Poly(lactic-co-glycolic acid) Nanoparticles Encapsulating the Prenylated Flavonoid, Xanthohumol, Protect Corneal Epithelial Cells from Dry Eye Disease-Associated Oxidative Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Articles, Antibodies and Chemicals
2.2. Cell Culture
2.3. Cell Viability Assays
2.4. Quantitative Immunoblotting
2.5. Generation and Characterization of PLGA NP
2.6. Bioanalytical High-Performance Liquid Chromatography (HPLC) Method for Xanthohumol Detection
2.7. Desiccating Stress/Scopolamine Model for Experimental Dry Eye Disease
2.8. 8-Hydroxy-2′-deoxyguanosine (8-OHdG) Staining
2.9. Data Analysis and Statistics
3. Results
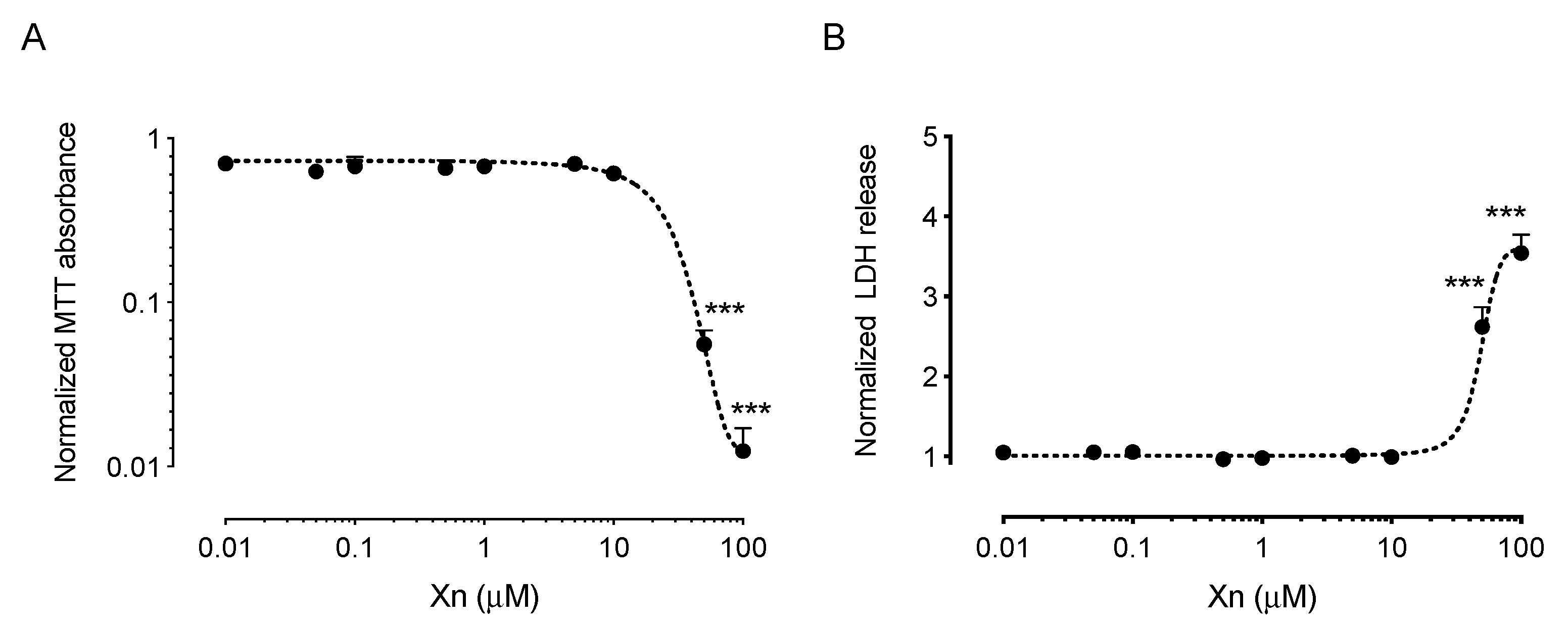
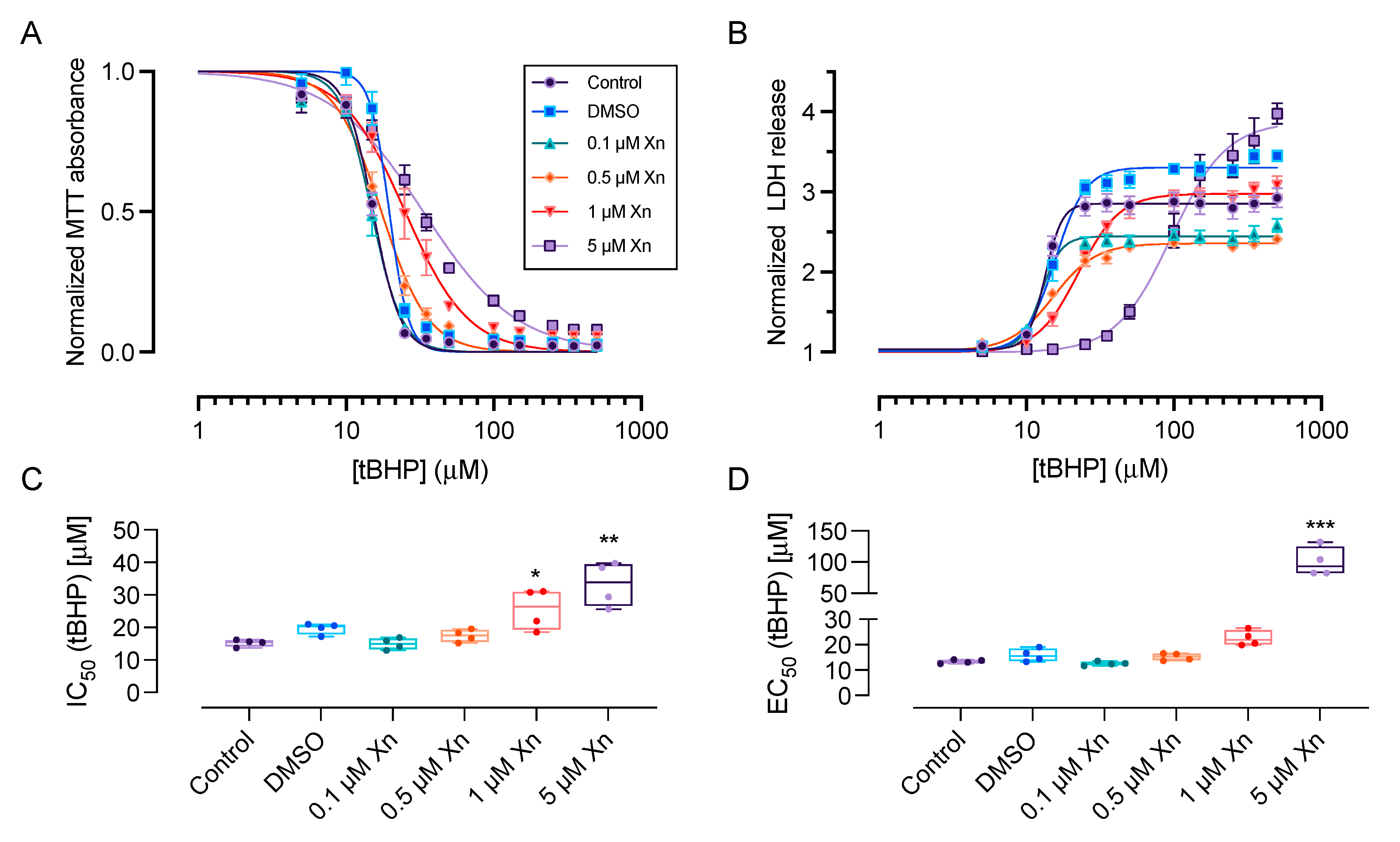
3.1. Xanthohumol Exerts Cytoprotective Effects against Chemically-Induced Oxidative Stress in HCE-T Cells
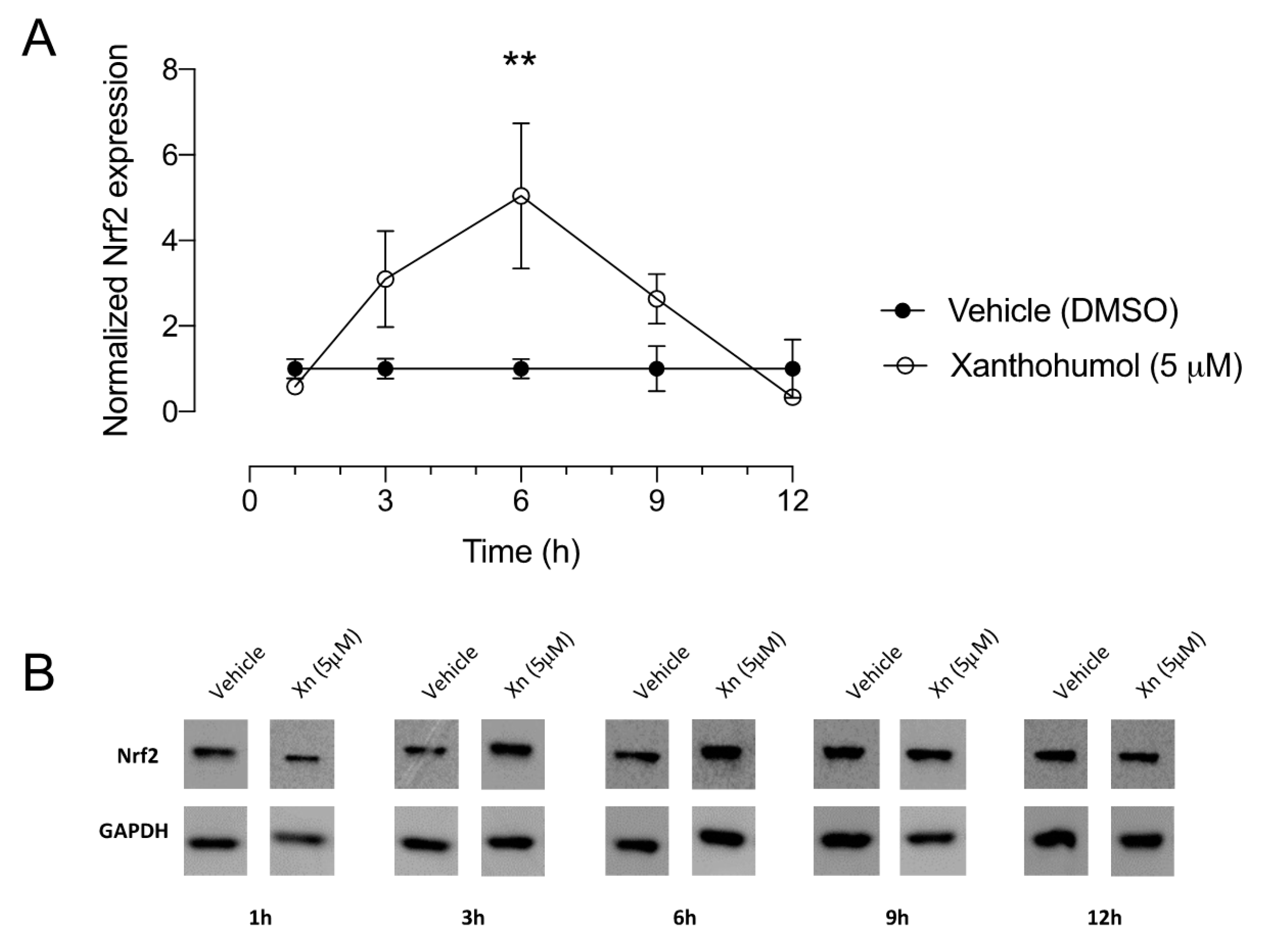
3.2. Xanthohumol Elicits Significant Increase in Nrf2 Protein Levels in Human Corneal Epithelial Cells
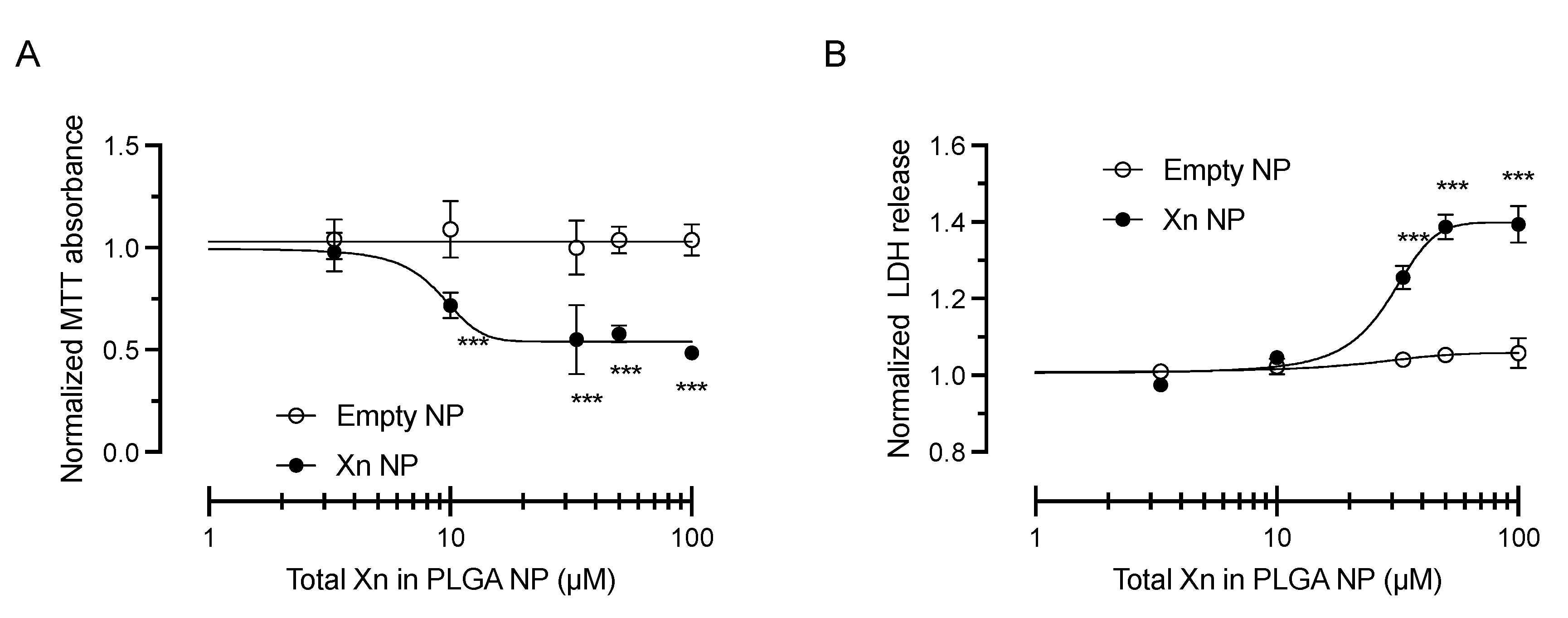
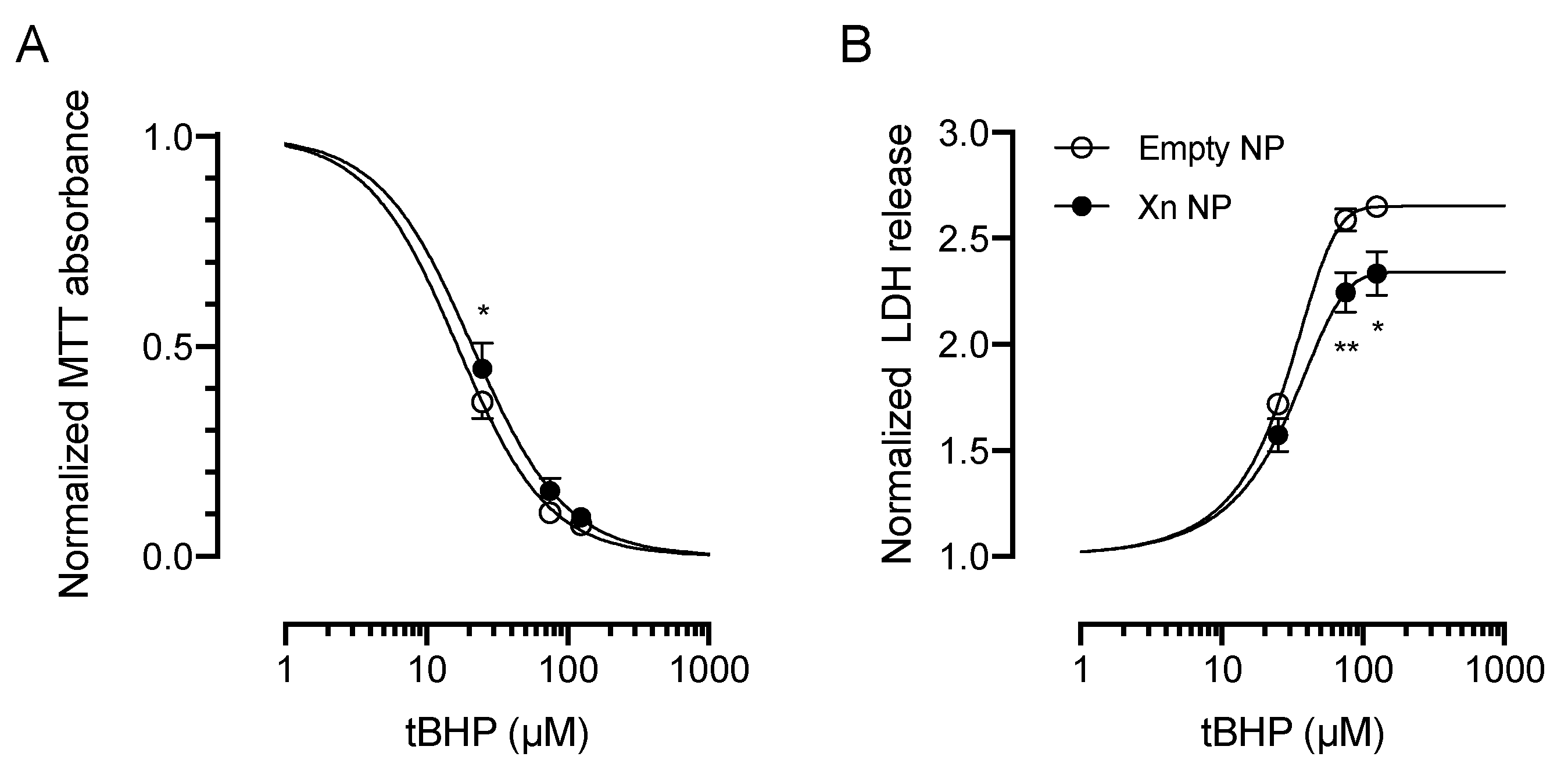
3.3. Xanthohumol-Encapsulating PLGA NP Are Cytoprotective against Oxidative Stress in HCE-T Cells
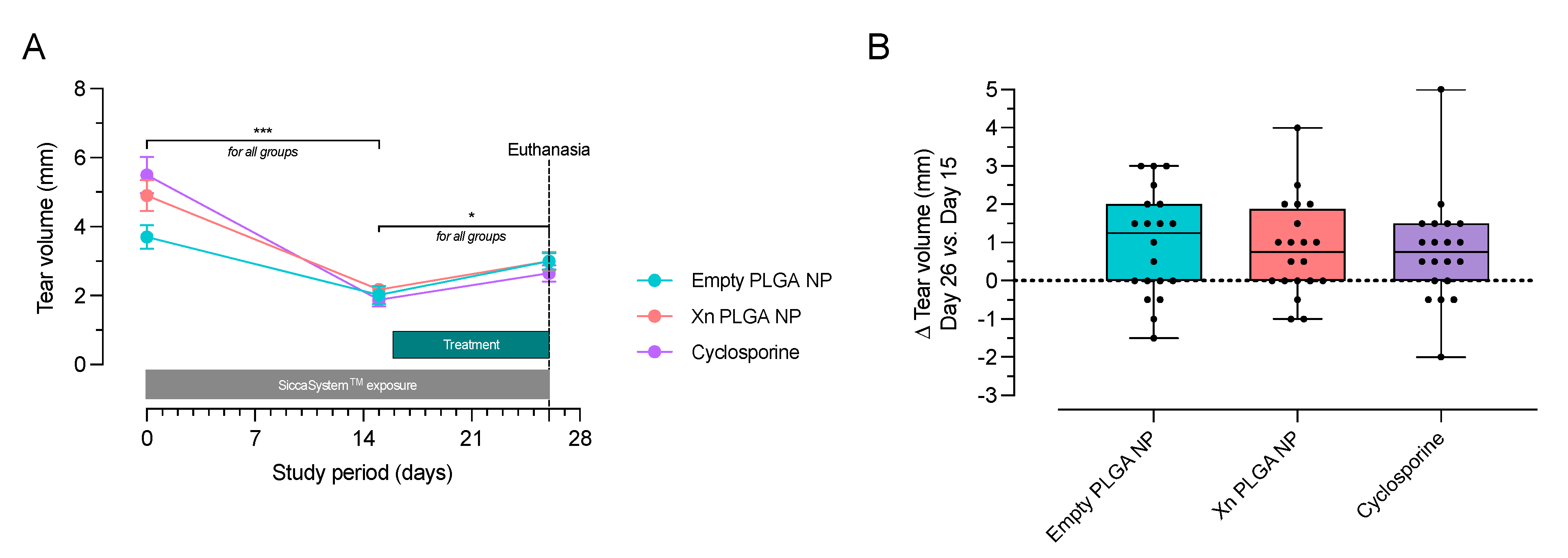
3.4. Xanthohumol-Encapsulating PLGA NP Reverse Ocular Surface Damage in the Desiccating Stress/Scopolamine Model for Dry Eye Disease
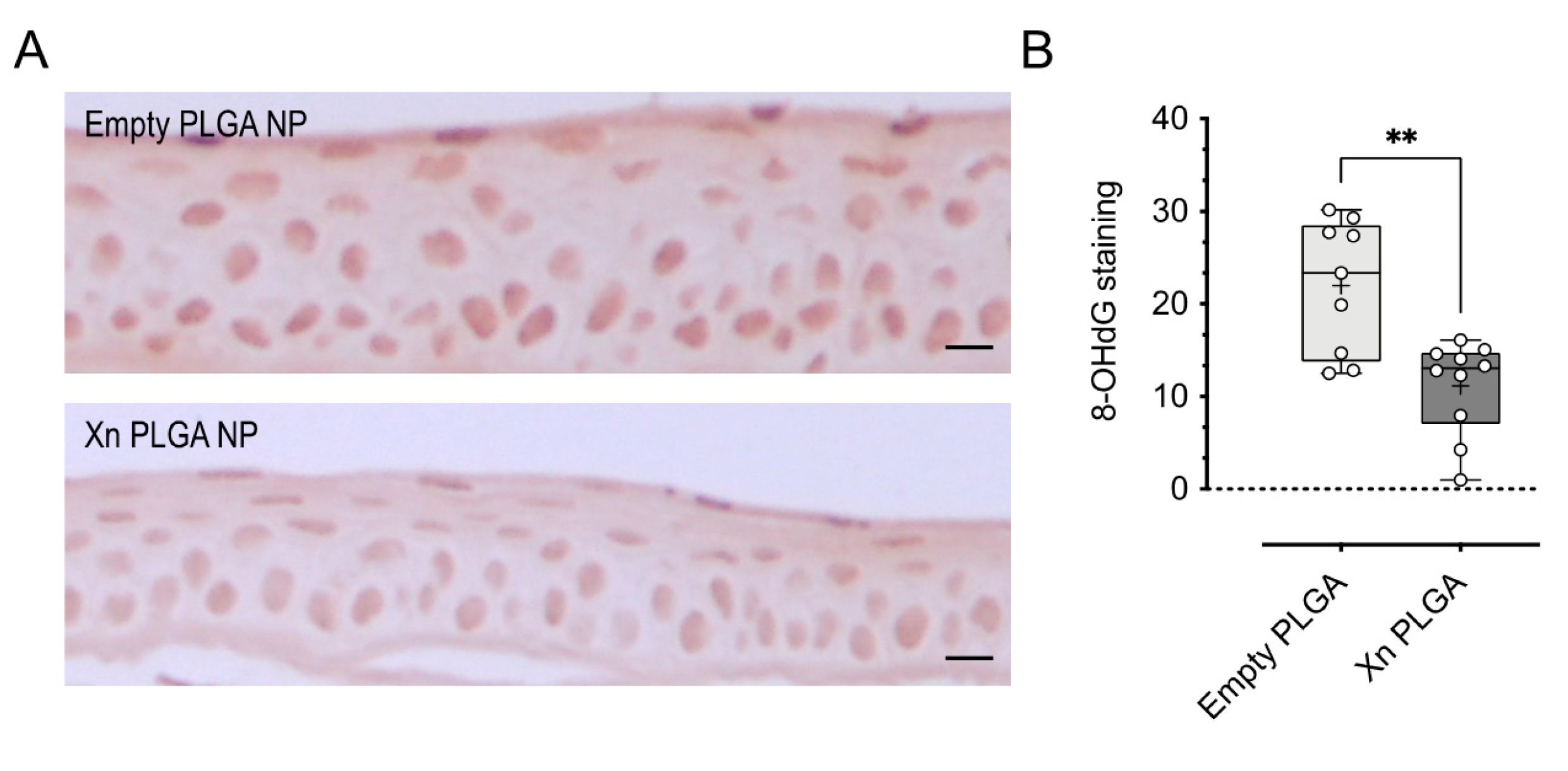
3.5. Topically-Delivered Xanthohumol-Encapsulating PLGA NP Reduce Oxidative DNA Damage in Corneal Epithelial Cells In Vivo after Induction of Dry Eye Disease by Desiccating Stress/Scopolamine
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 8-OHdG | 8-hydroxy-2’-deoxyguanosine |
| ANOVA | One-Way Analysis of Variance |
| GAPHDH | glyceraldehyde 3-phosphate dehydrogenase |
| Keap1 | Kelch-like ECH-associated protein 1 |
