Pharmacokinetic Evaluation of a Novel Transdermal Ketoprofen Formulation in Healthy Dogs
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vitro Experiments
2.1.1. Skin Samples
2.1.2. Drugs Used for Screening
2.1.3. In Vitro Experimentation
2.2. Pharmacokinetic Study
2.2.1. Animals
2.2.2. Drug Treatments and Sample Collection
2.3. Ketoprofen Analysis
2.4. Pharmacokinetic Parameters Calculation
2.5. Data Analysis
3. Results
3.1. In Vitro Screening
3.2. Pharmacokinetic Study
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lascelles, B.D.X.; King, S.; Roe, S.; Marcellin-Little, D.J.; Jones, S. Expression and Activity of COX-1 and 2 and 5-LOX in Joint Tissues from Dogs with Naturally Occurring Coxofemoral Joint Osteoarthritis. J. Orthop. Res. 2009, 27, 1204–1208. [Google Scholar] [CrossRef]
- Poradowski, D.; Obmińska-Mrukowicz, B. Effect of Selected Nonsteroidal Anti-Inflammatory Drugs on the Viability of Canine Osteosarcoma Cells of the D-17 Line: In Vitro Studies. J. Vet. Res. 2019, 63, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Rençber, S.; Karavana, S.Y.; Özyazici, M. Bioavailability File: Ketoprofen. FABAD J. Pharm. Sci. 2009, 34, 203–216. [Google Scholar]
- Plessers, E.; Watteyn, A.; Wyns, H.; Pardon, B.; De Baere, S.; De Backer, P.; Croubels, S. Enantioselective Pharmacokinetics of Ketoprofen in Calves after Intramuscular Administration of a Racemic Mixture. J. Vet. Pharmacol. Ther. 2015, 38, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Schellack, N.; Schellack, G.; Fourie, J. A Review of Nonsteroidal Anti-Inflammatory Drugs. SA Pharm. J. 2015, 82, 8–18. [Google Scholar]
- Narita, T.; Tomizawa, N.; Sato, R.; Goryo, M.; Hara, S. Effects of Long-Term Oral Administration of Ketoprofen in Clinically Healthy Beagle Dogs. J. Vet. Med. Sci. 2005, 67, 847–853. [Google Scholar] [CrossRef][Green Version]
