A Photoactive Supramolecular Complex Targeting PD-L1 Reveals a Weak Correlation between Photoactivation Efficiency and Receptor Expression Levels in Non-Small-Cell Lung Cancer Tumor Models
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Purification of Atezolizumab and Labeling with EITC or AlexaFluor647
2.3. Spectral Properties of the Conjugate and 1O2 Production
2.4. Cell Culture
2.5. Evaluation of PD-L1 Expression
2.5.1. Flow Cytometry
2.5.2. Microscope Setup
2.5.3. Sample Preparation and Acquisition for dSTORM Imaging
2.5.4. dSTORM Image Analysis
2.6. Photodynamic Inactivation of Cultured Cells
2.6.1. Analysis of Cell Viability and Cell Death
2.6.2. ROS Detection
2.6.3. Statistical Analyses
3. Results and Discussion
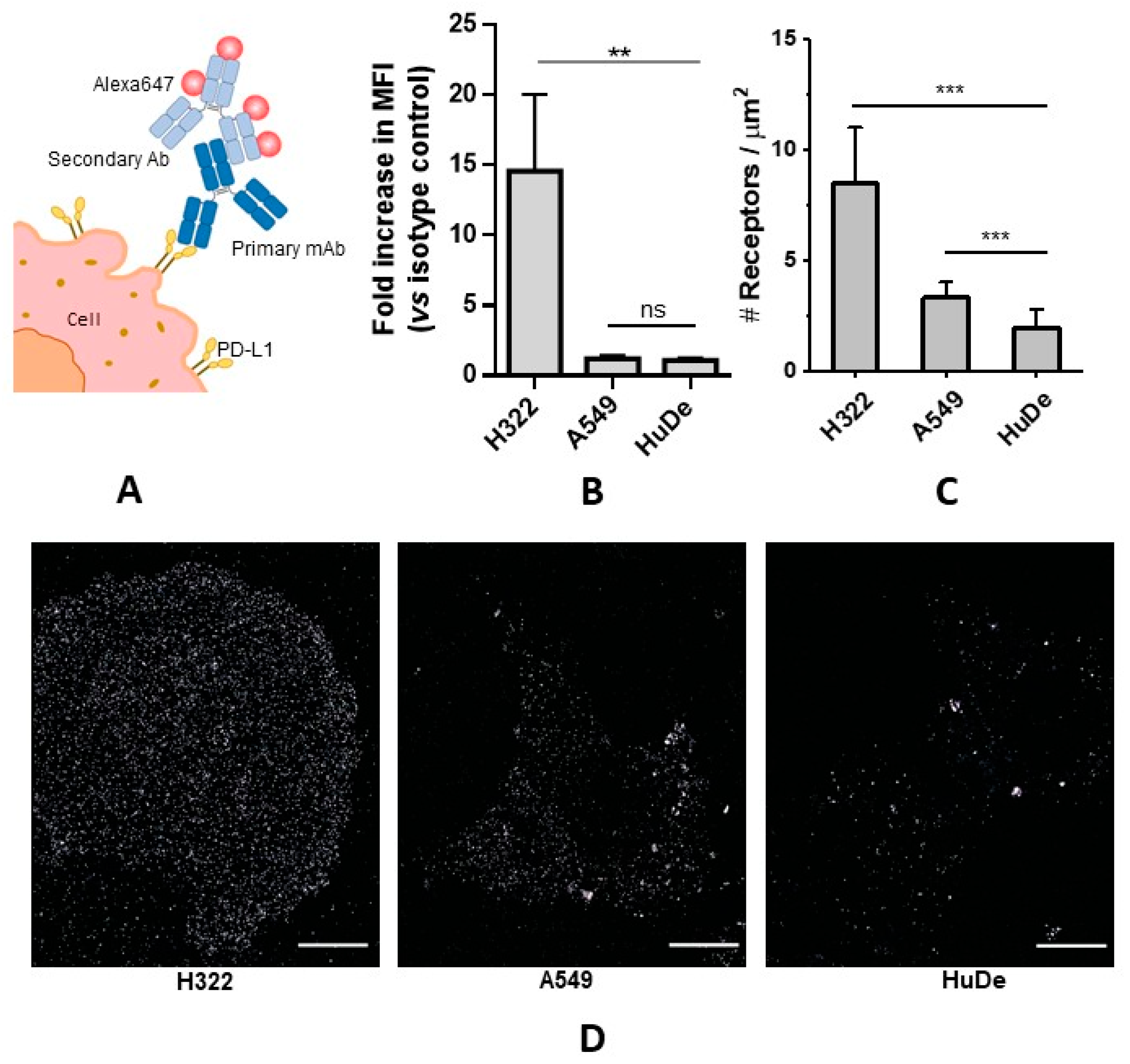
3.1. Quantitative Determination of PD-L1 Expression Levels
3.1.1. PD-L1 Receptor Density via Flow Cytometry
3.1.2. PD-L1 Receptor Density via dSTORM
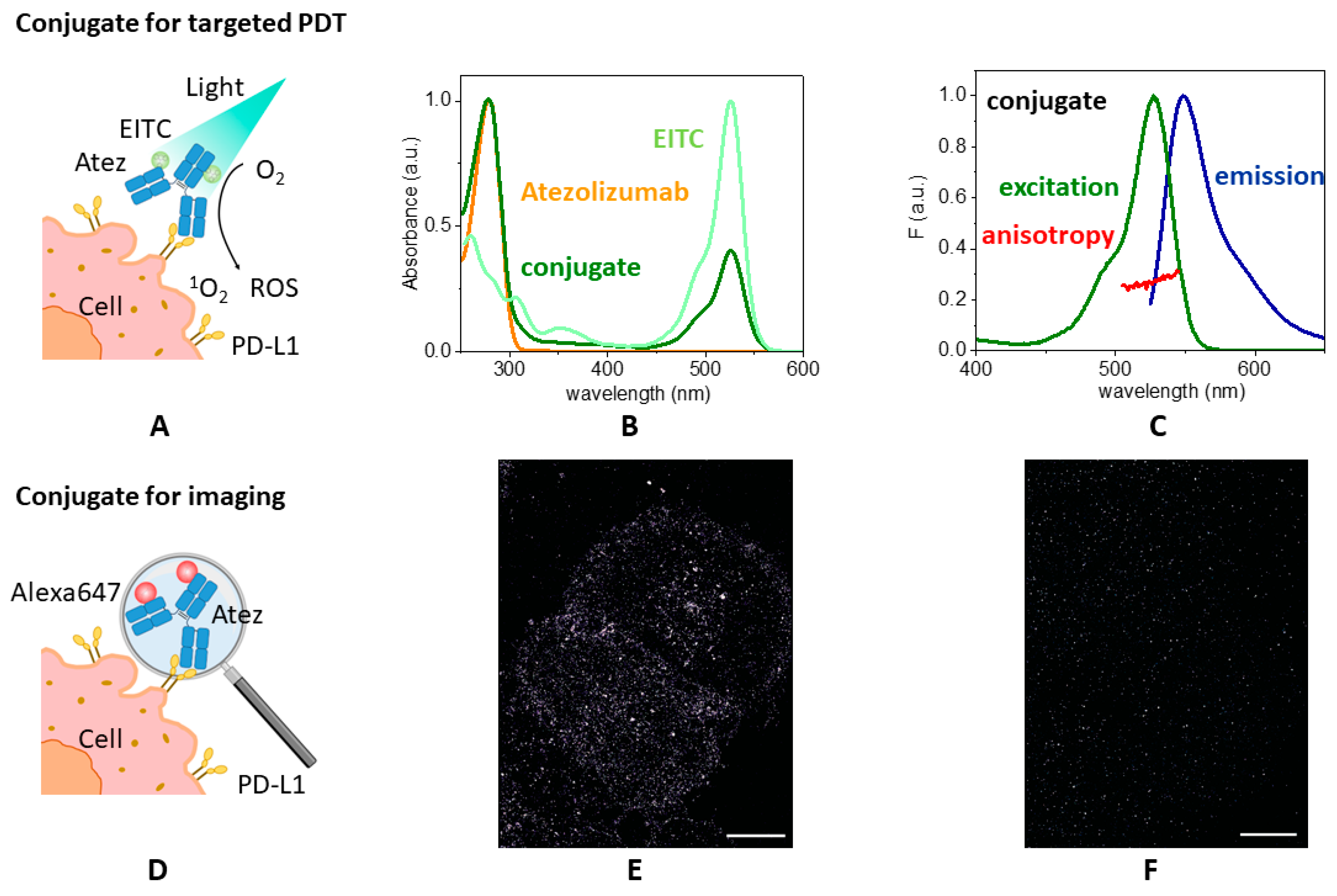
3.2. EITC–Atezolizumab Complex for Photo-Immunotherapy
3.2.1. Spectral Properties of EITC–Atezolizumab
3.2.2. Singlet Oxygen Production by EITC–Atezolizumab
3.2.3. Binding Capability of the Conjugate to Cell Lines Expressing PD-L1
3.3. Photoinactivation Treatment with the Conjugate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332. [Google Scholar] [CrossRef]
- Dąbrowski, J.M.; Arnaut, L.G. Photodynamic therapy (PDT) of cancer: From local to systemic treatment. Photochem. Photobiol. Sci. 2015, 14, 1765–1780. [Google Scholar] [CrossRef]
- Spring, B.Q.; Rizvi, I.; Xu, N.; Hasan, T. The role of photodynamic therapy in overcoming cancer drug resistance. Photochem. Photobiol. Sci. 2015, 14, 1476–1491. [Google Scholar] [CrossRef]
- Callaghan, S.; Senge, M.O. The good, the bad, and the ugly—Controlling singlet oxygen through design of photosensitizers and delivery systems for photodynamic therapy. Photochem. Photobiol. Sci. 2018, 17, 1490–1514. [Google Scholar] [CrossRef]
- Hwang, H.S.; Shin, H.; Han, J.; Na, K. Combination of photodynamic therapy (PDT) and anti-tumor immunity in cancer therapy. J. Pharm. Investig. 2018, 48, 143–151. [Google Scholar] [CrossRef]
- Baskaran, R.; Lee, J.; Yang, S.-G. Clinical development of photodynamic agents and therapeutic applications. Biomater. Res. 2018, 22, 25. [Google Scholar] [CrossRef]
- Qicai, X.; Juan, W.; Xin, P.; Yue, J.; Pan, W.; Albert, W.L.; Liqian, G.; Sheng, J.; Chuanshan, X. Discovery and Development of Natural Products and their Derivatives as Photosensitizers for Photodynamic Therapy. Curr. Med. Chem. 2018, 25, 839–860. [Google Scholar]