| LDH | lactate dehydrogenase |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| Mn-TM-2-PyP | manganese(III) tetrakis(1-methyl-4- pyridyl) porphyrin |
| NF-kB | nuclear factor kappa B |
| NP | nanoparticle |
| Nrf2 | nuclear factor erythroid 2-related factor 2 |
| P-gp | P-glycoprotein 1 |
| PBS | phosphate-buffered saline |
| PBS-T | phosphate-buffered saline supplemented with 0.2% v/v Tween-20 |
| PLGA | poly(lactic-co-glycolic acid) |
| ROS | Reactive oxygen species |
| tBHP | tert-butyl hydroperoxide |
| TEM | transmission electron microscopy |
| Xn | xanthohumol |
References
- Donnenfeld, E.D.; Karpecki, P.M.; Majmudar, P.A.; Nichols, K.K.; Raychaudhuri, A.; Roy, M.; Semba, C.P. Safety of Lifitegrast Ophthalmic Solution 5.0% in Patients with Dry Eye Disease: A 1-Year, Multicenter, Randomized, Placebo-Controlled Study. Cornea 2016, 35, 741–748. [Google Scholar] [CrossRef]
- Jones, L.; Downie, L.E.; Korb, D.; Benitez-Del-Castillo, J.M.; Dana, R.; Deng, S.X.; Dong, P.N.; Geerling, G.; Hida, R.Y.; Liu, Y.; et al. TFOS DEWS II Management and Therapy Report. Ocul. Surf. 2017, 15, 575–628. [Google Scholar] [CrossRef] [PubMed]
- de Paiva, C.S.; Pflugfelder, S.C.; Ng, S.M.; Akpek, E.K. Topical cyclosporine A therapy for dry eye syndrome. Cochrane Database Syst. Rev. 2019, 9, CD010051. [Google Scholar] [CrossRef] [PubMed]
- Seen, S.; Tong, L. Dry eye disease and oxidative stress. Acta Ophthalmol. 2018, 96, e412–e420. [Google Scholar] [CrossRef]
- Macri, A.; Scanarotti, C.; Bassi, A.M.; Giuffrida, S.; Sangalli, G.; Traverso, C.E.; Iester, M. Evaluation of oxidative stress levels in the conjunctival epithelium of patients with or without dry eye, and dry eye patients treated with preservative-free hyaluronic acid 0.15 % and vitamin B12 eye drops. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.; Lian, C.; Ying, L.; Kim, G.E.; You, I.C.; Park, S.H.; Yoon, K.C. Expression of Lipid Peroxidation Markers in the Tear Film and Ocular Surface of Patients with Non-Sjogren Syndrome: Potential Biomarkers for Dry Eye Disease. Curr. Eye Res. 2016, 41, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Deng, R.; Hua, X.; Li, J.; Chi, W.; Zhang, Z.; Lu, F.; Zhang, L.; Pflugfelder, S.C.; Li, D.Q. Oxidative stress markers induced by hyperosmolarity in primary human corneal epithelial cells. PLoS ONE 2015, 10, e0126561. [Google Scholar] [CrossRef]
- Ziniauskaite, A.; Ragauskas, S.; Ghosh, A.K.; Thapa, R.; Roessler, A.E.; Koulen, P.; Kalesnykas, G.; Hakkarainen, J.J.; Kaja, S. Manganese(III) tetrakis(1-methyl-4-pyridyl) porphyrin, a superoxide dismutase mimetic, reduces disease severity in in vitro and in vivo models for dry-eye disease. Ocul. Surf. 2019, 17, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Uchino, Y.; Kawakita, T.; Ishii, T.; Ishii, N.; Tsubota, K. A new mouse model of dry eye disease: Oxidative stress affects functional decline in the lacrimal gland. Cornea 2012, 31 (Suppl. 1), S63–S67. [Google Scholar] [CrossRef]
- Uchino, Y.; Kawakita, T.; Miyazawa, M.; Ishii, T.; Onouchi, H.; Yasuda, K.; Ogawa, Y.; Shimmura, S.; Ishii, N.; Tsubota, K. Oxidative stress induced inflammation initiates functional decline of tear production. PLoS ONE 2012, 7, e45805. [Google Scholar] [CrossRef]
- Wei, Y.; Troger, A.; Spahiu, V.; Perekhvatova, N.; Skulachev, M.; Petrov, A.; Chernyak, B.; Asbell, P. The Role of SKQ1 (Visomitin) in Inflammation and Wound Healing of the Ocular Surface. Ophthalmol. Ther. 2019, 8, 63–73. [Google Scholar] [CrossRef]
