Nanostructured Lipid Carriers Aimed to the Ocular Delivery of Mangiferin: In Vitro Evidence
Abstract
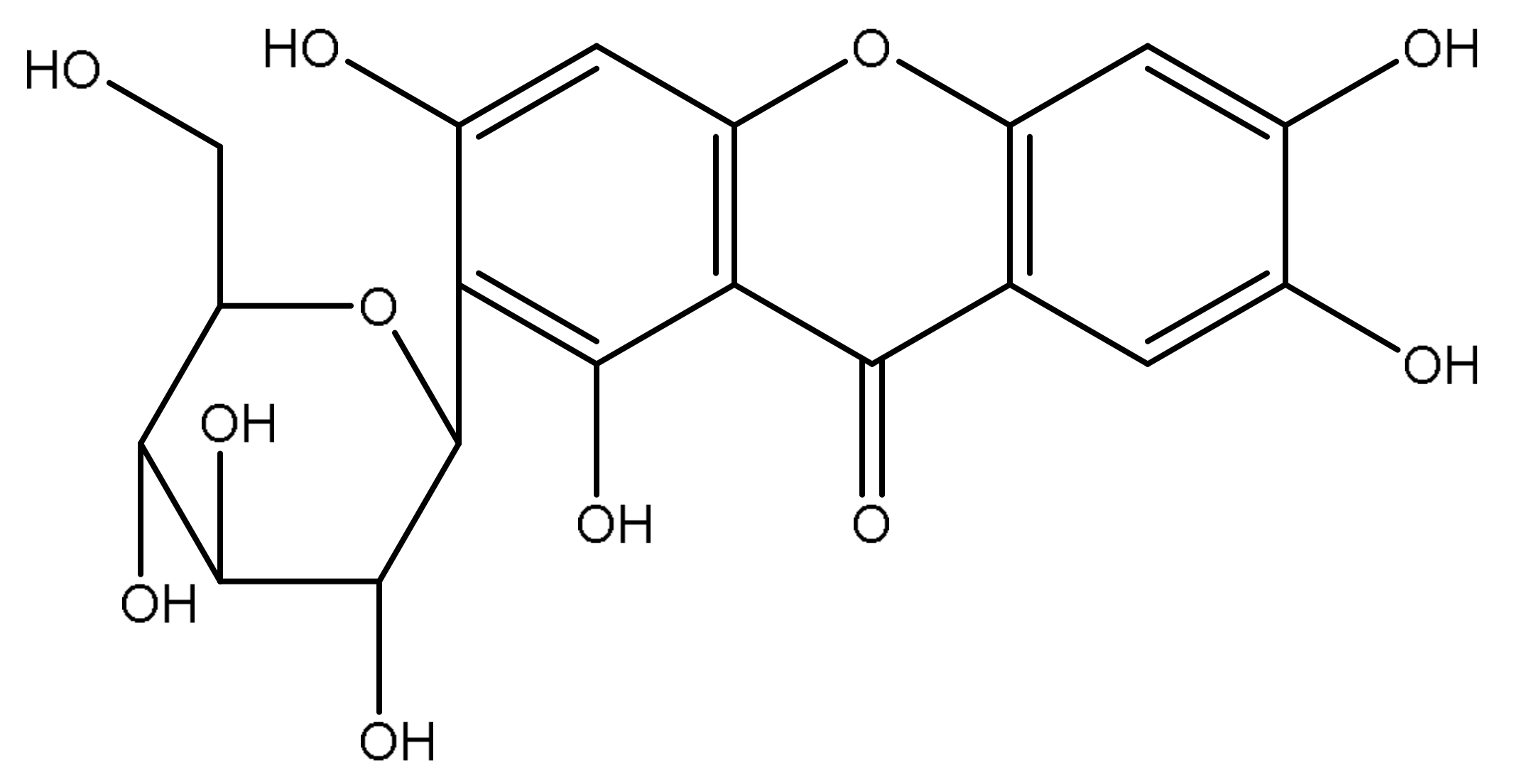
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. MGN–NLC Formulation
2.3. MGN–NLC Characterization
2.4. Encapsulation Efficiency
2.5. Lyophilization Stability
2.6. Corneal Permeability Assay
2.7. Cell Culture and Treatments
2.8. Cell Viability Assay
2.9. GSH Assay
2.10. ROS Measurement
2.11. Statistical Analysis
3. Results and Discussion
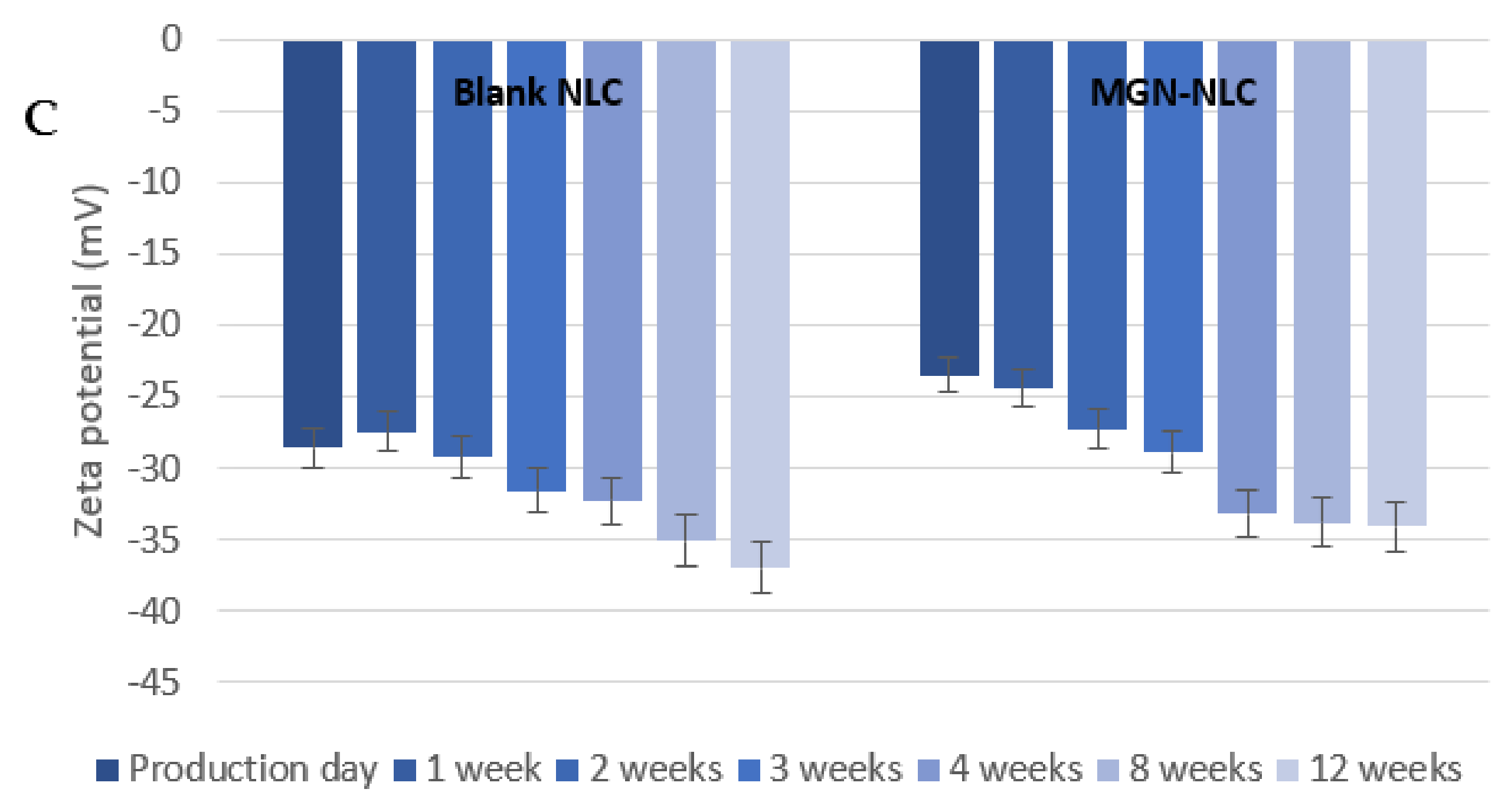
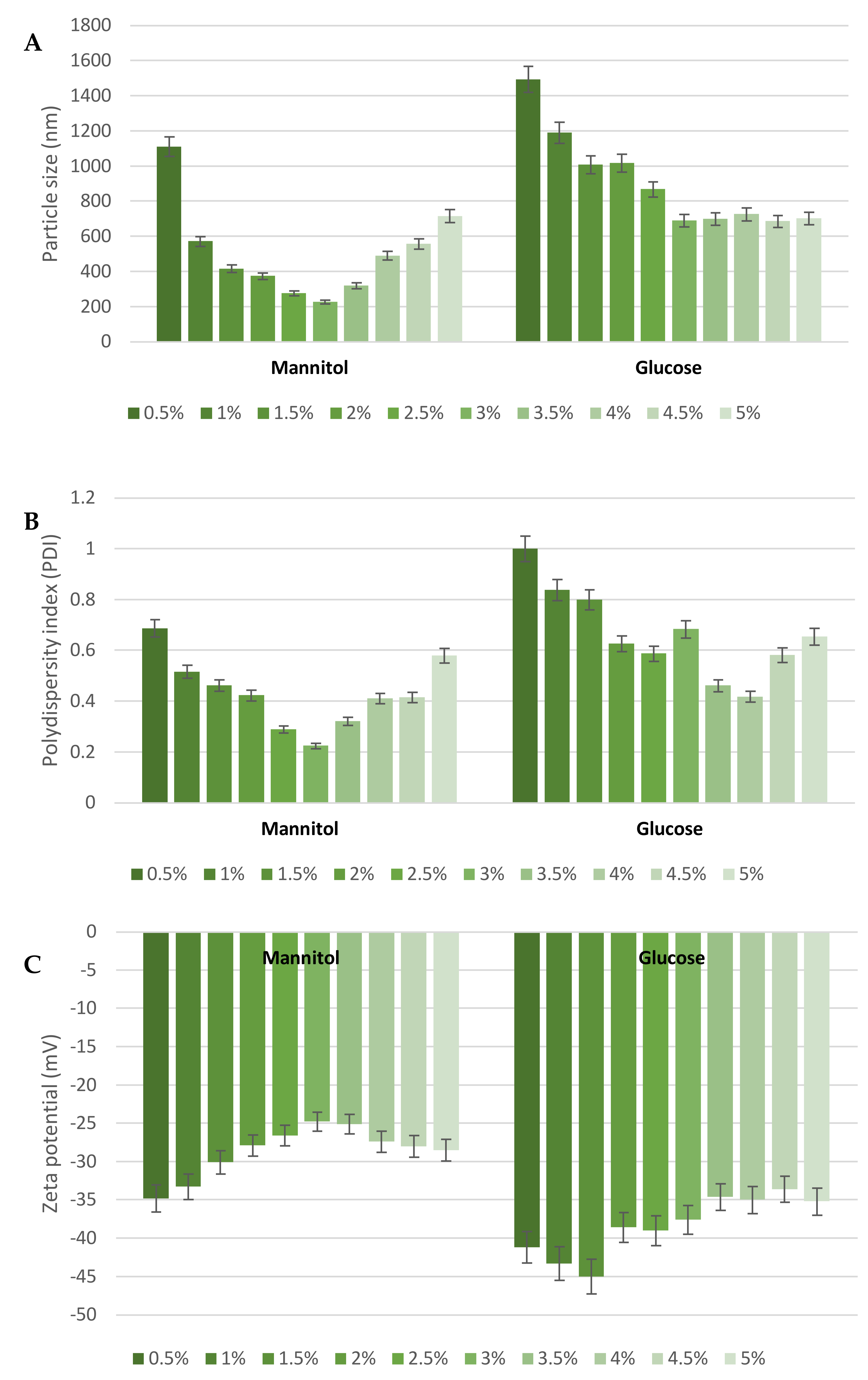
3.1. MGN–NLC Characterization
3.2. Lyophilization Effects
3.3. Corneal Permeability Assay
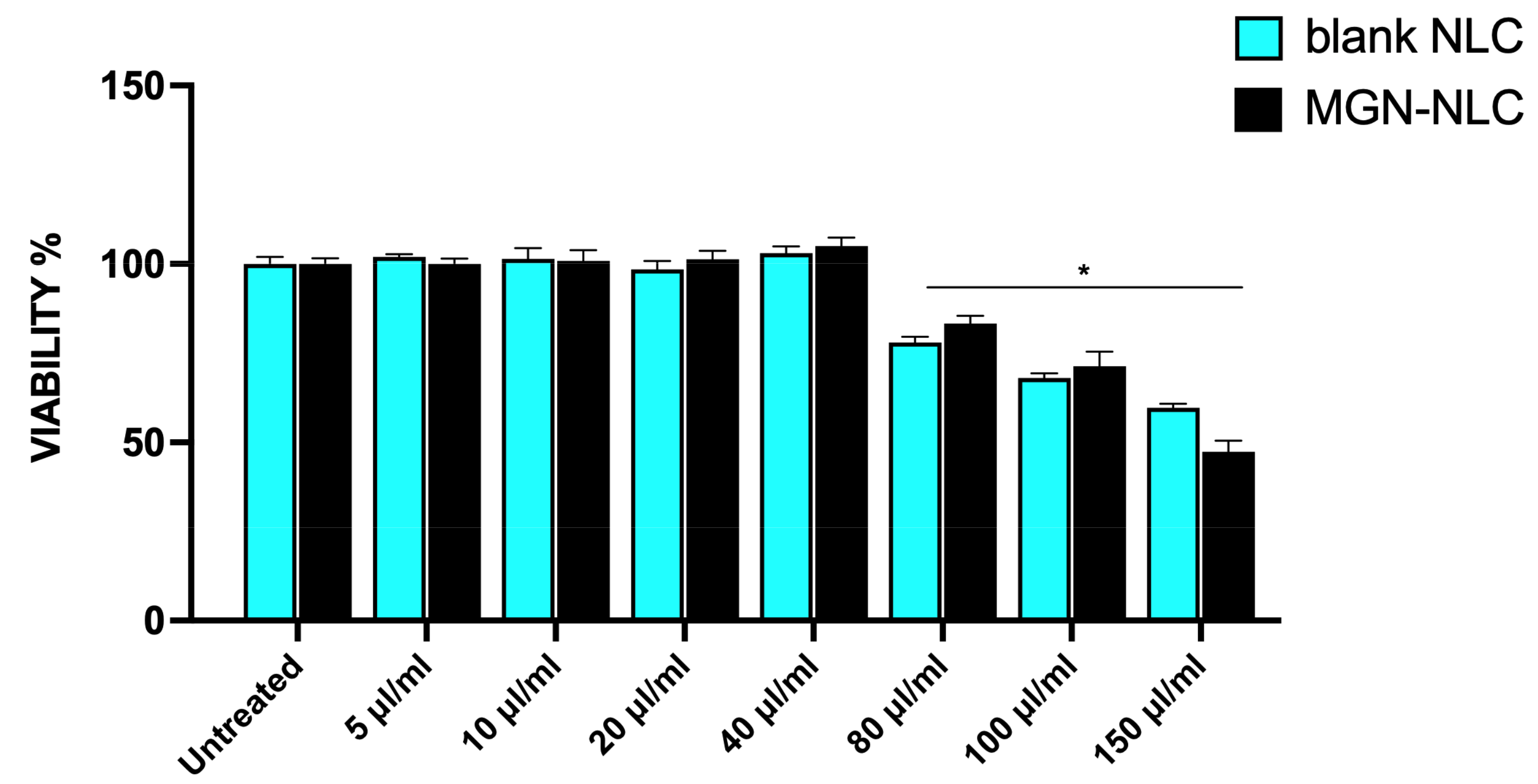
3.4. Effect of Blank NLC and MGN–NLC on Cell Viability
3.5. MGN–NLC Mitigates ROS Production
3.6. MGN–NLC Maintains Redox Balance by Increasing GSH Levels
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van der Pol, A.; Van Gilst, W.H.; Voors, A.A.; Van der Meer, P. Treating oxidative stress in heart failure: Past, present and future. Eur. J. Heart Fail. 2019, 21, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Homma, T.; Fujii, J. Application of Glutathione as Anti-Oxidative and Anti-Aging Drugs. Curr. Drug Metab. 2015, 16, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Ganea, E.; Harding, J.J. Glutathione-related enzymes and the eye. Curr. Eye Res. 2006, 31, 1–11. [Google Scholar] [CrossRef]
- Bonaccorso, A.; Pellitteri, R.; Ruozi, B.; Puglia, C.; Santonocito, D.; Pignatello, R.; Musumeci, T. Curcumin Loaded Polymeric vs. Lipid Nanoparticles: Antioxidant Effect on Normal and Hypoxic Olfactory Ensheathing Cells. Nanomaterials 2021, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol. Res. 2017, 39, 73–82. [Google Scholar] [CrossRef]
