Susceptibility of Dental Caries Microcosm Biofilms to Photodynamic Therapy Mediated by Fotoenticine
Abstract
:1. Introduction
2. Materials and Methods
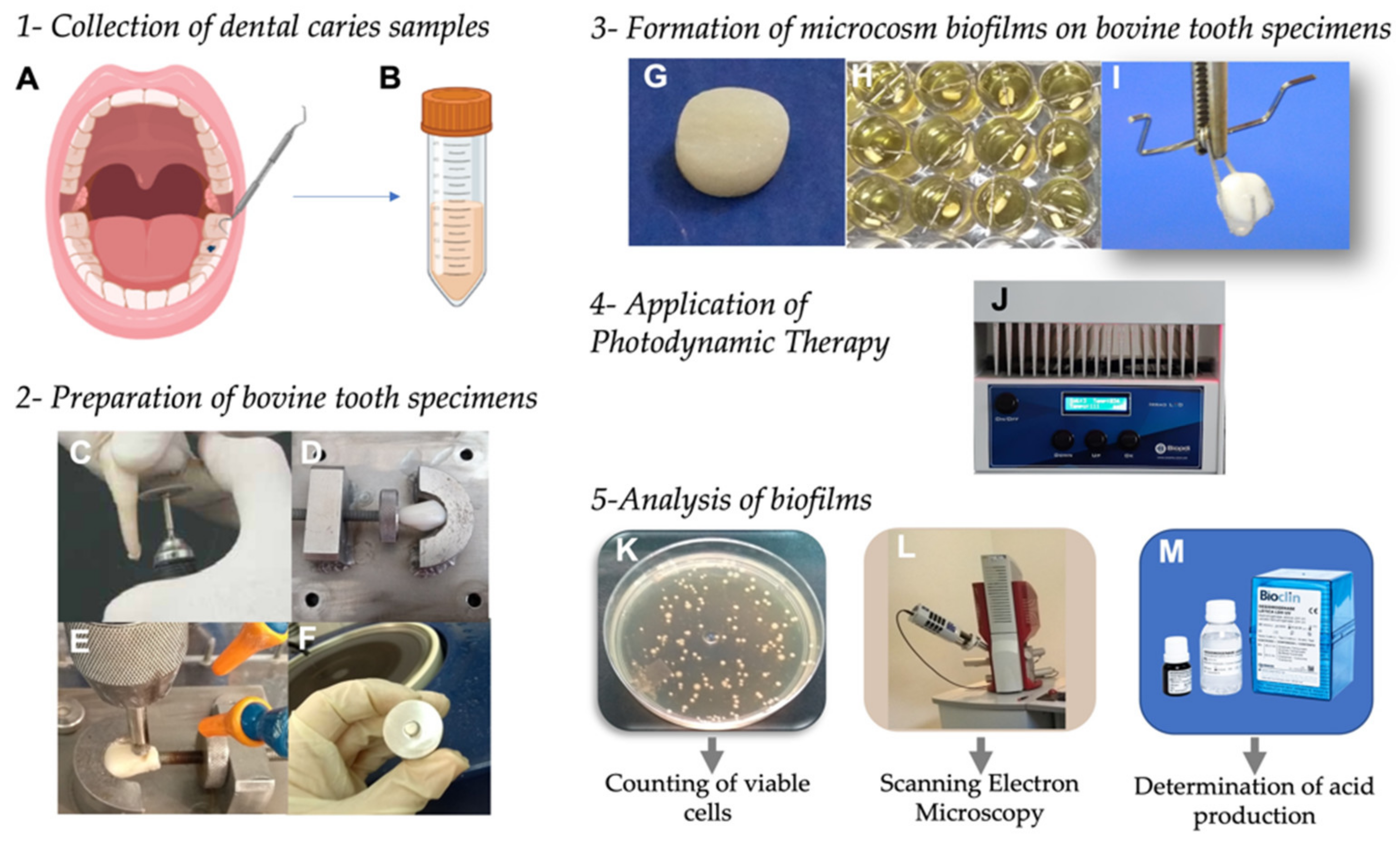
2.1. Collection of Dental Caries Samples
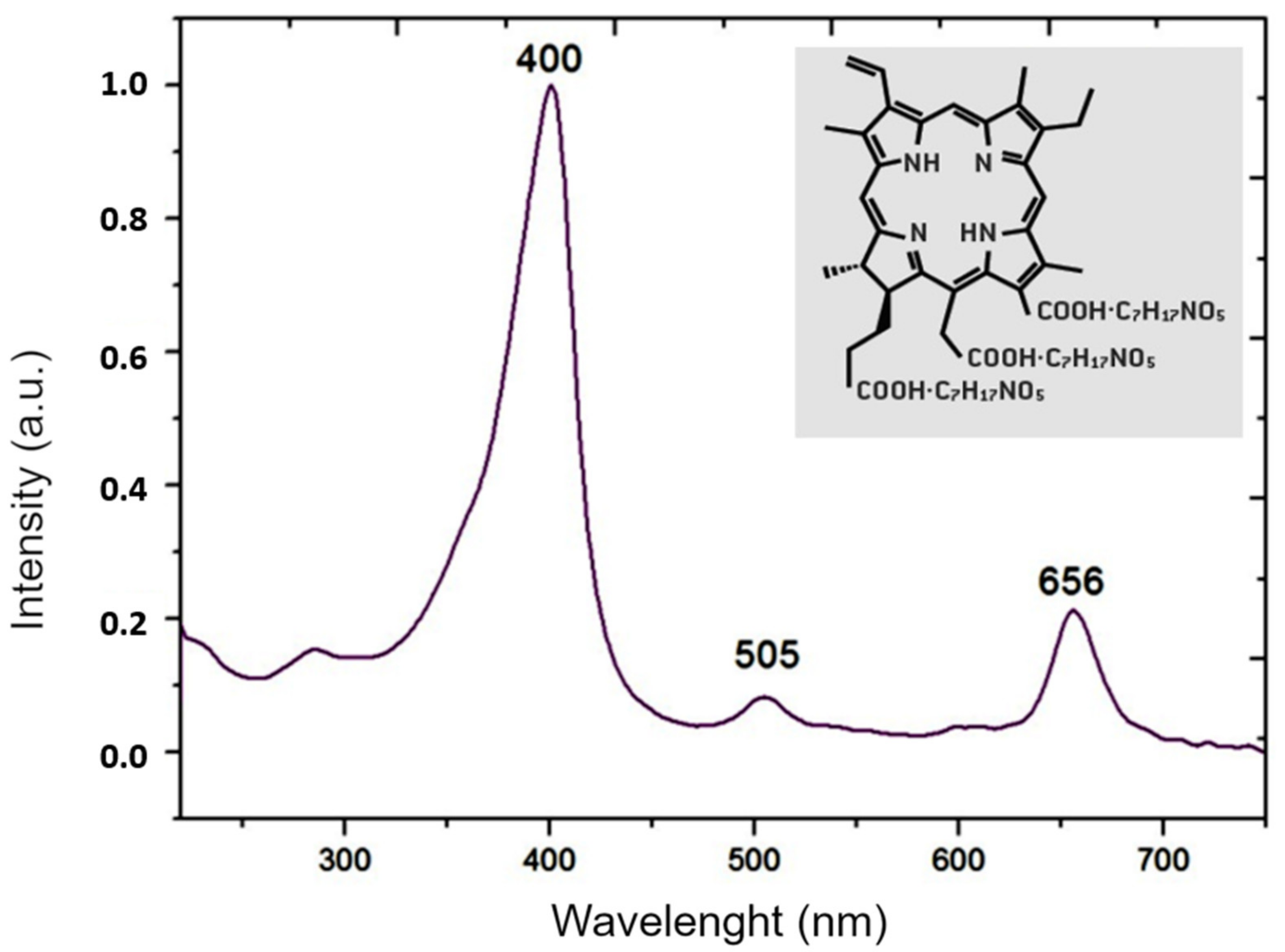
2.2. Preparation of Photosensitizer and Light Source
2.3. Preparation of Bovine Tooth Specimens
2.4. Formation of Microcosm Biofilms on Bovine Tooth Specimens
2.5. Application of Photodynamic Therapy on Microcosm Biofilms
2.6. Analysis of Biofilms by Counting the Number of Viable Cells
2.7. Analysis of Biofilms by Scanning Electron Microscopy (SEM)
2.8. Determination of Lactic Acid Production by the Microbial Cells in Biofilms
2.9. Statistical Analysis
3. Results
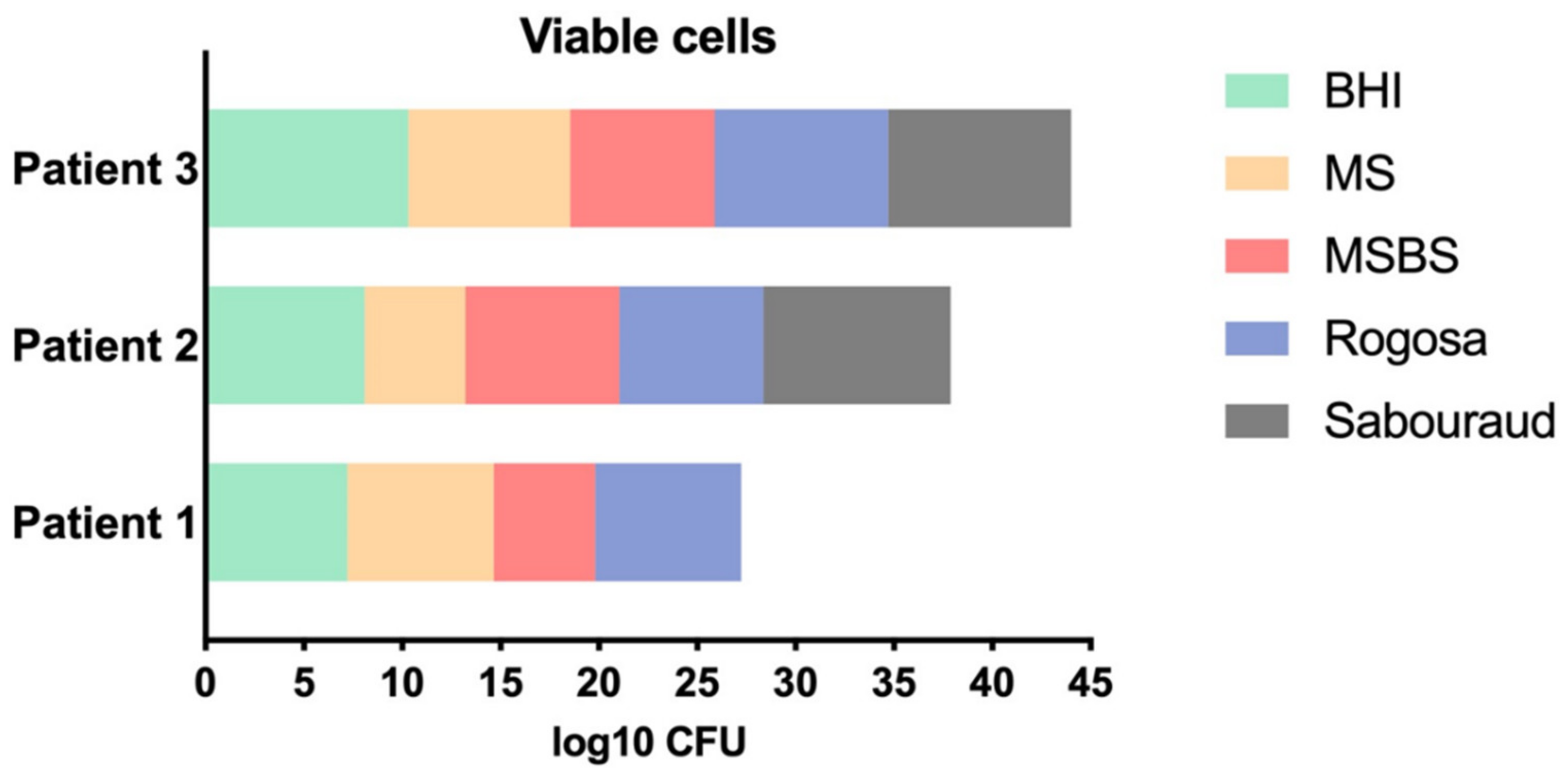
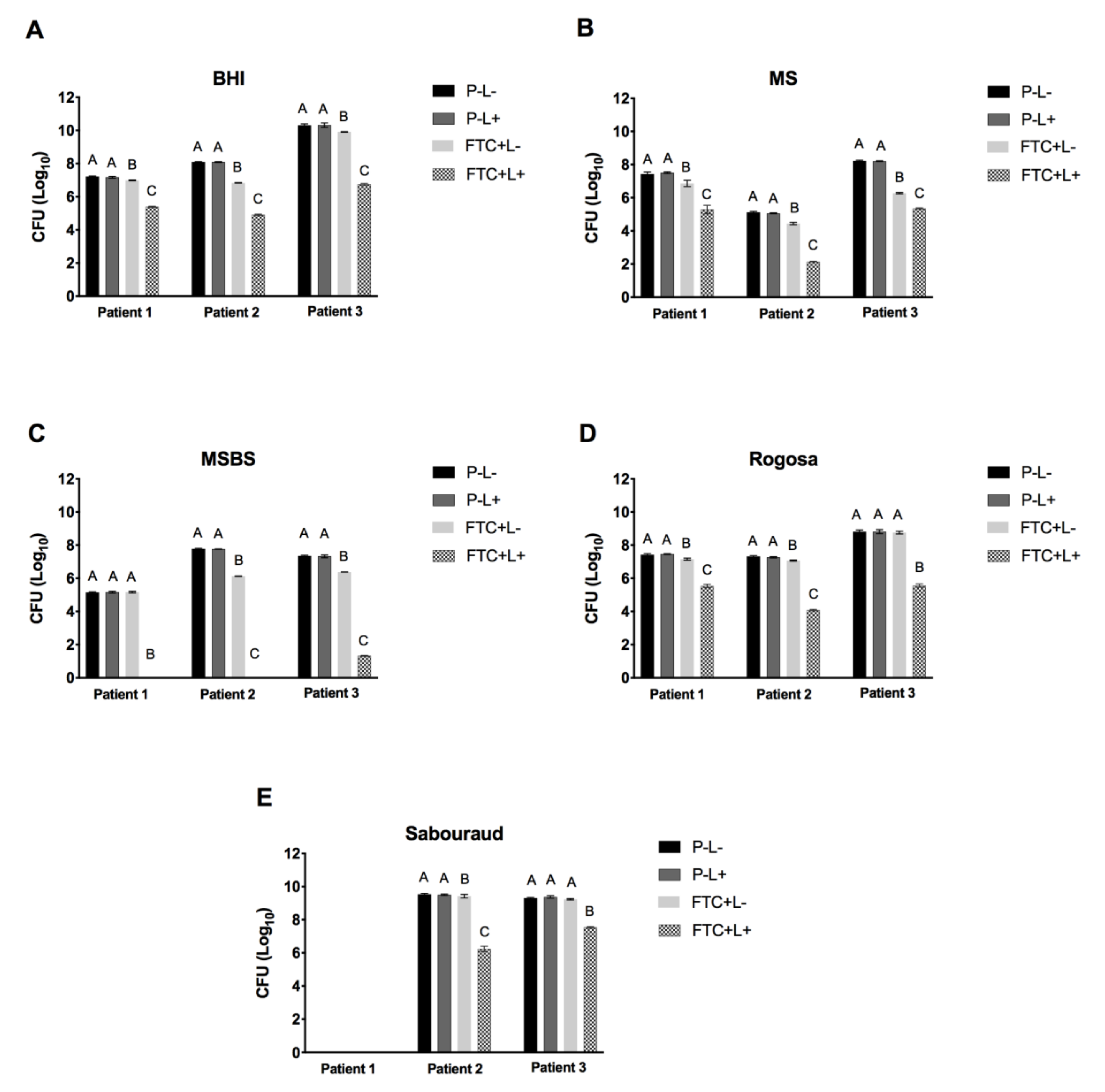
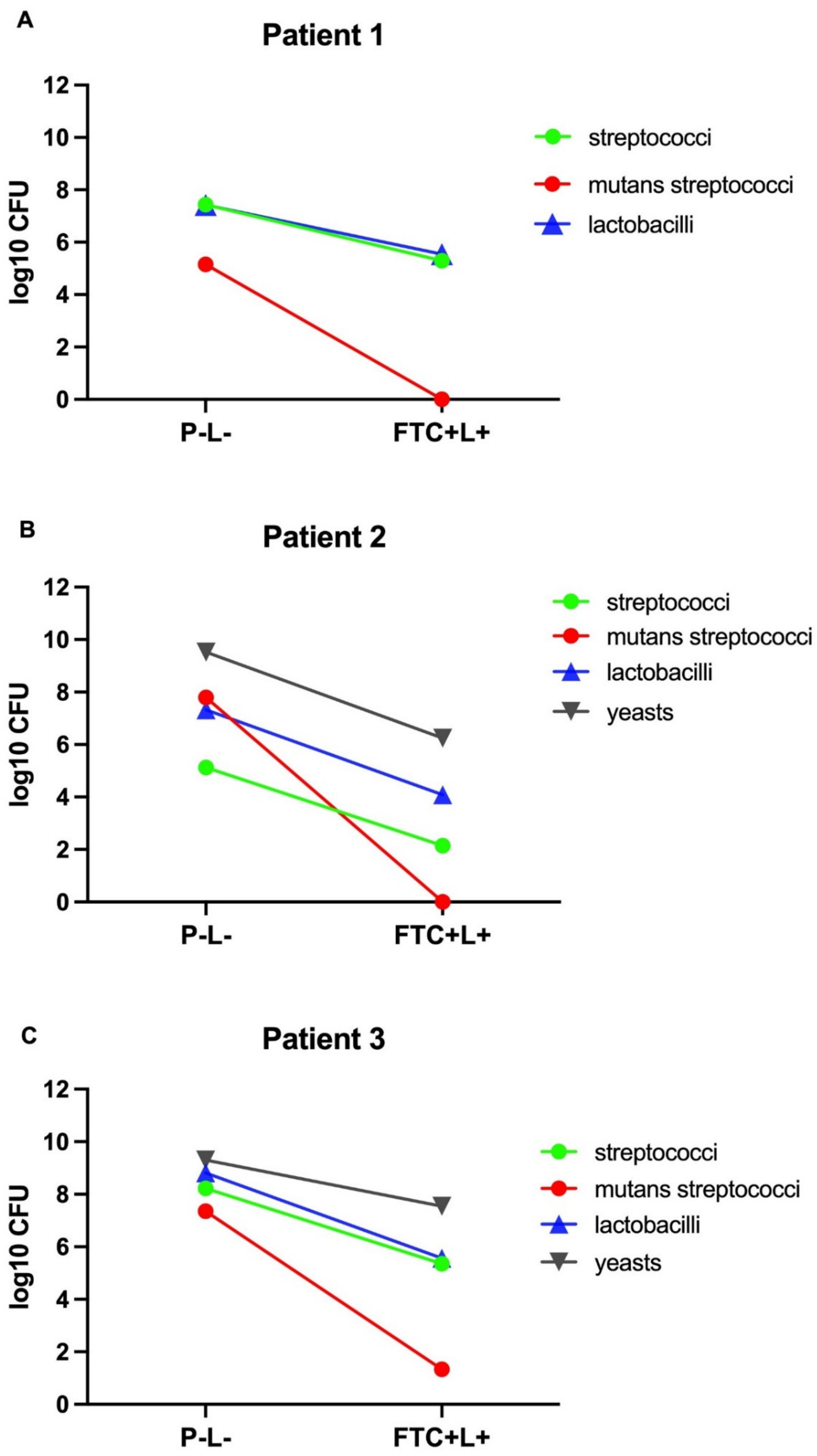
3.1. Effects of FTC-Mediated PDT on Viable Cells of Microcosm Biofilms
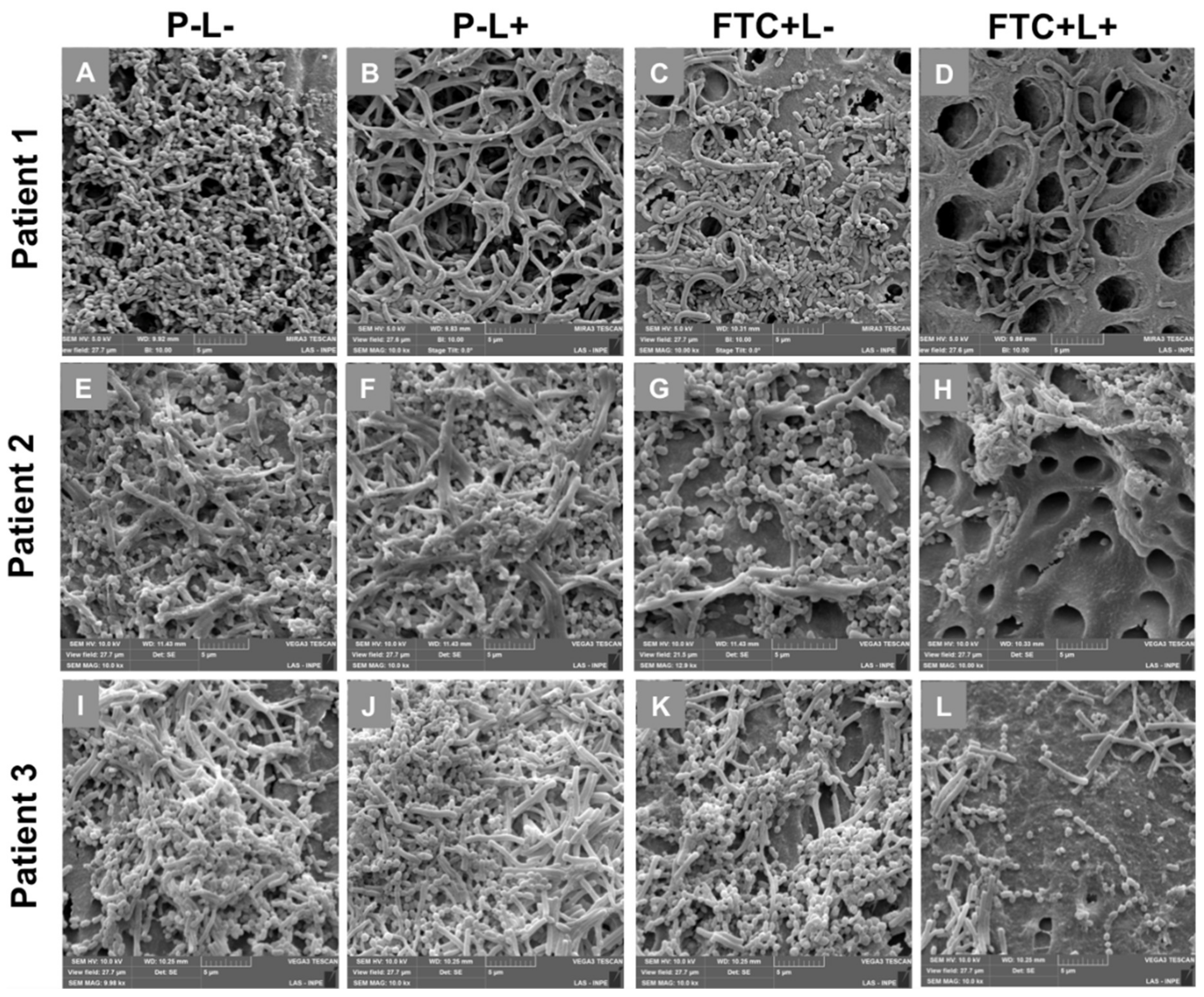
3.2. Effects of FTC-Mediated PDT on Microcosm Biofilm Structures
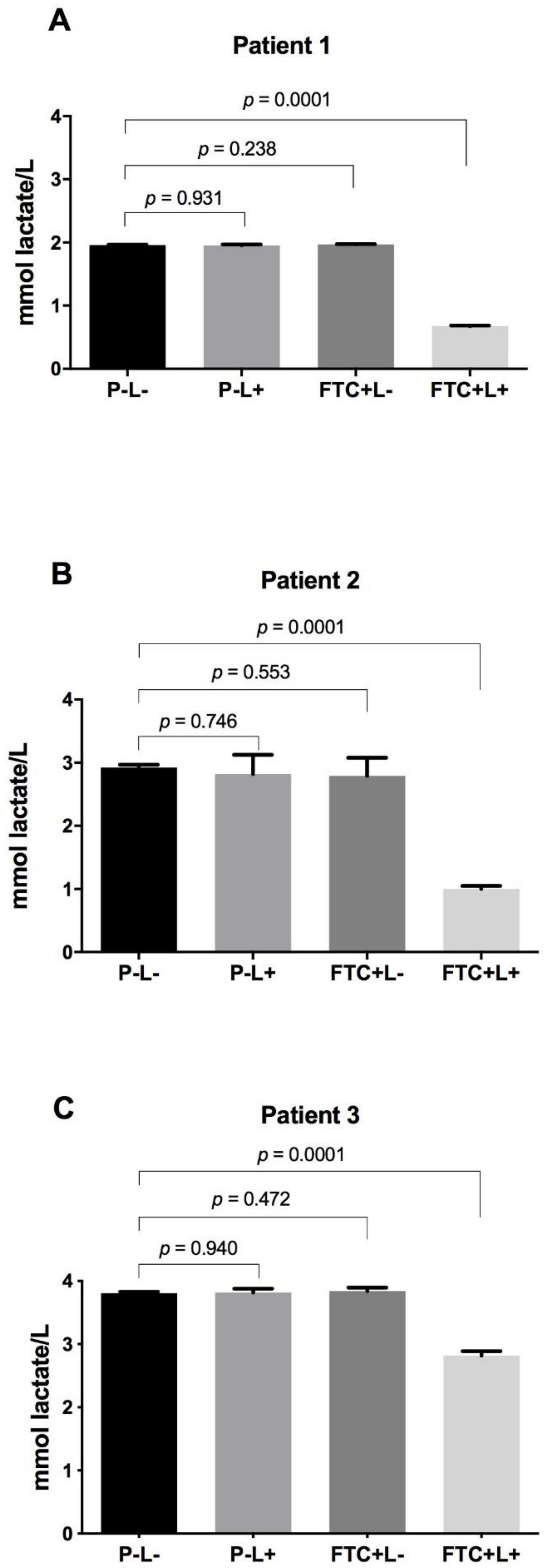
3.3. Influence of FTC-Mediated PDT on Acid Production by the Microbial Cells of Microcosm Biofilms
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Mathur, V.P.; Dhillon, J.K. Dental Caries: A Disease Which Needs Attention. Indian J. Pediatrics 2018, 85, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Daliri, E.B.; Tyagi, A.; Oh, D.H. Cariogenic Biofilm: Pathology-Related Phenotypes and Targeted Therapy. Microorganisms 2021, 9, 1311. [Google Scholar] [CrossRef] [PubMed]
