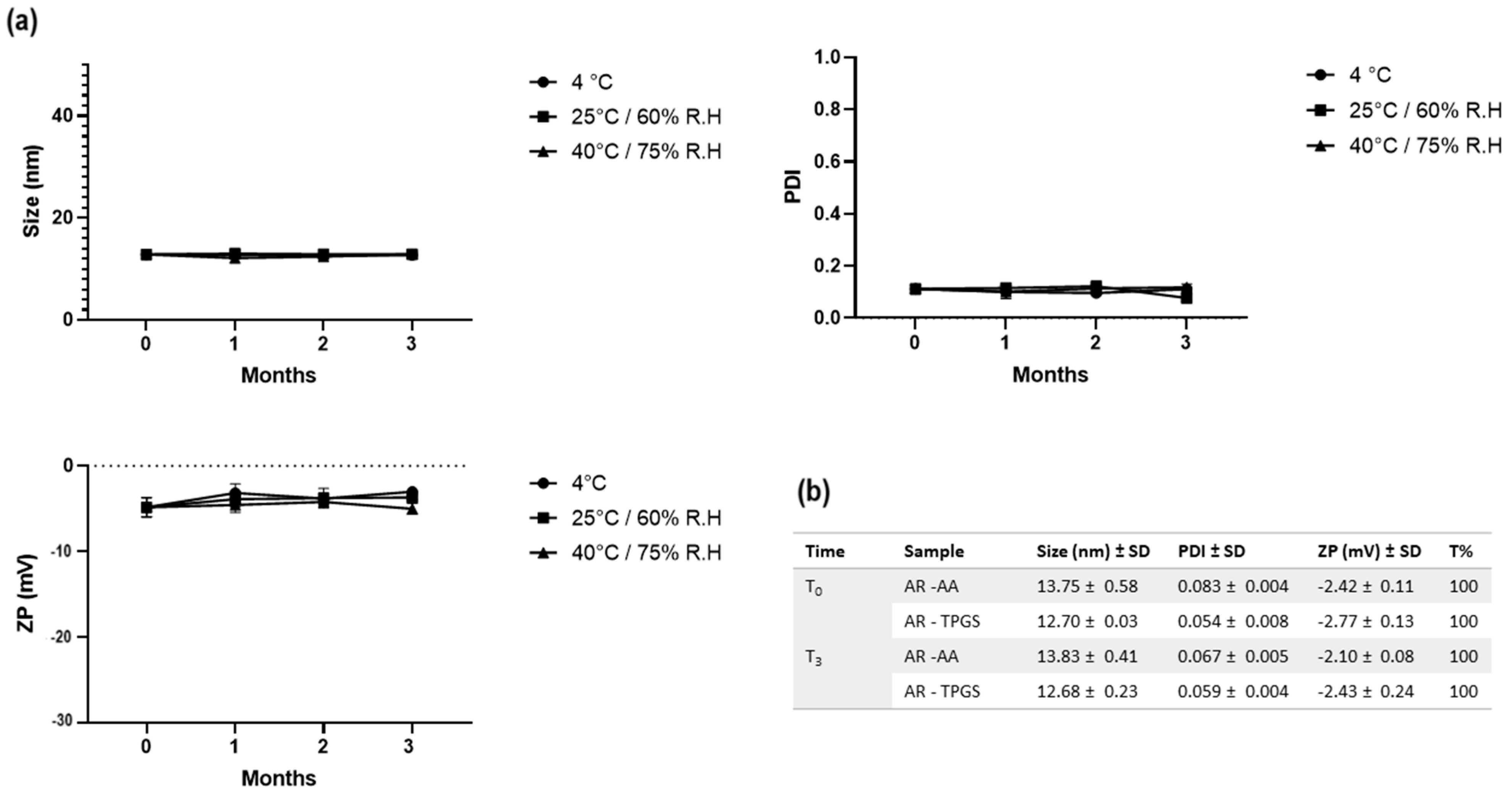
2.6. Stability Evaluation
2.7. Stability in Ocular Environment
2.8. pH, Osmolarity, and Viscosity Determination
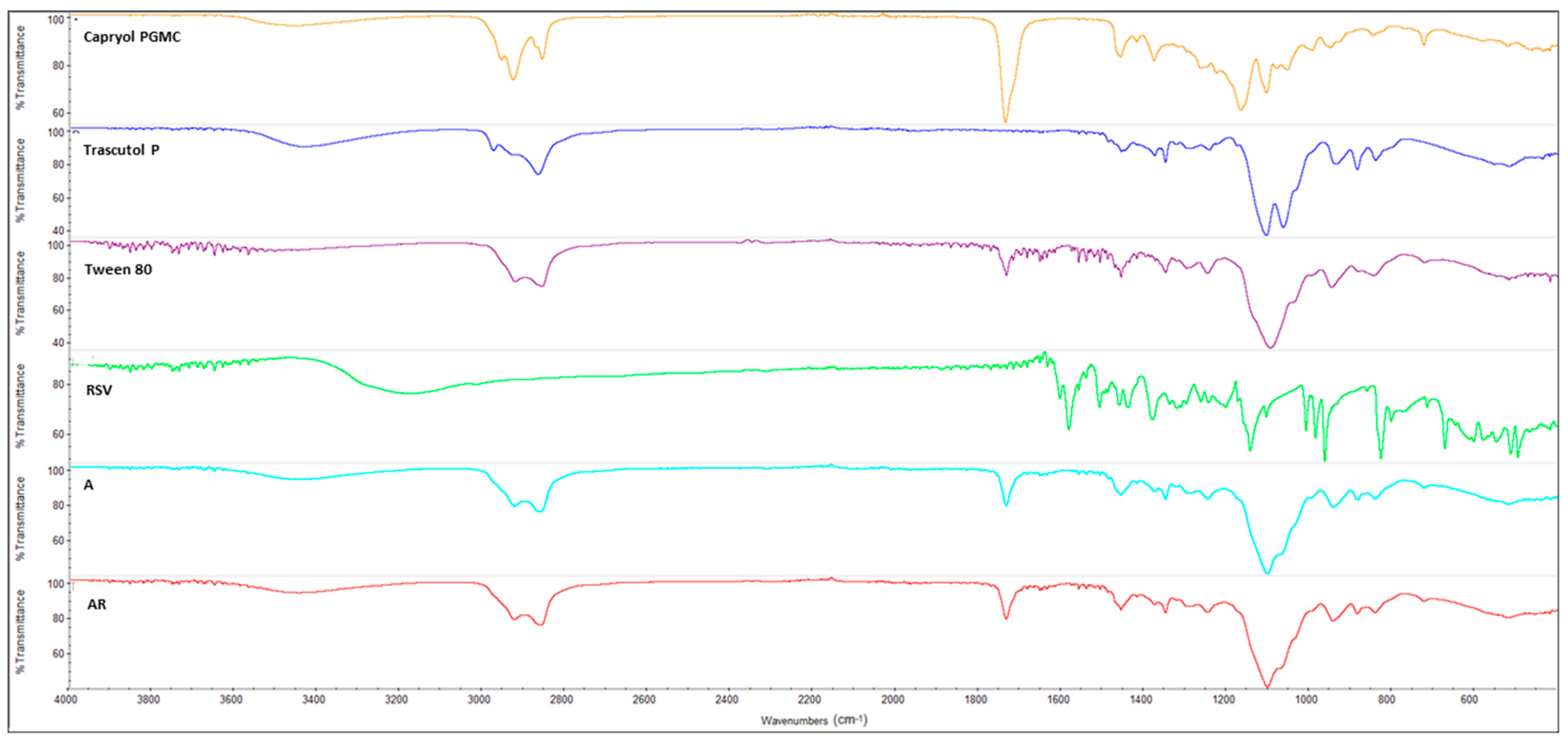
2.9. FT-IR Analysis
2.10. Mucoadhesion Study
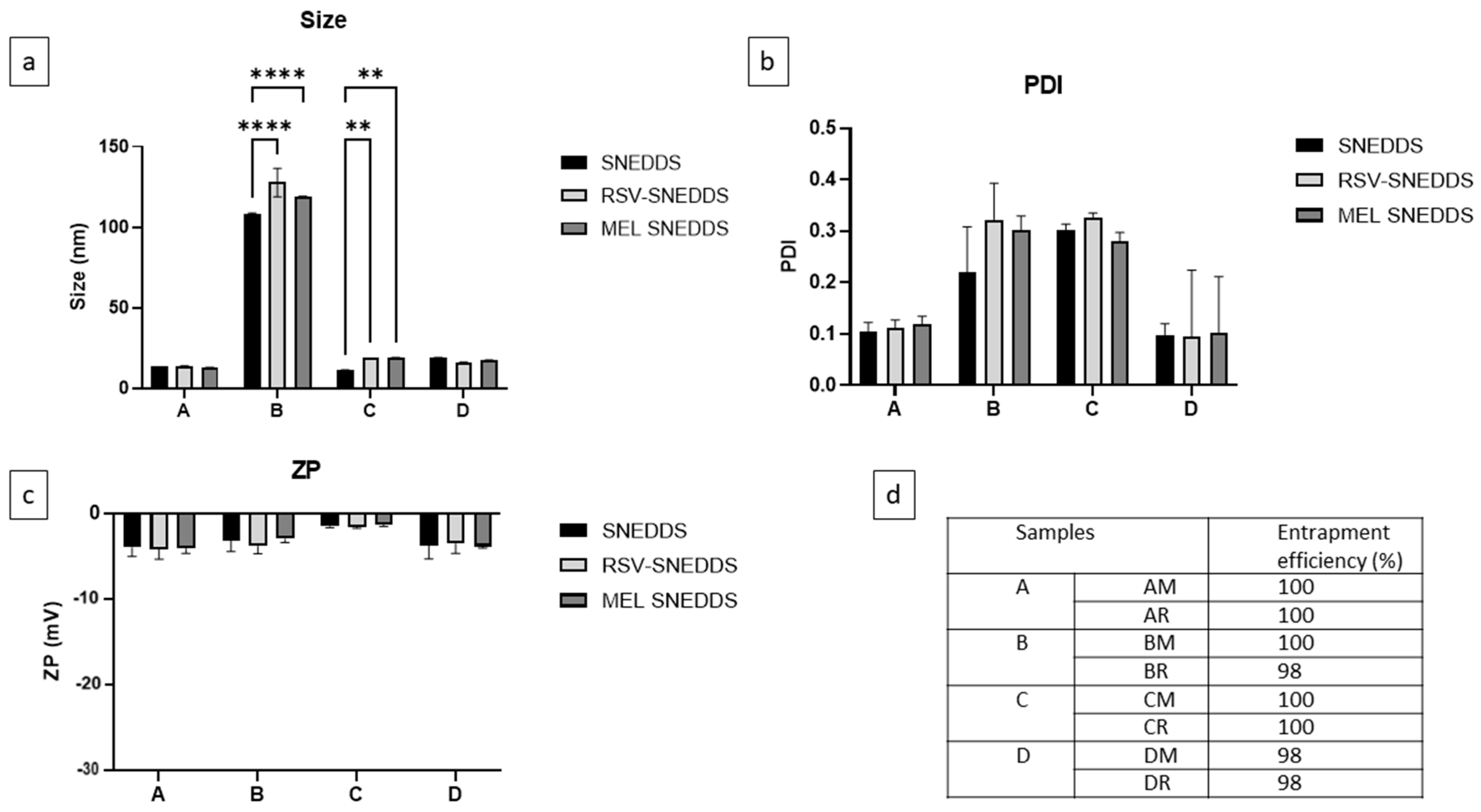
2.11. Drug Entrapment Efficiency (EE%)
2.12. Cloud Point Measurements
2.13. High-Performance Liquid Chromatography (HPLC) Method for the Quantification of MEL
2.14. High-Performance Liquid Chromatography (HPLC) Method for the Quantification of RSV
2.15. In Vitro Release Test
- Zero-order model: R = Kot
- First-order model: R = 1 − e − kt
- Higuchi model: R = KH t1/2
- Hixson–Crowell model: Wo1/3 − Wt1/3 = KHCt
- Korsmeyer–Peppas model: R = kKP tn
2.16. Cell Cultures and Viability Assay (MTT)
2.17. Statistical Analysis
3. Results and Discussion
3.1. Solubility of Drugs in Various Oils
3.2. Ternary Plot Diagram Construction
3.3. Construction of the Experimental Design
3.3.1. Effect of Independent Variables on Globule Size
3.3.2. Effect of Independent Variables on Time of Emulsification
3.3.3. Effect of Independent Variables on Transmittance%
3.4. Optimization Phase
3.5. Characterization of SNEDDS, RSV-SNEDDS, and MEL-SNEDDS
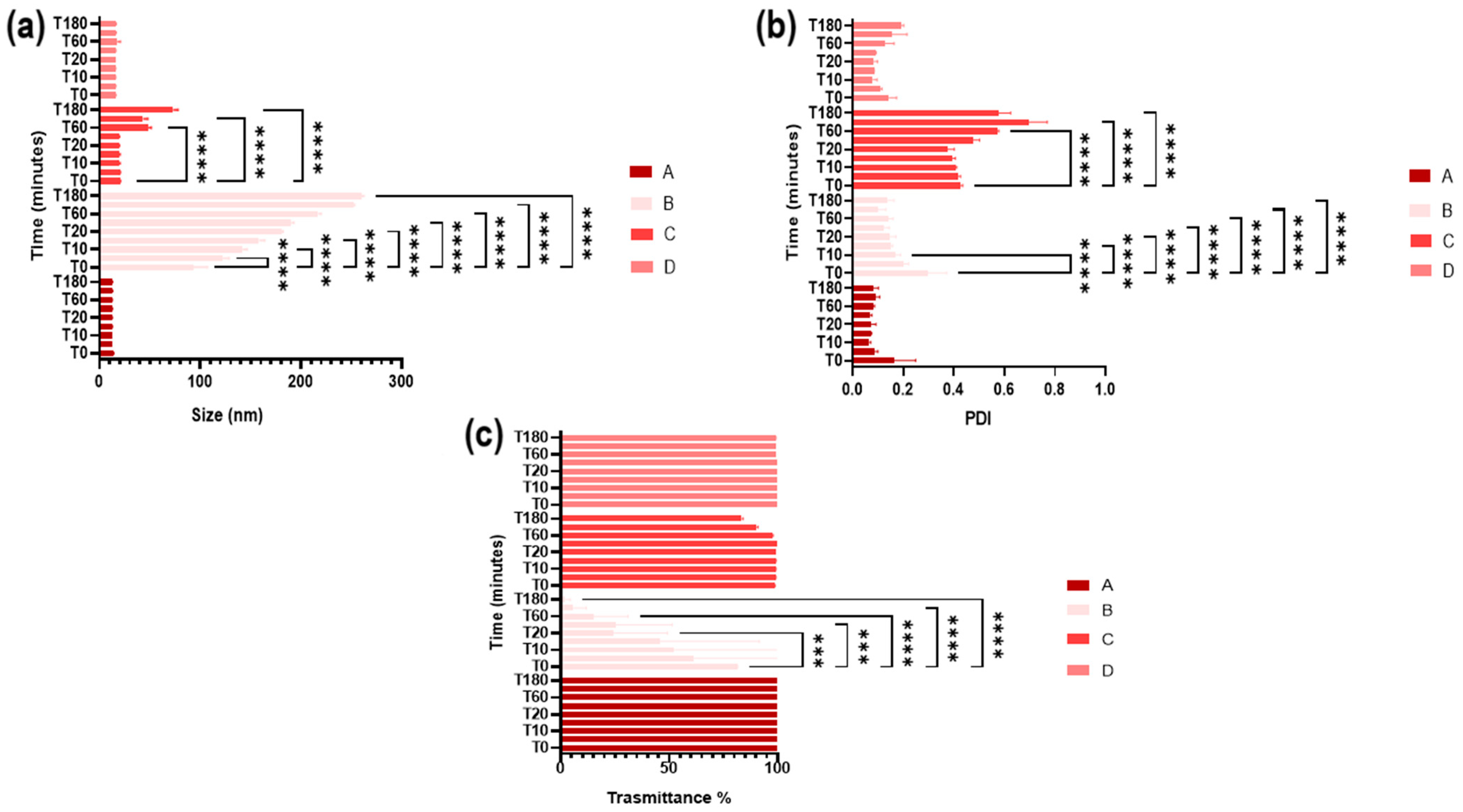
3.6. Stability in Simulated Ocular Environment
3.7. SNEDDS Stability Evaluation
Measurement of Cloud Point
3.8. FT-IR Spectroscopy
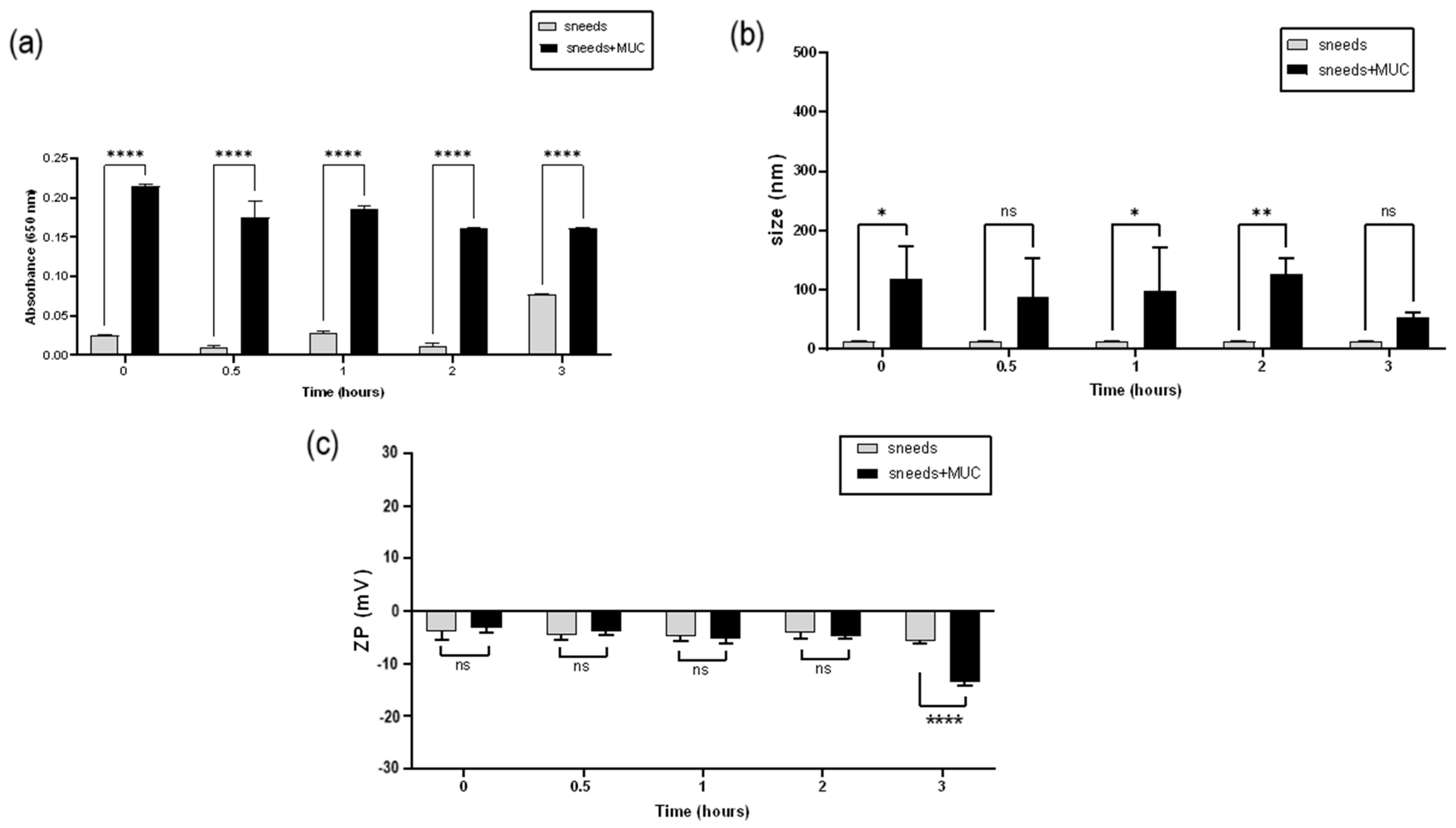
3.9. Mucoadhesion Study
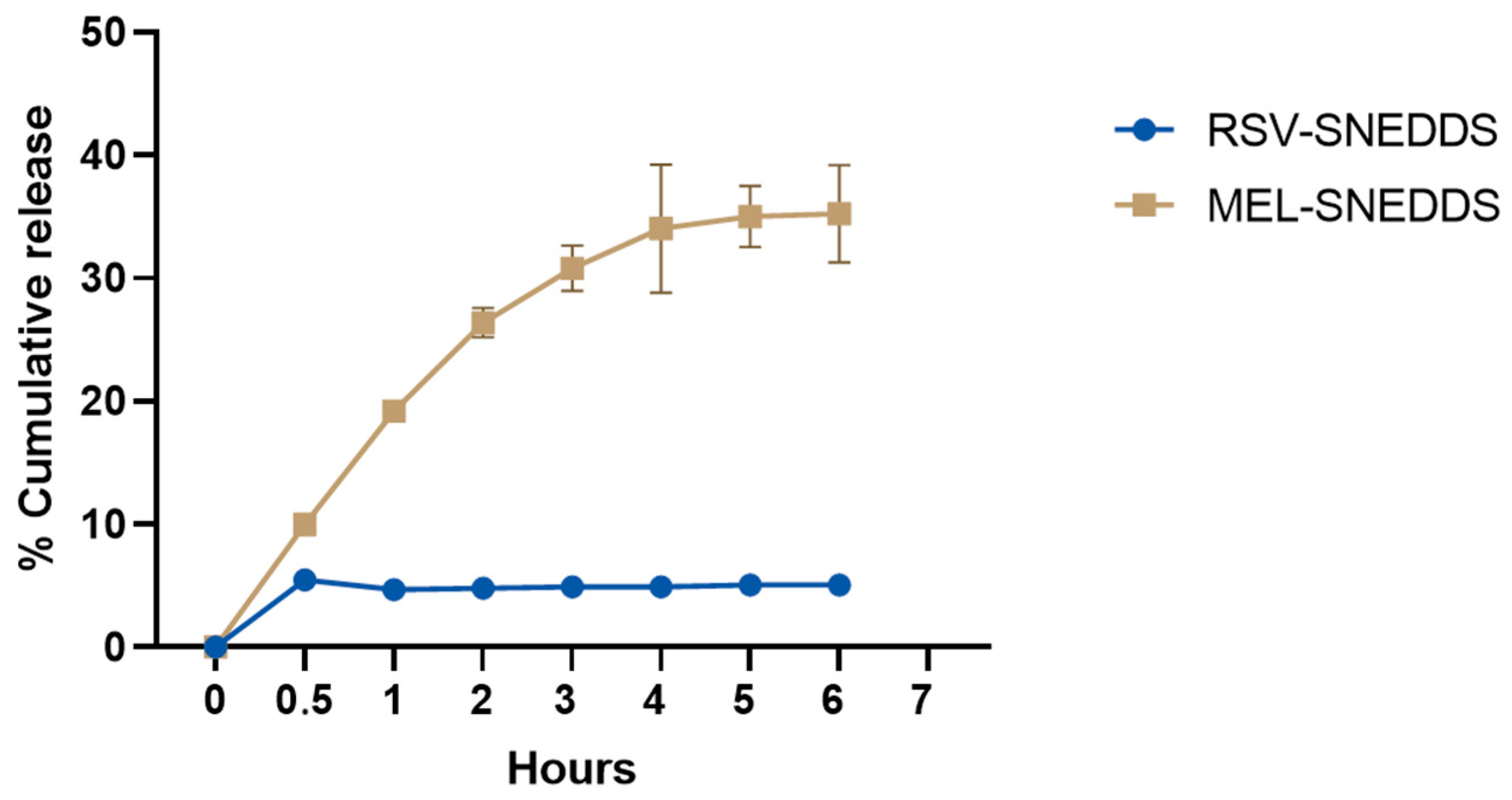
3.10. In Vitro Release in Simulated Ocular Environment
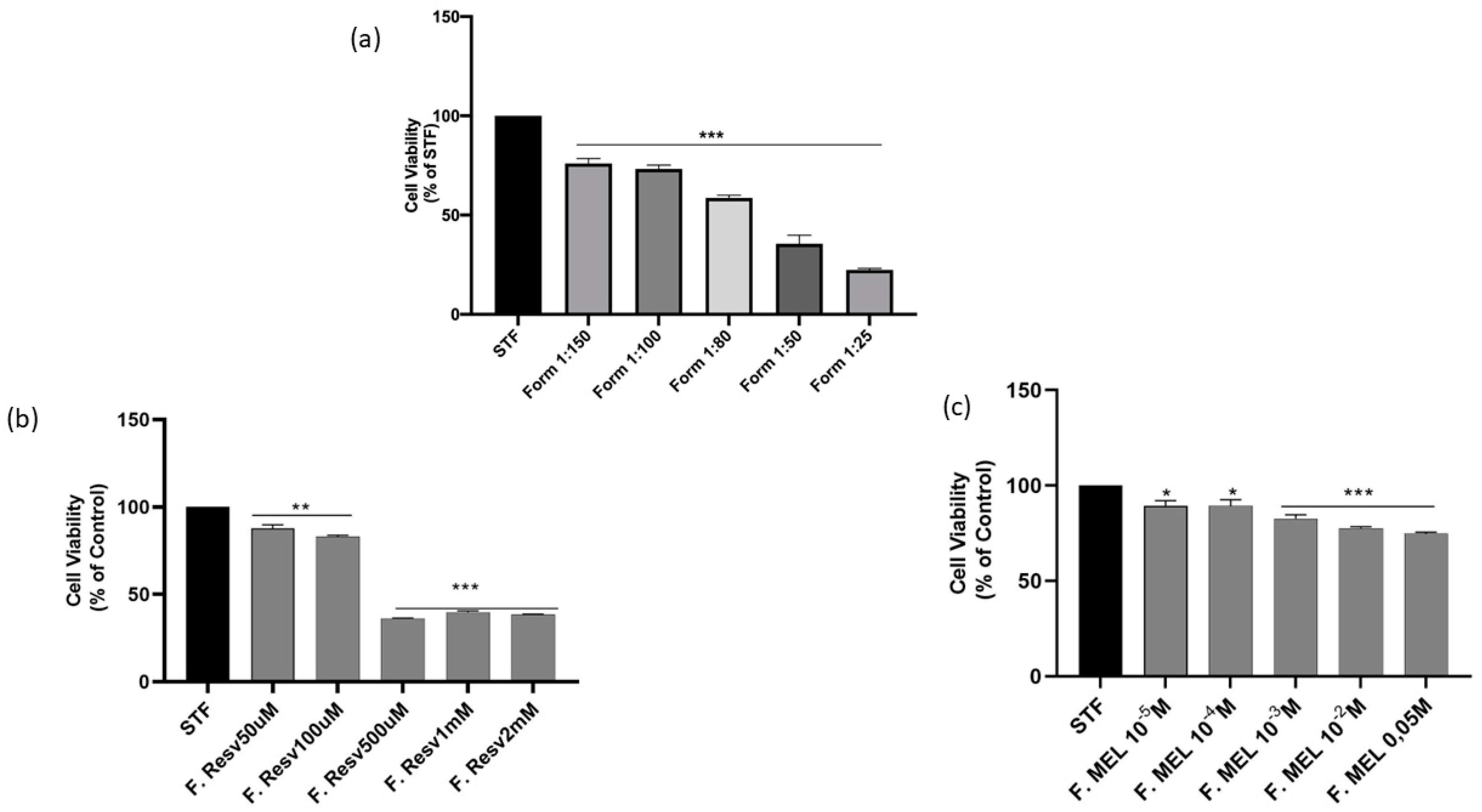
3.11. Short Time Exposure Test (STE)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, M.; Luo, J.; Zhang, H. Role of Sirtuin 1 in the Pathogenesis of Ocular Disease (Review). Int. J. Mol. Med. 2018, 42, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.-J.; Zhang, T.-N.; Chen, H.-H.; Yu, X.-F.; Lv, J.-L.; Liu, Y.-Y.; Liu, Y.-S.; Zheng, G.; Zhao, J.-Q.; Wei, Y.-F.; et al. The Sirtuin Family in Health and Disease. Signal Transduct. Target. Ther. 2022, 7, 402. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Zhou, Y.; Yang, Y.; Cai, L.; Xu, L.; Han, X.; Guo, Y.; Li, P.A. Activation of Sirtuin1 by Lyceum Barbarum Polysaccharides in Protection against Diabetic Cataract. J. Ethnopharmacol. 2020, 261, 113165. [Google Scholar] [CrossRef] [PubMed]
- Golestaneh, N.; Chu, Y.; Cheng, S.K.; Cao, H.; Poliakov, E.; Berinstein, D.M. Repressed SIRT1/PGC-1α Pathway and Mitochondrial Disintegration in iPSC-Derived RPE Disease Model of Age-Related Macular Degeneration. J. Transl. Med. 2016, 14, 344. [Google Scholar] [CrossRef]
- Yaman, D.; Takmaz, T.; Yüksel, N.; Dinçer, S.A.; Şahin, F.İ. Evaluation of Silent Information Regulator T (SIRT) 1 and Forkhead Box O (FOXO) Transcription Factor 1 and 3a Genes in Glaucoma. Mol. Biol. Rep. 2020, 47, 9337–9344. [Google Scholar] [CrossRef]
