Antibacterial Layer-by-Layer Coatings for Medical Implants
Abstract
1. Introduction
2. Nosocomial Infections
3. Coatings for Titanium Implants
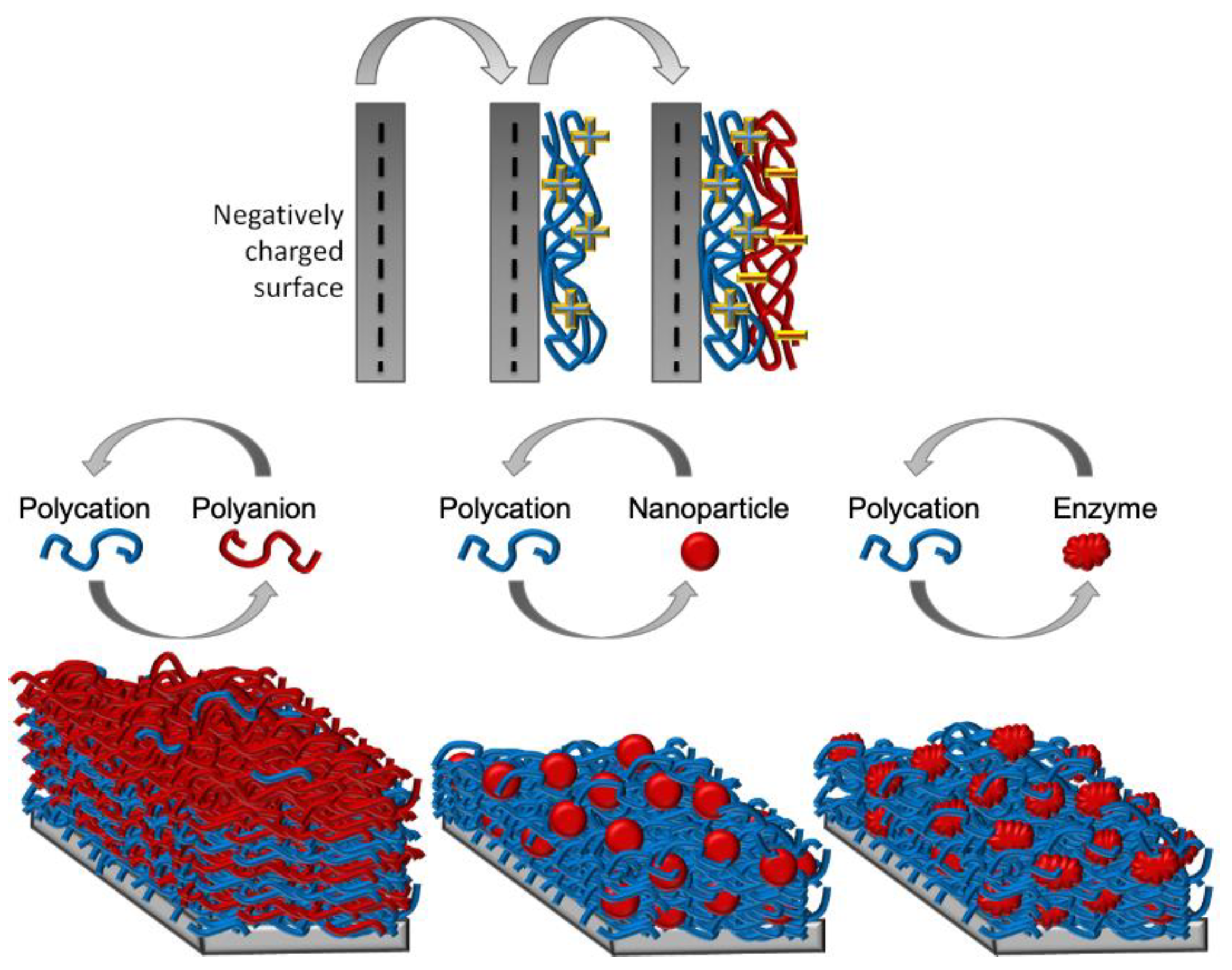
4. Layer-by-Layer Technique
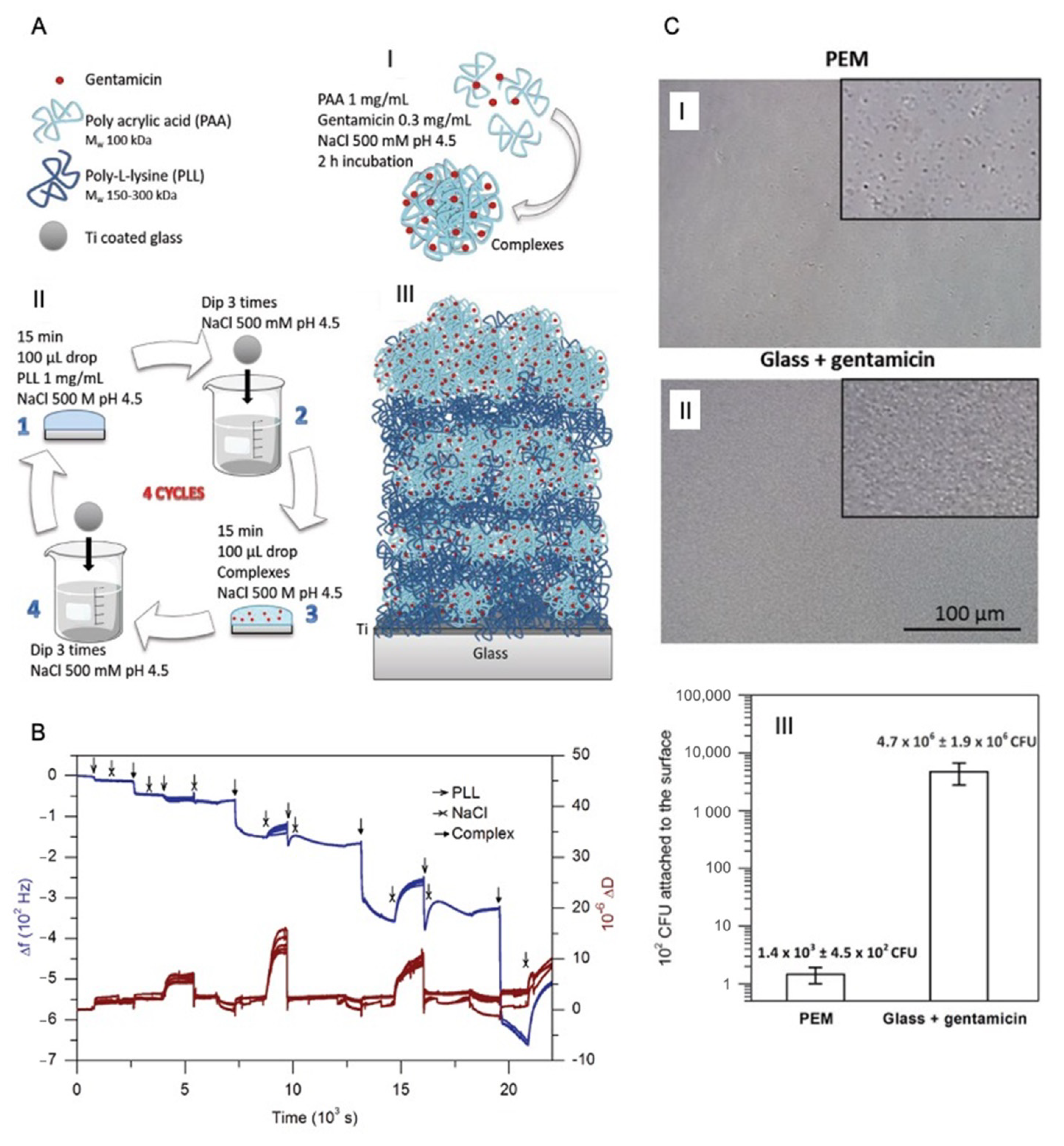
5. LbL for Antibiotic Encapsulation
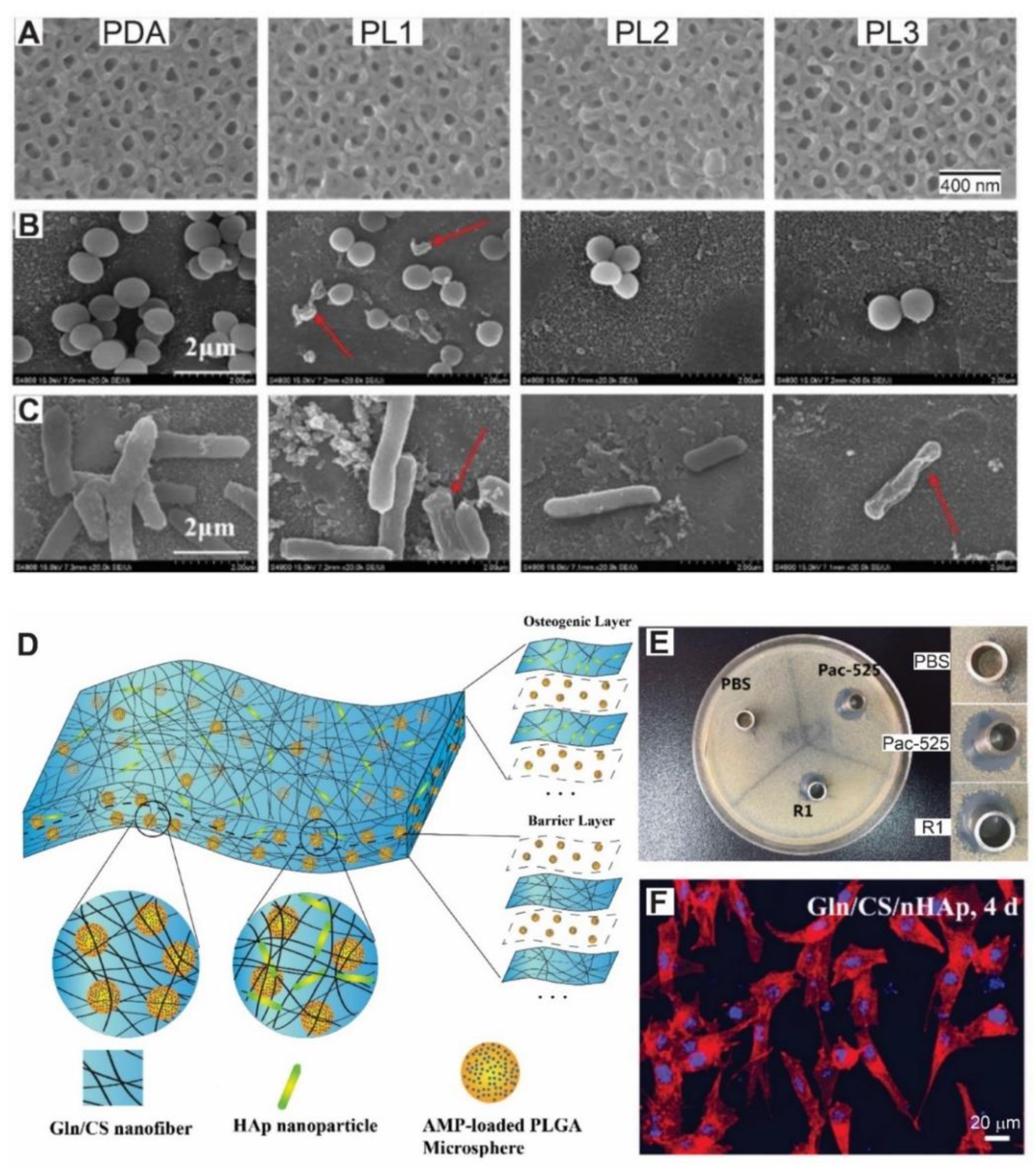
6. LbL for Antimicrobial Peptides Encapsulation
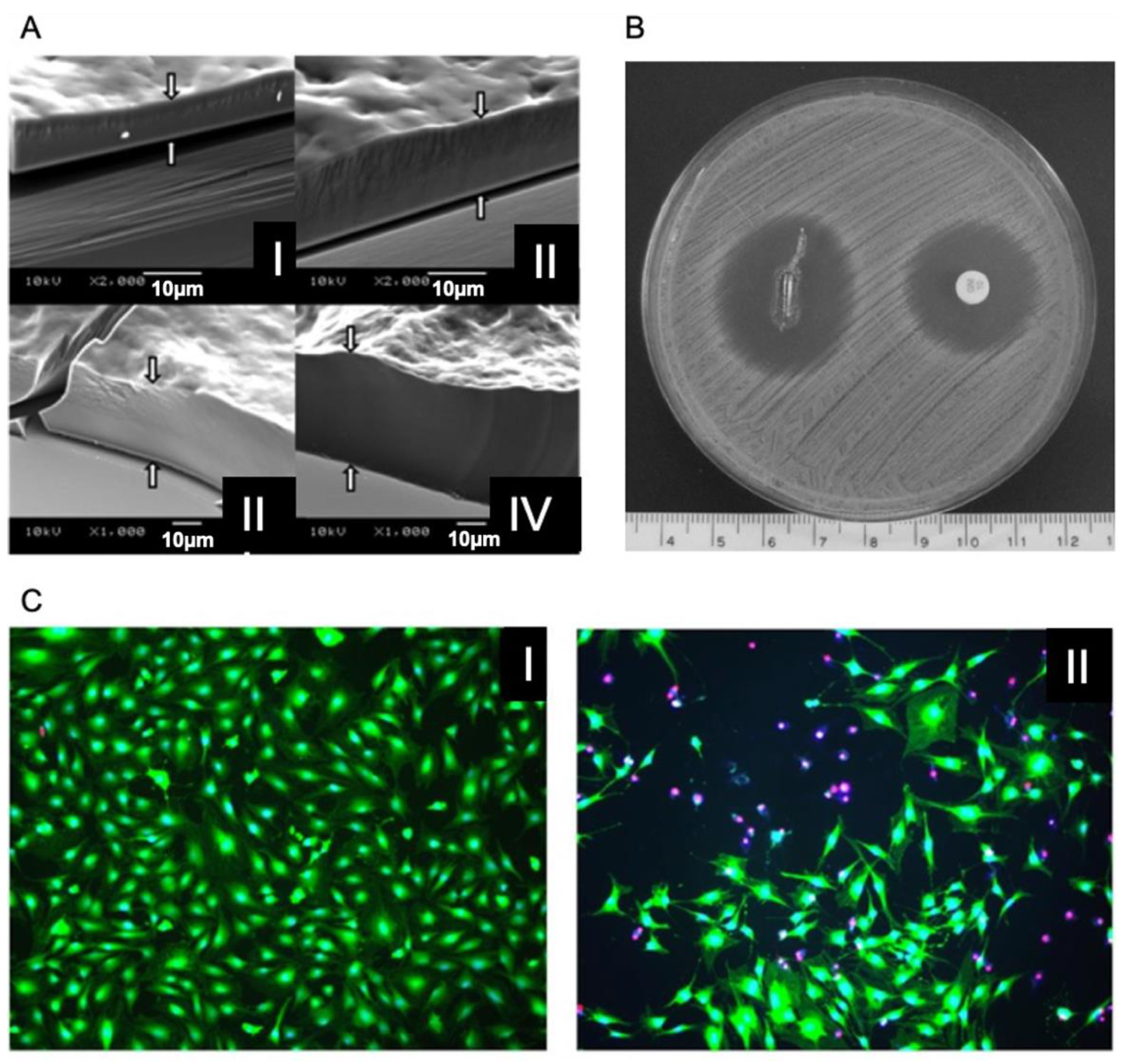
7. LbL for Antibacterial Nanoparticles Loading
8. LbL for Antiadhesive Surfaces
9. Multifunctional Antimicrobial Multilayers
10. Layer-by-Layer Assembly among Other Localized Antibacterial Strategies
11. Concluding Remarks and Future Perspectives
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pina, S.; Ribeiro, V.P.; Marques, C.F.; Maia, F.R.; Silva, T.H.; Reis, R.L.; Oliveira, J.M. Scaffolding strategies for tissue engineering and regenerative medicine applications. Materials 2019, 12, 1824. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, K.; Matsusaki, M.; Akashi, M. Control of cell surface and functions by layer-by-layer nanofilms. Langmuir 2010, 26, 5670–5678. [Google Scholar] [CrossRef] [PubMed]
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered biomaterials for in situ tissue regeneration. Nat. Rev. Mater. 2020, 5, 686–705. [Google Scholar] [CrossRef]
- Pados, G.; Makedos, A.; Tarlatzis, B. Endoscopy; Amornyotin, S., Ed.; InTech: London, UK, 2013; ISBN 978-953-51-1071-2. [Google Scholar]
- Park, H.; Baek, S.; Kang, H.; Lee, D. Biomaterials to Prevent Post-Operative Adhesion. Materials 2020, 13, 3056. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, M.; Mantovani, D.; Rosei, F. Antibacterial Coatings: Challenges, Perspectives, and Opportunities. Trends Biotechnol. 2015, 33, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, W.; Silva, P.; Silva, R.; Silva, G.; Machado, G.; Coelho, L.; Correia, M. Staphylococcus aureus and Staphylococcus epidermidis infections on implants. J. Hosp. Infect. 2018, 98, 111–117. [Google Scholar] [CrossRef]
- Dunne, W.M. Bacterial adhesion: Seen any good biofilms lately? Clin. Microbiol. Rev. 2002, 15, 155–166. [Google Scholar] [CrossRef]
- Lewis, K. Riddle of Biofilm Resistance. Antimicrob. Agents Chemother. 2001, 45, 999–1007. [Google Scholar] [CrossRef]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef]
- Moskowitz, J.S.; Blaisse, M.R.; Samuel, R.E.; Hsu, H.-P.; Harris, M.B.; Martin, S.D.; Lee, J.C.; Spector, M.; Hammond, P.T. The effectiveness of the controlled release of gentamicin from polyelectrolyte multilayers in the treatment of Staphylococcus aureus infection in a rabbit bone model. Biomaterials 2010, 31, 6019–6030. [Google Scholar] [CrossRef]
- Butterworth, M.; Payne, T. Surgical Infections. In Complications in Foot and Ankle Surgery; Springer International Publishing: Cham, Switzerland, 2017; pp. 69–87. [Google Scholar]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Pulido, L.; Ghanem, E.; Joshi, A.; Purtill, J.J.; Parvizi, J. Periprosthetic Joint Infection: The Incidence, Timing, and Predisposing Factors. Clin. Orthop. Relat. Res. 2008, 466, 1710–1715. [Google Scholar] [CrossRef] [PubMed]
- Blom, A.W.; Brown, J.; Taylor, A.H.; Pattison, G.; Whitehouse, S.; Bannister, G.C. Infection after total knee arthroplasty. J. Bone Jt. Surg. Br. 2004, 86, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, S.M.; Ong, K.L.; Lau, E.; Bozic, K.J.; Berry, D.; Parvizi, J. Prosthetic Joint Infection Risk after TKA in the Medicare Population. Clin. Orthop. Relat. Res. 2010, 468, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 91B, 470–480. [Google Scholar] [CrossRef]
- Brånemark, P.I.; Adell, R.; Breine, U.; Hansson, B.O.; Lindström, J.; Ohlsson, A. Intra-osseous anchorage of dental prostheses. I. Experimental studies. Scand. J. Plast. Reconstr. Surg. 1969, 3, 81–100. [Google Scholar] [CrossRef]
- Hetrick, E.M.; Schoenfisch, M.H. Reducing implant-related infections: Active release strategies. Chem. Soc. Rev. 2006, 35, 780. [Google Scholar] [CrossRef]
- Ribeiro, M.; Monteiro, F.J.; Ferraz, M.P. Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomatter 2012. [Google Scholar] [CrossRef]
- Tan, R.; Yoo, J.; Jang, Y. Engineering Approaches to Create Antibacterial Surfaces on Biomedical Implants and Devices. In Racing for the Surface; Springer International Publishing: Cham, Switzerland, 2020; pp. 313–340. [Google Scholar]
- Caldwell, M.D. Bacteria and Antibiotics in Wound Healing. Surg. Clin. N. Am. 2020, 100, 757–776. [Google Scholar] [CrossRef]
- Vasilev, K. Nanoengineered Antibacterial Coatings and Materials: A Perspective. Coatings 2019, 9, 654. [Google Scholar] [CrossRef]
- Ahmed, W.; Zhai, Z.; Gao, C. Adaptive antibacterial biomaterial surfaces and their applications. Mater. Today Bio 2019, 2, 100017. [Google Scholar] [CrossRef] [PubMed]
- Mas-Moruno, C.; Su, B.; Dalby, M.J. Multifunctional Coatings and Nanotopographies: Toward Cell Instructive and Antibacterial Implants. Adv. Healthc. Mater. 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.A.; Baig, F.K.; Mehboob, R. Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pac. J. Trop. Biomed. 2017, 7, 478–482. [Google Scholar] [CrossRef]
