Development and In Vivo Application of a Water-Soluble Anticancer Copper Ionophore System Using a Temperature-Sensitive Liposome Formulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Drug-Loaded Liposomes
2.3. Dynamic Light Scattering (DLS)
2.4. Microfluidic Resistive Pulse Sensing (MRPS)
2.5. IR Spectroscopy
2.6. Differential Scanning Calorimetry (DSC)
2.7. Stability of the Liposomes
2.8. Size Exclusion Chromatography (SEC)
2.9. Cell Lines
2.10. Cell Viability Assay
2.11. Sample Preparation for Determination of Intracellular Cu Levels
2.12. Total-Reflection X-Ray Fluorescence (TXRF) Analysis
2.13. In Vivo Anti-Tumor Efficacy of Drug-Containing Liposomes
2.14. Radiolabeling of Liposomes with Cu-64 for In Vivo PET Imaging
2.15. Small Animal Imaging
3. Results and Discussion
3.1. Optimization of the Liposomal Formulation
3.1.1. Optimal Copper(II) Concentration
3.1.2. Optimal Drug to Phospholipid Ratio
3.1.3. Optimization of the Lipid Composition of Thermosensitive Liposomes
3.1.4. Characterization of the Optimized Liposomal Formulations
3.1.5. Stability of Liposomal Preparations
3.2. Cytotoxic Effect of the Liposomes on Colorectal Cancer Cells
3.3. Cu Accumulation by the Liposomal Formulation In Vitro
3.4. In Vivo Effect of the Liposomal Formulations
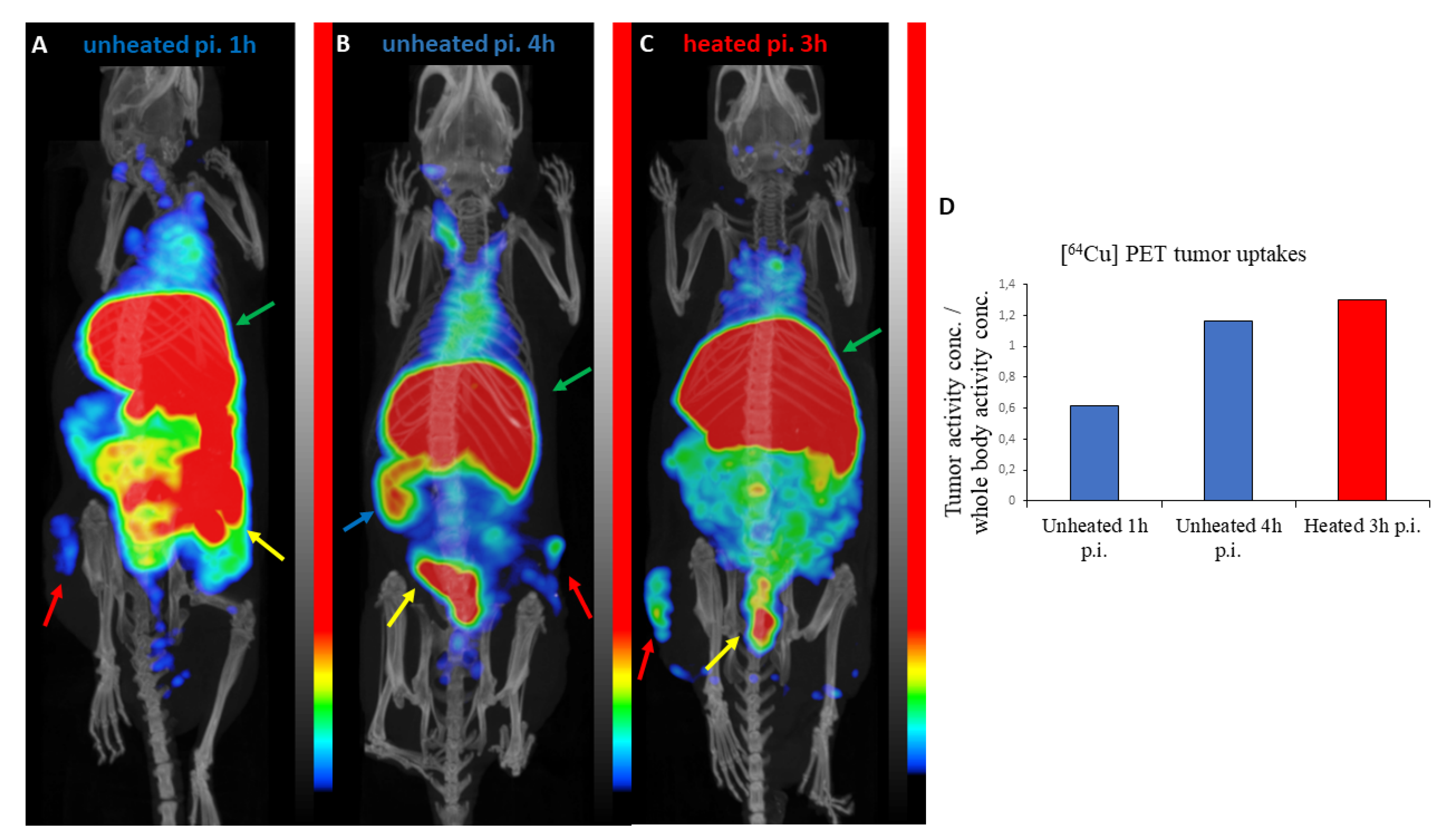
3.5. PET/CT Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Menyhárt, O.; Fekete, J.T.; Győrffy, B. Demographic shift disproportionately increases cancer burden in an aging nation: Current and expected incidence and mortality in Hungary up to 2030. Clin. Epidemiol. 2018, 10, 1093–1108. [Google Scholar] [CrossRef]
- Ghosh, S. Cisplatin: The first metal based anticancer drug. Bioorganic Chem. 2019, 88, 102925. [Google Scholar] [CrossRef]
- Galluzzi, L.; Senovilla, L.; Vitale, I.; Michels, J.; Martins, I.; Kepp, O.; Castedo, M.; Kroemer, G. Molecular mechanisms of cisplatin resistance. Oncogene 2012, 31, 1869–1883. [Google Scholar] [CrossRef]
- Dilruba, S.; Kalayda, G.V. Platinum-based drugs: Past, present and future. Cancer Chemother. Pharmacol. 2016, 77, 1103–1124. [Google Scholar] [CrossRef]
- Farrell, N.P. Multi-platinum anti-cancer agents. Substitution-inert compounds for tumor selectivity and new targets. Chem. Soc. Rev. 2015, 44, 8773–8785. [Google Scholar] [CrossRef]
- Ndagi, U.; Mhlongo, N.; Soliman, M.E. Metal complexes in cancer therapy—An update from drug design perspective. Drug Des. Devel Ther. 2017, 11, 599–616. [Google Scholar] [CrossRef]
- Duncan, C.; White, A.R. Copper complexes as therapeutic agents. Metallomics 2012, 4, 127–138. [Google Scholar] [CrossRef]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C. Advances in copper complexes as anticancer agents. Chem. Rev. 2014, 114, 815–862. [Google Scholar] [CrossRef]
- Denoyer, D.; Masaldan, S.; Fontaine, S.L.; Cater, M.A. Targeting copper in cancer therapy: ‘Copper That Cancer’. Metallomics 2015, 7, 1459–1476. [Google Scholar] [CrossRef]
