Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review
Abstract
1. Introduction
- Phages can be produced easily and cheaply in large quantities and easily purified. By only infecting a bacteria solution, one can obtain a large number of progeny phages.
- Bacteriophages are considered non-toxic to eukaryotes because structural elements of the virion cannot bind to eukaryotic cells [24]. There are, however, specific examples of phage internalization by eukaryotic cells. Lehti et al. demonstrated the penetration of eukaryotic cells by the E. coli phage PK1A2 in vitro [25]. The virus remained in the cells for up to 24 h but did not affect cell viability.
- The lysis of bacteria resulting from phage infection supports the inflammatory response against bacteria [26]. Therefore, phage therapies directly eliminate bacteria cells and indirectly activate humans’ immune systems.
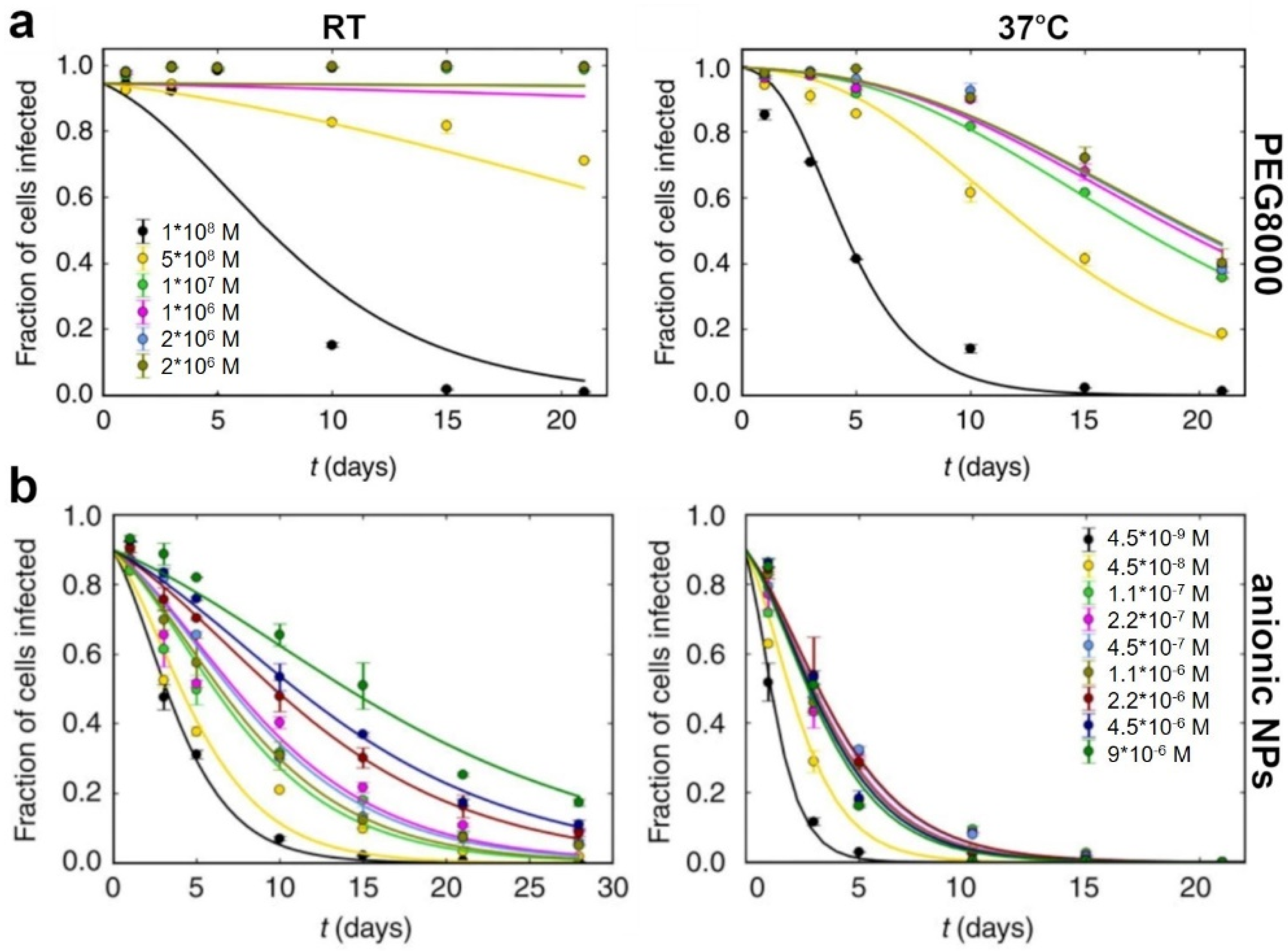
2. Polymer-Based Stabilization
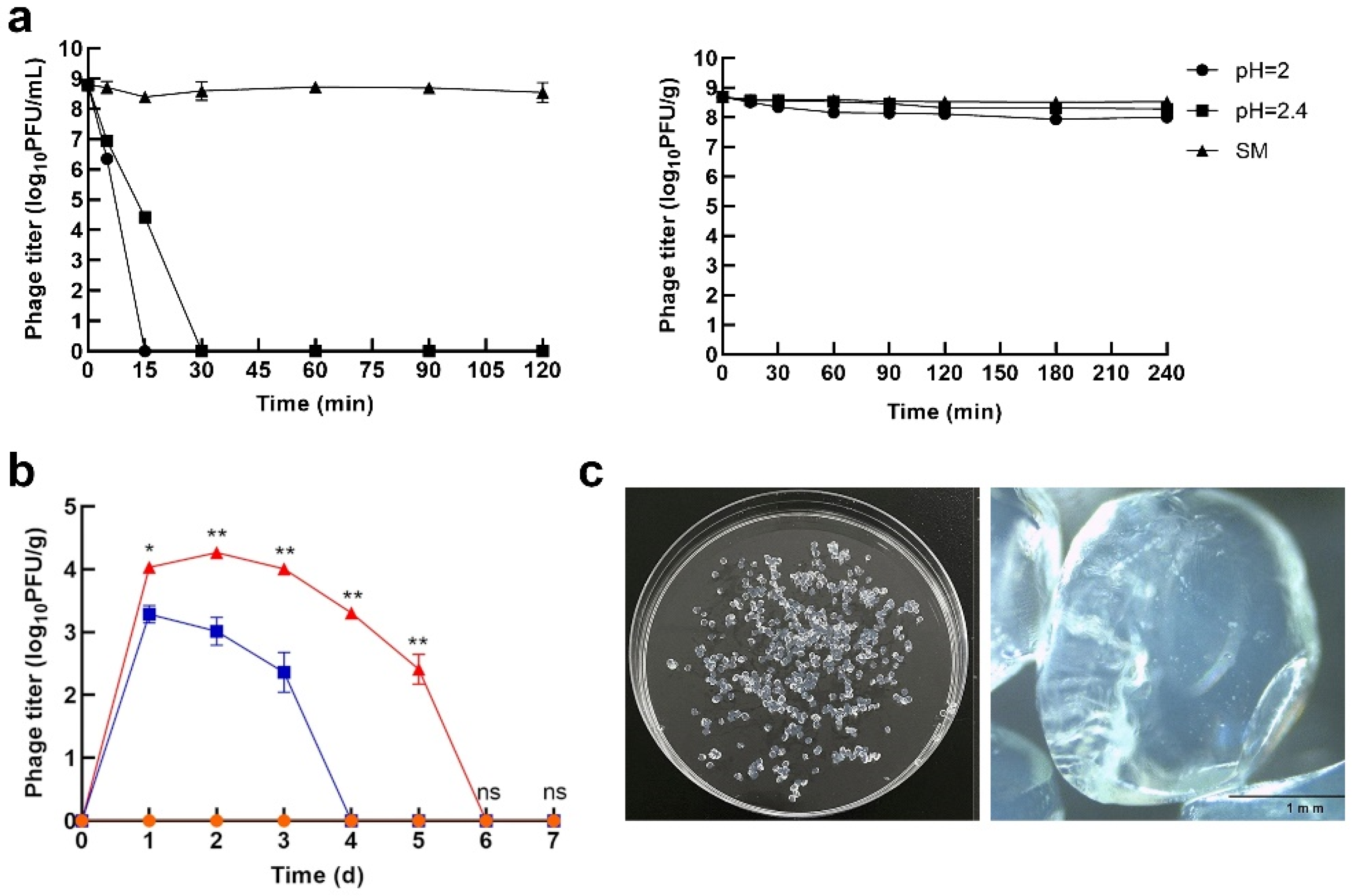
3. Encapsulation
4. Lyophilization
5. Nano-Assisted Stabilization
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hay, I.D.; Lithgow, T. Filamentous phages: Masters of a microbial sharing economy. EMBO Rep. 2019, 20, e47427. [Google Scholar] [CrossRef] [PubMed]
- Harper, D.R.; Kutter, E. Bacteriophage: Therapeutic Uses. In Encyclopedia of Life Sciences; John Wiley & Sons, Ltd.: Chichester, UK, 2003; ISBN 9780470015902. [Google Scholar]
- Cisek, A.A.; Dąbrowska, I.; Gregorczyk, K.P.; Wyżewski, Z. Phage Therapy in Bacterial Infections Treatment: One Hundred Years After the Discovery of Bacteriophages. Curr. Microbiol. 2017, 74, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Calap, P.; Georgel, P.; Bahram, S. Back to the future: Bacteriophages as promising therapeutic tools. Hla 2016, 87, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Bukhari, S.M.A.U.S.; Andleeb, S.; Ali, M.; Raza, S.; Nawaz, M.A.; Hussain, T.; Rahman, S.; Shah, S.S.A. Bacteriophages: An overview of the control strategies against multiple bacterial infections in different fields. J. Basic Microbiol. 2019, 59, 123–133. [Google Scholar] [CrossRef]
- Mackey, M.C.; Santillán, M.; Tyran-Kamińska, M.; Zeron, E.S. Lecture Notes on Mathematical Modelling in the Life Sciences Simple Mathematical Models of Gene Regulatory Dynamics; Springer: Cham, Switzerland, 2016; pp. 87–97. [Google Scholar] [CrossRef]
- Ackermann, H.W. Phage classification and characterization. Methods Mol. Biol. 2009, 501, 127–140. [Google Scholar] [CrossRef]
- Cochlan, W.P.; Wikner, J.; Steward, G.F.; Smith, D.C.; Azam, F. Spatial distribution of viruses, bacteria and chlorophyll a in neritic, oceanic and estuarine environments. Mar. Ecol. Prog. Ser. 1993, 92, 77–87. [Google Scholar] [CrossRef]
- Ackermann, H.-W. 5500 Phages examined in the electron microscope. Arch. Virol. 2007, 152, 227–243. [Google Scholar] [CrossRef]
- Dion, M.B.; Oechslin, F.; Moineau, S. Phage diversity, genomics and phylogeny. Nat. Rev. Microbiol. 2020, 18, 125–138. [Google Scholar] [CrossRef]
- Baran, G.J.; Bloomfield, V.A. Tail-fiber attachement in bacteriophage T4D studied by quasielastic light scattering–band electrophoresis. Biopolymers 1978, 17, 2015–2028. [Google Scholar] [CrossRef]
- Burton, D.R. Phage display. Immunotechnology 1995, 1, 87–94. [Google Scholar] [CrossRef]
- Loganathan, A.; Manohar, P.; Eniyan, K.; VinodKumar, C.S.; Leptihn, S.; Nachimuthu, R. Phage therapy as a revolutionary medicine against Gram-positive bacterial infections. Beni-Suef Univ. J. Basic Appl. Sci. 2021, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Mirshekari, H.; Moosavi Basri, S.M.; Bahrami, S.; Moghoofei, M.; Hamblin, M.R. Bacteriophages and phage-inspired nanocarriers for targeted delivery of therapeutic cargos. Adv. Drug Deliv. Rev. 2016, 106, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Paczesny, J.; Bielec, K. Application of bacteriophages in nanotechnology. Nanomaterials 2020, 10, 1944. [Google Scholar] [CrossRef] [PubMed]
- Bao, Q.; Li, X.; Han, G.; Zhu, Y.; Mao, C.; Yang, M. Phage-based vaccines. Adv. Drug Deliv. Rev. 2019, 145, 40–56. [Google Scholar] [CrossRef]
- Punina, N.V.; Makridakis, N.M.; Remnev, M.A.; Topunov, A.F. Whole-genome sequencing targets drug-resistant bacterial infections. Hum. Genom. 2015, 9, 19. [Google Scholar] [CrossRef]
- Elsland, D.; Neefjes, J. Bacterial infections and cancer. EMBO Rep. 2018, 19, e46632. [Google Scholar] [CrossRef]
- Castañer, O.; Schröder, H. Response to: Comment on “The Gut Microbiome Profile in Obesity: A Systematic Review”. Int. J. Endocrinol. 2018, 2018, 9109451. [Google Scholar] [CrossRef]
- World Health Organization. Report on the Burden of Endemic Health Care-Associated Infection Worldwide; WHO Press: Geneva, Switzerland, 2011. [Google Scholar]
- Cimiotti, J.P.; Aiken, L.H.; Sloane, D.M.; Wu, E.S. Nurse staffing, burnout, and health care-associated infection. Am. J. Infect. Control 2012, 40, 486–490. [Google Scholar] [CrossRef]
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; de Silva, N.R.; Gargouri, N.; et al. World Health Organization Global Estimates and Regional Comparisons of the Burden of Foodborne Disease in 2010. PLOS Med. 2015, 12, e1001923. [Google Scholar] [CrossRef]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States-Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef]
