Current Update of Collagen Nanomaterials—Fabrication, Characterisation and Its Applications: A Review
Abstract
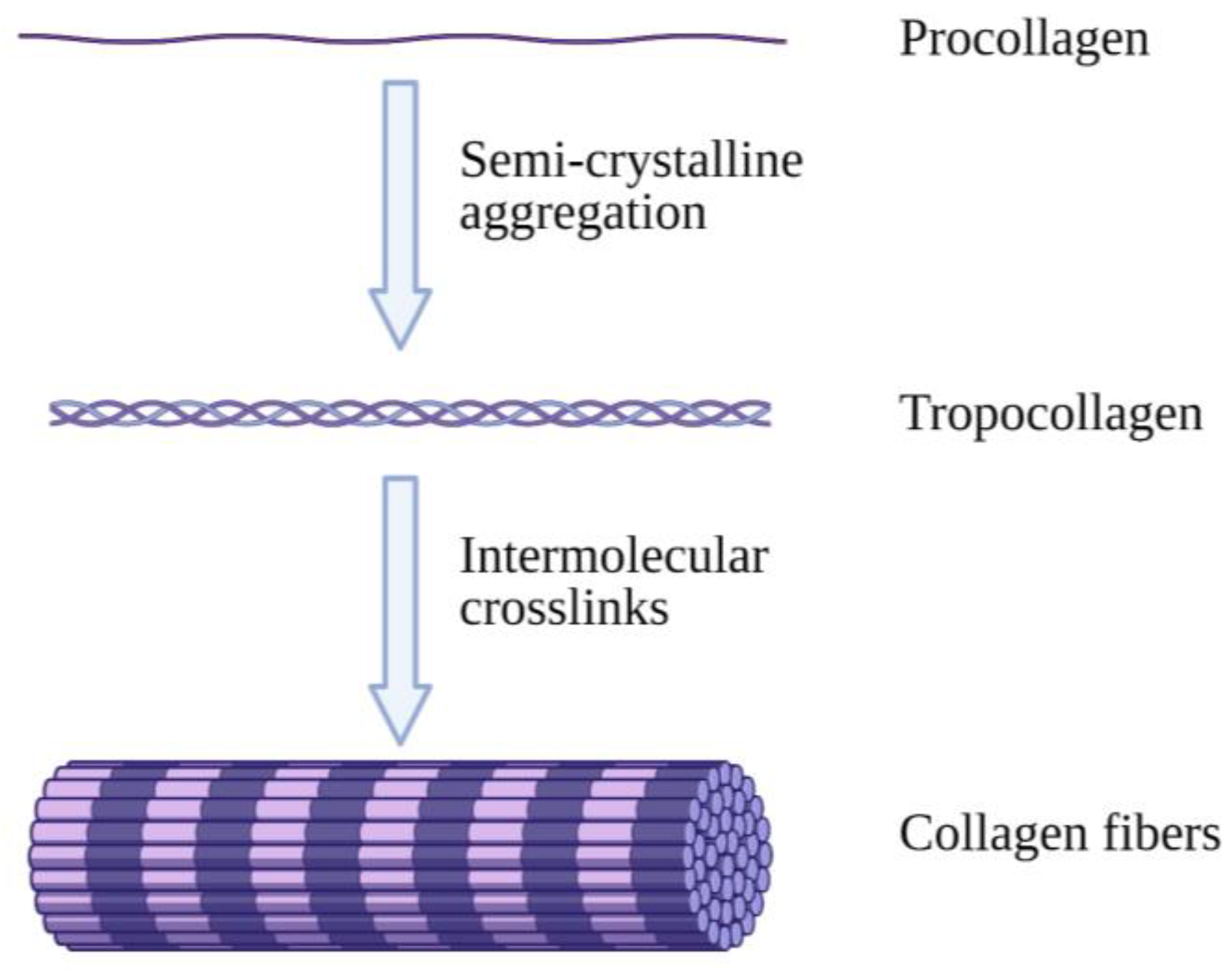
1. Introduction
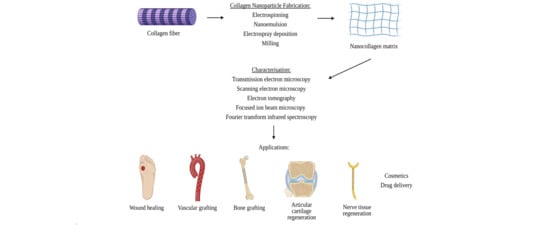
2. Nano Collagen Fabrication
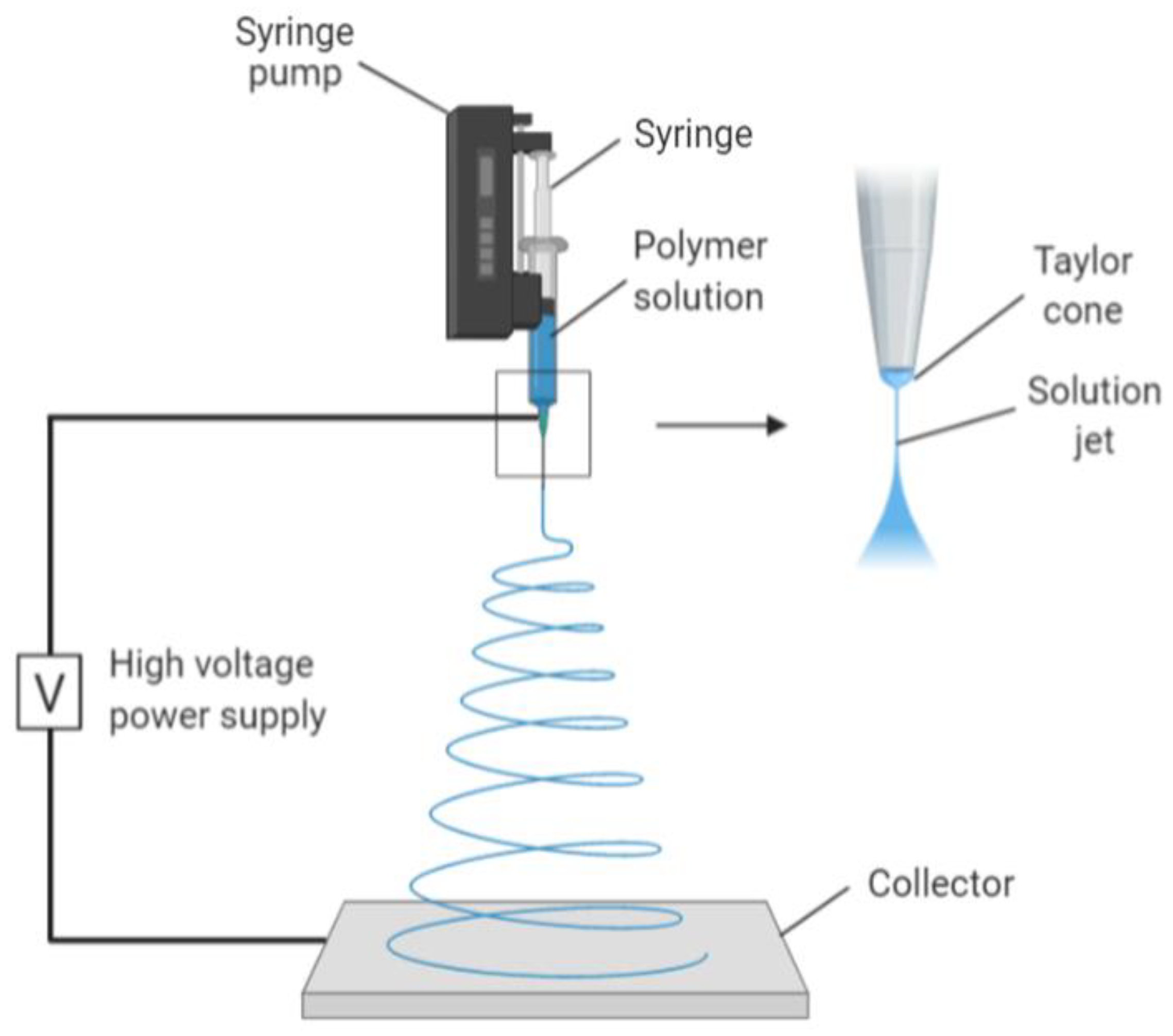
2.1. Electrospinning
2.2. Nanoemulsion
2.3. Electrospray Deposition
2.4. Milling
3. Characterisation Approaches
3.1. Transmission Electron Microscopy (TEM)
3.2. Electron Tomography (ET)
3.3. Scanning Electron Microscopy (SEM)
3.4. Focused Ion Beam (FIB) Microscopy
3.4.1. FIB-TEM
3.4.2. FIB-SEM
3.5. Fourier-Transform Infrared (FTIR) Spectroscopy
4. Applications
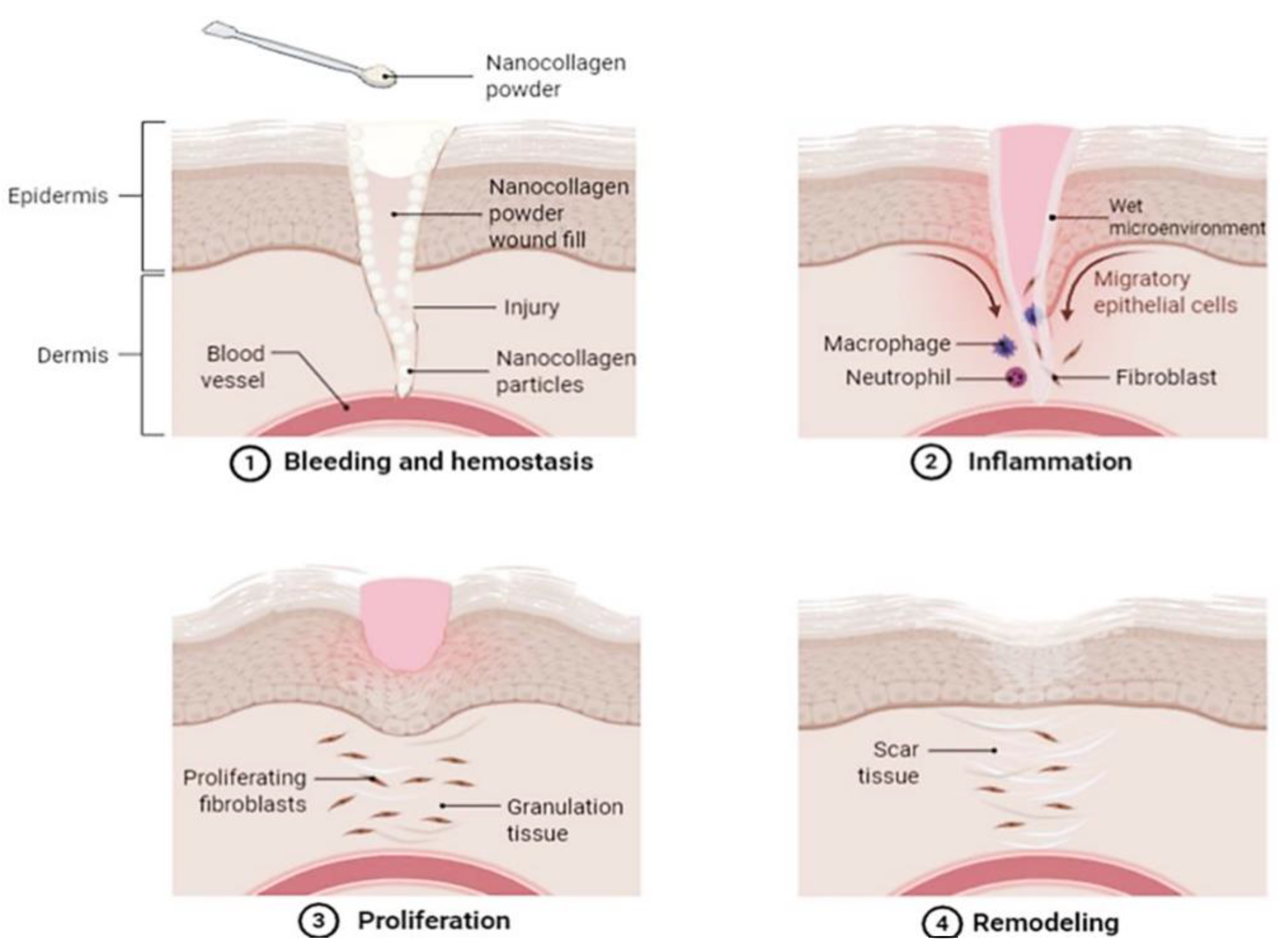
4.1. Skin Wound Healing
4.2. Bone Grafting
4.3. Drug Delivery
4.4. Nerve Tissue
4.5. Vascular Grafting
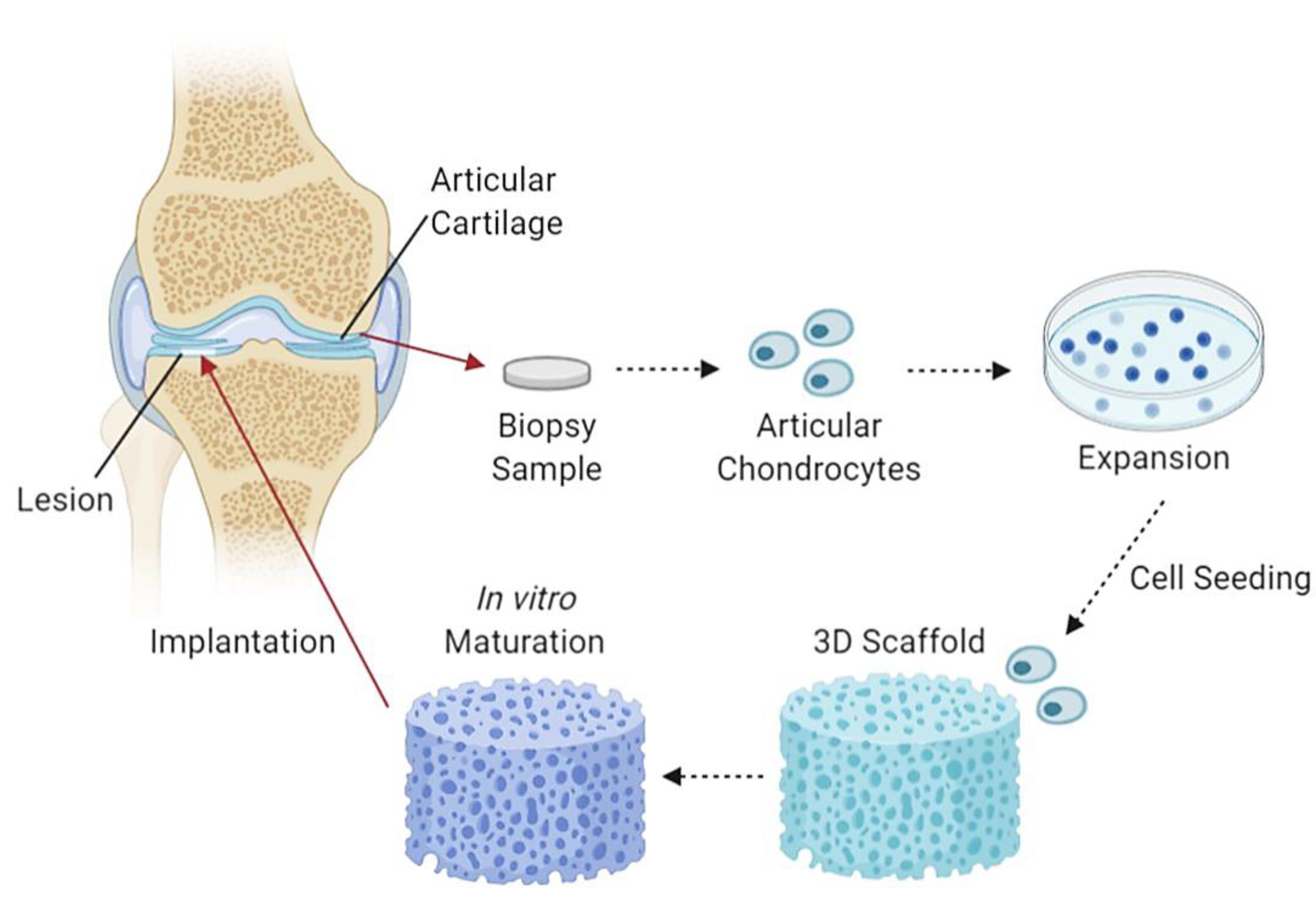
4.6. Articular Cartilage
4.7. Cosmetics
5. Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sionkowska, A.; Skrzyński, S.; Śmiechowski, K.; Kołodziejczak, A. The review of versatile application of collagen. Polym. Adv. Technol. 2017, 28, 4–9. [Google Scholar] [CrossRef]
- Jena, K.; Pandey, J.; Kumari, R.; Sinha, A.; Gupta, V.; Singh, G. Free radical scavenging potential of sericin obtained from various ecoraces of tasar cocoons and its cosmeceuticals implication. Int. J. Biol. Macromol. 2018, 120, 255–262. [Google Scholar] [CrossRef]
- Hussain, M.A.; Muhammad, G.; Jantan, I.; Bukhari, S.N.A. Psyllium Arabinoxylan: A Versatile Biomaterial for Potential Medicinal and Pharmaceutical Applications. Polym. Rev. 2015, 56, 1–30. [Google Scholar] [CrossRef]
- Moni, T.; Jose, D.; Paul, D.; Murali, A.; Jacob, J. Development of antibacterial biomaterial for medical application. AIP Conf. Proc. 2020, 2263, 030012. [Google Scholar]
- Echave, M.C.; Burgo, L.S.; Pedraz, J.L.; Orive, G. Gelatin as biomaterial for tissue engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Liu, W.; Zhang, M.; Si, C.; Zhang, X.; Li, B. Cellulose nanocrystals and cellulose nanofibrils based hydrogels for biomedical applications. Carbohydr. Polym. 2019, 209, 130–144. [Google Scholar] [CrossRef]
- Kelly, S.; Weinkamer, R.; Bertinetti, L.; Edmonds, R.; Sizeland, K.; Wells, H.; Fratzl, P.; Haverkamp, R. Effect of collagen packing and moisture content on leather stiffness. J. Mech. Behav. Biomed. Mater. 2019, 90, 1–10. [Google Scholar] [CrossRef]
- Milazzo, M.; Jung, G.S.; Danti, S.; Buehler, M.J. Wave Propagation and Energy Dissipation in Collagen Molecules. ACS Biomater. Sci. Eng. 2020, 6, 1367–1374. [Google Scholar] [CrossRef] [PubMed]
- Jung, G.S.; Buehler, M.J. Multiscale Modeling of Muscular-Skeletal Systems. Annu. Rev. Biomed. Eng. 2017, 19, 435–457. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Nagarajan, S.; Bechelany, M.; Kalkura, S.N. Collagen Based Biomaterials for Tissue Engineering Applications: A Review. In Processes and Phenomena on the Boundary between Biogenic and Abiogenic Nature; Frank-Kamenetskaya, O.V., Vlasov, D.Y., Panova, E.G., Lessovaia, S.N., Eds.; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Senadheera, T.R.; Dave, D.; Shahidi, F. Sea Cucumber Derived Type I Collagen: A Comprehensive Review. Mar. Drugs 2020, 18, 471. [Google Scholar] [CrossRef] [PubMed]
