1. Introduction
The main goal of smart drug delivery is the targeted nanocarrier transport of drugs, or immunomodulatory molecules, to the organ or cell type of interest without clearance or accumulation in other compartments [
1]. Controlling nanocarrier-based delivery is key to the reduction in systemic side effects and to the improvement of pharmacological profiles [
2]. Despite many years of research, tumor-targeting nanocarrier systems only achieve tumor accumulation rates with a median of 0.7% of the injected dose at the targeted site [
3]. Consequently, nanocarriers applied as nanomedicines have generally failed in clinical trials, with only a few exceptions. All of these nanomedicines have no targeting moiety incorporated into the nanocarrier. These nanocarriers include albumin-stabilized paclitaxel nanoparticles (ABRAXANE
®), or liposomal formulations of either doxorubicin (DOXIL
®) or irinotecan (ONIVYDE
®) [
4,
5,
6]. Nevertheless, nanocarriers inherit a great potential in revolutionizing targeted therapies, as demonstrated just recently by the use of lipid nanoparticle-based COVID-19 mRNA vaccines [
7].
Numerous factors influence successful clinical translation into the medical market and must be overcome to learn from and recognize previously failed nanocarrier systems [
5,
6]. In this field, the nano-bio interface represents a great challenge [
6]. In particular, we are talking about the role of the biomolecular corona and its influence, antibody-based nanocarrier targeting [
8], and drug release [
9]. In addition, many nanocarrier systems are rapidly cleared by the mononuclear phagocyte system (MPS), or the complement system [
10,
11,
12]. In this process, pristine nanocarriers are usually marked and identified by opsonins, such as complement proteins or immunoglobulins, which can bind to opsonin receptors expressed on macrophages leading to nanocarrier phagocytosis [
13]. Consequently, the immune system plays a critical role in the recognition and elimination of nanocarriers [
14].
Therefore, nanocarriers have been coated with an additional stealth layer that disguises the nanocarriers and prevents their recognition by opsonins, thus reducing phagocytic removal. Among the stealth polymers, poly-(ethylene glycol) (PEG) is the gold standard or benchmark for surface modification of nanocarriers resulting in reduced clearance by the MPS and complementary system [
15]. PEG has been shown to increase the hydrodynamic size and water solubility of nanocarrier systems, thereby decreasing their self-aggregation and interaction with blood components of the MPS by means of steric repulsion [
16,
17]. The use of PEGylated nanocarrier systems are rapidly increasing in the field of nanomedicine, and have now reached clinical trials, or have been licensed by pharmaceutical regulatory authorities [
18]. Among the PEGylated drugs or nanocarrier systems, attached PEGs differ in their molecular weight ranging from 2 to 60 kDa [
17]. However, these non-targeted nanocarriers demonstrate an improved pharmacokinetic profile compared to the administration of a free payload [
19]. However, as mentioned above, the average accumulation at the target site is low in the case of tumor therapy [
3].
Consequently, improving selective accumulation of the nanocarrier system requires active targeting ligands, such as surface decoration with antibody molecules. In the field of cancer vaccines and immunotherapy, a nanocarrier-based targeted delivery of tumor-associated antigens and immunomodulators (adjuvants) to dendritic cells (DCs) bears great potential since dendritic cells are professional antigen-presenting cells capable of generating robust antigen-specific anti-tumor immune responses [
20]. However, due to the poor success of DC-based anti-tumor vaccines in clinical trials, an efficient targeting of specific DC subsets is considered to be the prerequisite for inducing strong anti-tumor responses [
21]. In particular, conventional DCs type 1 (cDC1) inherit the potential to efficiently cross-present exogenous antigens enabling them to induce strong cytotoxic T cell responses against tumor cells [
22]. Therefore, delivering cargo to cDC1 via nanocarriers functionalized with antibodies, such as anti-CLEC9A [
23] and other targets, is a feasible approach. However, there are several limitations evidenced by the small number of antibody-functionalized nanocarrier formulations currently undergoing clinical trials [
24]. Most commonly, in the antibody surface-decoration of nanocarriers a PEG linker is applied to locate the antibody molecule at a certain distance from the nanocarrier surface. Hence, the PEG length can influence the interactions of the attached targeting ligand and the cell receptor of interest [
25,
26].
As investigated by Cruz et al., a PEG linker length comparison on antigen-loaded and antibody-coated PLGA nanoparticles highlighted the finding that short PEG constructs (2–3 kDa) result in stronger interactions with DCs, inducing higher T cell proliferation when compared to longer PEG constructs (6–20 kDa) [
27]. In contrast, Kapadia et al. demonstrated a higher particle uptake and antigen presentation of peptide-decorated hydrogels when using a 5 kDa PEG linker compared to 2 and 10 kDa [
28]. Given this complexity, we focused on investigating previously developed antibody-nanocarrier conjugates, precisely targeting DCs both in vitro [
29] and in vivo [
23], with respect to the influence of the applied PEG linker length (0.65, 2 and 5 kDa PEG). Our investigation focused particularly on the targeting of the CD11c receptor on DCs in immortalized (DC2.4), or primary DCs (BMDCs, splenocytic cDC1s, and the impact of the biomolecular corona. This study delineates the differences in DC targeting between the three PEG linker constructs from cell lines to primary cells, highlighting a greater antibody distance from the nanocarrier surface necessary for targeting DC subsets. In particular we have shown that the ability of nanocarriers to target cDC1 in primary cells required a 5 kDa PEG construct. Furthermore, our biomolecular corona analyses highlight the importance of the nanocarrier–antibody composition.
2. Materials and Methods
Cell culture. The murine RAW264.7 macrophages (DSMZ, Braunschweig, Germany) were cultured in Dulbecco’s modified eagle medium (DMEM, Thermo Fisher Scientific, Waltham, MA, USA containing 10% fetal bovine serum (FBS), 100 U mL−1 penicillin, 100 mg mL−1 streptomycin, and 2 mM glutamine (all Thermo Fisher Scientific, Waltham, MA, USA). The murine DC2.4 dendritic cells (Merck, Darmstadt, Germany) were cultured in Iscove’s Modified Dulbecco’s Medium (IMDM, Sigma-Aldrich, St. Louis, MO, USA), supplemented with 5% FBS, 100 U mL−1 penicillin, 100 mg mL−1 streptomycin, 2 mM glutamine, as well as 1% 2-Mercaptoethanol (100×). Both cell lines were maintained in an incubator at 37 °C and 5% CO2 humidity (CO2 Incubator C200, Labotect, Rosdorf, Germany). For the passaging of cells, the consumed media was discarded and the cells were briefly washed with 10 mL PBS. RAW264.7 cells were detached at 37 °C with Trypsin-EDTA (Thermo, Germany) and DC2.4 cells at 4 °C with PBS containing 2 mM EDTA for 10 min. The cells were detached from the flask by pipetting 10–20 mL of FBS-containing medium up and down, followed by centrifugation of the cell suspension at 300× g for 5 min. The resulting cell pellet was resuspended in medium with FBS and the cell viability was tested with trypan blue (1:1 dilution) using an automated cell counter (TC10, Bio-Rad, Hercules, CA, USA).
