Preparation of Magnetic–Luminescent Bifunctional Rapeseed Pod-Like Drug Delivery System for Sequential Release of Dual Drugs †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
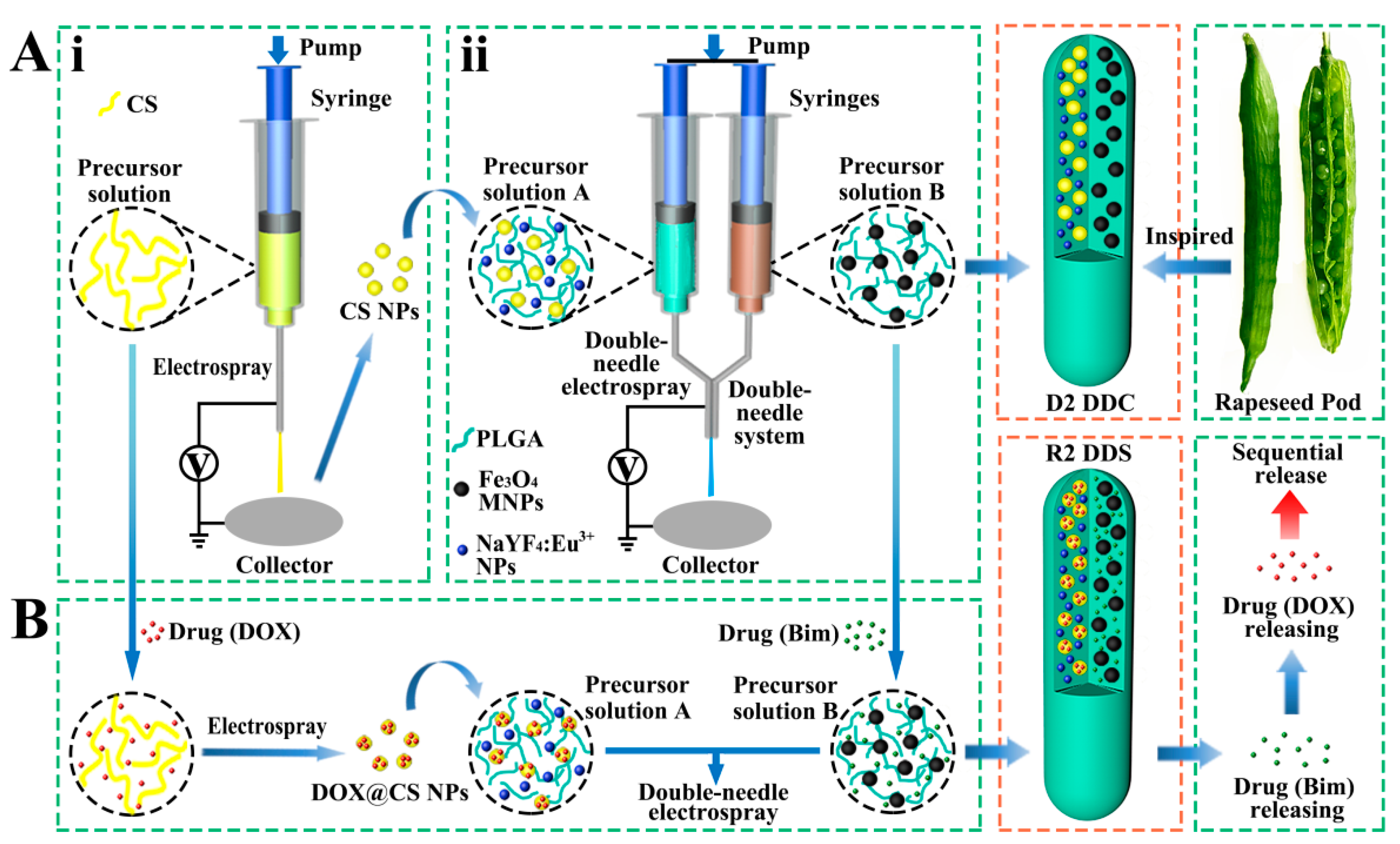
2.2. Preparation of Rapeseed Pod-Like DDCs
2.2.1. Synthesis of Fe3O4 MNPs
2.2.2. Synthesis of NaYF4:Eu3+ NPs
2.2.3. Preparation of CS NPs
2.2.4. Preparation of Rapeseed Pod-Like DDCs
2.3. Preparation of Rapeseed Pod-Like DDSs
2.4. Characterizations
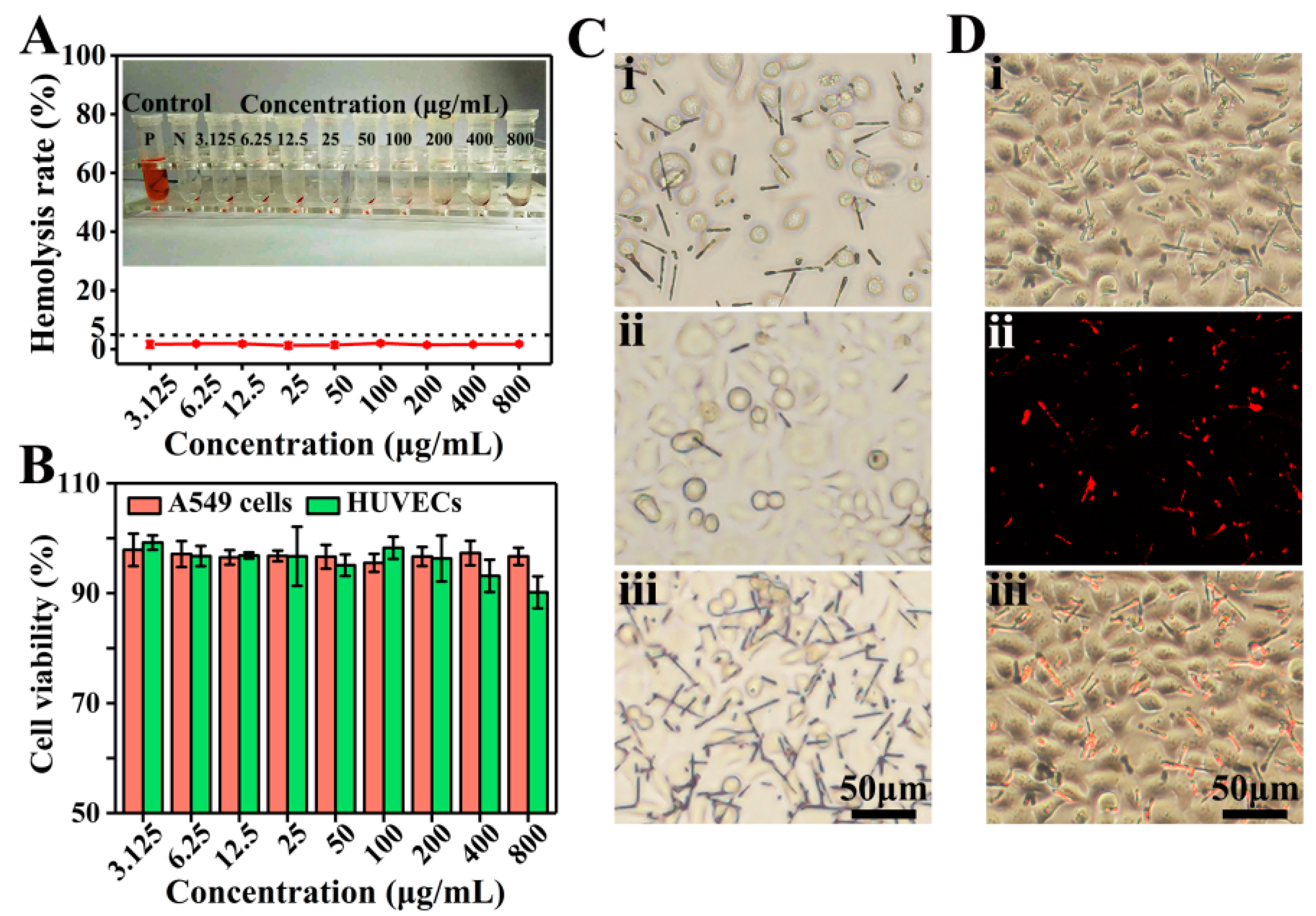
2.5. Hemocompatibility Evaluation
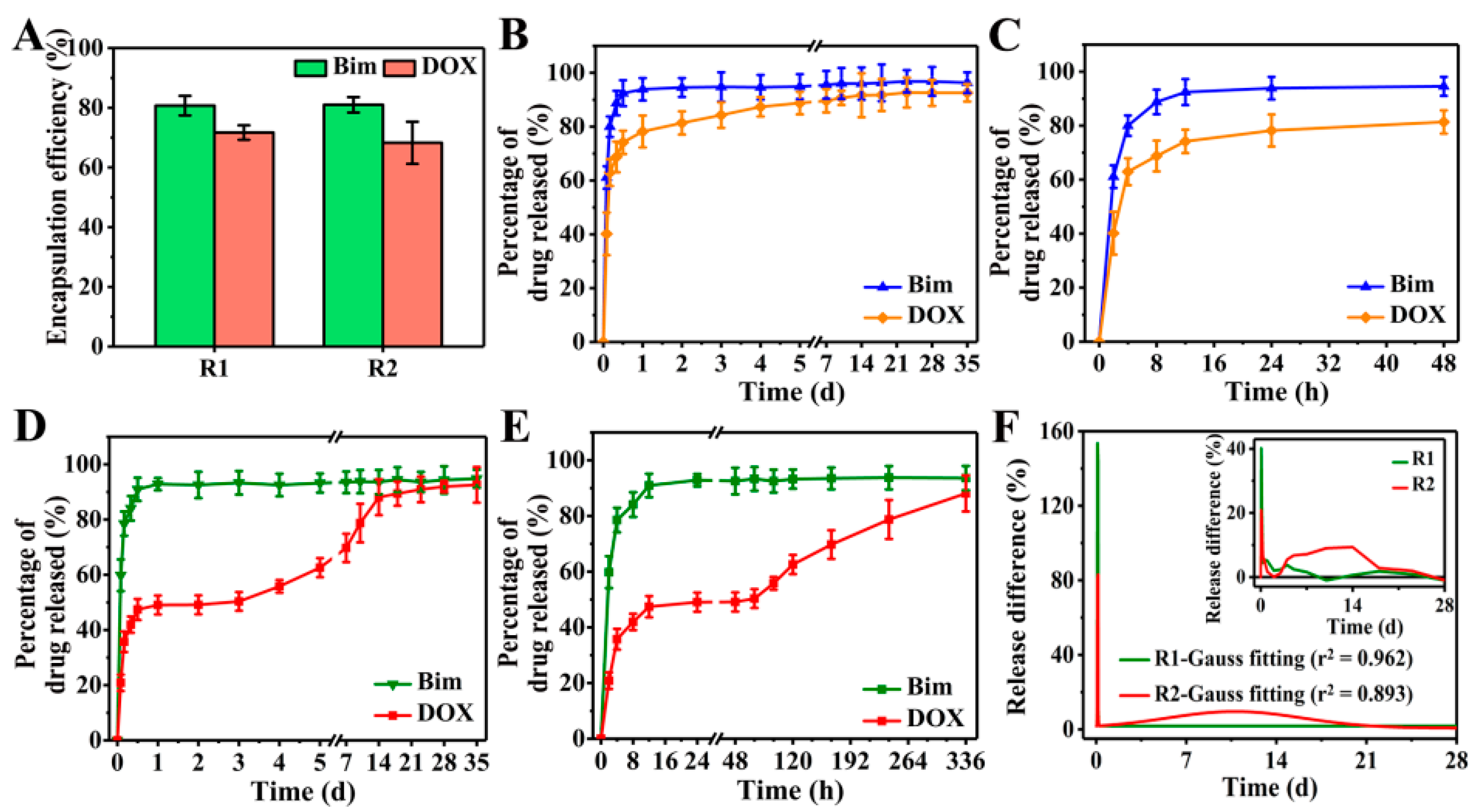
2.6. In Vitro Release Studies of Two Model Drugs
2.7. Cell Experiments
2.7.1. Cell Culture
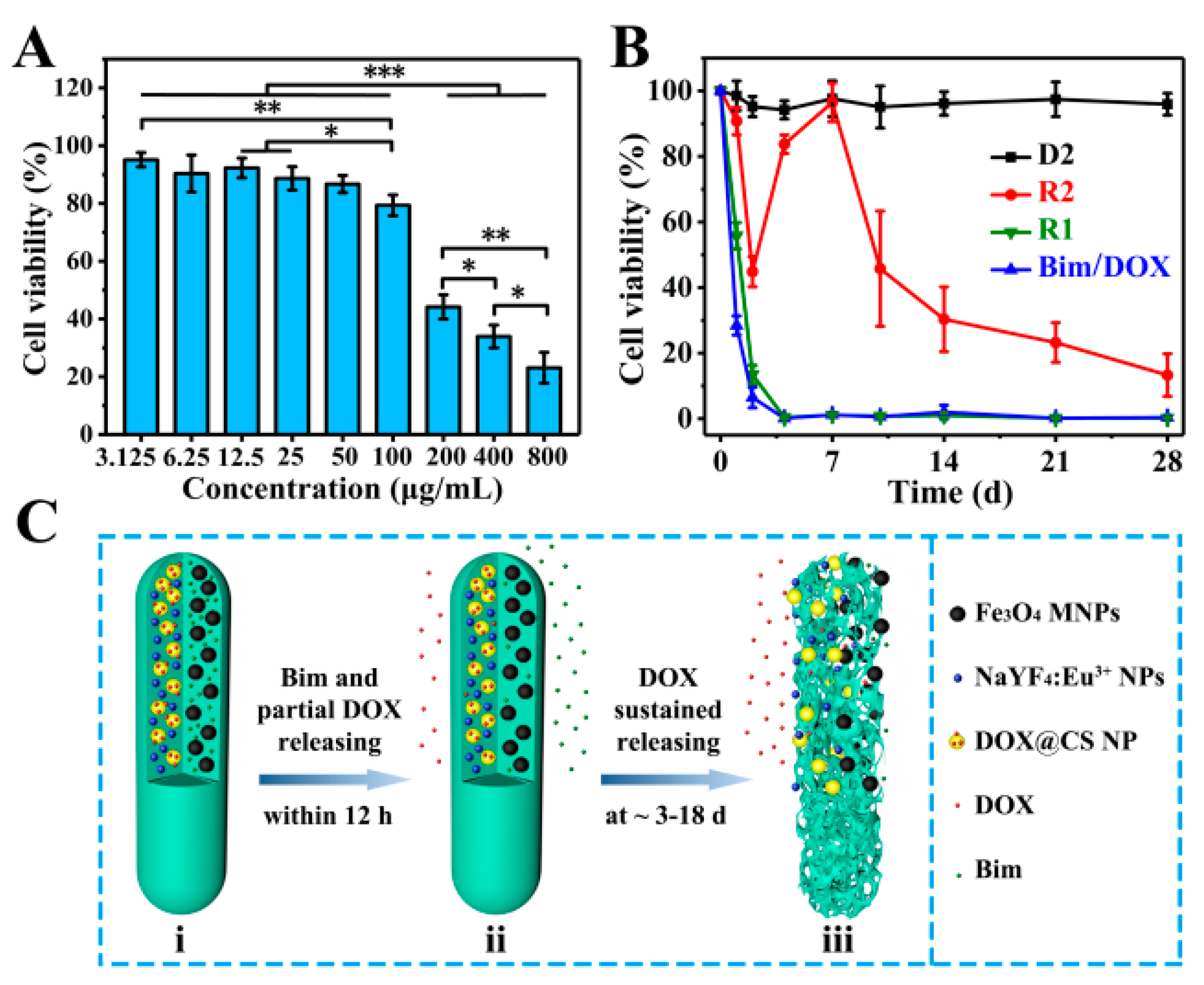
2.7.2. Cytotoxicity
2.7.3. Magnetic Responsiveness Assay
2.7.4. Cell Imaging Capability Assay
2.7.5. Drug Release for Killing A549 Cells
2.8. Characterizations
3. Results
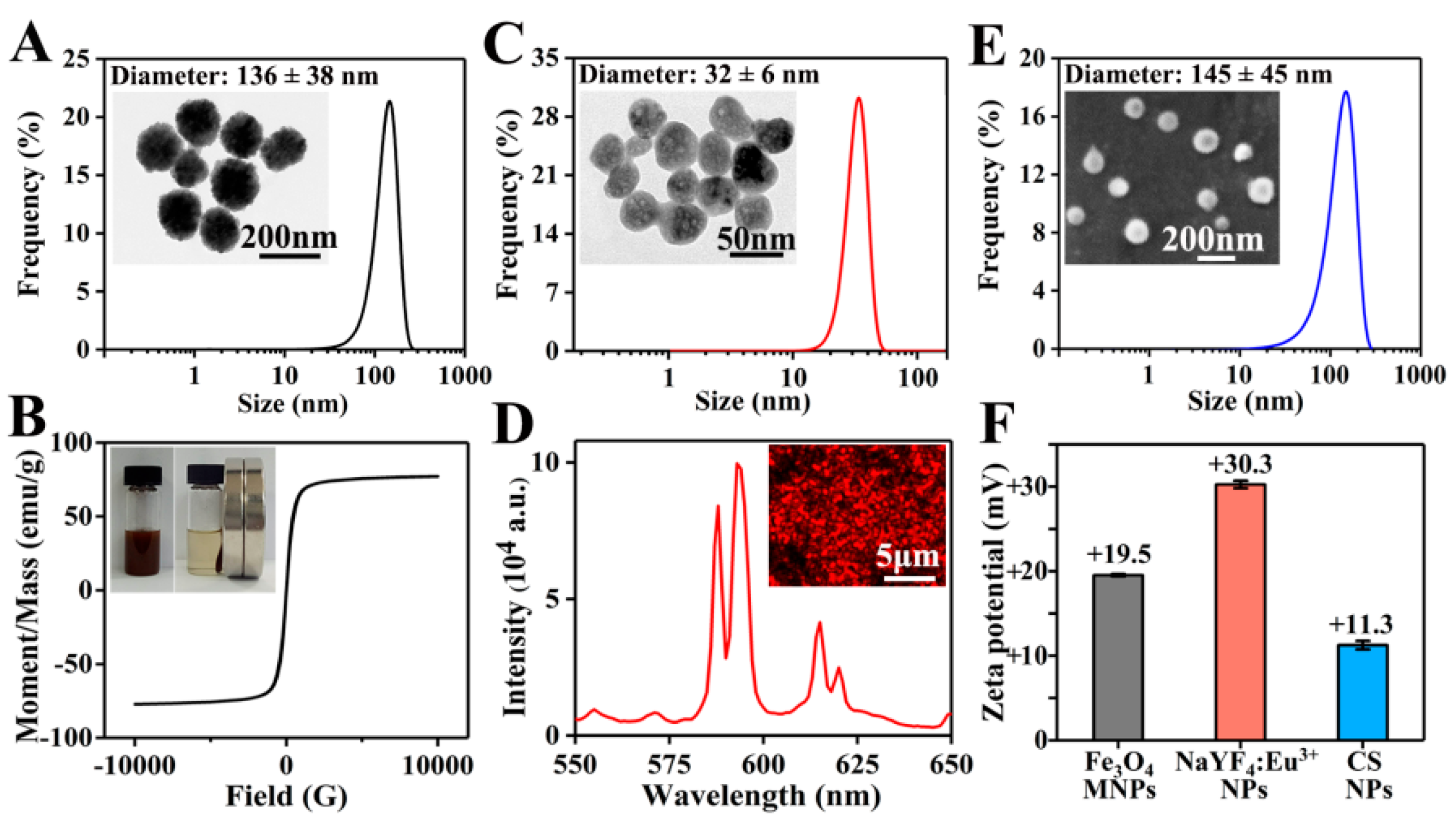
3.1. Preparation of NPs
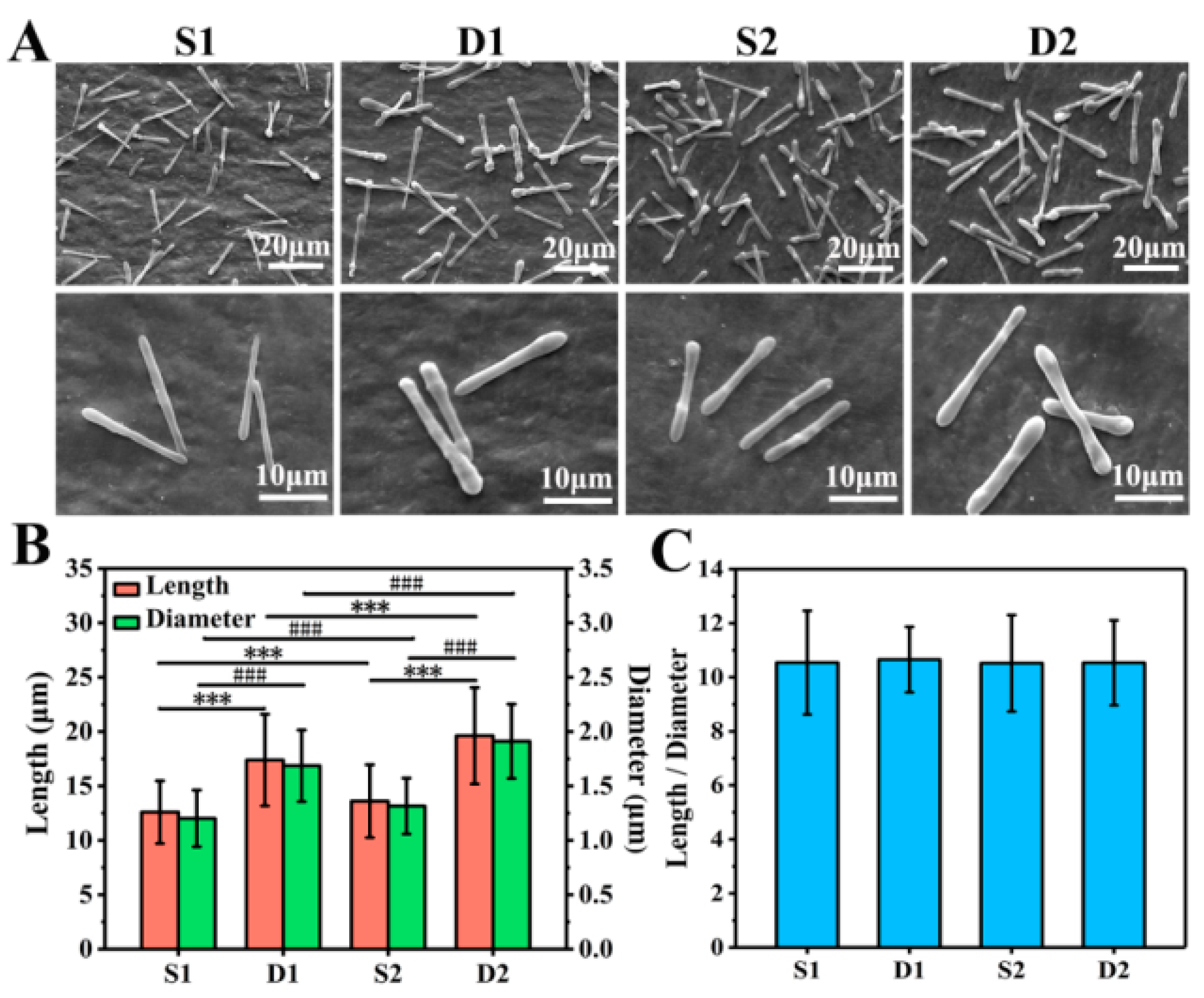
3.2. Morphological Observation
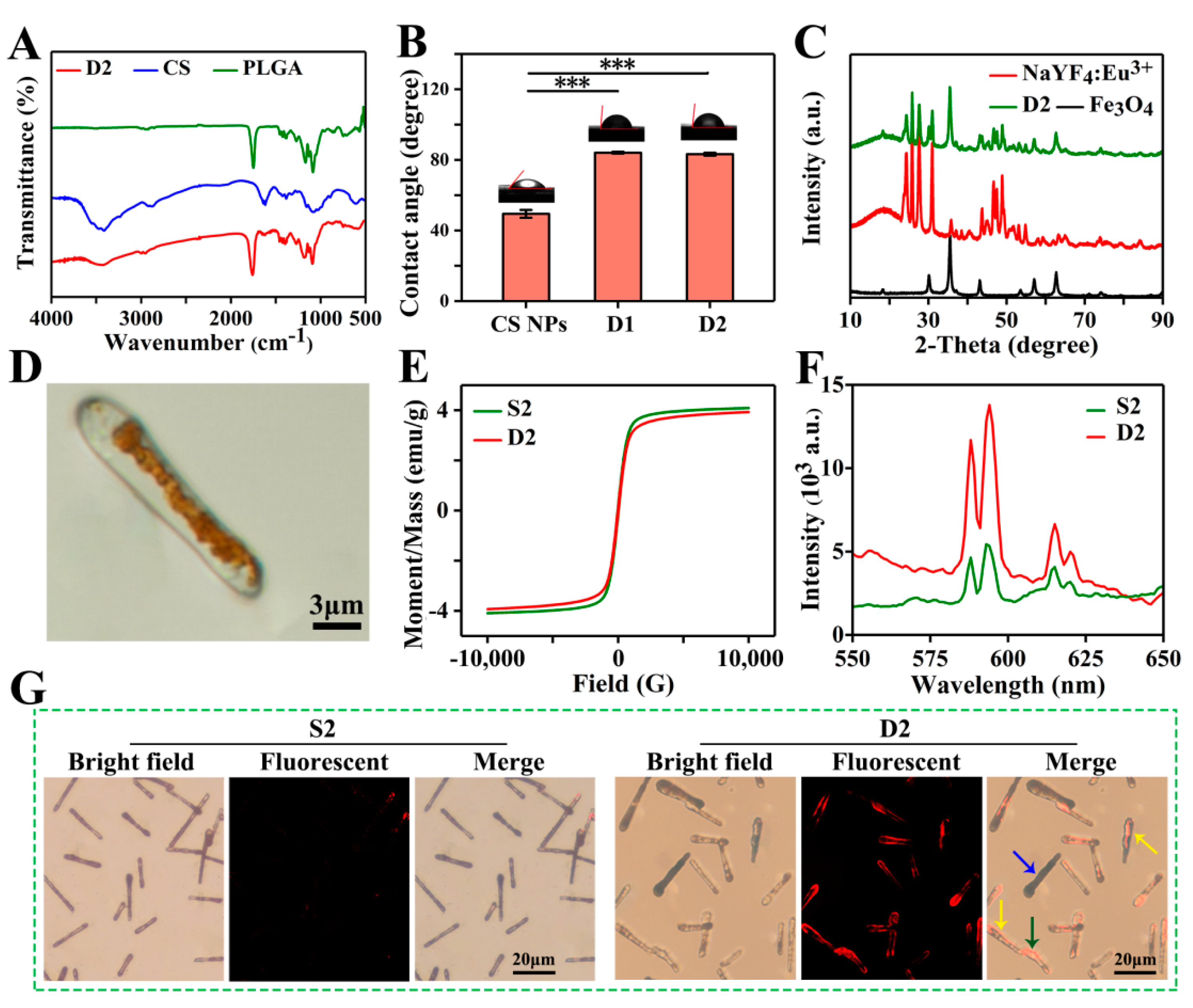
3.3. Physicochemical Characterizations
3.4. Biocompatible Evaluation
3.5. Drug Release Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kohane, D.S.; Langer, R. Biocompatibility and drug delivery systems. Chem. Sci. 2010, 1, 441–446. [Google Scholar] [CrossRef]
- Davoodi, P.; Lee, L.Y.; Xu, Q.; Sunil, V.; Sun, Y.; Soh, S.; Wang, C.H. Drug delivery systems for programmed and on-demand release. Adv. Drug. Deliver. Rev. 2018, 132, 104–138. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Bertleff-Zieschang, N.; Braunger, J.A.; Bjornmalm, M.; Cortez-Jugo, C.; Caruso, F. Particle Targeting in Complex Biological Media. Adv. Healthc. Mater. 