Accuracy of Dose Administered to Children Using Off-Labelled or Unlicensed Oral Dosage Forms
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
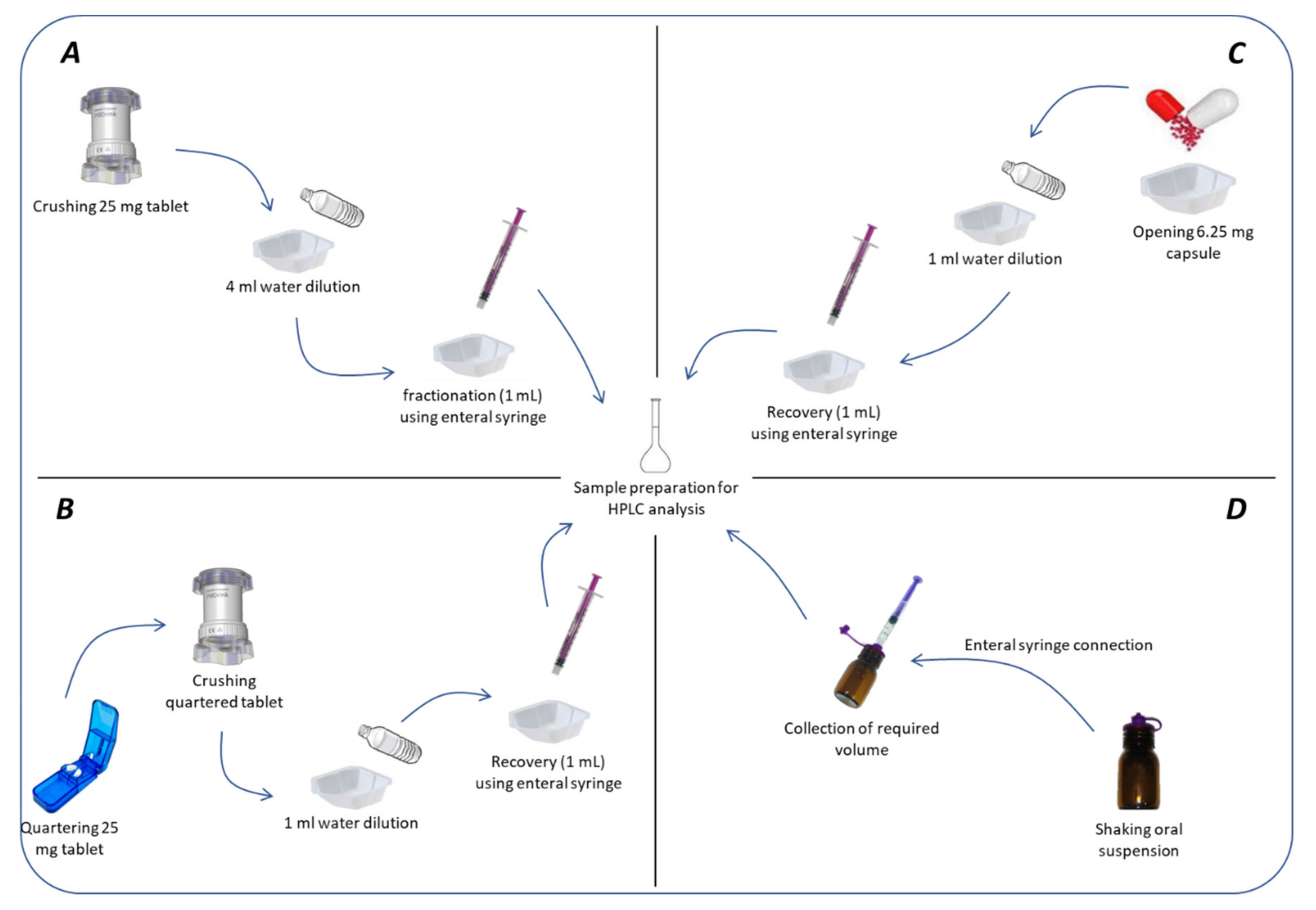
2.1.1. Dosage Forms
2.1.2. Mimicking Administration Modalities
2.1.3. Drug Content Determination
2.2. Statistical Analysis
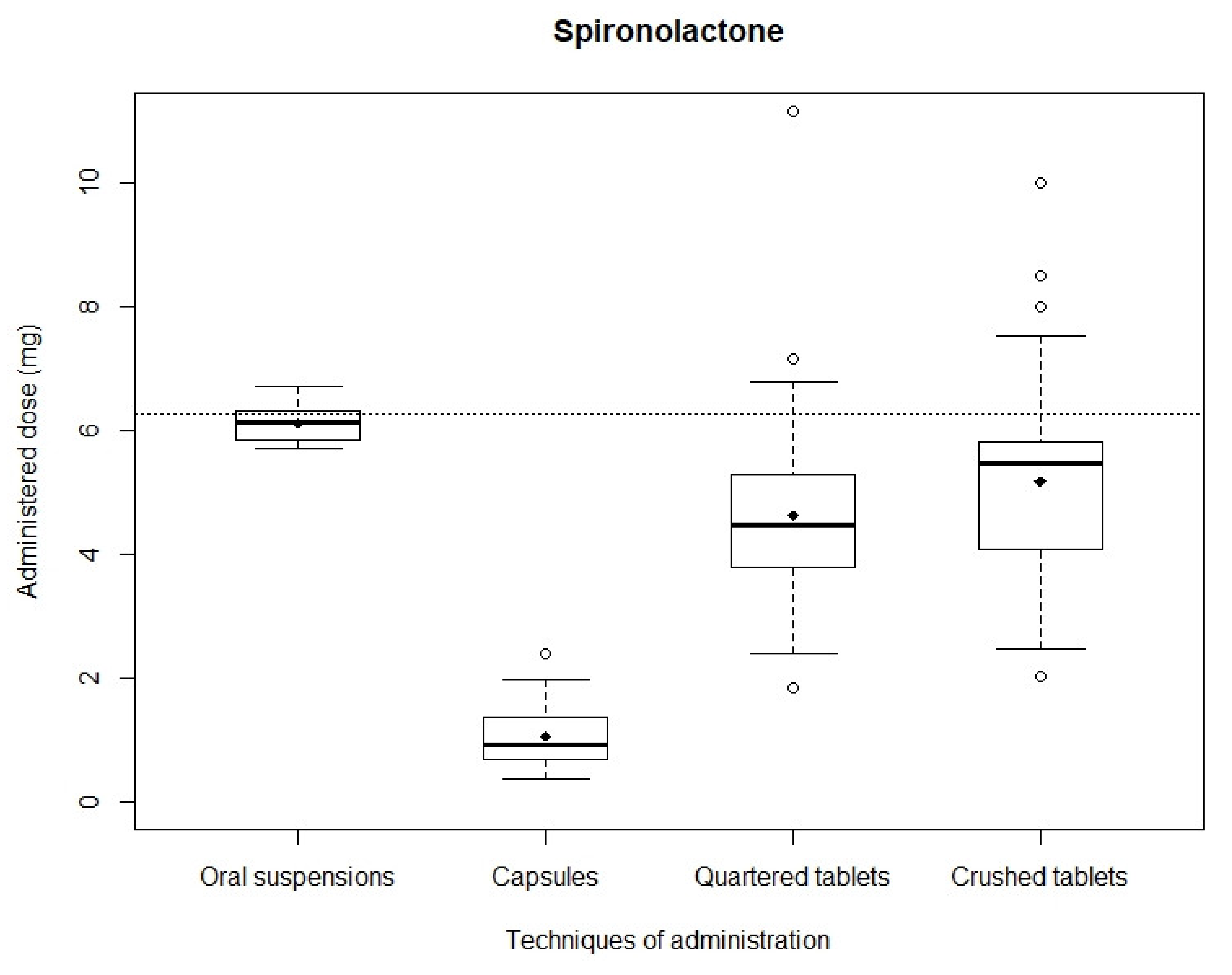
3. Results
3.1. Quality of Spironolactone and Hydrochlorothiazide Compounded Forms
3.2. Accuracy of Administered Doses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Conroy, S.; Choonara, I.; Impicciatore, P.; Mohn, A.; Arnell, H.; Rane, A.; Knoeppel, C.; Seyberth, H.; Pandolfini, C.; Raffaelli, M.P.; et al. Survey of Unlicensed and off Label Drug Use in Paediatric Wards in European Countries. Br. Med. J. 2000, 320, 79–82. [Google Scholar] [CrossRef] [Green Version]
- Mistry, P.; Batchelor, H. Evidence of Acceptability of Oral Paediatric Medicines: A Review. J. Pharm. Pharmacol. 2017, 69, 361–376. [Google Scholar] [CrossRef] [Green Version]
- Conroy, S. Paediatric Pharmacy—Drug Therapy. Hosp. Pharm. 2003, 10, 49–57. [Google Scholar]
- Czyzewski, D.I.; Runyan, R.D.; Lopez, M.A.; Calles, N.R. Teaching and Maintaining Pill Swallowing in HIV-Infected Children. AIDS Read. 2000, 10, 88–95. [Google Scholar]
- Klingmann, V.; Seitz, A.; Meissner, T.; Breitkreutz, J.; Moeltner, A.; Bosse, H.M. Acceptability of Uncoated Mini-Tablets in Neonates—A Randomized Controlled Trial. J. Pediatrics 2015, 167, 893–896.e2. [Google Scholar] [CrossRef]
- European Commission. State of Paediatric Medicines in the EU-10 Years of the EU Paediatric Regulation; European Commission: Brussels, Belgium, 2017. [Google Scholar]
- Nahata, M.C. Lack of Pediatric Drug Formulations. Pediatrics 1999, 104, 607–609. [Google Scholar] [PubMed]