Nanocarrier PEGylation and modification for the attachment of antibodies. The nanocarriers, namely bionized nanoferrite particles with a hydroxyethyl starch shell (Micromod, Rostock, Germany), were PEGylated to the surface amine groups via NHS chemistry based on the amount of NH2 given by the manufacturer (3 nmol mg−1). For this purpose, five different bifunctional linkers were investigated. These included: the NHS-PEG-DBCO linker of 0.65 kDa (Jena Biosciences, Jena, Germany), the 2 kDa NHS-PEG-MeO and 5 kDa NHS-PEG-MeO (both from Iris Biotech GmbH, Marktredwitz, Germany), and the 2 kDa NHS-PEG-DBCO and 5 kDa NHS-PEG-DBCO (both from Nanocs Inc., Boston, MA, USA). Each linker was conjugated onto the nanocarrier surface using three different molecular weight ratios (1:1, 1:10, 1:50) based on the amount of NH2. To do this, the number of NH2 groups per mg nanocarrier were multiplied by the respective molecular weight of the different linkers and the desired linker ratio. All linker solutions were freshly prepared in DMSO (Merck) for each experiment. After overnight reaction under constant shaking at room temperature, all nanocarrier linker conjugates were washed (3×) with PBS using a strong magnet (neodymium magnet) to remove excess linker species. The final nanocarrier linker products were resuspended in PBS and their concentration was determined by fluorescence calibration. The DBCO-functionalized nanocarriers were conjugated overnight with azide-modified antibodies (see site-directed antibody modification) at room temperature while shaking (in PBS). For this conjugation, the reaction was scaled based on a nanocarrier to azide-antibody ratio of 67:1. For example, 1 mg of nanocarrier was reacted with 14.93 µg of azide-antibody.
Fluorescence Calibration for Nanocarrier Quantification. The fluorescence calibration was performed to determine the amount and concentration of all nanocarrier conjugates. Pristine nanocarriers were used as a standard and diluted to a linear fit (100 µg mL−1 down to 3.125 µg mL−1). All probes were diluted based on the initial mass used. Measurements of the standard and samples were prepared as duplicates in PBS. The Infinite M1000 plate reader (Tecan, Männedorf, Switzerland), with an excitation wavelength of 552 nm and emission of 580 nm, recorded the fluorescence.
Validation of nanocarrier PEGylation. The surface PEGylation of the nanocarriers was qualitatively analyzed by two fluorescence-based methods via the plate reader (Infinite M1000, Tecan) and flow cytometry (Attune NxT, Thermo). For this purpose, an Alexa Fluor™ 647 NHS ester (Succinimidylester, Thermo) was used. For the preparation, 100 µg of pristine and PEGylated nanocarriers were reacted with a 50-fold molar excess of the NHS ester (18.75 ng, 1.9 µL of a 10 ng/µL stock) for 1 h at room temperature while shaking. After the incubation, all samples were washed (3×) with PBS using a strong magnet. Pristine nanocarriers incubated with and without the NHS ester were utilized to differentiate between the negative background signal and the positive NHS ester signal, representing the binding towards the primary amines on the nanocarriers. For the plate reader measurement, all samples were resuspended in a final volume of 0.1 mL PBS and 80 µL were pipetted into a black 96-well plate (n = 2). The fluorescence was measured with an excitation of 651 nm and an emission of 672 nm. Data sets were calculated from the positive control. For the flow cytometry measurement, 1 µL of nanocarrier sample (~10 µg) was diluted in 1 mL PBS and analyzed by the RL1 channel with an excitation laser of 633 nm and a 670/14 nm band pass filter for emission.
Site-directed antibody modification. Antibodies (α-CD11c, clone N418, and α-IgG isotype, clone HTK888, 0.5 mg/mL, both from Biolegend, San Diego, CA, USA) were azide-modified based on the manufacturer’s instructions (Site ClickTM Antibody Azido Modification Kit, Thermo). In summary, the concentrated antibody was first reacted overnight with ß-galactosidase at 37 °C to cleave the galactose residues present at the carbohydrate domains of the Fc region. Then the azide groups were attached by incubating the antibody with the GalT (Y289L) enzyme in combination with the UDP-GalNAz donor in an overnight reaction at 30 °C. Finally, the azide-modified antibody (1–2 mg/mL) was purified and concentrated. The modified antibodies were further analyzed for their concentration (Pierce 660 nm Protein Assay), for the validation of their azide-attachment, and for their conjugation onto DBCO-functionalized nanocarriers via copper-free click chemistry.
Antibody quantification. The antibody concentrations were specified by using the Pierce 660 nm protein assay according to the manufacturer’s instructions. BSA (bovine serum albumin) was applied as a standard by setting up a dilution series in PBS. The Infinite M1000 plate reader (Tecan) was used to measure the standard and all samples at an optical density of 660 nm, in duplicates.
Cell line uptake experiments. For the nanocarrier cell experiments, 1.5 × 105 cells mL−1 were cultivated in a 24-well plate in a humidified incubator at 37 °C and 5% CO2. After overnight attachment, the cell culture medium was exchanged before each experiment to a fresh medium with or without FBS for 1 to 2 h. This medium exchange was performed so that the cells could adapt to a serum-free environment. All samples were prepared in triplicates and incubated for 2 h with a concentration of 7.5 or 75 µg mL−1 in 250 µL of medium per well. The pre-incubation with mouse plasma (collected from 8–12-week-old sexually mature and parasite-free mice, GeneTex, Irvine, CA, USA) was performed for 1 h at 37 °C, shaking in a 1:1 nanocarrier to mouse plasma ratio. Afterwards, the mouse plasma was separated from the samples by using a magnet and 7.5 or 75 µg mL−1 in 250 µL medium without FBS were incubated with the cells. After the incubation, 1 mL PBS was used to wash the cells. Then, the cells were detached via 250 µL 2 mM PBS-EDTA for 10 min at 4 °C (DC2.4) or Trypsin-EDTA for 5 min at 37 °C (RAW264.7). Subsequently, 250 µL of medium with FBS were added and the cells were transferred from the well into a 1.5 mL micro tube. The cell viability was determined by using the Zombie Aqua assay (Biolegend, USA). For this assay, the cells were centrifuged at 500× g for 5 min and the cell pellet was resuspended in 100 µL Zombie Aqua solution (1:500 diluted in PBS). After an incubation of 15 min at 4 °C in the dark, the Zombie Aqua solution was separated from the cells by centrifugation at 500× g for 5 min and the cells were resuspended in 1 mL PBS for analysis using the flow cytometer.
Flow cytometry. The measurement of nanocarriers taken up by cells or the detection of covalently bound antibodies on the nanocarrier surface was performed by flow cytometry (Attune™ NxT, Thermo). The nanocarriers were detected by their red fluorescence with the YL1 channel and an excitation laser of 561 nm and a 585/16 nm band pass filter for emission. Viability of the cells was determined by using the Live/Dead fixable Zombie Aqua (Biolegend, USA) with the VL2 channel with an excitation laser of 405 nm and a 512/25 nm band pass filter for emission. The Attune™ NxT Software was applied for data analysis. In a first step, the cell population was selected using an FSC/SSC scatter plot, excluding cell debris. Only the gated events of viable cells were examined by the fluorescence signal expressed as the median fluorescence intensity (MFI), or as the percentage of gated events/cells. For the validation of a correct antibody surface modification of the nanocarriers, a secondary fluorophore-labeled antibody, which was directed against the Fc region of the bound antibody, was chosen. The testing was performed with 2 µg of sample and 1 µg of secondary antibody in 20 µL PBS, and was reacted at 4 °C in the dark for 30 min. The pristine nanocarriers with and without the secondary antibody were applied as adsorption controls. Following incubation, the probes were filled up to 1 mL PBS and analyzed by flow cytometry. After selecting the nanocarrier population by the FSC/SCC scatter plot, the red fluorescence (YL1) was plotted against the SSC. From this selected population, the MFI or the percentage of gated events of the secondary antibody channel were detected by using the BL1 channel with an excitation laser of 488 nm and a 530/30 nm band pass filter for emission.