- Van Belleghem, J.D.; Dąbrowska, K.; Vaneechoutte, M.; Barr, J.J.; Bollyky, P.L. Interactions between bacteriophage, bacteria, and the mammalian immune system. Viruses 2019, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Lehti, T.A.; Pajunen, M.I.; Skog, M.S.; Finne, J. Internalization of a polysialic acid-binding Escherichia coli bacteriophage into eukaryotic neuroblastoma cells. Nat. Commun. 2017, 8, 1915. [Google Scholar] [CrossRef] [PubMed]
- Hess, K.L.; Jewell, C.M. Phage display as a tool for vaccine and immunotherapy development. Bioeng. Transl. Med. 2020, 5, e10142. [Google Scholar] [CrossRef] [PubMed]
- de Jonge, P.A.; Nobrega, F.L.; Brouns, S.J.J.; Dutilh, B.E. Molecular and Evolutionary Determinants of Bacteriophage Host Range. Trends Microbiol. 2019, 27, 51–63. [Google Scholar] [CrossRef]
- Raza, S.; Matuła, K.; Karoń, S.; Paczesny, J. Resistance and adaptation of bacteria to non-antibiotic antibac-terial agents: Physical stressors, nanoparticles, and bacterio-phages. Antibiotics 2021, 10, 435. [Google Scholar] [CrossRef]
- Stanley, S.Y.; Maxwell, K.L. Phage-Encoded Anti-CRISPR Defenses. Annu. Rev. Genet. 2018, 52, 445–464. [Google Scholar] [CrossRef]
- Sulakvelidze, A.; Alavidze, Z.; Glenn Morris, J. Bacteriophage Therapy. Antimicrob. Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef]
- Summers, W.C. The strange history of phage therapy. Bacteriophage 2012, 2, 130–133. [Google Scholar] [CrossRef]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Ventola, C.L. The Antibiotic Resistance Crisis Part 2: Management Strategies and New Agents. Pharm. Ther. 2015, 40, 344–352. [Google Scholar]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, T. Georgia: An unlikely stronghold for bacteriophage therapy. Lancet 2005, 365, 2166–2167. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage treatment of human infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef] [PubMed]
- McCallin, S.; Alam Sarker, S.; Barretto, C.; Sultana, S.; Berger, B.; Huq, S.; Krause, L.; Bibiloni, R.; Schmitt, B.; Reuteler, G.; et al. Safety analysis of a Russian phage cocktail: From MetaGenomic analysis to oral application in healthy human subjects. Virology 2013, 443, 187–196. [Google Scholar] [CrossRef]
- Wright, A.; Hawkins, C.H.; Änggård, E.E.; Harper, D.R. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; A preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef]
- Speck, P.; Smithyman, A. Safety and efficacy of phage therapy via the intravenous route. FEMS Microbiol. Lett. 2015, 363, fnv242. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.-A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2018, 19, 35–45. [Google Scholar] [CrossRef]
- Voelker, R. FDA Approves Bacteriophage Trial. Jama 2019, 321, 638. [Google Scholar] [CrossRef]
- Łusiak-Szelachowska, M.; Zaczek, M.; Weber-Dabrowska, B.; Miȩdzybrodzki, R.; Kłak, M.; Fortuna, W.; Letkiewicz, S.; Rogóz, P.; Szufnarowski, K.; Jończyk-Matysiak, E.; et al. Phage neutralization by sera of patients receiving phage therapy. Viral Immunol. 2014, 27, 295–304. [Google Scholar] [CrossRef]
- Górski, A.; Miedzybrodzki, R.; Borysowski, J.; Dabrowska, K.; Wierzbicki, P.; Ohams, M.; Korczak-Kowalska, G.; Olszowska-Zaremba, N.; Łusiak-Szelachowska, M.; Kłak, M.; et al. Phage as a Modulator of Immune Responses. Practical Implications for Phage Therapy. Adv. Virus Res. 2012, 83, 41–71. [Google Scholar]
- Kazi, M.; Annapure, U.S. Bacteriophage biocontrol of foodborne pathogens. J. Food Sci. Technol. 2016, 53, 1355–1362. [Google Scholar] [CrossRef] [PubMed]
- Loc-Carrillo, C.; Abedon, S.T. Pros and cons of phage therapy. Bacteriophage 2011, 1, 111–114. [Google Scholar] [CrossRef]
- Pujato, S.A.; Quiberoni, A.; Mercanti, D.J. Bacteriophages on dairy foods. J. Appl. Microbiol. 2019, 126, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Vonasek, E.L.; Choi, A.H.; Sanchez, J.; Nitin, N. Incorporating Phage Therapy into WPI Dip Coatings for Applications on Fresh Whole and Cut Fruit and Vegetable Surfaces. J. Food Sci. 2018, 83, 1871–1879. [Google Scholar] [CrossRef] [PubMed]
- Cadieux, B.; Colavecchio, A.; Jeukens, J.; Freschi, L.; Emond-Rheault, J.G.; Kukavica-Ibrulj, I.; Levesque, R.C.; Bekal, S.; Chandler, J.C.; Coleman, S.M.; et al. Prophage induction reduces Shiga toxin producing Escherichia coli (STEC) and Salmonella enterica on tomatoes and spinach: A model study. Food Control 2018, 89, 250–259. [Google Scholar] [CrossRef]
- Alves, D.; Marques, A.; Milho, C.; Costa, M.J.; Pastrana, L.M.; Cerqueira, M.A.; Sillankorva, S.M. Bacteriophage ϕIBB-PF7A loaded on sodium alginate-based films to prevent microbial meat spoilage. Int. J. Food Microbiol. 2019, 291, 121–127. [Google Scholar] [CrossRef]
- Huang, K.; Nitin, N. Edible bacteriophage based antimicrobial coating on fish feed for enhanced treatment of bacterial infections in aquaculture industry. Aquaculture 2019, 502, 18–25. [Google Scholar] [CrossRef]
- Sarhan, W.A.; Azzazy, H.M.E. Phage approved in food, why not as a therapeutic? Expert Rev. Anti. Infect. Ther. 2015, 13, 91–101. [Google Scholar] [CrossRef]
- Moye, Z.D.; Woolston, J.; Sulakvelidze, A. Bacteriophage applications for food production and processing. Viruses 2018, 10, 205. [Google Scholar] [CrossRef]
- Svircev, A.; Roach, D.; Castle, A. Framing the future with bacteriophages in agriculture. Viruses 2018, 10, 218. [Google Scholar] [CrossRef]
- Mamane-Gravetz, H.; Linden, K.G.; Cabaj, A.; Sommer, R. Spectral sensitivity of Bacillus subtilis spores and MS2 coliphage for validation testing of ultraviolet reactors for water disinfection. Environ. Sci. Technol. 2005, 39, 7845–7852. [Google Scholar] [CrossRef] [PubMed]