- Mimura, T.; Kaji, Y.; Noma, H.; Funatsu, H.; Okamoto, S. The Role of SIRT1 in Ocular Aging. Exp. Eye Res. 2013, 116, 17–26. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; McCarty, M.F.; O’Keefe, J.H. Nutraceutical Activation of Sirt1: A Review. Open Heart 2022, 9, e002171. [Google Scholar] [CrossRef] [PubMed]
- Pop, R.; Daescu, A.; Rugina, D.; Pintea, A. Resveratrol: Its Path from Isolation to Therapeutic Action in Eye Diseases. Antioxidants 2022, 11, 2447. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, J.; Zhan, L.; Wang, M.; Shi, R.; Yuan, X.; Gao, X.; Liu, X.; Zang, J.; Liu, W.; et al. Resveratrol-Induced Sirt1 Phosphorylation by LKB1 Mediates Mitochondrial Metabolism. J. Biol. Chem. 2021, 297, 100929. [Google Scholar] [CrossRef]
- Wu, Y.; Pang, Y.; Wei, W.; Shao, A.; Deng, C.; Li, X.; Chang, H.; Hu, P.; Liu, X.; Zhang, X. Resveratrol Protects Retinal Ganglion Cell Axons through Regulation of the SIRT1-JNK Pathway. Exp. Eye Res. 2020, 200, 108249. [Google Scholar] [CrossRef]
- Zhang, H.; He, S.; Spee, C.; Ishikawa, K.; Hinton, D.R. SIRT1 Mediated Inhibition of VEGF/VEGFR2 Signaling by Resveratrol and Its Relevance to Choroidal Neovascularization. Cytokine 2015, 76, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Haldar, C.; Verma, R. Melatonin Ameliorates LPS-Induced Testicular Nitro-Oxidative Stress (iNOS/TNFα) and Inflammation (NF-kB/COX-2) via Modulation of SIRT-1. Reprod. Sci. 2021, 28, 3417–3430. [Google Scholar] [CrossRef]
- Pal, S.; Haldar, C.; Verma, R. Melatonin Attenuates LPS-Induced Ovarian Toxicity via Modulation of SIRT-1, PI3K/pAkt, pErk1/2 and NFĸB/COX-2 Expressions. Toxicol. Appl. Pharmacol. 2022, 451, 116173. [Google Scholar] [CrossRef] [PubMed]
- Savran, M.; Asci, H.; Ozmen, O.; Erzurumlu, Y.; Savas, H.; Sonmez, Y.; Sahin, Y. Melatonin Protects the Heart and Endothelium against High Fructose Corn Syrup Consumption–Induced Cardiovascular Toxicity via SIRT-1 Signaling. Hum. Exp. Toxicol. 2019, 38, 1212–1223. [Google Scholar] [CrossRef] [PubMed]
- Carloni, S.; Albertini, M.C.; Galluzzi, L.; Buonocore, G.; Proietti, F.; Balduini, W. Melatonin Reduces Endoplasmic Reticulum Stress and Preserves Sirtuin 1 Expression in Neuronal Cells of Newborn Rats after Hypoxia–Ischemia. J. Pineal Res. 2014, 57, 192–199. [Google Scholar] [CrossRef]
- Atacak, A.; Baltaci, S.B.; Akgun-Unal, N.; Mogulkoc, R.; Baltaci, A.K. Melatonin Protects Retinal Tissue Damage in Streptozotocin-Induced Aged Rats. Arch. Gerontol. Geriatr. 2023, 112, 105035. [Google Scholar] [CrossRef]
