Simultaneous Visualization of 161Tb- and 177Lu-Labeled Somatostatin Analogues Using Dual-Isotope SPECT Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Terbium-161 and Lutetium-177
2.2. Preparation and In Vitro Evaluation of the Radiopeptides
2.3. Cell Culture
2.4. In Vitro Studies
2.5. In Vivo Studies
2.6. Biodistribution Studies
2.7. Dual-Isotope SPECT/CT Imaging Studies
2.8. Statistical Analysis
3. Results and Discussion
3.1. Equal Labeling Conditions Can Be Applied for Terbium-161 and Lutetium-177
3.2. Radiolytic Stability of 161Tb- and 177Lu-Labeled SST Analogues Is Comparable
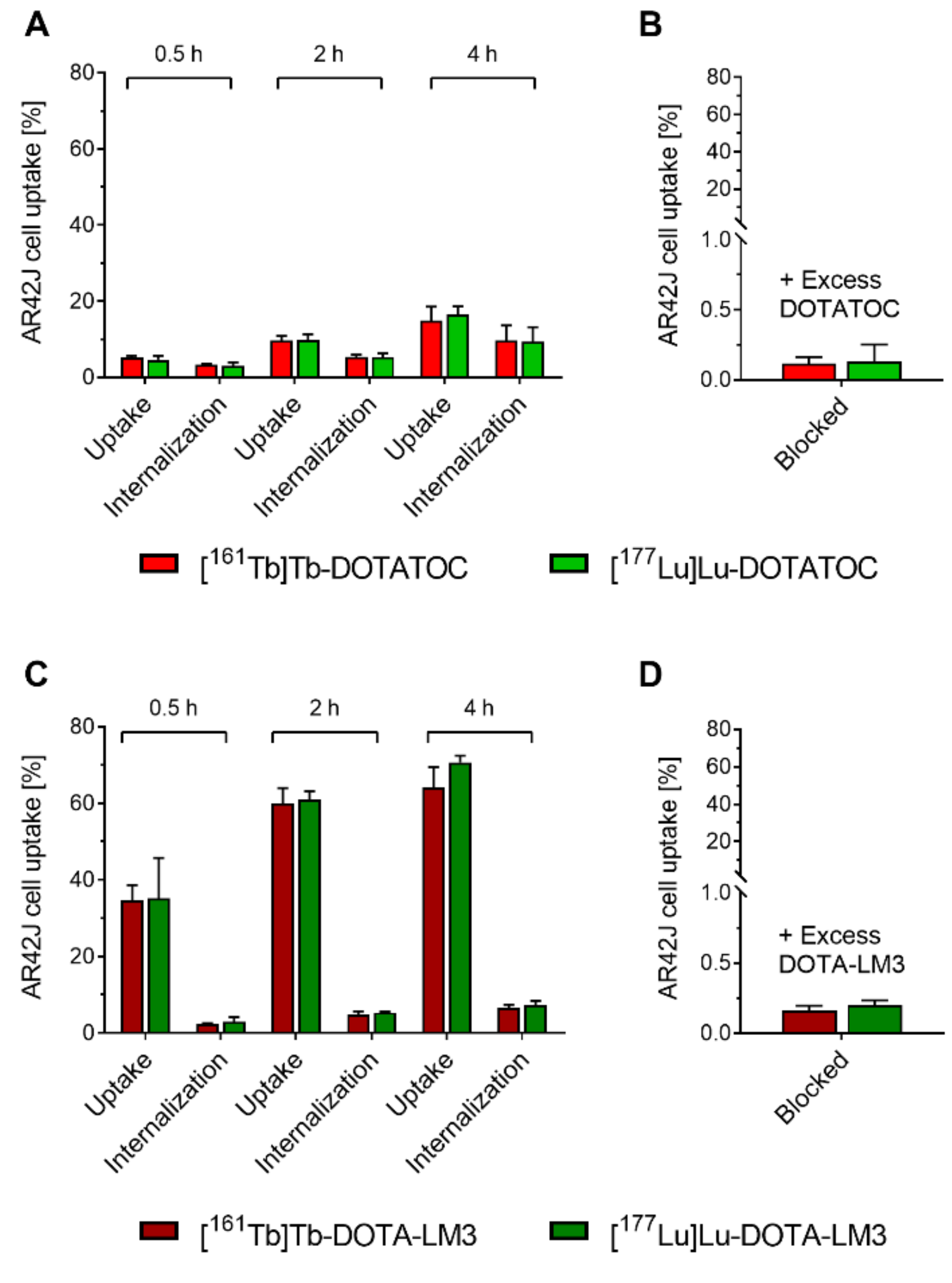
3.3. The In Vitro Properties of 161Tb- and 177Lu-Labeled Peptides Are Equal
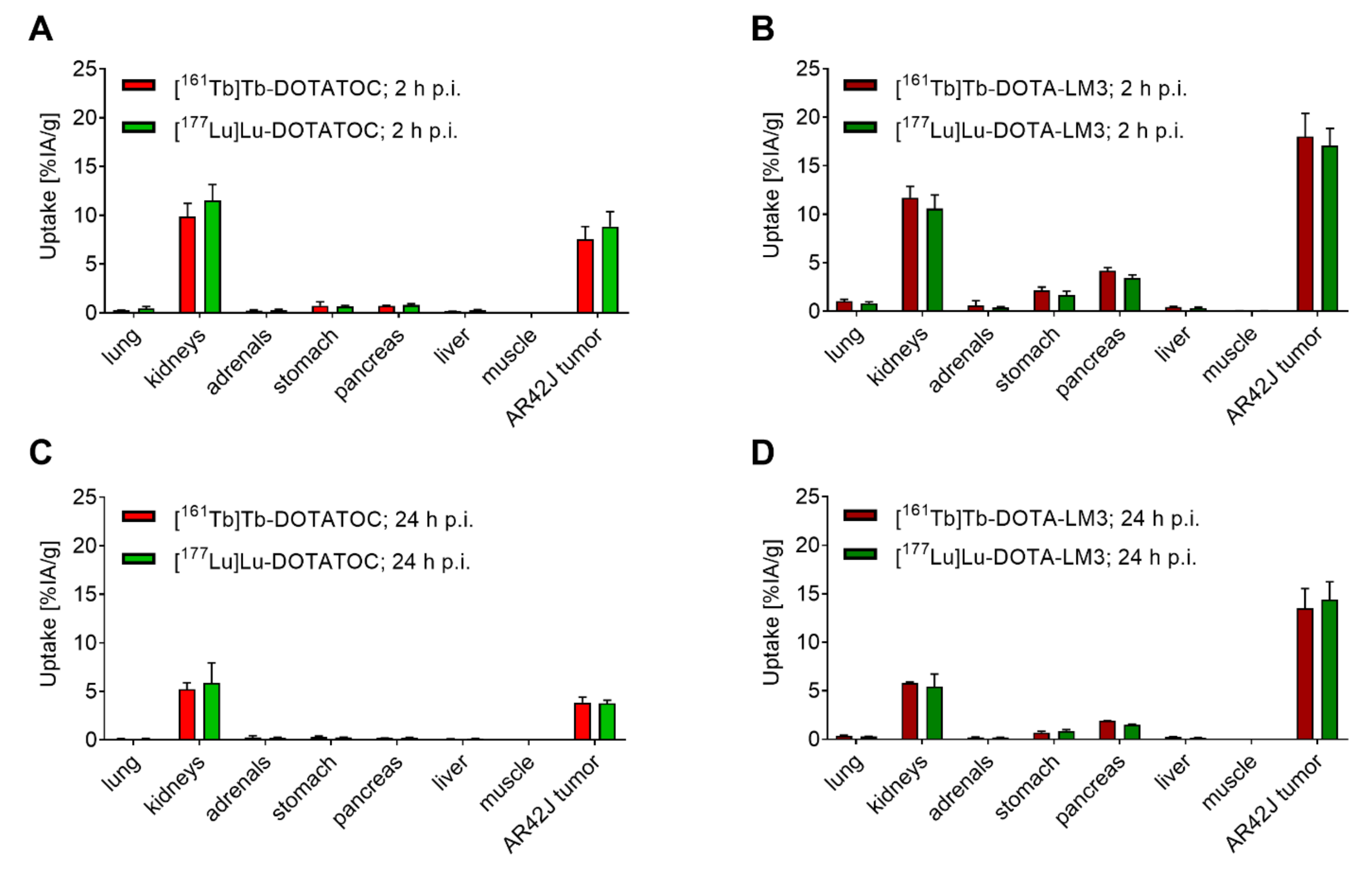
3.4. Terbium-161 Does Not Change the Biodistribution of the SST Analogues Otherwise Labeled with Lutetium-177
3.5. Variable Peptides (Agonist vs. Antagonist) Have Different Tissue Distribution Profiles
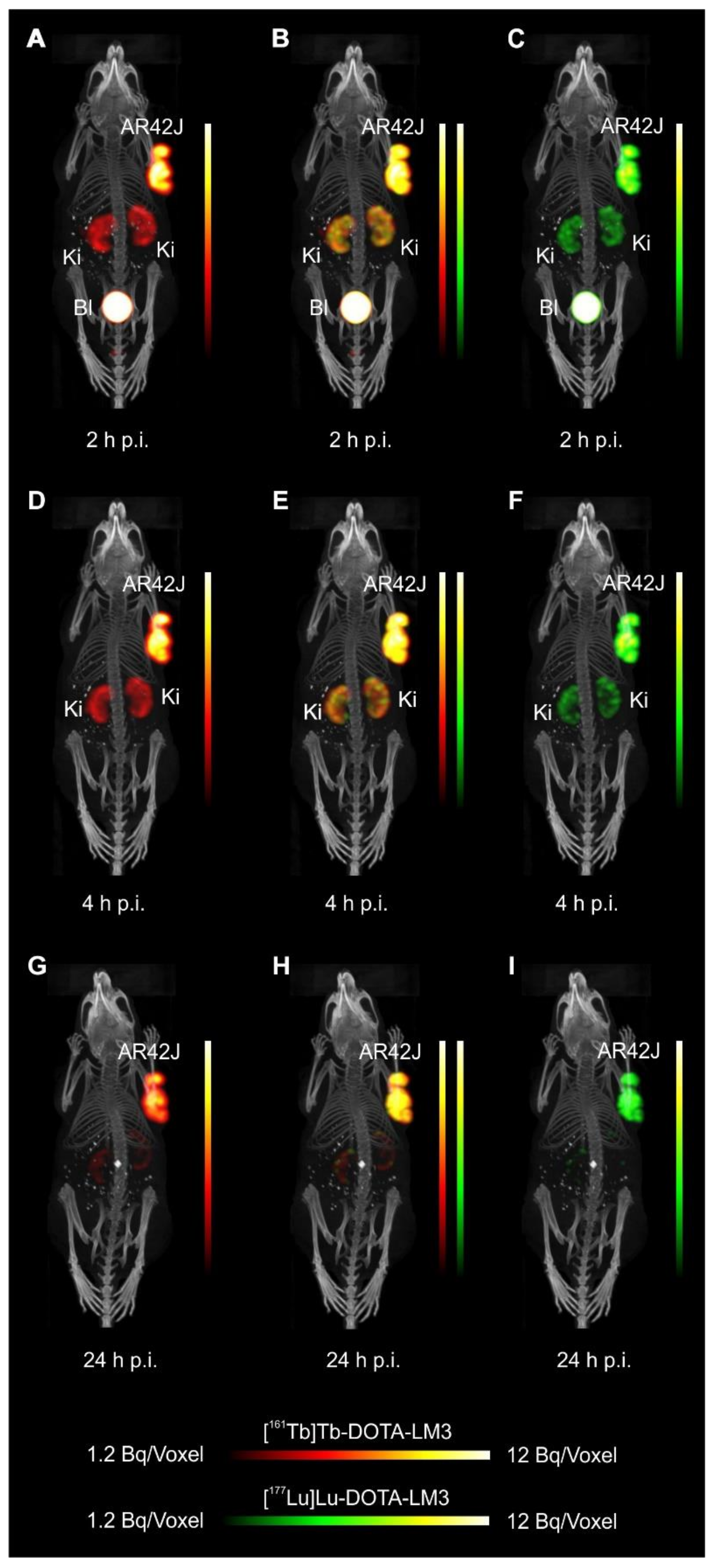
3.6. Simultaneous In Vivo Imaging Demonstrates Equal Biodistribution of 161Tb- and 177Lu-Labeled SST Analogues
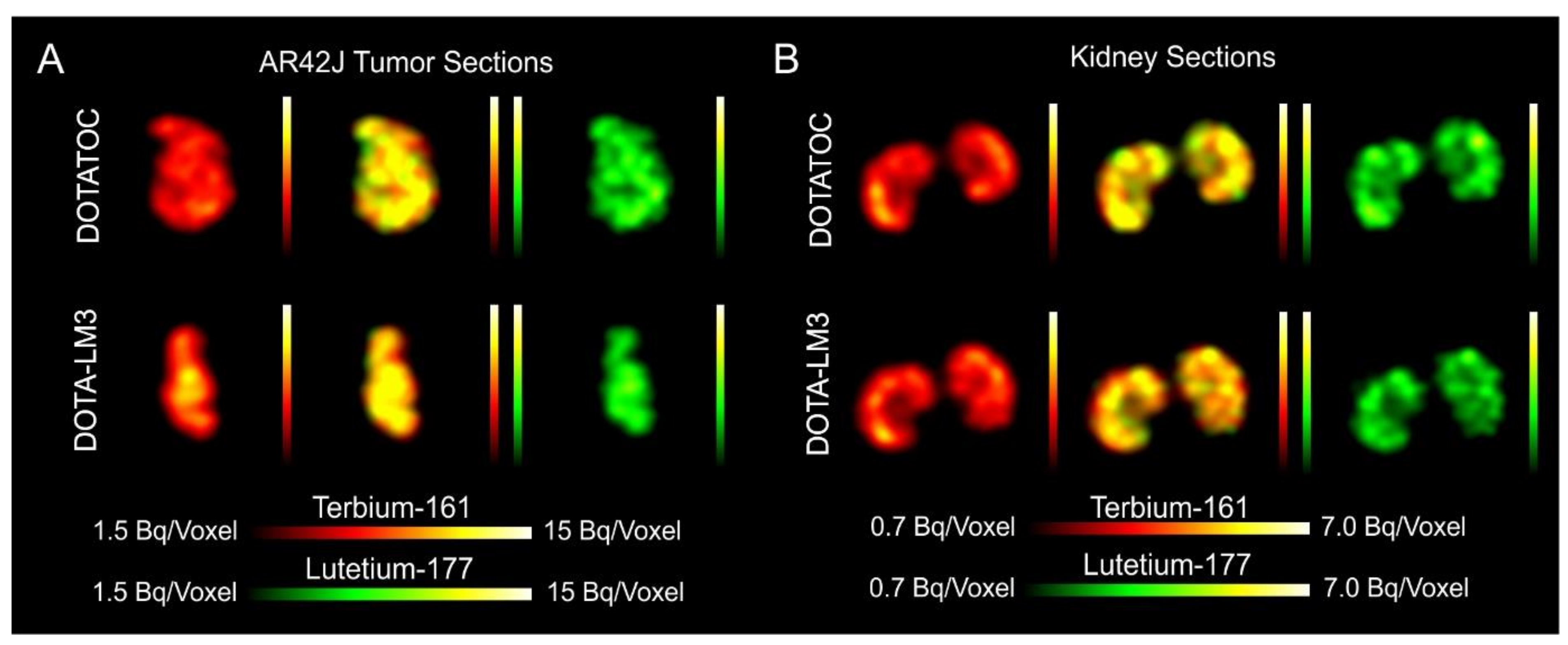
3.7. Simultaneous Imaging Demonstrates Equal Sub-Organ Distribution of 161Tb- and 177Lu-Labeled SST Analogues
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lehenberger, S.; Barkhausen, C.; Cohrs, S.; Fischer, E.; Grünberg, J.; Hohn, A.; Köster, U.; Schibli, R.; Türler, A.; Zhernosekov, K. The low-energy β− and electron emitter 161Tb as an alternative to 177Lu for targeted radionuclide therapy. Nucl. Med. Biol. 2011, 38, 917–924. [Google Scholar] [CrossRef]
- Müller, C.; Zhernosekov, K.; Köster, U.; Johnston, K.; Dorrer, H.; Hohn, A.; Van Der Walt, N.T.; Türler, A.; Schibli, R. A Unique Matched Quadruplet of Terbium Radioisotopes for PET and SPECT and for-and-Radionuclide Therapy: An in vivo proof-of-concept study with a new receptor-targeted folate derivative. J. Nucl. Med. 2012, 53, 1951–1959. [Google Scholar] [CrossRef]