2018, 7, 1700575. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Wu, J.; Lin, D.G. Construction of intelligent nano-drug delivery system for targeting extranodal nasal natural killer/thymus dependent lymphocyte. J. Biomed. Nanotechnol. 2021, 17, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Guan, X.; Zhong, S.; Chen, J.; Zhu, H.; Li, Z.; Xu, F.; Chen, P.; Wang, H. Multi-stimuli responsive smart chitosan-based microcapsules for targeted drug delivery and triggered drug release. Ultrason. Sonochem. 2017, 38, 145–153. [Google Scholar] [CrossRef]
- Zhao, T.; Xin, J.; Jiang, Y.; Zhao, Y.; Zhao, Y.; Zhao, Y.; Sun, W.; Yang, B.; Lin, Q. A novel strategy to synthesize dual blue fluorescent-magnetic EuCl2 nanocrystals via one-pot method with controlled morphologies using urea. Part. Part. Syst. Char. 2018, 35, 1800106. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, Y.; Ma, J.; Li, Q.; Li, Y.; Zhou, X.; Zhao, D.; Song, H.; Chen, Q.; Zhu, X. Light/magnetic hyperthermia triggered drug released from multi-functional thermo-sensitive magnetoliposomes for precise cancer synergetic theranostics. J. Control. Release 2018, 272, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, K.; Niu, X.; Fan, Y. Electrospraying magnetic-fluorescent bifunctional Janus PLGA microspheres with dual rare earth ions fluorescent-labeling drugs. RSC Adv. 2016, 6, 99034–99043. [Google Scholar] [CrossRef]
- Peng, H.; He, Y.; Tian, X.; Wen, J.; Zhang, L. LSPR effects in a magnetic–luminescent heterostructure for efficient enhanced luminescence performance. New J. Chem. 2019, 43, 304–311. [Google Scholar] [CrossRef]