- Rehman, F.U.; Shah, K.U.; Shah, S.U.; Khan, I.U.; Khan, G.M.; Khan, A. From Nanoemulsions to Self-Nanoemulsions, with Recent Advances in Self-Nanoemulsifying Drug Delivery Systems (SNEDDS). Expert. Opin. Drug Deliv. 2017, 14, 1325–1340. [Google Scholar] [CrossRef]
- Rasoanirina, B.N.V.; Lassoued, M.A.; Miladi, K.; Razafindrakoto, Z.; Chaâbane-Banaoues, R.; Ramanitrahasimbola, D.; Cornet, M.; Sfar, S. Self-Nanoemulsifying Drug Delivery System to Improve Transcorneal Permeability of Voriconazole: In-Vivo Studies. J. Pharm. Pharmacol. 2020, 72, 889–896. [Google Scholar] [CrossRef]
- Singh, G. Resveratrol: Nanocarrier-based delivery systems to enhance its therapeutic potential. Nanomedicine 2020, 15, 28. [Google Scholar] [CrossRef]
- ICH Q1A (R2) Guideline: ‘Stability Testing of New Drug Substances and Products’. Available online: https://ich.org/page/quality-guidelines (accessed on 31 December 2023).
- Singh, G.; Pai, R.S. Trans-Resveratrol Self-Nano-Emulsifying Drug Delivery System (SNEDDS) with Enhanced Bioavailability Potential: Optimization, Pharmacokinetics and in Situ Single Pass Intestinal Perfusion (SPIP) Studies. Drug Deliv. 2015, 22, 522–530. [Google Scholar] [CrossRef]
- Lombardo, R.; Ruponen, M.; Rautio, J.; Ghelardini, C.; Di Cesare Mannelli, L.; Calosi, L.; Bani, D.; Lampinen, R.; Kanninen, K.M.; Koivisto, A.M.; et al. Development of Lyophilised Eudragit® Retard Nanoparticles for the Sustained Release of Clozapine via Intranasal Administration. Pharmaceutics 2023, 15, 1554. [Google Scholar] [CrossRef]
- Takahashi, Y.; Koike, M.; Honda, H.; Ito, Y.; Sakaguchi, H.; Suzuki, H.; Nishiyama, N. Development of the Short Time Exposure (STE) Test: An in Vitro Eye Irritation Test Using SIRC Cells. Toxicol. Vitr. 2008, 22, 760–770. [Google Scholar] [CrossRef]
- Takahashi, Y.; Hayashi, K.; Abo, T.; Koike, M.; Sakaguchi, H.; Nishiyama, N. The Short Time Exposure (STE) Test for Predicting Eye Irritation Potential: Intra-Laboratory Reproducibility and Correspondence to Globally Harmonized System (GHS) and EU Eye Irritation Classification for 109 Chemicals. Toxicol. Vitr. 2011, 25, 1425–1434. [Google Scholar] [CrossRef]
- Maugeri, G.; D’Amico, A.G.; Giunta, S.; Giallongo, C.; Tibullo, D.; Bucolo, C.; Saccone, S.; Federico, C.; Scollo, D.; Longo, A.; et al. Activity-Dependent Neuroprotective Protein (ADNP)-Derived Peptide (NAP) Counteracts UV-B Radiation-Induced ROS Formation in Corneal Epithelium. Antioxidants 2022, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- Fallica, A.N.; Sorrenti, V.; D’Amico, A.G.; Salerno, L.; Romeo, G.; Intagliata, S.; Consoli, V.; Floresta, G.; Rescifina, A.; D’Agata, V.; et al. Discovery of Novel Acetamide-Based Heme Oxygenase-1 Inhibitors with Potent In Vitro Antiproliferative Activity. J. Med. Chem. 2021, 64, 13373–13393. [Google Scholar] [CrossRef] [PubMed]
