Plausible Role of Stem Cell Types for Treating and Understanding the Pathophysiology of Depression
Abstract
1. Introduction
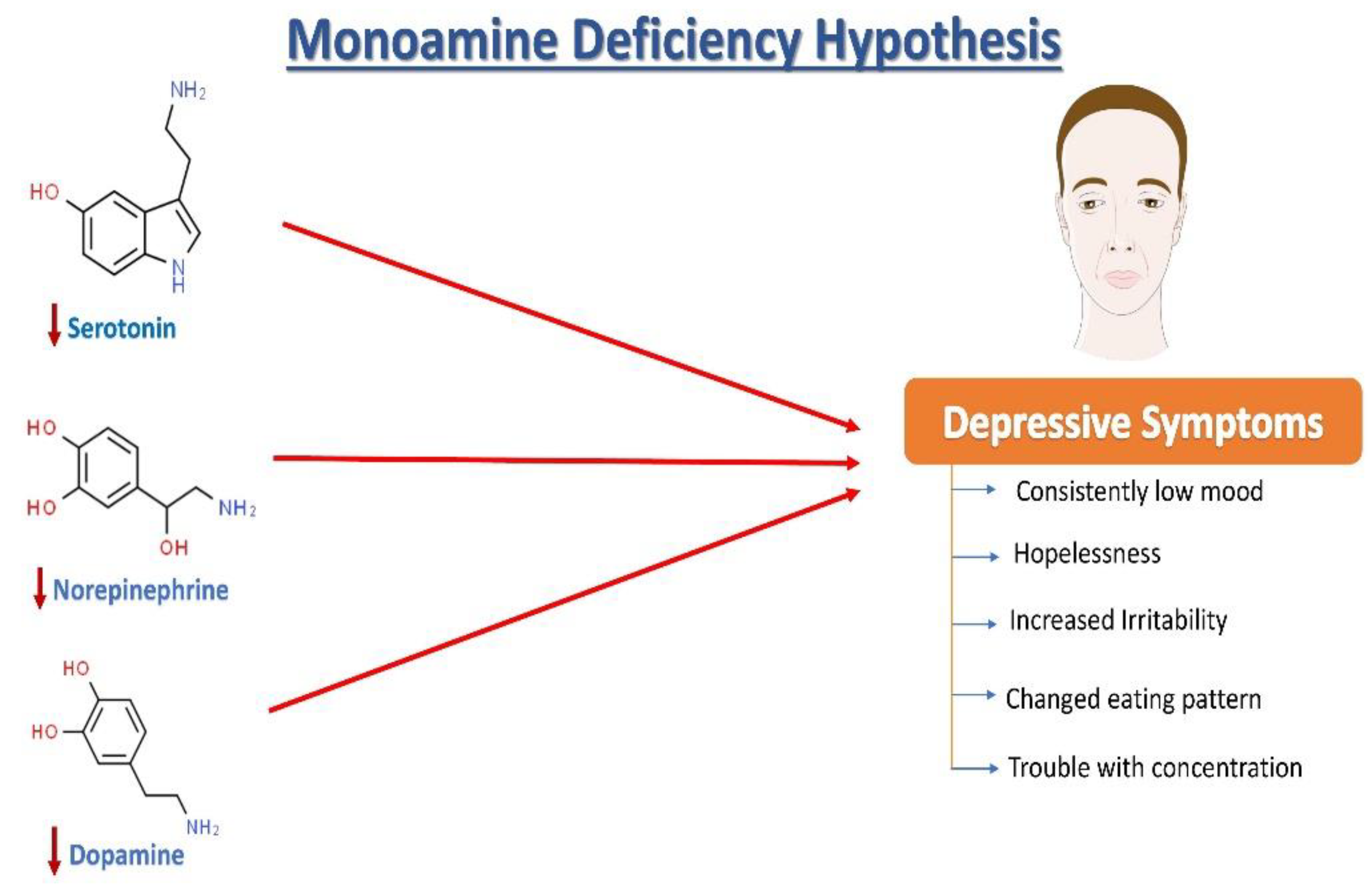
2. Neurochemistry of Depression: The Monoamine Hypothesis
3. Growth Factors Involved in Depression
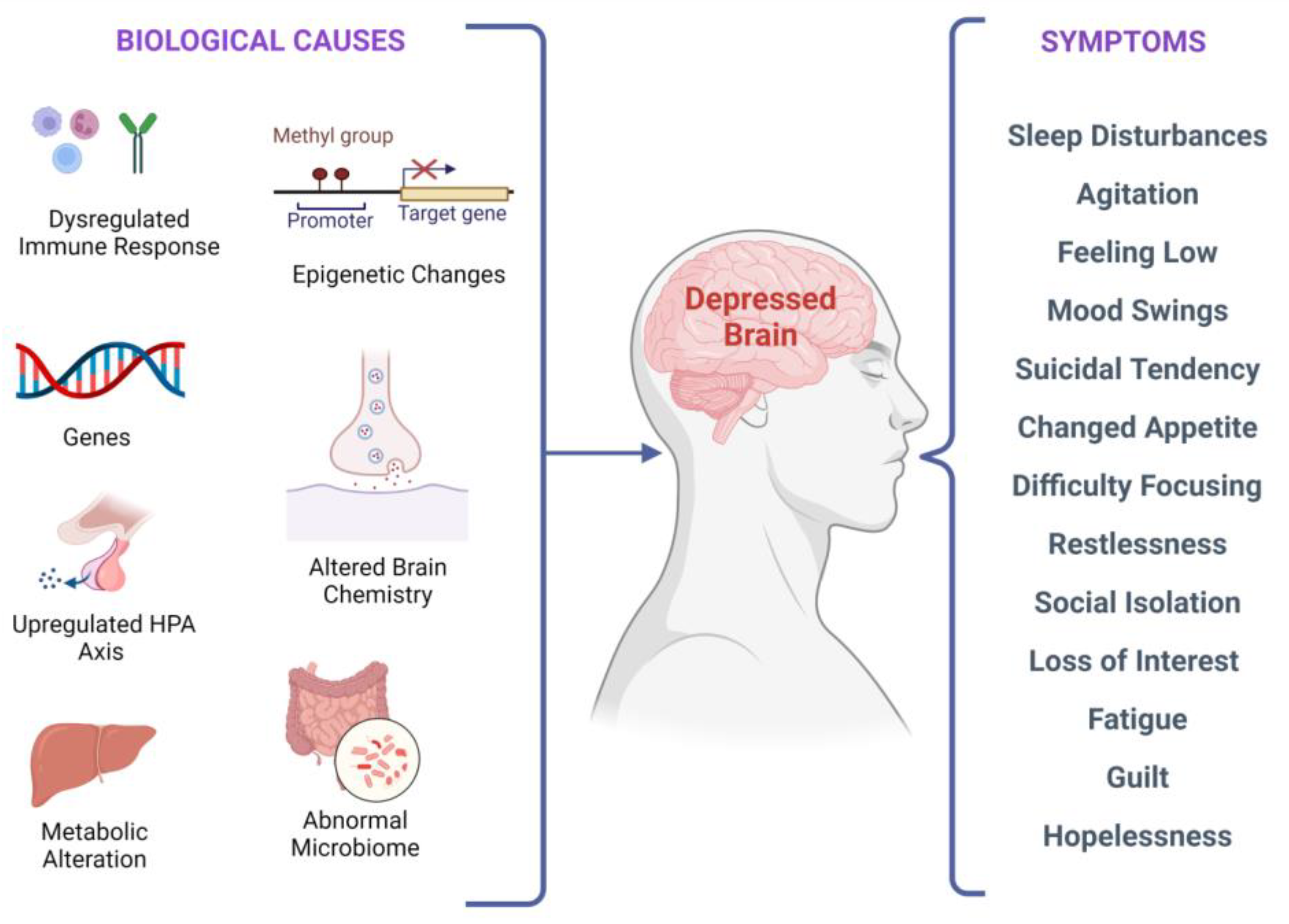
4. Depression as a Neurodegenerative Disorder
5. Stem Cells: An Insight
6. Intranasal Route for Stem Cells Administration
7. Utilizing the Induced Pluripotent Stem Cells (iPSCs) to Understand Depression Pathophysiology
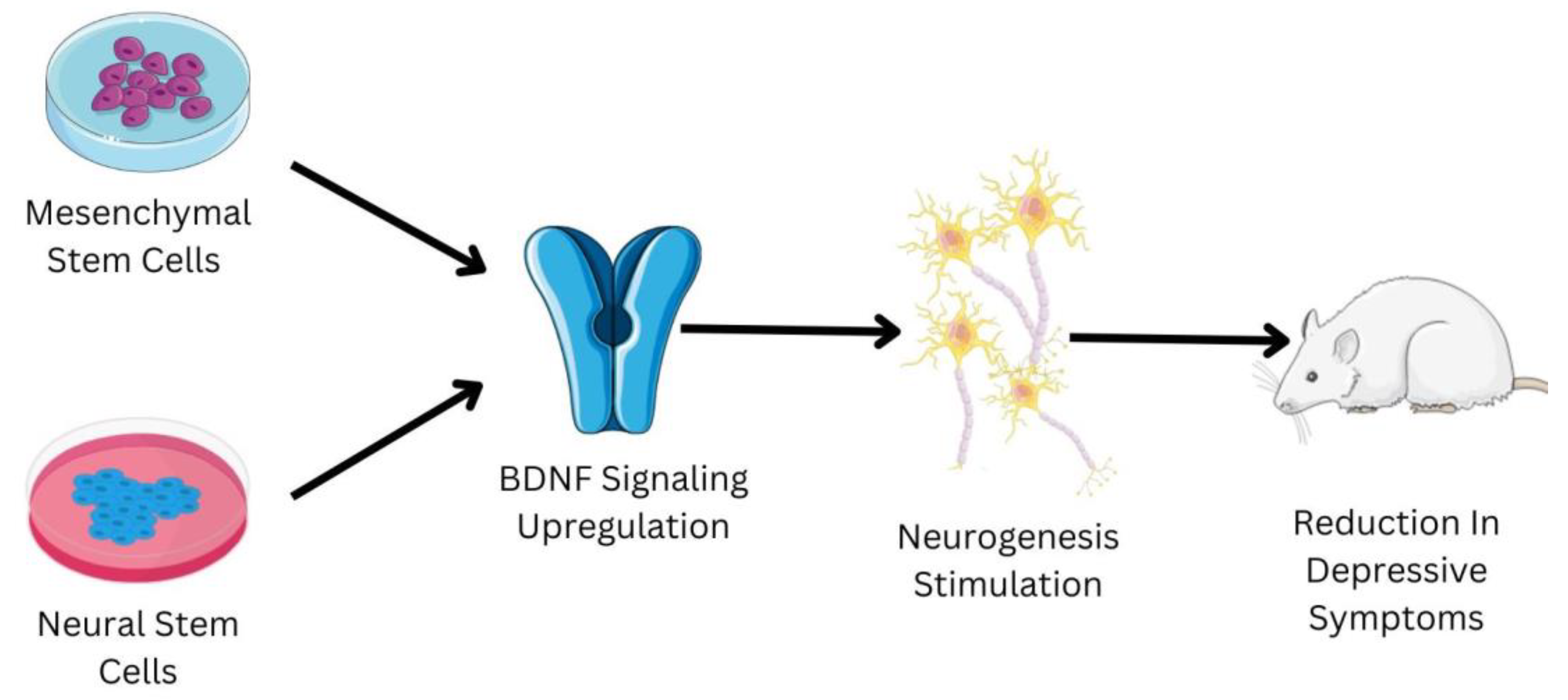
8. Mesenchymal Stem Cells and Depression: A Therapeutic Mechanism
9. Neural Stem Cells and Depression
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5HT | 5-hydroxytryptamine |
| ADSCs | Adipose-derived mesenchymal stem cells |
| BBB | Blood brain barrier |
| BDNF | Brain-derived neurotrophic factor |
| CNS | Central nervous system |
| CRP | C-reactive protein |
| CSF | Cerebrospinal fluid |
| DA | Dopamine |
| DAT | Dopamine transporter |
| DTI | Diffusion tensor imaging |
| BBB | Blood-brain barrier |
| EAAT | Excitatory amino acid transporter |
| FGF-2 | Fibroblast growth factor-2 |
| fMRI | functional MRI |
| FSL | Flinders sensitive line |
| FST | Forced swimming test |
| GDNF | Glial cell line-derived neurotrophic factor |
| GNPs | Gold nanoparticles |
| hiPSCs | Human iPSCs |
| HUC-MSCs | Human umbilical cord mesenchymal stem cells |
| iPSCs | Induced pluripotent stem cells |
| LC | Locus coeruleus |
| MDD | Major depressive disorder |
| MI | Myocardial Infarction |
| MRI | Magnetic resonance imaging |
| MSCs | Mesenchymal stem cells |
| NE | Norepinephrine |
| NETs | Norepinephrine transporters |
| NG | Neurogenesis |
| NGF | Nerve growth factor |
| NSCs | Neural stem cells |
| NT | 3-Neurotrophin-3 |
| PGE2 | Plasma prostaglandin E2 |
| PTSD | Post-traumatic stress disorder |
| SNRIs | Serotonin-norepinephrine reuptake inhibitors |
| SPT | Sucrose preference test |
| SSRIs | Selective serotonin reuptake inhibitors |
| TLR | Toll-like receptor |
| TRD | Treatment-resistant depression |
| TST | Tail suspension test |
| VGF | nonacronymic |
References
- Maj, M. When Does Depression Become a Mental Disorder? Br. J. Psychiatry 2011, 199, 85–86. [Google Scholar] [CrossRef] [PubMed]
- Lang, U.E.; Borgwardt, S. Molecular Mechanisms of Depression: Perspectives on New Treatment Strategies. Cell Physiol. Biochem. 2013, 31, 761–777. [Google Scholar] [CrossRef] [PubMed]
- Ramasubbu, R.; Patten, S.B. Effect of Depression on Stroke Morbidity and Mortality. Can. J. Psychiatry 2003, 48, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Van der Kooy, K.; van Hout, H.; Marwijk, H.; Marten, H.; Stehouwer, C.; Beekman, A. Depression and the Risk for Cardiovascular Diseases: Systematic Review and Meta Analysis. Int. J. Geriat. Psychiatry 2007, 22, 613–626. [Google Scholar] [CrossRef]
- Green, R.C.; Cupples, L.A.; Kurz, A.; Auerbach, S.; Go, R.; Sadovnick, D.; Duara, R.; Kukull, W.A.; Chui, H.; Edeki, T.; et al. Depression as a Risk Factor for Alzheimer Disease: The MIRAGE Study. Arch. Neurol. 2003, 60, 753. [Google Scholar] [CrossRef]
- Hesdorffer, D.C.; Hauser, W.A.; Annegers, J.F.; Cascino, G. Major Depression Is a Risk Factor for Seizures in Older Adults. Ann. Neurol. 2000, 47, 246–249. [Google Scholar] [CrossRef]
- Nouwen, A.; Lloyd, C.E.; Pouwer, F. Depression and Type 2 Diabetes Over the Lifespan: A Meta-Analysis. Diabetes Care 2009, 32, e56. [Google Scholar] [CrossRef]
- Penninx, B.W.J.H.; Guralnik, J.M.; Havlik, R.J.; Pahor, M.; Ferrucci, L.; Cerhan, J.R.; Wallace, R.B. Chronically Depressed Mood and Cancer Risk in Older Persons. JNCI J. Natl. Cancer Inst. 1998, 90, 1888–1893. [Google Scholar] [CrossRef]
- Kaur, J.; Ghosh, S.; Singh, P.; Dwivedi, A.K.; Sahani, A.K.; Sinha, J.K. Cervical Spinal Lesion, Completeness of Injury, Stress, and Depression Reduce the Efficiency of Mental Imagery in People With Spinal Cord Injury. Am. J. Phys. Med. Rehabil. 2022, 101, 513–519. [Google Scholar] [CrossRef]