- Yang, P.; Quan, Z.; Hou, Z.; Li, C.; Kang, X.; Cheng, Z.; Lin, J. A magnetic, luminescent and mesoporous core-shell structured composite material as drug carrier. Biomaterials 2009, 30, 4786–4795. [Google Scholar] [CrossRef]
- Sakamoto, N.; Hirai, Y.; Onodera, T.; Dezawa, T.; Shibata, Y.; Kasai, H.; Oikawa, H.; Yabu, H. Enhanced fluorescence emission and magnetic alignment control of biphasic functionalized composite Janus particles. Part. Part. Syst. Char. 2019, 36, 1800311. [Google Scholar] [CrossRef]
- Wang, L.; Dong, X.; Gai, G.; Zhao, L.; Xu, S.; Xiao, X. One-pot facile electrospinning construct of flexible Janus nanofibers with tunable and enhanced magnetism–photoluminescence bifunctionality. J. Nanopart. Res. 2015, 17, 91. [Google Scholar] [CrossRef]
- Li, P.; Xu, J.; Qi, B.; Li, K.; Gu, X.; Niu, X.; Fan, Y. One-step preparation of poly(lactide-co-glycolide) fiber rods embedding with luminescent materials as a drug delivery system via electrospray. Nanosci. Nanotech. Lett. 2018, 10, 1287–1291. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, J.F.; Dorn, H.C.; Zhang, C.M. Assembly of bio-nanoparticles for double controlled drug release. PLoS ONE 2013, 8, e74679. [Google Scholar] [CrossRef][Green Version]
- Yu, B.; Cong, H.; Peng, Q.; Gu, C.; Tang, Q.; Xu, X.; Tian, C.; Zhai, F. Current status and future developments in preparation and application of nonspherical polymer particles. Adv. Colloid. Interface. Sci. 2018, 256, 126–151. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Lu, W.; Qian, C.; Zeng, T.; Yin, L.; Wang, H.; Rao, L.; Liu, H.; Zeng, S. Urchin-like Ce/Tb co-doped GdPO4 hollow spheres for in vivo luminescence/X-ray bioimaging and drug delivery. Biomater. Sci. 2014, 2, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xiao, Y.; Huang, Y.; Xu, Y.; You, D.; Lu, W.; Yu, J. Rod-shaped micelles based on PHF-g-(PCL-PEG) with pH-triggered doxorubicin release and enhanced cellular uptake. Biomacromolecules 2019, 20, 1167–1177. [Google Scholar] [CrossRef]