- Durán, M.T.; Juget, F.; Nedjadi, Y.; Bochud, F.; Grundler, P.V.; Gracheva, N.; Müller, C.; Talip, Z.; Van Der Meulen, N.P.; Bailat, C. Determination of 161Tb half-life by three measurement methods. Appl. Radiat. Isot. 2020, 159, 109085. [Google Scholar] [CrossRef]
- Bernhardt, E.F.-A.P. Low-energy electron emitters for targeted radiotherapy of small tumours. ACTA Oncol. 2001, 40, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, P.; Benjegård, S.A.; Kölby, L.; Johanson, V.; Nilsson, O.; Ahlman, H.; Forssell-Aronsson, E. Dosimetric comparison of radionuclides for therapy of somatostatin receptor-expressing tumors. Int. J. Radiat. Oncol. 2001, 51, 514–524. [Google Scholar] [CrossRef]
- Hindié, E.; Zanotti-Fregonara, P.; Quinto, M.A.; Morgat, C.; Champion, C. Dose Deposits from 90Y, 177Lu, 111In, and 161Tb in micrometastases of various sizes: Implications for radiopharmaceutical therapy. J. Nucl. Med. 2016, 57, 759–764. [Google Scholar] [CrossRef]
- Champion, C.; Quinto, M.A.; Morgat, C.; Zanotti-Fregonara, P.; Hindié, E. Comparison between three promising ߯-emitting radionuclides, 67Cu, 47Sc and 161Tb, with emphasis on doses delivered to minimal residual disease. Theranostics 2016, 6, 1611–1618. [Google Scholar] [CrossRef]
- Alcocer-Ávila, M.E.; Ferreira, A.; Quinto, M.A.; Morgat, C.; Hindié, E.; Champion, C. Radiation doses from 161Tb and 177Lu in single tumour cells and micrometastases. EJNMMI Phys. 2020, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Reber, J.; Haller, S.; Dorrer, H.; Bernhardt, P.; Zhernosekov, K.; Türler, A.; Schibli, R. Direct in vitro and in vivo comparison of 161Tb and 177Lu using a tumour-targeting folate conjugate. Eur. J. Nucl. Med. Mol. Imaging 2013, 41, 476–485. [Google Scholar] [CrossRef]