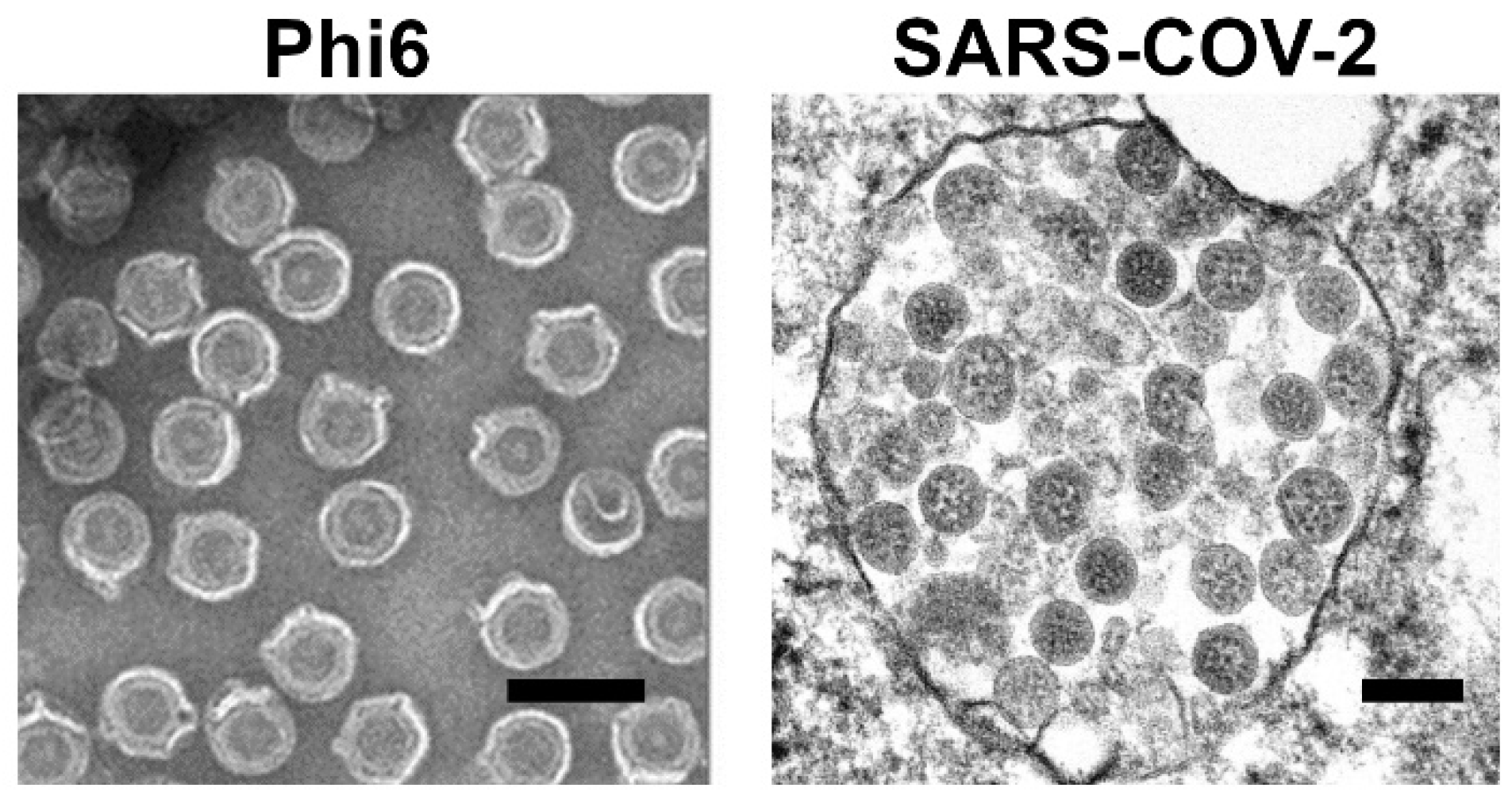
- Fedorenko, A.; Grinberg, M.; Orevi, T.; Kashtan, N. Survival of the enveloped bacteriophage Phi6 (a surrogate for SARS-CoV-2) in evaporated saliva microdroplets deposited on glass surfaces. Sci. Rep. 2020, 10, 22419. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, C.; Mu, Y.; Houston, H.; Martinez-Smith, M.; Noble-Wang, J.; Coulliette-Salmond, A.; Rose, L. Persistence of bacteriophage phi 6 on porous and nonporous surfaces and the potential for its use as an ebola virus or coronavirus surrogate. Appl. Environ. Microbiol. 2020, 86, e01482-20. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Cai, C.; Dai, X. Interactions between virus surrogates and sewage sludge vary by viral analyte: Recovery, persistence, and sorption. Water Res. 2022, 210, 117995. [Google Scholar] [CrossRef]
- Shirasaki, N.; Matsushita, T.; Matsui, Y.; Urasaki, T.; Ohno, K. Comparison of behaviors of two surrogates for pathogenic waterborne viruses, bacteriophages Qβ and MS2, during the aluminum coagulation process. Water Res. 2009, 43, 605–612. [Google Scholar] [CrossRef]
- Havelaar, A.H.; Meulemans, C.C.E.; Pot-Hogeeoom, W.M.; Koster, J. Inactivation of bacteriophage MS2 in wastewater effluent with monochromatic and polychromatic ultraviolet light. Water Res. 1990, 24, 1387–1393. [Google Scholar] [CrossRef]
- String, G.M.; White, M.R.; Gute, D.M.; Mühlberger, E.; Lantagne, D.S. Selection of a SARS-CoV-2 Surrogate for Use in Surface Disinfection Efficacy Studies with Chlorine and Antimicrobial Surfaces. Environ. Sci. Technol. Lett. 2021, 8, 995–1001. [Google Scholar] [CrossRef]
- Davis, R.; Zivanovic, S.; D’Souza, D.H.; Davidson, P.M. Effectiveness of chitosan on the inactivation of enteric viral surrogates. Food Microbiol. 2012, 32, 57–62. [Google Scholar] [CrossRef]
- de Vries, C.R.; Chen, Q.; Demirdjian, S.; Kaber, G.; Khosravi, A.; Liu, D.; Van Belleghem, J.D.; Bollyky, P.L. Phages in vaccine design and immunity; mechanisms and mysteries. Curr. Opin. Biotechnol. 2021, 68, 160–165. [Google Scholar] [CrossRef]
- Bazan, J.; Całkosiñski, I.; Gamian, A. Phage displaya powerful technique for immunotherapy: 1. Introduction and potential of therapeutic applications. Hum. Vaccines Immunother. 2012, 8, 1817–1828. [Google Scholar] [CrossRef]
- Wood, J.P.; Richter, W.; Sunderman, M.; Calfee, M.W.; Serre, S.; Mickelsen, L. Evaluating the Environmental Persistence and Inactivation of MS2 Bacteriophage and the Presumed Ebola Virus Surrogate Phi6 Using Low Concentration Hydrogen Peroxide Vapor. Environ. Sci. Technol. 2020, 54, 3581–3590. [Google Scholar] [CrossRef] [PubMed]
- Block, K.A.; Trusiak, A.; Katz, A.; Gottlieb, P.; Alimova, A.; Wei, H.; Morales, J.; Rice, W.J.; Steiner, J.C. Disassembly of the cystovirus φ{symbol}6 envelope by montmorillonite clay. Microbiologyopen 2014, 3, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, C.S.; Miller, S.E.; Martines, R.B.; Bullock, H.A.; Zaki, S.R. Electron microscopy of SARS-CoV-2: A challenging task. Lancet 2020, 395, e99. [Google Scholar] [CrossRef]
- Brigati, J.R.; Petrenko, V.A. Thermostability of landscape phage probes. Anal. Bioanal. Chem. 2005, 382, 1346–1350. [Google Scholar] [CrossRef] [PubMed]
- Nobrega, F.L.; Costa, A.R.; Santos, J.F.; Siliakus, M.F.; Van Lent, J.W.M.; Kengen, S.W.M.; Azeredo, J.; Kluskens, L.D. Genetically manipulated phages with improved pH resistance for oral administration in veterinary medicine. Sci. Rep. 2016, 6, 39235. [Google Scholar] [CrossRef]
- Olofsson, L.; Ankarloo, J.; Nicholls, I.A. Phage viability in organic media: Insights into phage stability. J. Mol. Recognit. 1998, 11, 91–93. [Google Scholar] [CrossRef]
- Olofsson, L.; Ankarloo, J.; Andersson, P.O.; Nicholls, I.A. Filamentous bacteriophage stability in non-aqueous media. Chem. Biol. 2001, 8, 661–671. [Google Scholar] [CrossRef]
- Jończyk-Matysiak, E.; Łodej, N.; Kula, D.; Owczarek, B.; Orwat, F.; Międzybrodzki, R.; Neuberg, J.; Bagińska, N.; Weber-Dąbrowska, B.; Górski, A. Factors determining phage stability/activity: Challenges in practical phage application. Expert Rev. Anti. Infect. Ther. 2019, 17, 583–606. [Google Scholar] [CrossRef]
- Malik, D.J.; Sokolov, I.J.; Vinner, G.K.; Mancuso, F.; Cinquerrui, S.; Vladisavljevic, G.T.; Clokie, M.R.J.; Garton, N.J.; Stapley, A.G.F.; Kirpichnikova, A. Formulation, stabilisation and encapsulation of bacteriophage for phage therapy. Adv. Colloid Interface Sci. 2017, 249, 100–133. [Google Scholar] [CrossRef]
- McCrum, N.G.; Buckley, C.P.; Buckley, C.P.; Bucknall, C.B. Principles of Polymer Engineering; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Walter, T.S.; Ren, J.; Tuthill, T.J.; Rowlands, D.J.; Stuart, D.I.; Fry, E.E. A plate-based high-throughput assay for virus stability and vaccine formulation. J. Virol. Methods 2012, 185, 166–170. [Google Scholar] [CrossRef]
- Timasheff, S.N. The control of protein stability and association by weak water interactions with water: How do solvents affect these processes? Annu. Rev. Biophys. Biomol. Struct. 1993, 22, 67–69. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Woods, C.E.; Litowski, J.R.; Roschen, L.A.; Gadgil, H.S.; Razinkov, V.I.; Kerwin, B.A. Effect of sugar molecules on the viscosity of high concentration monoclonal antibody solutions. Pharm. Res. 2011, 28, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Timasheff, S.N. The stabilization of proteins by sucrose. J. Biol. Chem. 1981, 256, 7193–7201. [Google Scholar] [CrossRef]