- Esberg, A.; Sheng, N.; Marell, L.; Claesson, R.; Persson, K.; Boren, T.; Stromberg, N. Streptococcus mutans Adhesin Biotypes that Match and Predict Individual Caries Development. EBioMedicine 2017, 24, 205–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Ma, Q.; Wang, Y.; Wu, H.; Zou, J. Molecular mechanisms of inhibiting glucosyltransferases for biofilm formation in Streptococcus mutans. Int. J. Oral Sci. 2021, 13, 30. [Google Scholar] [CrossRef]
- Avoaka-Boni, M.C.; Djolé, S.X.; Kaboré, W.A.D.; Gnagne-Koffi, Y.N.; Koffi, A.F. The causes of failure and the longevity of direct coronal restorations: A survey among dental surgeons of the town of Abidjan, Cote d’Ivoire. J. Conserv. Dent. 2019, 22, 270–274. [Google Scholar] [CrossRef]
- Godbole, E.; Tyagi, S.; Kulkarni, P.; Singla, S.; Mali, S.; Helge, S. Efficacy of Liquorice and Propolis Extract Used as Cavity Cleaning Agents against Streptococcus mutans in Deciduous Molars Using Confocal Microscopy: An In Vitro Study. Int. J. Clin. Pediatric Dent. 2019, 12, 194–200. [Google Scholar] [CrossRef]
- Galdino, D.Y.T.; da Rocha Leodido, G.; Pavani, C.; Goncalves, L.M.; Bussadori, S.K.; Benini Paschoal, M.A. Photodynamic optimization by combination of xanthene dyes on different forms of Streptococcus mutans: An in vitro study. Photodiagnosis Photodyn. Ther. 2021, 33, 102191. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy—What we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef] [Green Version]
- Terra Garcia, M.; Correia Pereira, A.H.; Figueiredo-Godoi, L.M.A.; Jorge, A.O.C.; Strixino, J.F.; Junqueira, J.C. Photodynamic therapy mediated by chlorin-type photosensitizers against Streptococcus mutans biofilms. Photodiagnosis Photodyn. Ther. 2018, 24, 256–261. [Google Scholar] [CrossRef]