- Santonocito, D.; Raciti, G.; Campisi, A.; Sposito, G.; Panico, A.; Siciliano, E.A.; Sarpietro, M.G.; Damiani, E.; Puglia, C. Astaxanthin-Loaded Stealth Lipid Nanoparticles (AST-SSLN) as Potential Carriers for the Treatment of Alzheimer’s Disease: Formulation Development and Optimization. Nanomaterials 2021, 11, 391. [Google Scholar] [CrossRef]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids Health Dis. 2017, 16, 84. [Google Scholar] [CrossRef]
- Walia, V.; Chaudhary, S.K.; Kumar Sethiya, N. Therapeutic potential of mangiferin in the treatment of various neuropsychiatric and neurodegenerative disorders. Neurochem. Int. 2021, 143, 104939. [Google Scholar] [CrossRef]
- Sferrazzo, G.; Palmeri, R.; Restuccia, C.; Parafati, L.; Siracusa, L.; Spampinato, M.; Carota, G.; Distefano, A.; Di Rosa, M.; Tomasello, B.; et al. Mangifera indica L. Leaves as a Potential Food Source of Phenolic Compounds with Biological Activity. Antioxidants 2022, 11, 1313. [Google Scholar] [CrossRef]
- Matkowski, A.; Kuś, P.; Góralska, E.; Woźniak, D. Mangiferin-a bioactive xanthonoid, not only from mango and not just antioxidant. Mini. Rev. Med. Chem. 2013, 13, 439–455. [Google Scholar]
- Santonocito, D.; Vivero-Lopez, M.; Lauro, M.R.; Torrisi, C.; Castelli, F.; Sarpietro, M.G.; Puglia, C. Design of Nanotechnological Carriers for Ocular Delivery of Mangiferin: Preformulation Study. Molecules 2022, 27, 1328. [Google Scholar] [CrossRef]