- Liu, F.F.; Ji, L.; Zhang, L.; Dong, X.Y.; Sun, Y. Molecular basis for polyol-induced protein stability revealed by molecular dynamics simulations. J. Chem. Phys. 2010, 132, 225103. [Google Scholar] [CrossRef]
- Alcock, R.; Cottingham, M.G.; Rollier, C.S.; Furze, J.; De Costa, S.D.; Hanlon, M.; Spencer, A.J.; Honeycutt, J.D.; Wyllie, D.H.; Gilbert, S.C.; et al. Long-term thermostabilization of live poxviral and adenoviral vaccine vectors at supraphysiological temperatures in carbohydrate glass. Sci. Transl. Med. 2010, 2, 19ra12. [Google Scholar] [CrossRef]
- Ausar, S.F.; Espina, M.; Brock, J.; Thyagarayapuran, N.; Repetto, R.; Khandke, L.; Middaugh, C.R. High-throughput screening of stabilizers for respirator/syncytial virus: Identification of stabilizers and their effects on the conformational thermostability of viral particles. Hum. Vaccin. 2007, 3, 94–103. [Google Scholar] [CrossRef]
- Wiggan, O.; Livengood, J.A.; Silengo, S.J.; Kinney, R.M.; Osorio, J.E.; Huang, C.Y.H.; Stinchcomb, D.T. Novel formulations enhance the thermal stability of live-attenuated flavivirus vaccines. Vaccine 2011, 29, 7456–7462. [Google Scholar] [CrossRef]
- Bailly, C.; Qu, X.; Anizon, F.; Prudhomme, M.; Riou, J.F.; Chaires, J.B. Enhanced binding to DNA and topoisomerase I inhibition by an analog of the antitumor antibiotic rebeccamycin containing an amino sugar residue. Mol. Pharmacol. 1999, 55, 377–385. [Google Scholar] [CrossRef]
- Kanta, C.; Siber, G.R.; Leszczynski, J.; Gupta, R.K. Stabilization of respiratory syncytial virus ( RSV ) against thermal inactivation and freeze-thaw cycles for development and control of RSV vaccines and immune globulin. Vaccine 1996, 14, 1417–1420. [Google Scholar]
- Rexroad, J.; Wiethoff, C.M.; Green, A.P.; Kierstead, T.D.; Scott, M.O.; Middaugh, C.R. Structural stability of adenovirus type 5. J. Pharm. Sci. 2003, 92, 665–678. [Google Scholar] [CrossRef]
- Stewart, M.; Ward, S.J.; Drew, J. Use of adenovirus as a model system to illustrate a simple method using standard equipment and inexpensive excipients to remove live virus dependence on the cold-chain. Vaccine 2014, 32, 2931–2938. [Google Scholar] [CrossRef] [PubMed]
- Balcão, V.M.; Moreira, A.R.; Moutinho, C.G.; Chaud, M.V.; Tubino, M.; Vila, M.M.D.C. Structural and functional stabilization of phage particles in carbohydrate matrices for bacterial biosensing. Enzym. Microb. Technol. 2013, 53, 55–69. [Google Scholar] [CrossRef] [PubMed]
- Leung, V.; Szewczyk, A.; Chau, J.; Hosseinidoust, Z.; Groves, L.; Hawsawi, H.; Anany, H.; Griffiths, M.W.; Monsur Ali, M.; Filipe, C.D.M. Long-Term Preservation of Bacteriophage Antimicrobials Using Sugar Glasses. ACS Biomater. Sci. Eng. 2018, 4, 3802–3808. [Google Scholar] [CrossRef] [PubMed]
- Trempe, J.F.; Morin, F.G.; Xia, Z.; Marchessault, R.H.; Gehring, K. Characterization of polyacrylamide-stabilized Pf1 phage liquid crystals for protein NMR spectroscopy. J. Biomol. NMR 2002, 22, 83–87. [Google Scholar] [CrossRef]
- Qi, Y.; Chilkoti, A. Protein-polymer conjugation-moving beyond PEGylation. Curr. Opin. Chem. Biol. 2015, 28, 181–193. [Google Scholar] [CrossRef]
- Lewis, G.D.; Metcalf, T.G. Polyethylene Glycol Precipitation for Recovery of Pathogenic Viruses, Including Hepatitis A Virus and Human Rotavirus, from Oyster, Water, and Sediment Samples. Appl. Environ. Microbiol. 1998, 54, 1983–1988. [Google Scholar] [CrossRef]
- Goodridge, L. Designing Phage Therapeutics. Curr. Pharm. Biotechnol. 2010, 11, 15–27. [Google Scholar] [CrossRef]
- Kim, K.P.; Cha, J.D.; Jang, E.H.; Klumpp, J.; Hagens, S.; Hardt, W.D.; Lee, K.Y.; Loessner, M.J. PEGylation of bacteriophages increases blood circulation time and reduces T-helper type 1 immune response. Microb. Biotechnol. 2008, 1, 247–257. [Google Scholar] [CrossRef]
- Viertel, T.M.; Ritter, K.; Horz, H.P. Viruses versus bacteria-novel approaches to phage therapy as a tool against multidrug-resistant pathogens. J. Antimicrob. Chemother. 2014, 69, 2326–2336. [Google Scholar] [CrossRef]
- Resch, G.; Moreillon, P.; Fischetti, V.A. PEGylating a bacteriophage endolysin inhibits its bactericidal activity. AMB Express 2011, 1, 29. [Google Scholar] [CrossRef]
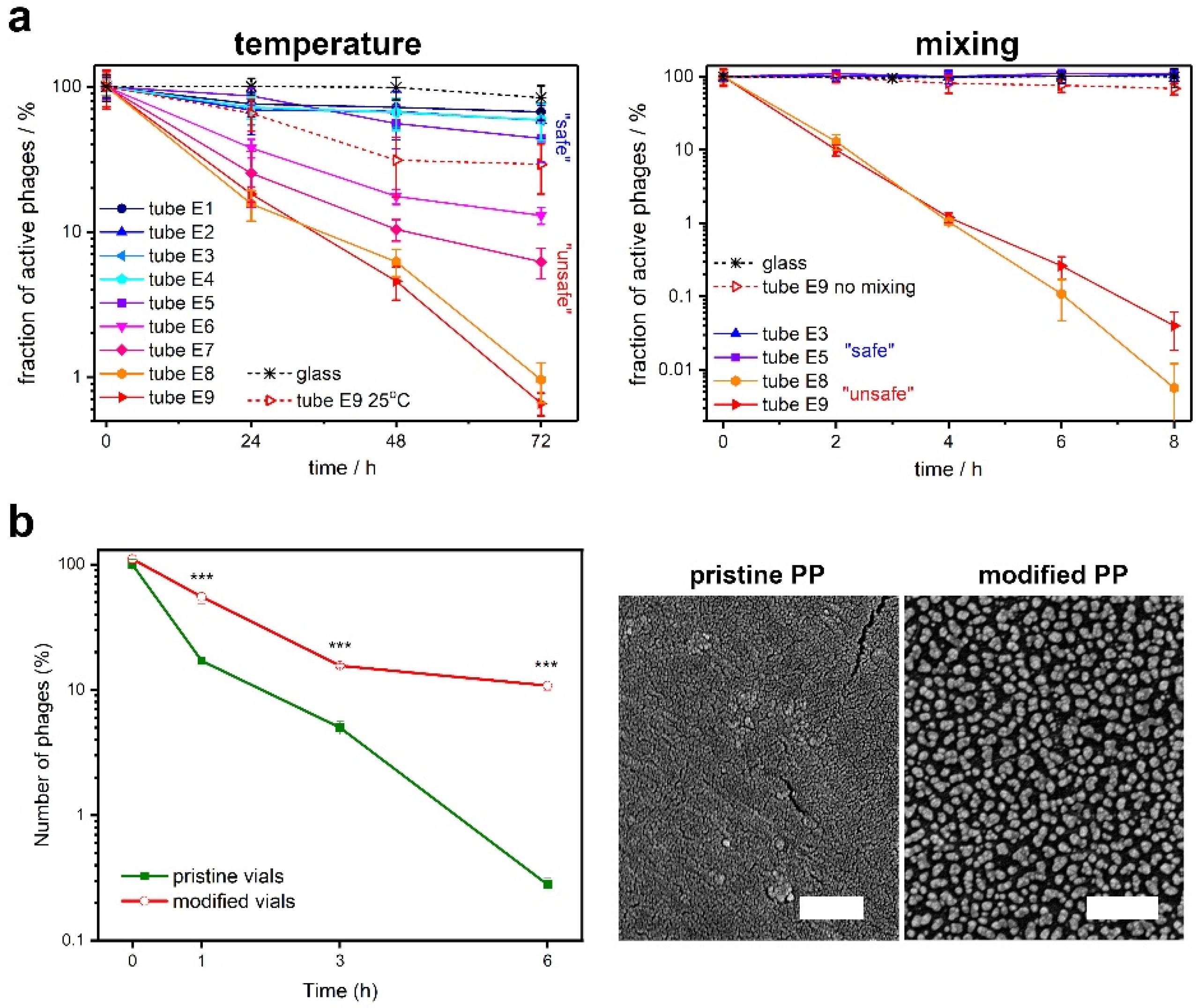
- Richter, Ł.; Księżarczyk, K.; Paszkowska, K.; Janczuk-Richter, M.; Niedziółka-Jönsson, J.; Gapiński, J.; Łoś, M.; Hołyst, R.; Paczesny, J.; Richter, Ł.; et al. Adsorption of bacteriophages on polypropylene labware affects reproducibility of phage research. W Recenzji 2021, 11, 7387. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, L.; Roupioz, Y.; Marcoux, P.R. Container Material Dictates Stability of Bacteriophage Suspensions: Light Scattering & Infectivity Measurements Reveal Mechanisms of Infectious Titer Decay. J. Appl. Microbiol. 2022, 133, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Wdowiak, M.; Ochirbat, E.; Paczesny, J. Gold—polyoxoborates nanocomposite prohibits adsorption of bacteriophages on inner surfaces of polypropylene labware and protects samples from bacterial and yeast infections. Viruses 2021, 13, 1206. [Google Scholar] [CrossRef] [PubMed]
- Loh, B.; Gondil, V.S.; Manohar, P.; Khan, F.M.; Yang, H.; Leptihn, S. Encapsulation and Delivery of Therapeutic Phages. Appl. Environ. Microbiol. 2021, 87, e01979-20. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Menendez, E.; Fernandez, L.; Gutierrez, D.; Rodríguez, A.; Martínez, B.; GarcíaI, P. Comparative analysis of different preservation techniques for the storage of Staphylococcus phages aimed for the industrial development of phage-based antimicrobial products. PLoS ONE 2018, 13, e0205728. [Google Scholar] [CrossRef]
