Purinergic P2Y2 and P2X4 Receptors Are Involved in the Epithelial-Mesenchymal Transition and Metastatic Potential of Gastric Cancer Derived Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Extracellular ATP Measurements
2.3. Real-Time qPCR Experiments to Determine Purinergic Receptors and EMT-Related Gene Expression
2.4. P2Y2R and P2X4R Molecular Interventions
2.5. Cell Viability Experiments
2.6. Transepithelial Electrical Resistance (TEER) Measurements
2.7. Immunofluorescence
2.8. Microarray Studies
2.9. Statistical Analysis
3. Results
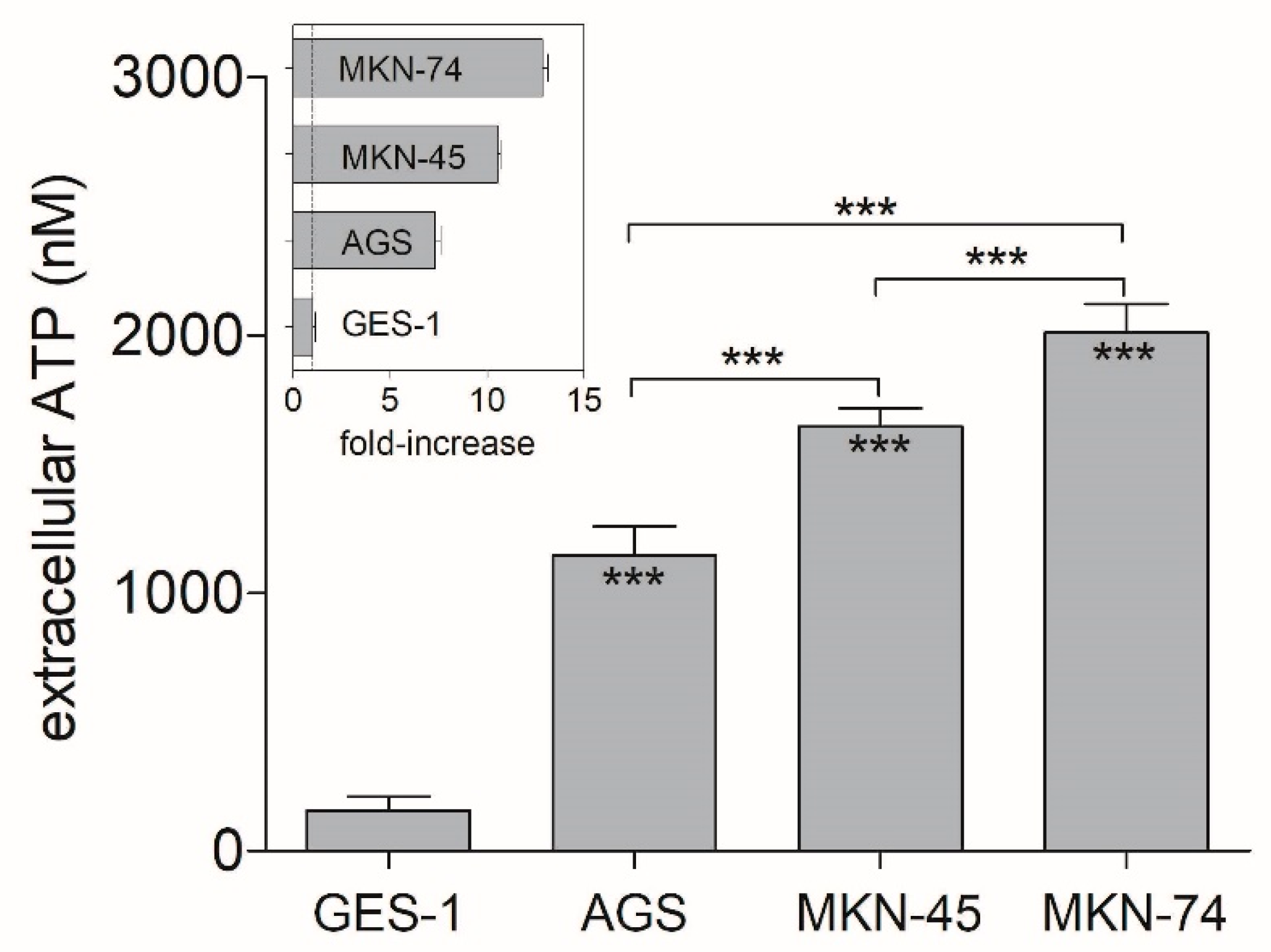
3.1. Extracellular ATP Concentrations on Non-Tumoral and GC Cell Lines
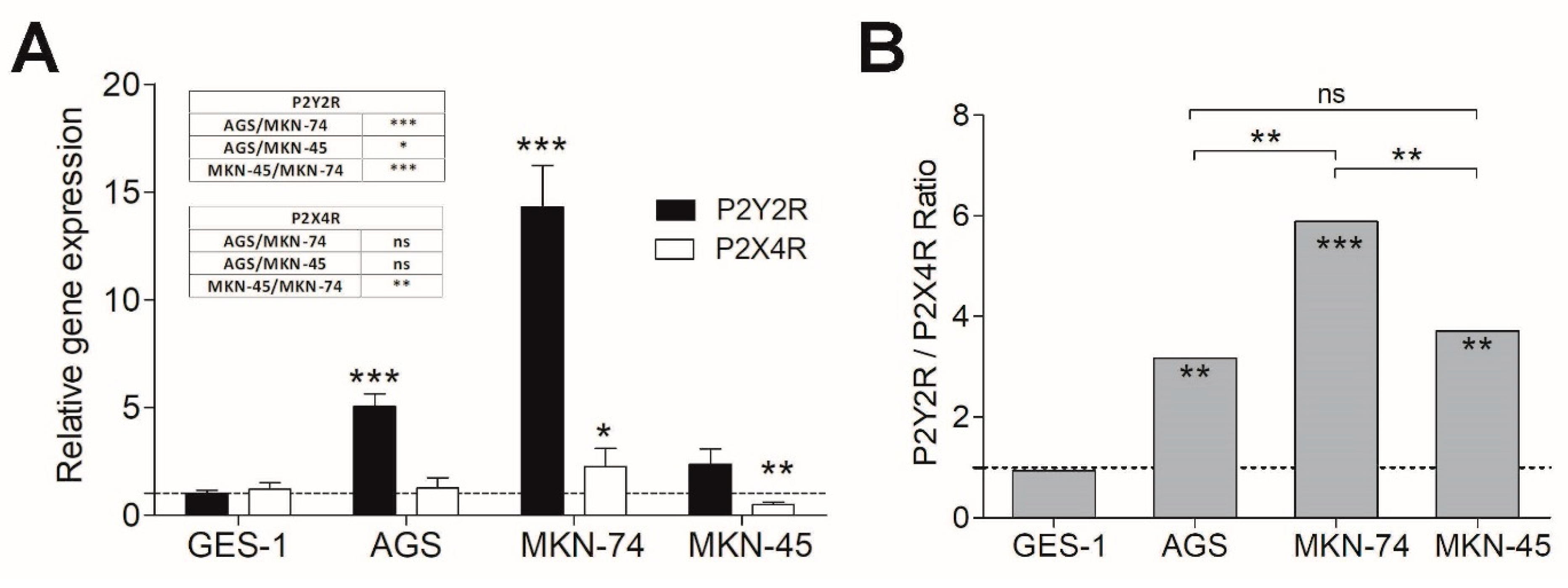
3.2. P2Y2R and P2X4R Expression Profile in Non-Tumoral and GC-Derived Cell Lines
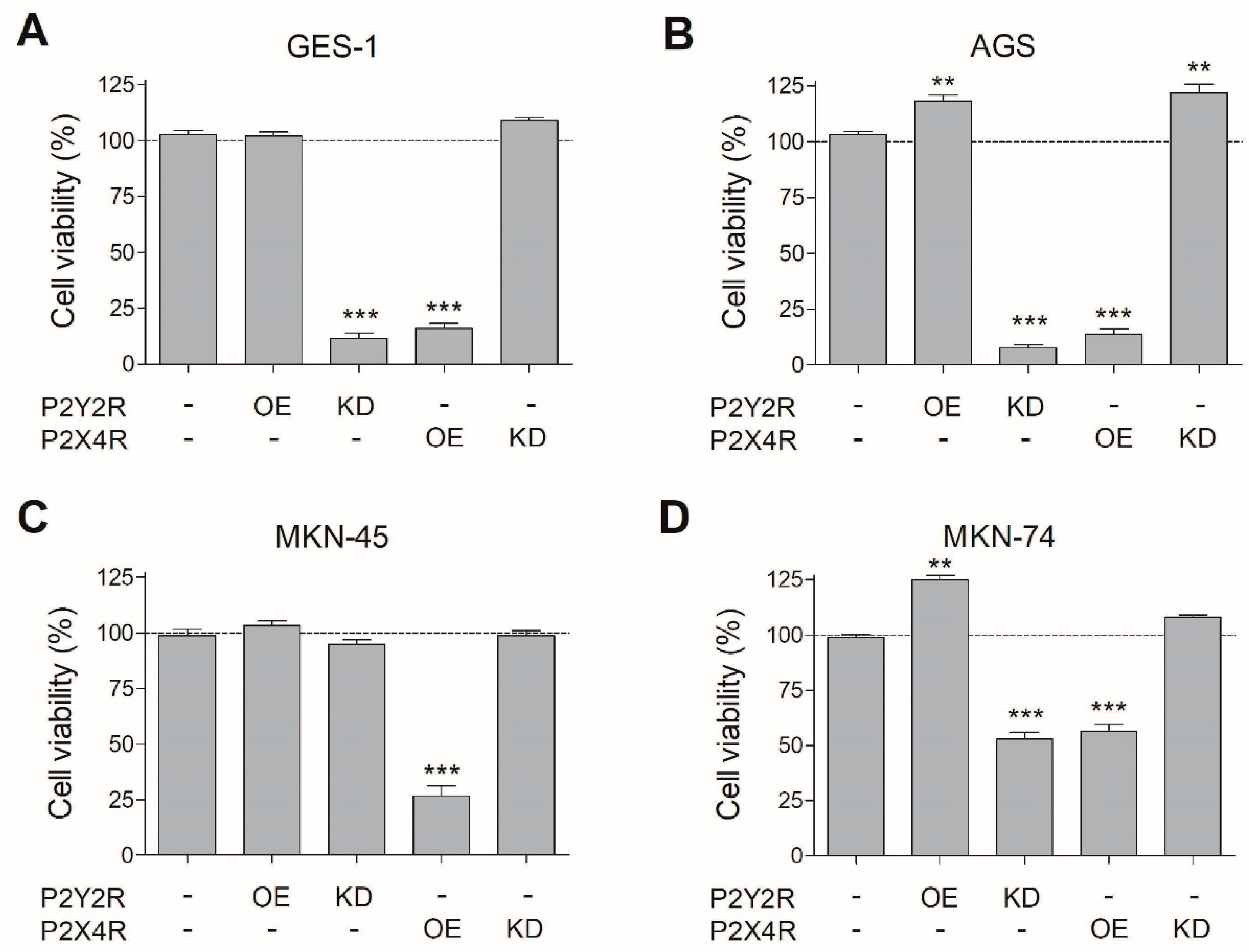
3.3. Changes in P2Y2R and P2X4R Expression Modify Cell Viability in Non-Tumoral and GC Cell Lines
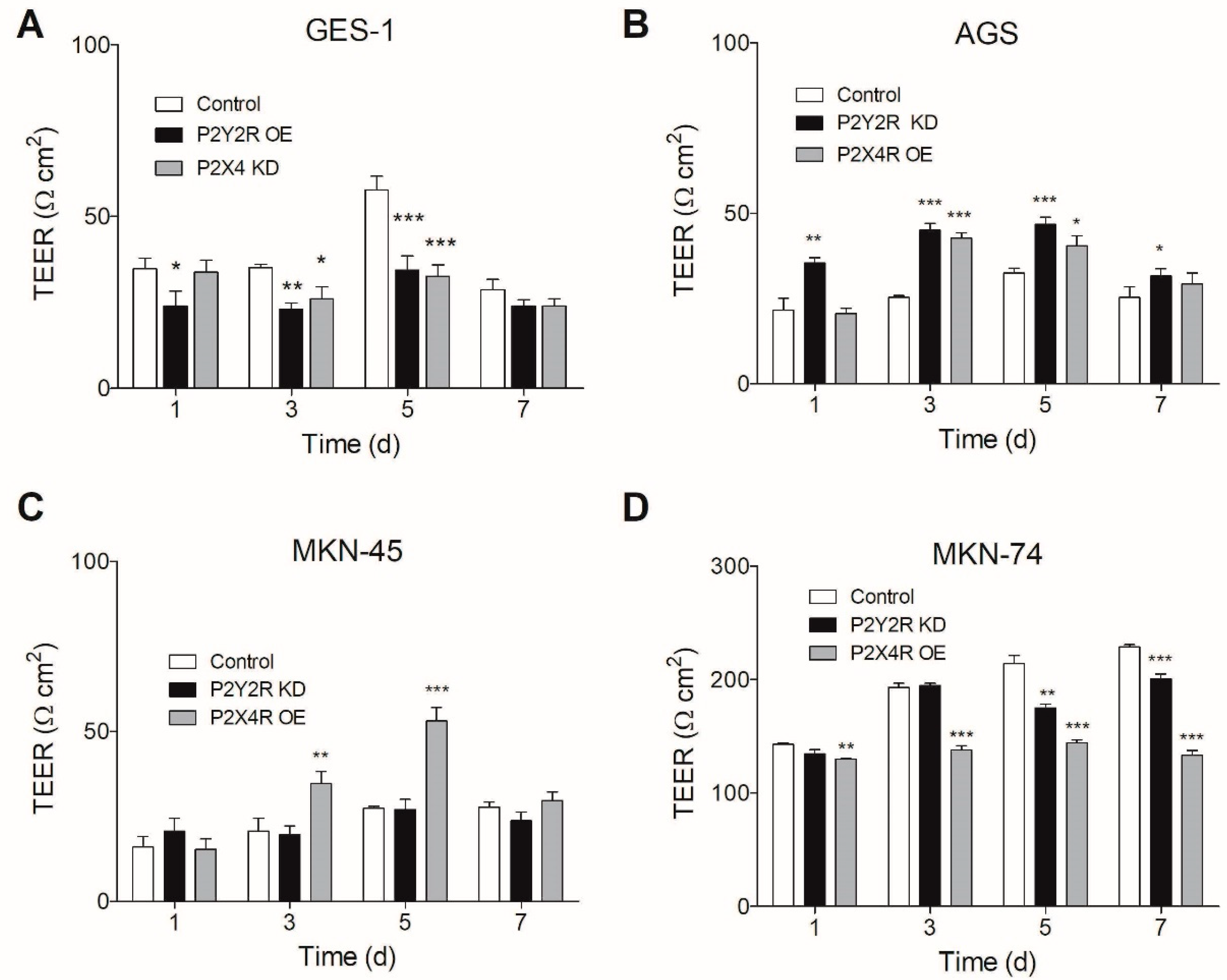
3.4. Changes in P2Y2R and P2X4R Expression Modify TEER in Non-Tumoral and GC Cell Lines
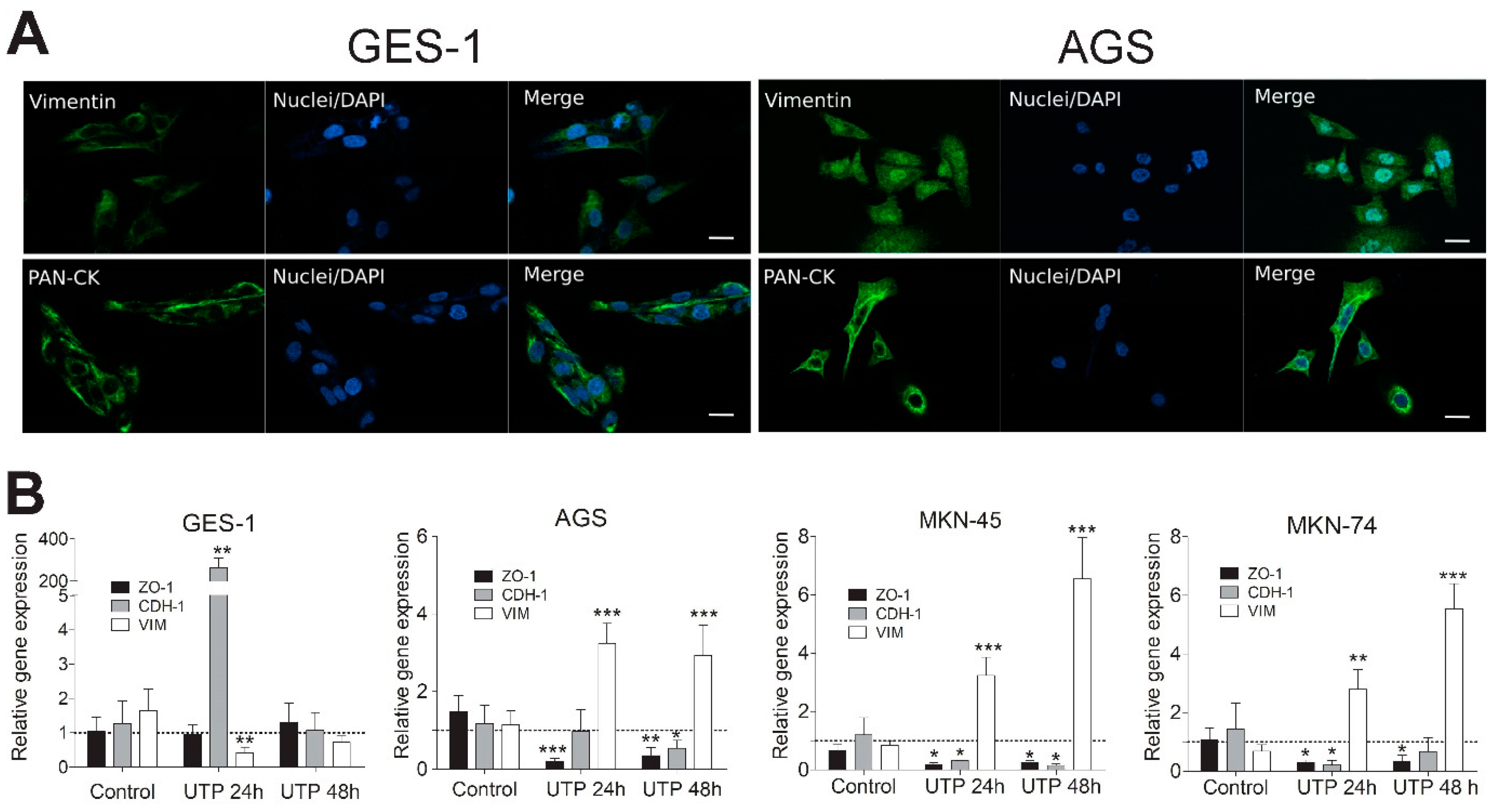
3.5. Purinergic Stimulation Changes the Expression of Epithelial and Mesenchymal Markers in Non-Tumoral and GC-Derived Cell Lines
3.6. Microarray Analysis of AGS Cells after UTP Stimulation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Sifri, R.; Gangadharappa, S.; Acheson, L.S. Identifying and testing for hereditary susceptibility to common cancers. CA A Cancer J. Clin. 2004, 54, 309–326. [Google Scholar] [CrossRef]
- Lewandowska, A.M.; Rudzki, M.; Rudzki, S.; Lewandowski, T.; Laskowska, B. Environmental risk factors for cancer—review paper. Ann. Agric. Environ. Med. 2019, 26, 1–7. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2014, 136, E359–E386. [Google Scholar] [CrossRef]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends—An Update. Cancer Epidemiol. Biomark. Prev. 2015, 25, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Caglevic, C.; Silva, S.; Mahave, M.; Rolfo, C.; Gallardo, J. The current situation for gastric cancer in Chile. Ecancermedicalscience 2016, 10, 707. [Google Scholar] [CrossRef][Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Padma, V.V. An overview of targeted cancer therapy. Biomedicine 2015, 5, 19. [Google Scholar] [CrossRef] [PubMed]
- Di Virgilio, F.; Sarti, A.C.; Falzoni, S.; De Marchi, E.; Adinolfi, E. Extracellular ATP and P2 purinergic signalling in the tumour microenvironment. Nat. Rev. Cancer 2018, 18, 601–618. [Google Scholar] [CrossRef]