- McBryde, E.S.S.; Bradley, L.C.C.; Whitby, M.; McElwain, D.L.S.L.S. An investigation of contact transmission of methicillin-resistant Staphylococcus aureus. J. Hosp. Infect. 2004, 58, 104–108. [Google Scholar] [CrossRef]
- Rutledge-Taylor, K.; Matlow, A.; Gravel, D.; Embree, J.; Le Saux, N.; Johnston, L.; Suh, K.; Embil, J.; Henderson, E.; John, M.; et al. A point prevalence survey of health care-associated infections in Canadian pediatric inpatients. Am. J. Infect. Control 2012, 40, 491–496. [Google Scholar] [CrossRef]
- Klevens, R.M.; Edwards, J.R.; Richards, C.L.; Horan, T.C.; Gaynes, R.P.; Pollock, D.A.; Cardo, D.M. Estimating Health Care-Associated Infections and Deaths in U.S. Hospitals, 2002. Public Health Rep. 2007, 122, 160–166. [Google Scholar] [CrossRef]
- Hudson, M.C.; Ramp, W.K.; Frankenburg, K.P. Staphylococcus aureus adhesion to bone matrix and bone-associated biomaterials. FEMS Microbiol. Lett. 1999, 173, 279–284. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Gamberini, S.; Donati, M.E.; Montanaro, L. Presence of fibrinogen-binding adhesin gene in Staphylococcus epidermidis isolates from central venous catheters-associated and orthopaedic implant-associated infections. Biomaterials 2004, 25, 4825–4829. [Google Scholar] [CrossRef]
- Harris, L.G.; Richards, R.G. Staphylococci and implant surfaces: A review. Injury 2006, 37, S3–S14. [Google Scholar] [CrossRef]
- Kalita, S.J.; Verma, S. Nanocrystalline hydroxyapatite bioceramic using microwave radiation: Synthesis and characterization. Mater. Sci. Eng. C 2010, 30, 295–303. [Google Scholar] [CrossRef]
- Tschernitschek, H.; Borchers, L.; Geurtsen, W. Nonalloyed titanium as a bioinert metal-a review. Quintessence Int. 2005, 36, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Karthik, K.; Sivakumar, S.; Thangaswamy, V. Evaluation of implant success: A review of past and present concepts. J. Pharm. Bioallied Sci. 2013, 5, S117–S119. [Google Scholar] [CrossRef] [PubMed]
- Elias, C.N.; Lima, J.H.C.; Valiev, R.; Meyers, M.A. Biomedical applications of titanium and its alloys. JOM 2008, 60, 46–49. [Google Scholar] [CrossRef]
- Vijayaraghavan, V.; Sabane, A.V.; Tejas, K. Hypersensitivity to Titanium: A Less Explored Area of Research. J. Indian Prosthodont. Soc. 2012, 12, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D. Biomaterials Science: An Introduction to Materials in Medicine; Academic Press: Cambridge, MA, USA, 2013; ISBN 9780123746269. [Google Scholar]
- Muzzio, N.E.; Pasquale, M.A.; Rios, X.; Azzaroni, O.; Llop, J.; Moya, S.E. Adsorption and Exchangeability of Fibronectin and Serum Albumin Protein Corona on Annealed Polyelectrolyte Multilayers and Their Consequences on Cell Adhesion. Adv. Mater. Interfaces 2019, 6, 1900008. [Google Scholar] [CrossRef]
- Muzzio, N.E.; Gregurec, D.; Diamanti, E.; Irigoyen, J.; Pasquale, M.A.; Azzaroni, O.; Moya, S.E. Thermal Annealing of Polyelectrolyte Multilayers: An Effective Approach for the Enhancement of Cell Adhesion. Adv. Mater. Interfaces 2017, 4, 1600126. [Google Scholar] [CrossRef]
- Chouirfa, H.; Bouloussa, H.; Migonney, V.; Falentin-Daudré, C. Review of titanium surface modification techniques and coatings for antibacterial applications. Acta Biomater. 2019, 83, 37–54. [Google Scholar] [CrossRef] [PubMed]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Li, S.; Dong, S.; Xu, W.; Tu, S.; Yan, L.; Zhao, C.; Ding, J.; Chen, X. Antibacterial Hydrogels. Adv. Sci. 2018, 5, 1700527. [Google Scholar] [CrossRef]
- Escobar, A.; Muzzio, N.; Coy, E.; Liu, H.; Bindini, E.; Andreozzi, P.; Wang, G.; Angelomé, P.; Delcea, M.; Grzelczak, M.; et al. Antibacterial Mesoporous Titania Films with Embedded Gentamicin and Surface Modified with Bone Morphogenetic Protein 2 to Promote Osseointegration in Bone Implants. Adv. Mater. Interfaces 2019, 6. [Google Scholar] [CrossRef]
- Yu, Q.; Wu, Z.; Chen, H. Dual-function antibacterial surfaces for biomedical applications. Acta Biomater. 2015, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Gallo, J.; Holinka, M.; Moucha, C.S. Antibacterial surface treatment for orthopaedic implants. Int. J. Mol. Sci. 2014, 15, 13849–13880. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, I.S.; Schiffman, J.D.; Kurtz, I.S.; Schiffman, J.D. Current and Emerging Approaches to Engineer Antibacterial and Antifouling Electrospun Nanofibers. Materials 2018, 11, 1059. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Owh, C.; Chee, P.L.; Leow, W.R.; Liu, X.; Wu, Y.-L.; Guo, P.; Loh, X.J.; Chen, X. Supramolecular hydrogels for antimicrobial therapy. Chem. Soc. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.X.; Duan, S.; Ding, X.X.; Liu, R.; Xu, F.-J.J. Versatile Antibacterial Materials: An Emerging Arsenal for Combatting Bacterial Pathogens. Adv. Funct. Mater. 2018, 28, 1802140. [Google Scholar] [CrossRef]
- Idrees, A.; Varela, P.; Ruini, F.; Vasquez, J.M.; Salber, J.; Greiser, U.; Wang, W.; McMahon, S.; Sartori, S.; Ciardelli, G.; et al. Drug-free antibacterial polymers for biomedical applications. Biomed. Sci. Eng. 2018, 2. [Google Scholar] [CrossRef]