Visualization of intracellular localization by cLSM. Nanocarrier cell interaction was visualized using confocal laser scanning microscopy (cLSM). The experiments were performed on the LSM SP5 STED Leica Laser Scanning Confocal Microscope (Leica, Wetzlar, Germany), composed of an inverse fluorescence microscope DMI 6000CS equipped with a multi-laser combination using a HCX PL APO CS 63 × 1.4 oil objective. The red fluorescence of the nanocarrier samples was excited with the excitation laser 561 nm and detected with an emission filter at 570–599 nm. The cell membrane of the cells was stained with CellMask DeepRed (Thermo) applying the excitation laser 633 nm with a detection at 660–700 nm. For the experimental setup, 5 × 104 RAW264.7 cells in 200 µL DMEM with 10% FBS were seeded in a well of a 15 µ-Slide 8-well glass bottom (ibidi). After overnight attachment in the incubator, the medium was exchanged for 60–90 min, followed by the sample incubation with 75 µg mL−1 for 2 h at 37 °C (DMEM medium with FBS, without FBS, and pre-incubated with mouse plasma and resuspended in medium without FBS). After the incubation, the cells were washed once with PBS and the cell membrane was stained for 5 min in the dark (1:1.000 in PBS) before live cell imaging. The image acquisition was performed by using ImageJ (v1.52n).
Incubation and Visualization of Mouse Plasma. The murine biomolecular corona proteins of the nanocarriers were analyzed by SDS-PAGE and silver staining (both from Thermo). For this analysis, the samples were incubated with the mouse plasma at a 1:1 volume ratio for 1 h at 37 °C while shaking. Subsequently, the mouse plasma was removed and the samples were washed (3×) with PBS by using a magnet. Adsorbed mouse plasma proteins were detached from the samples by resuspending the nanocarrier pellet in 100 µL desorption buffer (2% (w/v) SDS + 62.5 mM Tris-HCl in 3 mL deionized water) for 10 min at 70 °C while shaking. Desorbed proteins were recovered from the nanocarrier samples by applying a magnet. The total amount of denatured proteins in the supernatant was quantified by Pierce 660 nm protein assay and 2 µg were used for SDS-PAGE, visualized by silver staining.
Multi-angle dynamic light scattering. For the detection of the size (diameter in nm) and size distribution (polydispersity index, PDI), all nanocarriers were analyzed by multi-angle dynamic light scattering. The measurements were performed on an ALV spectrometer consisting of a goniometer and an ALV-5004 multiple-tau full-digital correlator (320 channels) at 20 °C and 7 (dynamic) angles ranging from 30° to 150°. The light source was a He-Ne Laser (wavelength of 632.8 nm). The samples were prepared by diluting 1 µL of unfiltered dispersion with 1 mL PBS, which was previously filtered through membrane filters (GS Millipore) with a pore size of 2.0 µm.
Zeta potential. The surface charge of the nanocarriers was detected using a Zeta Sizer Nano Series (Malvern Instruments GmbH, Herrenberg, Germany). All samples (10 µL) were diluted in a 1 mM potassium chloride solution (1 mL) and measured in technical triplicates.
TEM. The FEI Tecnai F20 transmission electron microscope (operating at a voltage of 200 kV) was used to investigate the morphology of the nanocarriers. To achieve this, all samples were diluted to a particle concentration of 75 µg mL−1 in IMDM medium containing 5% FBS, and 4 µL were pipetted on a carbon-coated grid. The excess solvent was blotted away with filter paper.
In solution digestion. SDS was removed from the protein corona samples by using Pierce
TM Detergent Removal Spin Columns (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. Next, protein samples were subjected to an in-solution tryptic digest as a preparation for the LC-MS proteomic measurements. The digestion protocol was carried out as described previously with modifications [
30,
31]. Briefly, 10 to 25 µg of protein were precipitated and washed twice by using the ProteoExtract Protein Precipitation Kit (CalBioChem, Darmstadt, Germany) according to the manufacturer’s instructions. After letting the protein pellet dry for 5 to 10 min, the protein was solubilized in 0.1% RapiGest SF surfactant (Waters Corporation, Germany, dissolved in 50 mM ammonium carbonate buffer), at 80 °C for 15 min. Disulfide bonds were reduced with a 5 mM final concentration of dithiothreitol (Sigma, Roedermark, Germany) at 56 °C for 45 min. Thiol alkylation was performed with a 500 mM final concentration of iodoacetamide (Sigma, Germany) at room temperature for 1 h in the dark. Subsequently, the digestion was carried out with trypsin in a mass ratio of 50:1 (protein:trypsin) at 37 °C for a maximum of 18 h. The digestion was stopped by lowering the pH of the peptide solution with 2 µL of hydrochloric acid (Sigma, Germany). To remove aggregates, the samples were centrifuged at 13,000×
g for 15 min at 4 °C and the supernatant was transferred to new tubes for measurement.
Liquid chromatography-electrospray ionization mass spectrometry (LC-MS). Peptide samples were spiked with 50 fmol μL
−1 HI3
E. coli Standard (Waters Corporation, Germany), and the total volume was adjusted with LC-MS grade water (Merck, Darmstadt, Germany). The absolute quantification performed in these measurements was based on a previously published protocol [
32]. The LC-MS measurements were carried out as previously described in the work of our group [
16,
31]. A nanoACQUITY UPLC system coupled to a Synapt G2Si mass spectrometer (both Waters Corporation, Germany) was utilized, operating the measurements with MassLynx 4.1 (Waters Corporation). The UPLC was equipped with a C18 nanoACQUITY trap column (5 μm, 180 μm × 20 mm) and a C18 analytical reversed-phase column (1.7 μm, 75 μm × 150 mm; both Waters Corporation, Germany). The samples were injected at a flow rate of 0.3 μL min
−1 and separated by a gradient of 2% to 37% of mobile phase B over 70 min. The solvents for the gradient consisted of (A) 0.1% (
v/
v) formic acid in LC-MS grade water and (B) 0.1% (
v/v) formic acid in acetonitrile (Biosolve, Neuss, Germany). The reference components Glu-Fibrinopeptide and LeuEnkephalin (both Sigma, Germany) were injected at a flow rate of 0.5 μL min
−1. The mass spectrometer was equipped with a NanoLockSpray for source by electrospray ionization (ESI) and configured for the positive mode. The measurements were conducted in the resolution mode with data-independent acquisition (MS
E) in a mass to charge range of 50–2000 Da, scan time of 1 s, ramped trap collision energy from 20 to 40 V, and data acquisition of 90 min. All samples were measured in technical triplicates.
Protein identification. Protein identification was performed by using the proteomics analysis software Progenesis QI 2.0 (Nonlinear Dynamics). The procedure was described previously [
16]. Following settings were applied to process the data: Noise reduction threshold for low energy defined as 120 counts, elevated energy as 25 counts, and peptide intensity as 750 counts. The utilized protein database consisted of the murine proteome with reviewed proteins, which were downloaded from uniport (swiss prot). The sequence information for the Hi3
E. coli standard for absolute quantification was added to the protein database file. The following settings were used to identify the proteins from the peptides: one missed cleavage, maximum protein mass of 600 kDa, fixed carbamidomethyl modification for cysteine, variable oxidation for methionine, a minimum of three assigned fragments per peptide, a minimum of two assigned peptides per protein, a minimum of five assigned fragments per protein, and a score parameter below 4. The protein quantification in fmol was calculated based on the TOP3/Hi3 approach [
33]. A table with all identified proteins is provided in the Supplementary Information section.
