Use of an In Vitro Skin Parallel Artificial Membrane Assay (Skin-PAMPA) as a Screening Tool to Compare Transdermal Permeability of Model Compound 4-Phenylethyl-Resorcinol Dissolved in Different Solvents
Abstract
1. Introduction
| Solvent Class | Code | Solvent | MW | PER Approximate Solubility 1 (mg/mL) | PER Equilibrium Solubility 2 (mg/mL) |
|---|---|---|---|---|---|
| Low-MW polar solvents | S1 | Water | 18.0 | 1 | 1.3 ± 0.2 |
| S2 | Ethanol | 46.1 | >1000 | 368 ± 52 | |
| S3 | Glycerol | 92.1 | 5 | - | |
| S4 | Dimethylisosorbide | 174.2 | 75 | 60 ± 5.7 | |
| S5 | Water/ethanol 80:20 (w/w) | NA | 10 | 8.1 ± 4.3 | |
| S6 | Water/dimethylisosorbide 90:10 (w/w) | NA | 1 | 1.1 ± 0.1 | |
| Low-MW polar “glycol” solvents | S7 | Propylene glycol | 76.1 | 500 | 350 ± 21 |
| S8 | Water:propylene glycol 80:20 (w/w) | NA | 10 | 5.1 ± 0.8 | |
| S9 | Water/propylenglycol/ethanol 10:30:60 (w/w/w) | NA | >1000 | 373 ± 49 | |
| High-MW non-polar solvents | S10 | Capric/caprylic triglycerides | 554.8/470.7 | 75 | 74 ± 5.1 |
| S11 | Octyl dodecanol | 298.6 | 1 | - | |
| S12 | Apricot kernel oil | NA | 1 | - | |
| S13 | Corn oil | NA | 1 | - |
2. Materials and Methods
2.1. Materials
2.2. Solubility Measurements
2.3. LogP Measurement
2.4. Permeability Measurements Using Skin-PAMPA Plates
2.5. Skin-PAMPA Membrane Integrity Study
2.6. Penetration Kinetics across Pig Ear Skin