- Daniel, K.G.; Chen, D.; Orlu, S.; Cui, Q.C.; Miller, F.R.; Dou, Q.P. Clioquinol and pyrrolidine dithiocarbamate complex with copper to form proteasome inhibitors and apoptosis inducers in human breast cancer cells. Breast Cancer Res. 2005, 7, R897–R908. [Google Scholar] [CrossRef] [PubMed]
- Daniel, K.G.; Gupta, P.; Harbach, R.H.; Guida, W.C.; Dou, Q.P. Organic copper complexes as a new class of proteasome inhibitors and apoptosis inducers in human cancer cells. Biochem. Pharmacol. 2004, 67, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Gaál, A.; Orgován, G.; Polgári, Z.; Réti, A.; Mihucz, V.G.; Bősze, S.; Szoboszlai, N.; Streli, C. Complex forming competition and in-vitro toxicity studies on the applicability of di-2-pyridylketone-4,4,-dimethyl-3-thiosemicarbazone (Dp44mT) as a metal chelator. J. Inorg. Biochem. 2014, 130, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Lovejoy, D.B.; Jansson, P.J.; Brunk, U.T.; Wong, J.; Ponka, P.; Richardson, D.R. Antitumor activity of metal-chelating compound Dp44mT is mediated by formation of a redox-active copper complex that accumulates in lysosomes. Cancer Res. 2011, 71, 5871–5880. [Google Scholar] [CrossRef] [PubMed]
- Gaál, A.; Mihucz, V.G.; Bősze, S.; Szabó, I.; Baranyi, M.; Horváth, P.; Streli, C.; Szoboszlai, N. Comparative in vitro investigation of anticancer copper chelating agents. Microchem. J. 2018, 136, 227–235. [Google Scholar] [CrossRef]
- Ganguly, A.; Basu, S.; Banerjee, K.; Chakraborty, P.; Sarkar, A.; Chatterjee, M.; Chaudhuri, S.K. Redox active copper chelate overcomes multidrug resistance in T-lymphoblastic leukemia cell by triggering apoptosis. Mol. BioSyst. 2011, 7, 1701–1712. [Google Scholar] [CrossRef]
- Rakshit, A.; Khatua, K.; Shanbhag, V.; Comba, P.; Datta, A. Cu2+ selective chelators relieve copper-induced oxidative stress in vivo. Chem. Sci. 2018, 9, 7916–7930. [Google Scholar] [CrossRef]
- Gaál, A.; Orgován, G.; Mihucz, V.G.; Pape, I.; Ingerle, D.; Streli, C.; Szoboszlai, N. Metal transport capabilities of anticancer copper chelators. J. Trace Elem. Med. Biol. 2018, 47, 79–88. [Google Scholar] [CrossRef]
- Kim, K.K.; Kawar, N.M.; Singh, R.K.; Lange, T.S.; Brard, L.; Moore, R.G. Tetrathiomolybdate induces doxorubicin sensitivity in resistant tumor cell lines. Gynecol. Oncol. 2011, 122, 183–189. [Google Scholar] [CrossRef]
- Roy, S.; Saha, S.; Majumdar, R.; Dighe, R.R.; Chakravarty, A.R. DNA photocleavage and anticancer activity of terpyridine copper(II) complexes having phenanthroline bases. Polyhedron 2010, 29, 2787–2794. [Google Scholar] [CrossRef]
- Kucková, L.; Jomová, K.; Švorcová, A.; Valko, M.; Segľa, P.; Moncoľ, J.; Kožíšek, J. Synthesis, Crystal Structure, Spectroscopic Properties and Potential Biological Activities of Salicylate‒Neocuproine Ternary Copper(II) Complexes. Molecules 2015, 20, 2115–2137. [Google Scholar] [CrossRef]