- Nimni, M.E. Collagen: Volume I: Biochemistry, 1st ed.; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Zhang, X.; Williams, D. Definitions of Biomaterials for the Twenty-First Century; Elsevier: Chennai, India, 2019. [Google Scholar]
- Arya, H.; Chauhan, C.; Sinha, V.B. Biomaterial—A review. J. Crit. Rev. 2020, 7, 681–685. [Google Scholar]
- An, Y.Z.; Kim, Y.K.; Lim, S.M.; Heo, Y.K.; Kwon, M.K.; Cha, J.K.; Lee, J.S.; Jung, U.W.; Choi, S.H. Physiochemical properties and re-sorption progress of porcine skin-derived collagen membranes: In vitro and in vivo analysis. Dent. Mater. J. 2018, 37, 2017–2065. [Google Scholar] [CrossRef]
- Sheehy, E.J.; Cunniffe, G.M.; O’Brien, F.J. Collagen-based biomaterials for tissue regeneration and repair. Pept. Proteins Biomater. Tissue Regen. Repair 2018, 2018, 127–150. [Google Scholar] [CrossRef]
- Lin, K.; Zhang, D.; Macedo, M.H.; Cui, W.; Sarmento, B.; Shen, G. Advanced collagen-based biomaterials for regenerative biomed-icine. Adv. Funct. Mater. 2019, 29, 1804943. [Google Scholar] [CrossRef]
- Scharf, R.E. Platelet Signaling in Primary Haemostasis and Arterial Thrombus Formation: Part 1. Hämostaseologie 2018, 38, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, B. Springer Handbook of Nanotechnology. Sens. Rev. 2004, 24, 319. [Google Scholar] [CrossRef]
- Wilson, A. The formation of dry, wet, spunlaid and other types of nonwovens. Appl. Nonwovens Tech. Text. 2010, 2010, 3–17. [Google Scholar] [CrossRef]
- Rohira, H.; Kumar, V.; Chugh, A. Nanotechnology: Revolutionizing Agricultural Sector. Botanica 2015, 64–65, 151–155. [Google Scholar]
- Naskar, A.; Kim, K.S. Recent Advances in nanomaterial-based wound-healing therapeutics. Pharmaceutics 2020, 12, 499. [Google Scholar] [CrossRef]
- Wang, X.; Yan, Y.; Yost, M.J.; Fann, S.A.; Dong, S.; Li, X. Nanomechanical characterization of micro/nanofiber reinforced type I col-lagens. J. Biomed. Mater. Res. A 2007, 83A, 130–135. [Google Scholar] [CrossRef]
- Law, J.X.; Liau, L.L.; Saim, A.; Yang, Y.; Idrus, R. Electrospun Collagen Nanofibers and Their Applications in Skin Tissue Engineering. Tissue Eng. Regen. Med. 2017, 14, 699–718. [Google Scholar] [CrossRef]
- Sundaramurthi, D.; Krishnan, U.M.; Sethuraman, S. Electrospun Nanofibers as Scaffolds for Skin Tissue Engineering. Polym. Rev. 2014, 54, 348–376. [Google Scholar] [CrossRef]
- Musazzi, U.M.; Franzè, S.; Minghetti, P.; Casiraghi, A. Emulsion versus nanoemulsion: How much is the formulative shift critical for a cosmetic product? Drug Deliv. Transl. Res. 2017, 8, 414–421. [Google Scholar] [CrossRef]
- Singh, Y.; Meher, J.G.; Raval, K.; Khan, F.A.; Chaurasia, M.; Jain, N.K.; Chourasia, M.K. Nanoemulsion: Concepts, development and applications in drug delivery. J. Control. Release 2017, 252, 28–49. [Google Scholar] [CrossRef] [PubMed]