- Xu, J.; Li, K.; Liu, M.; Gu, X.; Li, P.; Fan, Y. Studies on preparation and formation mechanism of poly(lactide-co-glycolide) microrods via one-step electrospray and an application for drug delivery system. Eur. Polym. J. 2021, 148, 110372. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, Z.F.; Peng, Y.L.; Wang, F.Y.; Deng, L. Gold nanomaterials as a promising integrated tool for diagnosis and treatment of pathogenic infections—A review. J. Biomed. Nanotechnol. 2021, 17, 744–770. [Google Scholar] [CrossRef]
- Moradi Kashkooli, F.; Soltani, M.; Souri, M.; Meaney, C.; Kohandel, M. Nexus between in silico and in vivo models to enhance clinical translation of nanomedicine. Nano Today 2021, 36, 101057. [Google Scholar] [CrossRef]
- Zhang, W.; Zheng, N.; Chen, L.; Xie, L.; Cui, M.; Li, S.; Xu, L. Effect of Shape on Mesoporous Silica Nanoparticles for Oral Delivery of Indomethacin. Pharmaceutics 2018, 11, 4. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.; Chen, M.; Luo, X.; Li, X. Shape effects of electrospun fiber rods on the tissue distribution and antitumor efficacy. J. Control. Release 2016, 244, 52–62. [Google Scholar] [CrossRef]
- Mehnath, S.; Arjama, M.; Rajan, M.; Annamalai, G.; Jeyaraj, M. Co-encapsulation of dual drug loaded in MLNPs: Implication on sustained drug release and effectively inducing apoptosis in oral carcinoma cells. Biomed. Pharmacother. 2018, 104, 661–671. [Google Scholar] [CrossRef]
