Revisiting the Cytotoxicity of Cationic Polyelectrolytes as a Principal Component in Layer-by-Layer Assembly Fabrication
Abstract
:1. Introduction
2. Materials and Methods
2.1. Polyelectrolytes
2.2. Cell Culture
2.3. Cytotoxicity Analysis
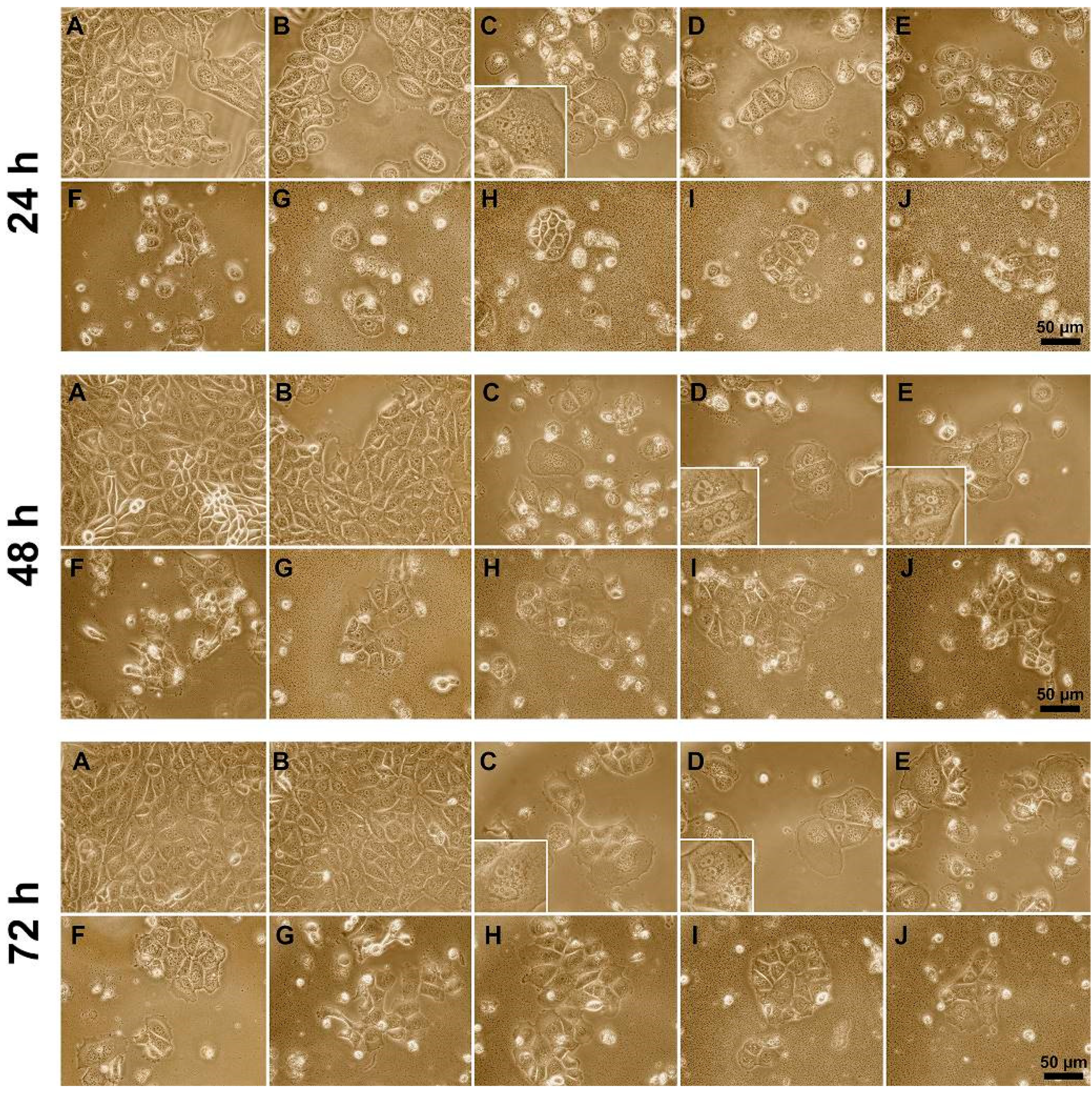
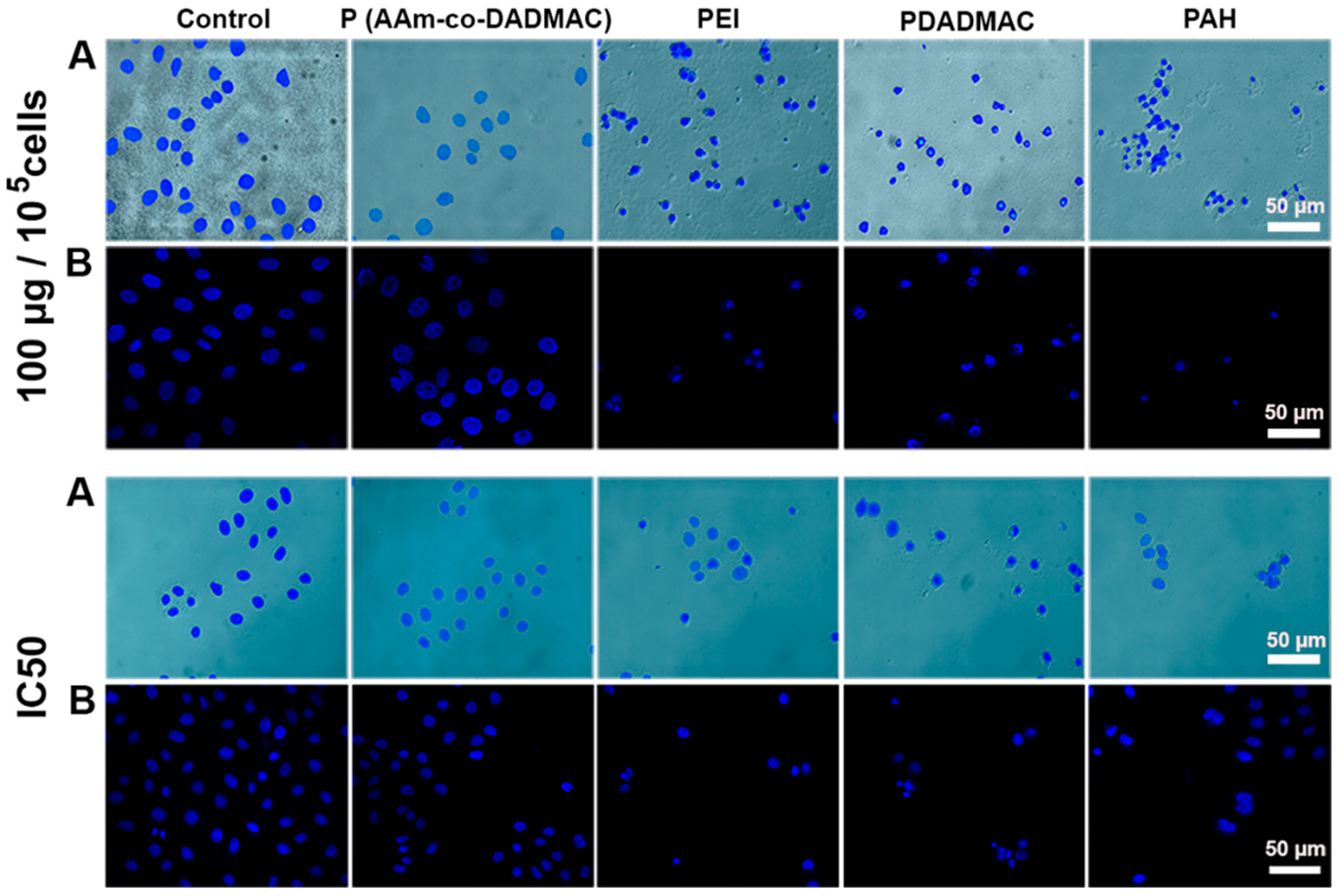
2.4. Cellular/Nuclear Morphology Study
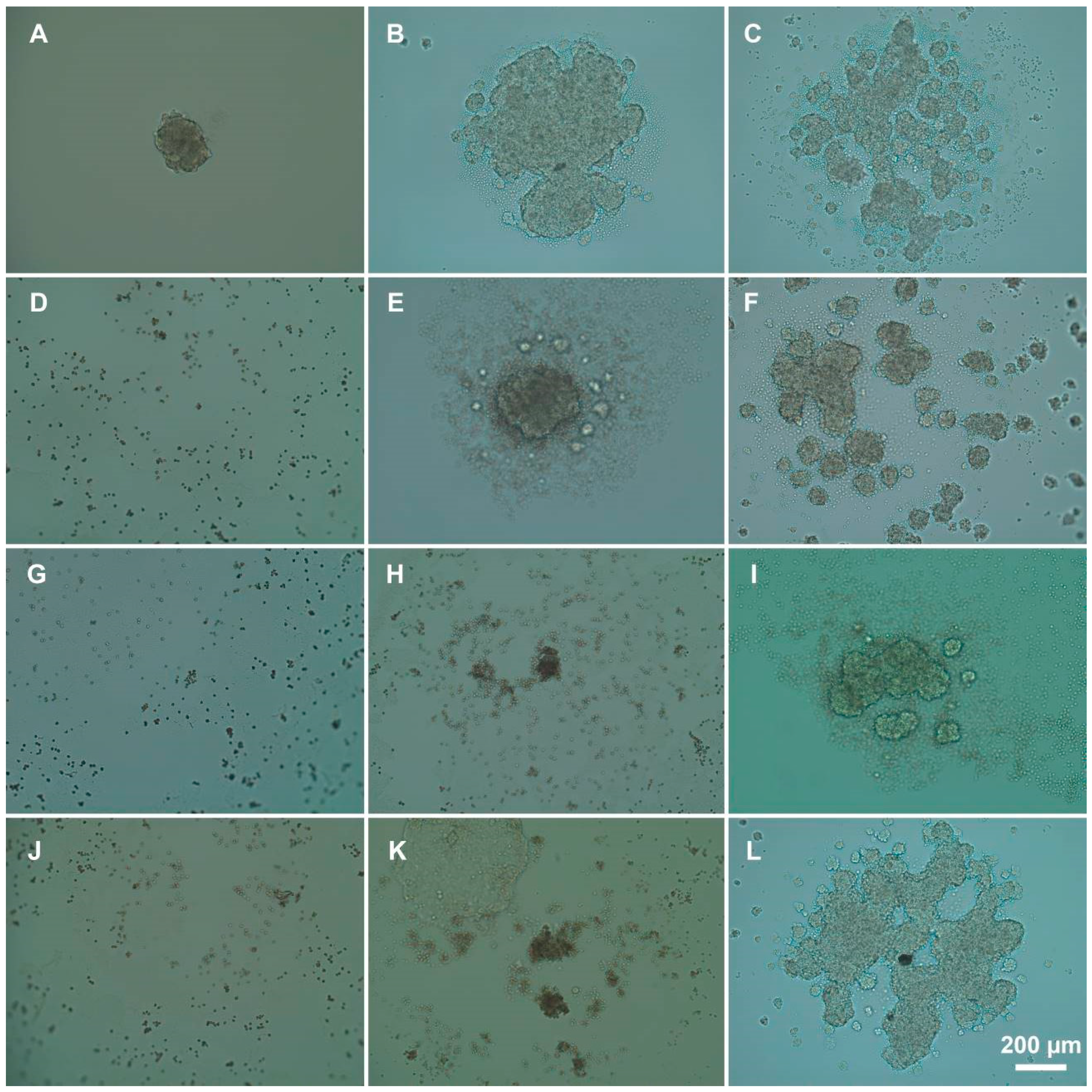
2.5. Multicellular Spheroids Formation
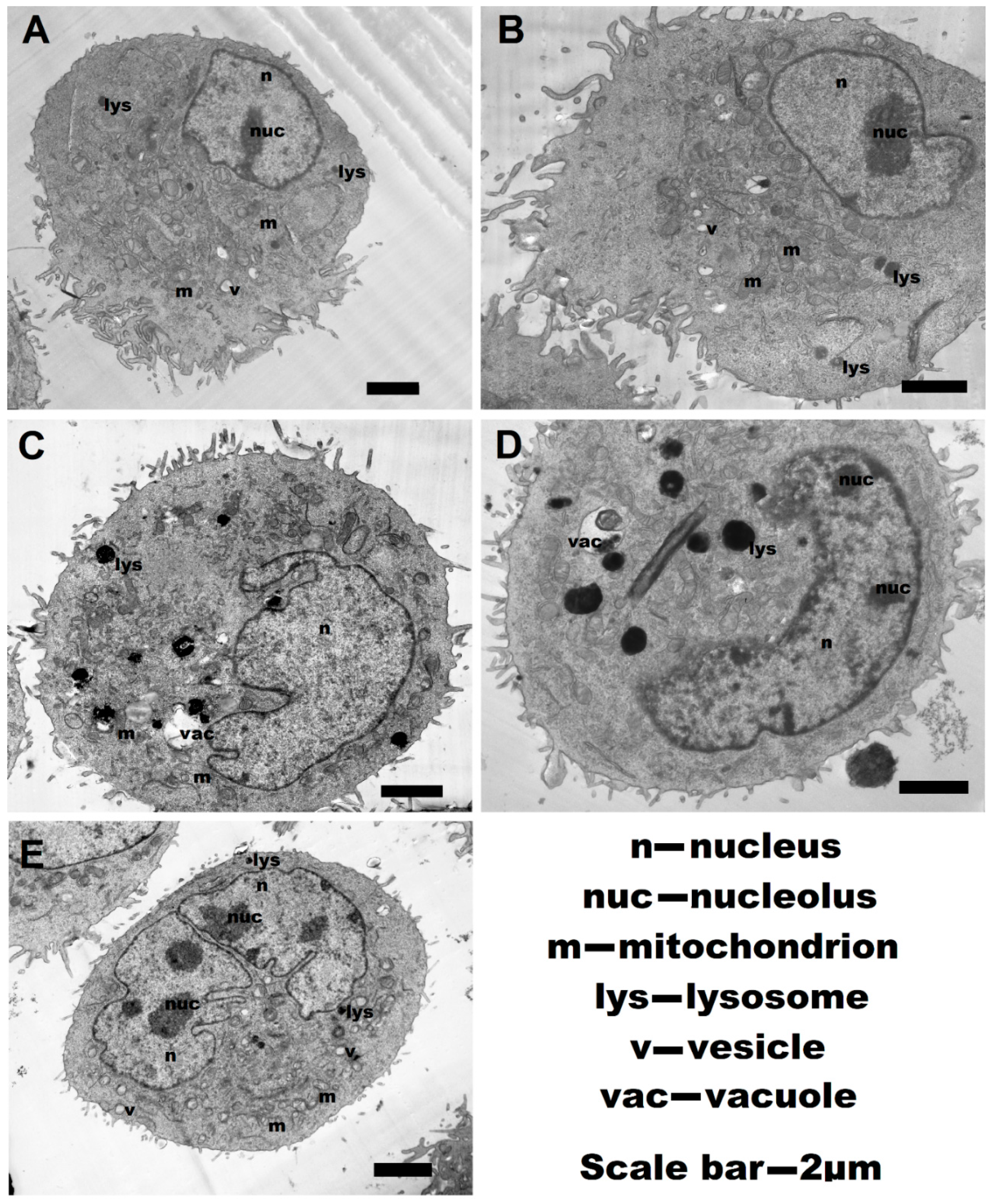
2.6. Transmission Electronic Microscopy (TEM)
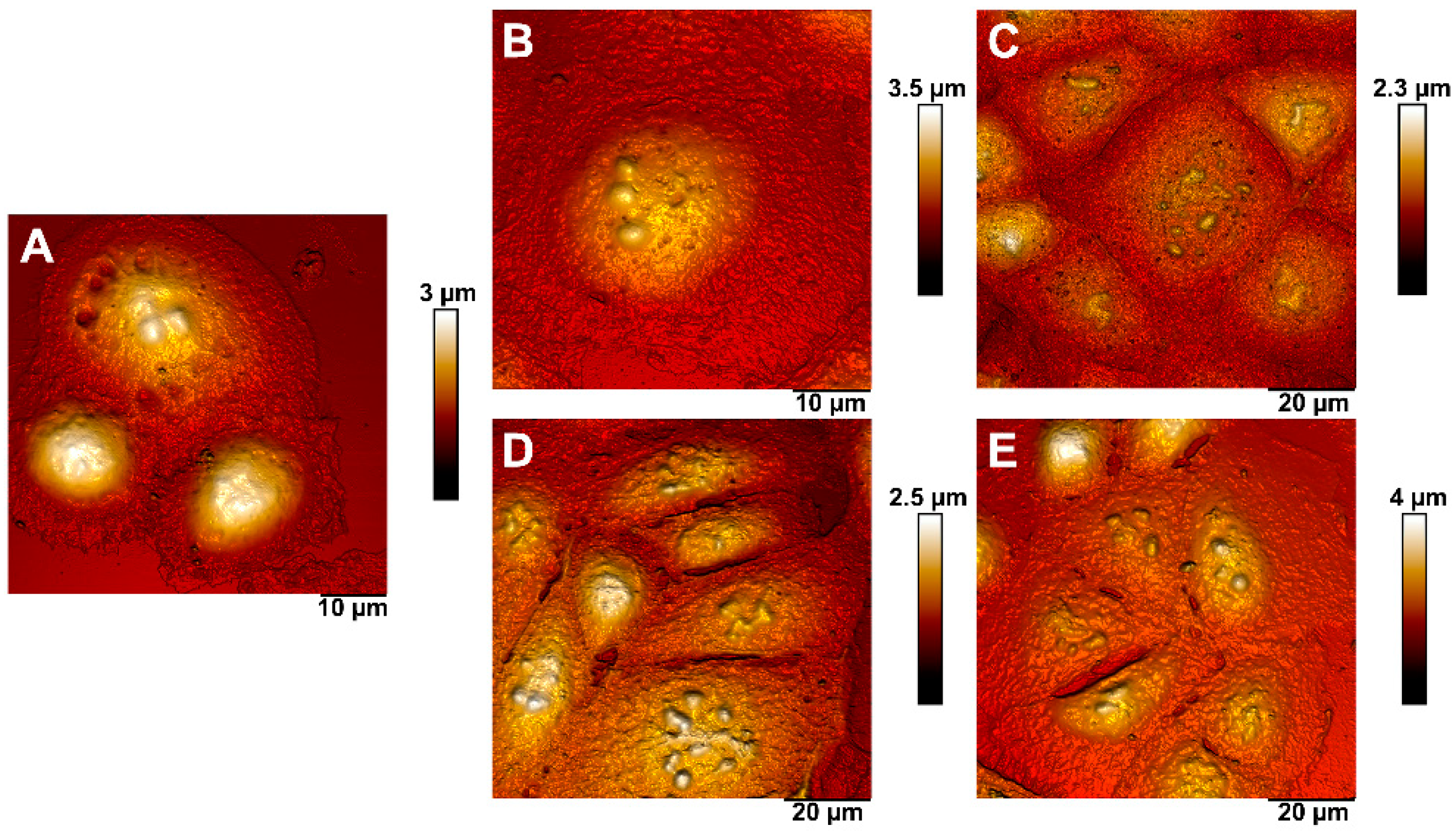
2.7. Atomic Force Microscopy (AFM)
2.8. Statistics
3. Results
3.1. Determination of Hydrodynamic Size and Zeta Potential
3.2. Cytotoxic Activity of Polyelectrolytes
3.3. Influence of Polyelectrolytes on 3D Cell Clusters Formation
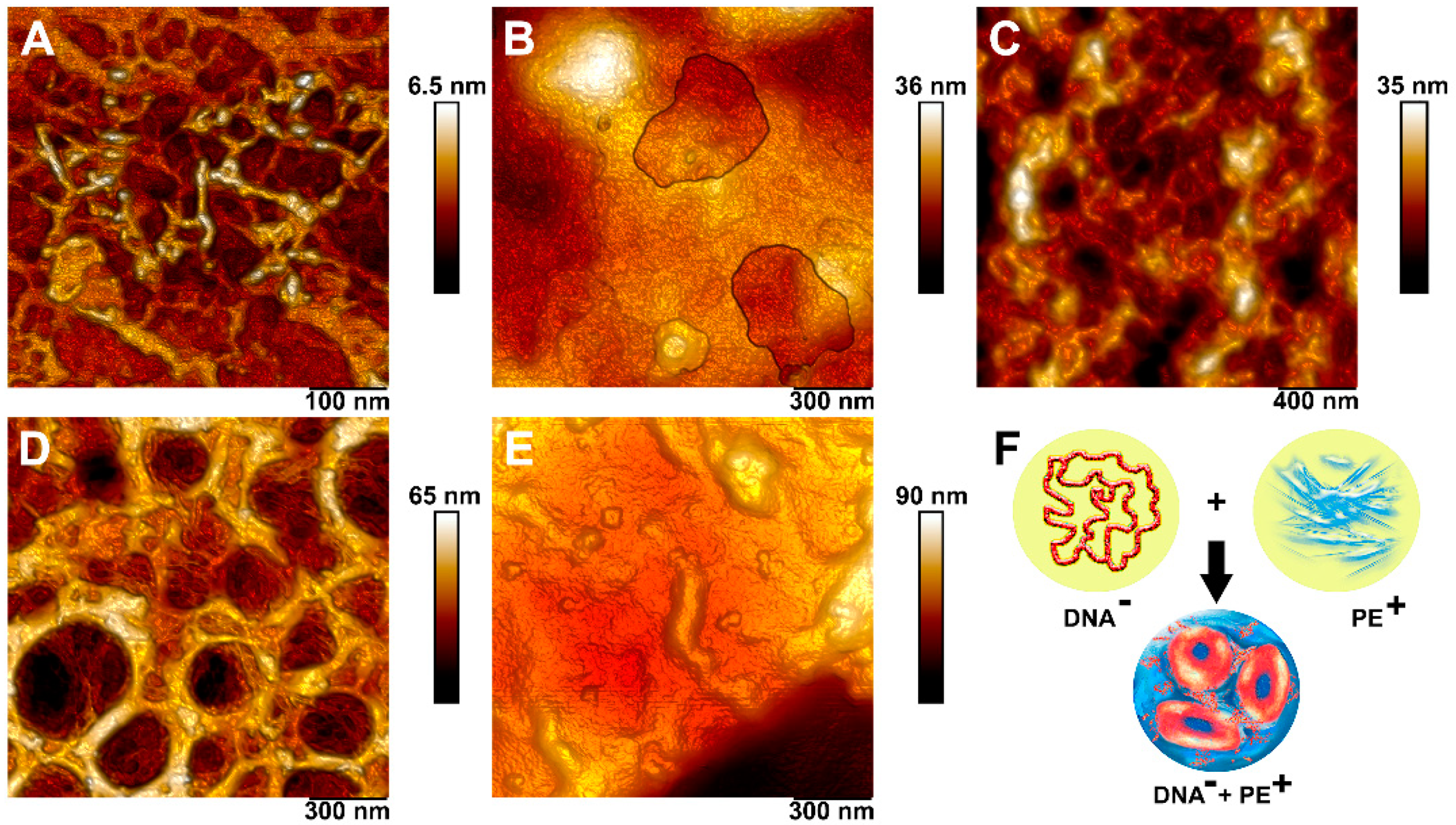
3.4. Complexation of DNA and Polyelectrolytes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, P.; Bookstaver, M.L.; Jewell, C.M. Engineering Cell Surfaces with Polyelectrolyte Materials for Translational Applications. Polymers 2017, 9, 40. [Google Scholar] [CrossRef] [Green Version]
- Rabiee, A.; Ershad-Langroudi, A.; Zeynali, M.E. A survey on cationic polyelectrolytes and their applications: Acrylamide derivatives. Rev. Chem. Eng. 2015, 31, 239–261. [Google Scholar] [CrossRef]
- Boudou, T.; Crouzier, T.; Ren, K.; Blin, G.; Picart, C. Multiple functionalities of polyelectrolyte multilayer films: New biomedical applications. Adv. Mater. 2010, 22, 441–467. [Google Scholar] [CrossRef]
- Lvov, Y.; Decher, G.; Moehwald, H. Assembly, structural characterization, and thermal behavior of layer-by-layer deposited ultrathin films of poly(vinyl sulfate) and poly(allylamine). Langmuir 1993, 9, 481–486. [Google Scholar] [CrossRef]