- Gurpret, K.; Singh, S.K. Review of Nanoemulsion Formulation and Characterization Techniques. Indian J. Pharm. Sci. 2018, 80, 781–789. [Google Scholar] [CrossRef]
- Che Marzuki, N.H.; Wahab, R.A.; Abdul Hamid, M. An overview of nanoemulsion: Concepts of development and cosmeceutical applications. Biotechnol. Biotechnol. Equip. 2019, 33, 779–797. [Google Scholar] [CrossRef]
- Care, O.; Corner, C.; Listing, J. Nanotechnology in cosmetics. Nanotechnology. 2017. Available online: http://www.thecosmeticchemist.com/news/nanotechnology_in_cosmetics.html (accessed on 26 February 2021).
- Katz, L.M.; Dewan, K.; Bronaugh, R.L. Nanotechnology in cosmetics. Food Chem. Toxicol. 2015, 85, 127–137. [Google Scholar] [CrossRef]
- Avilez, H.R.; Casadiego, D.C.; Avila, A.V.; Perez, O.P.; Almodovar, J. 11—Production of chitosan coatings on metal and ceramic biomaterials. In Chitosan Based Biomaterials; Jennings, J.A., Bumgardner, J.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 255–293. [Google Scholar]
- Boda, S.K.; Li, X.; Xie, J. Electrospraying an enabling technology for pharmaceutical and biomedical applications: A review. J. Aerosol. Sci. 2018, 125, 164–181. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, U.; Kawakami, K.; Zhang, S.; Chandrasekaran, B.; Nair, B.U. Fabrication of solid collagen nanoparticles using elec-trospray deposition. Chem. Pharm. Bull. 2014, 62, 422–428. [Google Scholar] [CrossRef]
- Saallah, S.; Naim, M.N.; Mokhtar, M.N.; Abu Bakar, N.F.; Gen, M.; Lenggoro, I.W. Transformation of cyclodextrin glucanotransferase (CGTase) from aqueous suspension to fine solid particles via electrospraying. Enzym. Microb. Technol. 2014, 65, 52–59. [Google Scholar] [CrossRef]
- Mhetar, S.K.; Nerle, A.A.; Patil, R.L.; Pawar, R.A.; Shinde, M.M.P.H.T. Cost effective ball milling machine for producing nanopowder. Int. J. Res. Eng. Technol. 2017, 4, 330–334. [Google Scholar]
- Kumar, M.; Xiong, X.; Wan, Z.; Sun, Y.; Tsang, D.C.; Gupta, J.; Gao, B.; Cao, X.; Tang, J.; Ok, Y.S. Ball milling as a mechanochemical technology for fabrication of novel biochar nanomaterials. Bioresour. Technol. 2020, 312, 123613. [Google Scholar] [CrossRef]
- Sanz-Fraile, H.; Amoros, S.; Mendizabal, M.I.I.; Galvez-Monton, C.; Prat-Vidal, C.; Bayes-Genis, A.; Navajas, D.; Farre, R.; Otero, J. Silk-Reinforced Collagen Hydrogels with Raised Multiscale Stiffness for Mesenchymal Cells 3D Culture. Tissue Eng. Part A 2020, 26, 358–370. [Google Scholar] [CrossRef] [PubMed]
- Starborg, T.; Kalson, N.S.; Lu, Y.; Mironov, A.; Cootes, T.F.; Holmes, D.F.; Kadler, K.E. Using transmission electron microscopy and 3View to determine collagen fibril size and three-dimensional organization. Nat. Protoc. 2013, 8, 1433–1448. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.J.F. Transmission Electron Microscopy of Carbon: A Brief History. C 2018, 4, 4. [Google Scholar] [CrossRef]
- Graham, B.J.; Hildebrand, D.G.C.; Kuan, A.T.; Maniates-Selvin, J.T.; Thomas, L.A.; Shanny, B.L.; Lee, W.C.A. High-throughput transmis-sion electron microscopy with automated serial sectioning. Biorxiv 2019, 657346. [Google Scholar] [CrossRef]