- Müller, C.; Umbricht, C.A.; Gracheva, N.; Tschan, V.J.; Pellegrini, G.; Bernhardt, P.; Zeevaart, J.R.; Köster, U.; Schibli, R.; van der Meulen, N.P. Terbium-161 for PSMA-targeted radionuclide therapy of prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1919–1930. [Google Scholar] [CrossRef] [PubMed]
- Marin, I.; Rydèn, T.; Van Essen, M.; Svensson, J.; Gracheva, N.; Köster, U.; Zeevaart, J.R.; van der Meulen, N.P.; Müller, C.; Bernhardt, P. Establishment of a clinical SPECT/CT protocol for imaging of 161Tb. EJNMMI Phys. 2020, 7, 1–16. [Google Scholar] [CrossRef]
- Baum, R.P.; Singh, A.; Kulkarni, H.R.; Bernhardt, P.; Rydén, T.; Schuchardt, C.; Gracheva, N.; Grundler, P.V.; Köster, U.; Müller, D.; et al. First-in-human application of terbium-161: A feasibility study using 161Tb-DOTATOC. J. Nucl. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Haller, S.; Pellegrini, G.; Vermeulen, C.; van der Meulen, N.P.; Köster, U.; Bernhardt, P.; Schibli, R.; Müller, C. Contribution of Auger/conversion electrons to renal side effects after radionuclide therapy: Preclinical comparison of 161Tb-folate and 177Lu-folate. EJNMMI Res. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- André, J.P.; Maecke, H.R.; Zehnder, M.; Macko, L.; Akyel, K.G. 1,4,7-Triazacyclononane-1-succinic acid-4,7-diacetic acid (NODASA): A new bifunctional chelator for radio gallium-labelling of biomolecules. Chem. Commun. 1998, 1998, 1301–1302. [Google Scholar] [CrossRef]
- Heppeler, A.; Froidevaux, S.; Macke, H.R.; Jermann, E.; Behe, M.; Powell, P. Radiometal-labelled macrocyclic chelator-derivatised somatostatin analogue with superb tumour-targeting properties and potential for receptor-mediated internal radiotherapy. Chem. Eur. J. 1999, 5, 1974–1981. [Google Scholar] [CrossRef]