- Coddou, C.; Yan, Z.; Obsil, T.; Huidobro-Toro, J.P.; Stojilkovic, S.S. Activation and Regulation of Purinergic P2X Receptor Channels. Pharmacol. Rev. 2011, 63, 641–683. [Google Scholar] [CrossRef]
- White, N. Burnstock G P2 receptors and cancer. Trends Pharmacol. Sci. 2006, 27, 211–217. [Google Scholar] [CrossRef]
- Aquea, G.; Bresky, G.; Lancellotti, D.; Madariaga, J.A.; Zaffiri, V.; Urzua, U.; Haberle, S.; Bernal, G. Increased Expression of P2RY2, CD248 and EphB1 in Gastric Cancers from Chilean Patients. Asian Pac. J. Cancer Prev. 2014, 15, 1931–1936. [Google Scholar] [CrossRef] [PubMed]
- Bellefeuille, S.D.; Molle, C.M.; Gendron, F.-P. Reviewing the role of P2Y receptors in specific gastrointestinal cancers. Purinergic Signal. 2019, 15, 451–463. [Google Scholar] [CrossRef]
- Shi, L.; Yang, L.; Wu, Z.; Xu, W.; Song, J.; Guan, W. Adenosine signaling: Next checkpoint for gastric cancer immunotherapy? Int. Immunopharmacol. 2018, 63, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Hevia, M.J.; Castro, P.; Pinto, K.; Reyna-Jeldes, M.; Rodríguez-Tirado, F.; Robles-Planells, C.; Ramírez-Rivera, S.; Madariaga, J.A.; Gutierrez, F.; López, J.; et al. Differential Effects of Purinergic Signaling in Gastric Cancer-Derived Cells Through P2Y and P2X Receptors. Front. Pharmacol. 2019, 10, 612. [Google Scholar] [CrossRef]
- Viebahn, C. Epithelio-mesenchymal transformation during formation of the mesoderm in the mammalian embryo. Acta Anat. 1995, 154, 79–97. [Google Scholar] [CrossRef]
- Brabletz, T.; Kalluri, R.; Nieto, M.A.; Weinberg, R.A. EMT in cancer. Nat. Rev. Cancer 2018, 18, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Ramena, G.; Elble, R.C. The role of cancer stem cells in relapse of solid tumors. Front. Biosci. 2012, 4, 1528–1541. [Google Scholar] [CrossRef]
- Martínez-Ramírez, A.S.; Díaz-Muñoz, M.; Butanda-Ochoa, A.; Vázquez-Cuevas, F.G. Nucleotides and nucleoside signaling in the regulation of the epithelium to mesenchymal transition (EMT). Purinergic Signal. 2016, 13, 1–12. [Google Scholar] [CrossRef]