- Marcolan De Mello, M.; De Barros, P.P.; de Cassia Bernardes, R.; Alves, S.R.; Ramanzini, N.P.; Figueiredo-Godoi, L.M.A.; Prado, A.C.C.; Jorge, A.O.C.; Junqueira, J.C. Antimicrobial photodynamic therapy against clinical isolates of carbapenem-susceptible and carbapenem-resistant Acinetobacter baumannii. Lasers Med. Sci. 2019, 34, 1755–1761. [Google Scholar] [CrossRef]
- De Souza, C.M.; Garcia, M.T.; de Barros, P.P.; Pedroso, L.L.C.; da Costa Ward, R.A.; Strixino, J.F.; Melo, V.M.M.; Junqueira, J.C. Chitosan enhances the Antimicrobial Photodynamic Inactivation mediated by Photoditazine(R) against Streptococcus mutans. Photodiagnosis Photodyn. Ther. 2020, 32, 102001. [Google Scholar] [CrossRef]
- Martins Antunes de Melo, W.C.; Celiesiute-Germaniene, R.; Simonis, P.; Stirke, A. Antimicrobial photodynamic therapy (aPDT) for biofilm treatments. Possible synergy between aPDT and pulsed electric fields. Virulence 2021, 12, 2247–2272. [Google Scholar] [CrossRef]
- Hu, X.; Huang, Y.Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial Photodynamic Therapy to Control Clinically Relevant Biofilm Infections. Front. Microbiol. 2018, 9, 1299. [Google Scholar] [CrossRef] [Green Version]
- Santin, G.C.; Oliveira, D.S.; Galo, R.; Borsatto, M.C.; Corona, S.A. Antimicrobial photodynamic therapy and dental plaque: A systematic review of the literature. Sci. World J. 2014, 2014, 824538. [Google Scholar] [CrossRef] [Green Version]
- Lacerda Rangel Esper, M.A.; Junqueira, J.C.; Uchoa, A.F.; Bresciani, E.; Nara de Souza Rastelli, A.; Navarro, R.S.; de Paiva Goncalves, S.E. Photodynamic inactivation of planktonic cultures and Streptococcus mutans biofilms for prevention of white spot lesions during orthodontic treatment: An in vitro investigation. Am. J. Orthod. Dentofac. Orthop. 2019, 155, 243–253. [Google Scholar] [CrossRef]