- Alvarez, N.; Viña, D.; Leite, C.M.; Mendes, L.F.S.; Batista, A.A.; Ellena, J.; Costa-Filho, A.J.; Facchin, G. Synthesis and structural characterization of a series of ternary copper(II)-L-dipeptide-neocuproine complexes. Study of their cytotoxicity against cancer cells including MDA-MB-231, triple negative breast cancer cells. J. Inorg. Biochem. 2020, 203, 110930. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-H.; Lin, J.-K.; Liu, S.-H.; Liang, Y.-C.; Lin-Shiau, S.-Y. Apoptosis of Cultured Astrocytes Induced by the Copper and Neocuproine Complex through Oxidative Stress and JNK Activation. Toxicol. Sci. 2008, 102, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Chen, D.; Zhang, X.; Cui, Q.; Fan, Y.; Bi, C.; Dou, Q.P. Molecular study on copper-mediated tumor proteasome inhibition and cell death. Int. J. Oncol. 2010, 37, 81–87. [Google Scholar]
- Mohindru, A.; Fisher, J.M.; Rabinovitz, M. 2,9-Dimethyl-1,10-phenanthroline (neocuproine): A potent, copper-dependent cytotoxin with anti-tumor activity. Biochem. Pharmacol. 1983, 32, 3627–3632. [Google Scholar] [CrossRef]
- Zhu, B.Z.; Chevion, M. Copper-mediated toxicity of 2,4,5-trichlorophenol: Biphasic effect of the copper(I)-specific chelator neocuproine. Arch. Biochem. Biophys. 2000, 380, 267–273. [Google Scholar] [CrossRef]
- Maeda, H.; Wu, J.; Sawa, T.; Matsumura, Y.; Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. J. Control Release 2000, 65, 271–284. [Google Scholar] [CrossRef]
- Shao, S.; Si, J.; Shen, Y. Copper as the Target for Anticancer Nanomedicine. Adv. Ther. 2019, 2, 1800147. [Google Scholar] [CrossRef]
- Needham, D.; Anyarambhatla, G.; Kong, G.; Dewhirst, M.W. A new temperature-sensitive liposome for use with mild hyperthermia: Characterization and testing in a human tumor xenograft model. Cancer Res. 2000, 60, 1197–1201. [Google Scholar] [PubMed]
- Wehbe, M.; Leung, A.W.Y.; Abrams, M.J.; Orvig, C.; Bally, M.B. A Perspective—Can copper complexes be developed as a novel class of therapeutics? Dalton Trans. 2017, 46, 10758–10773. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, S.; Lin, T.-M.; Krugner-Higby, L.; Lyman, D.; Steffen, D.; Xiong, M.P. Evaluating the Anticancer Properties of Liposomal Copper in a Nude Mouse Xenograft Model of Human Prostate Cancer: Formulation, In Vitro, In Vivo, Histology and Tissue Distribution Studies. Pharm. Res. 2014, 31, 3106–3119. [Google Scholar] [CrossRef] [PubMed]
- Marengo, A.; Forciniti, S.; Dando, I.; Dalla Pozza, E.; Stella, B.; Tsapis, N.; Yagoubi, N.; Fanelli, G.; Fattal, E.; Heeschen, C.; et al. Pancreatic cancer stem cell proliferation is strongly inhibited by diethyldithiocarbamate-copper complex loaded into hyaluronic acid decorated liposomes. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Wehbe, M.; Anantha, M.; Backstrom, I.; Leung, A.; Chen, K.; Malhotra, A.; Edwards, K.; Bally, M.B. Nanoscale Reaction Vessels Designed for Synthesis of Copper-Drug Complexes Suitable for Preclinical Development. PLoS ONE 2016, 11, e0153416. [Google Scholar] [CrossRef] [PubMed]
