Electrospun Nanofibers Loaded with Plantago major L. Extract for Potential Use in Cutaneous Wound Healing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Plantago major L. Extract Preparation
2.3. Polymeric Solutions
2.4. Electrospinning Process
2.5. Fhysico-Chemical Characterization
2.5.1. Determination of Phenolic Compounds in P. major Extract
2.5.2. Viscometry
2.5.3. Scanning Electron Microscopy—SEM
2.5.4. Wettability Assay
2.5.5. Fourier Transformed Infrared Spectroscopy—FTIR
2.5.6. Thermal Behavior using DSC and TGA/DTA
2.5.7. In Vitro Swelling Degree
2.5.8. Water Vapor Transpiration Rate Assay (WVTR)
2.6. In Vitro Release of the Extract
In Vitro Plantago major Extract Release Evaluation
2.7. In Vitro Microbiological Testing
2.7.1. In Vitro Minimum Inhibitory Concentration (MIC) Test
2.7.2. Antimicrobial Assay of P. major
3. Results and Discussion
3.1. Composition Evaluation of Plantago major Extract
3.2. Minimal Inhibition Concentration (MIC) Evaluation
3.3. Viscosity Evaluation
3.4. PCL/P. major Electrospun Samples
3.5. Fourier Transformation Infrared—FTIR
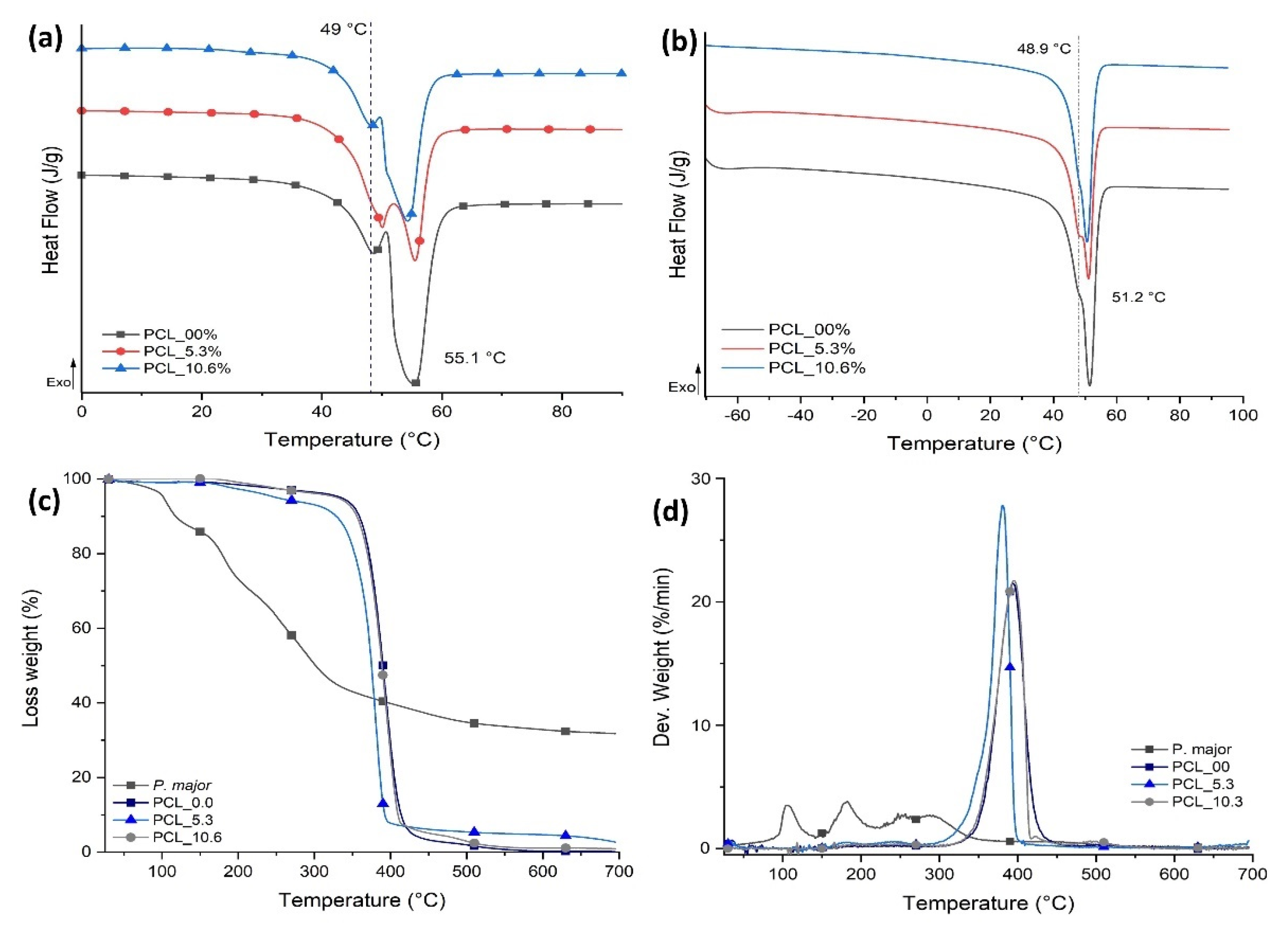
3.6. Thermal Behavior of Electrospun Mats
3.7. In Vitro Swelling Behavior of Electrospun Nanofibers
3.8. P. major Extract Encapsulation
3.9. Water Vapor Permeation Test
3.10. P. major Extract Release Assay
3.11. Antimicribial Assay from PCL/P. major Electrosun Nanofibers
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Canbolat, M.F.; Celebioglu, A.; Uyar, T. Drug delivery system based on cyclodextrin-naproxen inclusion complex incorporated in electrospun polycaprolactone nanofibers. Colloids Surf. B Biointerfaces 2014, 115, 15–21. [Google Scholar] [CrossRef]
- Turan, C.U.; Guvenilir, Y. Electrospun poly(ω-pentadecalactone-co-ε-caprolactone)/gelatin/chitosan ternary nanofibers with antibacterial activity for treatment of skin infections. Eur. J. Pharm. Sci. 2022, 170, 106113. [Google Scholar] [CrossRef]
- De Oliveira, B.G.R.B.; Oliveira, B.C.; Deutsch, G.; Pessanha, F.S.; Thiré, R.M.S.M.; de Castilho, S.R. rhEGR-loaded hydrogel in the treatment of chronic wounds in patients with diabetes: Clinical cases. Gels 2022, 8, 523. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Ding, L.; Chen, Y.; Chen, G.; Zhao, T.; Yu, Y. Nano-silver functionalized polysaccharides as platform for wound dressings: A review. Int. J. Biol. Macromol. 2022, 194, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Abrigo, M.; Mc Arthur, S.L.; Kinsgshott, P. Electrospun nanofibers as dressings for chronic wound care: Advances, Challenges and futures prospects. Macromol. Biosci. 2014, 14, 772–792. [Google Scholar] [CrossRef] [PubMed]
- Solovieva, E.V.; Teterina, A.Y.; Klein, O.I.; Komlev, V.S.; Alekseev, A.A.; Panteleyev, A.A. Sodium alginate-based composites as a collagen substitute for skin bioengineering. Biomed. Mater. 2020, 16, 015002. [Google Scholar] [CrossRef]