- Willer, J.-C.; De Broucker, T.; Bussel, B.; Roby-Brami, A.; Harrewyn, J.-M. Central Analgesic Effect of Ketoprofen in Humans: Electrophysiological Evidence for a Supraspinal Mechanism in a Double-Blind and Cross-over Study. Pain 1989, 438, 1–7. [Google Scholar] [CrossRef]
- De Beaurepaire, R.; Suaudeau, C.; Chait, A.; Cimetière, C. Anatomical Mapping of Brain Sites Involved in the Antinociceptive Effects of Ketoprofen. Brain Res. 1990, 536, 201–206. [Google Scholar] [CrossRef]
- Díaz-Reval, M.I.; Ventura-Martínez, R.; Déciga-Campos, M.; Terrón, J.A.; Cabré, F.; López-Muñoz, F.J. Evidence for a Central Mechanism of Action of S-(+)-Ketoprofen. Eur. J. Pharmacol. 2004, 483, 241–248. [Google Scholar] [CrossRef]
- Monteiro, B.P.; Lambert, C.; Bianchi, E.; Genevois, J.P.; Soldani, G.; Troncy, E. Safety and Efficacy of Reduced Dosage Ketoprofen with or without Tramadol for Long-Term Treatment of Osteoarthritis in Dogs: A Randomized Clinical Trial. BMC Vet. Res. 2019, 15, 213. [Google Scholar] [CrossRef]
- Baba, M.A.; Fazili, M.R.; Molvi, B.A.; Athar, H.; Khan, I. Comparison of the Analgesic Effects of Meloxicam with Those of Ketoprofen in Male Stray Dogs Undergoing Pinhole Castration. Asian J. Anim. Sci. 2012, 6, 164–173. [Google Scholar] [CrossRef][Green Version]
- Hazewinkel, H.A.W.; van den Brom, W.E.; Theijse, L.F.H.; Pollmeier, M.; Hanson, P.D. Reduced Dosage of Ketoprofen for the Short-Term and Long-Term Treatment of Joint Pain in Dogs. Vet. Rec. 2003, 152, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Deneuche, A.J.; Dufayet, C.; Goby, L.; Fayolle, P.; Desbois, C. Analgesic Comparison of Meloxicam or Ketoprofen for Orthopedic Surgery in Dogs. Vet. Surg. 2004, 33, 650–660. [Google Scholar] [CrossRef]
