Aqueous Prostaglandin Eye Drop Formulations
Abstract
1. Introduction
2. Physicochemical Properties and Eye Drop Formulations
3. Novel PGA Formulations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zukerman, R.; Harris, A.; Vercellin, A.V.; Siesky, B.; Pasquale, L.R.; Ciulla, T.A. Molecular genetics of glaucoma: Subtype and ethnicity considerations. Genes 2021, 12, 55. [Google Scholar] [CrossRef] [PubMed]
- Leske, M.C.; Heijl, A.; Hussein, M.; Bengtsson, B.; Hyman, L.; Komaroff, E. Factors for glaucoma progression and the effect of treatment: The early manifest glaucoma trial. Arch. Ophthalmol. 2003, 121, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Sali, T. Prostaglandins. In Encyclopedic Reference of Immunotoxicology; Assenmacher, M., Avraham, H.K., Avraham, S., Bala, S., Barnett, J., Basketter, D., Ben-David, Y., Berek, C., Blümel, J., Bolliger, A.P., et al., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 537–540. [Google Scholar]
- Shaw, J.E.; Ramwell, P.W. Prostaglandins: A general review. Res. Prostaglandins 1971, 1, 1–8. [Google Scholar] [PubMed]
- Camras, C.B.; Bito, L.Z.; Eakins, K.E. Reduction of intraocular pressure by prostaglandins applied topically to the eyes of conscious rabbits. Investig. Ophthalmol. Vis. Sci. 1977, 16, 1125–1134. [Google Scholar]
- Russo, A.; Riva, I.; Pizzolante, T.; Noto, F.; Quaranta, L. Latanoprost ophthalmic solution in the treatment of open angle glaucoma or raised intraocular pressure: A review. Clin. Ophthalmol. 2008, 2, 897–905. [Google Scholar] [PubMed]
- Wang, T.; Cao, L.; Jiang, Q.; Zhang, T. Topical medication therapy for glaucoma and ocular hypertension. Front. Pharmacol. 2021, 12, 749858. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, A.; Riva, I.; Bozkurt, B.; Holló, G.; Quaranta, L.; Oddone, F.; Irkec, M.; Dutton, G.N.; Konstas, A.G. A new look at the safety and tolerability of prostaglandin analogue eyedrops in glaucoma and ocular hypertension. Expert Opin. Drug Saf. 2022, 21, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.S.; Ju, W.-K.; Neufeld, A.H.; Abran, D.; Chemtob, S.; Roberts, L.J. Hydrolysis of Bimatoprost (Lumigan) to its free acid by ocular tissue in vitro. J. Ocul. Pharmacol. Ther. 2003, 19, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Moussa, W.G.E.H.; Farhat, R.G.; Nehme, J.C.; Sahyoun, M.A.; Schakal, A.R.; Jalkh, A.E.; Karam, M.P.A.; Azar, G.G. Comparison of efficacy and ocular surface disease index score between bimatoprost, latanoprost, travoprost, and tafluprost in glaucoma patients. J. Ophthalmol. 2018, 1319628. [Google Scholar] [CrossRef]
- Najjar, A.; Najjar, A.; Karaman, R. Newly developed prodrugs and prodrugs in development; an insight of the recent years. Molecules 2020, 25, 884. [Google Scholar] [CrossRef]