- Serbezeanu, D.; Bargan, A.; Homocianu, M.; Aflori, M.; Rimbu, C.M.; Enache, A.A.; Vlad-Bubulac, T. Electrospun polyvinyl alcohol loaded with phytotherapeutic agents for wound healing applications. Nanomaterials 2021, 11, 3336. [Google Scholar] [CrossRef] [PubMed]
- Gangwar, A.; Kumar, P.; Singh, R.; Kush, P. Recent advances in mupirocin delivery strategies for the treatment of bacterial skin and soft tissue infection. Future Pharmacol. 2021, 1, 80–103. [Google Scholar] [CrossRef]
- Krysiak, Z.J.; Stachewicz, U. Electrospun fibers as carriers for topical drug delivery and release in skin bandages and patches for atopic dermatitis treatment. WIREs Nanomed. Nanobiotechnol. 2022, 15, e1829. [Google Scholar] [CrossRef] [PubMed]
- Unalan, I.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Frank, G.; Boccaccinni, A.R. Physical and antibacterial properties of peppermint essential oil loaded poly(ε-caprolactone) (PCL) electrospun fiber mats for wound healing. Front. Bioeng. Biotechnol. 2019, 26, 346. [Google Scholar] [CrossRef]
- Jawhari, F.Z.; Moussaori, A.E.; Bourhia, M.; Imtara, H.; Mechchate, H.; Es-Safi, I.; Ullah, R.; Ezzeldin, E.; Mostafa, G.A.; Grafov, A.; et al. Anacyclus pyrethrum (L): Chemical composition, analgesic, anti-inflammatory, and wound healing properties. Molecules 2020, 25, 5469. [Google Scholar] [CrossRef]
- Shah, M.Z.; Guan, Z.H.; Din, A.U.; Ali, A.; Rehman, A.U.; Jan, K.; Faisal, S.; Saud, S.; Adnan, M.; Wahid, F.; et al. Synthesis of silver nanoparticles using Plantago lanceolata extract and assessing their antibacterial and antioxidant activities. Sci. Rep. 2021, 11, 20754. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, D.M.; Barbosa, W.S.; Rangel, W.S.P.; Valle, I.M.M.; Matos, A.P.S.; Melgaço, F.G.; Dias, M.L.; Ricci Júnior, E.; da Silva, L.C.P.; de Breu, L.C.L.; et al. Polymeric membrane based on polylactic acid and babassu oil for wound healing. Mater. Today Commun. 2021, 26, 102173. [Google Scholar] [CrossRef]
- Gorain, B.; Pandey, M.; Leng, N.H.; Yan, C.W.; Nie, K.W.; Kaur, S.J.; Marshall, V.; Sisinthy, S.P.; Panneerselvam, J.; Mologulu, N.; et al. Advanced drug delivery systems containing herbal components for wound healing. Int. J. Pharm. 2022, 617, 121617. [Google Scholar] [CrossRef] [PubMed]
- Ouedrhiri, W.; Mechchate, H.; Moja, S.; Baudino, S.; Saleh, A.; Al-Kamaly, O.M.; Grafov, A.; Greche, H. Optimized antibacterial effects in a designed mixture of essential oils of Myrtus communis, Artemisa herba-alba and Thymus serpyllum for wide range of applications. Foods 2022, 11, 132. [Google Scholar] [CrossRef]
- Mancipe, J.M.A.; Dias, M.L.; Thiré, R.M.S.M. Type I collagen—Poly (vinyl alcohol) electrospun nanofibers: FTIR study of the collagen helical structure preservation. Polym. Plast. Technol. Mater. 2022, 61, 846–860. [Google Scholar] [CrossRef]
- Raju, N.R.; Silina, E.; Stupin, V.; Manturova, N.; Chidambaram, S.B.; Achar, R.R. Multifunctional and smart wound dressing—A review on recent research advancements in skin regenerative medicine. Pharmaceutics 2022, 14, 1574. [Google Scholar] [CrossRef]