- Martins, T.L.; Kahvegian, M.A.P.; Noel-morgan, J.; Leon-román, M.A.; Otsuki, D.A.; Fantoni, D.T. Maxillectomy or Mandibulectomy. Am. J. Vet. Res. 2010, 71, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.P.; Basilio, A.C.; Steagall, P.V.M.; Machado, L.P.; Moutinho, F.Q.; Takahira, R.K.; BRandao, C.V.S. Evaluation of Adverse Effects of Long-Term Oral Administration of Carprofen, Etodolac, Flunixin Meglumine, Ketoprofen, and Meloxicam in Dogs. Am. J. Vet. Res. 2007, 68, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, S.F.; Guilford, W.G.; Haslett, S.J.; Godfrey, J. Endoscopy of the Gastroduodenal Mucosa after Carprofen, Meloxicam and Ketoprofen Administration in Dogs. J. Small Anim. Pract. 1998, 39, 421–424. [Google Scholar] [CrossRef]
- Forsyth, S.F.; Guilford, W.G.; Pfeiffer, D.U. Effect of NSAID Administration on Creatinine Clearance in Healthy Dogs Undergoing Anaesthesia and Surgery. J. Small Anim. Pract. 2000, 41, 547–550. [Google Scholar] [CrossRef]
- Roth, S.H.; Shainhouse, J.Z. Efficacy and Safety of a Topical Diclofenac Solution (Pennsaid) in the Treatment of Primary Osteoarthritis of the Knee: A Randomized, Double-Blind, Vehicle-Controlled Clinical Trial. Arch. Intern. Med. 2004, 164, 2017–2023. [Google Scholar] [CrossRef]
- Cannavino, C.R.; Abrams, J.; Palinkas, L.A.; Saglimbeni, A.; Bracker, M.D. Efficacy of Transdermal Ketoprofen for Delayed Onset Muscle Soreness. Clin. J. Sport Med. 2003, 13, 200–208. [Google Scholar] [CrossRef][Green Version]
- Jadhav, P.; Sinha, R.; Uppada, U.K.; Tiwari, P.K.; Subramanya Kumar, A.V.S.S. Pre-Emptive Diclofenac Versus Ketoprofen as a Transdermal Drug Delivery System: How They Face. J. Maxillofac. Oral Surg. 2018, 17, 488–494. [Google Scholar] [CrossRef]
- Chi, S.-C.; Jun, H.W. Anti-inflammatory Activity of Ketoprofen Gel on Carrageenan-induced Paw Edema in Rats. J. Pharmac. Sci. 1990, 79, 974–977. [Google Scholar] [CrossRef] [PubMed]
- Aliberti, A.L.M.; de Queiroz, A.C.; Praça, F.S.G.; Eloy, J.O.; Bentley, M.V.L.B.; Medina, W.S.G. Ketoprofen Microemulsion for Improved Skin Delivery and In Vivo Anti-Inflammatory Effect. AAPS PharmSciTech 2017, 18, 2783–2791. [Google Scholar] [CrossRef] [PubMed]