- Borhade, V.; Pathak, S.; Sharma, S.; Patravale, V. Clotrimazole Nanoemulsion for Malaria Chemotherapy. Part I: Preformulation Studies, Formulation Design and Physicochemical Evaluation. Int. J. Pharm. 2012, 431, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Lee, G.-W. Formulation Approaches for Improving the Dissolution Behavior and Bioavailability of Tolvaptan Using SMEDDS. Pharmaceutics 2022, 14, 415. [Google Scholar] [CrossRef]
- Seo, Y.G.; Kim, D.W.; Yousaf, A.M.; Park, J.H.; Chang, P.-S.; Baek, H.H.; Lim, S.-J.; Kim, J.O.; Yong, C.S.; Choi, H.-G. Solid Self-Nanoemulsifying Drug Delivery System (SNEDDS) for Enhanced Oral Bioavailability of Poorly Water-Soluble Tacrolimus: Physicochemical Characterisation and Pharmacokinetics. J. Microencapsul. 2015, 32, 503–510. [Google Scholar] [CrossRef]
- Ahmed, B.; Rizwanullah, M.; Mir, S.R.; Akhtar, M.S.; Amin, S. Development of Cannabidiol Nanoemulsion for Direct Nose to Brain Delivery: Statistical Optimization, in Vitro and in Vivo Evaluation. Biomed. Mater. 2022, 17, 065009. [Google Scholar] [CrossRef]
- Buya, A.B.; Terrasi, R.; Mbinze, J.K.; Muccioli, G.G.; Beloqui, A.; Memvanga, P.B.; Préat, V. Quality-by-Design-Based Development of a Voxelotor Self-Nanoemulsifying Drug-Delivery System with Improved Biopharmaceutical Attributes. Pharmaceutics 2021, 13, 1388. [Google Scholar] [CrossRef]
- Silva, A.E.; Barratt, G.; Chéron, M.; Egito, E.S.T. Development of Oil-in-Water Microemulsions for the Oral Delivery of Amphotericin B. Int. J. Pharm. 2013, 454, 641–648. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; Alanazi, F.K.; Alsarra, I.A. Impact of Various Nonionic Surfactants on Self-Nanoemulsification Efficiency of Two Grades of Capryol (Capryol-90 and Capryol-PGMC). J. Mol. Liq. 2013, 182, 57–63. [Google Scholar] [CrossRef]
- Leonardi, A.; Bucolo, C.; Romano, G.L.; Platania, C.B.M.; Drago, F.; Puglisi, G.; Pignatello, R. Influence of Different Surfactants on the Technological Properties and in Vivo Ocular Tolerability of Lipid Nanoparticles. Int. J. Pharm. 2014, 470, 133–140. [Google Scholar] [CrossRef]
- Buya, A.B.; Ucakar, B.; Beloqui, A.; Memvanga, P.B.; Préat, V. Design and Evaluation of Self-Nanoemulsifying Drug Delivery Systems (SNEDDSs) for Senicapoc. Int. J. Pharm. 2020, 580, 119180. [Google Scholar] [CrossRef] [PubMed]
- Sukmawati, A.; Utami, W.; Yuliani, R.; Da’i, M.; Nafarin, A. Effect of Tween 80 on Nanoparticle Preparation of Modified Chitosan for Targeted Delivery of Combination Doxorubicin and Curcumin Analogue. IOP Conf. Ser. Mater. Sci. Eng. 2018, 311, 012024. [Google Scholar] [CrossRef]
- Nasr, A.; Gardouh, A.; Ghonaim, H.; Abdelghany, E.; Ghorab, M. Effect of oils, surfactants and cosurfactants on phase behavior and physicochemical properties of self-nanoemulsifying drug delivery system (SNEDDS) for irbesartan and olmesartan. Int. J. Appl. Pharm. 2016, 8, 13–24. [Google Scholar]