- Toledo, A.L.M.M.; da Silva, T.N.; Vaucher, A.C.S.; Miranda, A.H.H.; Silva, G.C.C.; Vaz, M.E.R.; da Silva, L.V.; Barradas, T.N.; Picciani, P.H.S. Polymer nanofibers for biomedical applications: Advances in electrospinning. Curr. Appl. Polym. Sci. 2021, 4, 190–209. [Google Scholar] [CrossRef]
- Ghomi, E.R.; Khosravi, F.; Neisiany, R.E.; Shakiba, M.; Zare, M.; Lakshminarayanan, R.; Chellappan, V.; Adbouss, M.; Ramakrishna, S. Advances in electrospinning of aligned nanofiber scaffolds used for wound dressings. Curr. Opin. Biomed. Eng. 2022, 22, 100393. [Google Scholar] [CrossRef]
- Li, M.; Qiu, W.; Wang, Q.; Li, N.; Liu, L.; Wang, X.; Yu, J.; Li, X.; Li, F.; Wu, D. Nitric oxide-releasing tryptophan-bsed poly(ester uera)s electrospun composite nanofiber mats with antibacterial and antibiofilm activities for infected wound healing. ACS Appl. Mater. Interfaces 2022, 14, 15911–15926. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, C.; Wang, Q.; Zhou, F.; Li, T.; Wang, B.; Su, W.; Shag, D.; Wu, S. A simple, quick, and cost-effective strategy to fabricate polycaprolactone/silk fibroin nanofiber yarns for biotextile-based tissue scaffolds application. Eur. Polym. J. 2023, 186, 111863. [Google Scholar] [CrossRef]
- Keler, K.; Daglilar, S.; Gunduz, O.; Yuksek, M.; Sahin, Y.M.; Ekren, N.; Oktar, F.N.; Salman, S. Mechanical behavior of PCL nanofibers. Key Eng. Mater. 2016, 696, 196–201. [Google Scholar] [CrossRef]
- Altun, E.; Ahmed, J.; Aydogdu, M.O.; Harker, A.; Edirisinghe, M. The effect of solvent and pressure on polycaprolactone solutions for particle and fibre formation. Eur. Polym. J. 2022, 173, 111300. [Google Scholar] [CrossRef]
- Dias, J.R.; Sousa, A.; Augusto, A.; Bártolo, P.J.; Granja, P.L. Electrospun polycaprolactone (PCL) degradation: Na in vitro and in vivo study. Polymers 2022, 14, 3397. [Google Scholar] [CrossRef] [PubMed]
- Zubair, M.; Ekholm, A.; Nybom, H.; Renvert, S.; Widen, C.; Rumpunen, K. Effects of Plantago major L. leaf extracts on oral epithelial cells in a scratch assay. J. Ethnopharmacol. 2012, 141, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Samuelsen, A.B. The traditional uses, chemical constituents and biological activities of Plantago major L. A review. J. Ethnopharmacol. 2000, 71, 1–21. [Google Scholar] [CrossRef]
- Farid, A.; Sheibani, M.; Shojaii, A.; Noori, M.; Motevalian, M. Evaluation of anti-inflammatory effects of leaf and seed extracts of Plantago major on acetic acid-induced ulcerative colitis in rats. J. Ethnopharmacol. 2022, 298, 115595. [Google Scholar] [CrossRef]
- Soliman, M.A.; Galal, T.M.; Naim, M.A.; Khalafallah, A.A. Seasonal variation in the secondary metabolites and antimicrobial activity of Plantago major L. from Egyptian heterogenic habitats. Egypt. J. Bot. 2022, 62, 255–273. [Google Scholar] [CrossRef]
- Nikaeen, G.; Rahmdell, S.; Samari, F.; Mahdavinia, S. Central composite design for optimizing the biosynthesis of silver nanoparticles using Plantago major extract and investigation antibacterial, antifungal and antioxidant activity. Sci. Rep. 2020, 10, 9642. [Google Scholar] [CrossRef]