- Bullous, A.J.; Alonso, C.M.A.; Boyle, R.W. Photosensitiser–antibody conjugates for photodynamic therapy. Photochem. Photobiol. Sci. 2011, 10, 721–750. [Google Scholar] [CrossRef]
- Bugaj, A.M. Targeted photodynamic therapy—A promising strategy of tumor treatment. Photochem. Photobiol. Sci. 2011, 10, 1097–1109. [Google Scholar] [CrossRef]
- Giuntini, F.; Alonso, C.M.A.; Boyle, R.W. Synthetic approaches for the conjugation of porphyrins and related macrocycles to peptides and proteins. Photochem. Photobiol. Sci. 2011, 10, 759–791. [Google Scholar] [CrossRef]
- Giuntini, F.; Boyle, R.; Sibrian-Vazquez, M.; Vicente, M.G.H. Porphyrin Conjugates for Cancer Therapy. In Handbook of Porphyrin Science; World Scientific: Singapore, 2013; Volume 27, pp. 303–416. [Google Scholar]
- Planas, O.; Boix-Garriga, E.; Rodríguez-Amigo, T.J.B.; Bresolí-Obach, R.; Flors, C.; Viappiani, C.; Agut, M.; Ruiz-González, R.; Nonell, S. Newest approaches to singlet oxygen photosensitisation in biological media. In Photochemistry; Albini, A., Fasani, E., Eds.; The Royal Society of Chemistry: London, UK, 2014; Volume 42. [Google Scholar]
- Carsten, L.; Patrick, J.B. Photosensitizers for Photodynamic Therapy: Photochemistry in the Service of Oncology. Curr. Pharm. Des. 2016, 22, 6956–6974. [Google Scholar]
- Fernandes, S.R.G.; Fernandes, R.; Sarmento, B.; Pereira, P.M.R.; Tomé, J.P.C. Photoimmunoconjugates: Novel synthetic strategies to target and treat cancer by photodynamic therapy. Org. Biomol. Chem. 2019, 17, 2579–2593. [Google Scholar] [CrossRef]
- Mew, D.; Wat, C.K.; Towers, G.H.; Levy, J.G. Photoimmunotherapy: Treatment of animal tumors with tumor-specific monoclonal antibody-hematoporphyrin conjugates. J. Immunol. 1983, 130, 1473–1477. [Google Scholar] [CrossRef]
- Calixto, G.M.F.; Bernegossi, J.; De Freitas, L.M.; Fontana, C.R.; Chorilli, M. Nanotechnology-Based Drug Delivery Systems for Photodynamic Therapy of Cancer: A Review. Molecules 2016, 21, 342. [Google Scholar] [CrossRef]
- Mussini, A.; Uriati, E.; Bianchini, P.; Diaspro, A.; Cavanna, L.; Abbruzzetti, S.; Viappiani, C. Targeted photoimmunotherapy for cancer. Biomol. Concepts 2022, 13, 126–147. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, J.; Zhao, C.; Zhang, S.; Zhu, J. Recent Advancement of PD-L1 Detection Technologies and Clinical Applications in the Era of Precision Cancer Therapy. J. Cancer 2023, 14, 850–873. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, X.; Liu, X.; Yu, J.; Bai, X.; Wu, X.; Guo, X.; Liu, Z.; Liu, X. Combination of phototherapy with immune checkpoint blockade: Theory and practice in cancer. Front. Immunol. 2022, 13, 955920. [Google Scholar] [CrossRef]