- Wehbe, M.; Lo, C.; Leung, A.W.Y.; Dragowska, W.H.; Ryan, G.M.; Bally, M.B. Copper (II) complexes of bidentate ligands exhibit potent anti-cancer activity regardless of platinum sensitivity status. Investig. New Drugs 2017, 35, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, E.; Alnajim, J.; Anantha, M.; Taggar, A.; Thomas, A.; Edwards, K.; Karlsson, G.; Webb, M.; Bally, M. Transition metal-mediated liposomal encapsulation of irinotecan (CPT-11) stabilizes the drug in the therapeutically active lactone conformation. Pharm. Res. 2006, 23, 2799–2808. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.; van der Pol, E.; Pálmai, M.; Garcia-Diez, R.; Gollwitzer, C.; Krumrey, M.; Fraikin, J.-L.; Gasecka, A.; Hajji, N.; van Leeuwen, T.G.; et al. Hollow organosilica beads as reference particles for optical detection of extracellular vesicles. J. Thromb. Haemost. 2018. [Google Scholar] [CrossRef]
- Fraikin, J.-L.; Teesalu, T.; McKenney, C.M.; Ruoslahti, E.; Cleland, A.N. A high-throughput label-free nanoparticle analyser. Nat. Nanotechnol. 2011, 6, 308–313. [Google Scholar] [CrossRef]
- Grabarek, A.D.; Weinbuch, D.; Jiskoot, W.; Hawe, A. Critical Evaluation of Microfluidic Resistive Pulse Sensing for Quantification and Sizing of Nanometer- and Micrometer-Sized Particles in Biopharmaceutical Products. J. Pharm. Sci. 2019, 108, 563–573. [Google Scholar] [CrossRef]
- Barnett, G.V.; Perhacs, J.M.; Das, T.K.; Kar, S.R. Submicron Protein Particle Characterization using Resistive Pulse Sensing and Conventional Light Scattering Based Approaches. Pharm. Res. 2018, 35, 58. [Google Scholar] [CrossRef]
- Szoboszlai, N.; Polgári, Z.; Mihucz, V.G.; Záray, G. Recent trends in total reflection X-ray fluorescence spectrometry for biological applications. Anal. Chim. Acta 2009, 633, 1–18. [Google Scholar] [CrossRef]
- Polgári, Z.; Ajtony, Z.; Kregsamer, P.; Streli, C.; Mihucz, V.G.; Réti, A.; Budai, B.; Kralovánszky, J.; Szoboszlai, N.; Záray, G. Microanalytical method development for Fe, Cu and Zn determination in colorectal cancer cells. Talanta 2011, 85, 1959–1965. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Sayre, L.M. Oxidation of tertiary amine buffers by copper(II). Inorg. Chem. 1989, 28, 169–170. [Google Scholar] [CrossRef]
- Caffrey, M. Lipid Thermotropic Phase Transition Database (LIPIDAT). User’s Guide Version 1.0 Version 1.0; U.S. Department of Commerce, National Institute of Standards and Technology, Standard Reference Data Program: Gaithersburg, MD, USA, 1993.
- Silvius, J.R. Thermotropic Phase Transitions of Pure Lipids in Model Membranes and Their Modifications by Membrane Proteins. In Lipid-Protein Interactions; Jost, P.C., Griffith, O.H., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1982; pp. 239–281. Available online: http://www.oalib.com/references/8155109 (accessed on 8 March 2020).