- Sukhorukov, G.B.; Mohwald, H.; Decher, G.; Lvov, Y.M. Assembly of polyelectrolyte multilayer films by consecutively alternating adsorption of polynucleotides and polycations. Thin Solid Films 1996, 284–285, 220–223. [Google Scholar] [CrossRef]
- Strydom, S.J.; Otto, D.P.; Stieger, N.; Aucamp, M.E.; Liebenberg, W.; De Villiers, M.M. Self-assembled macromolecular nanocoatings to stabilize and control drug release from nanoparticles. Powder Technol. 2014, 256, 470–476. [Google Scholar] [CrossRef]
- De Villiers, M.M.; Otto, D.P.; Strydom, S.J.; Lvov, Y.M. Introduction to nanocoatings produced by layer-by-layer (LbL) self-assembly. Adv. Drug Deliv. Rev. 2011, 63, 701–715. [Google Scholar] [CrossRef]
- Otto, D.P.; de Villiers, M.M. Coarse-Grained molecular dynamics (CG-MD) simulation of the encapsulation of dexamethasone in PSS/PDDA Layer-by-Layer assembled polyelectrolyte nanocapsules. AAPS PharmSciTech 2020, 21, 292. [Google Scholar] [CrossRef] [PubMed]
- Prevot, M.; Cordeiro, A.L.; Sukhorukov, G.B.; Lvov, Y.; Besser, R.S.; Mohwald, H. Design of a microfluidic system to investigate the mechanical properties of Layer-by-Layer fabricated capsules. Macromol. Mat. Eng. 2003, 288, 915–919. [Google Scholar] [CrossRef]
- Ariga, K.; Hill, J.P.; Ji, Q. Layer-by-layer assembly as a versatile bottom-up nanofabrication technique for exploratory research and realistic application. Phys. Chem. Chem. Phys. 2007, 9, 2319–2340. [Google Scholar] [CrossRef] [PubMed]
- Ariga, K.; Nakanishi, T.; Michinobu, T. Immobilization of biomaterials to nano-assembled films (self-assembled monolayers, langmuir-blodgett films, and layer-by-layer assemblies) and their related functions. J. Nanosci. Nanotechnol. 2006, 6, 2278–2301. [Google Scholar] [CrossRef]