- Maniam, J.; Antoniadis, C.P.; Youngson, N.A.; Sinha, J.K.; Morris, M.J. Sugar Consumption Produces Effects Similar to Early Life Stress Exposure on Hippocampal Markers of Neurogenesis and Stress Response. Front. Mol. Neurosci. 2015, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Rush, A.J.; Trivedi, M.H.; Wisniewski, S.R.; Nierenberg, A.A.; Stewart, J.W.; Warden, D.; Niederehe, G.; Thase, M.E.; Lavori, P.W.; Lebowitz, B.D.; et al. Acute and Longer-Term Outcomes in Depressed Outpatients Requiring One or Several Treatment Steps: A STAR*D Report. Am. J. Psychiatry 2006, 163, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Akil, H.; Gordon, J.; Hen, R.; Javitch, J.; Mayberg, H.; McEwen, B.; Meaney, M.J.; Nestler, E.J. Treatment Resistant Depression: A Multi-Scale, Systems Biology Approach. Neurosci. Biobehav. Rev. 2018, 84, 272–288. [Google Scholar] [CrossRef] [PubMed]
- Salzman, C.; Wong, E.; Wright, B.C. Drug and ECT Treatment of Depression in the Elderly, 1996–2001: A Literature Review. Biol. Psychiatry 2002, 52, 265–284. [Google Scholar] [CrossRef] [PubMed]
- Keller, M.B.; Hirschfeld, R.M.A.; Demyttenaere, K.; Baldwin, D.S. Optimizing Outcomes in Depression: Focus on Antidepressant Compliance. Int. Clin. Psychopharmacol. 2002, 17, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J.; Carlezon, W.A. The Mesolimbic Dopamine Reward Circuit in Depression. Biol. Psychiatry 2006, 59, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Roy, A. Cerebrospinal Fluid Monoamine Metabolites and Suicidal Behavior in Depressed Patients: A 5-Year Follow-up Study. Arch. Gen. Psychiatry 1989, 46, 609. [Google Scholar] [CrossRef]
- Klimek, V.; Stockmeier, C.; Overholser, J.; Meltzer, H.Y.; Kalka, S.; Dilley, G.; Ordway, G.A. Reduced Levels of Norepinephrine Transporters in the Locus Coeruleus in Major Depression. J. Neurosci. 1997, 17, 8451–8458. [Google Scholar] [CrossRef]
- Marshe, V.S.; Maciukiewicz, M.; Rej, S.; Tiwari, A.K.; Sibille, E.; Blumberger, D.M.; Karp, J.F.; Lenze, E.J.; Reynolds, C.F.; Kennedy, J.L.; et al. Norepinephrine Transporter Gene Variants and Remission From Depression With Venlafaxine Treatment in Older Adults. Am. J. Psychiatry 2017, 174, 468–475. [Google Scholar] [CrossRef]
- Dunn, A.J. Effects of Cytokines and Infections on Brain Neurochemistry. Clin. Neurosci. Res. 2006, 6, 52–68. [Google Scholar] [CrossRef]
- Anacker, C.; Cattaneo, A.; Musaelyan, K.; Zunszain, P.A.; Horowitz, M.; Molteni, R.; Luoni, A.; Calabrese, F.; Tansey, K.; Gennarelli, M.; et al. Role for the Kinase SGK1 in Stress, Depression, and Glucocorticoid Effects on Hippocampal Neurogenesis. Proc. Natl. Acad. Sci. USA 2013, 110, 8708–8713. [Google Scholar] [CrossRef]
- Jokinen, J.; Nordström, A.-L.; Nordström, P. The Relationship Between CSF HVA/5-HIAA Ratio and Suicide Intent in Suicide Attempters. Arch. Suicide Res. 2007, 11, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Pizzagalli, D.A.; Berretta, S.; Wooten, D.; Goer, F.; Pilobello, K.T.; Kumar, P.; Murray, L.; Beltzer, M.; Boyer-Boiteau, A.; Alpert, N.; et al. Assessment of Striatal Dopamine Transporter Binding in Individuals With Major Depressive Disorder: In Vivo Positron Emission Tomography and Postmortem Evidence. JAMA Psychiatry 2019, 76, 854. [Google Scholar] [CrossRef]
- Cassano, P.; Lattanzi, L.; Fava, M.; Navari, S.; Battistini, G.; Abelli, M.; Cassano, G.B. Ropinirole in Treatment-Resistant Depression: A 16-Week Pilot Study. Can. J. Psychiatry 2005, 50, 357–360. [Google Scholar] [CrossRef]
- Descarries, L.; Watkins, K.C.; Garcia, S.; Beaudet, A. The Serotonin Neurons in Nucleus Raphe Dorsalis of Adult Rat: A Light and Electron Microscope Radioautographic Study. J. Comp. Neurol. 1982, 207, 239–254. [Google Scholar] [CrossRef] [PubMed]