- Lukova, P.; Nikolova, M.; Petit, E.; Elboutachfaiti, R.; Vasileva, T.; Katsarov, P.; Manev, H.; Gardarin, C.; Pierre, G.; Michaud, P.; et al. Prebiotic activity of poly- and oligosaccharides from Plantago major L. leaves. Appl. Sci. 2020, 10, 2648. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Rupasinghe, H.P.V. Invitro anti-inflammatory properties of selected green leafy vegetables. Biomedicines 2018, 6, 107. [Google Scholar] [CrossRef] [PubMed]
- Silva Neto, A.R.; Sousa, A.C.S.O.; Camboim, L.F.R.; da Silva, J.P.R.; Maia Filho, A.L.M.; Leal, F.R.; Costa, C.A.C.B.; Freitas, J.M.D.; de Freitas, J.D.; Marques, R.B.; et al. Phytochemical profile and analgesic Activity of the extract from Plantago major L. Rev. Virtual Quim. 2023, Preprints. [Google Scholar] [CrossRef]
- Keshavari, A.; Montaseri, H.; Akrami, R.; Saravestani, H.M.; Khosravi, F.; Foolad, S.; Zardosht, M.; Zareie, S.; Saharkhiz, M.J.; Shahriarirad, R. Therapeutuc efficacy of great plantain (Plantago major L.) in the treatment of second-degree burn wounds: A case-control study. Int. J. Clin. Pract. 2022, 2022, 4923277. [Google Scholar] [CrossRef]
- Ghanadian, M.; Soltani, R.; Homayouni, A.; Khorvash, F.; Jouabadi, S.M.; Abdollahzadeh, M. The effect of Plantago major hydroalcoholic extract on the healing of diabetic foot and pressure ulcers: A randomized open-label controlled clinical trial. Int. J. Low. Extrem. Wounds 2022, 2022, 1–7. [Google Scholar] [CrossRef]
- Albahri, G.; Badran, A.; Hijazi, A.; Daou, A.; Baydoun, E.; Nasser, M.; Merah, O. The therapeutic wound healing bioactivities of various medicinal plants. Life 2023, 13, 317. [Google Scholar] [CrossRef]
- Thomé, R.G.; dos Santos, H.B.; dos Santos, F.V.; Oliveira, R.J.S.; de Camargo, L.F.; Pereira, M.N.; Longatti, T.R.; Souto, C.M.; Franco, C.S.; Schüffner, R.O.A.; et al. Evaluation of healing wound and genotoxicity potential from extracts hydroalcoholic of Plantago major and Siparuna guianensis. Exp. Biol. Med. 2012, 237, 1379–1986. [Google Scholar] [CrossRef] [PubMed]
- Amini, M.; Kherad, M.; Mehrabani, D.; Azarpira, N.; Panjehshahin, M.R.; Tanideh, N. Effect of Plantago major on burn wound healing in rat. J. Appl. Anim. Res. 2010, 37, 53–56. [Google Scholar] [CrossRef]
- Reina, E.; Al-Shibani, N.; Allam, E.; Gregson, K.S.; Kowolik, M.; Windson, J. The effects of Plantago major on the activation of the neutrophil respiratory burst. J. Tradit. Complement. Med. 2013, 3, 268–272. [Google Scholar] [CrossRef]
- Golkar, P.; Kalani, S.; Allafchian, A.R.; Mohammadi, H.; Jalali, S.A.H. Fabrication and characterization of electrospun Plantago major seed mucilage/PVA nanofibers. J. Appl. Polym. Sci. 2019, 136, 47852. [Google Scholar] [CrossRef]
- De Castro, K.C.; Silva, E.K.; Campos, M.G.N.; Mei, L.H.I. Hyaluronic acid/polyvinyl alcohol electrospun nanofiber membranes loaded with Plantago major extract for smart wound dressings. ACS Appl. Nano Mater. 2022, 5, 12616–12625. [Google Scholar] [CrossRef]