- Chan, B.C.L.; Dharmaratne, P.; Wang, B.; Lau, K.M.; Lee, C.C.; Cheung, D.W.S.; Chan, J.Y.W.; Yue, G.G.L.; Lau, C.B.S.; Wong, C.K.; et al. Hypericin and Pheophorbide a Mediated Photodynamic Therapy Fighting MRSA Wound Infections: A Translational Study from In Vitro to In Vivo. Pharmaceutics 2021, 13, 1399. [Google Scholar] [CrossRef]
- Hamblin, M.R. Antimicrobial photodynamic inactivation: A bright new technique to kill resistant microbes. Curr. Opin. Microbiol. 2016, 33, 67–73. [Google Scholar] [CrossRef] [Green Version]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [Green Version]
- Nie, M.; Deng, D.M.; Wu, Y.; de Oliveira, K.T.; Bagnato, V.S.; Crielaard, W.; Rastelli, A.N.S. Photodynamic inactivation mediated by methylene blue or chlorin e6 against Streptococcus mutans biofilm. Photodiagnosis Photodyn. Ther. 2020, 31, 101817. [Google Scholar] [CrossRef]
- Terra-Garcia, M.; de Souza, C.M.; Ferreira Goncalves, N.M.; Pereira, A.H.C.; de Barros, P.P.; Borges, A.B.; Miyakawa, W.; Strixino, J.F.; Junqueira, J.C. Antimicrobial effects of photodynamic therapy with Fotoenticine on Streptococcus mutans isolated from dental caries. Photodiagnosis Photodyn. Ther. 2021, 34, 102303. [Google Scholar] [CrossRef]
- Salli, K.M.; Ouwehand, A.C. The use of in vitro model systems to study dental biofilms associated with caries: A short review. J. Oral Microbiol. 2015, 7, 26149. [Google Scholar] [CrossRef] [PubMed]
- Pires, J.G.; Braga, A.S.; Andrade, F.B.; Saldanha, L.L.; Dokkedal, A.L.; Oliveira, R.C.; Magalhaes, A.C. Effect of hydroalcoholic extract of Myracrodruon urundeuva All. and Qualea grandiflora Mart. leaves on the viability and activity of microcosm biofilm and on enamel demineralization. J. Appl. Oral Sci. 2019, 27, e20180514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chevalier, M.; Ranque, S.; Precheur, I. Oral fungal-bacterial biofilm models in vitro: A review. Med. Mycol. 