3. Results
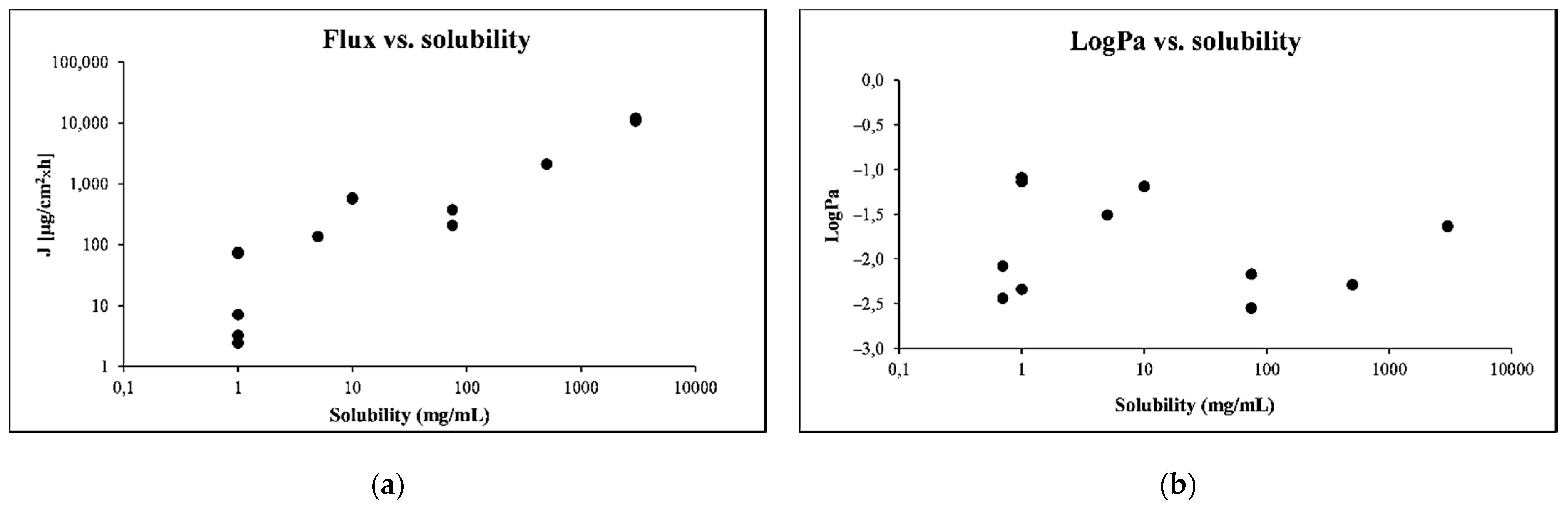
3.1. Solubility of PER in Different Solvents
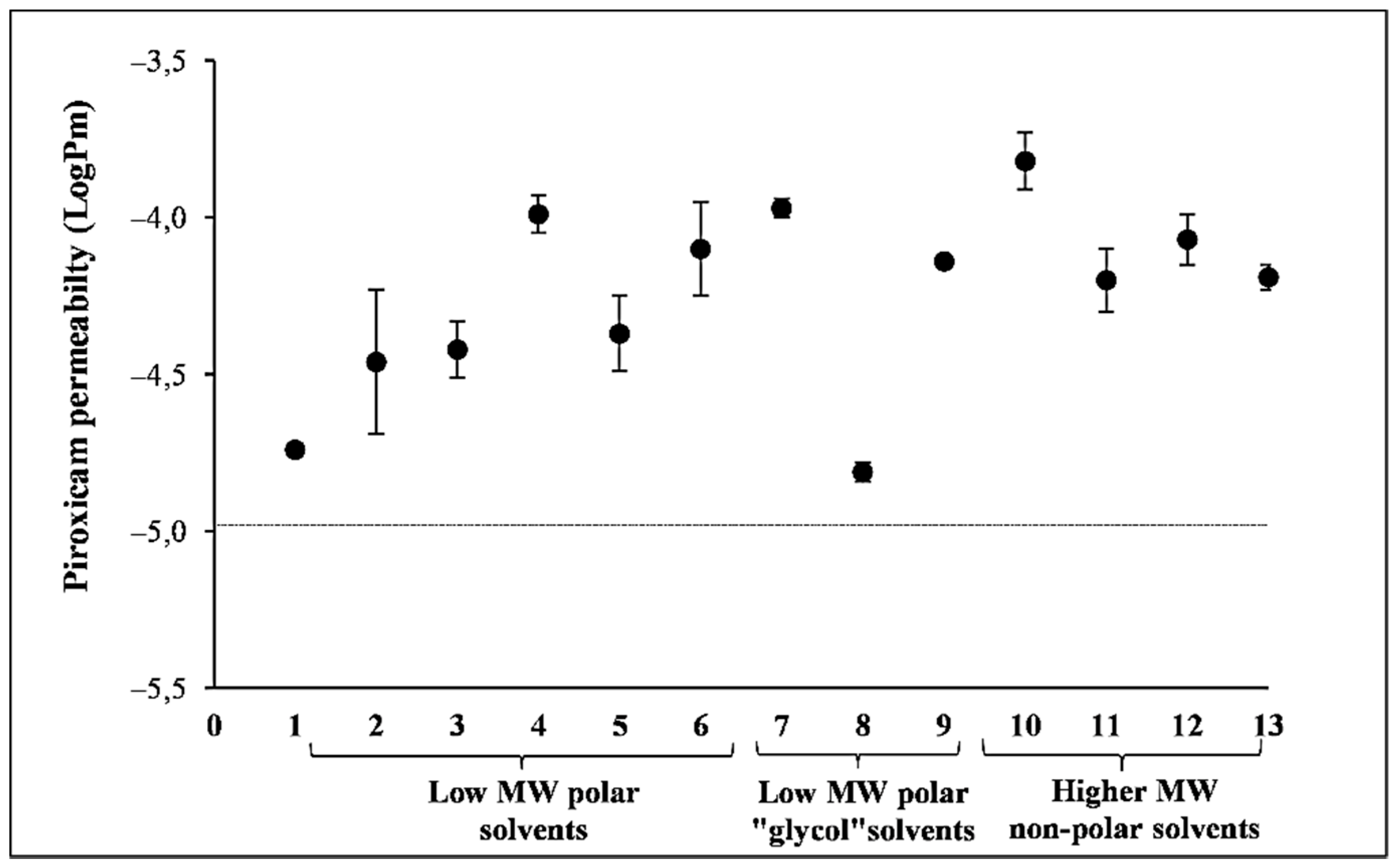
3.2. Effect of Solvents on PAMPA Membrane Integrity
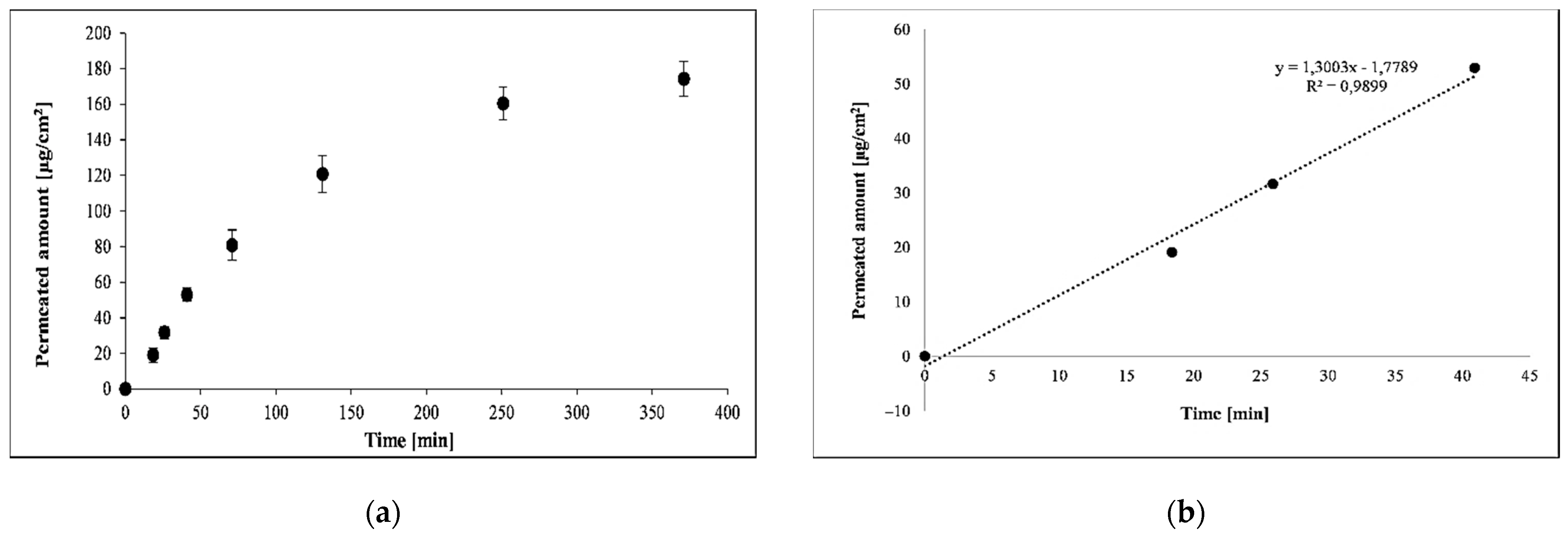
3.3. Effect of Solvent on the Permeability of PER Using PAMPA
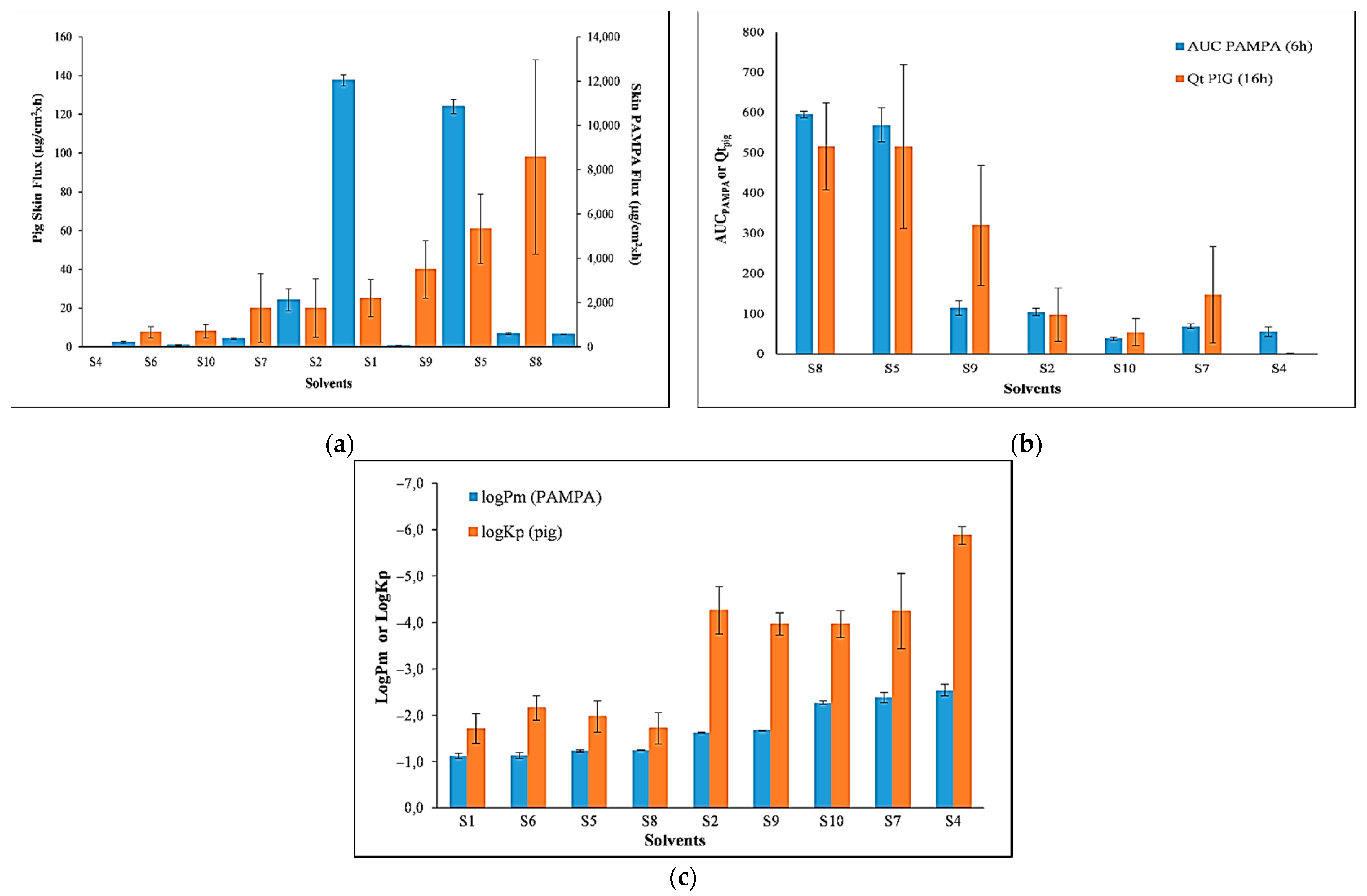
3.4. Comparison of the Permeability of PER Using Skin-PAMPA vs. Pig Skin
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Howes, D.; Guy, R.H.; Hadgraft, J.; Heylings, J.; Hoeck, U.; Kemper, F.; Maibach, H.; Marty, J.; Merk, H.; Parra, J.; et al. Methods for assessing percutaneous absorption—The report and recommendations of ECVAM workshop 13. ATLA—Altern. Lab. Anim. 1996, 24, 81–106. [Google Scholar] [CrossRef]
- Prausnitz, M.R.; Langer, R. Transdermal drug delivery(Review). Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef]
- Wiedersberg, S.; Guy, R.H. Transdermal drug delivery: 30 + years of war and still fighting! J. Control. Release 2014, 190, 150–156. [Google Scholar] [CrossRef]
- Scientific Committee on Consumer Safety Basic criteria for the in vitro assessment of dermal absorption of cosmetic ingredients. Eur. Comm. 2010, SCCS/1358, 1–14.
- OECD Test No. 428: Skin Absorption: In Vitro Method. In OECD Guidelines for the Testing of Chemicals, Section 4; OECD: Paris, France, 2004.