- Pusuluri, A.; Krishnan, V.; Wu, D.; Shields, C.W.t.; Wang, L.W.; Mitragotri, S. Role of synergy and immunostimulation in design of chemotherapy combinations: An analysis of doxorubicin and camptothecin. Bioeng. Transl. Med. 2019, 4, e10129. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Wang, P.; Huang, Z.; He, D.; Zhu, W.; Liu, H.; Chen, Z.; Wang, W.; Li, Y.; Shen, J.; et al. Construction of surface-modified polydopamine nanoparticles for sequential drug release and combined chemo-photothermal cancer therapy. Mol. Pharm. 2021, 18, 1327–1343. [Google Scholar] [CrossRef]
- Khuroo, T.; Verma, D.; Khuroo, A.; Ali, A.; Iqbal, Z. Simultaneous delivery of paclitaxel and erlotinib from dual drug loaded PLGA nanoparticles: Formulation development, thorough optimization and in vitro release. J. Mol. Liq. 2018, 257, 52–68. [Google Scholar] [CrossRef]
- Prabhakar, A.K.; Potroz, M.G.; Park, S.; Miyako, E.; Cho, N.-J. Spatially controlled molecular encapsulation in natural pine pollen picrocapsules. Part. Part. Syst. Char. 2018, 35, 1800151. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, J.; Zuo, T.; Ma, S.; Xokrat, N.; Hu, Z.; Wang, Z.; Xu, R.; Wei, Y.; Shen, Q. Heparanase-driven sequential released nanoparticles for ferroptosis and tumor microenvironment modulations synergism in breast cancer therapy. Biomaterials 2021, 266, 120429. [Google Scholar] [CrossRef] [PubMed]
- Moradi Kashkooli, F.; Soltani, M.; Souri, M. Controlled anti-cancer drug release through advanced nano-drug delivery systems: Static and dynamic targeting strategies. J. Control. Release 2020, 327, 316–349. [Google Scholar] [CrossRef]
- Zhou, F.; Gao, J.; Xu, Z.; Li, T.; Gao, A.; Sun, F.; Wang, F.; Wang, W.; Geng, Y.; Zhang, F.; et al. Overcoming immune resistance by sequential prodrug nanovesicles for promoting chemoimmunotherapy of cancer. Nano Today 2021, 36, 101025. [Google Scholar] [CrossRef]