- Bariguian Revel, F.; Fayet, M.; Hagen, M. Topical Diclofenac, an Efficacious Treatment for Osteoarthritis: A Narrative Review. Rheumatol. Ther. 2020, 7, 217–236. [Google Scholar] [CrossRef] [PubMed]
- Manoukian, M.A.C.; Migdal, C.W.; Tembhekar, A.R.; Harris, J.A.; DeMesa, C. Topical Administration of Ibuprofen for Injured Athletes: Considerations, Formulations, and Comparison to Oral Delivery. Sport. Med. Open 2017, 3, 36. [Google Scholar] [CrossRef]
- Higashi, Y.; Kiuchi, T.; Furuta, K. Efficacy and Safety Profile of a Topical Methyl Salicylate and Menthol Patch in Adult Patients with Mild to Moderate Muscle Strain: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled, Multicenter Study. Clin. Ther. 2010, 32, 34–43. [Google Scholar] [CrossRef]
- Mills, P.C.; Ghodasara, P.; Satake, N.; Alawneh, J.; Fraser, B.; Kopp, S.; McGowan, M. A Novel Transdermal Ketoprofen Formulation Provides Effective Analgesia to Calves Undergoing Amputation Dehorning. Animals 2020, 10, 2442. [Google Scholar] [CrossRef]
- Shinkai, N.; Korenaga, K.; Okumura, Y.; Mizu, H.; Yamauchi, H. Microdialysis Assessment of Percutaneous Penetration of Ketoprofen after Transdermal Administration to Hairless Rats and Domestic Pigs. Eur. J. Pharm. Biopharm. 2011, 78, 415–421. [Google Scholar] [CrossRef]
- Knych, H.K.; Arthur, R.M.; Gretler, S.R.; McKemie, D.S.; Goldin, S.; Kass, P.H. Pharmacokinetics of Transdermal Flunixin Meglumine and Effects on Biomarkers of Inflammation in Horses. J. Vet. Pharmacol. Ther. 2021, 44, 745–753. [Google Scholar] [CrossRef]
- Reppert, E.J.; Kleinhenz, M.D.; Montgomery, S.R.; Bornheim, H.N.; Magnin, G.; Sidhu, P.K.; Zhang, Y.; Joo, H.; Coetzee, J.F. Pharmacokinetics and Pharmacodynamics of Intravenous and Transdermal Flunixin Meglumine in Meat Goats. J. Vet. Pharmacol. Ther. 2019, 42, 309–317. [Google Scholar] [CrossRef]