- Petrov, A.; Perekhvatova, N.; Skulachev, M.; Stein, L.; Ousler, G. SkQ1 Ophthalmic Solution for Dry Eye Treatment: Results of a Phase 2 Safety and Efficacy Clinical Study in the Environment and During Challenge in the Controlled Adverse Environment Model. Adv. Ther. 2016, 33, 96–115. [Google Scholar] [CrossRef] [PubMed]
- Aragona, P.; Giannaccare, G.; Mencucci, R.; Rubino, P.; Cantera, E.; Rolando, M. Modern approach to the treatment of dry eye, a complex multifactorial disease: A PICASSO board review. Br. J. Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Wang, G.; Qiu, L.; Dong, J.; Yin, H.; Qian, Z.; Yang, M.; Miao, J. Pharmacological profile of xanthohumol, a prenylated flavonoid from hops (Humulus lupulus). Molecules 2015, 20, 754–779. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Zhang, B.; Ge, C.; Peng, S.; Fang, J. Xanthohumol, a polyphenol chalcone present in hops, activating Nrf2 enzymes to confer protection against oxidative damage in PC12 cells. J. Agric. Food Chem. 2015, 63, 1521–1531. [Google Scholar] [CrossRef]
- Araki-Sasaki, K.; Ohashi, Y.; Sasabe, T.; Hayashi, K.; Watanabe, H.; Tano, Y.; Handa, H. An SV40-immortalized human corneal epithelial cell line and its characterization. Investig. Ophthalmol. Vis. Sci 1995, 36, 614–621. [Google Scholar] [PubMed]
- Ghosh, A.K.; Rao, V.R.; Wisniewski, V.J.; Zigrossi, A.D.; Floss, J.; Koulen, P.; Stubbs, E.B., Jr.; Kaja, S. Differential Activation of Glioprotective Intracellular Signaling Pathways in Primary Optic Nerve Head Astrocytes after Treatment with Different Classes of Antioxidants. Antioxidants 2020, 9, 324. [Google Scholar] [CrossRef] [PubMed]
- Kaja, S.; Payne, A.J.; Naumchuk, Y.; Koulen, P. Quantification of Lactate Dehydrogenase for Cell Viability Testing Using Cell Lines and Primary Cultured Astrocytes. Curr. Protoc. Toxicol. 2017, 72, 2–26. [Google Scholar] [CrossRef] [PubMed]