- Bassegoda, A.; Ivanova, K.; Ramon, E.; Tzanov, T. Strategies to prevent the occurrence of resistance against antibiotics by using advanced materials. Appl. Microbiol. Biotechnol. 2018, 102, 2075–2089. [Google Scholar] [CrossRef]
- Santos, M.R.E.; Mendonça, P.V.; Almeida, M.C.; Branco, R.; Serra, A.C.; Morais, P.V.; Coelho, J.F.J. Increasing the antimicrobial activity of amphiphilic cationic copolymers by the facile synthesis of high molecular weight stars by SARA ATRP. Biomacromolecules 2019, 20, 1146–1156. [Google Scholar] [CrossRef]
- Baier, G.; Cavallaro, A.; Friedemann, K.; Müller, B.; Glasser, G.; Vasilev, K.; Landfester, K. Enzymatic degradation of poly(l-lactide) nanoparticles followed by the release of octenidine and their bactericidal effects. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 131–139. [Google Scholar] [CrossRef]
- Stigter, M.; de Groot, K.; Layrolle, P. Incorporation of tobramycin into biomimetic hydroxyapatite coating on titanium. Biomaterials 2002, 23, 4143–4153. [Google Scholar] [CrossRef]
- Stigter, M.; Bezemer, J.; de Groot, K.; Layrolle, P. Incorporation of different antibiotics into carbonated hydroxyapatite coatings on titanium implants, release and antibiotic efficacy. J. Control. Release 2004, 99, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Radin, S.; Campbell, J.T.; Ducheyne, P.; Cuckler, J.M. Calcium phosphate ceramic coatings as carriers of vancomycin. Biomaterials 1997, 18, 777–782. [Google Scholar] [CrossRef]
- Alt, V.; Bitschnau, A.; Osterling, J.; Sewing, A.; Meyer, C.; Kraus, R.; Meissner, S.; Wenisch, S.; Domann, E.; Schnettler, R. The effects of combined gentamicin–hydroxyapatite coating for cementless joint prostheses on the reduction of infection rates in a rabbit infection prophylaxis model. Biomaterials 2006, 27, 4627–4634. [Google Scholar] [CrossRef] [PubMed]
- Lewandrowski, K.-U.; Schollmeier, G.; Ekkernkamp, A.; Mankin, H.J.; Uhthoff, H.K.; Tomford, W.W. Enhancing Cortical Allograft Incorporation Processing by Partial Demineralization and Laser Perforation. In Biomaterials Engineering and Devices: Human Applications; Humana Press: Totowa, NJ, USA, 2000; pp. 111–132. [Google Scholar]
- Campbell, A.A.; Song, L.; Li, X.S.; Nelson, B.J.; Bottoni, C.; Brooks, D.E.; DeJong, E.S. Development, characterization, and anti-microbial efficacy of hydroxyapatite-chlorhexidine coatings produced by surface-induced mineralization. J. Biomed. Mater. Res. 2000, 53, 400–407. [Google Scholar] [CrossRef]
- Morra, M.; Cassinelli, C.; Cascardo, G.; Mazzucco, L.; Borzini, P.; Fini, M.; Giavaresi, G.; Giardino, R. Collagen I-coated titanium surfaces: Mesenchymal cell adhesion and in vivo evaluation in trabecular bone implants. J. Biomed. Mater. Res. 2006, 78, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Ewald, A.; Glückermann, S.K.; Thull, R.; Gbureck, U. Antimicrobial titanium/silver PVD coatings on titanium. Biomed. Eng. Online 2006, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liu, Y.; Courtney, H.; Bettenga, M.; Agrawal, C.M.; Bumgardner, J.D.; Ong, J.L. In vitro anti-bacterial and biological properties of magnetron co-sputtered silver-containing hydroxyapatite coating. Biomaterials 2006, 27, 5512–5517. [Google Scholar] [CrossRef]
- Gallardo-Moreno, A.M.; Pacha-Olivenza, M.A.; Saldaña, L.; Pérez-Giraldo, C.; Bruque, J.M.; Vilaboa, N.; González-Martín, M.L. In vitro biocompatibility and bacterial adhesion of physico-chemically modified Ti6Al4V surface by means of UV irradiation. Acta Biomater. 2009, 5, 181–192. [Google Scholar] [CrossRef]
- Nablo, B.J.; Schoenfisch, M.H. In vitro cytotoxicity of nitric oxide-releasing sol-gel derived materials. Biomaterials 2005, 26, 4405–4415. [Google Scholar] [CrossRef]
- Hetrick, E.M.; Schoenfisch, M.H. Antibacterial nitric oxide-releasing xerogels: Cell viability and parallel plate flow cell adhesion studies. Biomaterials 2007, 28, 1948–1956. [Google Scholar] [CrossRef]
- Muzzarelli, R.; Tarsi, R.; Filippini, O.; Giovanetti, E.; Biagini, G.; Varaldo, P.E. Antimicrobial properties of N-carboxybutyl chitosan. Antimicrob. Agents Chemother. 1990, 34, 2019–2023. [Google Scholar] [CrossRef] [PubMed]
- Decher, G.; Hong, J.D.D.; Schmitt, J. Buildup of ultrathin multilayer films by a self-assembly process: III. Consecutively alternating adsorption of anionic and cationic polyelectrolytes on charged surfaces. Thin Solid Film. 1992, 210–211, 831–835. [Google Scholar] [CrossRef]
- Decher, G.; Eckle, M.; Schmitt, J.; Struth, B. Layer-by-layer assembled multicomposite films. Curr. Opin. Colloid Interface Sci. 1998, 3, 32–39. [Google Scholar] [CrossRef]
- Holmes, C.; Daoud, J.; Bagnaninchi, P.O.; Tabrizian, M. Polyelectrolyte Multilayer Coating of 3D Scaffolds Enhances Tissue Growth and Gene Delivery: Non-Invasive and Label-Free Assessment. Adv. Healthc. Mater. 2014, 3, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.; Izquierdo, A.; Decher, G.; Voegel, J.C.; Schaaf, P.; Ball, V. Layer by layer self-assembled polyelectrolyte multilayers with embedded phospholipid vesicles obtained by spraying: Integrity of the vesicles. Langmuir 2005, 21, 7854–7859. [Google Scholar] [CrossRef] [PubMed]