- Kittrell, H.C.; Mochel, J.P.; Brown, J.T.; Forseth, A.M.K.; Hayman, K.P.; Rajewski, S.M.; Coetzee, J.F.; Schneider, B.K.; Ratliffe, B.; Skoland, K.J.; et al. Pharmacokinetics of Intravenous, Intramuscular, Oral, and Transdermal Administration of Flunixin Meglumine in Pre-Wean Piglets. Front. Vet. Sci. 2020, 7, 586. [Google Scholar] [CrossRef]
- Suh, H.; Jun, H.W.; Dzimianskl, M.T.; Lu, G.W. Pharmacokinetic and Local Tissue Disposition Studies of Naproxen Following Topical and Systemic Administration in Dogs and Rats. Biopharm. Drug Dispos. 1997, 18, 623–633. [Google Scholar] [CrossRef]
- Mills, P.C.; Magnusson, B.M.; Cross, S.E. Penetration of a Topically Applied Nonsteroidal Anti-Inflammatory Drug into Local Tissues and Synovial Fluid of Dogs. Am. J. Vet. Res. 2005, 66, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Chen, X.Y.; Li, S.M.; Wei, X.Y.; Yao, H.M.; Zhong, D.F. Pharmacokinetic Studies of Meloxicam Following Oral and Transdermal Administration in Beagle Dogs. Acta Pharmacol. Sin. 2009, 30, 1060–1064. [Google Scholar] [CrossRef] [PubMed]
- Baranowski, D.C.; Buchanan, B.; Dwyer, H.C.; Gabriele, J.P.; Kelly, S.; Araujo, J.A. Penetration and Efficacy of Transdermal NSAIDs in a Model of Acute Joint Inflammation. J. Pain Res. 2018, 11, 2809–2819. [Google Scholar] [CrossRef]
- Kukanich, B.; Clark, T.P. The History and Pharmacology of Fentanyl: Relevance to a Novel, Long-Acting Transdermal Fentanyl Solution Newly Approved for Use in Dogs. J. Vet. Pharmacol. Ther. 2012, 35, 3–19. [Google Scholar] [CrossRef]
- Pieper, K.; Schuster, T.; Levionnois, O.; Matis, U.; Bergadano, A. Antinociceptive Efficacy and Plasma Concentrations of Transdermal Buprenorphine in Dogs. Vet. J. 2011, 187, 335–341. [Google Scholar] [CrossRef]
- McNicol, E.; Strassels, S.; Goudas, L.; Lau, J.; Carr, D. Nonsteroidal Anti-Inflammatory Drugs, Alone or Combined with Opioids, for Cancer Pain: A Systematic Review. J. Clin. Oncol. 2004, 22, 1975–1992. [Google Scholar] [CrossRef]