- Fani, M.; Braun, F.; Waser, B.; Beetschen, K.; Cescato, R.; Erchegyi, J.; Rivier, J.E.; Weber, W.A.; Mäcke, H.R.; Reubi, J.C. Unexpected sensitivity of SST2 antagonists to N-terminal radiometal modifications. J. Nucl. Med. 2012, 53, 1481–1489. [Google Scholar] [CrossRef]
- Müller, C.; Vermeulen, C.; Johnston, K.; Köster, U.; Schmid, R.; Türler, A.; van der Meulen, N.P. Preclinical in vivo application of 152Tb-DOTANOC: A radiolanthanide for PET imaging. EJNMMI Res. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Baum, R.P.; Singh, A.; Benešová, M.; Vermeulen, C.; Gnesin, S.; Köster, U.; Johnston, K.; Müller, D.; Senftleben, S.; Kulkarni, H.R.; et al. Clinical evaluation of the radiolanthanide terbium-152: First-in-human PET/CT with 152Tb-DOTATOC. Dalton Trans. 2017, 46, 14638–14646. [Google Scholar] [CrossRef] [PubMed]
- Otte, A.; Jermann, E.; Behe, M.; Goetze, M.; Bucher, H.C.; Roser, H.W.; Heppeler, A.; Müller-Brand, J.; Mäcke, H.R. DOTATOC: A powerful new tool for receptor-mediated radionuclide therapy. Eur. J. Nucl. Med. Mol. Imaging 1997, 24, 792–795. [Google Scholar] [CrossRef][Green Version]
- Fani, M.; Del Pozzo, L.; Abiraj, K.; Mansi, R.; Tamma, M.L.; Cescato, R.; Waser, B.; Weber, W.A.; Reubi, J.C.; Mäcke, H.R. PET of Somatostatin receptor-positive tumors using 64Cu- and 68Ga-somatostatin antagonists: The chelate makes the difference. J. Nucl. Med. 2011, 52, 1110–1118. [Google Scholar] [CrossRef]
- Baum, R.P.; Kluge, A.W.; Kulkarni, H.; Schorr-Neufing, U.; Niepsch, K.; Bitterlich, N.; Van Echteld, C.J. [177Lu-DOTA]0-D-Phe1-Tyr3-Octreotide (177Lu-DOTATOC) for peptide receptor radiotherapy in patients with advanced neuroendocrine tumours: A Phase-II study. Theranostics 2016, 6, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-F.; Lin, L.; Wang, M.-J.; Li, Y. The therapeutic efficacy of 177Lu-DOTATATE/DOTATOC in advanced neuroendocrine tumors. Medicine 2020, 99, e19304. [Google Scholar] [CrossRef]