- Susi, T.; Meyer, J.C.; Kotakoski, J. Manipulating low-dimensional materials down to the level of single atoms with electron irradi-ation. Ultramicroscopy 2017, 180, 163–172. [Google Scholar] [CrossRef]
- Ishikawa, T. Electron Tomography. In Encyclopedia of Cell Biology; Bradshaw, R.A., Stahl, P.D., Eds.; Academic Press: Waltham, MA, USA, 2016; pp. 22–31. [Google Scholar]
- Song, H.; Yang, Y.; Geng, J.; Gu, Z.; Zou, J.; Yu, C. Electron tomography: A unique tool solving intricate hollow nanostructures. Adv. Mater. 2019, 31, 1801564. [Google Scholar] [CrossRef]
- Kováčik, L.; Kerïeche, S.; Höög, J.L.; Juda, P.; Matula, P.; Raška, I. A simple Fourier filter for suppression of the missing wedge ray artefacts in single-axis electron tomographic reconstructions. J. Struct. Biol. 2014, 186, 141–152. [Google Scholar] [CrossRef]
- Mohammed, A.; Abdullah, A. Scanning electron microscopy (SEM): A review. In Proceedings of the 2018 international conference on hydraulics and pneumatics–Hervex, Baile Govora, Romania, 7–9 November 2018. [Google Scholar]
- Ul-Hamid, A. A Beginners’ Guide to Scanning Electron Microscopy; Springer: Berlin, Germany, 2018. [Google Scholar]
- Akhtar, K.; Khan, S.A.; Khan, S.B.; Asiri, A.M. Scanning Electron Microscopy: Principle and Applications in Nanomaterials Char-acterization. In Handbook of Materials Characterization; Springer: Berlin, Germany, 2018; pp. 113–145. [Google Scholar]
- Van Duong, H.; Chau, T.T.L.; Dang, N.T.T.; Van Nguyen, D.; Le, S.L.; Ho, T.S.; Vu, T.P.; Tran, T.T.V.; Nguyen, T.D. Self-aggregation of wa-ter-dispersible nanocollagen helices. Biomater. Sci. 2018, 6, 651–660. [Google Scholar] [CrossRef]
- Xiao, Y.; Wehrs, J.; Ma, H.; Al-Samman, T.; Korte-Kerzel, S.; Göken, M.; Michler, J.; Spolenak, R.; Wheeler, J.M. Investigation of the de-formation behavior of aluminum micropillars produced by focused ion beam machining using Ga and Xe ions. Scr. Mater. 2017, 127, 191–194. [Google Scholar] [CrossRef]
- Joe, H.-E.; Lee, W.-S.; Jun, M.B.G.; Park, N.-C.; Min, B.-K. Material interface detection based on secondary electron images for focused ion beam machining. Ultramicroscopy 2018, 184, 37–43. [Google Scholar] [CrossRef]
- Cizmar, P.; Yuana, Y. Detection and Characterization of Extracellular Vesicles by Transmission and Cryo-Transmission Electron Microscopy. In Extracellular Vesicles: Methods and Protocols; Methods in Molecular Biology; Springer: New York, NY, USA, 2017; Volume 1660, pp. 221–232. [Google Scholar] [CrossRef]
- Gerlinde, H.; Habler, G.; Baldwin, L.C.; Abart, R. An improved FIB sample preparation technique for site-specific plan-view specimens: A new cutting geometry. Ultramicroscopy 2018, 184, 310–317. [Google Scholar] [CrossRef]