- Pathan, S.A.; Mitra, B.; Cameron, P.A. A Systematic Review and Meta-Analysis Comparing the Efficacy of Nonsteroidal Anti-Inflammatory Drugs, Opioids, and Paracetamol in the Treatment of Acute Renal Colic [Figure Presented]. Eur. Urol. 2018, 73, 583–595. [Google Scholar] [CrossRef]
- Praça, F.S.G.; Medina, W.S.G.; Eloy, J.O.; Petrilli, R.; Campos, P.M.; Ascenso, A.; Bentley, M.V.L.B. Evaluation of Critical Parameters for in Vitro Skin Permeation and Penetration Studies Using Animal Skin Models. Eur. J. Pharm. Sci. 2018, 111, 121–132. [Google Scholar] [CrossRef]
- Ahlstrom, L.A.; Cross, S.E.; Mills, P.C. The Effects of Freezing Skin on Transdermal Drug Penetration Kinetics. J. Vet. Pharmacol. Ther. 2007, 30, 456–463. [Google Scholar] [CrossRef]
- Mills, P.C.; Cross, S. Transdermal drug delivery: Basic principles for the veterinarian. Vet. J. 2006, 172, 218–233. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Uboh, C.E.; Guan, F.; Soma, L.R. Doping Control Analysis of 16 Non-Steroidal Anti-Inflammatory Drugs in Equine Plasma Using Liquid Chromatography-Tandem Mass Spectrometry. Am. J. Anal. Chem. 2014, 5, 1184–1199. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Xie, S. PKSolver: An Add-in Program for Pharmacokinetic and Pharmacodynamic Data Analysis in Microsoft Excel. Comput. Methods Programs Biomed. 2010, 99, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Toutain, P.L.; Bousquet-Mélou, A. Plasma Terminal Half-Life. J. Vet. Pharmacol. Ther. 2004, 27, 427–439. [Google Scholar] [CrossRef]
- Granero, G.E.; Amidon, G.L. Possibility of Enterohepatic Recycling of Ketoprofen in Dogs. Int. J. Pharm. 2008, 349, 166–171. [Google Scholar] [CrossRef]
- Serrano-Rodríguez, J.M.; Serrano, J.M.; Rodríguez, J.M.; Machuca, M.M.G.; Gómez-Villamandos, R.J.; Navarrete-Calvo, R. Pharmacokinetics of the Individual Enantiomer S-(+)-Ketoprofen after Intravenous and Oral Administration in Dogs at Two Dose Levels. Res. Vet. Sci. 2014, 96, 523–525. [Google Scholar] [CrossRef]