- Sguizzato, M.; Ferrara, F.; Hallan, S.S.; Baldisserotto, A.; Drechsler, M.; Malatesta, M.; Costanzo, M.; Cortesi, R.; Puglia, C.; Valacchi, G.; et al. Ethosomes and Transethosomes for Mangiferin Transdermal Delivery. Antioxidants 2021, 10, 768. [Google Scholar] [CrossRef]
- Frasca, G.; Cardile, V.; Puglia, C.; Bonina, C.; Bonina, F. Gelatin Tannate Reduces the Proinflammatory Effects of Lipopo-lysaccharide in Human Intestinal Epithelial Cells. Clin. Exp. Gastroenterol. 2012, 5, 61. [Google Scholar]
- Puglia, C.; Blasi, P.; Ostacolo, C.; Sommella, E.; Bucolo, C.; Platania, C.B.M.; Romano, G.L.; Geraci, F.; Drago, F.; Santonocito, D.; et al. Innovative Nanoparticles Enhance N-Palmitoylethanolamide Intraocular Delivery. Front. Pharmacol. 2018, 9, 285. [Google Scholar] [CrossRef]
- Puglia, C.; Santonocito, D.; Ostacolo, C.; Sommella, E.M.; Campiglia, P.; Carbone, C.; Drago, F.; Pignatello, R.; Bucolo, C. Ocular Formulation Based on Palmitoylethanolamide-Loaded Nanostructured Lipid Carriers: Technological and Pharmacological Profile. Nanomaterials 2020, 10, 287. [Google Scholar] [CrossRef]
- Puglia, C.; Santonocito, D.; Romeo, G.; Intagliata, S.; Romano, G.L.; Strettoi, E.; Novelli, E.; Ostacolo, C.; Campiglia, P.; Sommella, E.M.; et al. Lipid Nanoparticles Traverse Non-Corneal Path to Reach the Posterior Eye Segment: In Vivo Evidence. Molecules 2021, 26, 4673. [Google Scholar] [CrossRef]
- Carbone, C.; Fuochi, V.; Zielińska, A.; Musumeci, T.; Souto, E.B.; Bonaccorso, A.; Puglia, C.; Petronio Petronio, G.; Furneri, P.M. Dual-drugs delivery in solid lipid nanoparticles for the treatment of Candida albicans mycosis. Colloids Surf. B Biointerfaces 2020, 186, 110705. [Google Scholar] [CrossRef]