- Fujii, N.; Fujimoto, K.; Michinobu, T.; Akada, M.; Hill, J.P.; Shiratori, S.; Ariga, K.; Shigehara, K. The simplest layer-by-layer assembly structure: Best paired polymer electrolytes with one charge per main chain carbon atom for multilayered thin films. Macromolecules 2010, 43, 3947–3955. [Google Scholar] [CrossRef]
- Fakhrullin, R.F.; Brandy, M.-L.; Cayre, O.J.; Velev, O.D.; Paunov, V.N. Live celloidosome structures based on the assembly of individual cells by colloid interactions. Phys. Chem. Chem. Phys. 2010, 12, 11912–11922. [Google Scholar] [CrossRef]
- Naumenko, E.A.; Dzamukova, M.R.; Fakhrullina, G.I.; Akhatova, F.S.; Fakhrullin, R.F. Nano-labelled cells—A functional tool in biomedical applications. Curr. Opin. Pharmacol. 2014, 18, 84–90. [Google Scholar] [CrossRef]
- Van der Meeren, L.; Verduijn, J.; Li, J.; Parakhonskiy, B.V.; Skirtach, A.G. Encapsulation of cells in gold nanoparticle functionalized hybrid Layer-by-Layer (LbL) hybrid shells—Remote effect of laser light. Appl. Surf. Sci. Adv. 2021, 5, 100111. [Google Scholar] [CrossRef]
- Papagiannopoulos, A. Current Research on Polyelectrolyte Nanostructures: From Molecular Interactions to Biomedical Applications. Macromol 2021, 1, 155–172. [Google Scholar] [CrossRef]
- Liu, T.; Wang, Y.; Zhong, W.; Li, B.; Mequanint, K.; Luo, G.; Xing, M. Biomedical Applications of Layer-by-Layer Self-Assembly for Cell Encapsulation: Current Status and Future Perspectives. Adv. Health Mater. 2019, 8, 1800939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escobar, A.; Muzzio, N.; Moya, S.E. Antibacterial Layer-by-Layer Coatings for Medical Implants. Pharmaceutics 2021, 13, 16. [Google Scholar] [CrossRef]