- Bunin, M.A.; Wightman, R.M. Quantitative Evaluation of 5-Hydroxytryptamine (Serotonin) Neuronal Release and Uptake: An Investigation of Extrasynaptic Transmission. J. Neurosci. 1998, 18, 4854–4860. [Google Scholar] [CrossRef] [PubMed]
- Steinbusch, H.W.M. Distribution of Serotonin-Immunoreactivity in the Central Nervous System of the Rat—Cell Bodies and Terminals. Neuroscience 1981, 6, 557–618. [Google Scholar] [CrossRef] [PubMed]
- Mann, J. Role of the Serotonergic System in the Pathogenesis of Major Depression and Suicidal Behavior. Neuropsychopharmacology 1999, 21, 99S–105S. [Google Scholar] [CrossRef] [PubMed]
- Chaouloff, F. Serotonin and Stress. Neuropsychopharmacology 1999, 21, 28S–32S. [Google Scholar] [CrossRef]
- Andrews, P.W.; Bharwani, A.; Lee, K.R.; Fox, M.; Thomson, J.A. Is Serotonin an Upper or a Downer? The Evolution of the Serotonergic System and Its Role in Depression and the Antidepressant Response. Neurosci. Biobehav. Rev. 2015, 51, 164–188. [Google Scholar] [CrossRef]
- Bot, M.; Chan, M.K.; Jansen, R.; Lamers, F.; Vogelzangs, N.; Steiner, J.; Leweke, F.M.; Rothermundt, M.; Cooper, J.; Bahn, S.; et al. Serum Proteomic Profiling of Major Depressive Disorder. Transl. Psychiatry 2015, 5, e599. [Google Scholar] [CrossRef]
- Quintana, J. Platelet Serotonin and Plasma Tryptophan Decreases in Endogenous Depression. Clinical, Therapeutic, and Biological Correlations. J. Affect. Disord. 1992, 24, 55–62. [Google Scholar] [CrossRef]
- Park, C.; Rosenblat, J.D.; Brietzke, E.; Pan, Z.; Lee, Y.; Cao, B.; Zuckerman, H.; Kalantarova, A.; McIntyre, R.S. Stress, Epigenetics and Depression: A Systematic Review. Neurosci. Biobehav. Rev. 2019, 102, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Sinha, J.K.; Raghunath, M. “Obesageing”: Linking Obesity & Ageing. Indian J. Med. Res. 2019, 149, 610–615. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.; MacQueen, G. An Update on Regional Brain Volume Differences Associated with Mood Disorders. Curr. Opin. Psychiatry 2006, 19, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Videbech, P. Hippocampal Volume and Depression: A Meta-Analysis of MRI Studies. Am. J. Psychiatry 2004, 161, 1957–1966. [Google Scholar] [CrossRef]
- Mishra, P.; Mittal, A.K.; Kalonia, H.; Madan, S.; Ghosh, S.; Sinha, J.K.; Rajput, S.K. SIRT1 Promotes Neuronal Fortification in Neurodegenerative Diseases through Attenuation of Pathological Hallmarks and Enhancement of Cellular Lifespan. Curr. Neuropharmacol. 2021, 19, 1019–1037. [Google Scholar] [CrossRef] [PubMed]
- Moroi, K.; Sato, T. Comparison between Procaine and Isocarboxazid Metabolism in Vitro by a Liver Microsomal Amidase-Esterase. Biochem. Pharmacol. 1975, 24, 1517–1521. [Google Scholar] [CrossRef]
- Pittenger, C.; Duman, R.S. Stress, Depression, and Neuroplasticity: A Convergence of Mechanisms. Neuropsychopharmacology 2008, 33, 88–109. [Google Scholar] [CrossRef]
- Aydemir, O.; Deveci, A.; Taneli, F. The Effect of Chronic Antidepressant Treatment on Serum Brain-Derived Neurotrophic Factor Levels in Depressed Patients: A Preliminary Study. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2005, 29, 261–265. [Google Scholar] [CrossRef]
- Ricken, R.; Adli, M.; Lange, C.; Krusche, E.; Stamm, T.J.; Gaus, S.; Koehler, S.; Nase, S.; Bschor, T.; Richter, C.; et al. Brain-Derived Neurotrophic Factor Serum Concentrations in Acute Depressive Patients Increase During Lithium Augmentation of Antidepressants. J. Clin. Psychopharmacol. 2013, 33, 806–809. [Google Scholar] [CrossRef]
- Bauer, M.; Adli, M.; Bschor, T.; Pilhatsch, M.; Pfennig, A.; Sasse, J.; Schmid, R.; Lewitzka, U. Lithium’s Emerging Role in the Treatment of Refractory Major Depressive Episodes: Augmentation of Antidepressants. Neuropsychobiology 2010, 62, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Coradduzza, D.; Garroni, G.; Congiargiu, A.; Balzano, F.; Cruciani, S.; Sedda, S.; Nivoli, A.; Maioli, M. MicroRNAs, Stem Cells in Bipolar Disorder, and Lithium Therapeutic Approach. Int. J. Mol. Sci. 2022, 23, 10489. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.C.; Fatima, M. Direct and Indirect Evidences of BDNF and NGF as Key Modulators in Depression: Role of Antidepressants Treatment. Int. J. Neurosci. 2019, 129, 283–296. [Google Scholar] [CrossRef]