Generation of bone marrow-derived dendritic cells. Bone marrow-derived dendritic cells (BMDC) were differentiated from progenitor cells (BM cells) isolated from the bone marrow of 8- to 12-week-old C57BL/6J mice as previously described [
34]. Briefly, bone marrow was obtained by flushing the femur and tibia with cell culture medium (Iscove’s Modified Dulbecco’s Medium (IMDM) containing 5% FBS (PAN), 2 mM GlutaMax (Thermo Fisher Scientific), 100 U mL
−1 penicillin, 100 µg mL
−1 streptomycin, and 50 µM β-mercaptoethanol (Sigma-Aldrich) through a 26-gauge needle. BM cells were washed with cell culture medium and red blood cells were lysed using Gey’s lysis buffer (155 mM NH
4Cl, 10 mM KHCO
3, 100 µM EDTA) for 1 min at 4 °C. BM cells were washed again and seeded in non-TC-treated 10 cm-petri dishes (2 × 10
6 in 10 mL per dish). Fifty percent of cell culture medium was replenished on days 3 and 6. Nonadherent and loosely adherent immature BMDCs were harvested on day 7 and used for uptake experiments.
BMDC uptake experiments. Harvested BMDCs were washed with FBS-free cell culture medium and reseeded in 24-well suspension culture plates (105 per well) in 500 µL cell culture medium, either supplemented with 5% FBS or with 5% mouse plasma. Subsequently, cells were incubated with 3 µg mL−1 of different nanocarrier formulations for 20 h at 37 °C and 7.5% CO2, harvested, and analyzed by flow cytometry.
Conventional DC type 1 uptake experiment. Spleen cells were prepared as previously described [
35]. Briefly, spleens of C57BL/6J mice were dissected and mechanically ground through a 40 µm cell strainer. Red blood cells were subsequently lysed using a hypotonic buffer (155 mM NH
4Cl, 10 nM KHCO
3, 100 μM EDTA-disodium, pH 7.4) for 30 s and washed using cell culture medium (Iscove’s Modified Dulbecco’s Medium (IMDM) supplemented with 5% fetal bovine serum (FBS), 1% penicillin (100 U mL
−1), and streptomycin (100 µg mL
−1). Isolated spleen cells were subsequently seeded in 48-well plates (10
6 per well) in 500 µL cell culture medium without FBS supplemented with 5% mouse plasma. Different nanocarrier formulations were added (3 µg mL
−1) and cells were incubated for 20 h at 37 °C and 7.5% CO
2. Spleen cells were harvested and washed (1% FBS in PBS). Fc receptors were blocked with anti-CD16/CD32 (clone 2.4G2) for 10 min. To differentiate the cell types via flow cytometry, spleen cells were incubated with fluorophore-labelled cell type-specific antibodies for 30 min: anti-CD11c (clone N418), anti-CD11b (clone M1/70), anti-CD172a (clone P84), anti-CD8α (clone 53–6.7), anti-I-A/I-E (MHCII, clone M5/114.15.2), anti-Siglec-H (clone 551), anti-CD19 (clone 6D5), anti-CD3ε (clone 145–2C11), anti-CD14 (clone Sa14-2), anti-NK1.1 (clone PK136), and anti-Ly-6G (clone 1A8). Flow cytometric analysis was performed using the Attune NxT (Thermo Fisher Scientific) and analyzed with FlowJo software v10.7.1. Conventional DCs type 1 were defined as CD19
− CD3
− CD14
− NK1.1
− Ly-6G
− MHCII
+ CD11c
+ CD11b
− CD172a
− CD8α
+.
Statistical analysis. For the PEGylated nanocarrier uptake in the cell lines, the pristine nanocarriers of each medium condition (medium-FBS, medium+FBS, pre-incubated in mouse plasma) were statistically compared to the PEGylated samples from the corresponding medium condition using GraphPad Prism 9. One-way analysis of variance (ANOVA) was followed by a Dunnett’s multiple comparison test with a confidence interval as indicated in each figure. The CD11c/isotype fold changes were statistically compared to each other using the Brown–Forsythe and Welch ANOVA Test.
3. Results
The experimental workflow can be outlined as follows: we first investigated the stealth effect by coating our magnetic and red-fluorescent nanocarriers (magnetic hydroxyethyl starch particles or mgHES) with different PEG ratios. These PEGylated nanocarriers were then examined in cell culture with macrophages and DCs. Based on these findings, the PEG-induced stealth effect was then combined with the DC targeting ability of antibodies and termed stealth targeting. Both, the stealth effect and stealth targeting nanocarriers were analyzed based on their physicochemical characteristics (ζ-potential, MADLS, and TEM), their interaction with macrophages and DCs (flow cytometry and cLSM), and their proteomic biomolecular corona composition (SDS-PAGE and LC-MS).
Initially, two different PEG linkers (NHS-PEGx-MeO) of 2 and 5 kDa molecular weight were covalently immobilized onto the surface of the amine-functionalized nanocarriers. Three PEG excess ratios were then examined in order to achieve a sufficient stealth effect. This stealth effect, namely a reduced uptake of the PEGylated samples compared to the pristine nanocarriers, was investigated in highly phagocytic cells (macrophage cell line, RAW264.7), as well as in the cells of interest (DC line, DC2.4) and further analyzed by flow cytometry. In addition, primary DCs, such as bone marrow-derived DCs (BMDCs) and conventional DCs type 1 (cDC1) from the spleen, were analyzed for their potential to take up antibody-functionalized nanocarriers.
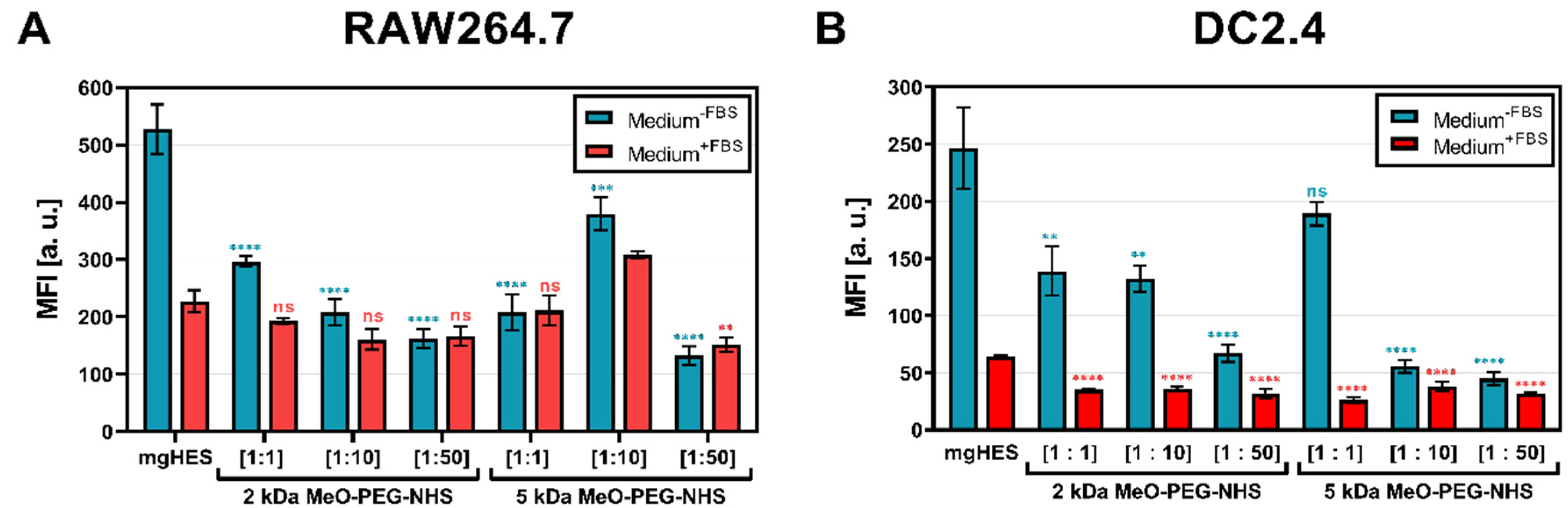
First, three molar ratios of the linkers were evaluated (
Figure 1A,B). Furthermore, the use of culture medium with and without fetal bovine serum (FBS) highlighted the role of additional proteins that can form a biomolecular corona on the nanocarrier surface, potentially influencing the nanocarrier cell interaction [
36]. Each PEG-modified (PEGylated) nanocarrier formulation was taken up to a lower rate by both cell lines when incubated in medium without FBS (blue bars). Given this medium condition, the stealth effect was found to be more pronounced for both cell lines at higher PEG ratios than at lower ones. In the medium supplemented with additional FBS proteins (red bars), the overall uptake of the samples was reduced compared to the medium without FBS. Here, only slightly lower values, or slightly increased values (1:10 ratio of the 5 kDa sample in RAW cells,
Figure 1A) were detected. A similar finding could be observed when analyzing the frequency of NC
+ cells (
Supporting Information Figure S1A,B).