- Jewell, C.M.; Lynn, D.M. Multilayered polyelectrolyte assemblies as platforms for the delivery of DNA and other nucleic acid-based therapeutics. Adv. Drug Deliv. Rev. 2008, 60, 979–999. [Google Scholar] [CrossRef] [Green Version]
- Goswami, R.; Subramanian, G.; Silayeva, L.; Newkirk, I.; Doctor, D.; Chawla, K.; Chattopadhyay, S.; Chandra, D.; Chilukuri, N.; Betapudi, V. Gene Therapy Leaves a Vicious Cycle. Front. Oncol. 2019, 9, 297. [Google Scholar] [CrossRef]
- Lagasse, H.A.; Alexaki, A.; Simhadri, V.L.; Katagiri, N.H.; Jankowski, W.; Sauna, Z.E.; Kimchi-Sarfaty, C. Recent advances in (therapeutic protein) drug development. F1000Research 2017, 6, 113. [Google Scholar] [CrossRef] [Green Version]
- Durymanov, M.; Reineke, J. Non-viral Delivery of Nucleic Acids: Insight into Mechanisms of Overcoming Intracellular Barriers. Front. Pharmacol. 2018, 9, 971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rozhina, E.; Batasheva, S.; Gomzikova, M.; Naumenko, E.; Fakhrullin, R. Multicellular spheroids formation: The synergistic effects of halloysite nanoclay and cationic magnetic nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2019, 565, 16–24. [Google Scholar] [CrossRef]
- Akhatova, F.; Fakhrullina, G.; Khakimova, E.; Fakhrullin, R. Atomic force microscopy for imaging and nanomechanical characterisation of live nematode epicuticle: A comparative Caenorhabditis elegans and Turbatrix aceti study. Ultramicroscopy 2018, 194, 40–47. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Odrobińska, J.; Skonieczna, M.; Neugebauer, D. PEG Graft Polymer Carriers of Antioxidants: In Vitro Evaluation for Transdermal Delivery. Pharmaceutics 2020, 12, 1178. [Google Scholar] [CrossRef]