- Varga, Z.; Fehér, B.; Kitka, D.; Wacha, A.; Bóta, A.; Berényi, S.; Pipich, V.; Fraikin, J.-L. Size Measurement of Extracellular Vesicles and Synthetic Liposomes: The Impact of the Hydration Shell and the Protein Corona. Colloids Surf. B Biointerfaces 2020, 192, 111053. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.N.; McElhaney, R.N.; Pohle, W.; Mantsch, H.H. Components of the carbonyl stretching band in the infrared spectra of hydrated 1,2-diacylglycerolipid bilayers: A reevaluation. Biophys. J. 1994, 67, 2367–2375. [Google Scholar] [CrossRef]
- Berényi, S.; Mihály, J.; Wacha, A.; Tőke, O.; Bóta, A. A mechanistic view of lipid membrane disrupting effect of PAMAM dendrimers. Colloids Surf. B Biointerfaces 2014, 118, 164–171. [Google Scholar] [CrossRef]
- Petersen, A.L.; Henriksen, J.R.; Binderup, T.; Elema, D.R.; Rasmussen, P.H.; Hag, A.M.; Kjær, A.; Andresen, T.L. In vivo evaluation of PEGylated 64Cu-liposomes with theranostic and radiotherapeutic potential using micro PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 941–952. [Google Scholar] [CrossRef]
- Man, F.; Gawne, P.J.; de Rosales, R.T. Nuclear imaging of liposomal drug delivery systems: A critical review of radiolabelling methods and applications in nanomedicine. Adv. Drug Deliv. Rev. 2019, 143, 134–160. [Google Scholar] [CrossRef]