- Mello, J.C.; Gonzalez, M.V.D.; Moraes, V.W.R.; Prieto, T.; Nascimento, O.R.; Rodrigues, T. Protective effect of Plantago major extract against t-BOOH-Induced mitochondrial oxidative damage and cytotoxicity. Molecules 2015, 20, 17747–17759. [Google Scholar] [CrossRef] [PubMed]
- Mancipe, J.M.A.; Dias, M.L.; Thiré, R.M.S.M. Morphological evaluation of electrospun polycaprolactone fibers depending on the type of solvent. Matéria (Rio J.) 2019, 24, e12400. [Google Scholar] [CrossRef]
- Rabello, L.G.; Ribeiro, R.C.C. Bio-based polyurethane resin: An ecological binder for a novel class of building materials-composites. Mater. Lett. 2022, 311, 131566. [Google Scholar] [CrossRef]
- Souza, J.P.L.M.; Pires, L.O.; dos Santos, R.F.; Prudêncio, E.R.; Sant’Ana, L.D.; Ferreira, D.A.S.; Castro, R.N. Estudo químico e potencial antimicrobiano da própolis brasileira produzida por diferentes espécies de abelhas. Rev. Virtual Quim. 2019, 11, 1480–1497. [Google Scholar] [CrossRef]
- Mouro, C.; Gomes, A.P.; Ahonen, M.; Fangueiro, R.; Gouveia, I.C. Chelidonium majus L. incorporated emulsion electrospun PCL/PVA_PEC nanofibrous meshes for antibacterial wound dressing applications. Nanomaterials 2021, 11, 1785. [Google Scholar] [CrossRef]
- Salgueiro, F.B.; Castro, R.N. Comparação entre a composição química e capacidade antioxidante de diferentes extratos de própolis verde. Quim. Nova 2016, 39, 1192–1199. [Google Scholar] [CrossRef]
- Anaya-Mancipe, J.M.; Pereira, L.C.B.; Borchio, P.G.M.; Dias, M.L.; Thiré, R.M.S.M. Novel polycaprolactone (PCL)-type I collagen core-shell electrospun nanofibers for wound healing applications. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 111, 366–381. [Google Scholar] [CrossRef]
- Snetkov, P.; Rogacheva, E.; Kremleva, A.; Morozkina, S.; Uspenskaya, M.; Kraeva, L. In-Vitro antibacterial activity of curcumin-loaded nanofibers based on hyaluronic acid against multidrug-resistant ESKAPE pathogens. Pharmaceutics 2022, 14, 1186. [Google Scholar] [CrossRef]
- Calatayud, M.; López-de-Dicastillo, C.; López-Carballo, G.; Vélez, D.; Muñoz, P.H.; Gavara, R. Active films based on cocoa extract with antioxidant, antimicrobial and biological applications. Food Chem. 2013, 139, 51–58. [Google Scholar] [CrossRef]
- CLASI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, Approved Standard, 9th ed.; Document M07-A9; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Anusree, S.S.; Nisha, V.M.; Priyanka, A.; Raghu, K.G. Insulin resistance by TNF-α is associated with mitochondrial dysfunction in 3T3-L1 adipocytes and is ameliorated by punicic acid, a PPARγ agonist. Mol. Cell. Endocrinol. 2015, 413, 120–128. [Google Scholar] [CrossRef]
- Chookalaii, H.; Riahi, H.; Shariatmadari, Z.; Mazarei, Z.; Hashtroudi, M.S. Enhancement of total flavonoid and phenolic contents in Plantago major L. with growth promoting cyanobacteria. J. Agric. Sci. Technol. 2020, 22, 505–518. Available online: http://jast.modares.ac.ir/article-23-21403-en.html (accessed on 22 February 2023).