- Puglia, C.; Santonocito, D.; Bonaccorso, A.; Musumeci, T.; Ruozi, B.; Pignatello, R.; Carbone, C.; Parenti, C.; Chiechio, S. Lipid Nanoparticle Inclusion Prevents Capsaicin-Induced TRPV1 Defunctionalization. Pharmaceutics 2020, 12, 339. [Google Scholar] [CrossRef]
- Liparulo, A.; Esposito, R.; Santonocito, D.; Muñoz-Ramírez, A.; Spaziano, G.; Bruno, F.; Xiao, J.; Puglia, C.; Filosa, R.; Berrino, L.; et al. Formulation and Characterization of Solid Lipid Nanoparticles Loading RF22-c, a Potent and Selective 5-LO Inhibitor, in a Monocrotaline-Induced Model of Pulmonary Hypertension. Front. Pharmacol. 2020, 11, 83. [Google Scholar] [CrossRef]
- Esposito, E.; Drechsler, M.; Mariani, P.; Panico, A.M.; Cardile, V.; Crascì, L.; Carducci, F.; Graziano, A.C.E.; Cortesi, R.; Puglia, C. Nanostructured lipid dispersions for topical administration of crocin, a potent antioxidant from saffron (Crocus sativus L.). Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 71, 669–677. [Google Scholar] [CrossRef]
- Santonocito, D.; Sarpietro, M.G.; Carbone, C.; Panico, A.; Campisi, A.; Siciliano, E.A.; Sposito, G.; Castelli, F.; Puglia, C. Curcumin Containing PEGylated Solid Lipid Nanoparticles for Systemic Administration: A Preliminary Study. Molecules 2020, 25, 2991. [Google Scholar] [CrossRef] [PubMed]
- Varela-Garcia, A.; Concheiro, A.; Alvarez-Lorenzo, C. Cytosine-functionalized bioinspired hydrogels for ocular delivery of antioxidant transferulic acid. Biomater. Sci. 2020, 8, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Naveen, P.; Lingaraju, H.B.; Shyam