- Song, W.; Xiao, Y. Sequential drug delivery of vancomycin and rhBMP-2 via pore-closed PLGA microparticles embedded photo-crosslinked chitosan hydrogel for enhanced osteointegration. Int. J. Biol. Macromol. 2021, 182, 612–625. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Wang, B.; Cao, Y.; Yu, Q.; Yin, T. Fabrication of core–shell micro/nanoparticles for programmable dual drug release by emulsion electrospraying. J. Nanopart. Res. 2013, 15, 1726. [Google Scholar] [CrossRef]
- He, D.; Wang, S.; Lei, L.; Hou, Z.; Shang, P.; He, X.; Nie, H. Core–shell particles for controllable release of drug. Chem. Eng. Sci. 2015, 125, 108–120. [Google Scholar] [CrossRef]
- Jahns, M.; Warwas, D.P.; Krey, M.R.; Nolte, K.; Konig, S.; Froba, M.; Behrens, P. Nanoporous hybrid core-shell nanoparticles for sequential release. J. Mater. Chem. B 2020, 8, 776–786. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, W.; Li, D.; Liu, T.; Zhang, Y.S.; Ding, J.; Chen, X. Locally Deployable Nanofiber Patch for Sequential Drug Delivery in Treatment of Primary and Advanced Orthotopic Hepatomas. ACS Nano 2018, 12, 6685–6699. [Google Scholar] [CrossRef] [PubMed]
- Nie, H.; Fu, Y.; Wang, C.H. Paclitaxel and suramin-loaded core/shell microspheres in the treatment of brain tumors. Biomaterials 2010, 31, 8732–8740. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Bu, L.L.; Xu, J.H.; Cai, B.; Yu, G.T.; Yu, X.; He, Z.; Huang, Q.; Li, A.; Guo, S.S.; et al. Red blood cell membrane as a biomimetic nanocoating for prolonged circulation time and reduced accelerated blood clearance. Small 2015, 11, 6225–6236. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.K.; Weng, X.X.; Huang, H.; Tao, X.Y. Enhanced luminescence properties of hexagonal-phase NaYF4: Eu3+ microrods by annealing treatment. Adv. Mater. Res. 2011, 335–336, 1009–1013. [Google Scholar] [CrossRef]