- Samaha, D.; Diaconu, V.; Bouchard, J.F.; Desalliers, C.; Dupont, A. Effect of latanoprostene bunod on optic nerve head blood flow. Optom. Vis. Sci. 2022, 99, 172–176. [Google Scholar] [CrossRef]
- Harasymowycz, P.; Royer, C.; Mathurin, K.; Lachaine, J.; Beauchemin, C.; Cui, A.X.; Barbeau, M.; Jobin-Gervais, K.; Mathurin, K.; Lachaine, J.; et al. Short-term efficacy of latanoprostene bunod for the treatment of open-angle glaucoma and ocular hypertension: A systematic literature review and a network meta-analysis. Br. J. Ophthalmol. 2022, 106, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Winkler, N.S.; Fautsch, M.P. Effects of prostaglandin analogues on aqueous humor outflow pathways. J. Ocul. Pharmacol. Ther. 2014, 30, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, F.; Topliss, J.G. Unified model for the corneal permeability of related and diverse compounds with respect to their physicochemical properties. J. Pharm. Sci. 1996, 85, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Loftsson, T. Topical drug delivery to the retina: Obstacles and routes to success. Expert Opin. Drug Deliv. 2022, 19, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Sripetch, S.; Loftsson, T. Topical drug delivery to the posterior segment of the eye: Thermodynamic considerations. Int. J. Pharm. 2021, 597, 120332. [Google Scholar] [CrossRef]
- Hageman, M.J. Prostaglandin E2. In Chemical Stability of Pharmaceuticals: A Handbook for Pharmacists, 2nd ed.; Connors, K.A., Amidon, G.L., Stella, V.J., Eds.; John Wiley & Sons: New York, NY, USA, 1986; pp. 719–727. [Google Scholar]
- Oesterling, T.O.; Morozowich, W.; Roseman, T.J. Prostaglandins. J. Pharm. Sci. 1972, 61, 1861–1895. [Google Scholar] [CrossRef] [PubMed]
- Stehle, R.G. Physical chemistry, stability, and handling of prostaglandins E2, F2α, D2, and I2: A critical summary. Methods Enzymol. 1982, 86, 436–458. [Google Scholar]
- Morgan, P.V.; Proniuk, S.; Blanchard, J.; Noecker, R.J. Effect of temperature and light on the stability of latanoprost and its clinical relevance. J. Glaucoma 2001, 10, 401–405. [Google Scholar] [CrossRef]
- Velpandian, T.; Kotnala, A.; Halder, N.; Ravi, A.K.; Archunan, V.; Sihota, R. Stability of latanoprost in generic formulations using controlled degradation and patient usage simulation studies. Curr. Eye Res. 2015, 40, 561–571. [Google Scholar] [CrossRef]
- Ochiai, A.; Iida, K.; Takabe, H.; Kawamura, E.; Sato, Y.; Kato, Y.; Ohkuma, M.; Danjo, K. Formulation design of latanoprost eye drops to improve the stability at room temperature. J. Pharm. Sci. Technol. 2010, 70, 324–332. [Google Scholar]
- Sawatdee, S.; Phetmung, H.; Srichana, T. Development of a stable latanoprost solution for use as eye drops. Chiang Mai J. Sci. 2013, 40, 656–668. [Google Scholar]