- Simon, P.; Grüner, D.; Worch, H.; Pompe, W.; Lichte, H.; El Khassawna, T.; Heiss, C.; Wenisch, S.; Kniep, R. First evidence of octacalcium phosphate@osteocalcin nanocomplex as skeletal bone component directing collagen triple–helix nanofibril mineralization. Sci. Rep. 2018, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Iacoviello, F.; Kirby, A.C.; Javanmardi, Y.; Moeendarbary, E.; Shabanli, M.; Tsolaki, E.; Sharp, A.C.; Hayes, M.J.; Keevend, K.; Li, J.-H.; et al. The multiscale hierarchical structure of Heloderma suspectum osteoderms and their mechanical properties. Acta Biomater. 2020, 107, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.P.; Farhang, N.; Poulson, R.; Parkman, G.; Weiss, J.A. Nanoscale Imaging of Collagen Gels with Focused Ion Beam Milling and Scanning Electron Microscopy. Biophys. J. 2016, 111, 1797–1804. [Google Scholar] [CrossRef] [PubMed]
- Nan, N.; Wang, J. FIB-SEM Three-Dimensional Tomography for Characterization of Carbon-Based Materials. Adv. Mater. Sci. Eng. 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Hu, C.; Aindow, M.; Wei, M. Focused ion beam sectioning studies of biomimetic hydroxyapatite coatings on Ti-6Al-4V substrates. Surf. Coat. Technol. 2017, 313, 255–262. [Google Scholar] [CrossRef]
- Hu, C.; Yu, L.; Wei, M. Sectioning studies of biomimetic collagen-hydroxyapatite coatings on Ti-6Al-4V substrates using focused ion beam. Appl. Surf. Sci. 2018, 444, 590–597. [Google Scholar] [CrossRef]
- Burnett, T.; Kelley, R.L.; Winiarski, B.; Contreras, L.; Daly, M.; Gholinia, A.; Burke, M.; Withers, P. Large volume serial section tomography by Xe Plasma FIB dual beam microscopy. Ultramicroscopy 2016, 161, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Shameer, P.M.; Nishath, P.M. Chapter 8—Exploration and enhancement on fuel stability of biodiesel: A step forward in the track of global commercialization. In Advanced Biofuels; Woodhead Publishing: Melbourne, Australia, 2019; pp. 181–213. [Google Scholar]
- Talari, A.C.S.; Martinez, M.A.G.; Movasaghi, Z.; Rehman, S.; Rehman, I.U. Advances in Fourier transform infrared [FTIR] spectroscopy of biological tissues. Appl. Spectrosc. Rev. 2017, 52, 456–506. [Google Scholar] [CrossRef]
- Sizeland, K.H.; Hofman, K.A.; Hallett, I.C.; Martin, D.E.; Potgieter, J.; Kirby, N.M.; Hawley, A.; Mudie, S.T.; Ryan, T.M.; Haverkamp, R.G.; et al. Nanostructure of electrospun collagen: Do electrospun collagen fibers form native structures? Materialia 2018, 3, 90–96. [Google Scholar] [CrossRef]
- Ingham, J.; Pilling, M.J.; Craig, T.; Siggel-King, M.R.F.; Smith, C.I.; Gardner, P.; Varro, A.; Pritchard, D.M.; Barrett, S.D.; Martin, D.S.; et al. An evaluation of the application of the aperture infrared SNOM technique to biomedical imaging. Biomed. Phys. Eng. Express 2018, 4, 025011. [Google Scholar] [CrossRef]
- Mester, L.; Govyadinov, A.A.; Chen, S.; Goikoetxea, M.; Hillenbrand, R. Subsurface chemical nanoidentification by nano-FTIR spectroscopy. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, O.P.; Kalita, P.C.; Doley, P.J.; Kalita, A. 2. Uses of Transmission Electron Microscope in Microscopy and its Advantages and Disadvantages. Available online: https://petsd.org/ojs/index.php/lifesciencesleaflets/article/view/1149 (accessed on 27 February 2021).