- Liang, X.; Hu, Y.; Ma, X.; Wang, Y.; Fan, J.; Hu, X. A novel template-free and one-step method to fabricate hollow mesoporous structured upconversion luminescent NaYF4: Yb3+, Er3+ nanoparticles. Mater. Let. 2014, 129, 107–110. [Google Scholar] [CrossRef]
- Pathak, T.K.; Kumar, A.; Erasmus, L.B.; Pandey, A.; Coetsee, E.; Swart, H.C.; Kroon, R.E. Highly efficient infrared to visible up-conversion emission tuning from red to white in Eu/Yb co-doped NaYF4 phosphor. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2019, 207, 23–30. [Google Scholar] [CrossRef]
- Min, B.-M.; Lee, S.W.; Lim, J.N.; You, Y.; Lee, T.S.; Kang, P.H.; Park, W.H. Chitin and chitosan nanofibers: Electrospinning of chitin and deacetylation of chitin nanofibers. Polymer 2004, 45, 7137–7142. [Google Scholar] [CrossRef]
- Wang, Z.; Hu, W.; Du, Y.; Xiao, Y.; Wang, X.; Zhang, S.; Wang, J.; Mao, C. Green gas-mediated cross-linking generates biomolecular hydrogels with enhanced strength and excellent hemostasis for wound healing. ACS Appl. Mater. Interfaces 2020, 12, 13622–13633. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Han, N.; Zhao, Q.; Bai, L.; Li, J.; Jiang, T.; Wang, S. Redox-responsive mesoporous silica as carriers for controlled drug delivery: A comparative study based on silica and PEG gatekeepers. Eur. J. Pharm. Sci. 2015, 72, 12–20. [Google Scholar] [CrossRef] [PubMed]
| DDC | Precursor Solution | Fe3O4 MNPs (mg) | NaYF4:Eu3+ NPs (mg) | CS NPs (mg) | PLGA (mg) | THF (mL) |