- Śliwka, P.; Mituła, P.; Mituła, A.; Skaradziński, G.; Choińska-Pulit, A.; Niezgoda, N.; Weber-Dąbrowska, B.; Żaczek, M.; Skaradzińska, A. Encapsulation of bacteriophage T4 in mannitol-alginate dry macrospheres and survival in simulated gastrointestinal conditions. Lwt 2019, 99, 238–243. [Google Scholar] [CrossRef]
- Ma, Y.; Pacan, J.C.; Wang, Q.; Sabour, P.M.; Huang, X.; Xu, Y. Enhanced alginate microspheres as means of oral delivery of bacteriophage for reducing staphylococcus aureus intestinal carriage. Food Hydrocoll. 2012, 26, 434–440. [Google Scholar] [CrossRef]
- Briot, T.; Kolenda, C.; Ferry, T.; Medina, M.; Laurent, F.; Leboucher, G.; Pirot, F. Paving the way for phage therapy using novel drug delivery approaches. J. Control. Release 2022, 347, 414–424. [Google Scholar] [CrossRef]
- Pacios-michelena, S.; Rodríguez-herrera, R.; Ramos-gonzález, R.; Ilina, A.; Flores-gallegos, A.C.; Enmanuel, J.; Peña, D. Effect of encapsulation and natural polyphenolic compounds on bacteriophage stability and activity on Escherichia coli in Lactuca sativa L. var. longifolia. J. Food Saf. 2022. [Google Scholar] [CrossRef]
- Abdelsattar, A.S.; Abdelrahman, F.; Dawoud, A.; Connerton, I.F.; El-Shibiny, A. Encapsulation of E. coli phage ZCEC5 in chitosan–alginate beads as a delivery system in phage therapy. AMB Express 2019, 9, 87. [Google Scholar] [CrossRef]
- Chadha, P.; Katare, O.P.; Chhibber, S. Liposome loaded phage cocktail: Enhanced therapeutic potential in resolving Klebsiella pneumoniae mediated burn wound infections. Burns 2017, 43, 1532–1543. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Xu, J.; Liu, B.; Zhao, Y.D.; Yu, L.; Chen, W. Controlled release of metal phenolic network protected phage for treating bacterial infection. Nanotechnology 2022, 33, 165102. [Google Scholar] [CrossRef] [PubMed]
- Colom, J.; Cano-Sarabia, M.; Otero, J.; Aríñez-Soriano, J.; Cortés, P.; Maspoch, D.; Llagostera, M. Microencapsulation with alginate/CaCO 3: A strategy for improved phage therapy. Sci. Rep. 2017, 7, 41441. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Li, J.; Huang, H.; Wang, Y.; Qian, X.; Ren, J.; Xue, F.; Dai, J.; Tang, F. Microencapsulated phages show prolonged stability in gastrointestinal environments and high therapeutic efficiency to treat Escherichia coli O157:H7 infection. Vet. Res. 2021, 52, 118. [Google Scholar] [CrossRef]
- Desobry, S.A.; Netto, F.M.; Labuza, T.P. Comparison of Spray-drying, Drum-drying and Freeze-drying for b -Carotene Encapsulation and Preservation. J. Food Sci. 1997, 62, 1158–1162. [Google Scholar] [CrossRef]
- Patel, R.P.; Patel, M.P.; Suthar, A.M. Spray drying technology: An Overview. Indian J. Sci. Technol. 2009, 2, 44–48. [Google Scholar] [CrossRef]
- Lewicki, P.P. Design of hot air drying for better foods. Trends Food Sci. Technol. 2006, 17, 153–163. [Google Scholar] [CrossRef]
- Pala, M. Hot-air drying characteristics of red pepper. J. Food Eng. 2002, 55, 331–335. [Google Scholar]
- Kaveh, M.; Abbaspour-Gilandeh, Y.; Chen, G. Drying kinetic, quality, energy and exergy performance of hot air-rotary drum drying of green peas using adaptive neuro-fuzzy inference system. Food Bioprod. Process. 2020, 124, 168–183. [Google Scholar] [CrossRef]
- Adali, M.B.; Barresi, A.A.; Boccardo, G.; Pisano, R. Spray freeze-drying as a solution to continuous manufacturing of pharmaceutical products in bulk. Processes 2020, 8, 709. [Google Scholar] [CrossRef]
- Ergin, F. Effect of freeze drying, spray drying and electrospraying on the morphological, thermal, and structural properties of powders containing phage Felix O1 and activity of phage Felix O1 during storage. Powder Technol. 2022, 404, 117516. [Google Scholar] [CrossRef]
- Engel HW, B.; Smith, L.; Berwald, L.G. The Preservation of Mycobacteriophages by Means of Freeze Drying. Am. Rev. Respir. Dis. 1973, 109, 561–566. [Google Scholar]
- Clark, W.A.; Geary, D. Preservation of bacteriophages by freezing and freeze-drying. Cryobiology 1973, 10, 351–360. [Google Scholar] [CrossRef]
- Cox, C.S.; Harris, W.J.; Lee, J. Viability and electron microscope studies of phages T3 and T7 subjected to freeze drying, freeze thawing and aerosolization. J. Gen. Microbiol. 1974, 81, 207–215. [Google Scholar] [CrossRef][Green Version]
- Shapira, A.; Kohn, A. The effects of freeze-drying on bacteriophage T4. Cryobiology 1974, 11, 452–464. [Google Scholar] [CrossRef]
- El Haddad, L.; Lemay, M.J.; Khalil, G.E.; Moineau, S.; Champagne, C.P. Microencapsulation of a Staphylococcus phage for concentration and long-term storage. Food Microbiol. 2018, 76, 304–309. [Google Scholar] [CrossRef]
- Merabishvili, M.; Vervaet, C.; Pirnay, J.; De Vos, D.; Verbeken, G.; Mast, J.; Chanishvili, N.; Vaneechoutte, M. Stability of Staphylococcus aureus Phage ISP after freeze-drying (lyophilization). PLoS ONE 2013, 8, e68797. [Google Scholar] [CrossRef]
- Dini, C.; de Urraza, P.J. Effect of buffer systems and disaccharides concentration on Podoviridae coliphage stability during freeze drying and storage. Cryobiology 2013, 66, 339–342. [Google Scholar] [CrossRef]
- Zhang, Y.; Peng, X.; Zhang, H.; Watts, A.B.; Ghosh, D. Manufacturing and ambient stability of shelf freeze dried bacteriophage powder formulations. Int. J. Pharm. 2018, 542, 1–7. [Google Scholar] [CrossRef]
- Puapermpoonsiri, U.; Ford, S.J.; van der Walle, C.F. Stabilization of bacteriophage during freeze drying. Int. J. Pharm. 2010, 389, 168–175. [Google Scholar] [CrossRef]