Prasad, K. Rapid Development and Validation of Improved Reversed-Phase High-performance Liquid Chromatography Method for the Quantification of Mangiferin, a Polyphenol Xanthone Glycoside in Mangifera indica. Pharmacognosy Res. 2017, 9, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Vivero-Lopez, M.; Muras, A.; Silva, D.; Serro, A.P.; Otero, A.; Concheiro, A.; Alvarez-Lorenzo, C. Resveratrol-Loaded Hydrogel Contact Lenses with Antioxidant and Antibiofilm Performance. Pharmaceutics 2021, 13, 532. [Google Scholar] [CrossRef]
- Varela-Garcia, A.; Concheiro, A.; Alvarez-Lorenzo, C. Soluplus micelles for acyclovir ocular delivery: Formulation and cornea and sclera permeability. Int. J. Pharm. 2018, 552, 39–47. [Google Scholar] [CrossRef]
- Jung, W.K.; Park, S.B.; Yu, H.Y.; Kim, Y.H.; Kim, J. Effect of Esculetin on Tert-Butyl Hydroperoxide-Induced Oxidative Injury in Retinal Pigment Epithelial Cells In Vitro. Molecules 2022, 27, 8970. [Google Scholar] [CrossRef]
- Yadav, M.; Schiavone, N.; Guzman-Aranguez, A.; Giansanti, F.; Papucci, L.; Perez de Lara, M.J.; Singh, M.; Kaur, I.P. Atorvastatin-loaded solid lipid nanoparticles as eye drops: Proposed treatment option for age-related macular degeneration (AMD). Drug Deliv. Transl. Res. 2020, 10, 919–944. [Google Scholar] [CrossRef]
- Shimizu, H.; Takayama, K.; Yamada, K.; Suzumura, A.; Sato, T.; Nishio, Y.; Ito, M.; Ushida, H.; Nishiguchi, K.M.; Takeuchi, M.; et al. Dimethyl Fumarate Protects Retinal Pigment Epithelium from Blue Light-Induced Oxidative Damage via the Nrf2 Pathway. Antioxidants 2022, 12, 45. [Google Scholar] [CrossRef]