- Goodman, A.; Patel, S.P.; Kurzrock, R. PD-1–PD-L1 immune-checkpoint blockade in B-cell lymphomas. Nat. Rev. Clin. Oncol. 2017, 14, 203–220. [Google Scholar] [CrossRef]
- Zhou, K.; Li, S.; Zhao, Y.; Cheng, K. Mechanisms of drug resistance to immune checkpoint inhibitors in non-small cell lung cancer. Front. Immunol. 2023, 14, 1758835920937902. [Google Scholar] [CrossRef]
- Topalian, S.L.; Taube, J.M.; Pardoll, D.M. Neoadjuvant checkpoint blockade for cancer immunotherapy. Science 2020, 367, eaax0182. [Google Scholar] [CrossRef]
- Picardo, S.L.; Doi, J.; Hansen, A.R. Structure and Optimization of Checkpoint Inhibitors. Cancers 2020, 12, 38. [Google Scholar] [CrossRef]
- Bilgin, B.; Sendur, M.A.N.; Bülent Akıncı, M.; Şener Dede, D.; Yalçın, B. Targeting the PD-1 pathway: A new hope for gastrointestinal cancers. Curr. Med. Res. Opin. 2017, 33, 749–759. [Google Scholar] [CrossRef]
- Chang, H.-N.; Liu, B.-Y.; Qi, Y.-K.; Zhou, Y.; Chen, Y.-P.; Pan, K.-M.; Li, W.-W.; Zhou, X.-M.; Ma, W.-W.; Fu, C.-Y.; et al. Blocking of the PD-1/PD-L1 Interaction by a D-Peptide Antagonist for Cancer Immunotherapy. Angew. Chem. Int. Ed. 2015, 54, 11760–11764. [Google Scholar] [CrossRef]
- Zhang, P.; Li, C.; Ji, X.; Gao, M.; Lyu, S.; Dai, X.; Du, J. In silico screening and surface plasma resonance-based verification of programmed death 1-targeted peptides. Chem. Biol. Drug Des. 2020, 95, 332–342. [Google Scholar] [CrossRef]
- Zhai, W.; Zhou, X.; Zhai, M.; Li, W.; Ran, Y.; Sun, Y.; Du, J.; Zhao, W.; Xing, L.; Qi, Y.; et al. Blocking of the PD-1/PD-L1 interaction by a novel cyclic peptide inhibitor for cancer immunotherapy. Sci. China Life Sci. 2020, 64, 548–562. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, Y.; Xi, G.; Zhang, F. HX008: A humanized PD-1 blocking antibody with potent antitumor activity and superior pharmacologic properties. Mabs 2020, 12, 1724751. [Google Scholar] [CrossRef]
- Davis, K.L.; Fox, E.; Merchant, M.S.; Reid, J.M.; Kudgus, R.A.; Liu, X.; Minard, C.G.; Voss, S.; Berg, S.L.; Weigel, B.J.; et al. Nivolumab in children and young adults with relapsed or refractory solid tumours or lymphoma (ADVL1412): A multicentre, open-label, single-arm, phase 1–2 trial. Lancet Oncol. 2020, 21, 541–550. [Google Scholar] [CrossRef]
- Carter, P.J.; Lazar, G.A. Next generation antibody drugs: Pursuit of the ‘high-hanging fruit’. Nat. Rev. Drug Discov. 2018, 17, 197–223. [Google Scholar] [CrossRef]