- Schlenoff, J.B.; Laurent, D.; Ly, H.; Stepp, J. Redox-Active Polyelectrolyte Multilayers. Adv. Mater. 1998, 10, 347–349. [Google Scholar] [CrossRef]
- Farhat, T.R.; Schlenoff, J.B. Ion Transport and Equilibria in Polyelectrolyte Multilayers. Langmuir 2001, 17, 1184–1192. [Google Scholar] [CrossRef]
- Onitsuka, O.; Fou, A.C.; Ferreira, M.; Hsieh, B.R.; Rubner, M.F. Enhancement of light emitting diodes based on self-assembled heterostructures of poly(p-phenylene vinylene). J. Appl. Phys. 1998, 80, 4067. [Google Scholar] [CrossRef]
- Ouyang, L.; Malaisamy, R.; Bruening, M.L. Multilayer polyelectrolyte films as nanofiltration membranes for separating monovalent and divalent cations. J. Memb. Sci. 2008, 310, 76–84. [Google Scholar] [CrossRef]
- Patel, I.F.; Kiryukhin, M.V.; Yakovlev, N.L.; Gupta, H.S.; Sukhorukov, G.B. Naturally inspired polyelectrolyte multilayer composite films synthesised through layer-by-layer assembly and chemically infiltrated with CaCO3. J. Mater. Chem. B 2015, 3, 4821–4830. [Google Scholar] [CrossRef]
- Mendelsohn, J.D.; Yang, S.Y.; Hiller, J.; Hochbaum, A.I.; Rubner, M.F. Rational Design of Cytophilic and Cytophobic Polyelectrolyte Multilayer Thin Films. Biomacromolecules 2003, 4, 96–106. [Google Scholar] [CrossRef]
- Bruening, M.; Dotzauer, D. Just spray it. Nat. Mater. 2009, 8, 449–450. [Google Scholar] [CrossRef]
- Lvov, Y.; Ariga, K.; Ichinose, I.; Kunitake, T. Assembly of Multicomponent Protein Films by Means of Electrostatic Layer-by-Layer Adsorption. J. Am. Chem. Soc. 1995, 117, 6117–6123. [Google Scholar] [CrossRef]
- Laurent, D.; Schlenoff, J.B. Multilayer Assemblies of Redox Polyelectrolytes. Langmuir 1997, 13, 1552–1557. [Google Scholar] [CrossRef]
- Dierendonck, M.; De Koker, S.; De Rycke, R.; De Geest, B.G. Just spray it-LbL assembly enters a new age. Soft Matter 2014, 10, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Krebs, T.; Tan, H.L.; Andersson, G.; Morgner, H.; Gregory Van Patten, P. Increased layer interdiffusion in polyelectrolyte films upon annealing in water and aqueous salt solutions. Phys. Chem. Chem. Phys. 2006, 8, 5462. [Google Scholar] [CrossRef]
- Shiratori, S.S.; Rubner, M.F. pH-Dependent Thickness Behavior of Sequentially Adsorbed Layers of Weak Polyelectrolytes. Macromolecules 2000, 33, 4213–4219. [Google Scholar] [CrossRef]
- Gregurec, D.; Olszyna, M.; Politakos, N.; Yate, L.; Dahne, L.; Moya, S.E. Stability of polyelectrolyte multilayers in oxidizing media: A critical issue for the development of multilayer based membranes for nanofiltration. Colloid Polym. Sci. 2015, 293, 381–388. [Google Scholar] [CrossRef]
- Decher, G.; Schmitt, J. Fine-Tuning of the film thickness of ultrathin multilayer films composed of consecutively alternating layers of anionic and cationic polyelectrolytes. In Trends in Colloid and Interface Science VI; Steinkopff: Darmstadt, Germany, 1992; pp. 160–164. [Google Scholar]
- De Villiers, M.M.; Otto, D.P.; Strydom, S.J.; Lvov, Y.M. Introduction to nanocoatings produced by layer-by-layer (LbL) self-assembly. Adv. Drug Deliv. Rev. 2011, 63, 701–715. [Google Scholar] [CrossRef]
- Xu, L.; Pristinski, D.; Zhuk, A.; Stoddart, C.; Ankner, J.F.; Sukhishvili, S.A. Linear versus exponential growth of weak polyelectrolyte multilayers: Correlation with polyelectrolyte complexes. Macromolecules 2012, 45, 3892–3901. [Google Scholar] [CrossRef]
- Richardson, J.J.; Bjornmalm, M.; Caruso, F. Technology-driven layer-by-layer assembly of nanofilms. Science 2015, 348, aaa2491. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.J.; Macdonald, M.L.; Beben, Y.M.; Padera, R.F.; Samuel, R.E.; Hammond, P.T. Tunable dual growth factor delivery from polyelectrolyte multilayer films. Biomaterials 2011, 32, 6183–6193. [Google Scholar] [CrossRef] [PubMed]
- Min, J.; Braatz, R.D.; Hammond, P.T. Tunable staged release of therapeutics from layer-by-layer coatings with clay interlayer barrier. Biomaterials 2014, 35, 2507–2517. [Google Scholar] [CrossRef]
- Macdonald, M.L.; Samuel, R.E.; Shah, N.J.; Padera, R.F.; Beben, Y.M.; Hammond, P.T. Tissue integration of growth factor-eluting layer-by-layer polyelectrolyte multilayer coated implants. Biomaterials 2011, 32, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Komeda, M.; Heo, J.; Hong, J.; Matsusaki, M.; Akashi, M. Vascular Endothelial Growth Factor Incorporated Multilayer Film Induces Preangiogenesis in Endothelial Cells. ACS Biomater. Sci. Eng. 2018, 4, 1833–1842. [Google Scholar] [CrossRef]
- Zhang, S.; Xing, M.; Li, B.; Zhang, S.; Xing, M.; Li, B. Biomimetic Layer-by-Layer Self-Assembly of Nanofilms, Nanocoatings, and 3D Scaffolds for Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 1641. [Google Scholar] [CrossRef]
- Goodman, S.B.; Yao, Z.; Keeney, M.; Yang, F. The future of biologic coatings for orthopaedic implants. Biomaterials 2013, 34, 3174–3183. [Google Scholar] [CrossRef]
- Romero, G.; Ochoteco, O.; Sanz, D.J.; Estrela-Lopis, I.; Donath, E.; Moya, S.E. Poly(Lactide- co -Glycolide) Nanoparticles, Layer by Layer Engineered for the Sustainable Delivery of AntiTNF-α. Macromol. Biosci. 2013, 13, 903–912. [Google Scholar] [CrossRef]