2018, 56, 653–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendez, D.A.C.; Gutierrez, E.; Dionisio, E.J.; Oliveira, T.M.; Buzalaf, M.A.R.; Rios, D.; Machado, M.; Cruvinel, T. Effect of methylene blue-mediated antimicrobial photodynamic therapy on dentin caries microcosms. Lasers Med. Sci. 2018, 33, 479–487. [Google Scholar] [CrossRef]
- De Almeida, R.M.S.; Fontana, L.C.; Dos Santos Vitorio, G.; Pereira, A.H.C.; Soares, C.P.; Pinto, J.G.; Ferreira-Strixino, J. Analysis of the effect of photodynamic therapy with Fotoenticine on gliosarcoma cells. Photodiagnosis Photodyn. Ther. 2020, 30, 101685. [Google Scholar] [CrossRef]
- Fysal, N.; Jose, S.; Kulshrestha, R.; Arora, D.; Hafiz, K.A.; Vasudevan, S. Antibiogram pattern of oral microflora in periodontic children of age group 6 to 12 years: A clinicomicrobiological study. J. Contemp. Dent. Pract. 2013, 14, 595–600. [Google Scholar] [CrossRef]
- Mosaddad, S.A.; Tahmasebi, E.; Yazdanian, A.; Rezvani, M.B.; Seifalian, A.; Yazdanian, M.; Tebyanian, H. Oral microbial biofilms: An update. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 2005–2019. [Google Scholar] [CrossRef]
- Kim, D.; Koo, H. Spatial Design of Polymicrobial Oral Biofilm in Its Native Disease State. J. Dent. Res. 2020, 99, 597–603. [Google Scholar] [CrossRef]
- Bhaumik, D.; Manikandan, D.; Foxman, B. Cariogenic and oral health taxa in the oral cavity among children and adults: A scoping review. Arch. Oral Biol. 2021, 129, 105204. [Google Scholar] [CrossRef]