- Reyna-Jeldes, M.; Díaz-Muñoz, M.; Madariaga, J.A.; Coddou, C.; Vázquez-Cuevas, F.G. Autocrine and paracrine purinergic signaling in the most lethal types of cancer. Purinergic Signal. 2021, 1–26. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Yan, Z.; Liang, Z.; Tomic, M.; Obsil, T.; Stojilkovic, S.S. Molecular determinants of the agonist binding domain of a P2X receptor channel. Mol. Pharmacol. 2005, 67, 1078–1088. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-M.; Kim, I.-H.; Nam, T.-J. Inhibition of AGS human gastric cancer cell invasion and proliferation by Capsosiphon fulvescens glycoprotein. Mol. Med. Rep. 2013, 8, 11–16. [Google Scholar] [CrossRef]
- Bay, A.E.P.; Schreiner, R.; Benedicto, I.; Marzolo, M.P.; Banfelder, J.; Weinstein, A.M.; Rodriguez-Boulan, E.J. The fast-recycling receptor Megalin defines the apical recycling pathway of epithelial cells. Nat. Commun. 2016, 7, 11550. [Google Scholar] [CrossRef]
- Di Paola, R.; Fusco, R.; Gugliandolo, E.; D’Amico, R.; Campolo, M.; Latteri, S.; Carughi, A.; Mandalari, G.; Cuzzocrea, S. The Antioxidant Activity of Pistachios Reduces Cardiac Tissue Injury of Acute Ischemia/Reperfusion (I/R) in Diabetic Streptozotocin (STZ)-Induced Hyperglycaemic Rats. Front. Pharmacol. 2018, 9, 51. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2018, 47, D607–D613. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A Software Environment for Integrated Models of Biomolecular Interaction Networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Kurata, A.; Tachibana, Y.; Takahashi, T.; Horiba, N. Novel AXL-specific inhibitor ameliorates kidney dysfunction through the inhibition of epithelial-to-mesenchymal transition of renal tubular cells. PLoS ONE 2020, 15, e0232055. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Chen, J.; Yang, X.; Zheng, W.; Wang, L.; Fang, M.; Wu, M.; Yao, M.; Yao, D. Ang-2 promotes lung cancer metastasis by increasing epithelial-mesenchymal transition. Oncotarget 2018, 9, 12705–12717. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.-M.; Chang, L.-Y.; Lin, S.-H.; Chou, J.-L.; Hsieh, H.-Y.; Zeng, L.-H.; Chuang, S.-Y.; Wang, H.-W.; Dittner, C.; Lin, C.-Y.; et al. Epigenetic silencing of the NR4A3 tumor suppressor, by aberrant JAK/STAT signaling, predicts prognosis in gastric cancer. Sci. Rep. 2016, 6, 31690. [Google Scholar] [CrossRef] [PubMed]
- Campos-Contreras, A.D.R.; Díaz-Muñoz, M.; Vázquez-Cuevas, F.G. Purinergic Signaling in the Hallmarks of Cancer. Cells 2020, 9, 1612. [Google Scholar] [CrossRef]
- Nienhüser, H.; Schmidt, T. Gastric cancer lymph node resection-the more the merrier? Transl. Gastroenterol. Hepatol. 2018, 3, 1. [Google Scholar] [CrossRef]