- Kayitare, E.; Vervaet, C.; Ntawukulilyayo, J.D.; Seminega, B.; Bortel, V.; Remon, J.P. Development of Fixed Dose Combination Tablets Containing Zidovudine and Lamivudine for Paediatric Applications. Int. J. Pharm. 2009, 370, 41–46. [Google Scholar] [CrossRef]
- Standing, J.F.; Tuleu, C. Paediatric Formulations—Getting to the Heart of the Problem. Int. J. Pharm. 2005, 300, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Richey, R.H.; Shah, U.U.; Peak, M.; Craig, J.V.; Ford, J.L.; Barker, C.E.; Nunn, A.J.; Turner, M.A. Manipulation of Drugs to Achieve the Required Dose Is Intrinsic to Paediatric Practice but Is not Supported by Guidelines or Evidence. BMC Pediatrics 2013, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watson, C.; Webb, E.A.; Kerr, S.; Davies, J.H.; Stirling, H.; Batchelor, H. How Close Is the Dose? Manipulation of 10 Mg Hydrocortisone Tablets to Provide Appropriate Doses to Children. Int. J. Pharm. 2018, 545, 57–63. [Google Scholar] [CrossRef] [Green Version]
- Verrue, C.; Mehuys, E.; Boussery, K.; Remon, J.P.; Petrovic, M. Tablet-Splitting: A Common yet not so Innocent Practice. J. Adv. Nurs. 2011, 67, 26–32. [Google Scholar] [CrossRef]
- Madathilethu, J.; Roberts, M.; Peak, M.; Blair, J.; Prescott, R.; Ford, J.L. Content Uniformity of Quartered Hydrocortisone Tablets in Comparison with Mini-Tablets for Paediatric Dosing. BMJ Paediatr. Open 2018, 2, e000198. [Google Scholar] [CrossRef]
- Bjerknes, K.; Bøyum, S.; Kristensen, S.; Brustugun, J.; Wang, S. Manipulating Tablets and Capsules given to Hospitalised Children in Norway Is Common Practice. Acta Paediatr. Int. J. Paediatr. 2017, 106, 503–508. [Google Scholar] [CrossRef]