- Zhang, J.; Kulkarni, H.R.; Singh, A.; Baum, R.P. Successful intra-arterial peptide receptor radionuclide therapy of DOTATOC-negative high-grade liver metastases of a pancreatic neuroendocrine neoplasm using 177Lu-DOTA-LM3. Clin. Nucl. Med. 2020, 45, e165–e168. [Google Scholar] [CrossRef]
- Baum, R.P.; Zhang, J.; Schuchardt, C.; Müller, D.; Mäcke, H. First-in-human study of novel SSTR antagonist 177Lu-DOTA-LM3 for peptide receptor radionuclide therapy in patients with metastatic neuroendocrine neoplasms: Dosimetry, safety and efficacy. J. Nucl. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Gracheva, N.; Müller, C.; Talip, Z.; Heinitz, S.; Köster, U.; Zeevaart, J.R.; Vögele, A.; Schibli, R.; van der meulen, N.P. Production and characterization of no-carrier-added 161Tb as an alternative to the clinically-applied 177Lu for radionuclide therapy. EJNMMI Radiopharm. Chem. 2019, 4, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Mindt, T.L.; De Jong, M.; Schibli, R. Evaluation of a novel radiofolate in tumour-bearing mice: Promising prospects for folate-based radionuclide therapy. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 938–946. [Google Scholar] [CrossRef]
- Hofsli, E.; Thommesen, L.; Nørsett, K.; Falkmer, S.; Syversen, U.; Sandvik, A.K.; Lægreid, A. Expression of chromogranin A and somatostatin receptors in pancreatic AR42J cells. Mol. Cell. Endocrinol. 2002, 194, 165–173. [Google Scholar] [CrossRef]
- Zaknun, J.J.; Bodei, L.; Müller-Brand, J.; Pavel, M.E.; Baum, R.P.; Hörsch, D.; O’Dorisio, M.S.; O’Dorisiol, T.M.; Howe, J.R.; Cremonesi, M.; et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 800–816. [Google Scholar] [CrossRef] [PubMed]