- De Jesus, V.C.; Khan, M.W.; Mittermuller, B.A.; Duan, K.; Hu, P.; Schroth, R.J.; Chelikani, P. Characterization of Supragingival Plaque and Oral Swab Microbiomes in Children with Severe Early Childhood Caries. Front. Microbiol. 2021, 12, 683685. [Google Scholar] [CrossRef]
- Astasov-Frauenhoffer, M.; Kulik, E.M. Cariogenic Biofilms and Caries from Birth to Old Age. Monogr. Oral Sci. 2021, 29, 53–64. [Google Scholar] [CrossRef]
- Rudney, J.D.; Chen, R.; Lenton, P.; Li, J.; Li, Y.; Jones, R.S.; Reilly, C.; Fok, A.S.; Aparicio, C. A reproducible oral microcosm biofilm model for testing dental materials. J. Appl. Microbiol. 2012, 113, 1540–1553. [Google Scholar] [CrossRef]
- Klinke, T.; Urban, M.; Luck, C.; Hannig, C.; Kuhn, M.; Kramer, N. Changes in Candida spp., mutans streptococci and lactobacilli following treatment of early childhood caries: A 1-year follow-up. Caries Res. 2014, 48, 24–31. [Google Scholar] [CrossRef]
- Meyer, F.; Enax, J.; Epple, M.; Amaechi, B.T.; Simader, B. Cariogenic Biofilms: Development, Properties, and Biomimetic Preventive Agents. Dent. J. 2021, 9, 88. [Google Scholar] [CrossRef]
- Alkhars, N.; Zeng, Y.; Alomeir, N.; Al Jallad, N.; Wu, T.T.; Aboelmagd, S.; Youssef, M.; Jang, H.; Fogarty, C.; Xiao, J. Oral Candida Predicts Streptococcus mutans Emergence in Underserved US Infants. J. Dent. Res. 2021, 220345211012385. [Google Scholar] [CrossRef]
- Zhang, Q.; Qin, S.; Xu, X.; Zhao, J.; Zhang, H.; Liu, Z.; Chen, W. Inhibitory Effect of Lactobacillus plantarum CCFM8724 towards Streptococcus mutans- and Candida albicans-Induced Caries in Rats. Oxidative Med. Cell. Longev. 2020, 2020, 4345804. [Google Scholar] [CrossRef]