- de Miranda, A.S.; de Barros, J.L.V.M.; Teixeira, A.L. Is Neurotrophin-3 (NT-3): A Potential Therapeutic Target for Depression and Anxiety? Expert Opin. Ther. Targets 2020, 24, 1225–1238. [Google Scholar] [CrossRef]
- Diniz, B.S.; Teixeira, A.L.; Miranda, A.S.; Talib, L.L.; Gattaz, W.F.; Forlenza, O.V. Circulating Glial-Derived Neurotrophic Factor Is Reduced in Late-Life Depression. J. Psychiatr. Res. 2012, 46, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.J.; Choudary, P.V.; Neal, C.R.; Li, J.Z.; Vawter, M.P.; Tomita, H.; Lopez, J.F.; Thompson, R.C.; Meng, F.; Stead, J.D.; et al. Dysregulation of the Fibroblast Growth Factor System in Major Depression. Proc. Natl. Acad. Sci. USA 2004, 101, 15506–15511. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.-M. A Role for Akt and Glycogen Synthase Kinase-3 as Integrators of Dopamine and Serotonin Neurotransmission in Mental Health. J. Psychiatry Neurosci. 2012, 37, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Miskowiak, K.W.; Vinberg, M.; Harmer, C.J.; Ehrenreich, H.; Knudsen, G.M.; Macoveanu, J.; Hansen, A.R.; Paulson, O.B.; Siebner, H.R.; Kessing, L.V. Effects of Erythropoietin on Depressive Symptoms and Neurocognitive Deficits in Depression and Bipolar Disorder. Trials 2010, 11, 97. [Google Scholar] [CrossRef]
- Eden Evins, A.; Demopulos, C.; Yovel, I.; Culhane, M.; Ogutha, J.; Grandin, L.D.; Nierenberg, A.A.; Sachs, G.S. Inositol Augmentation of Lithium or Valproate for Bipolar Depression. Bipolar Disord. 2006, 8, 168–174. [Google Scholar] [CrossRef]
- Cattaneo, A.; Sesta, A.; Calabrese, F.; Nielsen, G.; Riva, M.A.; Gennarelli, M. The Expression of VGF Is Reduced in Leukocytes of Depressed Patients and It Is Restored by Effective Antidepressant Treatment. Neuropsychopharmacology 2010, 35, 1423–1428. [Google Scholar] [CrossRef]
- Ahmad, F.; Sachdeva, P.; Sarkar, J.; Izhaar, R. Circadian Dysfunction and Alzheimer’s Disease—An Updated Review. Aging Med. 2022, 00:1–11. [Google Scholar] [CrossRef]
- Ahmad, F.; Sachdeva, P. Critical Appraisal on Mitochondrial Dysfunction in Alzheimer’s Disease. Aging Med. 2022, 5, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Mukerjee, N.; Al-Khafaji, K.; Maitra, S.; Suhail Wadi, J.; Sachdeva, P.; Ghosh, A.; Buchade, R.S.; Chaudhari, S.Y.; Jadhav, S.B.; Das, P.; et al. Recognizing Novel Drugs against Keap1 in Alzheimer’s Disease Using Machine Learning Grounded Computational Studies. Front. Mol. Neurosci. 2022, 15, 1036552. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, B.; Sachdeva, P. MXenes for Neurodegenerative Disorders. Mater. Today: Proc. 2023, 73, 294–296. [Google Scholar] [CrossRef]
- Madar, I.H.; Sultan, G.; Tayubi, I.A.; Hasan, A.N.; Pahi, B.; Rai, A.; Sivanandan, P.K.; Loganathan, T.; Begum, M.; Rai, S. Identification of Marker Genes in Alzheimer’s Disease Using a Machine-Learning Model. Bioinformation 2021, 17, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Liu, H.; Yan, R.; Hua, L.; Chen, Y.; Shi, J.; Lu, Q.; Yao, Z. Cortical Thickness and Subcortical Structure Volume Abnormalities in Patients with Major Depression with and without Anxious Symptoms. Brain Behav. 2017, 7, e00754. [Google Scholar] [CrossRef]
- Nagy, C.; Maitra, M.; Tanti, A.; Suderman, M.; Théroux, J.-F.; Davoli, M.A.; Perlman, K.; Yerko, V.; Wang, Y.C.; Tripathy, S.J.; et al. Single-Nucleus Transcriptomics of the Prefrontal Cortex in Major Depressive Disorder Implicates Oligodendrocyte Precursor Cells and Excitatory Neurons. Nat. Neurosci. 2020, 23, 771–781. [Google Scholar] [CrossRef]
