Design, Synthesis, Characterization, and In Vitro Evaluation of a New Cross-Linked Hyaluronic Acid for Pharmaceutical and Cosmetic Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
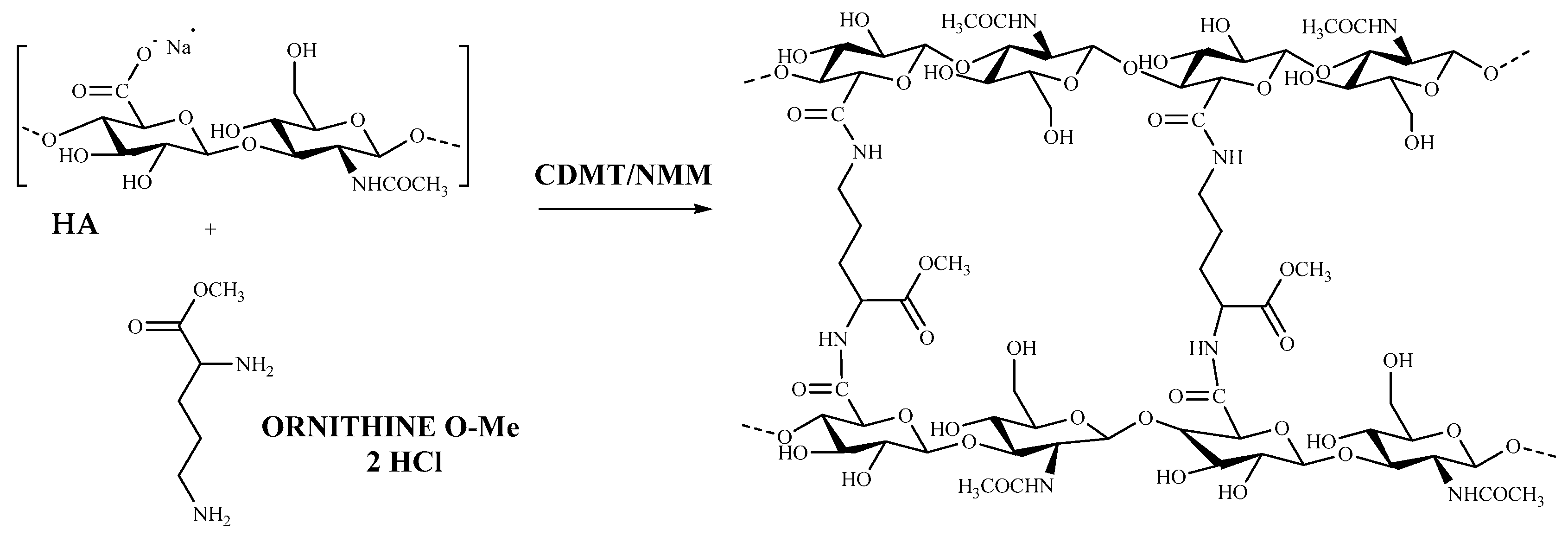
2.2. Cross-Linked HA–Orn
2.2.1. Processing Parameters
2.2.2. Synthesis of Cross-Linked HA–Orn
2.3. Chemical–Physical Characterization of HA–Orn
2.3.1. Fourier-Transform Infrared Spectroscopy (FTIR)
2.3.2. Differential Scanning Calorimetry (DSC)
2.3.3. Scanning Electron Microscopy (SEM)
2.3.4. Swelling Measurement
2.3.5. Rheological measurement
2.3.6. Water Content
2.4. Enzymatic Degradation Test
2.4.1. Sample Disc Preparation
2.4.2. In Vitro Degradation
2.4.3. Carbazole Assay
2.5. Biological Assays
2.5.1. Cells Culture
2.5.2. MTS Assay for Cytotoxicity
2.6. Statistical Analysis
3. Results
3.1. HA–Orn Cross-Linked Synthesis
3.2. Physico–Chemical Characterization Methods
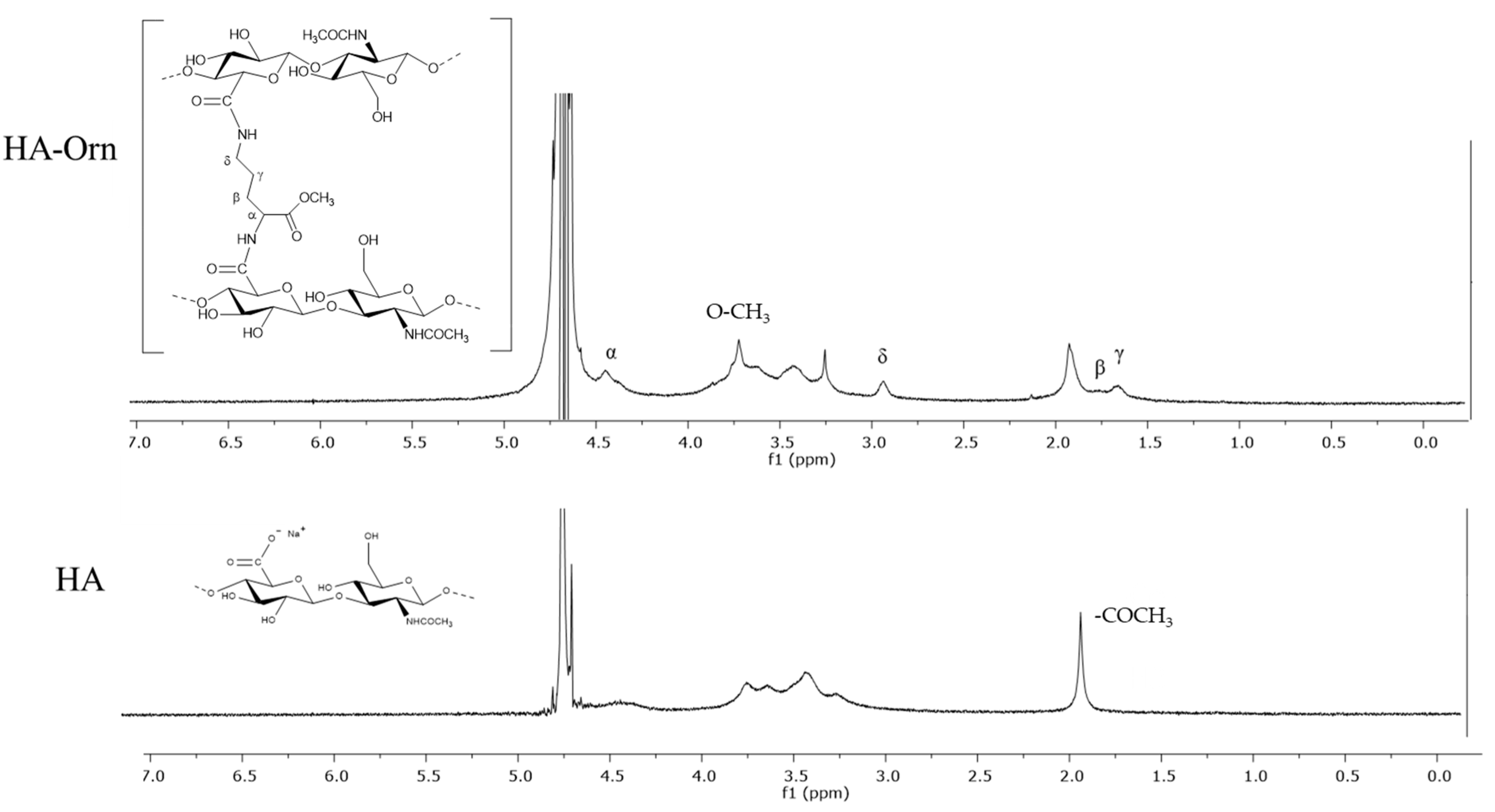
3.2.1. 1H-NMR
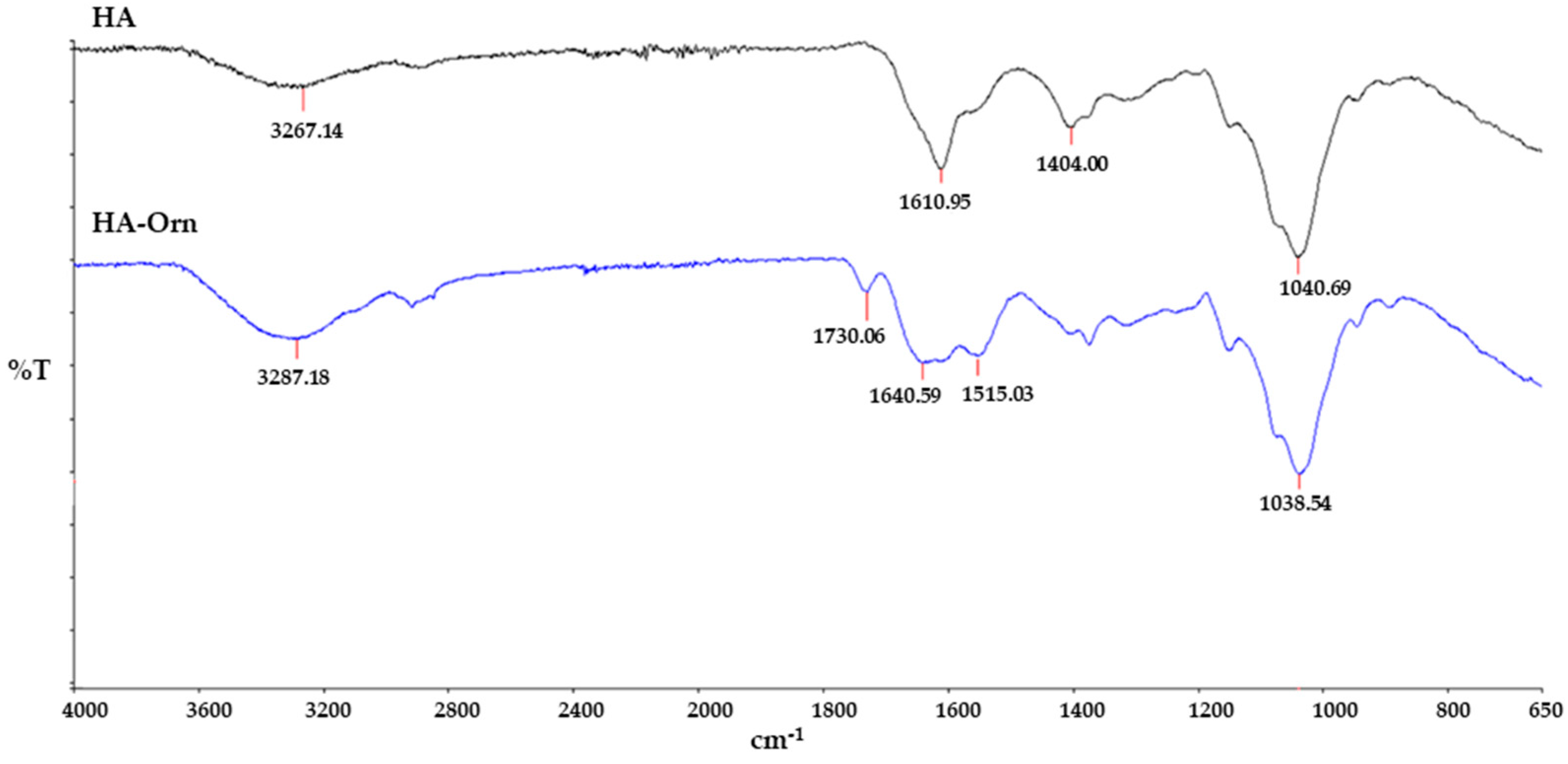
3.2.2. IR Spectroscopy
3.2.3. Thermal Analysis: DSC
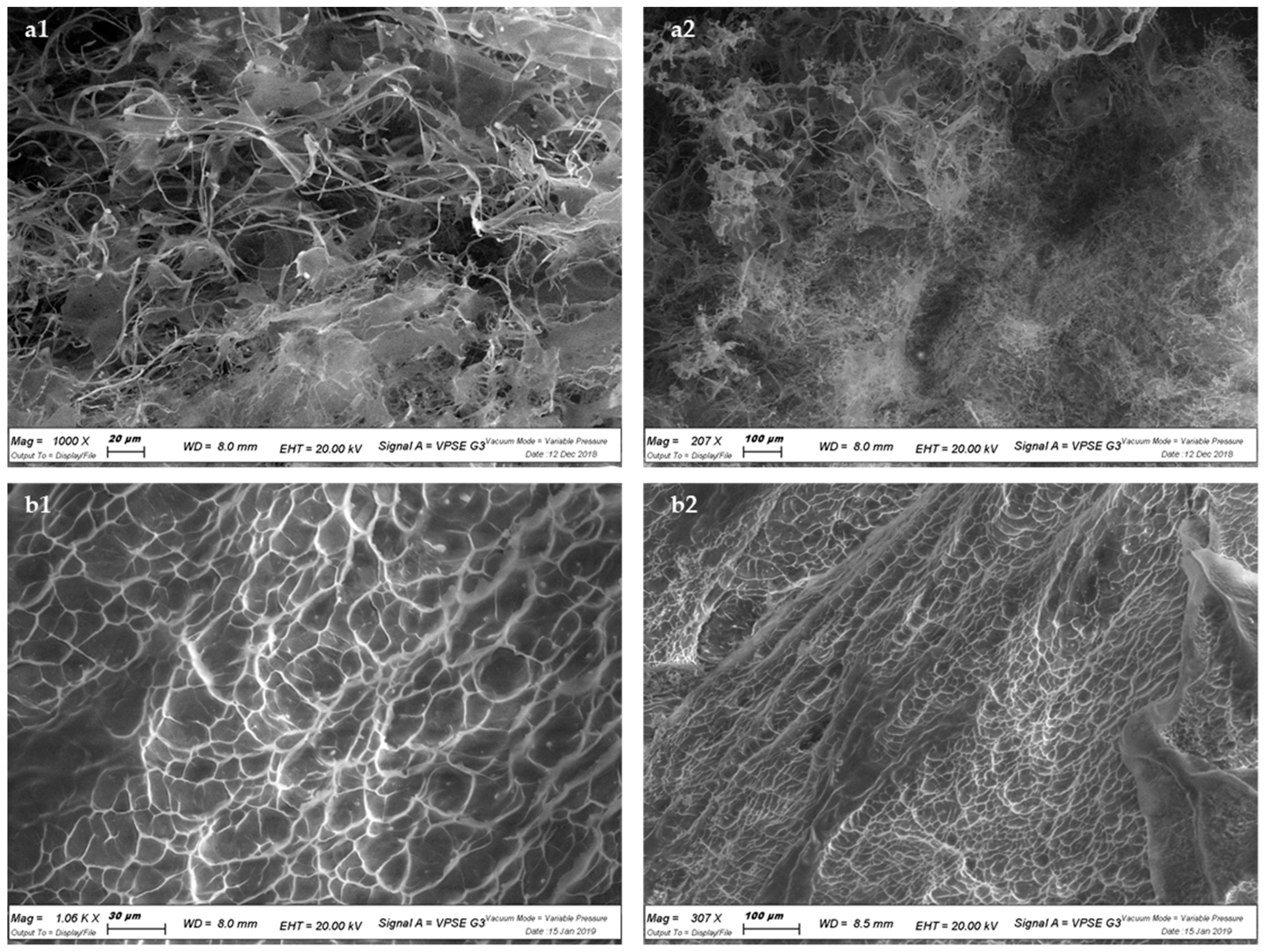
3.2.4. Scanning Electron Microscopy (SEM)
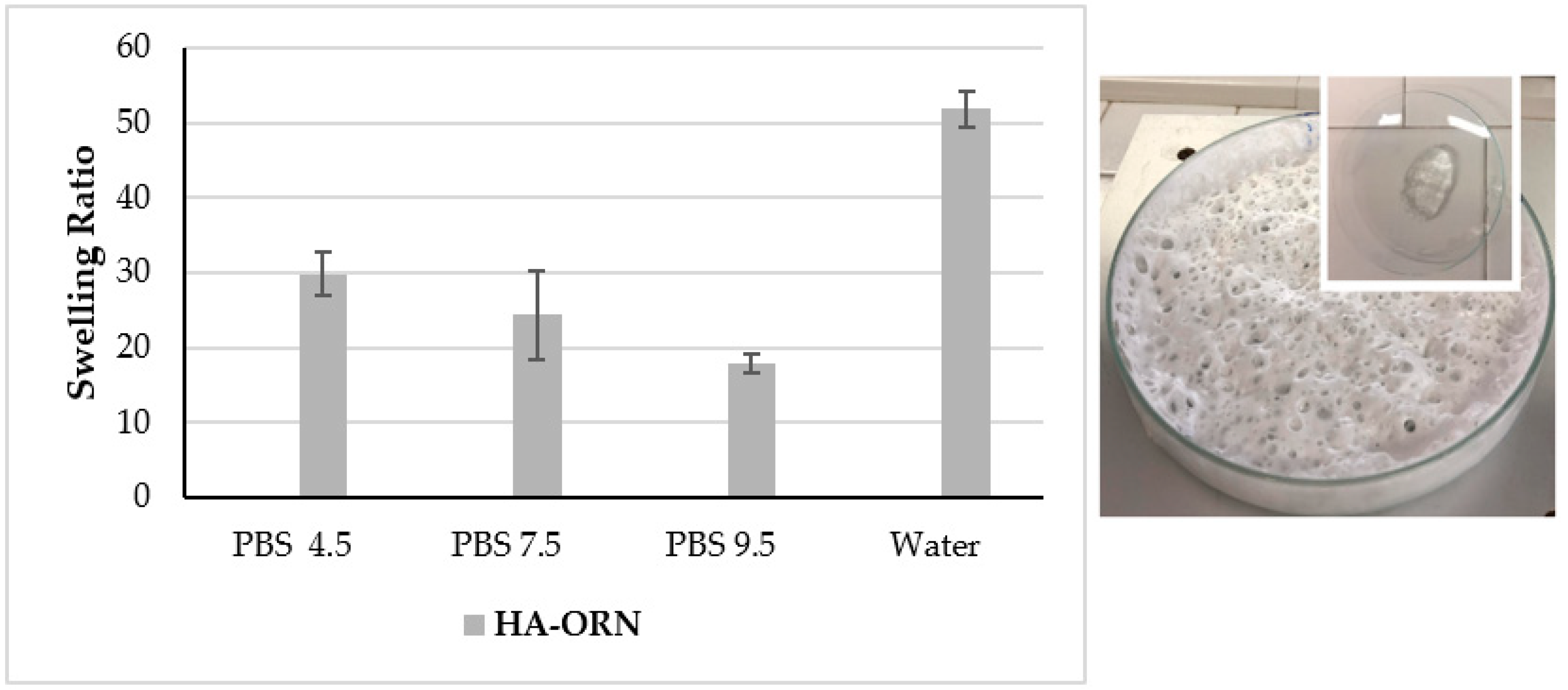
3.2.5. Swelling Ratio Measurement
3.2.6. Rheology
3.2.7. Dynamic Vapour Sorption
3.3. In Vitro Degradation
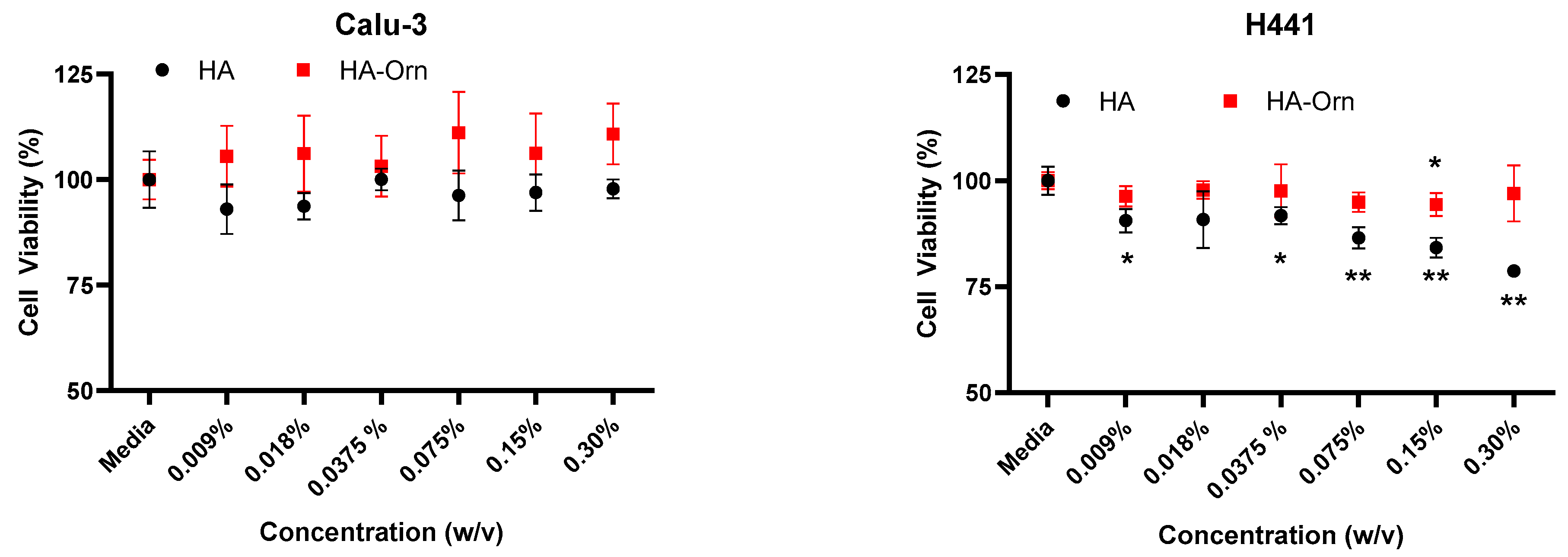
3.4. MTS Cytotoxicity Assay
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schanté, C.; Zuber, G.; Herlin, C.; Vandamme, F. Chemical modifications of hyaluronic acid for the synthesis of deriva-tives for a broad range of biomedical applications. Carbohydr. Polym. 2011, 85, 469–489. [Google Scholar] [CrossRef]
- Laffleur, F.; Dachs, S. Development of novel mucoadhesive hyaluronic acid derivate as lubricant for the treatment of dry eye syndrome. Ther. Deliv. 2015, 6, 1211–1219. [Google Scholar] [CrossRef]
- Weigel, P.; Fuller, G.; LeBoeuf, R.D. A model for the role of hyaluronic acid and fibrin in the early events during the in-flammatory response and wound healing. J. Theor. Biol. 1986, 119, 219–234. [Google Scholar] [CrossRef]