- Zhou, X.; Li, X.; Xu, J.; Cheng, Y.; Cao, F. Latanoprost-loaded cyclodextrin microaggregate suspension eye drops for enhanced bioavailability and stability. Eur. J. Pharm. Sci. 2021, 160, 105758. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Aller, M.; Guinchard, S.; Guillarme, D.; Pupier, M.; Jeannerat, D.; Rivara-Minten, E.; Veuthey, J.-L.; Gurny, R. New prostaglandin analog formulation for glaucoma treatment containing cyclodextrins for improved stability, solubility and ocular tolerance. Eur. J. Pharm. Biopharm. 2015, 95, 203–214. [Google Scholar] [CrossRef]
- Ochiai, A.; Danjo, K. The stabilization mechanism of latanoprost. Int. J. Pharm. 2011, 410, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Airy, S.; Chiou, J.; Nguyen, H.; Jani, R.; Gan, O.; Kabra, B.; Nguyen, H.; Weiner, A. Developmental preformulation studies in the design of travoprost ophthalmic solution 0.004% (TRAVANTAN®). In Proceedings of the American Association of Pharmaceutical Scientists (AAPS) Annual Meeting, Toronto, ON, Canada, 16–29 October 2002. [Google Scholar]
- Kahook, M.Y. Travoprost Z ophthalmic solution with sofZia: Clinical safety and efficacy. Expert Rev. Ophthalmol. 2007, 2, 363–368. [Google Scholar] [CrossRef]
- Alviset, G.; Corvis, Y.; Hammad, K.; Lemut, J.; Maury, M.; Mignet, N.; Boudy, V. New preservative-free formulation for the enhanced ocular bioavailability of prostaglandin analogues in glaucoma. Pharmaceutics 2022, 14, 453. [Google Scholar] [CrossRef]
- Loftson, T. Drug Stability for Pharmaceutical Scientists; Academic Press: Cambridge, CA, USA, 2014; p. 170. [Google Scholar]
- Johnson, T.V.; Gupta, P.K.; Vudathala, D.K.; Blair, I.A.; Tanna, A.P. Thermal stability of bimatoprost, latanoprost, and travoprost under simulated daily use. J. Ocul. Pharmacol. Ther. 2011, 27, 51–59. [Google Scholar] [CrossRef]
- Swymer, C.; Neville, M.W. Tafluprost: The first preservative-free prostaglandin to treat open-angle glaucoma and ocular hypertension. Ann. Pharmacother. 2012, 46, 1506–1510. [Google Scholar] [CrossRef]
- Kaufman, P.L. Latanoprostene bunod ophthalmic solution 0.024% for IOP lowering in glaucoma and ocular hypertension. Expert Opin. Pharmacother. 2017, 18, 433–444. [Google Scholar] [CrossRef]
- Baudouin, C.; Labbé, A.; Liang, H.; Pauly, A.; Brignole-Baudouin, F. Preservatives in eyedrops: The good, the bad and the ugly. Prog. Retin. Eye Res. 2010, 29, 312–334. [Google Scholar] [CrossRef]
- Goldstein, M.H.; Silva, F.Q.; Blender, N.; Tran, T.; Vantipalli, S. Ocular benzalkonium chloride exposure: Problems and solutions. Eye 2022, 36, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Johannsdottir, S.; Jansook, P.; Stefansson, E.; Kristinsdottir, I.M.; Asgrimsdottir, G.M.; Loftsson, T. Topical drug delivery to the posterior segment of the eye: The effect of benzalkonium chloride on topical dexamethasone penetration into the eye in vivo. J. Drug Deliv. Sci. Technol. 2018, 48, 125–127. [Google Scholar] [CrossRef]