- Repetto, G.; del Peso, A.; Zurita, J. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat. Protoc. 2008, 3, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Sgouras, D.; Duncan, R. Methods for the evaluation of biocompatibility of soluble synthetic polymers which have potential for biomedical use: 1—use of the tetrazolium-based colorimetric assay (MTT) as a preliminary screen for evaluation of in vitro cytotoxicity. J. Mater. Sci. Mater. Med. 1990, 1, 61–68. [Google Scholar] [CrossRef]
- Choksakulnimitr, S.; Masuda, S.; Tokuda, H.; Takakura, Y.; Hashida, M. In vitro cytotoxicity of macromolecules in different cell culture systems. J. Control. Release 1995, 34, 233–241. [Google Scholar] [CrossRef]
- Morgan, D.M.; Clover, J.; Pearson, J.D. Effects of synthetic polycations on leucine incorporation, lactate dehydrogenase release, and morphology of human umbilical vein endothelial cells. J. Cell Sci. 1988, 91, 231–238. [Google Scholar] [CrossRef]
- Guryanov, I.; Naumenko, E.; Konnova, S.; Lagarkova, M.; Kiselev, S.; Fakhrullin, R. Spatial manipulation of magnetically-responsive nanoparticle engineered human neuronal progenitor cells. Nanomed. Nanotechnol. Biol. Med. 2019, 20, 102038. [Google Scholar] [CrossRef]
- Dzamukova, M.; Naumenko, E.; Lannik, N.; Fakhrullin, R. Surface-modified magnetic human cells for scaffold-free tissue engineering. Biomater. Sci. 2013, 1, 810–813. [Google Scholar] [CrossRef]
- Rozhina, E.; Danilushkina, A.; Akhatova, F.; Fakhrullin, R.; Rozhin, A.; Batasheva, S. Biocompatibility of magnetic nanoparticles coating with polycations using A549 cells. J. Biotechnol. 2021, 325, 25–34. [Google Scholar] [CrossRef]
- Tarasova, E.; Naumenko, E.; Rozhina, E.; Akhatova, F.; Fakhrullin, R. Cytocompatibility and uptake of polycations-modified halloysite clay nanotubes. Appl. Clay Sci. 2019, 169, 21–30. [Google Scholar] [CrossRef]