- Mussini, A.; Uriati, E.; Hally, C.; Nonell, S.; Bianchini, P.; Diaspro, A.; Pongolini, S.; Delcanale, P.; Abbruzzetti, S.; Viappiani, C. Versatile Supramolecular Complex for Targeted Antimicrobial Photodynamic Inactivation. Bioconjugate Chem. 2022, 33, 666–676. [Google Scholar] [CrossRef]
- Fan, B.; Peng, W.; Zhang, Y.; Liu, P.; Shen, J. ROS conversion promotes the bactericidal efficiency of Eosin Y based photodynamic therapy. Biomater. Sci. 2023, 11, 4930–4937. [Google Scholar] [CrossRef]
- Johnson, G.A.; Muthukrishnan, N.; Pellois, J.P. Photoinactivation of Gram positive and Gram negative bacteria with the antimicrobial peptide (KLAKLAK)(2) conjugated to the hydrophilic photosensitizer eosin Y. Bioconjugate Chem. 2013, 24, 114–123. [Google Scholar] [CrossRef]
- López-Peña, G.; Simón-Fuente, S.; Ortgies, D.H.; Moliné, M.Á.; Martín Rodríguez, E.; Sanz-Rodríguez, F.; Ribagorda, M. Eosin Y-Functionalized Upconverting Nanoparticles: Nanophotosensitizers and Deep Tissue Bioimaging Agents for Simultaneous Therapeutic and Diagnostic Applications. Cancers 2023, 15, 102. [Google Scholar] [CrossRef]
- Zanello, A.; Bortolotti, M.; Maiello, S.; Bolognesi, A.; Polito, L. Anti-PD-L1 immunoconjugates for cancer therapy: Are available antibodies good carriers for toxic payload delivering? Front. Pharmacol. 2022, 13, 972046. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Li, K.; Cheng, X.; Chen, L.; Yu, Q.; Fan, H.; Zheng, L.; Yang, Z.; Ni, F. A photoactivatable antibody-Chlorin e6 conjugate enabling singlet oxygen production for tumor-targeting photodynamic therapy. Biomed. Mater 2021, 16, 045003. [Google Scholar] [CrossRef]
- Wong, J.J.W.; Selbo, P.K. Light-controlled elimination of PD-L1+ cells. J. Photochem. Photobiol. B Biol. 2021, 225, 112355. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.N.; Vajdos, F.; Fee, L.; Grimsley, G.; Gray, T. How to measure and predict the molar absorption coefficient of a protein. Protein Sci. 1995, 4, 2411–2423. [Google Scholar] [CrossRef] [PubMed]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: New York, NY, USA, 2006; p. 496. [Google Scholar]
- Zhang, X.-F.; Zhang, J.; Liu, L. Fluorescence Properties of Twenty Fluorescein Derivatives: Lifetime, Quantum Yield, Absorption and Emission Spectra. J. Fluor. 2014, 24, 819–826. [Google Scholar] [CrossRef]
- Bruno, S.; Faggiano, S.; Spyrakis, F.; Mozzarelli, A.; Cacciatori, E.; Dominici, P.; Grandi, E.; Abbruzzetti, S.; Viappiani, C. Different roles of protein dynamics and ligand migration in non-symbiotic hemoglobins AHb1 and AHb2 from Arabidopsis thaliana. Gene 2007, 398, 224–233. [Google Scholar] [CrossRef]