- Subhash, V.V.; Yeo, M.S.; Tan, W.L.; Yong, W.P. Strategies and Advancements in Harnessing the Immune System for Gastric Cancer Immunotherapy. J. Immunol. Res. 2015, 2015, 308574. [Google Scholar] [CrossRef]
- Marin, J.J.; Briz, O.; Monte, M.J.; Blazquez, A.G.; Macias, R.I. Genetic variants in genes involved in mechanisms of chemoresistance to anticancer drugs. Curr. Cancer Drug Targets 2012, 12, 402–438. [Google Scholar] [CrossRef]
- Utsugi, T. New Challenges and Inspired Answers for Anticancer Drug Discovery and Development. Jpn. J. Clin. Oncol. 2013, 43, 945–953. [Google Scholar] [CrossRef]
- Di Virgilio, F. Purines, Purinergic Receptors, and Cancer. Cancer Res. 2012, 72, 5441–5447. [Google Scholar] [CrossRef] [PubMed]
- Takai, E.; Tsukimoto, M.; Harada, H.; Kojima, S. Autocrine signaling via release of ATP and activation of P2X7 receptor influences motile activity of human lung cancer cells. Purinergic Signal. 2014, 10, 487–497. [Google Scholar] [CrossRef]
- Yang, H.; Geng, Y.; Wang, P.; Zhou, Y.; Yang, H.; Huo, Y.; Zhang, H.; Li, Y.; He, H.; Tian, X.; et al. Extracellular ATP promotes breast cancer invasion and epithelial-mesenchymal transition via hypoxia-inducible factor 2α signaling. Cancer Sci. 2019, 110, 2456–2470. [Google Scholar] [CrossRef] [PubMed]
- Busuttil, R.A.; Liu, D.S.; Di Costanzo, N.; Schroeder, J.; Mitchell, C.; Boussioutas, A. An orthotopic mouse model of gastric cancer invasion and metastasis. Sci. Rep. 2018, 8, 825. [Google Scholar] [CrossRef]
- Zhang, Y.; Ding, J.; Wang, L. The role of P2X7 receptor in prognosis and metastasis of colorectal cancer. Adv. Med Sci. 2019, 64, 388–394. [Google Scholar] [CrossRef]
- Lili, W.; Yun, L.; Tingran, W.; Xia, W.; Yanlei, S. P2RX7 functions as a putative biomarker of gastric cancer and contributes to worse prognosis. Exp. Biol. Med. 2019, 244, 734–742. [Google Scholar] [CrossRef]
- Katz, S.; Ayala, V.; Santillán, G.; Boland, R. Activation of the PI3K/Akt signaling pathway through P2Y2 receptors by extracellular ATP is involved in osteoblastic cell proliferation. Arch. Biochem. Biophys. 2011, 513, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Soltoff, S.P.; Avraham, H.; Avraham, S.; Cantley, L. Activation of P2Y2 Receptors by UTP and ATP Stimulates Mitogen-activated Kinase Activity through a Pathway That Involves Related Adhesion Focal Tyrosine Kinase and Protein Kinase C. J. Biol. Chem. 1998, 273, 2653–2660. [Google Scholar] [CrossRef] [PubMed]