- Preis, M. Orally Disintegrating Films and Mini-Tablets—Innovative Dosage Forms of Choice for Pediatric Use. AAPS PharmSciTech 2015, 16, 234–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaz, V.M.; Kumar, L. 3D Printing as a Promising Tool in Personalized Medicine. AAPS PharmSciTech 2021, 22, 49. [Google Scholar] [CrossRef] [PubMed]
- Brion, F.; Nunn, A.; Rieutord, A. Extemporaneous (Magistral) Preparation of Oral Medicines for Children in European Hospitals. Acta Paediatr. 2007, 92, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Cuzzolin, L.; Atzei, A.; Fanos, V. Off-Label and Unlicensed Prescribing for Newborns and Children in Different Settings: A Review of the Literature and a Consideration about Drug Safety. Expert Opin. Drug Saf. 2006, 5, 703–718. [Google Scholar] [CrossRef]
- Bellis, J.R.; Kirkham, J.J.; Thiesen, S.; Conroy, E.J.; Bracken, L.E.; Mannix, H.L.; Bird, K.A.; Duncan, J.C.; Peak, M.; Turner, M.A.; et al. Adverse Drug Reactions and Off-Label and Unlicensed Medicines in Children: A Nested Case? Control Study of Inpatients in a Pediatric Hospital. BMC Med. 2013, 11, 238. [Google Scholar] [CrossRef] [Green Version]
- Segar, J.L. Neonatal Diuretic Therapy: Furosemide, Thiazides, and Spironolactone. Clin. Perinatol. 2012, 39, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Van der Vorst, M.M.J.; Kist, J.E.; Van der Heijden, A.J.; Burggraaf, J. Diuretics in Pediatrics. Pediatr. Drugs 2006, 8, 245–264. [Google Scholar] [CrossRef] [PubMed]
- Roulet, L.; Maillard, N.; Dupuis, A. Bonnes Pratiques de Préparations: Le Projet de l’Afssaps. Actual. Pharm. Hosp. 2007, 3, 39–43. [Google Scholar] [CrossRef]
- European Pharmacopoeia Commission. 2.9.5. Uniformity of Mass of Single-Dose Preparations. In European Pharmacopoeia 9.2; Council Of Europe: Strasbourg, France, 2017. [Google Scholar]