- Kaja, S.; Payne, A.J.; Naumchuk, Y.; Levy, D.; Zaidi, D.H.; Altman, A.M.; Nawazish, S.; Ghuman, J.K.; Gerdes, B.C.; Moore, M.A.; et al. Plate reader-based cell viability assays for glioprotection using primary rat optic nerve head astrocytes. Exp. Eye Res. 2015, 138, 159–166. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Langert, K.A.; Goshu, B.; Stubbs, E.B., Jr. Attenuation of experimental autoimmune neuritis with locally administered lovastatin-encapsulating poly(lactic-co-glycolic) acid nanoparticles. J. Neurochem. 2017, 140, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Ziniauskaite, A.; Ragauskas, S.; Hakkarainen, J.J.; Rich, C.C.; Baumgartner, R.; Kalesnykas, G.; Albers, D.S.; Kaja, S. Efficacy of Trabodenoson in a Mouse Keratoconjunctivitis Sicca (KCS) Model for Dry-Eye Syndrome. Investig. Ophthalmol. Vis. Sci. 2018, 59, 3088–3093. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C.; Messmer, E.M.; Aragona, P.; Geerling, G.; Akova, Y.A.; Benitez-del-Castillo, J.; Boboridis, K.G.; Merayo-Lloves, J.; Rolando, M.; Labetoulle, M. Revisiting the vicious circle of dry eye disease: A focus on the pathophysiology of meibomian gland dysfunction. Br. J. Ophthalmol. 2016, 100, 300–306. [Google Scholar] [CrossRef]
- Lan, W.; Petznick, A.; Heryati, S.; Rifada, M.; Tong, L. Nuclear Factor-kappaB: Central regulator in ocular surface inflammation and diseases. Ocul. Surf. 2012, 10, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Hattori, T.; Park, E.Y.; Stevenson, W.; Chauhan, S.K.; Dana, R. Expression of toll-like receptor 4 contributes to corneal inflammation in experimental dry eye disease. Investig. Ophthalmol. Vis. Sci 2012, 53, 5632–5640. [Google Scholar] [CrossRef] [PubMed]