- Spampinato, M.; Carota, G.; Sferrazzo, G.; Fuochi, V.; Distefano, A.; Ronsisvalle, S.; Sipala, F.; Giuffrida, R.; Furneri, P.M.; Di Rosa, M.; et al. Effects of Mangiferin on LPS-Induced Inflammation and SARS-CoV-2 Viral Adsorption in Human Lung Cells. Pharmaceutics 2022, 14, 2845. [Google Scholar] [CrossRef]
- Li Volti, G.; Musumeci, T.; Pignatello, R.; Murabito, P.; Barbagallo, I.; Carbone, C.; Gullo, A.; Puglisi, G. Antioxidant potential of different melatonin-loaded nanomedicines in an experimental model of sepsis. Exp. Biol. Med. 2012, 237, 670–677. [Google Scholar] [CrossRef]
- Puglia, C.; Tropea, S.; Rizza, L.; Santagati, N.A.; Bonina, F. In vitro percutaneous absorption studies and in vivo evaluation of anti-inflammatory activity of essential fatty acids (EFA) from fish oil extracts. Int. J. Pharm. 2005, 299, 41–48. [Google Scholar] [CrossRef]
- Schwarz, C.; Mehnert, W. Freeze- drying of drug- free and drug- loaded solid lipid nanoparticles (SLNs). Int. J. Pharm. 1997, 157, 171–179. [Google Scholar] [CrossRef]
- Abdelwahed, W. Lyophilization of solid lipid nanoparticles for brain targeting. Int. J. Pharm. Pharm. Sci. 2015, 7, 381–385. [Google Scholar]
- Ranpise, N.S.; Korabu, S.S.; Ghodake, V.N. Second generation lipid nanoparticles (NLC) as an oral drug carrier for delivery of lercanidipine hydrochloride. Colloids Surf. B Biointerfaces 2014, 116, 81–87. [Google Scholar] [CrossRef]
- Alvarez-Rivera, F.; Fernández-Villanueva, D.; Concheiro, A.; Alvarez-Lorenzo, C. α-Lipoic Acid in Soluplus(®) Polymeric Nanomicelles for Ocular Treatment of Diabetes-Associated Corneal Diseases. J. Pharm. Sci. 2016, 105, 2855–2863. [Google Scholar] [CrossRef]