- Hammond, P.T. Building biomedical materials layer-by-layer. Mater. Today 2012, 15, 196–206. [Google Scholar] [CrossRef]
- Garza, J.M.; Jessel, N.; Ladam, G.; Dupray, V.; Muller, S.; Stoltz, J.-F.; Schaaf, P.; Voegel, J.-C.; Lavalle, P. Polyelectrolyte Multilayers and Degradable Polymer Layers as Multicompartment Films. Langmuir 2005, 21, 12372–12377. [Google Scholar] [CrossRef]
- Lavalle, P.; Gergely, C.; Cuisinier, F.J.G.; Decher, G.; Schaaf, P.; Voegel, J.C.; Picart, C. Comparison of the Structure of Polyelectrolyte Multilayer Films Exhibiting a Linear and an Exponential Growth Regime: An in Situ Atomic Force Microscopy Study. Macromolecules 2002, 35, 4458–4465. [Google Scholar] [CrossRef]
- Anderson, J.M. Biological Responses to Materials. Annu. Rev. Mater. Res. 2001, 31, 81–110. [Google Scholar] [CrossRef]
- Albright, V.; Zhuk, I.; Wang, Y.; Selin, V.; van de Belt-Gritter, B.; Busscher, H.J.; van der Mei, H.C.; Sukhishvili, S.A. Self-defensive antibiotic-loaded layer-by-layer coatings: Imaging of localized bacterial acidification and pH-triggering of antibiotic release. Acta Biomater. 2017, 61, 66–74. [Google Scholar] [CrossRef] [PubMed]
- de Avila, E.D.; Castro, A.G.B.; Tagit, O.; Krom, B.P.; Löwik, D.; van Well, A.A.; Bannenberg, L.J.; Vergani, C.E.; van den Beucken, J.J.J.P. Anti-bacterial efficacy via drug-delivery system from layer-by-layer coating for percutaneous dental implant components. Appl. Surf. Sci. 2019, 488, 194–204. [Google Scholar] [CrossRef]
- Escobar, A.; Muzzio, N.E.; Andreozzi, P.; Libertone, S.; Tasca, E.; Azzaroni, O.; Grzelczak, M.; Moya, S.E. Antibacterial Layer-by-Layer Films of Poly(acrylic acid)–Gentamicin Complexes with a Combined Burst and Sustainable Release of Gentamicin. Adv. Mater. Interfaces 2019, 6, 1901373. [Google Scholar] [CrossRef]
- Dwivedi, A.; Mazumder, A.; Nasongkla, N. Layer-by-layer nanocoating of antibacterial niosome on orthopedic implant. Int. J. Pharm. 2018, 547, 235–243. [Google Scholar] [CrossRef]
- Wongsuwan, N.; Dwivedi, A.; Tancharoen, S.; Nasongkla, N. Development of dental implant coating with minocycline-loaded niosome for antibacterial application. J. Drug Deliv. Sci. Technol. 2020, 56, 101555. [Google Scholar] [CrossRef]
- Horprasertkij, K.; Dwivedi, A.; Riansuwan, K.; Kiratisin, P.; Nasongkla, N. Spray coating of dual antibiotic-loaded nanospheres on orthopedic implant for prolonged release and enhanced antibacterial activity. J. Drug Deliv. Sci. Technol. 2019, 53, 101102. [Google Scholar] [CrossRef]
- Amin Yavari, S.; Croes, M.; Akhavan, B.; Jahanmard, F.; Eigenhuis, C.C.; Dadbakhsh, S.; Vogely, H.C.; Bilek, M.M.; Fluit, A.C.; Boel, C.H.E.; et al. Layer by layer coating for bio-functionalization of additively manufactured meta-biomaterials. Addit. Manuf. 2020, 32, 100991. [Google Scholar] [CrossRef]
- Min, J.; Choi, K.Y.; Dreaden, E.C.; Padera, R.F.; Braatz, R.D.; Spector, M.; Hammond, P.T. Designer Dual Therapy Nanolayered Implant Coatings Eradicate Biofilms and Accelerate Bone Tissue Repair. ACS Nano 2016. [Google Scholar] [CrossRef]
- Mahlapuu, M.; Håkansson, J.; Ringstad, L.; Björn, C. Antimicrobial Peptides: An Emerging Category of Therapeutic Agents. Front. Cell. Infect. Microbiol. 2016, 6, 194. [Google Scholar] [CrossRef] [PubMed]
- Magana, M.; Pushpanathan, M.; Santos, A.L.; Leanse, L.; Fernandez, M.; Ioannidis, A.; Giulianotti, M.A.; Apidianakis, Y.; Bradfute, S.; Ferguson, A.L.; et al. The value of antimicrobial peptides in the age of resistance. Lancet Infect. Dis. 2020, 20, e216–e230. [Google Scholar] [CrossRef]
- Lei, J.; Sun, L.C.; Huang, S.; Zhu, C.; Li, P.; He, J.; Mackey, V.; Coy, D.H.; He, Q.Y. The antimicrobial peptides and their potential clinical applications. Am. J. Transl. Res. 2019, 11, 3919–3931. [Google Scholar] [PubMed]
- Lázár, V.; Martins, A.; Spohn, R.; Daruka, L.; Grézal, G.; Fekete, G.; Számel, M.; Jangir, P.K.; Kintses, B.; Csörgő, B.; et al. Antibiotic-resistant bacteria show widespread collateral sensitivity to antimicrobial peptides. Nat. Microbiol. 2018, 3, 718–731. [Google Scholar] [CrossRef]
- Rezaei, N.; Hamidabadi, H.G.; Khosravimelal, S.; Zahiri, M.; Ahovan, Z.A.; Bojnordi, M.N.; Eftekhari, B.S.; Hashemi, A.; Ganji, F.; Darabi, S.; et al. Antimicrobial peptides-loaded smart chitosan hydrogel: Release behavior and antibacterial potential against antibiotic resistant clinical isolates. Int. J. Biol. Macromol. 2020, 164, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Felgueiras, H.P.; Amorim, M.T.P. Functionalization of electrospun polymeric wound dressings with antimicrobial peptides. Colloids Surf. B Biointerfaces 2017, 156, 133–148. [Google Scholar] [CrossRef]
- Zaccaria, S.; van Gaal, R.C.; Riool, M.; Zaat, S.A.J.; Dankers, P.Y.W. Antimicrobial peptide modification of biomaterials using supramolecular additives. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 1926–1934. [Google Scholar] [CrossRef]