- Bonaccorso, A.; Carbone, C.; Tomasello, B.; Italiani, P.; Musumeci, T.; Puglisi, G.; Pignatello, R. Optimization of Dextran Sulfate/Poly-l-Lysine Based Nanogels Polyelectrolyte Complex for Intranasal Ovalbumin Delivery. J. Drug Deliv. Sci. Technol. 2021, 65, 102678. [Google Scholar] [CrossRef]
- Tu, Y.; Zhu, M.; Wang, Z.; Wang, K.; Chen, L.; Liu, W.; Shi, Q.; Zhao, Q.; Sun, Y.; Wang, X.; et al. Melatonin Inhibits Müller Cell Activation and Pro-inflammatory Cytokine Production via Upregulating the MEG3/miR-204/Sirt1 Axis in Experimental Diabetic Retinopathy. J. Cell Physiol. 2020, 235, 8724–8735. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Song, E.; Wang, Z.; Ji, N.; Zhu, L.; Wang, K.; Sun, H.; Zhang, Y.; Zhu, Q.; Liu, X.; et al. Melatonin Attenuates Oxidative Stress and Inflammation of Müller Cells in Diabetic Retinopathy via Activating the Sirt1 Pathway. Biomed. Pharmacother. 2021, 137, 111274. [Google Scholar] [CrossRef]
- Ishikawa, K.; He, S.; Terasaki, H.; Nazari, H.; Zhang, H.; Spee, C.; Kannan, R.; Hinton, D.R. Resveratrol Inhibits Epithelial-Mesenchymal Transition of Retinal Pigment Epithelium and Development of Proliferative Vitreoretinopathy. Sci. Rep. 2015, 5, 16386. [Google Scholar] [CrossRef]
- Cao, K.; Ishida, T.; Fang, Y.; Shinohara, K.; Li, X.; Nagaoka, N.; Ohno-Matsui, K.; Yoshida, T. Protection of the Retinal Ganglion Cells: Intravitreal Injection of Resveratrol in Mouse Model of Ocular Hypertension. Invest. Ophthalmol. Vis. Sci. 2020, 61, 13. [Google Scholar] [CrossRef]
- Käsdorf, B.T.; Arends, F.; Lieleg, O. Diffusion Regulation in the Vitreous Humor. Biophys. J. 2015, 109, 2171–2181. [Google Scholar] [CrossRef]
- Tavakoli, S.; Peynshaert, K.; Lajunen, T.; Devoldere, J.; Del Amo, E.M.; Ruponen, M.; De Smedt, S.C.; Remaut, K.; Urtti, A. Ocular Barriers to Retinal Delivery of Intravitreal Liposomes: Impact of Vitreoretinal Interface. J. Control Release 2020, 328, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Hanieh, P.N.; Bonaccorso, A.; Zingale, E.; Cimarelli, S.; Souto, E.B.; Rinaldi, F.; Marianecci, C.; Pignatello, R.; Carafa, M. Almond Oil O/W Nanoemulsions: Potential Application for Ocular Delivery. J. Drug Deliv. Sci. Technol. 2022, 72, 103424. [Google Scholar] [CrossRef]
- Zhang, R.; Yang, J.; Luo, Q.; Shi, J.; Xu, H.; Zhang, J. Preparation and in Vitro and in Vivo Evaluation of an Isoliquiritigenin-Loaded Ophthalmic Nanoemulsion for the Treatment of Corneal Neovascularization. Drug Deliv. 2022, 29, 2217–2233. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorso, A.; Gigliobianco, M.R.; Lombardo, R.; Pellitteri, R.; Di Martino, P.; Mancuso, A.; Musumeci, T. Nanonized Carbamazepine for Nose-to-Brain Delivery: Pharmaceutical Formulation Development. Pharm. Dev. Technol. 2023, 28, 248–263. [Google Scholar] [CrossRef]