- Aya, K.L.; Stern, R. Hyaluronan in wound healing: Rediscovering a major player. Wound Repair Regen. 2014, 22, 579–593. [Google Scholar] [CrossRef]
- Borzacchiello, A.; Netti, P.A.; Ambrosio, L.; Nicolais, L. Hyaluronic acid derivatives mimic the rheological properties of vitreous body. In New Frontiers in Medical Sciences: Redefining Hyaluronan; Elsevier: Amsterdam, The Netherlands, 2000; pp. 192–202. [Google Scholar]
- Thambi, T.; Giang Phan, V.H.; Kim, S.H.; Le, T.; Lee, D. Hyaluronic acid decorated pH- and Temperature-induce injecta-ble bioconjugates for sustained delivery of bioactive factors and highly efficient wound regeneration. New J. Chem. 2019, 43, 18979. [Google Scholar] [CrossRef]
- Furnari, M.; Termini, L.; Traverso, G.; Barrale, S.; Bonaccorso, M.; Damiani, G.; Lo Piapro, C.; Collura, M. Nebulized hy-pertonic saline containing hyaluronic acid improves tolerability in patients with cystic fibrosis and lung disease compared with nebulized hypertonic saline alone: A prospective, randomized, double-blind, controlled study. Ther. Adv. Respir. Dis. 2012, 6, 315–322. [Google Scholar] [CrossRef] [Green Version]
- Garantziotis, S.; Brezina, M.; Castelnuovo, P.; Drago, L. The role of hyaluronan in the pathobiology and treatment of res-piratory disease. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 310, L785–L795. [Google Scholar] [CrossRef] [Green Version]
- Gavina, M.; Luciani, A.; Villella, V.R.; Esposito, S.; Ferrari, E.; Bressani, I.; Casale, A.; Bruscia, E.M.; Maiuri, L.; Raia, V. Nebulized Hyaluronan Ameliorates lung inflammation in cystic fibrosis mice. Pediatr. Pulmonol. 2012, 48, 761–771. [Google Scholar] [CrossRef] [Green Version]
- Petrigni, G.; Allegra, L. Aerosolised hyaluronic acid prevents exercise-induced bronchoconstriction, suggesting novel hy-potheses on the correction of matrix defects in asthma. Pulm. Pharmacol. Ther. 2006, 19, 166–171. [Google Scholar] [CrossRef]
- Rahman, I.; Adcock, I.M. Oxidative stress and redox regulation of lung inflammation in COPD. Eur. Respir. J. 2006, 28, 219–242. [Google Scholar] [CrossRef]