- Becker, U.; Ehrhardt, C.; Schneider, M.; Muys, L.; Gross, D.; Eschmann, K.; Schaefer, U.F.; Lehr, C.M. A comparative evaluation of corneal epithelial cell cultures for assessing ocular permeability. Altern Lab. Anim. 2008, 36, 33–44. [Google Scholar] [CrossRef]
- Reichl, S. Cell culture models of the human cornea—A comparative evaluation of their usefulness to determine ocular drug absorption in-vitro. J. Pharm. Pharmacol. 2008, 60, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Kawasaki, S.; Young, R.D.; Fukuoka, H.; Tanioka, H.; Nakatsukasa, M.; Quantock, A.J.; Kinoshita, S. Genomic aberrations and cellular heterogeneity in SV40-immortalized human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2009, 50, 604–613. [Google Scholar] [CrossRef][Green Version]
- Dursun, D.; Wang, M.; Monroy, D.; Li, D.Q.; Lokeshwar, B.L.; Stern, M.E.; Pflugfelder, S.C. A mouse model of keratoconjunctivitis sicca. Investig. Ophthalmol. Vis. Sci. 2002, 43, 632–638. [Google Scholar] [PubMed]
- Astete, C.E.; Sabliov, C.M. Synthesis and characterization of PLGA nanoparticles. J. Biomater. Sci. Polym. Ed. 2006, 17, 247–289. [Google Scholar] [CrossRef] [PubMed]
- Zigoneanu, I.G.; Astete, C.E.; Sabliov, C.M. Nanoparticles with entrapped alpha-tocopherol: Synthesis, characterization, and controlled release. Nanotechnology 2008, 19, 105606. [Google Scholar] [CrossRef] [PubMed]
- Alhowyan, A.A.; Altamimi, M.A.; Kalam, M.A.; Khan, A.A.; Badran, M.; Binkhathlan, Z.; Alkholief, M.; Alshamsan, A. Antifungal efficacy of Itraconazole loaded PLGA-nanoparticles stabilized by vitamin-E TPGS: In vitro and ex vivo studies. J. Microbiol. Methods 2019, 161, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Kong, Y.Y.; Sun, J.H.; Huo, S.J.; Zhou, M.; Gui, Y.L.; Mu, X.; Chen, H.; Yu, S.Q.; Xu, Q. Co-delivery nanoparticles with characteristics of intracellular precision release drugs for overcoming multidrug resistance. Int. J. Nanomed. 2017, 12, 2081–2108. [Google Scholar] [CrossRef] [PubMed]