- Soltoff, S.P. Related Adhesion Focal Tyrosine Kinase and the Epidermal Growth Factor Receptor Mediate the Stimulation of Mitogen-activated Protein Kinase by the G-protein-coupled P2Y2 Receptor. Phorbol ester or [Ca2+]i elevation can substitute for receptor activation. J. Biol. Chem. 1998, 273, 23110–23117. [Google Scholar] [CrossRef] [PubMed]
- Coddou, C.; Morales, B.; Gonzalez, J.; Grauso, M.; Gordillo, F.; Bull, P.; Rassendren, F.; Huidobro-Toro, J.P. Histidine 140 plays a key role in the inhibitory modulation of the P2X4 nucleotide receptor by copper but not zinc. J. Biol. Chem. 2003, 278, 36777–36785. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, X.-X.; Jiang, S.-H.; Tao, L.-Y.; Li, Q.; Zhu, L.-L.; Yang, M.; Huo, Y.-M.; Jiang, Y.-S.; Tian, G.-A.; et al. Targeting Purinergic Receptor P2Y2 Prevents the Growth of Pancreatic Ductal Adenocarcinoma by Inhibiting Cancer Cell Glycolysis. Clin. Cancer Res. 2018, 25, 1318–1330. [Google Scholar] [CrossRef] [PubMed]
- Zaparte, A.; Cappellari, A.R.; Brandão, C.A.; de Souza, J.B.; Borges, T.J.; Kist, L.W.; Bogo, M.R.; Zerbini, L.F.; Pinto, L.F.R.; Glaser, T.; et al. P2Y2 receptor activation promotes esophageal cancer cells proliferation via ERK1/2 pathway. Eur. J. Pharmacol. 2020, 891, 173687. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-J.; Wang, L.-Y.; Qu, H.-N.; Yu, L.-H.; Burnstock, G.; Ni, X.; Xu, M.; Ma, B. P2Y2 receptor-mediated modulation of estrogen-induced proliferation of breast cancer cells. Mol. Cell. Endocrinol. 2011, 338, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Draganov, D.; Gopalakrishna-Pillai, S.; Chen, Y.-R.; Zuckerman, N.S.; Moeller, S.; Wang, C.; Ann, D.; Lee, P.P. Modulation of P2X4/P2X7/Pannexin-1 sensitivity to extracellular ATP via Ivermectin induces a non-apoptotic and inflammatory form of cancer cell death. Sci. Rep. 2015, 5, 16222. [Google Scholar] [CrossRef]
- He, J.; Zhou, Y.; Carrera, H.M.A.; Sprules, A.; Neagu, R.; Zarkesh, S.A.; Eaton, C.; Luo, J.; Gartland, A.; Wang, N. Inhibiting the P2X4 Receptor Suppresses Prostate Cancer Growth In Vitro and In Vivo, Suggesting a Potential Clinical Target. Cells 2020, 9, 2511. [Google Scholar] [CrossRef]
- Wan, H.; Xie, R.; Xu, J.; He, J.; Tang, B.; Liu, Q.; Wang, S.; Guo, Y.; Yang, X.; Dong, T.X.; et al. Anti-proliferative Effects of Nucleotides on Gastric Cancer via a Novel P2Y6/SOCE/Ca2+/β-catenin Pathway. Sci. Rep. 2017, 7, 2459. [Google Scholar] [CrossRef] [PubMed]