- Carro, L.M.; Martínez-García, M.A. Use of Hyaluronic Acid (HA) in Chronic Airway Diseases. Cells 2020, 9, 2210. [Google Scholar] [CrossRef]
- Robert, L.; Robert, A.-M.; Renard, G. Biological effects of hyaluronan in connective tissues, eye, skin, venous wall. Role in aging. Pathol. Biol. 2010, 58, 187–198. [Google Scholar] [CrossRef]
- Baumann, L. Dermal fillers. J. Cosmet. Dermatol. 2004, 3, 249–250. [Google Scholar] [CrossRef]
- Gatej, I.; Popa, M.; Rinaudo, M. Role of the pH on Hyaluronan Behavior in Aqueous Solution. Biomacromolecules 2005, 6, 61–67. [Google Scholar] [CrossRef]
- Muhn, C.; Rosen, N.; Solish, N.; Bertucci, V.; Lupin, M.; Dansereau, A.; Weksberg, F.; Remington, B.K.; Swift, A. The evolving role of hyaluronic acid fillers for facial volume restoration and contouring: A Canadian overview. Clin. Cosmet. Investig. Dermatol. 2012, 5, 147–158. [Google Scholar] [CrossRef] [Green Version]
- Hašová, M.; Crhák, T.; Šafránková, B.; Dvořáková, J.; Muthný, T.; Velebný, V.; Kubala, L. Hyaluronan minimizes effects of UV irradiation on human keratinocytes. Arch. Dermatol. Res. 2011, 303, 277–284. [Google Scholar] [CrossRef]
- Trommer, H.; Wartewig, S.; Böttcher, R.; Pöppl, A.; Hoentsch, J.; Ozegowski, J.H.; Neubert, R.H. The effects of hyaluronan and its fragments on lipid models exposed to UV irradiation. Int. J. Pharm. 2003, 254, 223–234. [Google Scholar] [CrossRef]
- Girish, K.; Kemparaju, K. The magic glue hyaluronan and its eraser hyaluronidase: A biological overview. Life Sci. 2007, 80, 1921–1943. [Google Scholar] [CrossRef]
- Jedrzejas, M.J.; Stern, R. Structures of vertebrate hyaluronidases and their unique enzymatic mechanism of hydrolysis. Proteins Struct. Funct. Bioinform. 2005, 61, 227–238. [Google Scholar] [CrossRef]
- Knopf-Marques, H.; Pravda, M.; Wolfova, L.; Velebny, V.; Schaaf, P.; Vrana, N.; Lavalle, P. Hyaluronic Acid and Its De-rivatives in Coating and Delivery Systems: Applications in Tissue Engineering, Regenerative Medicine and Immunomod-ulation. Adv. Healthc. Mater. 2016, 5, 2841–2855. [Google Scholar] [CrossRef]