- Fischer, D.; Li, Y.; Ahlemeyer, B.; Krieglstein, J.; Kissel, T. In vitro cytotoxicity testing of polycations: Influence of polymer structure on cell viability and hemolysis. Biomaterials 2003, 24, 1121–1131. [Google Scholar] [CrossRef]
- Formigli, L.; Papucci, L.; Tani, A.; Schiavone, N.; Tempestini, A.; Orlandini, G.; Capaccioli, S.; Zecchi Orlandini, S. Aponecrosis: Morphological and biochemical exploration of a syncretic process of cell death sharing apoptosis and necrosis. J. Cell. Physiol. 2000, 182, 41–49. [Google Scholar] [CrossRef]
- Small, J.V.; Kaverina, I.; Krylyshkina, O.; Rottner, K. Cytoskeleton cross-talk during cell motility. FEBS Lett. 1999, 452, 96–99. [Google Scholar] [CrossRef] [Green Version]
- Alpin, A.E.; Howe, A.; Alahari, S.K.; Juliano, R.L. Signal transductions and signal modulations by cell adhesion receptors: The role of integrins, cadherins, immunoglobulin cell adhesion molecule and selectins. Pharmacol. Rev. 1998, 50, 197–263. [Google Scholar]
- Bershadsky, A.D.; Balaban, N.Q.; Geiger, B. Adhesion-dependent cell mechanosensitivity. Annu. Rev. Cell Dev. Biol. 2003, 19, 677–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cartagena-Rivera, A.X.; Van Itallie, C.M.; Anderson, J.M.; Chadwick, R.S. Apical surface supracellular mechanical properties in polarized epithelium using noninvasive acoustic force spectroscopy. Nat. Commun. 2017, 8, 1030. [Google Scholar] [CrossRef] [Green Version]
- Leong, D.T.; Kee, W.N. Probing the relevance of 3D cancer models in nanomedicine research. Adv. Drug Deliv. Rev. 2014, 79–80, 95–106. [Google Scholar] [CrossRef]