- Agteresch, H.J.; Burgers, S.A.; Van Der Gaast, A.; Wilson, J.H.P.; Dagnelie, P.C. Randomized clinical trial of adenosine 5′-triphosphate on tumor growth and survival in advanced lung cancer patients. Anti-Cancer Drugs 2003, 14, 639–644. [Google Scholar] [CrossRef]
- Nakamura, K.; Yoshikawa, N.; Yamaguchi, Y.; Kagota, S.; Shinozuka, K.; Kunitomo, M. Antitumor effect of cordycepin (3′-deoxyadenosine) on mouse melanoma and lung carcinoma cells involves adenosine A3 receptor stimulation. Anticancer Res. 2006, 26, 43–47. [Google Scholar]
- Fang, W.G.; Tian, X.X. Identification of a new pro-invasion factor in tumor microenvironment: Progress in function and mechanism of extracellular ATP. Beijing Da Xue Xue Bao Yi Xue Ban 2017, 49, 188–195. [Google Scholar]
- Lim, S.H.; Sa, J.K.; Lee, D.W.; Kim, J.; Kim, S.T.; Park, S.H.; Ku, B.; Park, J.O.; Park, Y.S.; Lim, H.; et al. Systematic Evaluation of Gastric Tumor Cell Index and Two-Drug Combination Therapy via 3-Dimensional High-Throughput Drug Screening. Front. Oncol. 2019, 9, 1327. [Google Scholar] [CrossRef] [PubMed]
- Yokozaki, H. Molecular characteristics of eight gastric cancer cell lines established in Japan. Pathol. Int. 2000, 50, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Kumar, A.; Faheem, M.M.; Katoch, A.; Kumar, A.; Jamwal, V.L.; Nayak, D.; Golani, A.; Rasool, R.U.; Ahmad, S.M.; et al. Vimentin activation in early apoptotic cancer cells errands survival pathways during DNA damage inducer CPT treatment in colon carcinoma model. Cell Death Dis. 2019, 10, 467. [Google Scholar] [CrossRef]
- Luo, W.; Fang, W.; Li, S.; Yao, K. Aberrant expression of nuclear vimentin and related epithelial-mesenchymal transition markers in nasopharyngeal carcinoma. Int. J. Cancer 2012, 131, 1863–1873. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, X.; Li, Y.; Evers, M.; Zhang, H.; Chen, X. Extracellular and macropinocytosis internalized ATP work together to induce epithelial–mesenchymal transition and other early metastatic activities in lung cancer. Cancer Cell Int. 2019, 19, 254. [Google Scholar] [CrossRef]
- Liu, D.; Shi, K.; Fu, M.; Chen, F. Placenta-specific protein 1 promotes cell proliferation via the AKT/GSK-3β/cyclin D1 signaling pathway in gastric cancer. IUBMB Life 2021. [Google Scholar] [CrossRef]