- Woythe, L.; Madhikar, P.; Feiner-Gracia, N.; Storm, C.; Albertazzi, L. A Single-Molecule View at Nanoparticle Targeting Selectivity: Correlating Ligand Functionality and Cell Receptor Density. ACS Nano 2022, 16, 3785–3796. [Google Scholar] [CrossRef]
- Delcanale, P.; Miret-Ontiveros, B.; Arista-Romero, M.; Pujals, S.; Albertazzi, L. Nanoscale Mapping Functional Sites on Nanoparticles by Points Accumulation for Imaging in Nanoscale Topography (PAINT). ACS Nano 2018, 12, 7629–7637. [Google Scholar] [CrossRef]
- Fumarola, C.; La Monica, S.; Alfieri, R.R.; Borra, E.; Guidotti, G.G. Cell size reduction induced by inhibition of the mTOR/S6K-signaling pathway protects Jurkat cells from apoptosis. Cell Death Differ. 2005, 12, 1344–1357. [Google Scholar] [CrossRef] [PubMed]
- Cavazzoni, A.; Digiacomo, G.; Alfieri, R.; La Monica, S.; Fumarola, C.; Galetti, M.; Bonelli, M.; Cretella, D.; Barili, V.; Zecca, A.; et al. Pemetrexed Enhances Membrane PD-L1 Expression and Potentiates T Cell-Mediated Cytotoxicity by Anti-PD-L1 Antibody Therapy in Non-Small-Cell Lung Cancer. Cancers 2020, 12, 666. [Google Scholar] [CrossRef] [PubMed]
- Beenen, A.C.; Sauerer, T.; Schaft, N.; Dörrie, J. Beyond Cancer: Regulation and Function of PD-L1 in Health and Immune-Related Diseases. Int. J. Mol. Sci. 2022, 23, 8599. [Google Scholar] [CrossRef] [PubMed]
- van de Linde, S.; Löschberger, A.; Klein, T.; Heidbreder, M.; Wolter, S.; Heilemann, M.; Sauer, M. Direct stochastic optical reconstruction microscopy with standard fluorescent probes. Nat. Protoc. 2011, 6, 991–1009. [Google Scholar] [CrossRef]
- Nerreter, T.; Letschert, S.; Götz, R.; Doose, S.; Danhof, S.; Einsele, H.; Sauer, M.; Hudecek, M. Super-resolution microscopy reveals ultra-low CD19 expression on myeloma cells that triggers elimination by CD19 CAR-T. Nat. Commun. 2019, 10, 3137. [Google Scholar] [CrossRef]
- Wäldchen, F.; Schlegel, J.; Götz, R.; Luciano, M.; Schnermann, M.; Doose, S.; Sauer, M. Whole-cell imaging of plasma membrane receptors by 3D lattice light-sheet dSTORM. Nat. Commun. 2020, 11, 887. [Google Scholar] [CrossRef]
- Letschert, S.; Göhler, A.; Franke, C.; Bertleff-Zieschang, N.; Memmel, E.; Doose, S.; Seibel, J.; Sauer, M. Super-Resolution Imaging of Plasma Membrane Glycans. Angew. Chem. Int. Ed. 2014, 53, 10921–10924. [Google Scholar] [CrossRef]
- Ehmann, N.; van de Linde, S.; Alon, A.; Ljaschenko, D.; Keung, X.Z.; Holm, T.; Rings, A.; DiAntonio, A.; Hallermann, S.; Ashery, U.; et al. Quantitative super-resolution imaging of Bruchpilot distinguishes active zone states. Nat. Commun. 2014, 5, 4650. [Google Scholar] [CrossRef]