- Kraeling, M.E.K.; Bronaugh, R. In vitro percutaneous absorption of alpha hydroxy acids in human skin. J. Soc. Cosmet. Chem. 1997, 48, 187–197. [Google Scholar]
- Gerstel, D.; Jacques-Jamin, C.; Schepky, A.; Cubberley, R.; Eilstein, J.; Grégoire, S.; Hewitt, N.; Klaric, M.; Rothe, H.; Duplan, H. Comparison of protocols for measuring cosmetic ingredient distribution in human and pig skin. Toxicol. Vitr. 2016, 34, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Wargniez, W.; Jungman, E.; Wilkinson, S.; Seyler, N.; Grégoire, S. Inter-laboratory skin distribution study of 4-n-butyl resorcinol: The importance of liquid chromatography/mass spectrometry (HPLC–MS/MS) bioanalytical validation. J. Chromatogr. B 2017, 1060, 416–423. [Google Scholar] [CrossRef]
- Lotte, C.; Patouillet, C.; Zanini, M.; Messager, A.; Roguet, R. Permeation and Skin Absorption: Reproducibility of Various Industrial Reconstructed Human Skin Models. Ski. Pharmacol. Appl. Ski. Physiol. 2002, 15, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Schäfer-Korting, M.; Bock, U.; Diembeck, W.; Düsing, H.-J.; Gamer, A.; Haltner-Ukomadu, E.; Hoffmann, C.; Kaca, M.; Kamp, H.; Kersen, S.; et al. The Use of Reconstructed Human Epidermis for Skin Absorption Testing: Results of the Validation Study. Altern. Lab. Anim. 2008, 36, 161–187. [Google Scholar] [CrossRef] [PubMed]
- De Jager, M.W.; Gooris, G.; Ponec, M.; Bouwstra, J. Lipid mixtures prepared with well-defined synthetic ceramides closely mimic the unique stratum corneum lipid phase behavior. J. Lipid. Res. 2005, 46, 2649–2656. [Google Scholar] [CrossRef]
- Ottaviani, G.; Martel, A.S.; Carrupt, P.-A. Parallel Artificial Membrane Permeability Assay: A New Membrane for the Fast Prediction of Passive Human Skin Permeability. J. Med. Chem. 2006, 49, 3948–3954. [Google Scholar] [CrossRef]
- Joshi, V.; Brewster, D.; Colonero, P. In vitro diffusion studies in transdermal research: A synthetic membrane model in place of human skin. Drug Dev. Deliv. 2012, 12, 40–42. [Google Scholar]
- Provin, C.; Nicolas, A.; Grégoire, S.; Fujii, T. A Microfluidic Diffusion Cell for Fast and Easy Percutaneous Absorption Assays. Pharm. Res. 2015, 32, 2704–2712. [Google Scholar] [CrossRef]
- Sinkó, B.; Garrigues, T.M.; Balogh, G.T.; Nagy, Z.K.; Tsinman, O.; Avdeef, A.; Takács-Novák, K. Skin–PAMPA: A new method for fast prediction of skin penetration. Eur. J. Pharm. Sci. 2012, 45, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and function of the epidermis related to barrier properties. Clin. Dermatol. 2012, 30, 257–262. [Google Scholar] [CrossRef]
- Karadzovska, D.; Riviere, J.E. Assessing vehicle effects on skin absorption using artificial membrane assays. Eur. J. Pharm. Sci. 2013, 50, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Haq, A.; Goodyear, B.; Ameen, D.; Joshi, V.; Michniak-Kohn, B. Strat-M® synthetic membrane: Permeability comparison to human cadaver skin. Int. J. Pharm. 2018, 547, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Sinkó, B.; Vizserálek, G.; Novák, K.T. Skin PAMPA: Application in practice. ADMET DMPK 2015, 2, 191–198. [Google Scholar] [CrossRef][Green Version]