- Xu, K.M.; Cho, R.; Chan, T.Y.B. Retrospective analysis of switching bimatoprost 0.01% to bimatoprost 0.03% in patients with various types of glaucoma and ocular hypertension. Clin. Ophthalmol. 2022, 16, 2385–2390. [Google Scholar] [CrossRef] [PubMed]
- Rosin, L.M.; Bell, N.P. Preservative toxicity in glaucoma medication: Clinical evaluation of benzalkonium chloride-free 0.5% timolol eye drops. Clin. Ophthalmol. 2013, 7, 2131–2135. [Google Scholar] [CrossRef]
- Pellinen, P.; Lokkila, J. Corneal penetration into rabbit aqueous humor is comparable between preserved and preservative-free tafluprost. Ophthalmic Res. 2009, 41, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Economou, M.A.; Laukeland, H.K.; Grabska-Liberek, I.; Rouland, J.F. Better tolerance of preservative-free latanoprost compared to preserved glaucoma eye drops: The 12-month real-life FREE study. Clin. Ophthalmol. 2018, 12, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Park, S.W.; Seong, M.; Ha, S.J.; Lee, J.W.; Rho, S.; Lee, C.E.; Kim, K.N.; Kim, T.W.; Sung, K.R.; et al. Comparison of the safety and efficacy between preserved and preservative-free latanoprost and preservative-free tafluprost. Pharmaceuticals 2021, 14, 501. [Google Scholar] [CrossRef]
- Uekama, K.; Hirayama, F.; Fujise, A.; Otagiri, M.; Inaba, K.; Saito, H. Inclusion complexation of prostaglandin F2 alpha with gamma-cyclodextrin in solution and solid phases. J. Pharm. Sci. 1984, 73, 382–384. [Google Scholar] [CrossRef]
- Gu, F.-G.; Cui, F.-D.; Gao, Y.-L. Effect of complexation with hydroxylpropyl-β-cyclodextrin on solubility, dissolution rate and chemical stability of prostaglandin E1. J. Chin. Pharm. Sci. 2004, 13, 158–165. [Google Scholar]
- Uekama, K.; Hirayama, F.; Yamada, Y.; Inaba, K.; Ikeda, K. Improvements of dissolution characteristics and chemical stability of 16,16-dimethyl-trans-Δ2-prostaglandin E1 methyl ester by cyclodextrin complexation. J. Pharm. Sci. 1979, 68, 1059–1060. [Google Scholar] [CrossRef]
- Inaba, K.; Wakuda, T.; Uekama, K. Prostaglandins and their cyclodextrin complexes. J. Incl. Phenom. 1984, 2, 467–474. [Google Scholar] [CrossRef]
- Uekama, K.; Hieda, Y.; Hirayama, F.; Arima, H.; Sudoh, M.; Yagi, A.; Terashima, H. Stabilizing and solubilizing effects of sulfobutyl ether β-cyclodextrin on prostaglandin E1 analog. Pharm. Res. 2001, 18, 1578–1585. [Google Scholar] [CrossRef]
- Kumar, P.V.T.; Kumar, A.A.; Rao, N.S. Formulation and evaluation of tafluprost ophthalmic solution. J. Drug Deliv. Ther. 2019, 9, 18–24. [Google Scholar] [CrossRef]
- Gonzalez, J.R.; Baiza-Duran, L.; Quintana-Hau, J.; Tornero-Montaño, R.; Castaneda-Hernandez, G.; Ortiz, M.; Alarcon-Oceguera, F.; Beltran-Loustaunau, M.; Cortez-Gastelum, M.; Garcidueñas-Mejia, J.; et al. Comparison of the stability, efficacy, and adverse effect profile of the innovator 0.005% latanoprost ophthalmic solution and a novel cyclodextrin-containing formulation. J. Clin. Pharmacol. 2007, 47, 121–126. [Google Scholar] [CrossRef]