- Neirinckx, E.; Croubels, S.; De Boever, S.; Remon, J.P.; Bosmans, T.; Daminet, S.; De Backer, P.; Vervaet, C. Species Comparison of Enantioselective Oral Bioavailability and Pharmacokinetics of Ketoprofen. Res. Vet. Sci. 2011, 91, 415–421. [Google Scholar] [CrossRef]
- Martinez, M.N.; Hunter, R.P. Current Challenges Facing the Determination of Product Bioequivalence in Veterinary Medicine. J. Vet. Pharmacol. Ther. 2010, 33, 418–433. [Google Scholar] [CrossRef]
- Wsol, V.; Skalova, L.; Szotakova, B. Chiral Inversion of Drugs: Coincidence or Principle? Curr. Drug Metab. 2004, 5, 517–533. [Google Scholar] [CrossRef]
- Jamali, F.; Brocks, D.R. Clinical Pharmacokinetics of Ketoprofen and Its Enantiomers. Clin. Pharmacokinet. 1990, 19, 197–217. [Google Scholar] [CrossRef]
- Andaluz, A.; Moll, X.; Ventura, R.; Abellán, R.; Fresno, L.; García, F. Plasma Buprenorphine Concentrations after the Application of a 70 Μg/h Transdermal Patch in Dogs. Preliminary Report. J. Vet. Pharmacol. Ther. 2009, 32, 503–505. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.K.; Wei, G.; Lanman, R.C.; Bhargava, V.O.; Weir, S.J. Percutaneous Absorption of Ketoprofen from Different Anatomical Sites. Pharm. Res. 1996, 13, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Dowling, T.C.; Arjomand, M.; Lin, E.T.; Allen, L.V.; McPherson, M.L. Relative Bioavailability of Ketoprofen 20% in a Poloxamer-Lecithin Organogel. Am. J. Health Pharm. 2004, 61, 2541–2544. [Google Scholar] [CrossRef] [PubMed]
- Montoya, L.; Ambros, L.; Kreil, V.; Bonafine, R.; Albarellos, G.; Hallu, R.; Soraci, A. A Pharmacokinetic Comparison of Meloxicam and Ketoprofen Following Oral Administration to Healthy Dogs. Vet. Res. Commun. 2004, 28, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Mathews, K.A. Non-Steroidal Anti-Inflammatory Analgesics for Acute Pain Management in Dogs and Cats. Can. Vet. J. 1996, 37, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Heyneman, C.A.; Lawless-Liday, C.; Wall, G.C. Oral versus Topical NSAIDs in Rheumatic Diseases: A Comparison. Drugs 2000, 60, 555–574. [Google Scholar] [CrossRef]