- Wilkinson, F.; Helman, W.P.; Ross, A.B. Quantum yields for the photosensitized production of the lowest electronically excited singlet state of molecular oxygen in solution. J. Phys. Chem. Ref. Data 1993, 22, 113–263. [Google Scholar] [CrossRef]
- Deerinck, T.J.; Martone, M.E.; Lev-Ram, V.; Green, D.P.; Tsien, R.Y.; Spector, D.L.; Huang, S.; Ellisman, M.H. Fluorescence photooxidation with eosin: A method for high resolution immunolocalization and in situ hybridization detection for light and electron microscopy. J. Cell Biol. 1994, 126, 901–910. [Google Scholar] [CrossRef]
- Comas-Barceló, J.; Rodríguez-Amigo, B.; Abbruzzetti, S.; Rey-Puech, P.d.; Agut, M.; Nonell, S.; Viappiani, C. A self-assembled nanostructured material with photosensitising properties. RSC Adv. 2013, 3, 17874–17879. [Google Scholar] [CrossRef]
- Bianchini, P.; Cozzolino, M.; Oneto, M.; Pesce, L.; Pennacchietti, F.; Tognolini, M.; Giorgio, C.; Nonell, S.; Cavanna, L.; Delcanale, P.; et al. Hypericin—Apomyoglobin an enhanced photosensitizer complex for the treatment of tumour cells. Biomacromolecules 2019, 20, 2024–2033. [Google Scholar] [CrossRef]
- Brilkina, A.A.; Peskova, N.N.; Dudenkova, V.V.; Gorokhova, A.A.; Sokolova, E.A.; Balalaeva, I.V. Monitoring of hydrogen peroxide production under photodynamic treatment using protein sensor HyPer. J. Photochem. Photobiol. B Biol. 2018, 178, 296–301. [Google Scholar] [CrossRef]
- Ulukaya, E.; Ozdikicioglu, F.; Oral, A.Y.; Demirci, M. The MTT assay yields a relatively lower result of growth inhibition than the ATP assay depending on the chemotherapeutic drugs tested. Toxicol. In Vitro 2008, 22, 232–239. [Google Scholar] [CrossRef]
- Peng, W.; de Bruijn, H.S.; Farrell, E.; Sioud, M.; Mashayekhi, V.; Oliveira, S.; van Dam, G.M.; Roodenburg, J.L.N.; Witjes, M.J.H.; Robinson, D.J. Epidermal growth factor receptor (EGFR) density may not be the only determinant for the efficacy of EGFR-targeted photoimmunotherapy in human head and neck cancer cell lines. Lasers Surg. Med. 2018, 50, 513–522. [Google Scholar] [CrossRef]
- Shui, S.; Zhao, Z.; Wang, H.; Conrad, M.; Liu, G. Non-enzymatic lipid peroxidation initiated by photodynamic therapy drives a distinct ferroptosis-like cell death pathway. Redox Biol. 2021, 45, 102056. [Google Scholar] [CrossRef] [PubMed]
- Mishchenko, T.; Balalaeva, I.; Gorokhova, A.; Vedunova, M.; Krysko, D.V. Which cell death modality wins the contest for photodynamic therapy of cancer? Cell Death Dis. 2022, 13, 455. [Google Scholar] [CrossRef]