- Muthu, M.S.; Feng, S.-S. Pharmaceutical stability aspects of nanomedicines. Nanomedicine 2009, 4, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Zupančič, Š.; Lavrič, Z.; Kristl, J. Stability and Solubility of Trans-Resveratrol Are Strongly Influenced by pH and Temperature. Eur. J. Pharm. Biopharm. 2015, 93, 196–204. [Google Scholar] [CrossRef]
- Sessa, M.; Tsao, R.; Liu, R.; Ferrari, G.; Donsì, F. Evaluation of the Stability and Antioxidant Activity of Nanoencapsulated Resveratrol during in Vitro Digestion. J. Agric. Food Chem. 2011, 59, 12352–12360. [Google Scholar] [CrossRef]
- Ferreyra, S.; Bottini, R.; Fontana, A. Temperature and Light Conditions Affect Stability of Phenolic Compounds of Stored Grape Cane Extracts. Food Chem. 2023, 405, 134718. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Lee, S.H.; Chow, P.S.; Macbeath, C. Microemulsion Composed of Combination of Skin Beneficial Oils as Vehicle: Development of Resveratrol-Loaded Microemulsion Based Formulations for Skin Care Applications. Colloids Surf. B Biointerfaces 2020, 194, 111161. [Google Scholar] [CrossRef]
- Agustin-Salazar, S.; Gamez-Meza, N.; Medina-Juàrez, L.À.; Soto-Valdez, H.; Cerruti, P. From Nutraceutics to Materials: Effect of Resveratrol on the Stability of Polylactide. ACS Sustain. Chem. Eng. 2014, 2, 1534–1542. [Google Scholar] [CrossRef]
- Bancuta, O.R.; Chilian, A.; Bancuta, I.; Setnescu, R.; Setnescu, T.; Ion, R.M. Thermal Characterization of Resveratrol. Rev. Chim. 2018, 69, 1346–1351. [Google Scholar] [CrossRef]
- Lin, Y.C.; Hu, S.C.S.; Huang, P.H.; Lin, T.C.; Yen, F.L. Electrospun resveratrol-loaded polyvinylpyrrolidone/cyclodextrin nanofibers and their biomedical applications. Pharmaceutics 2020, 12, 552. [Google Scholar] [CrossRef] [PubMed]
- Topal, B.; Altındal, D.Ç.; Gümüşderelioğlu, M. Melatonin/HPβCD complex: Microwave synthesis, integration with chitosan scaffolds and inhibitory effects on MG-63CELLS. Int. J. Pharm. 2015, 496, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.S.; Mandal, S.K.; Malik, A.; Choudhary, A.; Agarwal, S.; Sarkar, S.; Dey, S. Nano micelle: Novel Approach for Targeted Ocular Drug Delivery System. Egypt. J. Chem. 2022, 65, 337–355. [Google Scholar] [CrossRef]
- Corsaro, R.; Lombardo, R.; Ghelardini, C.; Di Cesare Mannelli, L.; Bani, D.; Bonaccorso, A.; Pignatello, R. Development of Eudragit® Nanoparticles for Intranasal Drug Delivery: Preliminary Technological and Toxicological Evaluation. Appl. Sci. 2022, 12, 2373. [Google Scholar] [CrossRef]