- Luo, Y.; Prestwich, G.D. Synthesis and Selective Cytotoxicity of a Hyaluronic Acid−Antitumor Bioconjugate. Bioconjugate Chem. 1999, 10, 755–763. [Google Scholar] [CrossRef]
- Drobnik, J. Hyaluronan in drug delivery. Adv. Drug Deliv. Rev. 1991, 7, 295–308. [Google Scholar] [CrossRef]
- Choi, K.Y.; Min, K.H.; Na, J.H.; Choi, K.; Ki, K.; Park, J.H.; Kwon, I.C.; Jeong, S.Y. Self-assembled hyaluronic acid nano-particles as a potential drug carrier for cancer therapy: Synthesis, characterization, and in vivo biodistribution. J. Mater. Chem. 2009, 19, 4102–4107. [Google Scholar] [CrossRef]
- Shimojo, A.A.M.; Pires, A.M.B.; Lichy, R.; Rodrigues, A.A.; Santana, M.H.A. The crosslinking degree controls the mechanical, rheological, and swelling properties of hyaluronic acid microparticles. J. Biomed. Mater. Res. Part A 2014, 103, 730–737. [Google Scholar] [CrossRef]
- La Gatta, A.; Schiraldi, C.; Papa, A.; D’Agostino, A.; Cammarota, M.; Rosa, A.; Rosa, M. Hyaluronan scaffolds via di-glycidyl ether cross-linking: Toward improvements in composition and performance. Carbohydr. Polym. 2013, 96, 536–544. [Google Scholar] [CrossRef]
- Sung, H.; Huang, R.; Huang, L.; Tsai, C.; Chiu, C. Feasibility study of a natural crosslinking reagent for biologi-cal tissue fixation. J. Biomed. Mater. Res. 1998, 42, 560–567. [Google Scholar] [CrossRef]
- Ferretti, M.; Marra, K.K.; Defail, A.; Chu, C. Controlled in vivo degradation of genipin crosslinked polyethylene glycol hy-drogels within osteochondral defects. Tissue Eng. 2006, 12, 2657–2663. [Google Scholar] [CrossRef]
- Tan, H.; Chu, C.R.; Payne, K.; Marra, K.G. Injectable in situ forming biodegradable chitosan–hyaluronic acid based hydrogels for cartilage tissue engineering. Biomaterials 2009, 30, 2499–2506. [Google Scholar] [CrossRef] [Green Version]
- Ye, D.; Chang, C.; Zhang, L. High-strenght and tought cellulose hydrogels chemically dual cross-linked by using low- and high-molecular-weight cross-linkers. Biomacromolecules 2019, 20, 1989–1995. [Google Scholar] [CrossRef]