- Balázs, B.; Vizserálek, G.; Berkó, S.; Budai-Szűcs, M.; Kelemen, A.; Sinkó, B.; Takács-Novák, K.; Szabó-Révész, P.; Csányi, E. Investigation of the Efficacy of Transdermal Pene-tration Enhancers Through the Use of Human Skin and a Skin Mimic Artificial Membrane. J. Pharm. Sci. 2016, 105, 1134–1140. [Google Scholar] [CrossRef]
- Vizserálek, G.; Balogh, T.; Takács-Novák, K.; Sinkó, B. PAMPA study of the temperature effect on permeability. Eur. J. Pharm. Sci. 2014, 53, 45–49. [Google Scholar] [CrossRef]
- Vizserálek, G.; Berkó, S.; Tóth, G.; Balogh, R.; Budai-Szűcs, M.; Csányi, E.; Sinkó, B.; Takács-Novák, K. Permeability test for transdermal and local therapeutic patches using Skin PAMPA method. Eur. J. Pharm. Sci. 2015, 76, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sil, B.C.; Kung, C.; Hadgraft, J.; Heinrich, M.; Sinko, B.; Lane, M.E. Characterization and topical delivery of phenylethyl resorcinol. Int. J. Cosmet. Sci. 2019, 41, 479–488. [Google Scholar] [CrossRef]
- Hadgraft, J.; Guy, R.H. Feasibility Assessment in Topical and Transdermal Delivery: Mathematical Models and in Vitro Studies. In Transdermal Drug Delivery; CRC Press: Boca Raton, FL, USA, 2002; pp. 1–23. [Google Scholar]
- OECD Test No. 105: Water Solubility. In OECD Guidelines for the Testing of Chemicals, Section 1; Organisation for Economic Co-Operation and Development (OECD): Paris, France, 1995.
- Baka, E.; Comer, J.E.; Takács-Novák, K. Study of equilibrium solubility measurement by saturation shake-flask method using hydrochlorothiazide as model compound. J. Pharm. Biomed. Anal. 2008, 46, 335–341. [Google Scholar] [CrossRef]
- Avdeef, A.; Fuguet, E.; Llinas, A.; Ràfols, C.; Bosch, E.; Völgyi, G.; Verbic, T.; Boldyreva, E.; Takács-Novák, K. Equilibrium solubility measurement of ionizable drugs—consensus recommendations for improving data quality. ADMET DMPK 2016, 4, 117–178. [Google Scholar] [CrossRef]
- Takács-Novák, K.; Józan, M.; Hermecz, I.; Szász, G. Lipophilicity of antibacterial fluoroquinolones. Int. J. Pharm. 1992, 79, 89–96. [Google Scholar] [CrossRef]
- Takács-Novák, K.; Józan, M.; Szász, G. Lipophilicity of amphoteric molecules expressed by the true partition coefficient. Int. J. Pharm. 1995, 113, 47–55. [Google Scholar] [CrossRef]
| PER | |
|---|---|
| Structure | 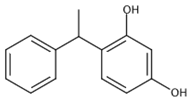 |
| Chemical Name | 4-phenylethyl-resorcinol |
| CAS Number | 94-77-9 |
| Molecular Weight | 214.3 (g/mol) |
| logP | 2.98 1 |
| pKa | 9.77–10.77 (AH/A−) 2 |
| Solubilty in Prisma Buffer | 3.45 1 (mg/mL) |
| Water Solubility | 3.85 (mg/mL) |
| Code | CD [mg/mL] | J [µg/cm2 × h] | Lag Time [min] | Permeated Amount (6 h) [µg/cm2] | AUC Normalized to CD | logPm |
|---|---|---|---|---|---|---|
| S1 | 1 | 72.4 ± 7.8 | 1.4 | 169 ± 4 | 647 | −1.12 ± 0.06 |
| S2 | 500 | 12,033 ± 252 | 0.0 | 13,575 ± 1710 | 105 | −1.62 ± 0.01 |