- Swetledge, S.; Jung, J.P.; Carter, R.; Sabliov, C. Distribution of polymeric nanoparticles in the eye: Implications in ocular disease therapy. J. Nanobiotechnology 2021, 19, 10. [Google Scholar] [CrossRef]
- Amann, L.C.; Gandal, M.J.; Lin, R.; Liang, Y.; Siegel, S.J. In vitro-in vivo correlations of scalable PLGA-risperidone implants for the treatment of schizophrenia. Pharm. Res. 2010, 27, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, B.; Kompella, U.B. Ocular Drug Delivery. Handb. Exp. Pharmacol. 2017, 242, 57–93. [Google Scholar] [CrossRef] [PubMed]
- Wagh, V.D.; Apar, D.U. Cyclosporine A Loaded PLGA Nanoparticles for Dry Eye Disease: In Vitro Characterization Studies. J. Nanotechnol. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Ames, P.; Galor, A. Cyclosporine ophthalmic emulsions for the treatment of dry eye: A review of the clinical evidence. Clin. Investig. 2015, 5, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; An, C.; Gao, Y.; Leak, R.K.; Chen, J.; Zhang, F. Emerging roles of Nrf2 and phase II antioxidant enzymes in neuroprotection. Prog. Neurobiol. 2013, 100, 30–47. [Google Scholar] [CrossRef]


| Parameter | Empty PLGA NP | Xn PLGA NP |
|---|---|---|
| Size (nm) | 201.9 ± 0.1 | 191.0 ± 0.8 |
| Polydispersity Index (PDI) | 0.045 ± 0.009 | 0.029 ± 0.007 |
| Zeta (ζ) potential (mV) | −21.6 ± 0.3 | −24.8 ± 0.2 |
| Encapsulation efficiency | N/A | 13.1 ± 0.06% |
| Parameter | Empty PLGA NP | Xn PLGA NP | Statistics |
|---|---|---|---|
| Thickness, corneal epithelium (µm) * | 35.5 ± 3.3 | 30.8 ± 2.2 | n = 10, p = 0.24 |
| Thickness, corneal stroma (µm) * | 178.9 ± 12.9 | 159.4 ± 7.8 | n = 10, p = 0.21 |