- Carter, M.; Shieh, J.C. Chapter 5—Microscopy. In Guide to Research Techniques in Neuroscience; Academic Press: New York, NY, USA, 2010. [Google Scholar]
- Ortega, D.R.; Oikonomou, C.M.; Ding, H.J.; Rees-Lee, P.; Jensen, G.J. Alexandria ETDB-Caltech: A blockchain-based distributed public database for electron tomography. PLoS ONE 2019, 14, e0215531. [Google Scholar] [CrossRef]
- Moll, P.J. Focused Ion Beam Microstructuring of Quantum Matter. Annu. Rev. Condens. Matter Phys. 2018, 9, 147–162. [Google Scholar] [CrossRef]
- Wirth, R. Focused Ion Beam (FIB) combined with SEM and TEM: Advanced analytical tools for studies of chemical composition, microstructure and crystal structure in geomaterials on a nanometre scale. Chem. Geol. 2009, 261, 217–229. [Google Scholar] [CrossRef]
- Gerke, K.M.; Korostilev, E.V.; Romanenko, K.A.; Karsanina, M.V. Going submicron in the precise analysis of soil structure: A FIB-SEM imaging study at nanoscale. Geoderma 2021, 383, 114739. [Google Scholar] [CrossRef]
- Dutta, A. Chapter 4—Fourier Transform Infrared Spectroscopy. In Spectroscopic Methods for Nanomaterials Characterization; Thomas, S., Thomas, R., Zachariah, A.K., Mishra, R.K., Eds.; Elsevier: Aurangabad, India, 2017; pp. 73–93. [Google Scholar]
- Wallace, H.A.; Basehore, B.M.; Zito, P.M. Wound Healing Phases; StatPearls Publishing LLC: Treasure Island, FL, USA, 2019. [Google Scholar]
- Barki, K.G.; Das, A.; Dixith, S.; Das Ghatak, P.; Mathew-Steiner, S.; Schwab, E.; Khanna, S.; Wozniak, D.J.; Roy, S.; Sen, C.K. Electric Field Based Dressing Disrupts Mixed-Species Bacterial Biofilm Infection and Restores Functional Wound Healing. Ann. Surg. 2019, 269, 756–766. [Google Scholar] [CrossRef]
- Schimek, K.; Hsu, H.-H.; Boehme, M.; Kornet, J.J.; Marx, U.; Lauster, R.; Pörtner, R.; Lindner, G. Bioengineering of a Full-Thickness Skin Equivalent in a 96-Well Insert Format for Substance Permeation Studies and Organ-On-A-Chip Applications. Bioengineering 2018, 5, 43. [Google Scholar] [CrossRef]
- Dhakshinamurthy, I. A Comparative Study of Novel Stem Cell Therapy-Based Treatments for Diabetic Foot Ulcers. Master’s Thesis, Yale University, New Haven, CT, USA, 19 June 2020. [Google Scholar]
- Widjaja, W.; Tan, J.; Maitz, P.K.M. Efficacy of dermal substitute on deep dermal to full thickness burn injury: A systematic review. ANZ J. Surg. 2017, 87, 446–452. [Google Scholar] [CrossRef]
- Munish, T.; Ramneesh, G.; Sanjeev, S.; Jasdeep, S.; Jaspal, S.; Nikhil, G. Comparative Study of Collagen Based Dressing and Standard Dressing in Diabetic Foot Ulcer. J. Evol. Med. Dent. Sci. 2015, 4, 3614–3621. [Google Scholar] [CrossRef]
- Sahana, T.G.; Rekha, P.D. Biopolymers: Applications in wound healing and skin tissue engineering. Mol. Biol. Rep. 2018, 45, 2857–2867. [Google Scholar] [CrossRef] [PubMed]
- Ovais, M.; Ahmad, I.; Khalil, A.T.; Mukherjee, S.; Javed, R.; Ayaz, M.; Raza, A.; Shinwari, Z.K. Wound healing applications of biogenic colloidal silver and gold nanoparticles: Recent trends and future prospects. Appl. Microbiol. Biotechnol. 2018, 102, 4305–4318. [Google Scholar] [CrossRef]
- Marcato, P.D.; De Paula, L.B.; Melo, P.S.; Ferreira, I.R.; Almeida, A.B.A.; Torsoni, A.S.; Alves, O.L. In Vivo Evaluation of Complex Biogenic Silver Nanoparticle and Enoxaparin in Wound Healing. J. Nanomater. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Paladini, F.; Pollini, M. Antimicrobial Silver Nanoparticles for Wound Healing Application: Progress and Future Trends. Materials 2019, 12, 2540. [Google Scholar] [CrossRef]
- Natarajan, D.; Kiran, M.S. Fabrication of juglone functionalized silver nanoparticle stabilized collagen scaffolds for pro-wound healing activities. Int. J. Biol. Macromol. 2019, 124, 1002–1015. [Google Scholar] [CrossRef]
- You, C.; Liping, Z.; Wang, X.; Wu, P.; Ho, J.K.; Jin, R.; Zhang, L.; Shao, H.; Han, C. Silver nanoparticle loaded collagen/chitosan scaffolds promote wound healing via regulating fibroblast migration and macrophage activation. Sci. Rep. 2017, 7, 10489. [Google Scholar] [CrossRef] [PubMed]
- Navani, N.K.; Lambadi, P.R.; Sharma, T.K.; Kumar, P.; Vasnani, P.; Thalluri, S.M.; Bisht, N.; Pathania, R. Facile biofunctionalization of silver nanoparticles for enhanced antibacterial properties, endotoxin removal, and biofilm control. Int. J. Nanomed. 2015, 10, 2155–2171. [Google Scholar] [CrossRef]
- Singh, B.R.; Singh, B.N.; Singh, A.; Khan, W.A.; Naqvi, A.H.; Singh, H.B. Mycofabricated biosilver nanoparticles interrupt Pseudomonas aeruginosa quorum sensing systems. Sci. Rep. 2015, 5, srep13719. [Google Scholar] [CrossRef] [PubMed]
- Karri, V.V.S.R.; Kuppusamy, G.; Talluri, S.V.; Mannemala, S.S.; Kollipara, R.; Wadhwani, A.D.; Mulukutla, S.; Raju, K.R.S.; Malayandi, R. Curcumin loaded chitosan nanoparticles impregnated into collagen-alginate scaffolds for diabetic wound healing. Int. J. Biol. Macromol. 2016, 93, 1519–1529. [Google Scholar] [CrossRef]
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound dressings–a review. Biomedicine 2015, 5, 22. [Google Scholar] [CrossRef]
- Pringgandini, L.A.; Indarti, G.Y.; Melinda, M.; Sari, M. Efektivitas spray nanokolagen limbah sisik ikan mas (Cyprinus carpio) untuk mempercepat proses penyembuhan luka insisiEffectiveness of nano-collagen spray of goldfish (Cyprinus carpio) scales waste in accelerating the incision wound healing process. J. Kedokt. Gigi Univ. Padjadjaran 2018, 30, 113–120. [Google Scholar] [CrossRef]