- Theodoraki, M.A.; Rezende, C.O., Jr.; Chantarasriwong, O.; Corben, A.D.; Theodorakis, E.A.; Alpaugh, M.L. Spontaneously-forming spheroids as an in vitro cancer cell model for anticancer drug screening. Oncotarget 2015, 6, 21255–21267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evtugyn, G.; Stepanova, V.; Porfireva, A.; Zamaleeva, A.; Fakhrullin, R. Electrochemical DNA sensors based on nanostructured organic dyes/DNA/polyelectrolyte complexes. J. Nanosci. Nanotechnol. 2014, 14, 6738–6747. [Google Scholar] [CrossRef]
- Wolfert, M.A.; Seymour, L.W. Atomic force microscopic analysis of the influence of the molecular weight of poly(L)lysine on the size of polyelectrolyte complexes formed with DNA. Gene Ther. 1996, 3, 269–273. [Google Scholar] [PubMed]
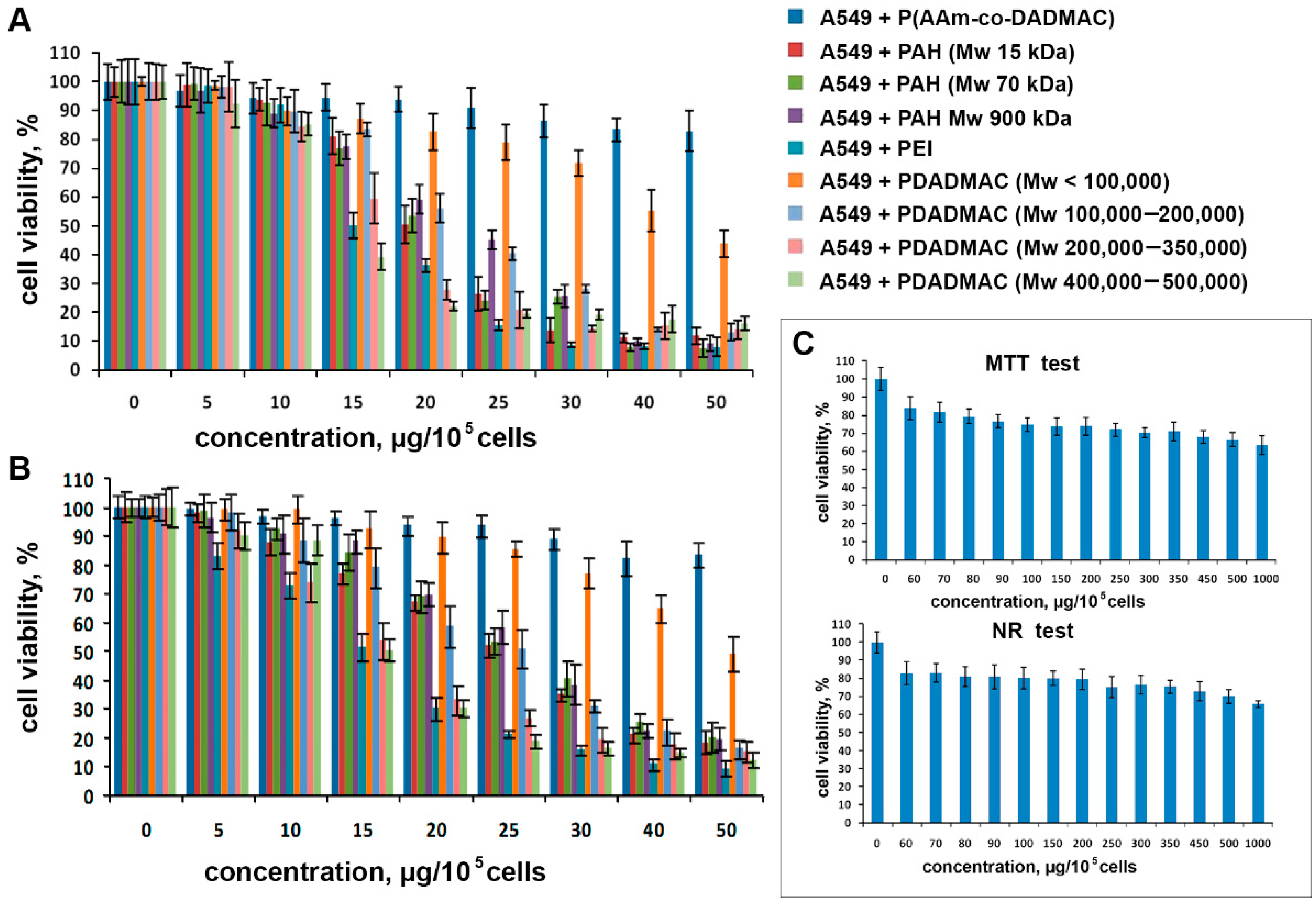
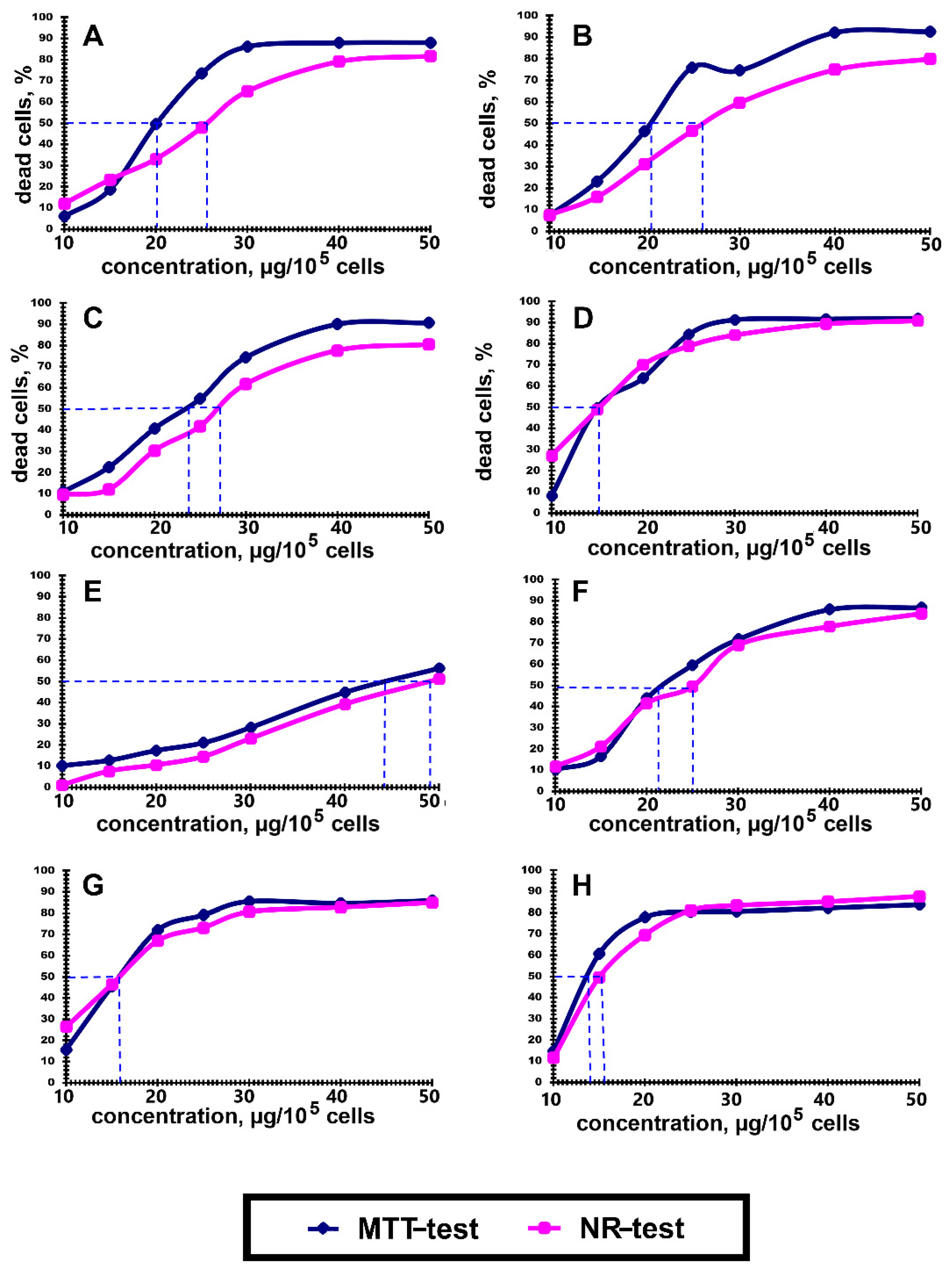

| Polyelectrolyte | Size (d.nm) | Zeta Potential (mV) |
|---|---|---|
| Poly(acrylamide-co-diallyldimethyl-ammonium chloride) 10 wt% solution in H2O P(AAm-co-DADMAC) | 83.9 ± 0.4 | 7.0 ± 0.4 |
| Poly(allylamine hydrochloride) PAH Mw15 kDa | 160.3 ± 12.2 | 20.1 ± 2.5 |
| Poly(allylamine hydrochloride) PAH Mw 70 kDa | 306.2 ± 32.7 | 38.0 ± 3.8 |
| Poly(allylamine hydrochloride) PAH Mw 900 kDa | 196.8 ± 19.4 | 68.9 ± 6.6 |
| Poly(ethyleneimine) 50 wt% solution in H2O PEI | 85.7 ± 0.7 | 21.1 ± 1.0 |
| Poly(diallyldimethhylammonium chloride) solution 35 wt% in H2O PDADMAC average Mw < 100,000 (very low molecular weight) | 120.2 ± 6.8 | 31.1 ± 2.3 |
| Poly(diallyldimethhylammonium chloride) solution 20 wt% in H2O PDADMAC average Mw 100,000–200,000 (low molecular weight) | 150.8 ± 14.8 | 34.0 ± 2.1 |
| Poly(diallyldimethhylammonium chloride) solution 20 wt% in H2O PDADMAC average Mw 200,000–350,000 (medium molecular weight) | 350.6 ± 33.6 | 55.3 ± 5.5 |
| Poly(diallyldimethhylammonium chloride) solution 20 wt% in H2O PDADMAC average Mw 400,000–500,000 (high molecular weight) | 700.8 ± 40.2 | 56.4 ± 4.3 |
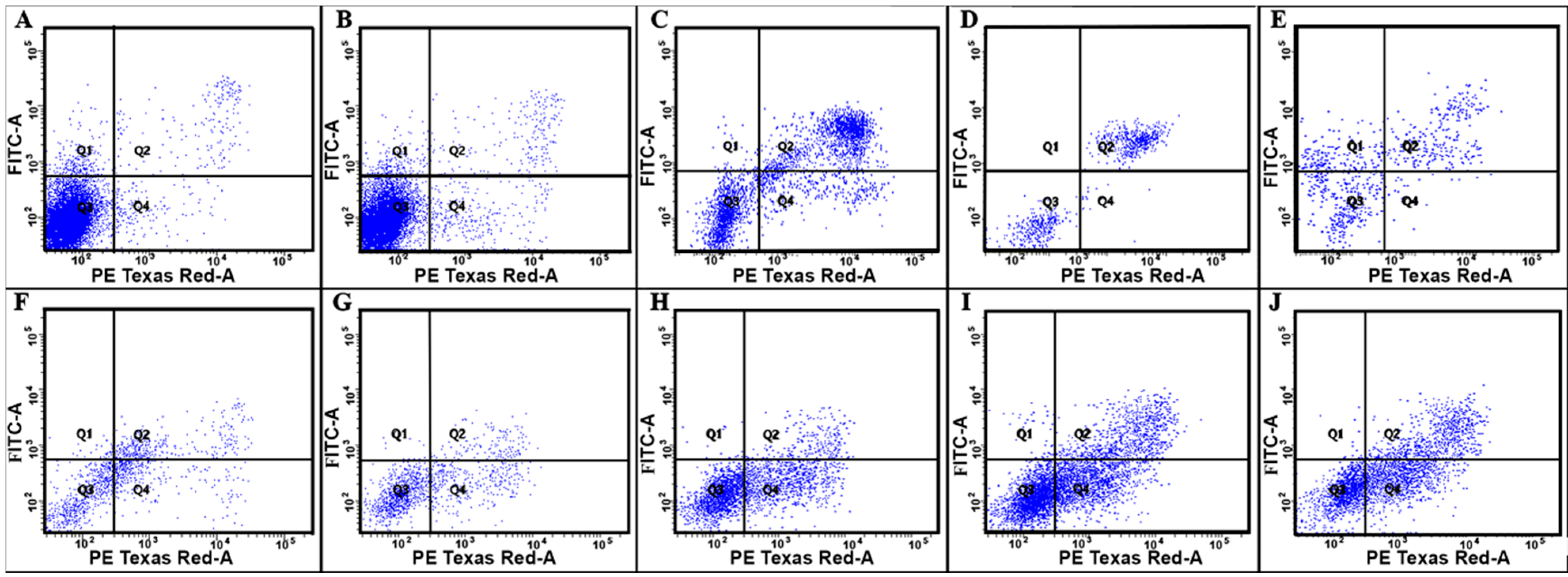
| Polyelectrolyte | Parameters | |||