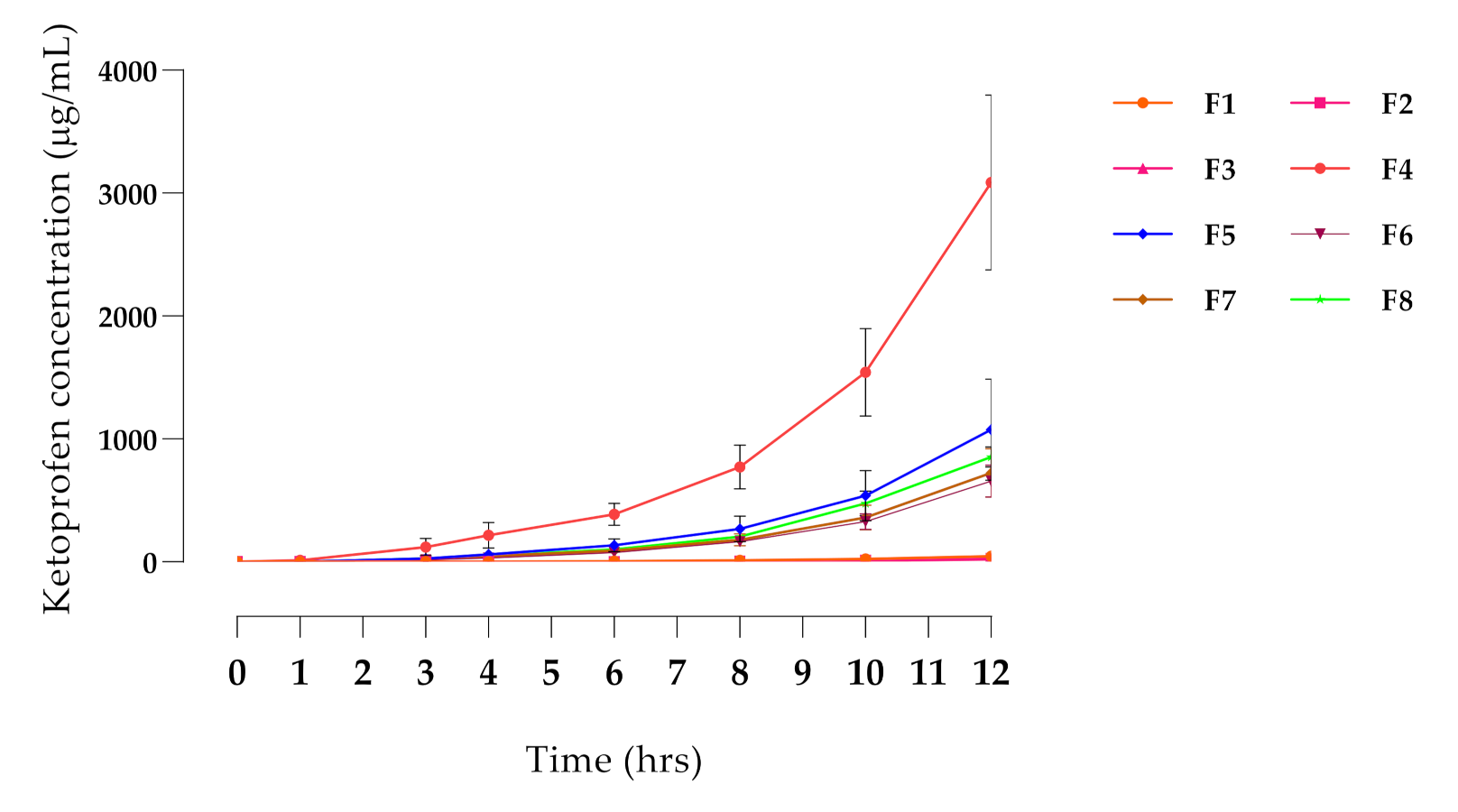
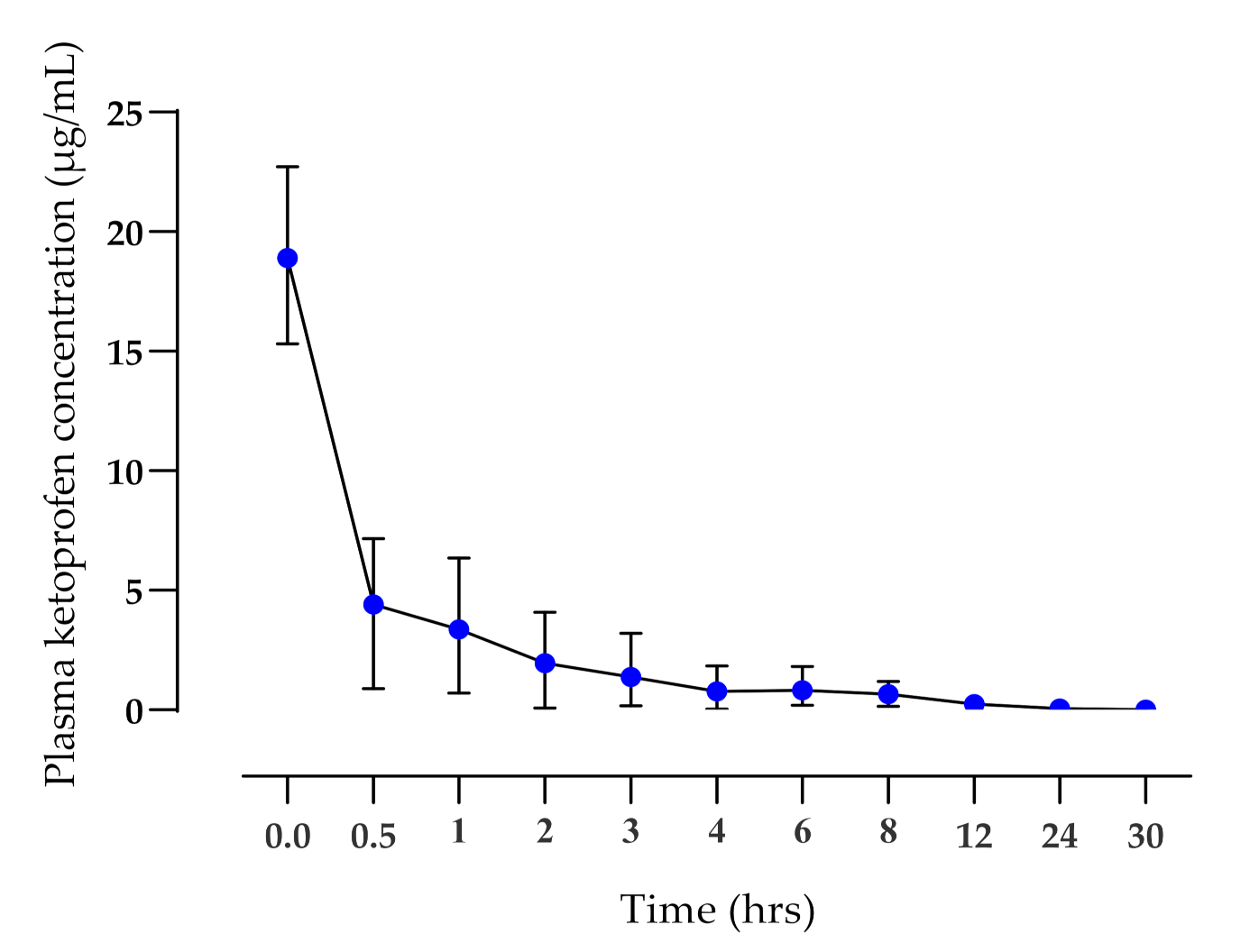
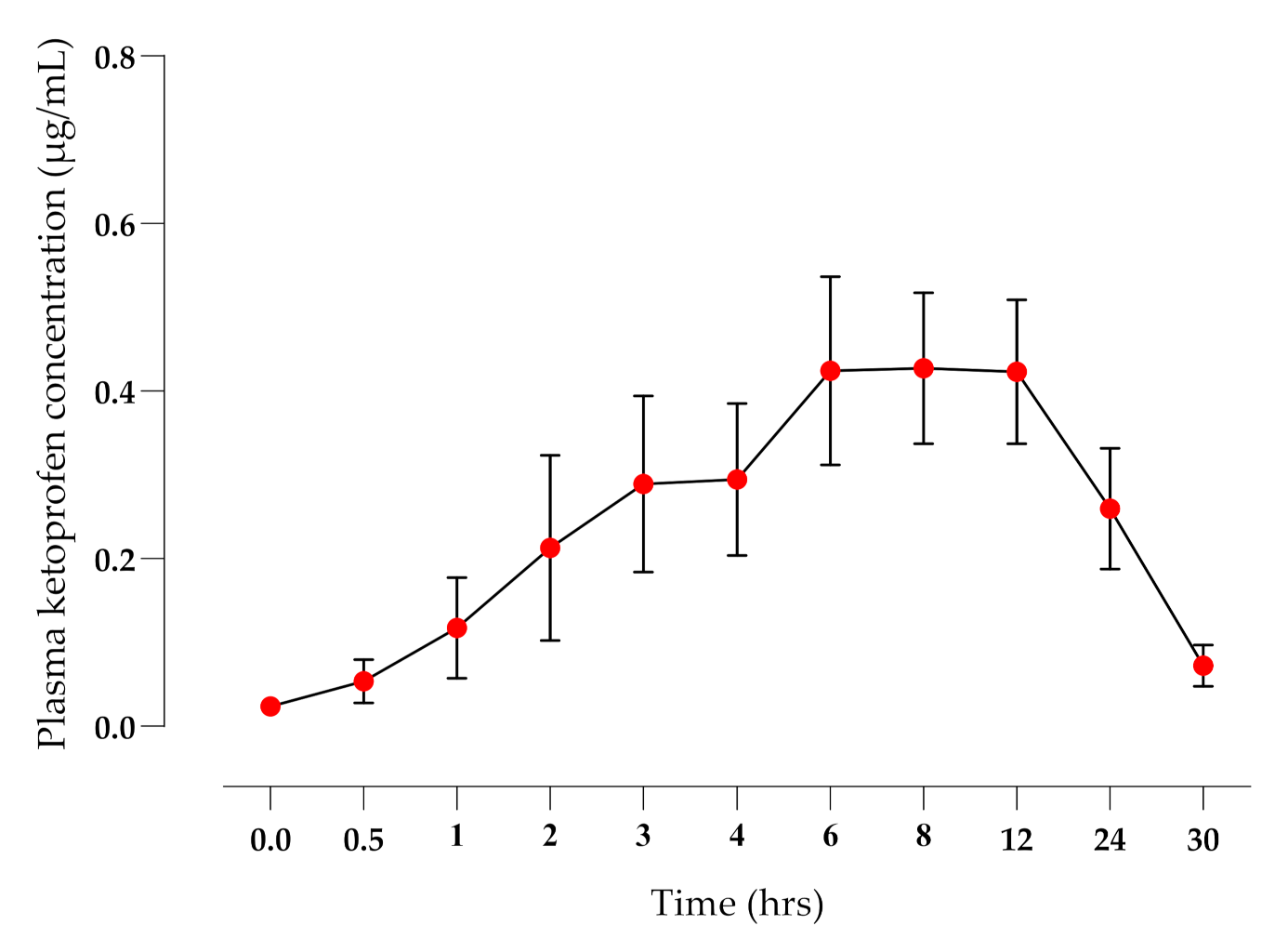
| Parameter | Unit | IV Route (Mean ± SD) | TD Route (Mean ± SD) |
|---|---|---|---|
| Lambda_z (λz) | L/h | 0.16 ± 0.04 | 0.05 ± 0.06 * |
| T½ | h | 4.69 ± 1.33 | 25.77 ± 22.14 * |
| TMAX | h | 0.03 ± 0.0 | 9.14 ± 3.98 * |
| CMAX | µg/mL | 16.43 ± 5.32 | 0.52 ± 0.28 * |
| AUC0-t | µg·h/mL | 15.75 ± 7.72 | 8.13 ± 4.28 * |
| AUC0-∞ | µg·h/mL | 16.71 ± 8.46 | 19.61 ± 16.94 |
| MRT | h | 4.86 ± 1.81 | 41.63 ± 32.33 * |
| Vd | L/kg | 0.48 ± 0.18 | 21.69 ± 15.64 * |
| Cl | L/h kg | 0.08 ± 0.04 | 0.77 ± 0.43 * |
| F (bioavailability) | % | NA | 7.16 ± 7.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravuri, H.G.; Satake, N.; Balmanno, A.; Skinner, J.; Kempster, S.; Mills, P.C. Pharmacokinetic Evaluation of a Novel Transdermal Ketoprofen Formulation in Healthy Dogs. Pharmaceutics 2022, 14, 646. https://doi.org/10.3390/pharmaceutics14030646
Ravuri HG, Satake N, Balmanno A, Skinner J, Kempster S, Mills PC. Pharmacokinetic Evaluation of a Novel Transdermal Ketoprofen Formulation in Healthy Dogs. Pharmaceutics. 2022; 14(3):646. https://doi.org/10.3390/pharmaceutics14030646
Chicago/Turabian StyleRavuri, Halley Gora, Nana Satake, Alexandra Balmanno, Jazmine Skinner, Samantha Kempster, and Paul C. Mills. 2022. "Pharmacokinetic Evaluation of a Novel Transdermal Ketoprofen Formulation in Healthy Dogs" Pharmaceutics 14, no. 3: 646. https://doi.org/10.3390/pharmaceutics14030646
APA StyleRavuri, H. G., Satake, N., Balmanno, A., Skinner, J., Kempster, S., & Mills, P. C. (2022). Pharmacokinetic Evaluation of a Novel Transdermal Ketoprofen Formulation in Healthy Dogs. Pharmaceutics, 14(3), 646. https://doi.org/10.3390/pharmaceutics14030646