- European Pharmacopoeia Commission. 2.9.40. Uniformity of Dosage Units. In European Pharmacopoeia 9.2; Council Of Europe: Strasbourg, France, 2017. [Google Scholar]
- Binson, G.; Beuzit, K.; Migeot, V.; Marco, L.; Troussier, B.; Venisse, N.; Dupuis, A. Preparation and Physicochemical Stability of Liquid Oral Dosage Forms Free of Potentially Harmful Excipient Designed for Pediatric Patients. Pharmaceutics 2019, 11, 190. [Google Scholar] [CrossRef] [Green Version]
- Allen, L.V.; Bassani, G.S.; Elder, E.J.; Parr, A.F. Strength and Stability Testing for Compounded Preparations; US Pharmacopeia: Rockville, MD, USA, 2014; pp. 1–7. [Google Scholar]
- Richey, R.H.; Hughes, C.; Craig, J.V.; Shah, U.U.; Ford, J.L.; Barker, C.E.; Peak, M.; Nunn, A.J.; Turner, M.A. A Systematic Review of the Use of Dosage Form Manipulation to Obtain Required Doses to Inform Use of Manipulation in Paediatric Practice. Int. J. Pharm. 2017, 518, 155–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walch, A.C.; Henin, E.; Berthiller, J.; Dode, X.; Abel, B.; Kassai, B.; Lajoinie, A. Oral Dosage Form Administration Practice in Children under 6 Years of Age: A Survey Study of Paediatric Nurses. Int. J. Pharm. 2016, 511, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.; Ranmal, S.R.; Ernest, T.B.; Liu, F. Patient Acceptability, Safety and Access: A Balancing Act for Selecting Age-Appropriate Oral Dosage Forms for Paediatric and Geriatric Populations. Int. J. Pharm. 2018, 536, 547–562. [Google Scholar] [CrossRef]
- ICH. Validation of Analytical Procedures: Text and Methodology Q2(R1). International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use 2005. Available online: http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q2_R1/Step4/Q2_R1__Guideline.pdf (accessed on 27 August 2019).