- Battaglia, L.; Serpe, L.; Foglietta, F.; Muntoni, E.; Gallarate, M.; Del Pozo Rodriguez, A.; Solinis, M.A. Application of lipid nanoparticles to ocular drug delivery. Expert Opin. Drug Deliv. 2016, 13, 1743–1757. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Nair, A.B.; Shah, J.; Gupta, S.; Boddu, S.H.S.; Sreeharsha, N.; Joseph, A.; Shinu, P.; Morsy, M.A. Lipid nanoparticles as a promising drug delivery carrier for topical ocular therapy-An overview on recent advances. Pharmaceutics 2022, 14, 533. [Google Scholar] [CrossRef]
- Habib, F.; El-Mahdy, M.; Maher, S. Microemulsions for ocular delivery: Evaluation and characterization. J. Drug Deliv. Sci. Technol. 2011, 21, 485–489. [Google Scholar] [CrossRef]
- Singh, M.; Bharadwaj, S.; Lee, K.E.; Kang, S.G. Therapeutic nanoemulsions in ophthalmic drug administration: Concept in formulations and characterization techniques for ocular drug delivery. J. Control. Release 2020, 328, 895–916. [Google Scholar] [CrossRef] [PubMed]
- Tamilvanan, S.; Benita, S. The potential of lipid emulsion for ocular delivery of lipophilic drugs. Eur. J. Pharm. Biopharm. 2004, 58, 357–368. [Google Scholar] [CrossRef]
- Liang, H.; Baudouin, C.; Faure, M.O.; Lambert, G.; Brignole-Baudouin, F. Comparison of the ocular tolerability of a latanoprost cationic emulsion versus conventional formulations of prostaglandins: An in vivo toxicity assay. Mol. Vis. 2009, 15, 1690–1699. [Google Scholar]
- Daull, P.; Buggage, R.; Lambert, G.; Faure, M.O.; Serle, J.; Wang, R.F.; Garrigue, J.S. A comparative study of a preservative-free latanoprost cationic emulsion (Catioprost) and a BAK-preserved latanoprost solution in animal models. J. Ocul. Pharmacol. Ther. 2012, 28, 515–523. [Google Scholar] [CrossRef]
- Liang, H.; Baudouin, C.; Daull, P.; Garrigue, J.-S.; Buggage, R.; Brignole-Baudouin, F. In vitro and In vivo evaluation of a preservative-free cationic emulsion of latanoprost in corneal wound healing models. Cornea 2012, 31, 1319–1329. [Google Scholar] [CrossRef] [PubMed]
- Daull, P.; Amrane, M.; Garrigue, J.-S. Novasorb® cationic nanoemulsion and latanoprost: The ideal combination for glaucoma management? Glaucoma 2017, 2, 107. [Google Scholar] [CrossRef]
- Ismail, A.; Nasr, M.; Sammour, O. Nanoemulsion as a feasible and biocompatible carrier for ocular delivery of travoprost: Improved pharmacokinetic/pharmacodynamic properties. Int. J. Pharm. 2020, 583, 119402. [Google Scholar] [CrossRef]
- Tau, J.; Passerini, M.S.; Del Papa, M.; Aguilar, A.; Berra, A. A novel ophthalmic latanoprost 0.005% nanoemulsion: A cytotoxicity study. Graefes Arch. Clin. Exp. Ophthalmol. 2022, 260, 1941–1946. [Google Scholar] [CrossRef]