- Kochar, M.P.; Singh, S.P. Role of nano-collagen particles dressing in the management of chronic ulcer: A prospective non-randomized trial on 100 cases. Int. Surg. J. 2020, 7, 802–806. [Google Scholar] [CrossRef][Green Version]
- Katsimbri, P. The biology of normal bone remodelling. Eur. J. Cancer Care 2017, 26, e12740. [Google Scholar] [CrossRef] [PubMed]
- De Melo Pereira, D.; Eischen-Loges, M.; Birgani, Z.T.; Habibovic, P. Proliferation and Osteogenic Differentiation of hMSCs on Bio-mineralized Collagen. Front. Bioeng. Biotechnol. 2020, 8, 1248. [Google Scholar] [CrossRef]
- Winkler, T.; Sass, F.; Duda, G.; Schmidt-Bleek, K. A review of biomaterials in bone defect healing, remaining shortcomings and future opportunities for bone tissue engineering: The unsolved challenge. Bone Joint J. 2018, 7, 232–243. [Google Scholar] [CrossRef]
- Sun, C.Y.; Che, Y.J.; Lu, S.J. Preparation and application of collagen scaffold-encapsulated silver nanoparticles and bone mor-phogenetic protein 2 for enhancing the repair of infected bone. Biotechnol. Lett. 2015, 37, 467–473. [Google Scholar] [CrossRef]
- Ma, S.; Adayi, A.; Liu, Z.; Li, M.; Wu, M.; Xiao, L.; Sun, Y.; Cai, Q.; Yang, X.; Zhang, X.; et al. Asymmetric Collagen/chitosan Membrane Containing Minocycline-loaded Chitosan Nanoparticles for Guided Bone Regeneration. Sci. Rep. 2016, 6, 31822. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Zhao, Y.; Yang, M.; Wang, S.F. Nano-collagen artificial bone for alveolar ridge preservation in the Kazakh from Xinjiang Tacheng Region. Chin. J. Tissue Eng. Res. 2015, 19, 1864–1871. [Google Scholar]
- Shen, L.; Wang, X.; Chen, P.; Tong, Y. Allogeneic adipose-derived stem cells combined with nano collagen-based bone for repair of ulna bone defects. Chin. J. Tissue Eng. Res. 2015, 13, 5162–5166. [Google Scholar]
- Jahangirian, H.; Lemraski, E.G.; Webster, T.J.; Rafiee-Moghaddam, R.; Abdollahi, Y. A review of drug delivery systems based on nanotechnology and green chemistry: Green nanomedicine. Int. J. Nanomed. 2017, 12, 2957–2978. [Google Scholar] [CrossRef] [PubMed]
- DeFrates, K.; Markiewicz, T.; Gallo, P.; Rack, A.; Weyhmiller, A.; Jarmusik, B.; Hu, X. Protein Polymer-Based Nanoparticles: Fabrication and Medical Applications. Int. J. Mol. Sci. 2018, 19, 1717. [Google Scholar] [CrossRef]
- Le, V.-M.; Lang, M.-D.; Shi, W.-B.; Liu, J.-W. A collagen-based multicellular tumor spheroid model for evaluation of the efficiency of nanoparticle drug delivery. Artif. Cells Nanomed. Biotechnol. 2014, 44, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Hoang, G.; Manivasagan, P.; Moorthy, M.S.; Phan, T.T.V.; Kim, H.H.; Nguyen, T.P.; Oh, J. Rapid microwave-assisted synthesis of gold loaded hydroxyapatite collagen nano-bio materials for drug delivery and tissue engineering application. Ceram. Int. 2019, 45, 2977–2988. [Google Scholar] [CrossRef]
- Anandhakumar, S.; Krishnamoorthy, G.; Ramkumar, K.; Raichur, A. Preparation of collagen peptide functionalized chitosan nanoparticles by ionic gelation method: An effective carrier system for encapsulation and release of doxorubicin for cancer drug delivery. Mater. Sci. Eng. C 2017, 70, 378–385. [Google Scholar] [CrossRef]
- Orasugh, J.T.; Dutta, S.; Das, D.; Pal, C.; Zaman, A.; Das, S.; Dutta, K.; Banerjee, R.; Ghosh, S.K.; Chattopadhyay, D. Sustained release of ketorolac tromethamine from poloxamer 407/cellulose nanofibrils graft nanocollagen based ophthalmic formulations. Int. J. Biol. Macromol. 2019, 140, 441–453. [Google Scholar] [CrossRef]
- Mohamadi, F.; Ebrahimi-Barough, S.; Nourani, M.R.; Derakhshan, M.A.; Goodarzi, V.; Nazockdast, M.S.; Farokhi, M.; Tajerian, R.; Majidi, R.F.; Ai, J. Electrospun nerve guide scaffold of poly[ε-caprolactone]/collagen/nanobioglass: An in vitro study in peripheral nerve tissue engineering. J. Biomed. Mater. Res. Part A 2017, 105, 1960–1972. [Google Scholar] [CrossRef]
- Bhangra, K.S. Delivery and Integration of Engineered Neural Tissue for Peripheral Nerve Repair. Ph.D. Thesis, University College London, London, UK, 2020. [Google Scholar]
- Wei, D.; Sun, J.; Yang, Y.; Wu, C.; Chen, S.; Guo, Z.; Fan, H.; Zhang, X. Cell alignment guided by nano/micro oriented collagen fibers and the synergistic vascularization for nervous cell functional expression. Mater. Today Chem. 2018, 8, 85–95. [Google Scholar] [CrossRef]
- Humpel, C.; Ucar, B. Collagen for brain repair: Therapeutic perspectives. Neural Regen. Res. 2018, 13, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, X.; Li, Z.; Bai, Y.; Liao, G.; Pan, J.; Zhang, C. Collagen/nano-sized β-tricalcium phosphate conduits combined with col-lagen filaments and nerve growth factor promote facial nerve regeneration in miniature swine: An in vivo study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 128, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Pashneh-Tala, S.; MacNeil, S.; Claeyssens, F. The Tissue-Engineered Vascular Graft—Past, Present, and Future. Tissue Eng. Part B Rev. 2016, 22, 68–100. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, J.; Lee, M.-K.; Park, C.; Jung, H.-D.; Kim, H.-E.; Jang, T.-S. Fabrication of strong, bioactive vascular grafts with PCL/collagen and PCL/silica bilayers for small-diameter vascular applications. Mater. Des. 2019, 181. [Google Scholar] [CrossRef]
- Fouad, H.; Al-Shammari, B.A.; Alrez, M.F.; Al-Fotawi, R.; Mahmood, A. Modified Bi-Layered Polycaprolactone Nanofiber Scaffolds for Vascular Tissue Engineering Applications. Nanosci. Nanotechnol. Lett. 2019, 11, 1–10. [Google Scholar] [CrossRef]