- Sabet, A.; Nagarajah, J.; Dogan, A.S.; Biersack, H.-J.; Sabet, A.; Guhlke, S.; Ezziddin, S. Does PRRT with standard activities of 177Lu-octreotate really achieve relevant somatostatin receptor saturation in target tumor lesions?: Insights from intra-therapeutic receptor imaging in patients with metastatic gastroenteropancreatic neuroendocrine tumors. EJNMMI Res. 2013, 3, 82. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Das, T.; Bhadwal, M.; Banerjee, S.; Sarma, H.D.; Shinto, A.; Kamaleshwaran, K.K. Preparation of DOTA-TATE and DOTA-NOC freeze-dried kits for formulation of patient doses of 177Lu-labeled agents and their comparison for peptide receptor radionuclide therapy application. J. Radioanal. Nucl. Chem. 2014, 299, 1389–1398. [Google Scholar] [CrossRef]
- Ku, A.; Facca, V.J.; Cai, Z.; Reilly, R.M. Auger electrons for cancer therapy–a review. EJNMMI Radiopharm. Chem. 2019, 4, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Iori, M.; Capponi, P.C.; Rubagotti, S.; Esposizione, L.R.; Seemann, J.; Pitzschler, R.; Dreger, T.; Formisano, D.; Grassi, E.; Fioroni, F.; et al. Labelling of 90Y- and 177Lu-DOTA-bioconjugates for targeted radionuclide therapy: A comparison among manual, semiautomated, and fully automated synthesis. Contrast Media Mol. Imaging 2017, 2017, 1–12. [Google Scholar] [CrossRef]
- Cescato, R.; Schulz, S.; Waser, B.; Eltschinger, V.; Rivier, E.J.; Wester, H.-J.; Culler, M.; Ginj, M.; Liu, Q.; Schonbrunn, A.; et al. Internalization of SST2, SST3, and SST5 receptors: Effects of somatostatin agonists and antagonists. J. Nucl. Med. 2006, 47, 502–511. [Google Scholar] [PubMed]
- Wadas, T.J.; Eiblmaier, M.; Zheleznyak, A.; Sherman, C.D.; Ferdani, R.; Liang, K.; Achilefu, S.; Anderson, C.J. Preparation and biological evaluation of 64Cu-CB-TE2A-SST2-ANT, a somatostatin antagonist for PET imaging of somatostatin receptor-positive tumors. J. Nucl. Med. 2008, 49, 1819–1827. [Google Scholar] [CrossRef]