- Agarwal, R.; Iezhitsa, I.; Agarwal, P.; Abdul Nasir, N.A.; Razali, N.; Alyautdin, R.; Ismail, N.M. Liposomes in topical ophthalmic drug delivery: An update. Drug Deliv. 2016, 23, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, J.V.; Chattopadhyay, S.; Ang, M.; Darwitan, A.; Foo, S.; Zhen, M.; Koo, M.; Wong, T.T.; Venkatraman, S.S. Sustained release of an anti-glaucoma drug: Demonstration of efficacy of a liposomal formulation in the rabbit eye. PLoS ONE 2011, 6, e24513. [Google Scholar] [CrossRef]
- Natarajan, J.V.; Ang, M.; Darwitan, A.; Chattopadhyay, S.; Wong, T.T.; Venkatraman, S.S. Nanomedicine for glaucoma: Liposomes provide sustained release of latanoprost in the eye. Int. J. Nanomed. 2012, 7, 123–131. [Google Scholar] [CrossRef]
- Natarajan, J.V.; Darwitan, A.; Barathi, V.A.; Ang, M.; Htoon, H.M.; Boey, F.; Tam, K.C.; Wong, T.T.; Venkatraman, S.S. Sustained Drug Release in Nanomedicine: A Long-Acting Nanocarrier-Based Formulation for Glaucoma. ACS Nano 2014, 8, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Tiwari, A.; Saraf, S.; Panda, P.K.; Jain, A.; Jain, S.K. Emerging potential of niosomes in ocular delivery. Expert Opin. Drug Deliv. 2021, 18, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Durak, S.; Esmaeili Rad, M.; Alp Yetisgin, A.; Eda Sutova, H.; Kutlu, O.; Cetinel, S.; Zarrabi, A. Niosomal drug delivery systems for ocular disease-recent advances and future prospects. Nanomaterials 2020, 10, 1191. [Google Scholar] [CrossRef]
- Fathalla, D.; Fouad, E.A.; Soliman, G.M. Latanoprost niosomes as a sustained release ocular delivery system for the management of glaucoma. Drug Dev. Ind. Pharm. 2020, 46, 806–813. [Google Scholar] [CrossRef]
- Cheng, Y.-H.; Tsai, T.-H.; Jhan, Y.-Y.; Chiu, A.W.-H.; Tsai, K.-L.; Chien, C.-S.; Chiou, S.-H.; Liu, C.J.-L. Thermosensitive chitosan-based hydrogel as a topical ocular drug delivery system of latanoprost for glaucoma treatment. Carbohydr. Polym. 2016, 144, 390–399. [Google Scholar] [CrossRef]
- Khattab, A.; Marzok, S.; Ibrahim, M. Development of optimized mucoadhesive thermosensitive pluronic based in situ gel for controlled delivery of Latanoprost: Antiglaucoma efficacy and stability approaches. J. Drug Deliv. Sci. Technol. 2019, 53, 101134. [Google Scholar] [CrossRef]
- Moosa, R.M.; Choonara, Y.E.; du Toit, L.C.; Kumar, P.; Carmichael, T.; Tomar, L.K.; Tyagi, C.; Pillay, V. A review of topically administered mini- tablets for drug delivery to the anterior segment of the eye. J. Pharm. Pharmacol. 2014, 66, 490–506. [Google Scholar] [CrossRef]
- Rubenicia, A.M.L.; Cubillan, L.D.P.; Sicam, V.A.D.P.; Macabeo, A.P.G.; Villaflores, O.B.; Castillo, A.L. Intraocular pressure reduction effect of 0.005% latanoprost eye drops in a hyaluronic acid-chitosan nanoparticle drug delivery system in albino rabbits. Transl. Vis. Sci. Technol. 2021, 10, 2. [Google Scholar] [CrossRef]