- Cevenini, A.; Orrù, S.; Mancini, A.; Alfieri, A.; Buono, P.; Imperlini, E. Molecular Signatures of the Insulin-like Growth Factor 1-mediated Epithelial-Mesenchymal Transition in Breast, Lung and Gastric Cancers. Int. J. Mol. Sci. 2018, 19, 2411. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tian, F.; Chen, X.; Liu, Z.; Wu, C.; Zhao, Z. Ras-ERK1/2 signaling participates in the progression of gastric cancer through repressing Aurora B-mediated H1.4 phosphorylation at Ser27. J. Cell. Physiol. 2020. [Google Scholar] [CrossRef]
- Pari, A.A.A.; Singhal, M.; Hübers, C.; Mogler, C.; Schieb, B.; Gampp, A.; Gengenbacher, N.; Reynolds, L.E.; Terhardt, D.; Géraud, C.; et al. Tumor Cell–Derived Angiopoietin-2 Promotes Metastasis in Melanoma. Cancer Res. 2020, 80, 2586–2598. [Google Scholar] [CrossRef] [PubMed]
- Munakata, S.; Ueyama, T.; Ishihara, H.; Komiyama, H.; Tsukamoto, R.; Kawai, M.; Takahashi, M.; Kojima, Y.; Tomiki, Y.; Sakamoto, K. Angiopoietin-2 as a Prognostic Factor in Patients with Incurable Stage IV Colorectal Cancer. J. Gastrointest. Cancer 2020, 52, 237–242. [Google Scholar] [CrossRef]
- Du, Z.; Tang, C.-H.; Li, L.-J.; Kang, L.; Zhao, J.; Jin, L.; Wang, C.-Q.; Su, C.-M. Angiopoietin-2 gene polymorphisms are biomarkers for the development and progression of colorectal cancer in Han Chinese. Int. J. Med. Sci. 2020, 17, 97–102. [Google Scholar] [CrossRef]
- Colavito, S.A. AXL as a Target in Breast Cancer Therapy. J. Oncol. 2020, 2020, 5291952. [Google Scholar] [CrossRef]
- Ikushima, H.; Miyazono, K. TGFβ signalling: A complex web in cancer progression. Nat. Rev. Cancer 2010, 10, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Filippone, N.R.; Reiner, T.; Roberts, S. Sensors and Inhibitors for the Detection of Ataxia Telangiectasia Mutated (ATM) Protein Kinase. Mol. Pharm. 2021, 18, 2470–2481. [Google Scholar] [CrossRef]
- Cheng, X.; Wang, J.; Liu, C.; Jiang, T.; Yang, N.; Liu, D.; Zhao, H.; Xu, Z. Zinc transporter SLC39A13/ZIP13 facilitates the metastasis of human ovarian cancer cells via activating Src/FAK signaling pathway. J. Exp. Clin. Cancer Res. 2021, 40, 199. [Google Scholar] [CrossRef] [PubMed]
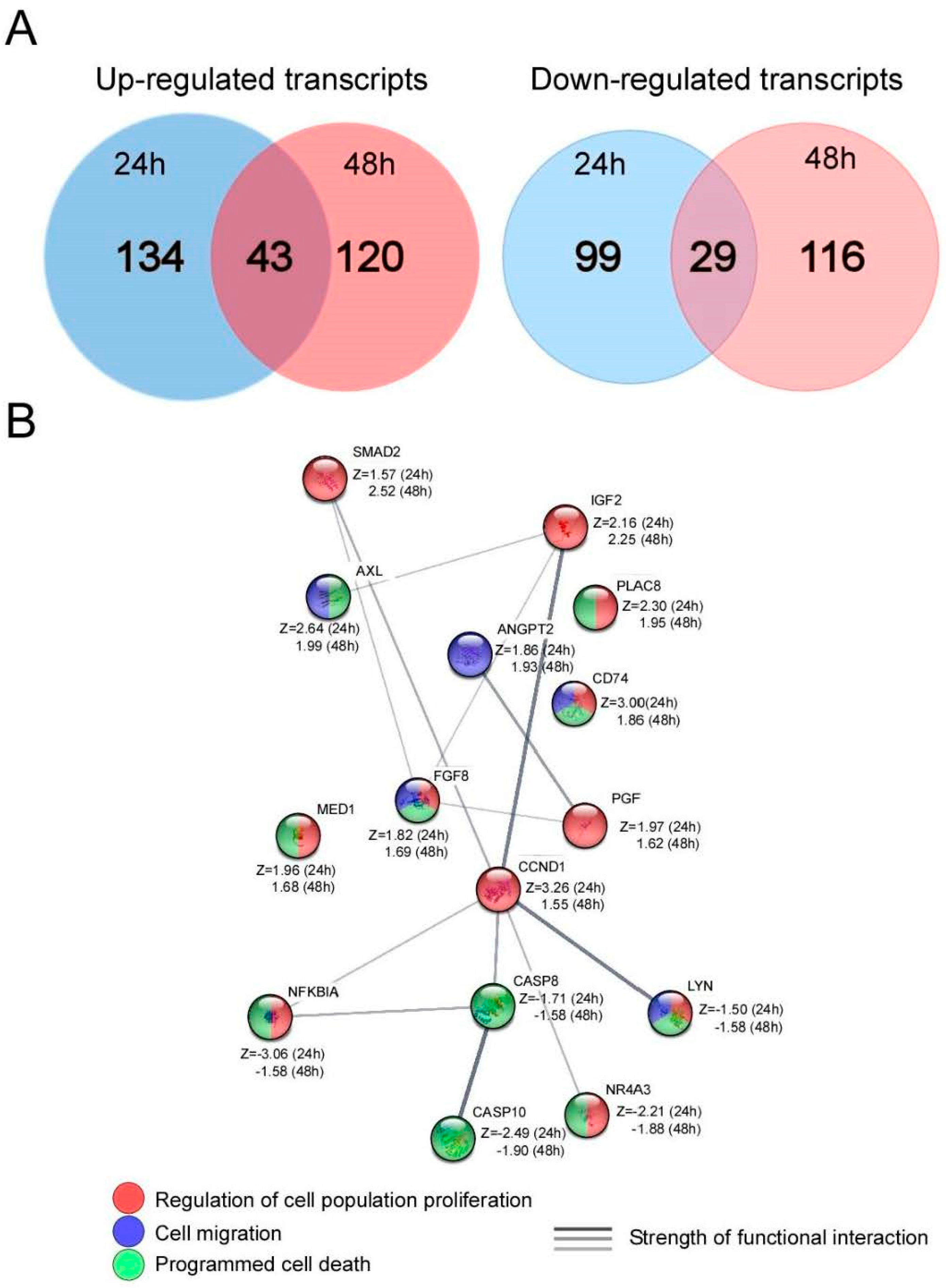
| Up-Regulated Genes in Networks | |||||
| GO Term | Description | Count in Network | FDR | ||
| 24 h | 48 h | 24 h | 48 h | ||
| GO:0030154 | Cell differentiation | 45 of 3457 | 44 of 3457 | 0.00011 | 2.04 × 10−5 |
| GO:0042127 | Regulation of cell population proliferation | 40 of 1594 | 33 of 1594 | 2.47 × 10−10 | 7.65 × 10−8 |