- Vertuan, M.; Machado, P.F.; de Souza, B.M.; Braga, A.S.; Magalhaes, A.C. Effect of TiF4/NaF and chitosan solutions on the development of enamel caries under a microcosm biofilm model. J. Dent. 2021, 111, 103732. [Google Scholar] [CrossRef]
- Frazao Camara, J.V.; Araujo, T.T.; Mendez, D.A.C.; da Silva, N.D.G.; de Medeiros, F.F.; Santos, L.A.; de Souza Carvalho, T.; Reis, F.N.; Martini, T.; Moraes, S.M.; et al. Effect of a sugarcane cystatin on the profile and viability of microcosm biofilm and on dentin demineralization. Arch. Microbiol. 2021, 203, 4133–4139. [Google Scholar] [CrossRef]
- Cusicanqui Mendez, D.A.; Gutierres, E.; Jose Dionisio, E.; Afonso Rabelo Buzalaf, M.; Cardoso Oliveira, R.; Andrade Moreira Machado, M.A.; Cruvinel, T. Curcumin-mediated antimicrobial photodynamic therapy reduces the viability and vitality of infected dentin caries microcosms. Photodiagnosis Photodyn. Ther. 2018, 24, 102–108. [Google Scholar] [CrossRef]
- Llena, C.; Almarche, A.; Mira, A.; Lopez, M.A. Antimicrobial efficacy of the supernatant of Streptococcus dentisani against microorganisms implicated in root canal infections. J. Oral Sci. 2019, 61, 184–194. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.K.; Dai, T.; Kharkwal, G.B.; Huang, Y.Y.; Huang, L.; De Arce, V.J.; Tegos, G.P.; Hamblin, M.R. Drug discovery of antimicrobial photosensitizers using animal models. Curr. Pharm. Des. 2011, 17, 1303–1319. [Google Scholar] [CrossRef]
- Kim, D.; Barraza, J.P.; Arthur, R.A.; Hara, A.; Lewis, K.; Liu, Y.; Scisci, E.L.; Hajishengallis, E.; Whiteley, M.; Koo, H. Spatial mapping of polymicrobial communities reveals a precise biogeography associated with human dental caries. Proc. Natl. Acad. Sci. USA 2020, 117, 12375–12386. [Google Scholar] [CrossRef]