- Abatangelo, G.; Vindigni, V.; Avruscio, G.; Pandis, L.; Brun, P. Hyaluronic Acid: Redefining Its Role. Cells 2020, 9, 1743. [Google Scholar] [CrossRef]
- Zamboni, F.; Ryan, E.; Culebras, M.; Collins, M.N. Labile crosslinked hyaluronic acid via urethane formation using bis(β-isocyanatoethyl) disulphide with tuneable physicochemical and immunomodulatory properties. Carbohydr. Polym. 2020, 245, 116501. [Google Scholar] [CrossRef]
- Zamboni, F.; Okoroafor, C.; Ryan, M.P.; Pembroke, J.T.; Strozyk, M.; Culebras, M.; Collins, M.N. On the bacteriostatic ac-tivity of hyaluronic acid composite films. Carbohydr. Polym. 2021, 260, 117803. [Google Scholar] [CrossRef]
- How, K.N.; Yap, W.H.; Lim, C.L.H.; Goh, B.H.; Lai, Z.W. Hyaluronic acid-mediated drug delivery system targeting for in-flammatory skin diseases: A mini review. Front. Pharmacol. 2020, 11, 1105. [Google Scholar] [CrossRef]
- Chang, K.C.; Chen, W.C.; Haung, S.M.; Liu, S.M.; Lin, C.L. Effects of Hinokitiol and dicalcium phosphate on the oste-oconduction and antibacterial activity of gelatin-hyaluronic acid crosslinked hydrogel membrane in vitro. Pharmaceuticals 2021, 14, 802. [Google Scholar] [CrossRef]
- Arezki, N.R.; Williams, A.C.; Cobb, A.J.A.; Brown, M. Design, synthesis and characterization of linear unnatural amino acids for skin moisturization. Int. J. Cosmet. Sci. 2016, 39, 72–82. [Google Scholar] [CrossRef] [Green Version]
- Thambi, T.; Giang Phan, V.H.; Kim, S.H.; Le, T.; Doung, H.T.D.; Lee, D. Smart injectable biogels based on hyaluronic acid bioconiugates finely substituted with poly(β-amino ester urethane) for cancer therapy. Biomater. Sci. 2019, 7, 5424. [Google Scholar] [CrossRef]
- Silva, A.C.; Rodrigues, S.C.; Caldeira, J.; Nunes, A.M.; Sampaio-Pinto, V.; Resende, T.P.; Oliveira, M.J.; Barbosa, M.A.; Thorsteinsdóttir, S.; Nascimento, D.S.; et al. Three-dimensional scaffolds of fetal decellularized hearts exhibit en-hanced potential to support cardiac cells in comparison to the adult. Biomaterials 2016, 104, 52–64. [Google Scholar] [CrossRef]