- Tang, D.; Chen, X.; Kang, R.; Kroemer, G. Ferroptosis: Molecular mechanisms and health implications. Cell Res. 2021, 31, 107–125. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Bao, Y.; Meng, B.; Xu, Z.; Li, S.; Wang, X.; Hou, R.; Ma, W.; Liu, D.; Zheng, J.; et al. From rough to precise: PD-L1 evaluation for predicting the efficacy of PD-1/PD-L1 blockades. Front. Immunol. 2022, 13, 920021. [Google Scholar] [CrossRef]
- Hirata, H.; Kuwatani, M.; Nakajima, K.; Kodama, Y.; Yoshikawa, Y.; Ogawa, M.; Sakamoto, N. Near-infrared photoimmunotherapy (NIR-PIT) on cholangiocarcinoma using a novel catheter device with light emitting diodes. Cancer Sci. 2021, 112, 828–838. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, H.; Pantarat, N.; Suzuki, T.; Evdokiou, A. Near-Infrared Photoimmunotherapy Using a Small Protein Mimetic for HER2-Overexpressing Breast Cancer. Int. J. Mol. Sci. 2019, 20, 5835. [Google Scholar] [CrossRef] [PubMed]
- Heryanto, Y.-D.; Hanaoka, H.; Nakajima, T.; Yamaguchi, A.; Tsushima, Y. Applying near-infrared photoimmunotherapy to B-cell lymphoma: Comparative evaluation with radioimmunotherapy in tumor xenografts. Ann. Nucl. Med. 2017, 31, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Elekonawo, F.M.K.; Bos, D.L.; Goldenberg, D.M.; Boerman, O.C.; Rijpkema, M. Carcinoembryonic antigen-targeted photodynamic therapy in colorectal cancer models. EJNMMI Res. 2019, 9, 108. [Google Scholar] [CrossRef]
- Mączyńska, J.; Raes, F.; Da Pieve, C.; Turnock, S.; Boult, J.K.R.; Hoebart, J.; Niedbala, M.; Robinson, S.P.; Harrington, K.J.; Kaspera, W.; et al. Triggering anti-GBM immune response with EGFR-mediated photoimmunotherapy. BMC Med. 2022, 20, 16. [Google Scholar]
- Burley, T.A.; Mączyńska, J.; Shah, A.; Szopa, W.; Harrington, K.J.; Boult, J.K.R.; Mrozek-Wilczkiewicz, A.; Vinci, M.; Bamber, J.C.; Kaspera, W.; et al. Near-infrared photoimmunotherapy targeting EGFR-Shedding new light on glioblastoma treatment. Int. J. Cancer 2018, 142, 2363–2374. [Google Scholar] [CrossRef]
- Driehuis, E.; Spelier, S.; Beltrán Hernández, I.; de Bree, R.; Willems, S.M.; Clevers, H.; Oliveira, S. Patient-Derived Head and Neck Cancer Organoids Recapitulate EGFR Expression Levels of Respective Tissues and Are Responsive to EGFR-Targeted Photodynamic Therapy. J. Clin. Med. 2019, 8, 1880. [Google Scholar] [CrossRef]
- Taki, S.; Matsuoka, K.; Nishinaga, Y.; Takahashi, K.; Yasui, H.; Koike, C.; Shimizu, M.; Sato, M.; Sato, K. Spatiotemporal depletion of tumor-associated immune checkpoint PD-L1 with near-infrared photoimmunotherapy promotes antitumor immunity. J. Immunother. Cancer 2021, 9, e003036. [Google Scholar] [CrossRef]