- Metwalli, K.H.; Khan, S.A.; Krom, B.P.; Jabra-Rizk, M.A. Streptococcus mutans, Candida albicans, and the human mouth: A sticky situation. PLoS Pathog. 2013, 9, e1003616. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, J.O.; Rossoni, R.D.; Vilela, S.F.; de Alvarenga, J.A.; Velloso Mdos, S.; Prata, M.C.; Jorge, A.O.; Junqueira, J.C. Streptococcus mutans Can Modulate Biofilm Formation and Attenuate the Virulence of Candida albicans. PLoS ONE 2016, 11, e0150457. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, M.T.; Ward, R.A.d.C.; Gonçalves, N.M.F.; Pedroso, L.L.C.; Neto, J.V.d.S.; Strixino, J.F.; Junqueira, J.C. Susceptibility of Dental Caries Microcosm Biofilms to Photodynamic Therapy Mediated by Fotoenticine. Pharmaceutics 2021, 13, 1907. https://doi.org/10.3390/pharmaceutics13111907
Garcia MT, Ward RAdC, Gonçalves NMF, Pedroso LLC, Neto JVdS, Strixino JF, Junqueira JC. Susceptibility of Dental Caries Microcosm Biofilms to Photodynamic Therapy Mediated by Fotoenticine. Pharmaceutics. 2021; 13(11):1907. https://doi.org/10.3390/pharmaceutics13111907
Chicago/Turabian StyleGarcia, Maíra Terra, Rafael Araújo da Costa Ward, Nathália Maria Ferreira Gonçalves, Lara Luise Castro Pedroso, José Vieira da Silva Neto, Juliana Ferreira Strixino, and Juliana Campos Junqueira. 2021. "Susceptibility of Dental Caries Microcosm Biofilms to Photodynamic Therapy Mediated by Fotoenticine" Pharmaceutics 13, no. 11: 1907. https://doi.org/10.3390/pharmaceutics13111907
APA StyleGarcia, M. T., Ward, R. A. d. C., Gonçalves, N. M. F., Pedroso, L. L. C., Neto, J. V. d. S., Strixino, J. F., & Junqueira, J. C. (2021). Susceptibility of Dental Caries Microcosm Biofilms to Photodynamic Therapy Mediated by Fotoenticine. Pharmaceutics, 13(11), 1907. https://doi.org/10.3390/pharmaceutics13111907