|---|---|---|---|---|---|---|
| S1 | - | 50 | 50 | - | 150 | 5.0 |
| D1 | A | - | 50 | - | 75 | 2.5 |
| B | 50 | - | - | 75 | 2.5 | |
| S2 | - | 50 | 50 | 25 | 150 | 5.0 |
| D2 | A | - | 50 | 25 | 75 | 2.5 |
| B | 50 | - | - | 75 | 2.5 |
| DDC | Precursor Solution | Fe3O4 MNPs (mg) | NaYF4:Eu3+ NPs (mg) | Drug (Bim) | Drug (DOX) | PLGA (mg) | THF (mL) |
|---|---|---|---|---|---|---|---|
| R1 | A | - | 50 | - | DOX: 5mg, CS NPs: 25mg | 75 | 2.5 |
| B | 50 | - | 10 mg | - | 75 | 2.5 | |
| R2 | A | - | 50 | - | DOX@CS NPs: 30 mg | 75 | 2.5 |
| B | 50 | - | 10 mg | - | 75 | 2.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Jia, Y.; Liu, M.; Gu, X.; Li, P.; Fan, Y. Preparation of Magnetic–Luminescent Bifunctional Rapeseed Pod-Like Drug Delivery System for Sequential Release of Dual Drugs. Pharmaceutics 2021, 13, 1116. https://doi.org/10.3390/pharmaceutics13081116
Xu J, Jia Y, Liu M, Gu X, Li P, Fan Y. Preparation of Magnetic–Luminescent Bifunctional Rapeseed Pod-Like Drug Delivery System for Sequential Release of Dual Drugs. Pharmaceutics. 2021; 13(8):1116. https://doi.org/10.3390/pharmaceutics13081116
Chicago/Turabian StyleXu, Junwei, Yunxue Jia, Meili Liu, Xuenan Gu, Ping Li, and Yubo Fan. 2021. "Preparation of Magnetic–Luminescent Bifunctional Rapeseed Pod-Like Drug Delivery System for Sequential Release of Dual Drugs" Pharmaceutics 13, no. 8: 1116. https://doi.org/10.3390/pharmaceutics13081116
APA StyleXu, J., Jia, Y., Liu, M., Gu, X., Li, P., & Fan, Y. (2021). Preparation of Magnetic–Luminescent Bifunctional Rapeseed Pod-Like Drug Delivery System for Sequential Release of Dual Drugs. Pharmaceutics, 13(8), 1116. https://doi.org/10.3390/pharmaceutics13081116