| Number of epithelial cell layers ** | 5 (4; 5.5) | 4 (4; 5) | n = 10, p = 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghosh, A.K.; Thapa, R.; Hariani, H.N.; Volyanyuk, M.; Ogle, S.D.; Orloff, K.A.; Ankireddy, S.; Lai, K.; Žiniauskaitė, A.; Stubbs, E.B., Jr.; et al. Poly(lactic-co-glycolic acid) Nanoparticles Encapsulating the Prenylated Flavonoid, Xanthohumol, Protect Corneal Epithelial Cells from Dry Eye Disease-Associated Oxidative Stress. Pharmaceutics 2021, 13, 1362. https://doi.org/10.3390/pharmaceutics13091362
Ghosh AK, Thapa R, Hariani HN, Volyanyuk M, Ogle SD, Orloff KA, Ankireddy S, Lai K, Žiniauskaitė A, Stubbs EB Jr., et al. Poly(lactic-co-glycolic acid) Nanoparticles Encapsulating the Prenylated Flavonoid, Xanthohumol, Protect Corneal Epithelial Cells from Dry Eye Disease-Associated Oxidative Stress. Pharmaceutics. 2021; 13(9):1362. https://doi.org/10.3390/pharmaceutics13091362
Chicago/Turabian StyleGhosh, Anita Kirti, Rubina Thapa, Harsh Nilesh Hariani, Michael Volyanyuk, Sean David Ogle, Karoline Anne Orloff, Samatha Ankireddy, Karen Lai, Agnė Žiniauskaitė, Evan Benjamin Stubbs, Jr., and et al. 2021. "Poly(lactic-co-glycolic acid) Nanoparticles Encapsulating the Prenylated Flavonoid, Xanthohumol, Protect Corneal Epithelial Cells from Dry Eye Disease-Associated Oxidative Stress" Pharmaceutics 13, no. 9: 1362. https://doi.org/10.3390/pharmaceutics13091362
APA StyleGhosh, A. K., Thapa, R., Hariani, H. N., Volyanyuk, M., Ogle, S. D., Orloff, K. A., Ankireddy, S., Lai, K., Žiniauskaitė, A., Stubbs, E. B., Jr., Kalesnykas, G., Hakkarainen, J. J., Langert, K. A., & Kaja, S. (2021). Poly(lactic-co-glycolic acid) Nanoparticles Encapsulating the Prenylated Flavonoid, Xanthohumol, Protect Corneal Epithelial Cells from Dry Eye Disease-Associated Oxidative Stress. Pharmaceutics, 13(9), 1362. https://doi.org/10.3390/pharmaceutics13091362