- Karima, S.; Farida, S.; Mihoub, Z.M. Antioxidant and antimicrobial activities of Plantago major. Int. Pharm. Pharm Sci. 2015, 5, 58–64. [Google Scholar]
- Süntar, I.; Akkol, K.E.; Nahar, L.; Sarker, S.D. Wound healing and antioxidant properties do they coexist in plants? Free Radic. Antioxid. 2012, 2, 1–7. [Google Scholar] [CrossRef]
- Adom, M.B.; Taher, M.; Mutalabisin, M.F.; Amri, M.S.; Kudos, M.B.A.; Sulaiman, M.W.A.W.; Sengupta, P.; Susanti, D. Chemical constituents and medical benefits of Plantago major. Biomed. Pharmacother. 2017, 96, 348–360. [Google Scholar] [CrossRef]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Naghavi, M. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Carlos, A.L.M.; Mancipe, J.M.A.; Dias, M.L.; Thiré, R.M.S.M. Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) core-shell spun fibers produced by solution blow spinning for bioactive agent’s encapsulation. J. Appl. Polym. Sci. 2022, 139, 52081. [Google Scholar] [CrossRef]
- Figueiredo, A.C.; Anaya-Mancipe, J.M.; de Barros, A.O.S.; Santos-Oliveira, R.; Dias, M.L.; Thiré, R.M.S.M. Nanostructured electrospun polycaprolactone—Propolis mats composed of different morphologies for potential use in wound healing. Molecules 2022, 27, 5351. [Google Scholar] [CrossRef]
- Mohajeri, P.; Sourki, A.H.; Nikoo, A.M.; Ertas, Y.N. Fabrication, characterization and antimicrobial activity of electrospun Plantago psyllium L. seed gum/gelatine nanofibres incorporated with Curcumim cymium essential oil nanoemulsion. Int. J. Food Sci. Technol. 2023, 58, 1832–1840. [Google Scholar] [CrossRef]
- Oviedo, M.; Montoya, Y.; Agudelo, W.; García-García, A.; Bustamante, J. Effect of molecular weight and nanoarchitecture of chitosan and polycaprolactone electrospun membranes on physicochemical and hemocompatible properties for wound dressing. Polymers 2021, 13, 4320. [Google Scholar] [CrossRef]
- Kulkarni, D.; Musale, S.; Panzade, P.; Paiva-Santos, A.C.; Sonwane, P.; Madine, M.; Choundhe, P.; Giram, P.; Cavalu, S. Surface functionalization of nanofibers: The multifaceted approach for advanced biomedical applications. Nanomaterials 2022, 12, 3899. [Google Scholar] [CrossRef]
- Toledo, A.L.M.M.; Ramalho, B.S.; Picciani, L.S.; Baptista, L.S.; Martinez, A.M.B.; Dias, M.L. Effect of three different amines on the surface properties of electrospun polycaprolactone mats. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 1258–1270. [Google Scholar] [CrossRef]
- Elzein, T.; Nasser-Eddine, M.; Delaite, C.; Bistac, S.; Dumas, P. FTIR study of polycaprolactone chain organization at interfaces. J. Colloid Interface Sci. 2004, 273, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Mancini, M.C.; de Oliveira, F.C.S.; Passos, T.M.; Quilty, B.; Thiré, R.M.S.M.; McGuinness, G.B. FTIR analysis and quantification of phenols and flavonoids of five commercially available plant extracts used in wound healing. Matéria (Rio J.) 2016, 21, 11743. [Google Scholar] [CrossRef]
- Dewi, A.F.; Prajitno, A.; Yuniarti, A. Phytochemicals and the ability of Plantago major Linn. extract to inhibit the growth of Aeromonas hydrophila. J. Exp. Life Sci. 2019, 9, 70–75. [Google Scholar] [CrossRef]
- Behbahani, B.A.; Yazdi, F.T.; Shahidi, F.; Hesarinejad, M.A.; Mortazavi, S.A.; Mohebbi, M. Plantago major seed mucilage: Optimization of extract and some physicochemical and rheological aspects. Carbohydr. Polym. 2017, 155, 68–77. [Google Scholar] [CrossRef]
- Sukweenadhi, J.; Setiawan, K.I.; Avanti, C.; Kartini, K.; Rupa, E.J.; Yang, D.C. Scale-up of green synthesis and characterization of silver nanoparticles using ethanol extract of Plantago major L. leaf and its antibacterial potential. S. Afr. J. Chem. Eng. 2021, 38, 1–8. [Google Scholar] [CrossRef]
- Mohammed, N.K. Phytochemical screening by FTIR spectroscopy analysis and anti-bacterial activity of methanolic extract of selected medicinal plant of Anethum graveolens and Plantago major. Ann. Romanian Soc. Cell Biol. 2021, 25, 3110–3122. Available online: https://www.annalsofrscb.ro/index.php/journal/article/view/2852 (accessed on 14 March 2023).