- Yasuda, K.; Armstrong, R.; Cohen, R. Shear flow properties of concentrated solutions of linear and star branched polysty-renes. Rheol. Acta. 1981, 20, 163–178. [Google Scholar] [CrossRef]
- Lapasin, R.; Pricl, S. Rheology of Industrial Polysaccharides: Theory and Applications; Blackie Academic & Professional: London, UK, 1995. [Google Scholar]
- Tadros, T. Application of rheology for assessment and prediction of the long-term physical stability of emulsions. Adv. Colloid Interface Sci. 2004, 108, 227–258. [Google Scholar] [CrossRef]
- Bergman, K.; Elvingson, C.; Hilborn, J.; Svensk, G.; Bowden, T. Hyaluronic Acid Derivatives Prepared in Aqueous Media by Triazine-Activated Amidation. Biomacromolecules 2007, 8, 2190–2195. [Google Scholar] [CrossRef]
- Ford, J.; Timmins, P. Pharmaceutical Thermal Analysis: Techniques and Applications; Ellis Horwood: Chichester, UK, 1989. [Google Scholar]
- Collins, M.; Birkinshaw, C. Comparison of the effectiveness of four different crosslinking agents with hyaluronic acid hy-drogel films for tissue-culture applications. J. Appl. Polym. Sci. 2007, 104, 3183–3191. [Google Scholar] [CrossRef]
- Collins, N.; Birkinshaw, C. Physical properties of crosslinked hyaluronic acid hydrogels. J. Mater. Sci. Mater. Med. 2008, 19, 3335–3343. [Google Scholar] [CrossRef]
- Kafedjiiski, K.; Jetti, R.K.; Föger, F.; Hoyer, H.; Werle, M.; Hoffer, M.; Bernkop-Schnürch, A. Synthesis and in vitro evaluation of thiolated hyaluronic acid for mucoadhesive drug delivery. Int. J. Pharm. 2007, 343, 48–58. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Funda-mental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef]
- Smart, J.D. The basics and underlying mechanisms of mucoadhesion. Adv. Drug Deliv. Rev. 2005, 57, 1556–1568. [Google Scholar] [CrossRef]
- Luo, Y.; Kirker, K.R.; Prestwich, G.D. Cross-linked hyaluronic acid hydrogel films: New biomaterials for drug delivery. J. Control. Release 2000, 69, 169–184. [Google Scholar] [CrossRef]