- Crane, N.A.; Jenkins, L.M.; Dion, C.; Meyers, K.K.; Weldon, A.L.; Gabriel, L.B.; Walker, S.J.; Hsu, D.T.; Noll, D.C.; Klumpp, H.; et al. Comorbid Anxiety Increases Cognitive Control Activation in Major Depressive Disorder: Crane et Al. Depress. Anxiety 2016, 33, 967–977. [Google Scholar] [CrossRef]
- Zhang, F.-F.; Peng, W.; Sweeney, J.A.; Jia, Z.-Y.; Gong, Q.-Y. Brain Structure Alterations in Depression: Psychoradiological Evidence. CNS Neurosci. Ther. 2018, 24, 994–1003. [Google Scholar] [CrossRef]
- Hussain, M.; Kumar, P.; Khan, S.; Gordon, D.K.; Khan, S. Similarities Between Depression and Neurodegenerative Diseases: Pathophysiology, Challenges in Diagnosis and Treatment Options. Cureus 2020, 12, e11613. [Google Scholar] [CrossRef]
- Borsini, A.; Zunszain, P.A. Advances in Stem Cells Biology: New Approaches to Understand Depression. In Stem Cells in Neuroendocrinology; Pfaff, D., Christen, Y., Eds.; Springer: Cham, Switzerland, 2016; ISBN 978-3-319-41602-1. [Google Scholar]
- Vats, A.; Bielby, R.; Tolley, N.; Nerem, R.; Polak, J. Stem Cells. Lancet 2005, 366, 592–602. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V. Stem Cells in Tissue Repair and Regeneration. J. Investig. Dermatol. 2012, 132, 1538–1541. [Google Scholar] [CrossRef] [PubMed]
- Congdon, C.C. Bone Marrow Transplantation in Animals Exposed to Whole-Body Radiation. J. Cell. Comp. Physiol. 1957, 50, 103–108. [Google Scholar] [CrossRef]
- Kolios, G.; Moodley, Y. Introduction to Stem Cells and Regenerative Medicine. Respiration 2013, 85, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Mora, C.; Serzanti, M.; Consiglio, A.; Memo, M.; Dell’Era, P. Clinical Potentials of Human Pluripotent Stem Cells. Cell Biol. Toxicol. 2017, 33, 351–360. [Google Scholar] [CrossRef]
- Zhang, Y.-T.; He, K.-J.; Zhang, J.-B.; Ma, Q.-H.; Wang, F.; Liu, C.-F. Advances in Intranasal Application of Stem Cells in the Treatment of Central Nervous System Diseases. Stem Cell Res. Ther. 2021, 12, 210. [Google Scholar] [CrossRef]
- Villar-Gómez, N.; Ojeda-Hernandez, D.D.; López-Muguruza, E.; García-Flores, S.; Bonel-García, N.; Benito-Martín, M.S.; Selma-Calvo, B.; Canales-Aguirre, A.A.; Mateos-Díaz, J.C.; Montero-Escribano, P.; et al. Nose-to-Brain: The Next Step for Stem Cell and Biomaterial Therapy in Neurological Disorders. Cells 2022, 11, 3095. [Google Scholar] [CrossRef]
- Marcatili, M.; Sala, C.; Dakanalis, A.; Colmegna, F.; D’Agostino, A.; Gambini, O.; Dell’Osso, B.; Benatti, B.; Conti, L.; Clerici, M. Human Induced Pluripotent Stem Cells Technology in Treatment Resistant Depression: Novel Strategies and Opportunities to Unravel Ketamine’s Fast-Acting Antidepressant Mechanisms. Ther. Adv. Psychopharmacol. 2020, 10, 204512532096833. [Google Scholar] [CrossRef]
- Soliman, M.A.; Aboharb, F.; Zeltner, N.; Studer, L. Pluripotent Stem Cells in Neuropsychiatric Disorders. Mol. Psychiatry 2017, 22, 1241–1249. [Google Scholar] [CrossRef]
- Correia-Melo, F.S.; Leal, G.C.; Carvalho, M.S.; Jesus-Nunes, A.P.; Ferreira, C.B.N.; Vieira, F.; Magnavita, G.; Vale, L.A.S.; Mello, R.P.; Nakahira, C.; et al. Comparative Study of Esketamine and Racemic Ketamine in Treatment-Resistant Depression: Protocol for a Non-Inferiority Clinical Trial. Medicine 2018, 97, e12414. [Google Scholar] [CrossRef]
- Licinio, J.; Wong, M.-L. Serotonergic Neurons Derived from Induced Pluripotent Stem Cells (IPSCs): A New Pathway for Research on the Biology and Pharmacology of Major Depression. Mol. Psychiatry 2016, 21, 1–2. [Google Scholar] [CrossRef]
- Xu, Z.; Jiang, H.; Zhong, P.; Yan, Z.; Chen, S.; Feng, J. Direct Conversion of Human Fibroblasts to Induced Serotonergic Neurons. Mol. Psychiatry 2016, 21, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Vadodaria, K.C.; Mertens, J.; Paquola, A.; Bardy, C.; Li, X.; Jappelli, R.; Fung, L.; Marchetto, M.C.; Hamm, M.; Gorris, M.; et al. Generation of Functional Human Serotonergic Neurons from Fibroblasts. Mol. Psychiatry 2016, 21, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal Stem Cells in Health and Disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef]