- Petsong, K.; Benjakul, S.; Vongkamjan, K. Optimization of wall material for phage encapsulation via freeze-drying and antimicrobial efficacy of microencapsulated phage against Salmonella. J. Food Sci. Technol. 2021, 58, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.Y.; Wong, J.; Mathai, A.; Morales, S.; Kutter, E.; Britton, W.; Li, J.; Chan, H.K. Production of highly stable spray dried phage formulations for treatment of Pseudomonas aeruginosa lung infection. Eur. J. Pharm. Biopharm. 2017, 121, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Vandenheuvel, D.; Singh, A.; Vandersteegen, K.; Klumpp, J.; Lavigne, R.; Van Den Mooter, G. Feasibility of spray drying bacteriophages into respirable powders to combat pulmonary bacterial infections. Eur. J. Pharm. Biopharm. 2013, 84, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Vandenheuvel, D.; Meeus, J.; Lavigne, R.; Van Den Mooter, G. Instability of bacteriophages in spray-dried trehalose powders is caused by crystallization of the matrix. Int. J. Pharm. 2014, 472, 202–205. [Google Scholar] [CrossRef]
- Leung, S.S.Y.; Parumasivam, T.; Nguyen, A.; Gengenbach, T.; Carter, E.A.; Carrigy, N.B.; Wang, H.; Vehring, R.; Finlay, W.H.; Morales, S.; et al. Effect of storage temperature on the stability of spray dried bacteriophage powders. Eur. J. Pharm. Biopharm. 2018, 127, 213–222. [Google Scholar] [CrossRef]
- Chang, R.Y.K.; Kwok, P.C.L.; Khanal, D.; Morales, S.; Kutter, E.; Li, J.; Chan, H.K. Inhalable bacteriophage powders: Glass transition temperature and bioactivity stabilization. Bioeng. Transl. Med. 2020, 5, e10159. [Google Scholar] [CrossRef]
- Carrigy, N.B.; Liang, L.; Wang, H.; Kariuki, S.; Nagel, T.E.; Connerton, I.F.; Vehring, R. Trileucine and Pullulan Improve Anti-Campylobacter Bacteriophage Stability in Engineered Spray-Dried Microparticles. Ann. Biomed. Eng. 2020, 48, 1169–1180. [Google Scholar] [CrossRef]
- Anany, H.; Chen, W.; Pelton, R.; Griffiths, M.W. Biocontrol of Listeria monocytogenes and Escherichia coli O157:H7 in meat by using phages immobilized on modified cellulose membranes. Appl. Environ. Microbiol. 2011, 77, 6379–6387. [Google Scholar] [CrossRef]
- Malik, D.J. Bacteriophage Encapsulation Using Spray Drying for Phage Therapy. Bact. Viruses Exploit. Biocontrol Ther. 2021, 40, 303–316. [Google Scholar] [CrossRef]
- Ly, A.; Carrigy, N.B.; Wang, H.; Harrison, M.; Sauvageau, D.; Martin, A.R.; Vehring, R.; Finlay, W.H. Atmospheric spray freeze drying of sugar solution with phage D29. Front. Microbiol. 2019, 10, 488. [Google Scholar] [CrossRef]
- Mumenthaler, M.; Leuenberger, H. Atmospheric spray-freeze drying: A suitable alternative in freeze-drying technology. Int. J. Pharm. 1991, 72, 97–110. [Google Scholar] [CrossRef]
- Chang, R.Y.K.; Wallin, M.; Lin, Y.; Leung, S.S.Y.; Wang, H.; Morales, S.; Chan, H.K. Phage therapy for respiratory infections. Adv. Drug Deliv. Rev. 2018, 133, 76–86. [Google Scholar] [CrossRef]
- Leung, S.S.Y.; Parumasivam, T.; Gao, F.G.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Britton, W.J.; Chan, H. Production of inhalation phage powders using spray freeze drying and spray drying techniques for treatment of respiratory infections1. Friman VP, Ghoul M, Molin S, Johansen HK, Buckling, A. Pseudomonas aeruginosa Adaptation to Lungs of Cystic Fibrosis Pati. PLoS ONE 2017, 33, 1486–1496. [Google Scholar] [CrossRef]
- Li, M.; Chang RY, K.; Lin, Y.; Morales, S.; Kutter, E.; Chan, H.K. Phage Cocktail Powders for Psedomonas aeruginosa respiratory infections Phage Cocktail Powder for Pseudomonas aeruginosa Respiratory Infections. Int. J. Pharm. 2021, 596, 120200. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.Y.K.; Wallin, M.; Kutter, E.; Morales, S.; Britton, W.; Li, J.; Chan, H.K. Storage stability of inhalable phage powders containing lactose at ambient conditions. Int. J. Pharm. 2019, 560, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.Y.K.; Chen, K.; Wang, J.; Wallin, M.; Britton, W.; Morales, S.; Kutter, E.; Li, J.; Chana, H.-K. Proof-of-principle study in a murine lung infection model of antipseudomonal activity of phage PEV20 in a dry-powder formulation. Antimicrob. Agents Chemother 2018, 62, e01714-17. [Google Scholar] [CrossRef]
- Lin, Y.; Quan, D.; Chang, R.Y.K.; Chow, M.Y.T.; Wang, Y.; Li, M.; Morales, S.; Britton, W.J.; Kutter, E.; Li, J.; et al. Synergistic activity of phage PEV20-ciprofloxacin combination powder formulation—A proof-of-principle study in a P. aeruginosa lung infection model. Eur. J. Pharm. Biopharm. 2021, 158, 166–171. [Google Scholar] [CrossRef]
- Lin, Y.; Chang, R.Y.K.; Britton, W.J.; Morales, S.; Kutter, E.; Li, J.; Chan, H.K. Inhalable combination powder formulations of phage and ciprofloxacin for P. aeruginosa respiratory infections. Eur. J. Pharm. Biopharm. 2019, 142, 543–552. [Google Scholar] [CrossRef]
- Tabare, E.; Glonti, T.; Cochez, C.; Ngassam, C.; Pirnay, J.P.; Amighi, K.; Goole, J. A design of experiment approach to optimize spray-dried powders containing pseudomonas aeruginosa podoviridae and myoviridae bacteriophages. Viruses 2021, 13, 1926. [Google Scholar] [CrossRef]
- Lin, Y.; Yoon Kyung Chang, R.; Britton, W.J.; Morales, S.; Kutter, E.; Li, J.; Chan, H.K. Storage stability of phage-ciprofloxacin combination powders against Pseudomonas aeruginosa respiratory infections. Int. J. Pharm. 2020, 591, 119952. [Google Scholar] [CrossRef]