- Bonaccorso, A.; Pepe, V.; Zappulla, C.; Cimino, C.; Pricoco, A.; Puglisi, G.; Giuliano, F.; Pignatello, R.; Carbone, C. Sorafenib Repurposing for Ophthalmic Delivery by Lipid Nanoparticles: A Preliminary Study. Pharmaceutics 2021, 13, 1956. [Google Scholar] [CrossRef]
- Cimino, C.; Leotta, C.G.; Marrazzo, A.; Musumeci, T.; Pitari, G.M.; Pignatello, R.; Bonaccorso, A.; Amata, E.; Barbaraci, C.; Carbone, C. Nanostructured Lipid Carrier for the Ophthalmic Delivery of Haloperidol Metabolite II Valproate Ester (±)-MRJF22: A Potential Strategy in the Treatment of Uveal Melanoma. J. Drug Deliv. Sci. Technol. 2023, 87, 104811. [Google Scholar] [CrossRef]
- Vikash, B.; Shashi; Pandey, N.K.; Kumar, B.; Wadhwa, S.; Goutam, U.; Alam, A.; Al-Otaibi, F.; Chaubey, P.; Mustafa, G.; et al. Formulation and Evaluation of Ocular Self-Nanoemulsifying Drug Delivery System of Brimonidine Tartrate. J. Drug Deliv. Sci. Technol. 2023, 81, 104226. [Google Scholar] [CrossRef]
- Gu, H.; Chen, P.; Liu, X.; Lian, Y.; Xi, J.; Li, J.; Song, J.; Li, X. Trimethylated Chitosan-Coated Flexible Liposomes with Resveratrol for Topical Drug Delivery to Reduce Blue-Light-Induced Retinal Damage. Int. J. Biol. Macromol. 2023, 252, 126480. [Google Scholar] [CrossRef]
- Paul, D.R. Elaborations on the Higuchi Model for Drug Delivery. Int. J. Pharm. 2011, 418, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Ekenna, I.C.; Abali, S.O. Comparison of the Use of Kinetic Model Plots and DD Solver Software to Evaluate the Drug Release from Griseofulvin Tablets. J. Drug Deliv. Ther. 2022, 12, 5–13. [Google Scholar] [CrossRef]
- Rüweler, M.; Gülden, M.; Maser, E.; Murias, M.; Seibert, H. Cytotoxic, Cytoprotective and Antioxidant Activities of Resveratrol and Analogues in C6 Astroglioma Cells in Vitro. Chem. Biol. Interact. 2009, 182, 128–135. [Google Scholar] [CrossRef]
- Soo, E.; Thakur, S.; Qu, Z.; Jambhrunkar, S.; Parekh, H.S.; Popat, A. Enhancing Delivery and Cytotoxicity of Resveratrol through a Dual Nanoencapsulation Approach. J. Colloid. Interface Sci. 2016, 462, 368–374. [Google Scholar] [CrossRef]
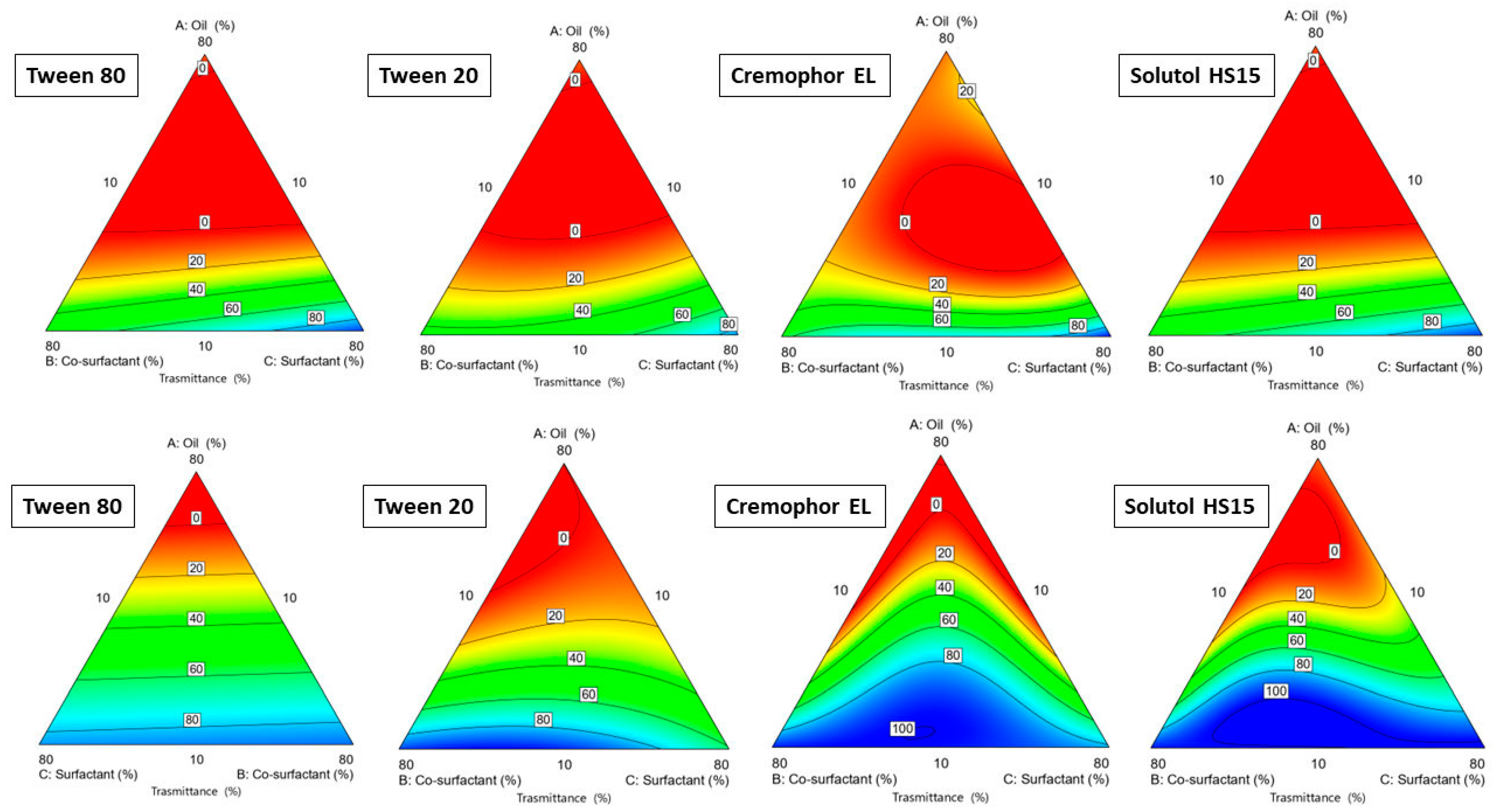




| Component | Units | Type | Minimum | Maximum |
|---|---|---|---|---|
| A (oil) | % (w/w) | Mixture | 10 | 80 |
| B (surfactant) | % (w/w) | Mixture | 10 | 80 |
| C (co-surfactant) | % (w/w) | Mixture | 10 | 80 |
| Constraints | Total (A + B + C) = 100 | |||
| Transmittance | T% | Response | ||
| Factors | Name | Units | Type | Levels | |
|---|---|---|---|---|---|
| Low | High | ||||
| X1 | Oil concn. | % (w/w) | Numeric | 10 | 30 |
| X2 | Surfactant concn. | % (w/w) | Numeric | 10 | 70 |
| X3 | Co-surfactant concn. | % (w/w) | Numeric | 10 | 70 |
| X4 | Surfactant type | Categoric | Tween® 80 Tween® 20 Cremophor® EL Solutol® HS15 | ||
| Constraints: X1 + X2 + X3 = 100 | |||||
| Y1 | Size | nm | |||