- Zhu, X.; Jun Loh, X.; Neoh, K.-G.; Kang, E.-T.; Teo, S.L.-M.; Rittschof, D.; Zhao, C.; Li, X.; Egles, C.; Detrembleur, C.; et al. Layer-by-layer assemblies for antibacterial applications. Biomater. Sci. 2015, 3, 1505–1518. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Huang, Q.; Patil, A.B.; Hu, J.; Fan, L.; Yang, Y.; Duan, H.; Dong, X.; Lin, C. Layer-by-layer immobilizing of polydopamine-assisted ε-polylysine and gum Arabic on titanium: Tailoring of antibacterial and osteogenic properties. Mater. Sci. Eng. C 2020, 110, 110690. [Google Scholar] [CrossRef]
- He, Y.; Jin, Y.; Wang, X.; Yao, S.; Li, Y.; Wu, Q.; Ma, G.; Cui, F.; Liu, H. An Antimicrobial Peptide-Loaded Gelatin/Chitosan Nanofibrous Membrane Fabricated by Sequential Layer-by-Layer Electrospinning and Electrospraying Techniques. Nanomaterials 2018, 8, 327. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Zhou, Z.; Lai, H.; Xu, P.; Liao, L.; Wei, J. Biodegradable polymer membranes applied in guided bone/tissue regeneration: A review. Polymers 2016, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Pranantyo, D.; Zhang, B.; Xu, L.; Neoh, K.G.; Kang, E.T. Tannic acid anchored layer-by-layer covalent deposition of parasin i peptide for antifouling and antimicrobial coatings. RSC Adv. 2016, 6, 14809–14818. [Google Scholar] [CrossRef]
- Shi, J.; Liu, Y.; Wang, Y.; Zhang, J.; Zhao, S.; Yang, G. Biological and immunotoxicity evaluation of antimicrobial peptide-loaded coatings using a layer-by-layer process on titanium. Sci. Rep. 2015, 5, 16336. [Google Scholar] [CrossRef] [PubMed]
- Dorner, F.; Malek-Luz, A.; Saar, J.S.; Bonaus, S.; Al-Ahmad, A.; Lienkamp, K. Synthetic Mimics of Antimicrobial Peptides (SMAMPs) in Layer-by-Layer Architectures: Possibilities and Limitations. Macromol. Chem. Phys. 2016, 217, 2154–2164. [Google Scholar] [CrossRef]
- Hemeg, H. Nanomaterials for alternative antibacterial therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Zheng, J. Antibacterial Activity of Silver Nanoparticles: Structural Effects. Adv. Healthc. Mater. 2018, 7. [Google Scholar] [CrossRef]
- Carvalho, A.L.; Vale, A.C.; Sousa, M.P.; Barbosa, A.M.; Torrado, E.; Mano, J.F.; Alves, N.M. Antibacterial bioadhesive layer-by-layer coatings for orthopedic applications. J. Mater. Chem. B 2016, 4, 5385–5393. [Google Scholar] [CrossRef]
- Kruk, T.; Gołda-Cępa, M.; Szczepanowicz, K.; Szyk-Warszyńska, L.; Brzychczy-Włoch, M.; Kotarba, A.; Warszyński, P. Nanocomposite multifunctional polyelectrolyte thin films with copper nanoparticles as the antimicrobial coatings. Colloids Surf. B Biointerfaces 2019, 181, 112–118. [Google Scholar] [CrossRef]
- Gadkari, R.R.; Ali, S.W.; Joshi, M.; Rajendran, S.; Das, A.; Alagirusamy, R. Leveraging antibacterial efficacy of silver loaded chitosan nanoparticles on layer-by-layer self-assembled coated cotton fabric. Int. J. Biol. Macromol. 2020, 162, 548–560. [Google Scholar] [CrossRef]
- Liu, Z.; Qi, L.; An, X.; Liu, C.; Hu, Y. Surface Engineering of Thin Film Composite Polyamide Membranes with Silver Nanoparticles through Layer-by-Layer Interfacial Polymerization for Antibacterial Properties. ACS Appl. Mater. Interfaces 2017, 9, 40987–40997. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; He, H.; Xiao, J.; Zhao, P.; Xie, J.; Lu, Z. Controllable in situ synthesis of silver nanoparticles on multilayered film-coated silk fibers for antibacterial application. J. Colloid Interface Sci. 2016, 461, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Montelongo, J.; Lucchesi, E.G.; Gonzalez, I.; Macedo, W.A.A.; Nascimento, V.F.; Moraes, A.M.; Beppu, M.M.; Cotta, M.A. Hyaluronan/chitosan nanofilms assembled layer-by-layer and their antibacterial effect: A study using Staphylococcus aureus and Pseudomonas aeruginosa. Colloids Surf. B Biointerfaces 2016, 141, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Muzzio, N.E.; Pasquale, M.A.; Diamanti, E.; Gregurec, D.; Martinez Moro, M.; Azzaroni, O.; Moya, S.E. Enhanced antiadhesive properties of chitosan/hyaluronic acid polyelectrolyte multilayers driven by thermal annealing: Low adherence for mammalian cells and selective decrease in adhesion for Gram-positive bacteria. Mater. Sci. Eng. C 2017, 80, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Muzzio, N.E.; Pasquale, M.A.; Moya, S.E.; Azzaroni, O. Tailored polyelectrolyte thin film multilayers to modulate cell adhesion. Biointerphases 2017, 12, 04E403. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Kwek, M.Y.; Toh, Z.Q.; Pranantyo, D.; Kang, E.T.; Loh, X.J.; Zhu, X.; Jańczewski, D.; Neoh, K.G. Tailoring Polyelectrolyte Architecture to Promote Cell Growth and Inhibit Bacterial Adhesion. ACS Appl. Mater. Interfaces 2018, 10, 7882–7891. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, Y.; Liang, J.; Libera, M.R.; Sukhishvili, S.A. Self-defensive antibacterial layer-by-layer hydrogel coatings with pH-triggered hydrophobicity. Biomaterials 2015, 45, 64–71. [Google Scholar] [CrossRef]
- Xu, G.; Pranantyo, D.; Xu, L.; Neoh, K.G.; Kang, E.T.; Teo, S.L.M. Antifouling, Antimicrobial, and Antibiocorrosion Multilayer Coatings Assembled by Layer-by-layer Deposition Involving Host-Guest Interaction. Ind. Eng. Chem. Res. 2016, 55, 10906–10915. [Google Scholar] [CrossRef]