- Ahmad, F.; Sachdeva, P. A Consolidated Review on Stem Cell Therapy for Treatment and Management of Alzheimer’s Disease. Aging Med. 2022, 5, 182–190. [Google Scholar] [CrossRef]
- Ding, D.-C.; Shyu, W.-C.; Lin, S.-Z. Mesenchymal Stem Cells. Cell Transpl. 2011, 20, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Hao, H.; Han, Q.; Song, X.; Liu, J.; Dong, L.; Han, W.; Mu, Y. Human Umbilical Cord-Derived Mesenchymal Stem Cells Ameliorate Insulin Resistance by Suppressing NLRP3 Inflammasome-Mediated Inflammation in Type 2 Diabetes Rats. Stem Cell Res. Ther. 2017, 8, 241. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Li, Y.; Liu, R.; Pan, J.; Tang, X.; Sun, S.; Liu, J.; Ma, W. Human Umbilical Cord Mesenchymal Stem Cells Ameliorate Depression by Regulating Jmjd3 and Microglia Polarization in Myocardial Infarction Mice. Psychopharmacology 2021, 238, 2973–2984. [Google Scholar] [CrossRef]
- Mathews, D.C.; Henter, I.D.; Zarate, C.A. Targeting the Glutamatergic System to Treat Major Depressive Disorder: Rationale and Progress to Date. Drugs 2012, 72, 1313–1333. [Google Scholar] [CrossRef]
- Mauri, M.C.; Ferrara, A.; Boscati, L.; Bravin, S.; Zamberlan, F.; Alecci, M.; Invernizzi, G. Plasma and Platelet Amino Acid Concentrations in Patients Affected by Major Depression and under Fluvoxamine Treatment. Neuropsychobiology 1998, 37, 124–129. [Google Scholar] [CrossRef]
- Kim, J.S.; Schmid-Burgk, W.; Claus, D.; Kornhuber, H.H. Increased Serum Glutamate in Depressed Patients. Arch. Psychiatr. Nervenkr. 1982, 232, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Mitani, H.; Shirayama, Y.; Yamada, T.; Maeda, K.; Ashby, C.R.; Kawahara, R. Correlation between Plasma Levels of Glutamate, Alanine and Serine with Severity of Depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2006, 30, 1155–1158. [Google Scholar] [CrossRef]
- Frye, M.A.; Tsai, G.E.; Huggins, T.; Coyle, J.T.; Post, R.M. Low Cerebrospinal Fluid Glutamate and Glycine in Refractory Affective Disorder. Biol. Psychiatry 2007, 61, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Francis, P.T.; Poynton, A.; Lowe, S.L.; Najlerahim, A.; Bridges, P.K.; Bartlett, J.R.; Procter, A.W.; Bruton, C.J.; Bowen, D.M. Brain Amino Acid Concentrations and Ca2+-Dependent Release in Intractable Depression Assessed Antemortem. Brain Res. 1989, 494, 315–324. [Google Scholar] [CrossRef]
- Nowak, G.; Ordway, G.A.; Paul, I.A. Alterations in the N-Methyl-d-Asparatate (NMDA) Receptor Complex in the Frontal Cortex of Suicide Victims. Brain Res. 1995, 675, 157–164. [Google Scholar] [CrossRef]
- Holemans, S.; De Paermentier, F.; Horton, R.W.; Crompton, M.R.; Katona, C.L.E.; Maloteaux, J.-M. NMDA Glutamatergic Receptors, Labelled with [3H]MK-801, in Brain Samples from Drug-Free Depressed Suicides. Brain Res. 1993, 616, 138–143. [Google Scholar] [CrossRef]
- Hascup, K.N.; Hascup, E.R.; Stephens, M.L.; Glaser, P.E.; Yoshitake, T.; Mathé, A.A.; Gerhardt, G.A.; Kehr, J. Resting Glutamate Levels and Rapid Glutamate Transients in the Prefrontal Cortex of the Flinders Sensitive Line Rat: A Genetic Rodent Model of Depression. Neuropsychopharmacology 2011, 36, 1769–1777. [Google Scholar] [CrossRef]
- Rajkowska, G.; Miguel-Hidalgo, J.J.; Wei, J.; Dilley, G.; Pittman, S.D.; Meltzer, H.Y.; Overholser, J.C.; Roth, B.L.; Stockmeier, C.A. Morphometric Evidence for Neuronal and Glial Prefrontal Cell Pathology in Major Depression∗∗See Accompanying Editorial, in This Issue. Biol. Psychiatry 1999, 45, 1085–1098. [Google Scholar] [CrossRef]
- Öngür, D.; Drevets, W.C.; Price, J.L. Glial Reduction in the Subgenual Prefrontal Cortex in Mood Disorders. Proc. Natl. Acad. Sci. USA 1998, 95, 13290–13295. [Google Scholar] [CrossRef]