- The Five Rights: A Destination without a Map. Available online: https://www.ismp.org/resources/five-rights-destination-without-map (accessed on 27 August 2019).
- Yalkowsky, S.H.; He, Y.; Jain, P. Handbook of Aqueous Solubility Data; CRC Press: Boca Raton, FL, USA, 2010; ISBN 978-85-7811-079-6. [Google Scholar]
- Rowe, R.C. (Ed.) Handbook of Pharmaceutical Excipients, 6th ed.; APhA, (PhP) Pharmaceutical Press: London, UK, 2009; ISBN 978-0-85369-792-3. [Google Scholar]
- Tuleu, C.; Grangé, J.; Seurin, S. The Need for Pædiatric Formulation: Oral Administration of Nifedipine in Children, a Proof of Concept. J. Drug Deliv. Sci. Technol. 2005, 15, 319–324. [Google Scholar] [CrossRef]
- 5.1.4. Microbiological Quality of Non-Sterile Products for Pharmaceutical Use. Eur. Pharm. 2017, 2, 507.

| Batch 1 | Batch 2 | Batch 3 | Mean | |
|---|---|---|---|---|
| Hydrochlorothiazide | 5.91 ± 0.51 | 5.85 ± 0.57 | 6.11 ± 0.28 | 5.93 ± 0.44 |
| Spironolactone | 6.35 ± 0.45 | 6.12 ± 0.51 | 5.75 ± 0.46 | 6.07 ± 0.47 |
| Batch 1 | Batch 2 | Batch 3 | Mean | |
|---|---|---|---|---|
| Hydrochlorothiazide | 2.02 ± 0.04 | 1.96 ± 0,03 | 1.97 ± 0.08 | 1.98 ± 0.06 |
| Spironolactone | 5.10 ± 0.04 | 5.35 ± 0.13 | 4.74 ± 0.13 | 5.06 ± 0.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binson, G.; Sanchez, C.; Waton, K.; Chanat, A.; Di Maio, M.; Beuzit, K.; Dupuis, A. Accuracy of Dose Administered to Children Using Off-Labelled or Unlicensed Oral Dosage Forms. Pharmaceutics 2021, 13, 1014. https://doi.org/10.3390/pharmaceutics13071014
Binson G, Sanchez C, Waton K, Chanat A, Di Maio M, Beuzit K, Dupuis A. Accuracy of Dose Administered to Children Using Off-Labelled or Unlicensed Oral Dosage Forms. Pharmaceutics. 2021; 13(7):1014. https://doi.org/10.3390/pharmaceutics13071014
Chicago/Turabian StyleBinson, Guillaume, Cécile Sanchez, Karen Waton, Adeline Chanat, Massimo Di Maio, Karine Beuzit, and Antoine Dupuis. 2021. "Accuracy of Dose Administered to Children Using Off-Labelled or Unlicensed Oral Dosage Forms" Pharmaceutics 13, no. 7: 1014. https://doi.org/10.3390/pharmaceutics13071014
APA StyleBinson, G., Sanchez, C., Waton, K., Chanat, A., Di Maio, M., Beuzit, K., & Dupuis, A. (2021). Accuracy of Dose Administered to Children Using Off-Labelled or Unlicensed Oral Dosage Forms. Pharmaceutics, 13(7), 1014. https://doi.org/10.3390/pharmaceutics13071014