- Tang, L.; Huynh, K.A.; Fleming, M.L.; Larronde-Larretche, M.; Chen, K.L. Imparting antimicrobial and anti-adhesive properties to polysulfone membranes through modification with silver nanoparticles and polyelectrolyte multilayers. J. Colloid Interface Sci. 2015, 451, 125–133. [Google Scholar] [CrossRef]
- Vaterrodt, A.; Thallinger, B.; Daumann, K.; Koch, D.; Guebitz, G.M.; Ulbricht, M. Antifouling and Antibacterial Multifunctional Polyzwitterion/Enzyme Coating on Silicone Catheter Material Prepared by Electrostatic Layer-by-Layer Assembly. Langmuir 2016, 32, 1347–1359. [Google Scholar] [CrossRef]
- Kurapati, R.; Vaidyanathan, M.; Raichur, A.M. Synergistic photothermal antimicrobial therapy using graphene oxide/polymer composite layer-by-layer thin films. RSC Adv. 2016, 6, 39852–39860. [Google Scholar] [CrossRef]
- Wang, M.; Tang, T. Surface treatment strategies to combat implant-related infection from the beginning. J. Orthop. Transl. 2019, 17, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, M.; Siove, A.; Gueguen, V.; Gamble, L.J.; Castner, D.G.; Migonney, V. Radical Graft Polymerization of Styrene Sulfonate on Poly(ethylene terephthalate) Films for ACL Applications: “Grafting From” and Chemical Characterization. Biomacromolecules 2006. [Google Scholar] [CrossRef] [PubMed]
- Alcheikh, A.; Pavon-Djavid, G.; Helary, G.; Petite, H.; Migonney, V.; Anagnostou, F. PolyNaSS grafting on titanium surfaces enhances osteoblast differentiation and inhibits Staphylococcus aureus adhesion. J. Mater. Sci. Mater. Med. 2013, 24, 1745–1754. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, T.D.; Rao, M.S.; Koh, C.Y.; Stupp, S.I. Covalent functionalization of NiTi surfaces with bioactive peptide amphiphile nanofibers. Biomaterials 2008, 29, 1085–1098. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.; Abke, J.; Schnell, E.; Scharnweber, D.; Kujat, R.; Englert, C.; Taheri, D.; Nerlich, M.; Angele, P. Influence of surface pretreatment of titanium- and cobalt-based biomaterials on covalent immobilization of fibrillar collagen. Biomaterials 2006, 27, 4059–4068. [Google Scholar] [CrossRef]
- Ercan, B.; Taylor, E.; Alpaslan, E.; Webster, T.J. Diameter of titanium nanotubes influences anti-bacterial efficacy. Nanotechnology 2011, 22. [Google Scholar] [CrossRef]
- Puckett, S.D.; Taylor, E.; Raimondo, T.; Webster, T.J. The relationship between the nanostructure of titanium surfaces and bacterial attachment. Biomaterials 2010, 31, 706–713. [Google Scholar] [CrossRef]
- Harris, L.G.; Tosatti, S.; Wieland, M.; Textor, M.; Richards, R.G. Staphylococcus aureus adhesion to titanium oxide surfaces coated with non-functionalized and peptide-functionalized poly(L-lysine)-grafted- poly(ethylene glycol) copolymers. Biomaterials 2004, 25, 4135–4148. [Google Scholar] [CrossRef]
- Bräuer, G.; Szyszka, B.; Vergöhl, M.; Bandorf, R. Magnetron sputtering—Milestones of 30 years. Vacuum 2010, 84, 1354–1359. [Google Scholar] [CrossRef]
- Jemat, A.; Ghazali, M.J.; Razali, M.; Otsuka, Y. Surface modifications and their effects on titanium dental implants. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, R.; Muñoz-Piña, S.; González, M.U.; Izquierdo-Barba, I.; Fernández-Martínez, I.; Rico, V.; Arcos, D.; García-Valenzuela, A.; Palmero, A.; Vallet-Regi, M.; et al. Antibacterial Nanostructured Ti Coatings by Magnetron Sputtering: From Laboratory Scales to Industrial Reactors. Nanomaterials 2019, 9, 1217. [Google Scholar] [CrossRef] [PubMed]
- Qadir, M.; Li, Y.; Wen, C. Ion-substituted calcium phosphate coatings by physical vapor deposition magnetron sputtering for biomedical applications: A review. Acta Biomater. 2019, 89, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Shirai, T.; Shimizu, T.; Ohtani, K.; Zen, Y.; Takaya, M.; Tsuchiya, H. Antibacterial iodine-supported titanium implants. Acta Biomater. 2011, 7, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Iturri Ramos, J.J.; Stahl, S.; Richter, R.P.; Moya, S.E. Water content and buildup of poly(diallyldimethylammonium chloride)/poly(sodium 4-styrenesulfonate) and poly(allylamine hydrochloride)/poly(sodium 4-styrenesulfonate) polyelectrolyte multilayers studied by an in situ combination of a quartz crystal microb. Macromolecules 2010, 43, 9063–9070. [Google Scholar] [CrossRef]

| Antibacterial Mechanism | Composition | Bacteria Tested | Findings | Reference |
|---|---|---|---|---|
| Antibiotic Encapsulation | Gentamicin sulphate, PAA, Poly 1 | S. aureus (Gram+) | Bactericidal effect and nontoxic towards MC3T3-E1 pre-osteoblasts | [11] |
| Antibiotic Encapsulation | Poly(methacrylic acid), SNARF-1, gentamicin or polymyxin B | S. aureus (Gram+) and E. coli (Gram−) | Antibiotic release is triggered by pH changes. High bactericidal efficiency | [99] |
| Antibiotic Encapsulation | PAA, PLL and tetracycline | Porphyromonas gingivalis (Gram−) | Initial burst release of tetracycline, which antibacterial effects | [100] |
| Antibiotic Encapsulation | PAA-Gentamicin complexes and PLL | S. aureus (Gram+) | Prevents bacteria proliferation with low number of bilayers. Antibiotic burst release followed by a prolonged release | [101] |
| Antibiotic Encapsulation | Vancomycin-loaded niosomes and PLA | S. aureus (Gram+) | Antibacterial activity with no cytotoxic effects on L929 mouse fibroblast cells | [102] |