- Yang, C.; Hu, Y.-M.; Zhou, Z.-Q.; Zhang, G.-F.; Yang, J.-J. Acute Administration of Ketamine in Rats Increases Hippocampal BDNF and MTOR Levels during Forced Swimming Test. Upsala J. Med. Sci. 2013, 118, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Sachdeva, B.; Sachdeva, P.; Chaudhary, V.; Rani, G.M.; Sinha, J.K. Graphene Quantum Dots as a Potential Diagnostic and Therapeutic Tool for the Management of Alzheimer’s Disease. Carbon Lett. 2022, 32, 1381–1394. [Google Scholar] [CrossRef]
- Shwartz, A.; Betzer, O.; Kronfeld, N.; Kazimirsky, G.; Cazacu, S.; Finniss, S.; Lee, H.K.; Motiei, M.; Dagan, S.Y.; Popovtzer, R.; et al. Therapeutic Effect of Astroglia-like Mesenchymal Stem Cells Expressing Glutamate Transporter in a Genetic Rat Model of Depression. Theranostics 2017, 7, 2690–2703. [Google Scholar] [CrossRef] [PubMed]
- Friedman, A.; Frankel, M.; Flaumenhaft, Y.; Merenlender, A.; Pinhasov, A.; Feder, Y.; Taler, M.; Gil-Ad, I.; Abeles, M.; Yadid, G. Programmed Acute Electrical Stimulation of Ventral Tegmental Area Alleviates Depressive-Like Behavior. Neuropsychopharmacology 2009, 34, 1057–1066. [Google Scholar] [CrossRef]
- Jia, K.-K.; Ding, H.; Yu, H.-W.; Dong, T.-J.; Pan, Y.; Kong, L.-D. Huanglian-Wendan Decoction Inhibits NF- κ B/NLRP3 Inflammasome Activation in Liver and Brain of Rats Exposed to Chronic Unpredictable Mild Stress. Mediat. Inflamm. 2018, 2018, 3093516. [Google Scholar] [CrossRef]
- Huang, X.; Fei, G.; Liu, W.; Ding, J.; Wang, Y.; Wang, H.; Ji, J.; Wang, X. Adipose-Derived Mesenchymal Stem Cells Protect against CMS-Induced Depression-like Behaviors in Mice via Regulating the Nrf2/HO-1 and TLR4/NF-ΚB Signaling Pathways. Acta Pharmacol. Sin. 2020, 41, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Choi, S.H.; Kim, B.S.; Choi, J.Y.; Park, G.-B.; Kwon, T.G.; Chun, S.Y. Advanced Properties of Urine Derived Stem Cells Compared to Adipose Tissue Derived Stem Cells in Terms of Cell Proliferation, Immune Modulation and Multi Differentiation. J. Korean Med. Sci. 2015, 30, 1764. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Sheng, W.; Han, L.; He, Q.; Ji, X.; Liu, K. Activation of BDNF-TrkB Signaling Pathway-Regulated Brain Inflammation in Pentylenetetrazole-Induced Seizures in Zebrafish. Fish Shellfish Immunol. 2018, 83, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Xiong, B.; Zhang, B.; Li, S.; Huang, N.; Zhan, G.; Jiang, R.; Yang, L.; Wu, Y.; Miao, L.; et al. Sulforaphane Alleviates Lipopolysaccharide-Induced Spatial Learning and Memory Dysfunction in Mice: The Role of BDNF-MTOR Signaling Pathway. Neuroscience 2018, 388, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Jahromi, M.; Razavi, S.; Amirpour, N.; Khosravizadeh, Z. Paroxetine Can Enhance Neurogenesis during Neurogenic Differentiation of Human Adipose-Derived Stem Cells. Avicenna J. Med. Biotechnol. 2016, 8, 152–158. [Google Scholar]
- Wang, L.-J.; Liu, L.-P.; Gu, X.L.; Wang, M.; Liu, L.-M. Implantation of Adipose-Derived Stem Cells Cures the Optic Nerve Injury on Rats through Inhibiting the Expression of Inflammation Factors in the TLR4 Signaling Pathway. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 1196–1202. [Google Scholar] [CrossRef]
- Putcha, G.V.; Le, S.; Frank, S.; Besirli, C.G.; Clark, K.; Chu, B.; Alix, S.; Youle, R.J.; LaMarche, A.; Maroney, A.C.; et al. JNK-Mediated BIM Phosphorylation Potentiates BAX-Dependent Apoptosis. Neuron 2003, 38, 899–914. [Google Scholar] [CrossRef]
- Tfilin, M.; Sudai, E.; Merenlender, A.; Gispan, I.; Yadid, G.; Turgeman, G. Mesenchymal Stem Cells Increase Hippocampal Neurogenesis and Counteract Depressive-like Behavior. Mol. Psychiatry 2010, 15, 1164–1175. [Google Scholar] [CrossRef]
- Kin, K.; Yasuhara, T.; Kameda, M.; Tomita, Y.; Umakoshi, M.; Kuwahara, K.; Kin, I.; Kidani, N.; Morimoto, J.; Okazaki, M.; et al. Cell Encapsulation Enhances Antidepressant Effect of the Mesenchymal Stem Cells and Counteracts Depressive-like Behavior of Treatment-Resistant Depressed Rats. Mol. Psychiatry 2020, 25, 1202–1214. [Google Scholar] [CrossRef]
- Urbán, N.; Blomfield, I.M.; Guillemot, F. Quiescence of Adult Mammalian Neural Stem Cells: A Highly Regulated Rest. Neuron 2019, 104, 834–848. [Google Scholar] [CrossRef] [PubMed]