- Acosta, J.; Sevilla, I.; Salomón, S.; Nuevas, L.; Romero, A.; Amaro, D. Determination of mangiferin solubility in solvents used in the biopharmaceutical industry. J. Pharm. Pharmacogn. Res. 2016, 4, 49–53. [Google Scholar]
- Lorenzo-Veiga, B.; Sigurdsson, H.H.; Loftsson, T.; Alvarez-Lorenzo, C. Cyclodextrin–amphiphilic Copolymer Supramolecular Assemblies for the Ocular Delivery of Natamycin. Nanomaterials 2019, 9, 745. [Google Scholar] [CrossRef]
- Sferrazzo, G.; Palmeri, R.; Vanella, L.; Parafati, L.; Ronsisvalle, S.; Biondi, A.; Basile, F.; Li Volti, G.; Barbagallo, I. Mangifera indica L. Leaf Extract Induces Adiponectin and Regulates Adipogenesis. Int. J. Mol. Sci. 2019, 20, 3211. [Google Scholar] [CrossRef]
- Barbagallo, I.; Marrazzo, G.; Frigiola, A.; Zappala, A.; Li Volti, G. Role of carbon monoxide in vascular diseases. Curr. Pharm. Biotechnol. 2012, 13, 787–796. [Google Scholar] [CrossRef]
- Salomone, F.; Barbagallo, I.; Godos, J.; Lembo, V.; Currenti, W.; Cinà, D.; Avola, R.; D’Orazio, N.; Morisco, F.; Galvano, F.; et al. Silibinin Restores NAD+ Levels and Induces the SIRT1/AMPK Pathway in Non-Alcoholic Fatty Liver. Nutrients 2017, 9, 1086. [Google Scholar] [CrossRef]
- Cho, H.M.; Lee, S.J.; Choung, S.Y. Protective effects of Panax ginseng berry extract on blue light-induced retinal damage in ARPE-19 cells and mouse retina. J. Ginseng Res. 2023, 47, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Sellamuthu, P.S.; Arulselvan, P.; Muniappan, B.P.; Fakurazi, S.; Kandasamy, M. Mangiferin from Salacia chinensis