In this case, the nanocarrier PEGylation was limited to the number of surface-NH
2 sites (3 nmol mg
−1) accessible for conjugation. Nevertheless, the cell uptake studies in both cell lines revealed that higher PEG ratios with sufficient reaction rates of NHS to NH
2 were better able to convey the stealth effect. Therefore, higher PEG to NH
2 ratios were able to fully saturate the NHS reaction on the nanocarrier surface. The difference in the PEGylation ratios could be demonstrated by using an Alexa Fluor
TM 647 NHS-ester. For this purpose, the difference in the fluorescence labeling between PEGylated and pristine nanocarriers was evaluated (see Material and Methods section for a detailed description). Following the conjugation, the Alexa Fluor
TM 647-based nanocarrier fluorescence was measured by flow cytometry and the plate reader (
Figure S2A,B). A decreasing fluorescence signal was observed for increasing PEG ratios (10× and 50×) indicating that less primary amines were available to react with the Alexa Fluor
TM 647 NHS-ester compared to the equimolar ratio (1:1). Given the 3 nmol NH
2 per mg nanocarrier and assuming a sphere shape, in theory, the mgHES nanocarriers are equipped with 0.4 NH
2 target sites per nm
2. Therefore, at a full conversion of amines to NHS linker reactions, 0.4 PEG chains per nm
2 can be assumed. In summary, the best stealth properties could be achieved when PEGylation was performed at the molar ratio of NH
2 on nanocarriers to PEG linker ratio of 1:50. Li et al. showed that a PEG grafting density of 0.2 to 2.5 chains nm
−2 on polymeric ovalbumin nanocapsules with 2 kDa PEG gave rise to the formation of an intermediate mushroom and to a dense brush conformation [
37]. The ovalbumin nanocapsules evaluated in the aforementioned study, that were modified with 5 kDa PEG, acquired the brush conformation even at a low density of 0.2 chains nm
−2 due to the higher molecular weight. In comparison to the ovalbumin nanocapsules, the PEGylated nanocarriers analyzed here also seem to differ in their PEG conformation, with a transition from the less-favorable mushroom conformation to the desired brush conformation, resulting in the greatest stealth effect for a complete amine to PEG conversion in macrophages and DCs. Previous PEGylation studies showed that the PEG-induced stealth effect is highly dependent on the density and conformation of the surface PEG chains. These parameters affect the formation of a PEG barrier on the nanocarrier surface, and therefore decrease opsonization and increase circulation time [
38,
39]. While the mushroom conformation with a larger PEG chain motion and unoccupied spaces on the surface is more susceptible to opsonins, the brush conformation with a limited PEG mobility and an increased surface coverage has been shown to reduce the interaction between the nanocarrier and the opsonins [
14,
40].
In addition, the shortest PEG linker (0.65 kDa) applied in our previous studies [
29], was tested in terms of RAW264.7 cell uptake (
Figure S1C,D). In comparison to the 2 and 5 kDa PEG linkers (
Figure 1), the 0.65 kDa PEG linker showed higher uptake values (both for the MFI and frequency of NC
+ cells) when FBS was present in the medium. This less pronounced stealth effect, compared to the 2 and 5 kDa PEGylated nanocarriers (50x excess), could also be influenced by the difference in the reactive group pointing to the cells. Namely, the methoxy (MeO) or the dibenzocyclooctyne (DBCO) groups, of which the latter one is necessary for clicking an azide-functionalized antibody onto the nanocarrier’s surface.
With that in mind and according to the findings described above, we decided to further investigate the 50-fold molar excess ratio of the different PEGylated nanocarrier formulations with either the MeO (for the 2 and 5 kDa linkers), or the DBCO-reactive end group (for the 0.65, 2, and 5 kDa linkers) with respect to RAW264.7 cell uptake. Additionally, the PEGylated samples were pre-incubated with mouse plasma for 1 h at 37 °C in order to mimic a more physiological condition (
Figure 2).
As a result, we found that the PEGylated samples in medium without FBS and pre-incubated in mouse plasma showed almost no differences, indicating that the murine biomolecular corona is hardly involved in conveying or preventing the stealth effect. Overall, the uptake analysis showed that in every sample group the frequency of NC
+ cells was above 80% (
Figure S3). However, based on the MFI, we observed that the PEGylated samples were taken up to a much lower degree. For both, the medium without FBS and the mouse plasma pre-incubation, the greatest stealth effect could be observed for the higher molecular weight PEG linker (5 kDa) with the MeO end group. Carrying DBCO at the end group resulted in a generally higher uptake compared to MeO. This influence of the DBCO group was particularly evident for the shortest PEG linker. The FBS-containing medium showed weaker but comparable results to the other two medium conditions. However, the use of bovine-derived serum in a murine cell uptake study is relatively artificial, since it does not resemble a physiological environment and can bias the cell uptake results [
41].