- Ginj, M.; Zhang, H.; Waser, B.; Cescato, R.; Wild, D.; Wang, X.; Erchegyi, J.; Rivier, J.; Mäcke, H.R.; Reubi, J.C. Radiolabeled somatostatin receptor antagonists are preferable to agonists for in vivo peptide receptor targeting of tumors. Proc. Natl. Acad. Sci. USA 2006, 103, 16436–16441. [Google Scholar] [CrossRef] [PubMed]
- Reubi, J.C.; Schär, J.-C.; Waser, B.; Wenger, S.; Heppeler, A.; Schmitt, J.S.; Mäcke, H.R. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur. J. Nucl. Med. Mol. Imaging 2000, 27, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Ginj, M.; Hinni, K.; Tschumi, S.; Schulz, S.; Mäcke, H.R. Trifunctional somatostatin-based derivatives designed for targeted radiotherapy using Auger electron emitters. J. Nucl. Med. 2005, 46, 2097–2103. [Google Scholar]
- Nicolas, G.P.; Mansi, R.; McDougall, L.; Kaufmann, J.; Bouterfa, H.; Wild, D.; Fani, M. Biodistribution, pharmacokinetics, and dosimetry of 177Lu-, 90Y-, and 111In-labeled somatostatin receptor antagonist OPS201 in comparison to the agonist 177Lu-DOTATATE: The mass effect. J. Nucl. Med. 2017, 58, 1435–1441. [Google Scholar] [CrossRef]
- Dude, I.; Zhang, Z.; Rousseau, J.; Hundal-Jabal, N.; Colpo, N.; Merkens, H.; Lin, K.-S.; Bénard, F. Evaluation of agonist and antagonist radioligands for somatostatin receptor imaging of breast cancer using positron emission tomography. EJNMMI Radiopharm. Chem. 2017, 2, 1–16. [Google Scholar] [CrossRef]
- Velikyan, I.; Xu, H.; Nair, M.; Hall, H. Robust labeling and comparative preclinical characterization of DOTA-TOC and DOTA-TATE. Nucl. Med. Biol. 2012, 39, 628–639. [Google Scholar] [CrossRef]
- Vegt, E.; Melis, M.; Eek, A.; De Visser, M.; Brom, M.; Oyen, W.J.G.; Gotthardt, M.; De Jong, M.; Boerman, O.C. Renal uptake of different radiolabelled peptides is mediated by megalin: SPECT and biodistribution studies in megalin-deficient mice. Eur. J. Nucl. Med. Mol. Imaging 2010, 38, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Balster, D.A.; O’Dorisio, M.S.; Summers, M.A.; Turman, M.A. Segmental expression of somatostatin receptor subtypes SST1 and SST2 in tubules and glomeruli of human kidney. Am. J. Renal Physiol. 