| Antibiotic Encapsulation | Gelatin, BMP-2, CHI, Vancomycin | S. aureus (Gram+) | Bactericidal effect and enhanced osteogenic differentiation of mesenchymal stem cells | [105] |
| Antimicrobial Peptides Encapsulation | Polydopamine, ε-polylysine, gum Arabic | S. aureus (Gram+) and E. coli (Gram−) | Long-term antibacterial properties and improved proliferation and osteogenic differentiation of rBMSCs | [115] |
| Antimicrobial Peptides Encapsulation | Gelatin, CHI, (Pac-525)-loaded PLGA microspheres | S. aureus (Gram+) and E. coli (Gram−) | Bactericidal activity for one week and bacteriostatic activity for up to a month. Good adhesion, proliferation and osteogenic differentiation of rBMSCs | [116] |
| Antimicrobial Peptides Encapsulation | Polyphenolic tannic acid and parasin I | S. aureus, S. epidermidis (Gram+) and E. coli, Pseudomonas sp. (Gram−) | The coating exhibits good resistance to bacteria adhesion | [118] |
| Antimicrobial Peptides Encapsulation | CHI, HA and Tet213 linked to collagen IV | S. aureus (Gram+) and P. gingivalis (Gram−) | Bacteria growth inhibition for up to a month. Multilayer presents good adhesion of keratinocyte cell line and non-cytotoxicity | [119] |
| Antibacterial Nanoparticles Loading | CHI, dopamine-modified HA, silver doped bioactive glass NPs | S. aureus (Gram+) and E. coli (Gram−) | Antibacterial properties against both bacteria. The film supports L929 fibroblast cells adhesion and proliferation. Good adhesion strength to other materials. | [124] |
| Antibacterial Nanoparticles Loading | PSS, PAH, silver nanoparticle-loaded chitosan | S. aureus (Gram+) and E. coli (Gram−) | Antibacterial effect due to due to a barrier mechanism/antifouling effect and silver ions release | [126] |
| Antibacterial Nanoparticles Loading | BSA capped Ag NPs, m-phenylenediamine, trimesoyl chloride | E. coli (Gram−) | Film assembled through Layer-by-Layer interfacial polymerization with good antimicrobial activity | [127] |
| Antibacterial Nanoparticles Loading | PAA, PDDA, in situ growth of silver NPs | S. aureus (Gram+) and E. coli (Gram−) | Antibacterial efficacy tuned by varying the number of PAA/PDDA bilayers | [128] |
| Antibacterial Nanoparticles Loading | PDDA and copper NPs | S. aureus (Gram+) | Antibacterial action caused by cell lysis upon contact of the bacteria with the surface of the multilayer | [125] |
| Antiadhesive Surface | CHI, HA | S. aureus (Gram+) and P. aeruginosa (Gram−) | Antibacterial properties can be tuned by changing the pH of the polymer solutions and number of layers. Total suppression of S. aureus’s growth, little effect on P. aeruginosa. | [129] |
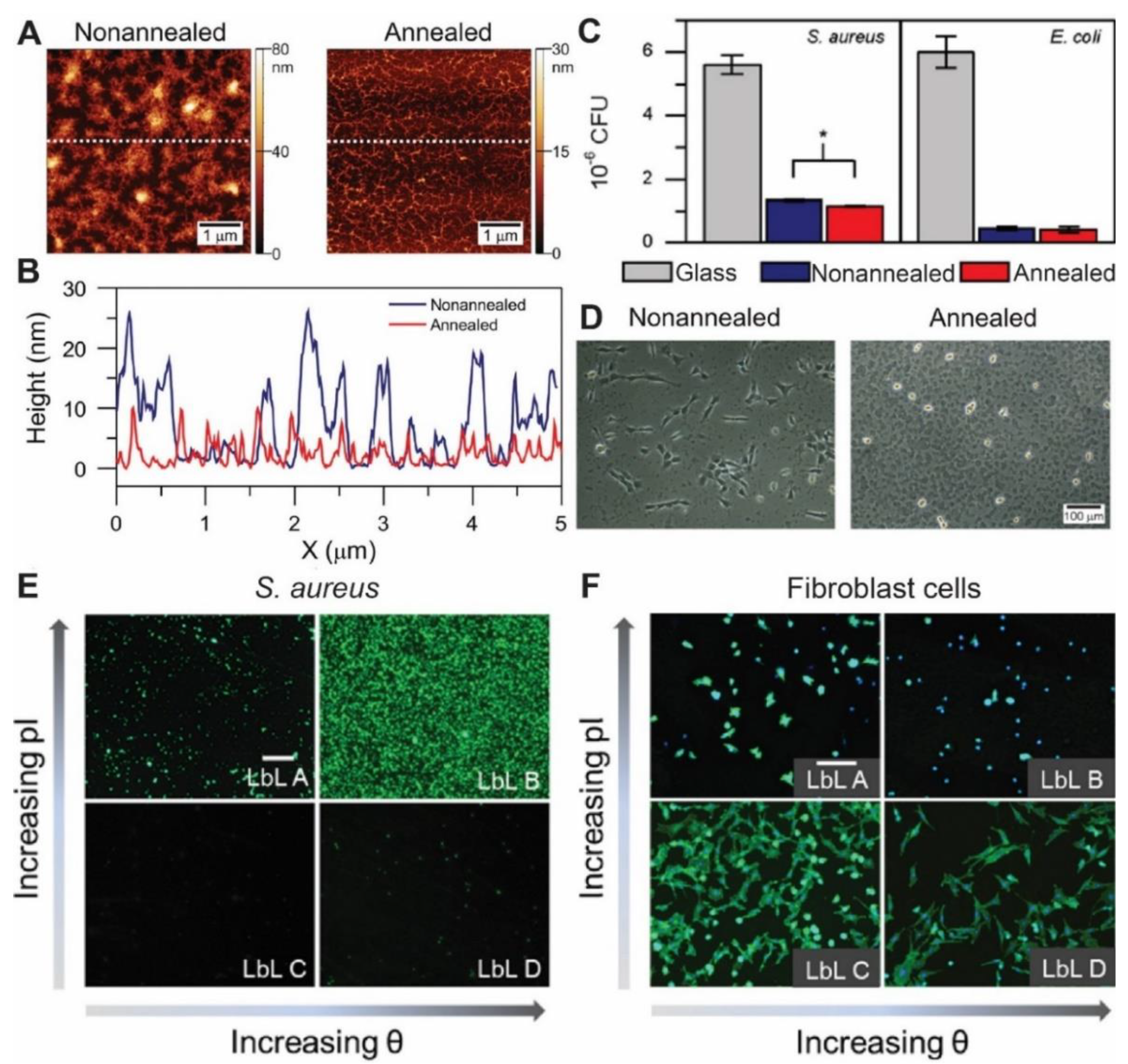
| Antiadhesive Surface | CHI, HA | S. aureus (Gram+) and E. coli (Gram−) | Low bacteria adhesion on films. After thermal annealing of the films, S. aureus’ adhesion further decreases | [130] |
| Antiadhesive Surface | Branched PEI and custom-synthesized polyanions | S. aureus (Gram+) and E. coli (Gram−) | Surface charge and wettability control bacteria and 3T3 fibroblasts adhesion on non-cross-linked soft films | [132] |
| Antiadhesive Surface | PVPON and different polyacids with increasing alkyl side chain length | S. epidermidis (Gram+) | Film with pH-triggered hydrophobicity with antiadhesive and bactericidal properties. | [133] |
| Antiadhesive Surface | PEI-β-cyclodextrin and synthetic ferrocene-modified CHI | S. aureus (Gram+) and Pseudomonas sp. (Gram−) | Antifouling and antimicrobial performance increase with the number of bilayers | [134] |
| Multifunctional Antibacterial Multilayer | PAH, PAA, silver NPs | E. coli (Gram−) | Antiadhesive properties due to PAH/PAA and bactericidal properties due to silver NPs | [135] |
| Multifunctional Antibacterial Multilayer | Cellobiose dehydrogenase, synthetic antifouling copolymer and PSS | S. aureus (Gram+) | The location of the hydrogen peroxide-producing enzyme in the multilayer was found to influence its activity. The films presented both antifouling and antimicrobial properties | [136] |
| Multifunctional Antibacterial Multilayer | PAH and graphene oxide | E. coli (Gram−) | Combined antimicrobial effects (membrane stress + photothermal heating) cause increased bacteria lysis in comparison to the individual effects | [137] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Escobar, A.; Muzzio, N.; Moya, S.E. Antibacterial Layer-by-Layer Coatings for Medical Implants. Pharmaceutics 2021, 13, 16. https://doi.org/10.3390/pharmaceutics13010016
Escobar A, Muzzio N, Moya SE. Antibacterial Layer-by-Layer Coatings for Medical Implants. Pharmaceutics. 2021; 13(1):16. https://doi.org/10.3390/pharmaceutics13010016
Chicago/Turabian StyleEscobar, Ane, Nicolas Muzzio, and Sergio Enrique Moya. 2021. "Antibacterial Layer-by-Layer Coatings for Medical Implants" Pharmaceutics 13, no. 1: 16. https://doi.org/10.3390/pharmaceutics13010016
APA StyleEscobar, A., Muzzio, N., & Moya, S. E. (2021). Antibacterial Layer-by-Layer Coatings for Medical Implants. Pharmaceutics, 13(1), 16. https://doi.org/10.3390/pharmaceutics13010016