prevents oxidative stress and protects pancreatic β-cells in streptozotocin-induced diabetic rats. J. Med. Food 2013, 16, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, S.; Jainu, M.; Sabitha, K.E.; Devi, C.S. Role of mangiferin on biochemical alterations and antioxidant status in isoproterenol-induced myocardial infarction in rats. J. Ethnopharmacol. 2006, 107, 126–133. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santonocito, D.; Barbagallo, I.; Distefano, A.; Sferrazzo, G.; Vivero-Lopez, M.; Sarpietro, M.G.; Puglia, C. Nanostructured Lipid Carriers Aimed to the Ocular Delivery of Mangiferin: In Vitro Evidence. Pharmaceutics 2023, 15, 951. https://doi.org/10.3390/pharmaceutics15030951
Santonocito D, Barbagallo I, Distefano A, Sferrazzo G, Vivero-Lopez M, Sarpietro MG, Puglia C. Nanostructured Lipid Carriers Aimed to the Ocular Delivery of Mangiferin: In Vitro Evidence. Pharmaceutics. 2023; 15(3):951. https://doi.org/10.3390/pharmaceutics15030951
Chicago/Turabian StyleSantonocito, Debora, Ignazio Barbagallo, Alfio Distefano, Giuseppe Sferrazzo, Maria Vivero-Lopez, Maria Grazia Sarpietro, and Carmelo Puglia. 2023. "Nanostructured Lipid Carriers Aimed to the Ocular Delivery of Mangiferin: In Vitro Evidence" Pharmaceutics 15, no. 3: 951. https://doi.org/10.3390/pharmaceutics15030951
APA StyleSantonocito, D., Barbagallo, I., Distefano, A., Sferrazzo, G., Vivero-Lopez, M., Sarpietro, M. G., & Puglia, C. (2023). Nanostructured Lipid Carriers Aimed to the Ocular Delivery of Mangiferin: In Vitro Evidence. Pharmaceutics, 15(3), 951. https://doi.org/10.3390/pharmaceutics15030951