2001, 280, F457–F465. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.M.; Kegg, H.; Petrevski, C.; Grady, S. Expression of somatostatin receptors 3, 4, and 5 in mouse kidney proximal tubules. Kidney Int. 2003, 63, 53–63. [Google Scholar] [CrossRef] [PubMed][Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borgna, F.; Barritt, P.; Grundler, P.V.; Talip, Z.; Cohrs, S.; Zeevaart, J.R.; Köster, U.; Schibli, R.; van der Meulen, N.P.; Müller, C. Simultaneous Visualization of 161Tb- and 177Lu-Labeled Somatostatin Analogues Using Dual-Isotope SPECT Imaging. Pharmaceutics 2021, 13, 536. https://doi.org/10.3390/pharmaceutics13040536
Borgna F, Barritt P, Grundler PV, Talip Z, Cohrs S, Zeevaart JR, Köster U, Schibli R, van der Meulen NP, Müller C. Simultaneous Visualization of 161Tb- and 177Lu-Labeled Somatostatin Analogues Using Dual-Isotope SPECT Imaging. Pharmaceutics. 2021; 13(4):536. https://doi.org/10.3390/pharmaceutics13040536
Chicago/Turabian StyleBorgna, Francesca, Patrick Barritt, Pascal V. Grundler, Zeynep Talip, Susan Cohrs, Jan Rijn Zeevaart, Ulli Köster, Roger Schibli, Nicholas P. van der Meulen, and Cristina Müller. 2021. "Simultaneous Visualization of 161Tb- and 177Lu-Labeled Somatostatin Analogues Using Dual-Isotope SPECT Imaging" Pharmaceutics 13, no. 4: 536. https://doi.org/10.3390/pharmaceutics13040536
APA StyleBorgna, F., Barritt, P., Grundler, P. V., Talip, Z., Cohrs, S., Zeevaart, J. R., Köster, U., Schibli, R., van der Meulen, N. P., & Müller, C. (2021). Simultaneous Visualization of 161Tb- and 177Lu-Labeled Somatostatin Analogues Using Dual-Isotope SPECT Imaging. Pharmaceutics, 13(4), 536. https://doi.org/10.3390/pharmaceutics13040536