- Kim, S.N.; Min, C.H.; Kim, Y.K.; Ha, A.; Park, C.G.; Lee, S.H.; Park, K.H.; Choy, Y.B. Iontophoretic ocular delivery of latanoprost-loaded nanoparticles via skin-attached electrodes. Acta Biomater. 2022, 144, 32–41. [Google Scholar] [CrossRef]
- Schnichels, S.; Hurst, J.; de Vries, J.W.; Ullah, S.; Gruszka, A.; Kwak, M.; Löscher, M.; Dammeier, S.; Bartz-Schmidt, K.-U.; Spitzer, M.S.; et al. Self-assembled DNA nanoparticles loaded with travoprost for glaucoma-treatment. Nanomed. Nanotechnol. Biol. Med. 2020, 29, 102260. [Google Scholar] [CrossRef]
- González-Cela-Casamayor, M.A.; López-Cano, J.J.; Bravo-Osuna, I.; Andrés-Guerrero, V.; Vicario-de-la-Torre, M.; Guzmán-Navarro, M.; Benítez-Del-Castillo, J.M.; Herrero-Vanrell, R.; Molina-Martínez, I.T. Novel osmoprotective DOPC-DMPC liposomes loaded with antihypertensive drugs as potential strategy for glaucoma treatment. Pharmaceutics 2022, 14, 1405. [Google Scholar] [CrossRef]
- Ciolino, J.B.; Stefanescu, C.F.; Ross, A.E.; Salvador-Culla, B.; Cortez, P.; Ford, E.M.; Wymbs, K.A.; Sprague, S.L.; Mascoop, D.R.; Rudina, S.S.; et al. In vivo performance of a drug-eluting contact lens to treat glaucoma for a month. Biomaterials 2014, 35, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Gao, Q.; Zhang, L. Extended ocular delivery of latanoprost from niosome-laden contact lenses: In vitro characterization and in vivo studies. J. Drug Deliv. Sci. Technol. 2022, 68, 103044. [Google Scholar] [CrossRef]
- Dang, H.; Dong, C.; Zhang, L. Sustained latanoprost release from PEGylated solid lipid nanoparticle-laden soft contact lens to treat glaucoma. Pharm. Dev. Technol. 2022, 27, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Liu, Y.; Han, S.; Zhao, Q.; Liu, N. Bimatoprost imprinted silicone contact lens to treat glaucoma. AAPS PharmSciTech 2020, 21, 63. [Google Scholar] [CrossRef] [PubMed]
- Franca, J.R.; Foureaux, G.; Fuscaldi, L.L.; Ribeiro, T.G.; Rodrigues, L.B.; Bravo, R.; Castilho, R.O.; Yoshida, M.I.; Cardoso, V.N.; Fernandes, S.O.; et al. Bimatoprost-loaded ocular inserts as sustained release drug delivery systems for glaucoma treatment: In vitro and in vivo evaluation. PLoS ONE 2014, 9, e95461. [Google Scholar] [CrossRef]
- Brandt, J.D.; Sall, K.; DuBiner, H.; Benza, R.; Alster, Y.; Walker, G.; Semba, C.P. Six-month intraocular pressure reduction with a topical bimatoprost ocular insert: Results of a Phase II Randomized Controlled study. Ophthalmology 2016, 123, 1685–1694. [Google Scholar] [CrossRef]
- Shukr, M.H.; Ismail, S.; El-Hossary, G.G.; El-Shazly, A.H. Spanlastics nanovesicular ocular insert as a novel ocular delivery of travoprost: Optimization using Box-Behnken design and in vivo evaluation. J. Liposome Res. 2022, 1–11. [Google Scholar] [CrossRef]

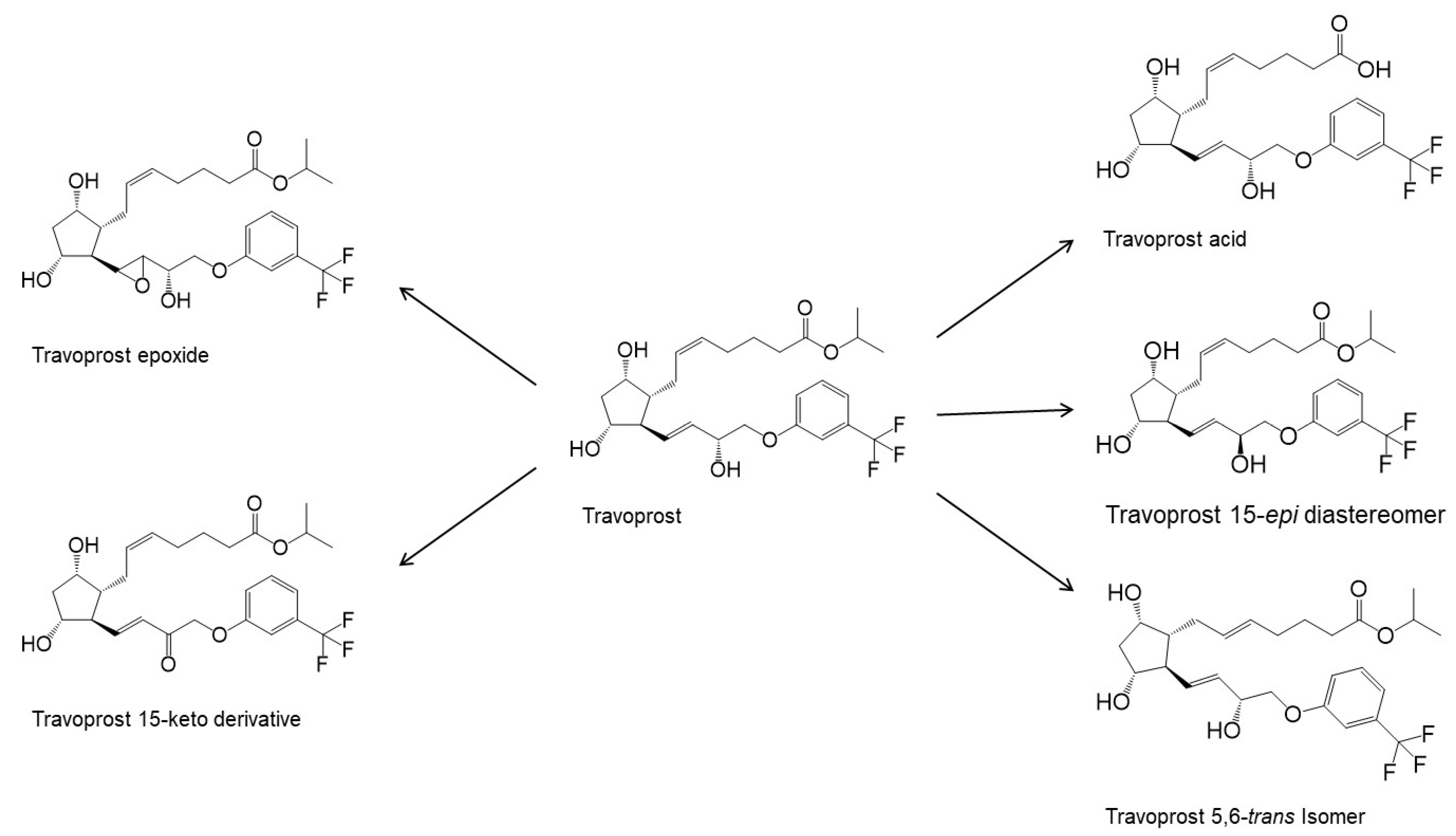