- Carballo, C.B.; Nakagawa, Y.; Sekiya, I.; Rodeo, S.A. Basic Science of Articular Cartilage. Clin. Sports Med. 2017, 36, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Armiento, A.; Stoddart, M.; Alini, M.; Eglin, D. Biomaterials for articular cartilage tissue engineering: Learning from biology. Acta Biomater. 2018, 65, 1–20. [Google Scholar] [CrossRef]
- Kaviani, A.; Zebarjad, S.M.; Javadpour, S.; Ayatollahi, M.; Bazargan-Lari, R. Fabrication and characterization of low-cost freeze-gelated chitosan/collagen/hydroxyapatite hydrogel nanocomposite scaffold. Int. J. Polym. Anal. Charact. 2019, 24, 191–203. [Google Scholar] [CrossRef]
- Jiang, X.; Zhong, Y.; Zheng, L.; Zhao, J. Nano-hydroxyapatite/collagen film as a favorable substrate to maintain the phenotype and promote the growth of chondrocytes cultured in�vitro. Int. J. Mol. Med. 2018, 41, 2150–2158. [Google Scholar] [CrossRef]
- Irawan, V.; Sung, T.-C.; Higuchi, A.; Ikoma, T. Collagen Scaffolds in Cartilage Tissue Engineering and Relevant Approaches for Future Development. Tissue Eng. Regen. Med. 2018, 15, 673–697. [Google Scholar] [CrossRef]
- Trilaksani, W.; Adnyane, I.K.M.; Riyanto, B.; Safitri, N. Nano collagen of the grouper swim bladder in compliance with quality standard of cosmetics materials. In Proceedings of the IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2019; Volume 404, p. 012050. [Google Scholar]
- Shekhter, A.B.; Fayzullin, A.L.; Vukolova, M.N.; Rudenko, T.G.; Osipycheva, V.D.; Litvitsky, P.F. Medical Applications of Collagen and Collagen-Based Materials. Curr. Med. Chem. 2019, 26, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Avila Rodríguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, X.; Li, T.; Yang, H.; Zhang, H.; Regenstein, J.M.; Zhou, P. Extraction and characterization of acid- and pepsin-soluble collagens from the scales, skins and swim-bladders of grass carp [Ctenopharyngodon idella]. Food Biosci. 2015, 9, 68–74. [Google Scholar] [CrossRef]
- Rauta, P.R.; Mohanta, Y.K.; Nayak, D. Nanotechnology in Biology and Medicine: Research Advancements & Future Perspectives; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Hamdan, S.; Pastar, I.; Drakulich, S.; Dikici, E.; Tomic-Canic, M.; Deo, S.; Daunert, S. Nanotechnology-Driven Therapeutic Interventions in Wound Healing: Potential Uses and Applications. ACS Cent. Sci. 2017, 3, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Senturk, B.; Uzunalli, G.; Mammadov, R.; Guler, M.O.; Tekinay, A.B. Wound Healing Applications of Nanomaterials. Ther. Nanomater. 2016, 2016, 87–117. [Google Scholar] [CrossRef]
| Characterisation Techniques | Advantages | Disadvantages |
|---|---|---|
| Transmission electron microscopy (TEM) | Straightforward sample preparation technique. Unsusceptible to radiation. | Presents structural artifacts from staining and dehydration [66]. |
| Electron tomography (ET) | Produces 3D structural organisation data of sample. | Cannot be tested on live specimens [67]. Produces low-quality reconstructions of data [68]. |
| Scanning electron microscopy (SEM) | Images 3D topographic data of samples. | Can cause structural abnormalities in samples from harsh preparation processes [67]. |
| Focused ion beam (FIB) spectroscopy | 2D samples can be imaged at nanoscale. | Ion beam emission causes surface damage on samples [69]. |
| FIB-TEM | Allows site-specific sample preparation. | Sample surface damage from ion implantation [70]. |
| FIB-SEM | Produces detailed surface images of 2D flat milled samples. | Limited sample processing methods. Expensive to conduct [71]. |
| Fourier transform infrared (FTIR) spectroscopy | Fast and sensitive quantitative analysis technique. | Uses only a single beam for quantitative analysis [72]. |
| Author(s) | Nano Collagen Source | Summary of Intervention | Results |
|---|---|---|---|
| Pringgandini LA et al., 2018 [89] | Goldfish scales (Cyprinus carpio) | Collagen extraction and purification followed by freeze-drying, producing nano collagen powder spray. | Accelerated healing of incision wound in mice. |
| Kochar MP et al., 2020 [90] | Collofiber-MM™, collagen type I, naturally sourced (not specified) | Fabrication process not stated, powder product used consists of collagen nano particles, mupirocin and metronidazole and was applied onto chronic ulcers of 100 patients. | Re-epithelialisation and early ulcer healing, reduces exudate secretion and bacterial colonisation. |
| Van Duong H et al., 2018 [49] | Catfish skin waste (Pangasius bocourti) | Fabrication of water-dispersible nano collagen helices, can be made into sponge-like aerogels. | Highly versatile applications as wound dressings, bandages and tissue-engineering scaffolds. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo, S.; Fauzi, M.B. Current Update of Collagen Nanomaterials—Fabrication, Characterisation and Its Applications: A Review. Pharmaceutics 2021, 13, 316. https://doi.org/10.3390/pharmaceutics13030316
Lo S, Fauzi MB. Current Update of Collagen Nanomaterials—Fabrication, Characterisation and Its Applications: A Review. Pharmaceutics. 2021; 13(3):316. https://doi.org/10.3390/pharmaceutics13030316
Chicago/Turabian StyleLo, Samantha, and Mh Busra Fauzi. 2021. "Current Update of Collagen Nanomaterials—Fabrication, Characterisation and Its Applications: A Review" Pharmaceutics 13, no. 3: 316. https://doi.org/10.3390/pharmaceutics13030316
APA StyleLo, S., & Fauzi, M. B. (2021). Current Update of Collagen Nanomaterials—Fabrication, Characterisation and Its Applications: A Review. Pharmaceutics, 13(3), 316. https://doi.org/10.3390/pharmaceutics13030316