- Yan, W.; He, R.; Tang, X.; Tian, B.; Liu, Y.; Tong, Y.; Kenneth, K.W.T.; Leung, S.S.Y. The influence of formulation components and environmental humidity on spray-dried phage powders for treatment of respiratory infections caused by acinetobacter baumannii. Pharmaceutics 2021, 13, 1162. [Google Scholar] [CrossRef] [PubMed]
- Matinkhoo, S.; Lynch, K.H.; Dennis, J.J.; Finlay, W.H.; Vehring, R. Spray-Dried Respirable Powders Containing Bacteriophages for the Treatment of Pulmonary Infections. J. Pharm. Sci. 2011, 101, 5197–5205. [Google Scholar] [CrossRef]
- Vinner, G.K.; Rezaie-Yazdi, Z.; Leppanen, M.; Stapley, A.G.F.; Leaper, M.C.; Malik, D.J. Microencapsulation of Salmonella-specific bacteriophage felix o1 using spray-drying in a ph-responsive formulation and direct compression tableting of powders into a solid oral dosage form. Pharmaceuticals 2019, 12, 43. [Google Scholar] [CrossRef] [PubMed]
- Carrigy, N.B.; Liang, L.; Wang, H.; Kariuki, S.; Nagel, T.E.; Connerton, I.F.; Vehring, R. Spray-dried anti-Campylobacter bacteriophage CP30A powder suitable for global distribution without cold chain infrastructure. Int. J. Pharm. 2019, 569, 118601. [Google Scholar] [CrossRef]
- Carrigy, N.B.; Ly, A.; Harrison, M.; Sauvageau, D.; Martin, A.; Finlay, W.H.; Vehring, R. Comparison of Spray Drying and Atmospheric Spray Freeze Drying for the Production of Active Anti-tuberculosis Bacteriophage D29 Dry Powder for Inhalation. Respir. Drug Deliv. Eur. 2019, 421–424. [Google Scholar]
- Leung, S.S.Y.; Parumasivan, T.; Gao, F.G.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Warwick, W.J.; Kutter, E.; Chan, H.-K. Effects of storage conditions on the stability of spray dried, inhalable bacteriophage powders. Int. J. Pharm. 2017, 521, 141–149. [Google Scholar] [CrossRef]
- Golec, P.; Dabrowski Kamil, K.; Hejnowicz, M.S.; Gozdek, A.; Łoś, J.M.; Wegrzyn, G.; Łobocka, M.B.; Łoś, M. A reliable method for storage of tailed phages. J. Microbiol. Methods 2011, 84, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Jin, P.; Sha, R.; Zhang, Y.; Liu, L.; Bian, Y.; Qian, J.; Qian, J.; Lin, J.; Ishimwe, N.; Hu, Y.; et al. Blood Circulation-Prolonging Peptides for Engineered Nanoparticles Identified via Phage Display. Nano Lett. 2019, 19, 1467–1478. [Google Scholar] [CrossRef]
- Anjum, S.; Ishaque, S.; Fatima, H.; Farooq, W.; Hano, C.; Abbasi, B.H.; Anjum, I. Emerging applications of nanotechnology in healthcare systems: Grand challenges and perspectives. Pharmaceuticals 2021, 14, 707. [Google Scholar] [CrossRef]
- Jaime, E.A.; Tolibia, S.M.; Rodelo, C.G.; Salinas, R.A.; Galdámez-Martínez, A.; Dutt, A. Interaction of virus-like particles and nanoparticles with inorganic materials for biosensing: An exciting approach. Mater. Lett. 2022, 307, 131088. [Google Scholar] [CrossRef]
- Farooq, U.; Yang, Q.; Wajid Ullah, M.; Wang, S. Principle and Development of Phage-Based Biosensors. Biosens. Environ. Monit. 2019, 1, 1–18. [Google Scholar] [CrossRef]
- Wang, D.B.; Cui, M.M.; Li, M.; Zhang, X.E. Biosensors for the Detection of Bacillus anthracis. Acc. Chem. Res. 2021, 54, 4451–4461. [Google Scholar] [CrossRef] [PubMed]
- Paramasivam, K.; Shen, Y.; Yuan, J.; Waheed, I.; Mao, C.; Zhou, X. Advances in the Development of Phage-Based Probes for Detection of Bio-Species. Biosensors 2022, 12, 30. [Google Scholar] [CrossRef]
- Witeof, A.E.; McClary, W.D.; Rea, L.T.; Yang, Q.; Davis, M.M.; Funke, H.H.; Catalano, C.E.; Randolph, T.W. Atomic-Layer Deposition Processes Applied to Phage λ and a Phage-like Particle Platform Yield Thermostable, Single-Shot Vaccines. J. Pharm. Sci. 2022, 111, 1354–1362. [Google Scholar] [CrossRef]
- Pelliccia, M.; Andreozzi, P.; Paulose, J.; D’Alicarnasso, M.; Cagno, V.; Donalisio, M.; Civra, A.; Broeckel, R.M.; Haese, N.; Silva, P.J.; et al. Additives for vaccine storage to improve thermal stability of adenoviruses from hours to months. Nat. Commun. 2016, 7, 13520. [Google Scholar] [CrossRef]
- Ali, Q.; Ahmar, S.; Sohail, M.A.; Kamran, M.; Ali, M.; Saleem, M.H.; Rizwan, M.; Ahmed, A.M.; Mora-Poblete, F.; do Amaral Júnior, A.T.; et al. Research advances and applications of biosensing technology for the diagnosis of pathogens in sustainable agriculture. Environ. Sci. Pollut. Res. 2021, 28, 9002–9019. [Google Scholar] [CrossRef]
- Sedki, M.; Chen, X.; Chen, C.; Ge, X.; Mulchandani, A. Non-lytic M13 phage-based highly sensitive impedimetric cytosensor for detection of coliforms. Biosens. Bioelectron. 2020, 148, 111794. [Google Scholar] [CrossRef]
- He, Y.; Shi, Y.; Liu, M.; Wang, Y.; Wang, L.; Lu, S.; Fu, Z. Nonlytic Recombinant Phage Tail Fiber Protein for Specific Recognition of Pseudomonas aeruginosa. Anal. Chem. 2018, 90, 14462–14468. [Google Scholar] [CrossRef]
- Wang, Y.; He, Y.; Bhattacharyya, S.; Lu, S.; Fu, Z. Recombinant Bacteriophage Cell-Binding Domain Proteins for Broad-Spectrum Recognition of Methicillin-Resistant Staphylococcus aureus Strains. Anal. Chem. 2020, 92, 3340–3345. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Fromm, K.M.; Akbar Ashkarran, A.; Jimenez de Aberasturi, D.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, A.; Safwat, A.; Abdelsattar, A.S.; Essam, K.; Makky, S.; El-shibiny, A.; Elsayed, A.; Safwat, A.; Abdelsattar, A.S.; Essam, K. The antibacterial and biofilm inhibition activity of encapsulated silver nanoparticles in emulsions and its synergistic effect with E. coli bacteriophage. Inorg. Nano-Met. Chem. 2022, 1–11. [Google Scholar] [CrossRef]
- Chegini, Z.; Khoshbayan, A.; Taati Moghadam, M.; Farahani, I.; Jazireian, P.; Shariati, A. Bacteriophage therapy against Pseudomonas aeruginosa biofilms: A review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 45. [Google Scholar] [CrossRef] [PubMed]