- Manconi, M.; Manca, M.; Valenti, D.; Escribano, D.; Hillaireau, E.; Fadda, A.; Fattal, E. Chitosan and hyaluronan coated liposomes for pulmonary administration of curcum. Int. J. Pharm. 2017, 525, 203–210. [Google Scholar] [CrossRef]
- Egbu, R.; Brocchini, S.; Khaw, P.; Awwad, S. Antibody loaded collapsible hyaluronic acid hydrogels for intraocular deliv-ery. Eur. J. Pharm. Biopharm. 2018, 124, 95–103. [Google Scholar] [CrossRef]
- Schneider, L.; Korber, A.; Grabbe, S.; Dissemond, J. Influence of pH on wound-healing: A new perspective for wound-therapy? Arch. Dermatol. Res. 2007, 298, 413–420. [Google Scholar] [CrossRef]
- Shukla, V.; Shukla, D.; Tiwary, S.; Agrawal, S.; Rastogi, A. Evaluation of pH measurement as a method of wound assess-ment. J. Wound Care 2007, 16, 291–294. [Google Scholar] [CrossRef]
- Panagopoulou, A.; Molina, J.V.; Kyritsis, A.; Pradas, M.M.; Lluch, A.V.; Ferrer, G.G.; Pissis, P. Glass Transition and Water Dynamics in Hyaluronic Acid Hydrogels. Food Biophys. 2013, 8, 192–202. [Google Scholar] [CrossRef]
- Ghosh, K.; Shu, X.M.; Mou, R.; Lombardi, J.; Prestwich, G.D.; Rafailovich, M.H.; Clark, R.A.F. Rheological characteriza-tion of in situ cross-linkable hysluronan hydrogels. Biomacromolecules 2005, 6, 2857–2865. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, M.R.; Campese, G.M.; Radovanovic, E.; Rubira, A.; Feitosa, J.P.; Muniz, E.C. Morphology and water affinity of superabsorbent hydrogels composed of methacrylated cashew gum and acrylamide with good mechanical properties. Polymer 2005, 46, 7867–7873. [Google Scholar] [CrossRef]
- Noh, I.; Kim, G.C.; Kim, M.; Choi, Y.; Kim, M.; Park, Y.; Lee, K.; Kim, I.; Hwang, S.; Tae, G. Effects of cross-linking molec-ular weights in a hyaluronic acid–poly(ethylene oxide) hydrogel network on its properties. Biomed. Mater. 2006, 1, 116–123. [Google Scholar] [CrossRef] [PubMed]
- La Gatta, A.; Schiraldi, C.; Papa, A.; De Rosa, M. Comparative analysis of commercial dermal fillers based on crosslinked hyaluronan: Physical characterization and in vitro enzymatic degradation. Polym. Degrad. Stab. 2011, 96, 630–636. [Google Scholar] [CrossRef]