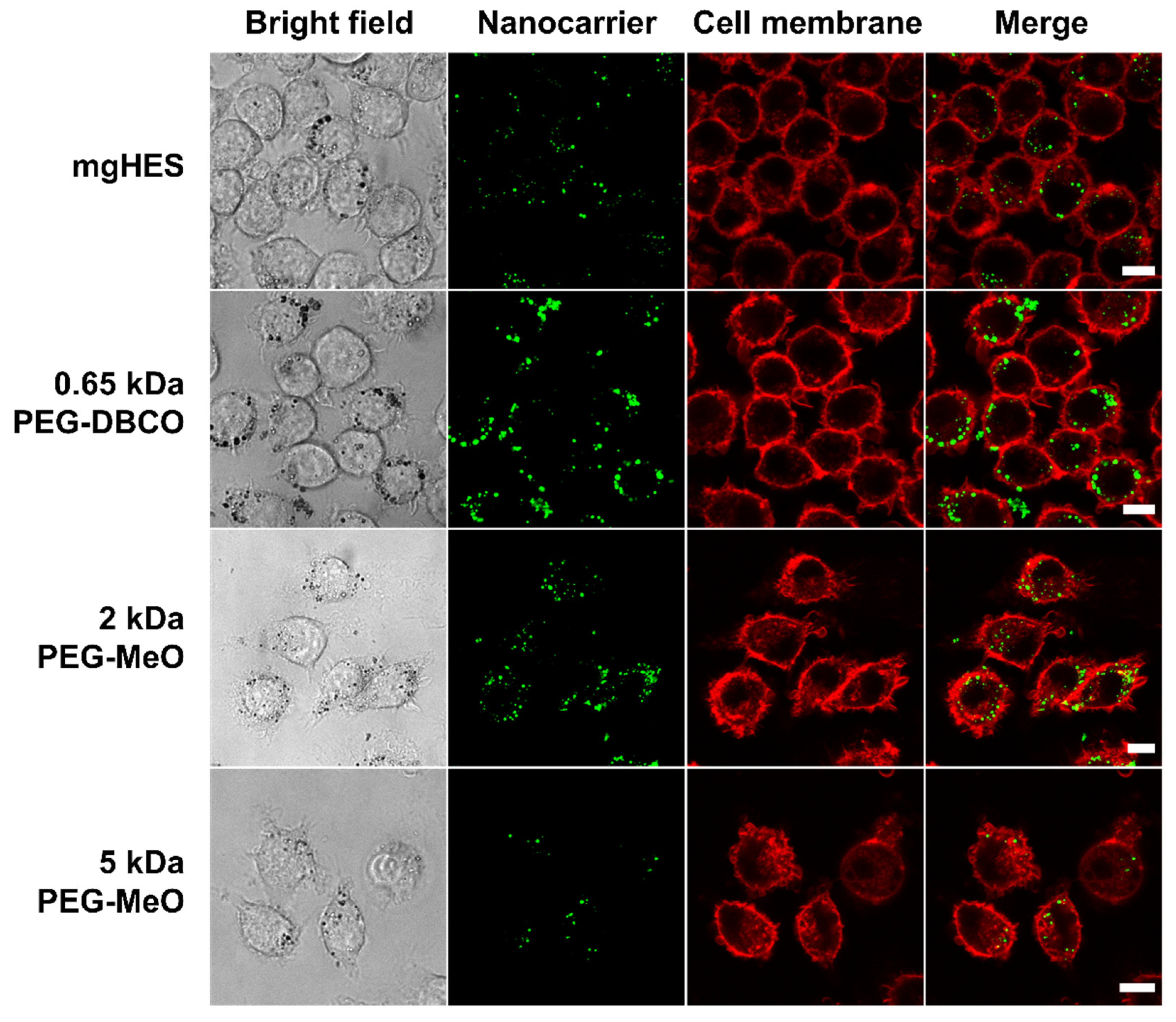
Confocal laser scanning microscopy was used to further depict the stealth effect and the cellular localization of our nanocarriers that were either sticking to the cell membrane or being internalized (
Figure 3). Therefore, the uptake of pristine and PEGylated nanocarriers, either pre-incubated in mouse plasma (no FBS present in medium), or in medium with 10% FBS was investigated in RAW264.7 cells (
Figure S4). Confocal laser scanning microscopy imaging on dendritic cells (DC2.4) was performed in a previous study showing preferential uptake of anti-CD11c-functionalized mgHES nanocarriers [
29].
Visually, almost all nanocarrier formulations were internalized by the cells with only a few localized at the cell membrane. Only the 5 kDa PEGylated sample could be clearly distinguished from the other sample groups. Here, we found a lower number of nanocarriers as well as a weaker fluorescence signal. In comparison to the uptake in FBS-containing medium, a generally lower nanocarrier uptake of all samples could be observed. Here, the longer PEG chains with 2 and 5 kDa led to a reduction in cell internalization. The stealth properties of the PEG linkers with the DBCO-reactive group could not be visualized microscopically, and were therefore not included in the 2 and 5 kDa linkers.
In a next step, the influence of the biomolecular corona on the stealth effect was determined by quantitatively analyzing the murine proteome of pre-incubated, PEGylated nanocarriers (all with a 50-fold molar excess of linker) via label-free mass spectrometry (LC-MS). All identified proteins could be correlated to eight protein groups and the respective values were plotted as the percentage based on all identified proteins (
Figure 4A). In comparison to the composition of the mouse plasma control, the percentage of serum albumin within the protein corona of the pristine nanocarriers (mgHES) increased significantly while the frequency of proteins of all other groups slightly decreased. A similar finding could be observed for all PEGylated samples irrespective of the PEG linker length used, even though the elevation in serum albumin content was not as pronounced as for the pristine nanocarriers. Qualitative analysis using SDS-PAGE further revealed serum albumin to be the strongest band with a molecular weight of about 62 kDa as detected by silver staining (
Figure S5A).
Additionally, the top seven most abundant corona proteins detected in each group were plotted as a heatmap (
Figure 4B). In parallel to the protein group analysis, serum albumin was found to be by far the most abundant corona protein for every nanocarrier formulation. While serum albumin presence was detected in the range of 48% (5 kDa PEG-DBCO) to 72% (mgHES), the other six most abundant proteins were observed with a relative abundance of less than 7% each. The whole mouse plasma control group showed a comparable distribution with the most abundant proteins (
Figure S6).
Apart from complement C3, none of the other abundant murine proteins is thought to influence uptake by macrophages. C3 coated on nanocarriers, however, can lead to an increased internalization of NCs by macrophages through complement receptor ligation when being activated by pathway-specific C3 convertases [
42,
43]. However, the relatively low abundance of C3 on each nanocarrier formulation (~2%) indicates that C3-induced uptake by macrophages only plays a minor role in this carrier system. Walkey et al. demonstrated that higher PEG grafting densities control a decrease and change in the composition of the protein corona, where the nanoparticle size and the PEG density together determine the macrophage uptake [
44]. In contrast, PEGylation of the nanocarrier formulations described here increased the rate of protein adsorption in comparison to the pristine nanocarriers (
Figure S7). Nevertheless, this increase in corona protein adsorption did not diminish the stealth effect of PEGylated nanocarriers. In addition, the physicochemical properties of the PEGylation with and without a protein corona were evaluated with respect to nanocarrier size and surface charge (
Table S1). Multi-angle dynamic light scattering revealed a larger size of pristine nanocarriers compared to the PEGylated formulations in protein-free media, and a smaller particle size under protein corona conditions. This might be due to changes in the light scattering properties and/or a reduction in aggregation after PEGylation. Regarding the surface charge of the nanocarriers, all samples presented a rather neutral zeta potential, which consistently appeared slightly negative for all mouse plasma pre-incubated nanocarrier formulations. In addition, TEM was used to visually confirm the nanocarrier size and morphology (
Figure S8). There were no visual differences between the pristine and PEGylated nanocarriers detected. All formulations appeared to form small cluster duplets with a diameter of about 200 nm. In summary, the experiments conducted during this study exhibited a reduced but not fully diminished protein corona formation following PEGylation of mgHES nanocarriers, which in turn did not have an influence on the PEG-induced stealth properties.
In addition to avoiding non-specific clearance of nanocarriers by phagocytes, conjugating active targeting moieties onto nanocarriers is the main prerequisite for a precise targeting of distinct DC subsets. Each DC subset is characterized by its ability to migrate, cytokine secretion, and antigen presentation. Hence, the selection of a distinct subtype critically affects a realistic DC vaccination approach. Applying the pan-DC marker CD11c as a target receptor has already underlined the role of DCs in enhancing humoral responses [
45,
46,
47,
48]. Prior to the investigation of primary DCs, the influence of the length of the different DBCO-containing PEG linkers on the antibody-based targeting ability of the nanocarrier formulations used here were investigated in the DC2.4 cell line. In order to evaluate the stealth targeting, azide-modified CD11c antibodies were covalently bound onto the DBCO-functionalized surface as described previously [
29]. Additionally, the corresponding isotype control antibody was included as a control for non-specific, Fc-mediated uptake. This control antibody was conjugated to each NC-PEG
X-DBCO linker group to differentiate between specific Fab- and undesired Fc-mediated cell binding. Conjugates with the 0.65 kDa PEG-DBCO linker were generated using a 10-fold molar excess of the linker, while the 2 kDa and 5 kDa molecular weight NC-PEG
X-DBCO linkers were conjugated with a 50-fold molar excess. Azide-modified antibodies were conjugated at a nanocarrier to antibody weight ratio of 67:1.