| S3 | 5 | 137 ± 22 | 5.5 | 869 ± 112 | 491 | −1.57 ± 0.07 |
| S4 | 70 | 209 ± 53 | 3.6 | 1342 ± 298 | 55.2 | −2.54 ± 0.13 |
| S5 | 10 | 589 ± 25 | 0.9 | 1662 ± 169 | 569 | −1.23 ± 0.03 |
| S6 | 1 | 76.4 ± 15.2 | 0.0 | 175 ± 22 | 551 | −1.13 ± 0.07 |
| S7 | 500 | 2118 ± 502 | 3.8 | 11,142 ± 729 | 69.1 | −2.38 ± 0.11 |
| S8 | 10 | 570 ± 5 | 1.1 | 1772 ± 98 | 595 | −1.24 ± 0.01 |
| S9 | 500 | 10,846 ± 326 | 1.4 | 14,926 ± 2431 | 114 | −1.66 ± 0.01 |
| S10 | 70 | 377 ± 36 | 3.0 | 749 ± 92 | 38.2 | −2.27 ± 0.04 |
| S11 | 1 | 7.18 ± 1.6 | 3.9 | 19 ± 0.4 | 64.5 | −2.16 ± 0.08 |
| S12 | 1 | 2.45 ± 0.57 | 3.3 | 13 ± 1.9 | 36.7 | −2.63 ± 0.07 |
| S13 | 1 | 3.26 ± 0.41 | 1.4 | 13 ± 1.3 | 38.6 | −2.48 ± 0.06 |
| Code | CD [mg/mL] | J [µg/cm2 × h] | Permeated Amount (16 h) [µg/cm2] | logKp |
|---|---|---|---|---|
| S1 | 1 | 25 ± 9.6 | 261 ± 100 | −1.71 ± 0.32 |
| S2 | 368 | 20 ± 15 | 98 ± 67 | −4.26 ± 0.51 |
| S4 | 60 | 0.08 ± 0.024 | 1.09 ± 0,39 | −5.88 ± 0.19 |
| S5 | 8 | 61 ± 18 | 515 ± 203 | −1.97 ± 0.34 |
| S6 | 1 | 7.5 ± 3 | 100 ± 37 | −2.16 ± 0.26 |
| S7 | 350 | 20 ± 17.6 | 147 ± 120 | −4.24 ± 0.81 |
| S8 | 51 | 98 ± 50 | 516 ± 109 | −1.72 ± 0.34 |
| S9 | 373 | 40 ± 15 | 320 ± 149 | −3.97 ± 0.24 |
| S10 | 75 | 8 ± 3.5 | 54 ± 34 | −3.97 ± 0.29 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinkó, B.; Bárdos, V.; Vesztergombi, D.; Kádár, S.; Malcsiner, P.; Moustie, A.; Jouy, C.; Takács-Novák, K.; Grégoire, S. Use of an In Vitro Skin Parallel Artificial Membrane Assay (Skin-PAMPA) as a Screening Tool to Compare Transdermal Permeability of Model Compound 4-Phenylethyl-Resorcinol Dissolved in Different Solvents. Pharmaceutics 2021, 13, 1758. https://doi.org/10.3390/pharmaceutics13111758
Sinkó B, Bárdos V, Vesztergombi D, Kádár S, Malcsiner P, Moustie A, Jouy C, Takács-Novák K, Grégoire S. Use of an In Vitro Skin Parallel Artificial Membrane Assay (Skin-PAMPA) as a Screening Tool to Compare Transdermal Permeability of Model Compound 4-Phenylethyl-Resorcinol Dissolved in Different Solvents. Pharmaceutics. 2021; 13(11):1758. https://doi.org/10.3390/pharmaceutics13111758
Chicago/Turabian StyleSinkó, Bálint, Vivien Bárdos, Dániel Vesztergombi, Szabina Kádár, Petra Malcsiner, Anne Moustie, Chantal Jouy, Krisztina Takács-Novák, and Sebastien Grégoire. 2021. "Use of an In Vitro Skin Parallel Artificial Membrane Assay (Skin-PAMPA) as a Screening Tool to Compare Transdermal Permeability of Model Compound 4-Phenylethyl-Resorcinol Dissolved in Different Solvents" Pharmaceutics 13, no. 11: 1758. https://doi.org/10.3390/pharmaceutics13111758
APA StyleSinkó, B., Bárdos, V., Vesztergombi, D., Kádár, S., Malcsiner, P., Moustie, A., Jouy, C., Takács-Novák, K., & Grégoire, S. (2021). Use of an In Vitro Skin Parallel Artificial Membrane Assay (Skin-PAMPA) as a Screening Tool to Compare Transdermal Permeability of Model Compound 4-Phenylethyl-Resorcinol Dissolved in Different Solvents. Pharmaceutics, 13(11), 1758. https://doi.org/10.3390/pharmaceutics13111758