- Bikiaris, N.D.; Koumentakou, I.; Michailidou, G.; Kostoglou, M.; Vlachou, M.; Barmpalexis, P.; Karavas, E.; Papageorgiou, G.Z. Investigation of molecular weight, polymer concentration and process parameters factors on the sustained release of the anti-multiple-sclerosis agent teriflunomide from poly(ε-caprolactone) electrospun nanofibrous matrices. Pharmaceutics 2022, 14, 1693. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, H.; Turng, L.S.; Li, Q. Crystalline morphology of electrospun poly(ε-caprolactone) (PCL) nanofibers. Ind. Eng. Chem. Res. 2013, 52, 4939–4949. [Google Scholar] [CrossRef]
- Krysiak, Z.J.; Stachewicz, U. Urea-based patches with controlled release for potential atopic dermatitis treatment. Pharmaceutics 2022, 14, 1494. [Google Scholar] [CrossRef]
- Pedrosa, M.C.G.; dos Anjos, S.A.; Mavropoulos, E.; Bernardo, P.L.; Granjeiro, J.M.; Rossi, A.M.; Dias, M.L. Structural and biological compatibility of polycaprolactone/zinc-hydroxyapatite electrospun nanofibers for tissue regeneration. J. Bioact. Compat. Polym. 2021, 36, 314–333. [Google Scholar] [CrossRef]
- Saraiva, M.M.; Campelo, M.S.; Câmara-Neto, J.F.; Lima, A.B.N.; Silva, G.A.; Dias, A.T.F.F.; Ricardo, N.M.P.S.; Kaplan, D.L.; Ribeiro, M.E.N.P. Alginate/polyvinyl alcohol films for wound healing: Advantages and challenges. J. Biomed. Mater. Res. B 2022, 111, 351465. [Google Scholar] [CrossRef] [PubMed]
- Karizmenh, M.S.; Poursamar, S.A.; Kefayat, A.; Farahbakhsh, Z.; Rafienia, M. An in vitro and in vivo study of PCL/chitosan electrospun mat on polyurethane/propolis foam as a bilayer wound dressing. Biomater. Adv. 2022, 135, 112667. [Google Scholar] [CrossRef] [PubMed]
- Nuutila, K.; Eriksson, E. Moist wound healing with commonly available dressing. Adv. Wound Care 2021, 10, 685–698. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, L.; Zhang, C.; Danhua, L.; Meng, S.; Zhang, W.; Meng, S. Effect on polymer permeability and solvent removal rate on in situ forming implants: Drug burst release and microstructure. Pharmaceutics 2019, 11, 520. [Google Scholar] [CrossRef]
- Shirley, K.P.; Windsor, J.; Eckert, G.J.; Gregory, R.L. In vitro effects of Plantago major extract, Aucubin, and Baicalein on candida albicans biofilm formation, metabolic activity, and cell surface hydrophobicity. J. Prosthodont. 2017, 26, 508–515. [Google Scholar] [CrossRef]
- Araújo, J.; Dobrovolski, M.; Kozlowski-Júnior, V.A.; Rezende, M. Use of Plantago major for the control of dental biofilm: Case report. Res. Soc. Dev. 2021, 10, e29610111491. [Google Scholar] [CrossRef]
- Alizadeh, B.B.; Shahidi, F.; Tabatabaei, Y.F.; Mortazavi, S.A.; Mohebbi, M. The antimicrobial effect and interaction between the aqueous and ethanolic extract of Plantago major on Staphylococcus aureus, Listeria innocua, Escherichia coli and Pseudomonas aeruginosa In vitro. Iran. J. Infect Dis. Trop. Med. 2017, 21, 1–8. [Google Scholar]
- Trinidade, G.O.; Alves, V.H.; Mariño, P.A.; Maldaner, G.; Menezes, A.P.S.; dos Reis, R.O. Triagem fitoquímica e avaliação do potencial antibacteriano de extratos das folhas de Plantago major L. Rev. Iniciação Cient. Univ. Val. Rio Verde 2018, 9, 41–48. [Google Scholar]