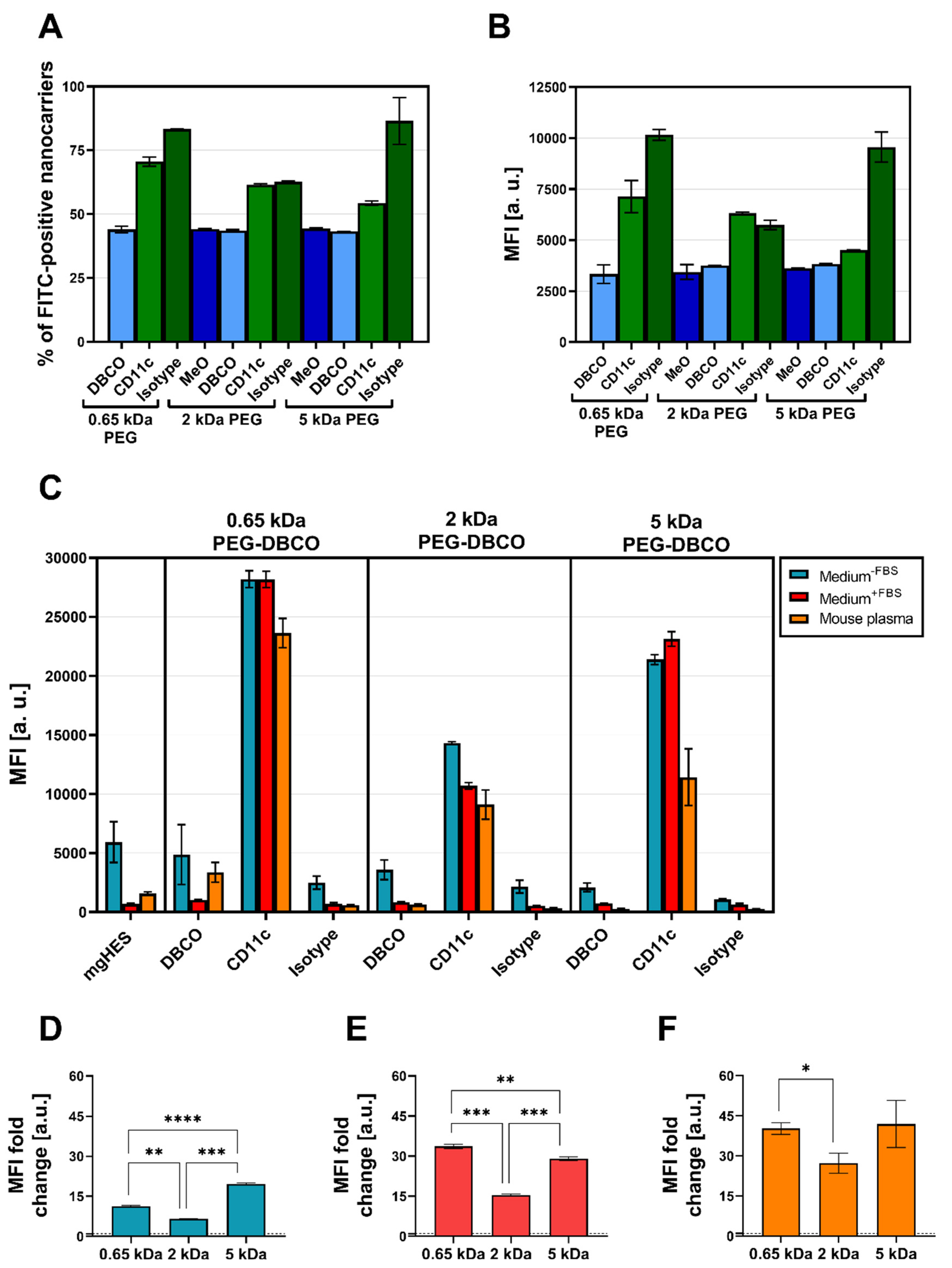
Flow cytometry was used to validate the successful covalent binding of the CD11c and isotype antibodies on the PEG-DBCO linker-modified nanocarrier surface (
Figure 5A,B). To achieve this, antibodies immobilized on the nanocarrier surface were stained with an anti-hamster FITC secondary antibody. The binding of the secondary antibody, either via adsorption onto the nanocarrier’s surface (no covalent-bound antibody present), or a positive, non-covalent binding towards the Fc region of the covalently attached antibodies, enabled a qualitative detection method. In this way, we confirmed the presence of the covalently bound antibodies on the surface for each formulation. Subsequently, the CD11c targeting ability of functionalized nanocarriers was examined in terms of uptake by DC2.4 cells under three different medium conditions via flow cytometry (
Figure 5C). For each condition, the targeting ability of anti-CD11c-modified nanocarriers was clearly distinguishable from the isotype control-functionalized formulations. Only the CD11c nanocarrier conjugates showed a significant uptake by the DC2.4 cells, while the pristine nanocarriers, the corresponding DBCO linker conjugates, and the isotype conjugates remained at lower values. Comparing the three CD11c nanocarrier conjugate groups, the highest uptake was observed for the 0.65 kDa PEG-CD11c, followed by the 5 kDa PEG-CD11c, and 2 kDa PEG-CD11c being the weakest for culture medium with or without FBS. Applying mouse plasma pre-incubated nanocarriers, only a minor difference between the 2 and 5 kDa PEG-CD11c could be observed, while the 0.65 kDa PEG-CD11c exhibited a substantial uptake. The anti-CD11c/isotype control ratio based on the respective MFI values was calculated in order to evaluate the influence of different PEG chain lengths on the specificity of antibody-mediated targeting (
Figure 5D–F). Among the three medium conditions, the ratio demonstrated a particularly low influence of the Fc region when applying the 5 kDa CD11c-functionalized nanocarriers, which was even more pronounced under medium conditions that led to protein corona formation (Medium
+FBS E, or mouse plasma pre-incubated, F).
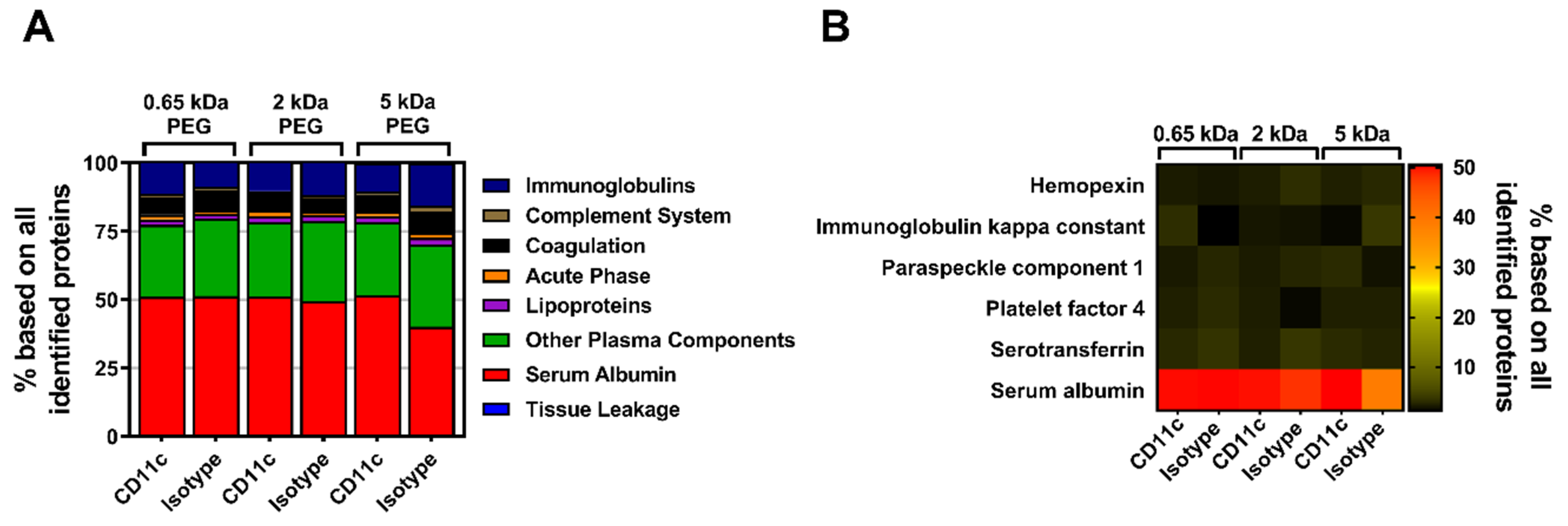
In parallel to the MeO-PEG-coupled nanocarriers, the influence of the biomolecular corona of anti-CD11c-functionalized nanocarriers on their targeting ability was analyzed and quantitative LC-MS was performed. Again, all identified proteins were assigned to eight protein groups and the respective values were plotted as the percentage based on all identified proteins (
Figure 6A). No major differences between the three molecular weight PEG groups with either the CD11c targeting or the isotype control antibody could be detected. All of the individual groups exhibited serum albumin, which had been highly adsorbed onto the nanocarrier’s surface.