- Kukekov, V.G.; Laywell, E.D.; Suslov, O.; Davies, K.; Scheffler, B.; Thomas, L.B.; O’Brien, T.F.; Kusakabe, M.; Steindler, D.A. Multipotent Stem/Progenitor Cells with Similar Properties Arise from Two Neurogenic Regions of Adult Human Brain. Exp. Neurol. 1999, 156, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Buylla, A.; García-Verdugo, J.M. Neurogenesis in Adult Subventricular Zone. J. Neurosci. 2002, 22, 629–634. [Google Scholar] [CrossRef]
- Liu, Z.; Martin, L.J. Olfactory Bulb Core Is a Rich Source of Neural Progenitor and Stem Cells in Adult Rodent and Human. J. Comp. Neurol. 2003, 459, 368–391. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G. Regulation of Adult Hippocampal Neurogenesis—Implications for Novel Theories of Major Depression 1: Regulation of Adult Hippocampal Neurogenesis. Bipolar Disord. 2002, 4, 17–33. [Google Scholar] [CrossRef]
- Mansoor, A.K.; Thomas, S.; Sinha, J.K.; Alladi, P.A.; Ravi, V.; Raju, T.R. Olfactory tract transection reveals robust tissue-level plasticity by cellular numbers and neurotrophic factor expression in olfactory bulb. Indian J. Exp. Biol. 2012, 50, 765–770. [Google Scholar]
- Feldmann, R.E.; Mattern, R. The Human Brain and Its Neural Stem Cells Postmortem: From Dead Brains to Live Therapy. Int. J. Leg. Med. 2006, 120, 201–211. [Google Scholar] [CrossRef]
- Lindvall, O.; Kokaia, Z. Recovery and Rehabilitation in Stroke: Stem Cells. Stroke 2004, 35, 2691–2694. [Google Scholar] [CrossRef]
- Sachdeva, P.; Ghosh, S.; Ghosh, S.; Han, S.; Banerjee, J.; Bhaskar, R.; Sinha, J.K. Childhood Obesity: A Potential Key Factor in the Development of Glioblastoma Multiforme. Life 2022, 12, 1673. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Manchala, S.; Raghunath, M.; Sharma, G.; Singh, A.K.; Sinha, J.K. Role of Phytomolecules in the Treatment of Obesity: Targets, Mechanisms and Limitations. Curr. Top. Med. Chem. 2021, 21, 863–877. [Google Scholar] [CrossRef]
- Goldman, S. Stem and Progenitor Cell–Based Therapy of the Human Central Nervous System. Nat. Biotechnol. 2005, 23, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Lipska, B.K. Using Animal Models to Test a Neurodevelopmental Hypothesis of Schizophrenia. J. Psychiatry Neurosci. 2004, 29, 282–286. [Google Scholar] [PubMed]
- Feldmann, R.E.; Sawa, A.; Seidler, G.H. Causality of Stem Cell Based Neurogenesis and Depression—To Be or Not to Be, Is That the Question? J. Psychiatr. Res. 2007, 41, 713–723. [Google Scholar] [CrossRef]
- Zhao, C. Distinct Morphological Stages of Dentate Granule Neuron Maturation in the Adult Mouse Hippocampus. J. Neurosci. 2006, 26, 3–11. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sachdeva, P.; Ji, S.; Ghosh, S.; Ghosh, S.; Raghunath, M.; Kim, H.; Bhaskar, R.; Sinha, J.K.; Han, S.S. Plausible Role of Stem Cell Types for Treating and Understanding the Pathophysiology of Depression. Pharmaceutics 2023, 15, 814. https://doi.org/10.3390/pharmaceutics15030814
Sachdeva P, Ji S, Ghosh S, Ghosh S, Raghunath M, Kim H, Bhaskar R, Sinha JK, Han SS. Plausible Role of Stem Cell Types for Treating and Understanding the Pathophysiology of Depression. Pharmaceutics. 2023; 15(3):814. https://doi.org/10.3390/pharmaceutics15030814
Chicago/Turabian StyleSachdeva, Punya, Seongmin Ji, Shampa Ghosh, Soumya Ghosh, Manchala Raghunath, Hyunjin Kim, Rakesh Bhaskar, Jitendra Kumar Sinha, and Sung Soo Han. 2023. "Plausible Role of Stem Cell Types for Treating and Understanding the Pathophysiology of Depression" Pharmaceutics 15, no. 3: 814. https://doi.org/10.3390/pharmaceutics15030814
APA StyleSachdeva, P., Ji, S., Ghosh, S., Ghosh, S., Raghunath, M., Kim, H., Bhaskar, R., Sinha, J. K., & Han, S. S. (2023). Plausible Role of Stem Cell Types for Treating and Understanding the Pathophysiology of Depression. Pharmaceutics, 15(3), 814. https://doi.org/10.3390/pharmaceutics15030814