|---|---|---|---|---|
| Apoptosis, % * (Q1) | Aponecrosis, % * (Q2) | Alive Cells, % * (Q3) | Necrosis, % * (Q4) | |
| Control | 1.5 | 1.1 | 96.5 | 1.0 |
| P(AAm-co-DADMAC) | 1.2 | 1.2 | 96.0 | 1.6 |
| PAH Mw 15 kDa | 1.3 | 43.6 | 44.5 | 10.6 |
| PAH Mw 70 kDa | - | 56.0 | 42.3 | 1.6 |
| PAH Mw 900 kDa | 21.2 | 31.6 | 43.9 | 3.3 |
| PEI | 3.0 | 21.9 | 52.1 | 23.0 |
| PDADMAC average Mw < 100,000 | 0.7 | 8.9 | 68.3 | 22.1 |
| PDADMAC average Mw 100,000–200,000 | 0.5 | 7.6 | 59.9 | 32.0 |
| PDADMAC average Mw 200,000–350,000 | 0.8 | 15.7 | 51.0 | 32.5 |
| PDADMAC average Mw 400,000–500,000 | 0.6 | 23.9 | 38.5 | 37.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naumenko, E.; Akhatova, F.; Rozhina, E.; Fakhrullin, R. Revisiting the Cytotoxicity of Cationic Polyelectrolytes as a Principal Component in Layer-by-Layer Assembly Fabrication. Pharmaceutics 2021, 13, 1230. https://doi.org/10.3390/pharmaceutics13081230
Naumenko E, Akhatova F, Rozhina E, Fakhrullin R. Revisiting the Cytotoxicity of Cationic Polyelectrolytes as a Principal Component in Layer-by-Layer Assembly Fabrication. Pharmaceutics. 2021; 13(8):1230. https://doi.org/10.3390/pharmaceutics13081230
Chicago/Turabian StyleNaumenko, Ekaterina, Farida Akhatova, Elvira Rozhina, and Rawil Fakhrullin. 2021. "Revisiting the Cytotoxicity of Cationic Polyelectrolytes as a Principal Component in Layer-by-Layer Assembly Fabrication" Pharmaceutics 13, no. 8: 1230. https://doi.org/10.3390/pharmaceutics13081230
APA StyleNaumenko, E., Akhatova, F., Rozhina, E., & Fakhrullin, R. (2021). Revisiting the Cytotoxicity of Cationic Polyelectrolytes as a Principal Component in Layer-by-Layer Assembly Fabrication. Pharmaceutics, 13(8), 1230. https://doi.org/10.3390/pharmaceutics13081230