Likewise, the qualitative SDS-PAGE detection of desorbed corona proteins showed serum albumin as the most abundant protein as visualized by silver staining (
Figure S5B). Moreover, by depicting the top six corona proteins we further highlighted that serum albumin has the highest abundance of all identified proteins (
Figure 6B). As seen before with PEGylation, the analysis of the murine biomolecular corona showed that the surface-modification of the nanocarriers with PEG, or in combination with antibodies only, slightly influenced the protein adsorption profile. In general, only a low number of proteins adsorbed onto the nanocarrier’s surface with very little influence on the stealth targeting ability.
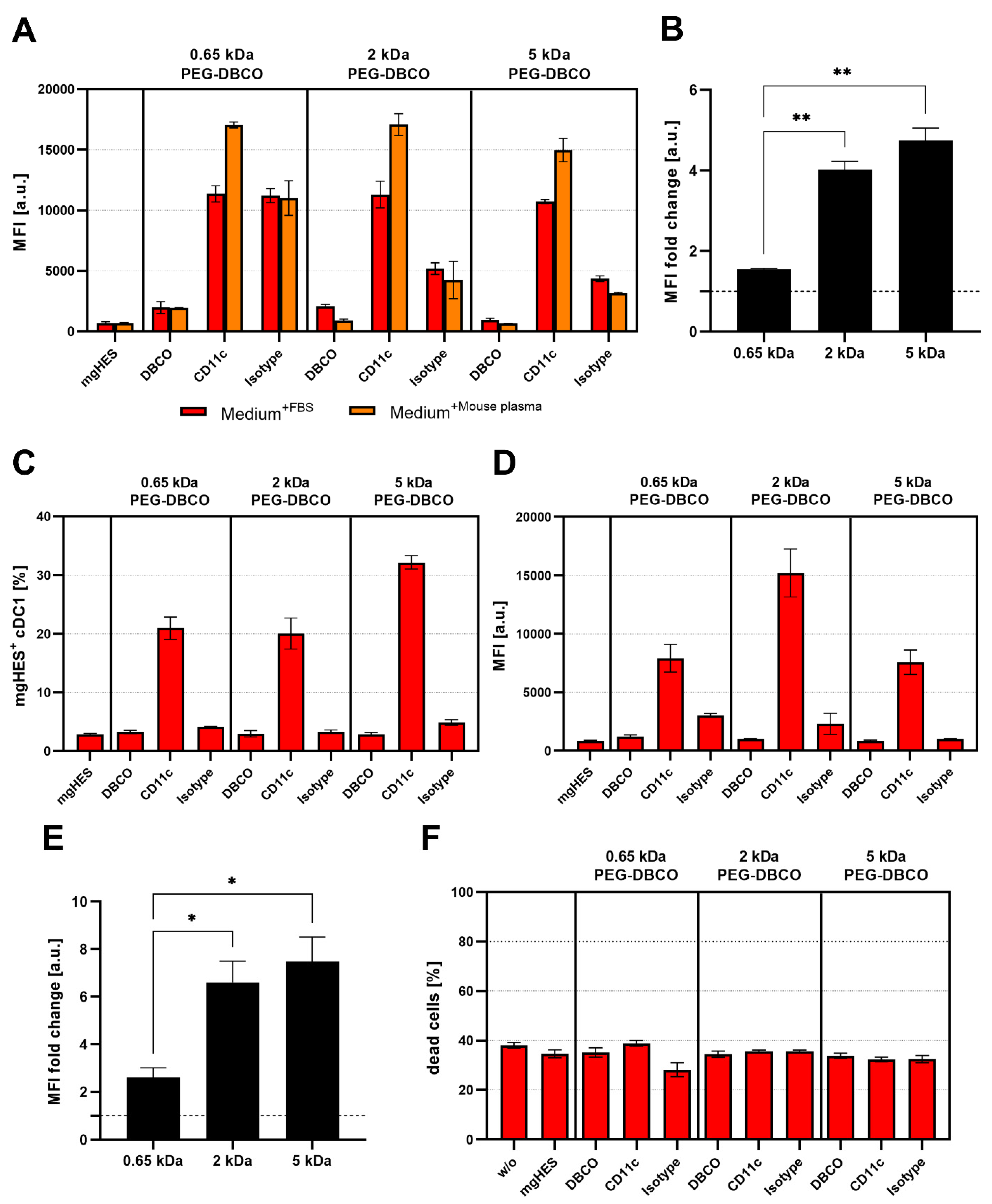
While conducting the uptake studies using the DC2.4 cell line, we also analyzed the influence of the PEG linker length on the uptake of anti-CD11c-functionalized nanocarriers in primary DCs, such as BMDCs and cDC1s of the spleen (
Figure 7A–E). In contrast to the uptake studies in DC2.4 cells, the targeting ability of anti-CD11c-functionalized nanocarriers was reduced when incubated with BMDCs (
Figure 7A). This was even more pronounced for incubation in the presence of FBS. However, the targeting ability, in terms of the anti-CD11c/isotype control ratio, was significantly increased for the longer PEG-DBCO linkers 2 kDa and 5 kDa by 4.0-fold and 4.75-fold, respectively, compared to 0.65 kDa PEG-DBCO (1.55-fold) (
Figure 7B). When analyzing cDC1s within whole splenocytes cultures, anti-CD11c-functionalized nanocarriers exhibited a substantial targeting ability compared to pristine, DBCO-, or isotype-functionalized nanocarriers (
Figure 7C–E). The uptake of IgG-functionalized nanocarriers was almost reduced to background levels in contrast to BMDC cultures, in terms of frequency of NC
+ cells (
Figure 7C), as well as MFI (
Figure 7D). In line with the data obtained from the incubation with DC2.4 cells and BMDCs, the PEG-DBCO linker length significantly influenced the anti-CD11c-mediated uptake of nanocarriers. While the 0.65 kDa linker only led to a 2.6-fold higher uptake rate compared to the isotype control, 2 kDa and 5 kDa resulted in significantly higher specific uptake by 6.6-fold and 7.5-fold, respectively (
Figure 7E). Additionally, the toxicity of different nanocarrier formulations was evaluated by co-incubation with splenocytes for 20 h and staining of dead cells with LIVE/DEAD Aqua Dead Cell Stain (
Figure 7F). Flow cytometric analyses revealed no significant changes in the percentage of dead cells when comparing all nanocarrier formulations under study.