| GO:0016477 | Cell migration | 15 of 812 | 19 of 812 | 0.0046 | 2.62 × 10−5 |
| Down-Regulated Genes in Networks | |||||
| GO:0042493 | Response to drug | 20 of 900 | 15 of 900 | 1.85 × 10−6 | 0.0037 |
| GO:0012501 | Programmed cell death | 14 of 1042 | 15 of 1042 | 0.0072 | 0.0108 |
| GO:0045785 | Positive regulation of cell adhesion | 9 of 375 | 11 of 375 | 0.0018 | 0.00041 |
| Up-Regulated Genes in SHARED Network | |||
| GO Term | Description | Count in Network | FDR |
| GO:0030154 | Cell differentiation | 15 of 3457 | 0.0415 |
| GO:0042127 | Regulation of cell population proliferation | 14 of 1594 | 0.00078 |
| GO:007162 | Negative regulation of cell adhesion | 4 of 245 | 0.0233 |
| Up-Regulated Genes in SHARED Network | |||
| GO:0042493 | Response to drug | 7 of 900 | 0.0069 |
| GO:0012501 | Programmed cell death | 6 of 1042 | 0.0336 |
| GO:0045785 | Positive regulation of cell adhesion | 4 of 375 | 0.0243 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyna-Jeldes, M.; De la Fuente-Ortega, E.; Cerda, D.; Velázquez-Miranda, E.; Pinto, K.; Vázquez-Cuevas, F.G.; Coddou, C. Purinergic P2Y2 and P2X4 Receptors Are Involved in the Epithelial-Mesenchymal Transition and Metastatic Potential of Gastric Cancer Derived Cell Lines. Pharmaceutics 2021, 13, 1234. https://doi.org/10.3390/pharmaceutics13081234
Reyna-Jeldes M, De la Fuente-Ortega E, Cerda D, Velázquez-Miranda E, Pinto K, Vázquez-Cuevas FG, Coddou C. Purinergic P2Y2 and P2X4 Receptors Are Involved in the Epithelial-Mesenchymal Transition and Metastatic Potential of Gastric Cancer Derived Cell Lines. Pharmaceutics. 2021; 13(8):1234. https://doi.org/10.3390/pharmaceutics13081234
Chicago/Turabian StyleReyna-Jeldes, Mauricio, Erwin De la Fuente-Ortega, Daniela Cerda, Erandi Velázquez-Miranda, Katherine Pinto, Francisco G. Vázquez-Cuevas, and Claudio Coddou. 2021. "Purinergic P2Y2 and P2X4 Receptors Are Involved in the Epithelial-Mesenchymal Transition and Metastatic Potential of Gastric Cancer Derived Cell Lines" Pharmaceutics 13, no. 8: 1234. https://doi.org/10.3390/pharmaceutics13081234
APA StyleReyna-Jeldes, M., De la Fuente-Ortega, E., Cerda, D., Velázquez-Miranda, E., Pinto, K., Vázquez-Cuevas, F. G., & Coddou, C. (2021). Purinergic P2Y2 and P2X4 Receptors Are Involved in the Epithelial-Mesenchymal Transition and Metastatic Potential of Gastric Cancer Derived Cell Lines. Pharmaceutics, 13(8), 1234. https://doi.org/10.3390/pharmaceutics13081234