| Y2 | Time of emulsification | s | |||
| Y3 | Transmittance | % | |||
| Factors and Responses | Goal | Lower Limit | Upper Limit | |
|---|---|---|---|---|
| X1 | Oil concn. % (w/w) | Minimize | 10 | 30 |
| X2 | Surfactant concn. % (w/w) | Maximize | 20 | 70 |
| X3 | Co-surfactant concn. % (w/w) | Minimize | 20 | 70 |
| X4 | Type of surfactant | In range | Tween® 80, Tween® 20, Cremophor® EL, Solutol® HS15 | |
| Y1 | Mean particle size (nm) | Close to 20 nm | 13.29 | 455.1 |
| Y2 | Time of emulsification (s) | Minimize | 31 | 134 |
| Y3 | % Transmittance | Maximize | 0.1 | 100 |
| Sample | Type of Oil | Oil Concn. % | Surfactant Concn. % | Co-Surfactant Concn. % | Desirability |
|---|---|---|---|---|---|
| A | Tween® 80 | 15.041 | 55.181 | 28.211 | 0.886 |
| B | Tween® 20 | 15.456 | 52.471 | 30.133 | 0.723 |
| C | Cremophor® EL | 14.351 | 58.025 | 23.358 | 0.761 |
| D | Solutol® HS15 | 14.351 | 58.025 | 23.358 | 0.868 |
| Sample | Size (nm) ± SD | % Transmittance | Time of Emulsification (s) |
|---|---|---|---|
| A | 13.26 ± 0.07 | 100 | 12.04 |
| B | 127.29 ± 1.12 | 88 | 12.18 |
| C | 11.29 ± 0.11 | 100 | 17.85 |
| D | 18.82 ± 0.41 | 100 | 15.76 |
| Ratio SNEDDS/STF | Temperature (°C) | Size (nm) ± SD | PDI |
|---|---|---|---|
| 1:10 | 47.2 | 210 ± 17.5 | 0.263 ± 0.090 |
| 1:50 | 60.5 | 201.1 ± 11.73 | 0.311 ± 0.049 |
| 1:100 | 70.0 | 120.1 ± 5.208 | 0.289 ± 0.024 |
| 1:200 | 79.9 | 443.8 ± 286 | 0.575 ± 0.601 |
| 1:300 | 82.2 | 1228.6 ± 16.09 | 0.561 ± 0.105 |
| Sample | Zero Order | First Order | Higuchi | Hixson–Crowell | Korsmeyer–Peppas |
|---|---|---|---|---|---|
| MEL | 0.7465 | 0.6142 | 0.8685 | 0.7692 | 0.8863 |
| RSV | 0.0116 | 0.0072 | 0.0456 | 0.0117 | 0.9736 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zingale, E.; Bonaccorso, A.; D’Amico, A.G.; Lombardo, R.; D’Agata, V.; Rautio, J.; Pignatello, R. Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments. Pharmaceutics 2024, 16, 125. https://doi.org/10.3390/pharmaceutics16010125
Zingale E, Bonaccorso A, D’Amico AG, Lombardo R, D’Agata V, Rautio J, Pignatello R. Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments. Pharmaceutics. 2024; 16(1):125. https://doi.org/10.3390/pharmaceutics16010125
Chicago/Turabian StyleZingale, Elide, Angela Bonaccorso, Agata Grazia D’Amico, Rosamaria Lombardo, Velia D’Agata, Jarkko Rautio, and Rosario Pignatello. 2024. "Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments" Pharmaceutics 16, no. 1: 125. https://doi.org/10.3390/pharmaceutics16010125
APA StyleZingale, E., Bonaccorso, A., D’Amico, A. G., Lombardo, R., D’Agata, V., Rautio, J., & Pignatello, R. (2024). Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments. Pharmaceutics, 16(1), 125. https://doi.org/10.3390/pharmaceutics16010125