- Petrović, M.; Jovanović, M.; Lević, S.; Nedović, V.; Mitić-Ćulafić, D.; Semren, T.Ž.; Veljović, S. Valorization potential of Plantago major L. solid waste remaining after industrial tincture production: Insight into the chemical composition and bioactive properties. Waste Biomass Valorization 2022, 13, 1639–1651. [Google Scholar] [CrossRef]
- Razik, B.M.A.; Hasan, H.A.; Murtadha, M.K. The study of antibacterial activity of Plantago major and Ceratonia siliqua. Iraqi Postgrad. Med. J. 2012, 11, 130–135. [Google Scholar]









| Sample | 1st Heating Cycle | 2nd Heating Cycle | ||||
|---|---|---|---|---|---|---|
| Tm (°C) | ΔHm (J/g) | Xc | Tm (°C) | ΔHm (J/g) | Xc | |
| PCL_0.0 | 55.4 | 123.0 | 81.1 | 51.4 | 117.0 | 77.1 |
| PCL_5.3 | 55.4 | 80.5 | 53.1 | 51.1 | 74.2 | 48.9 |
| PCL_10.6 | 54.3 | 88.5 | 58.3 | 50.6 | 86.0 | 56.7 |
| Degradation Event | Tonset (°C) | Tmax (°C) | Tendset (°C) | Mass Loss (%) | |
|---|---|---|---|---|---|
| P. major | 1° | 45 | 103.0 | 167.1 | 11.9 |
| 2° | 167.1 | 182.3 | 239.6 | 18.1 | |
| 3° | 239.7 | 287.1 | 438.8 | 38.2 | |
| PCL_00 | 1° | 368.7 | 394.0 | 445.3 | 99.9 |
| PCL_5.3 | 1° | 170.0 | 180.6 | 237.2 | 04.8 |
| 2° | 360.2 | 380.7 | 430.4 | 92.5 | |
| PCL_10.6 | 1° | 170.7 | 192.4 | 367.9 | 05.0 |
| 2° | 367.9 | 394.6 | 457.5 | 94.0 | |
| Sample | Fiber Diameters (nm) | Mass Variation | |
|---|---|---|---|
| Before | After | (%) | |
| PCL_00 | 233.2 ± 84.2 | 258.3 ± 77.8 | 3.7 ± 0.38 |
| PCL_5.3 | 235.5 ± 72.2 | 261.9 ± 84.4 | 7.1 ± 0.15 |
| PCL_10.6 | 378.5 ± 118.5 | 428.1 ± 136.9 | 30.4 ± 10.2 |
| Sample | Drug-Loading Capacity (DL) | Encapsulation Efficiency (EE) | ||
|---|---|---|---|---|
| Theoretical (%) | Experimental (%) | Theoretical (%) | Experimental (%) | |
| PCL_5.3 | 25 | 13.02 | ~100 | 63.52 |
| PCL_10.6 | 50 | 31.01 | ~100 | 60.31 |
| Sample | J = Δm/Δt (g/min) | Water Vapor Transpiration Rate (g/m2 per day) |
|---|---|---|
| PCL_00 | 0.01008 ± 0.00083 | 1471.34 |
| PCL_5.3 | 0.01213 ± 0.00047 | 1545.98 |
| PCL_10.6 | 0.01122 ± 0.00045 | 1492.83 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anaya-Mancipe, J.M.; Queiroz, V.M.; dos Santos, R.F.; Castro, R.N.; Cardoso, V.S.; Vermelho, A.B.; Dias, M.L.; Thiré, R.M.S.M. Electrospun Nanofibers Loaded with Plantago major L. Extract for Potential Use in Cutaneous Wound Healing. Pharmaceutics 2023, 15, 1047. https://doi.org/10.3390/pharmaceutics15041047
Anaya-Mancipe JM, Queiroz VM, dos Santos RF, Castro RN, Cardoso VS, Vermelho AB, Dias ML, Thiré RMSM. Electrospun Nanofibers Loaded with Plantago major L. Extract for Potential Use in Cutaneous Wound Healing. Pharmaceutics. 2023; 15(4):1047. https://doi.org/10.3390/pharmaceutics15041047
Chicago/Turabian StyleAnaya-Mancipe, Javier M., Vanessa M. Queiroz, Rafael F. dos Santos, Rosane N. Castro, Verônica S. Cardoso, Alane B. Vermelho, Marcos L. Dias, and Rossana M. S. M. Thiré. 2023. "Electrospun Nanofibers Loaded with Plantago major L. Extract for Potential Use in Cutaneous Wound Healing" Pharmaceutics 15, no. 4: 1047. https://doi.org/10.3390/pharmaceutics15041047
APA StyleAnaya-Mancipe, J. M., Queiroz, V. M., dos Santos, R. F., Castro, R. N., Cardoso, V. S., Vermelho, A. B., Dias, M. L., & Thiré, R. M. S. M. (2023). Electrospun Nanofibers Loaded with Plantago major L. Extract for Potential Use in Cutaneous Wound Healing. Pharmaceutics, 15(4), 1047. https://doi.org/10.3390/pharmaceutics15041047