- Gong, J.; Chehrazi-Raffle, A.; Reddi, S.; Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J. Immunother. Cancer 2018, 6, 8. [Google Scholar] [CrossRef]
- Srivastava, M.K.; Zhu, L.; Harris-White, M.; Kar, U.K.; Huang, M.; Johnson, M.F.; Lee, J.M.; Elashoff, D.; Strieter, R.; Dubinett, S.; et al. Myeloid suppressor cell depletion augments antitumor activity in lung cancer. PLoS ONE 2012, 7, e40677. [Google Scholar] [CrossRef]
- Iclozan, C.; Antonia, S.; Chiappori, A.; Chen, D.T.; Gabrilovich, D. Therapeutic regulation of myeloid-derived suppressor cells and immune response to cancer vaccine in patients with extensive stage small cell lung cancer. Cancer Immunol. Immunother. 2013, 62, 909–918. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delcanale, P.; Alampi, M.M.; Mussini, A.; Fumarola, C.; Galetti, M.; Petronini, P.G.; Viappiani, C.; Bruno, S.; Abbruzzetti, S. A Photoactive Supramolecular Complex Targeting PD-L1 Reveals a Weak Correlation between Photoactivation Efficiency and Receptor Expression Levels in Non-Small-Cell Lung Cancer Tumor Models. Pharmaceutics 2023, 15, 2776. https://doi.org/10.3390/pharmaceutics15122776
Delcanale P, Alampi MM, Mussini A, Fumarola C, Galetti M, Petronini PG, Viappiani C, Bruno S, Abbruzzetti S. A Photoactive Supramolecular Complex Targeting PD-L1 Reveals a Weak Correlation between Photoactivation Efficiency and Receptor Expression Levels in Non-Small-Cell Lung Cancer Tumor Models. Pharmaceutics. 2023; 15(12):2776. https://doi.org/10.3390/pharmaceutics15122776
Chicago/Turabian StyleDelcanale, Pietro, Manuela Maria Alampi, Andrea Mussini, Claudia Fumarola, Maricla Galetti, Pier Giorgio Petronini, Cristiano Viappiani, Stefano Bruno, and Stefania Abbruzzetti. 2023. "A Photoactive Supramolecular Complex Targeting PD-L1 Reveals a Weak Correlation between Photoactivation Efficiency and Receptor Expression Levels in Non-Small-Cell Lung Cancer Tumor Models" Pharmaceutics 15, no. 12: 2776. https://doi.org/10.3390/pharmaceutics15122776
APA StyleDelcanale, P., Alampi, M. M., Mussini, A., Fumarola, C., Galetti, M., Petronini, P. G., Viappiani, C., Bruno, S., & Abbruzzetti, S. (2023). A Photoactive Supramolecular Complex Targeting PD-L1 Reveals a Weak Correlation between Photoactivation Efficiency and Receptor Expression Levels in Non-Small-Cell Lung Cancer Tumor Models. Pharmaceutics, 15(12), 2776. https://doi.org/10.3390/pharmaceutics15122776