- La Gatta, A.; Papa, A.; Schiraldi, C.; De Rosa, M. Hyaluronan dermal fillers via crosslinking with 1,4-butandiol diglyc-idyl ether: Exploitation of heterogeneous reaction conditions. J. Biomed. Mater Res. B Appl. Biomater. 2016, 104, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Bitter, T.; Muir, H. A modified uronic acid carbazole reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciabica, S.; Tafuro, G.; Semenzato, A.; Traini, D.; Silva, D.M.; Reis, L.G.D.; Canilli, L.; Terno, M.; Durini, E.; Vertuani, S.; et al. Design, Synthesis, Characterization, and In Vitro Evaluation of a New Cross-Linked Hyaluronic Acid for Pharmaceutical and Cosmetic Applications. Pharmaceutics 2021, 13, 1672. https://doi.org/10.3390/pharmaceutics13101672
Sciabica S, Tafuro G, Semenzato A, Traini D, Silva DM, Reis LGD, Canilli L, Terno M, Durini E, Vertuani S, et al. Design, Synthesis, Characterization, and In Vitro Evaluation of a New Cross-Linked Hyaluronic Acid for Pharmaceutical and Cosmetic Applications. Pharmaceutics. 2021; 13(10):1672. https://doi.org/10.3390/pharmaceutics13101672
Chicago/Turabian StyleSciabica, Sabrina, Giovanni Tafuro, Alessandra Semenzato, Daniela Traini, Dina M. Silva, Larissa Gomes Dos Reis, Luisa Canilli, Massimo Terno, Elisa Durini, Silvia Vertuani, and et al. 2021. "Design, Synthesis, Characterization, and In Vitro Evaluation of a New Cross-Linked Hyaluronic Acid for Pharmaceutical and Cosmetic Applications" Pharmaceutics 13, no. 10: 1672. https://doi.org/10.3390/pharmaceutics13101672
APA StyleSciabica, S., Tafuro, G., Semenzato, A., Traini, D., Silva, D. M., Reis, L. G. D., Canilli, L., Terno, M., Durini, E., Vertuani, S., Baldisserotto, A., & Manfredini, S. (2021). Design, Synthesis, Characterization, and In Vitro Evaluation of a New Cross-Linked Hyaluronic Acid for Pharmaceutical and Cosmetic Applications. Pharmaceutics, 13(10), 1672. https://doi.org/10.3390/pharmaceutics13101672