| Prostaglandin Analog | Structure | Molecular Weight | Calculated Values a | |
|---|---|---|---|---|
| LogP(o/w) b | Solubility in Water c | |||
| Prostaglandin F2α (pKa 4.76) | 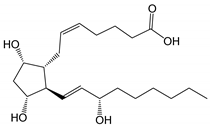 | 354.48 | 2.6 (LogD7.0 0.4) | 30 mg/mL (at pH 7.0) |
| Latanoprost acid |  | 390.51 | 2.8 (LogD7.0 0.6) | 7 mg/mL (at pH 7.0) |
| Bimatoprost (Lumigan®) |  | 415.57 | 2.8 | 40 µg/mL |
| Latanoprost (Xalatan®, Xelpros®) | 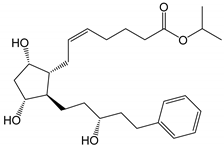 | 432.59 | 4.3 | 6 µg/mL |
| Latanoprostene bunod (Vyzulta®) |  | 507.62 | 4.8 | 1 µg/mL |
| Tafluprost (Taflotan®, Zioptan®) |  | 452.53 | 3.8 | 10 µg/mL |
| Travoprost (Travatan Z®) | 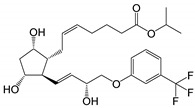 | 500.55 | 4.1 | 4 µg/mL |
| Prostaglandin Analog | Ophthalmic Preparation | Study | Main Observation | Refs. |
|---|---|---|---|---|
| Nanotechnology platforms | ||||
| Latanoprost | Niosome loaded in situ gel | In vitro release and in vivo in rabbits |
| [67] |
| Liposome | In vitro release, in vivo in rabbits and in human primate model |
| [62,63,64] | |
| Hyaluronic acid-chitosan nanoparticles | In vivo in albino rats. |
| [71] | |
| Poly(lactic-co-glycolic acid) nanoparticles | In vitro release and in vivo in rabbits |
| [72] | |
| Travoprost | DNA nanoparticles | Ex vivo in porcine cornea, in vivo in rats and mice |
| [73] |
| Liposome | In vitro release and in vivo in rabbits |
| [74] | |
| Ocular biomaterials | ||||
| Latanoprost | Poly(lactic-co-glycolic acid) film contact lens | In vitro release and in vivo in rabbits |
| [75] |
| Niosome laden contact lens | In vitro release and in vivo in rabbits |
| [76] | |
| PEGylated solid lipid nanoparticle-laden soft contact lens | In vitro release and in vivo in rabbits |
| [77] | |
| Bimatoprost | Molecular imprinted silicone contact lens | In vitro release and in vivo in rabbits |
| [78] |
| Chitosan polymeric inserts | In vitro release and in vivo in Wistar rats |
| [79] | |
| Ocular insert | Phase II clinical study |
| [80] | |
| Travoprost | Spanlastic nano-vesicles ocular insert | In vitro release and in vivo pharmacokinetic in rabbits |
| [81] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jansook, P.; Loftsson, T. Aqueous Prostaglandin Eye Drop Formulations. Pharmaceutics 2022, 14, 2142. https://doi.org/10.3390/pharmaceutics14102142
Jansook P, Loftsson T. Aqueous Prostaglandin Eye Drop Formulations. Pharmaceutics. 2022; 14(10):2142. https://doi.org/10.3390/pharmaceutics14102142
Chicago/Turabian StyleJansook, Phatsawee, and Thorsteinn Loftsson. 2022. "Aqueous Prostaglandin Eye Drop Formulations" Pharmaceutics 14, no. 10: 2142. https://doi.org/10.3390/pharmaceutics14102142
APA StyleJansook, P., & Loftsson, T. (2022). Aqueous Prostaglandin Eye Drop Formulations. Pharmaceutics, 14(10), 2142. https://doi.org/10.3390/pharmaceutics14102142