- Abdelsattar, A.S.; Nofal, R.; Makky, S.; Safwat, A.; Taha, A.; El-Shibiny, A. The synergistic effect of biosynthesized silver nanoparticles and phage zcse2 as a novel approach to combat multidrug-resistant salmonella enterica. Antibiotics 2021, 10, 678. [Google Scholar] [CrossRef]
- Li, L.L.; Yu, P.; Wang, X.; Yu, S.S.; Mathieu, J.; Yu, H.Q.; Alvarez, P.J.J. Enhanced biofilm penetration for microbial control by polyvalent phages conjugated with magnetic colloidal nanoparticle clusters (CNCs). Environ. Sci. Nano 2017, 4, 1817–1826. [Google Scholar] [CrossRef]
- Ahiwale, S.S.; Bankar, A.V.; Tagunde, S.; Kapadnis, B.P. A Bacteriophage Mediated Gold Nanoparticles Synthesis and Their Anti-biofilm Activity. Indian J. Microbiol. 2017, 57, 188–194. [Google Scholar] [CrossRef]
- Abdelsattar, A.S.; Farouk, W.M.; Mohamed Gouda, S.; Safwat, A.; Hakim, T.A.; El-Shibiny, A. Utilization of Ocimum basilicum extracts for zinc oxide nanoparticles synthesis and their antibacterial activity after a novel combination with phage. Mater. Lett. 2022, 309, 131344. [Google Scholar] [CrossRef]
- Hussain, S.; Joo, J.; Kang, J.; Kim, B.; Braun, G.B.; She, Z.; Kim, D.; Mann, A.P.; Mölder, T.; Teesalu, T.; et al. Antibiotic-loaded nanoparticles targeted to the site of infection enhance antibacterial efficacy. Nat. Biomed. Eng. 2018, 2, 95–103. [Google Scholar] [CrossRef]
- Brandau, D.T.; Jones, L.S.; Wiethoff, C.M.; Rexroad, J.; Middaugh, C.R. Thermal stability of vaccines. J. Pharm. Sci. 2003, 92, 218–231. [Google Scholar] [CrossRef]
- Knezevic, I. Stability evaluation of vaccines: WHO approach. Biologicals 2009, 37, 357–359. [Google Scholar] [CrossRef]
- Pardi, N.; Hogan, M.J.; Weissman, D. Recent advances in mRNA vaccine technology. Curr. Opin. Immunol. 2020, 65, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Jamaledin, R.; Sartorius, R.; Di Natale, C.; Veccione, R.; De Berardinis, P.; Netti, P.A. Recombinant Filamentous Bacteriophages Encapsulated in Biodegradable Polymeric Microparticles for Stimulation of Innate and Adaptive Immune Responses. Microorganisms 2020, 8, 650. [Google Scholar] [CrossRef]
- Robbins, A.; Freeman, P. Obstacles to Developing Vaccines for the Third World. Sci. Am. 1988, 259, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.M.; Silva, M.D.; Pastrana, L.M.; Bañobre-López, M.; Sillankorva, S. The clinical path to deliver encapsulated phages and lysins. FEMS Microbiol. Rev. 2021, 45, fuab019. [Google Scholar] [CrossRef]
- Manohar, P.; Ramesh, N. Improved lyophilization conditions for long-term storage of bacteriophages. Sci. Rep. 2019, 9, 15242. [Google Scholar] [CrossRef]
- Lavenburg, V.M.; Liao, Y.T.; Salvador, A.; Hsu, A.L.; Harden, L.A.; Wu, V.C.H. Effects of lyophilization on the stability of bacteriophages against different serogroups of Shiga toxin-producing Escherichia coli. Cryobiology 2020, 96, 85–91. [Google Scholar] [CrossRef]
- Duyvejonck, H.; Merabishvili, M.; Vaneechoutte, M.; de Soir, S.; Wright, R.; Friman, V.P.; Verbeken, G.; De Vos, D.; Pirnay, J.P.; Van Mechelen, E.; et al. Evaluation of the stability of bacteriophages in different solutions suitable for the production of magistral preparations in Belgium. Viruses 2021, 13, 865. [Google Scholar] [CrossRef]
- Cui, H.; Yuan, L.; Lin, L. Novel chitosan film embedded with liposome-encapsulated phage for biocontrol of Escherichia coli O157:H7 in beef. Carbohydr. Polym. 2017, 177, 156–164. [Google Scholar] [CrossRef]
- Born, Y.; Bosshard, L.; Duffy, B.; Loessner, M.J.; Fieseler, L. Protection of Erwinia amylovora bacteriophage Y2 from UV-induced damage by natural compounds. Bacteriophage 2015, 5, e1074330. [Google Scholar] [CrossRef]
| Technique | Stability Enhancers | Phage | Duration | Reference |
|---|---|---|---|---|
| Polymer-based | Pullulan and Trehalose | LISTEX P100 | 60 days | [87] |
| Polyethylene glycol (PEG) | Felix-O1, A511 | 24 h within bloodstream | [92] | |
| Polyoxoborate composite | T4, MS2, M13 | 6 h of incubation | [97] | |
| Encapsulation | phiIPLA88, phiIPLA35, phiIPLA-RODI, phiIPLA-C1C | 2 months | [99] | |
| Liposome | KØ1, KØ2, KØ3, KØ4, KØ5 | 48 h within bloodstream | [102] | |
| Freeze-dried powder | Lactose, trehalose, sucrose | ISP | 37 months | [121] |
| Lecithin | PEV2, PEV20 | 250 days | [130] | |
| Spray-dried powder | Trehalose, dextran, lactose | LUZ19, Romulus | 12 min of atomizing | [127] |
| Freezing inside bacterial cells | Glycerol | Tailed-phages | ~10 months | [151] |
| Nano-assisted | Nanolayers of aluminum oxide | λ | ~1 month | [159] |
| Carbon-based nanoparticles, gold nanoparticles | M13 | 16 days | [162] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wdowiak, M.; Paczesny, J.; Raza, S. Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review. Pharmaceutics 2022, 14, 1936. https://doi.org/10.3390/pharmaceutics14091936
Wdowiak M, Paczesny J, Raza S. Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review. Pharmaceutics. 2022; 14(9):1936. https://doi.org/10.3390/pharmaceutics14091936
Chicago/Turabian StyleWdowiak, Mateusz, Jan Paczesny, and Sada Raza. 2022. "Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review" Pharmaceutics 14, no. 9: 1936. https://doi.org/10.3390/pharmaceutics14091936
APA StyleWdowiak, M., Paczesny, J., & Raza, S. (2022). Enhancing the Stability of Bacteriophages Using Physical, Chemical, and Nano-Based Approaches: A Review. Pharmaceutics, 14(9), 1936. https://doi.org/10.3390/pharmaceutics14091936