- Bar-David, S.; Larush, L.; Goder, N.; Aizic, A.; Zigmond, E.; Varol, C.; Klausner, J.; Magdassi, S.; Nizri, E. Size and lipid modification determine liposomal Indocyanine green performance for tumor imaging in a model of rectal cancer. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef]
- Caracciolo, G.; Pozzi, D.; Capriotti, A.L.; Cavaliere, C.; Piovesana, S.; Amenitsch, H.; Laganà, A. Lipid composition: A “key factor” for the rational manipulation of the liposome–protein corona by liposome design. RSC Adv. 2014, 5, 5967–5975. [Google Scholar] [CrossRef]
- Chakravarty, R.; Chakraborty, S.; Dash, A. 64Cu2+ Ions as PET Probe: An Emerging Paradigm in Molecular Imaging of Cancer. Mol. Pharm. 2016, 13, 3601–3612. [Google Scholar] [CrossRef]
- Peng, F.; Lu, X.; Janisse, J.; Muzik, O.; Shields, A.F. PET of human prostate cancer xenografts in mice with increased uptake of 64CuCl2. J. Nucl. Med. 2006, 47, 1649–1652. [Google Scholar] [PubMed]
- Phillips, null Delivery of gamma-imaging agents by liposomes. Adv. Drug Deliv. Rev. 1999, 37, 13–32. [CrossRef]
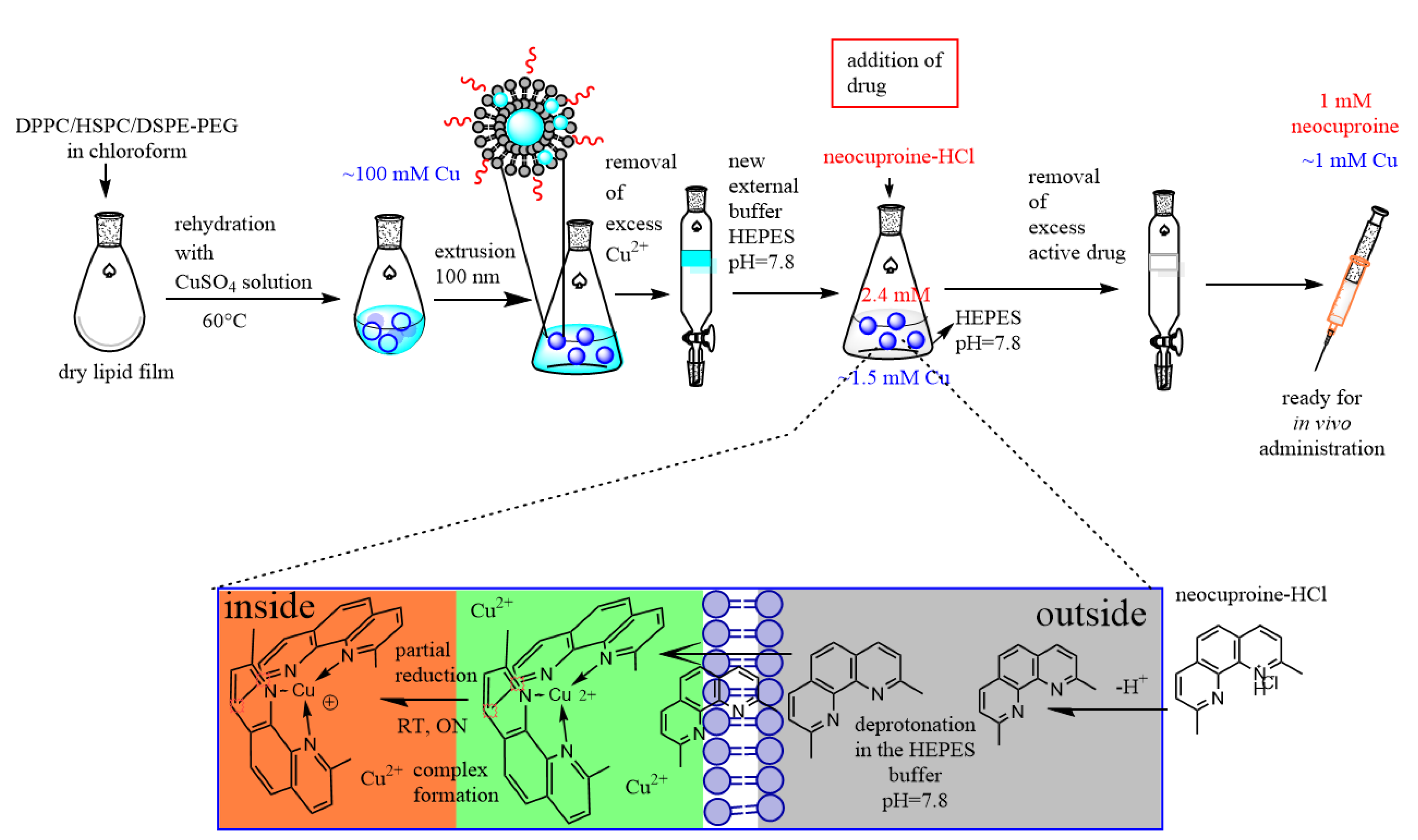
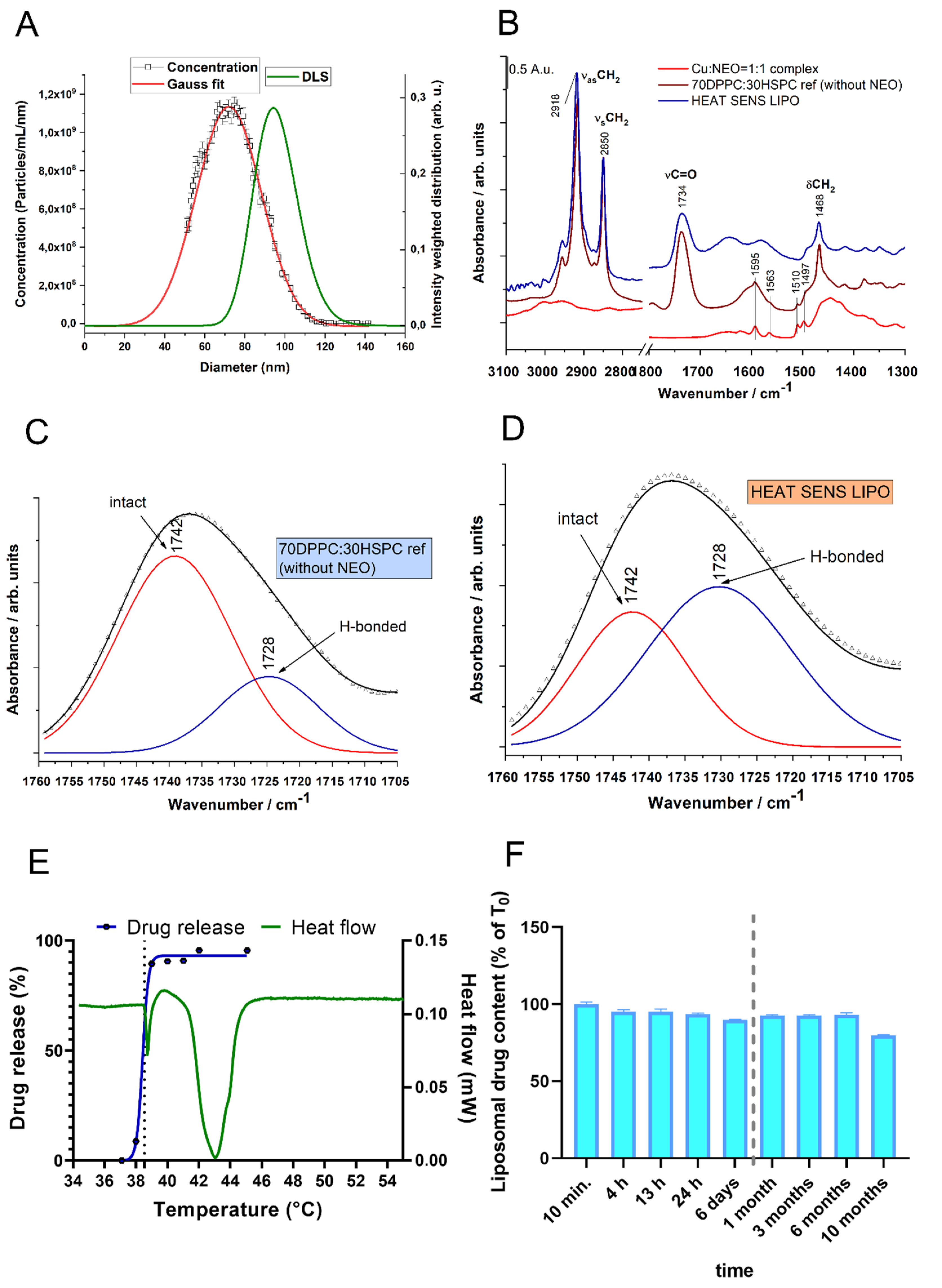
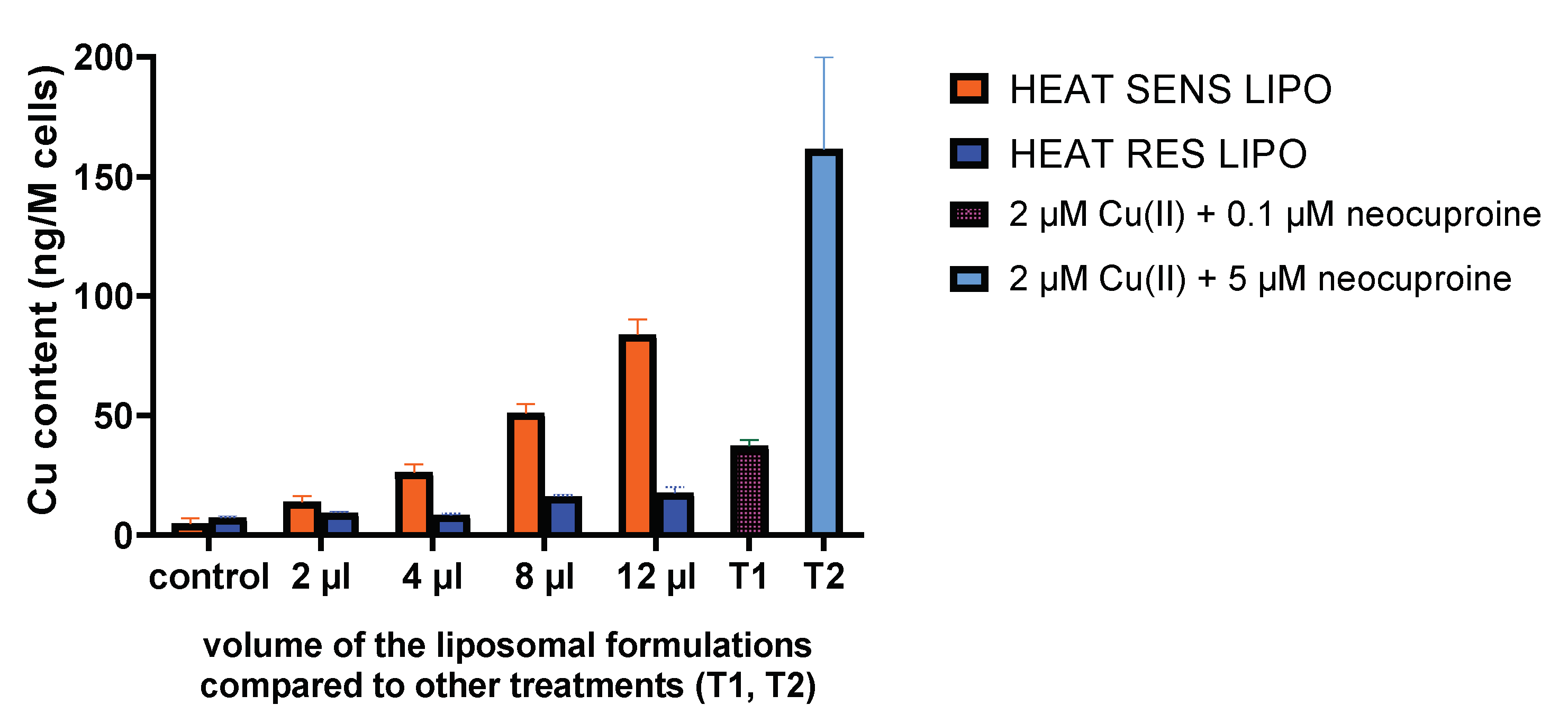
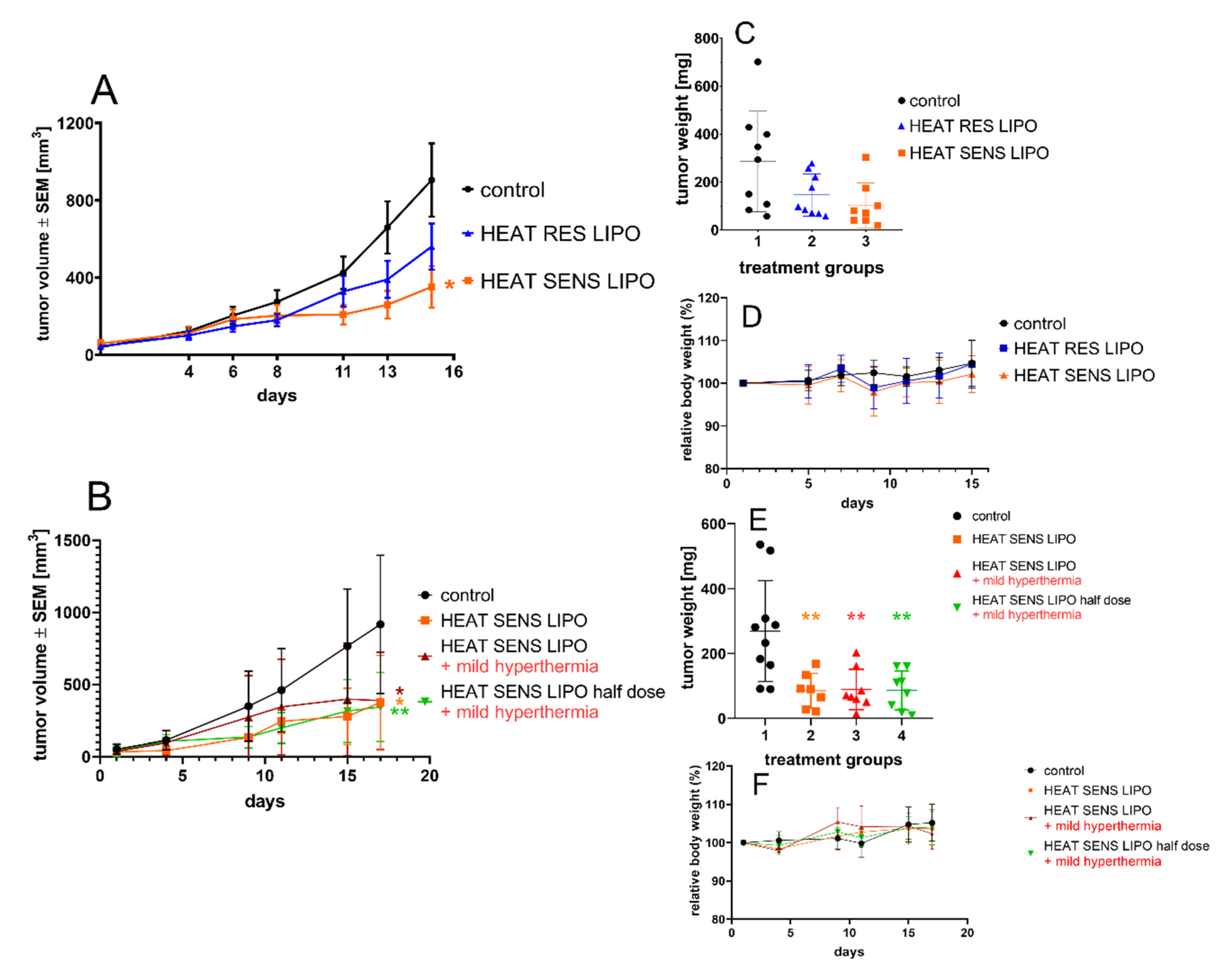
| Sample Name | DPPC (mg) | HSPC (mg) | DSPE-PEG2000 (mg) | w/w% of DPPC and HSPC |
|---|---|---|---|---|
| LIPO1 hereinafter HEAT RES LIPO | 0 | 30 | 5 | 100%HSPC:PEG |
| LIPO2 | 15 | 15 | 5 | 50%DPPC:50%HSPC:PEG |
| LIPO3 hereinafter HEAT SENS LIPO | 21 | 9 | 5 | 70%DPPC:30%HSPC:PEG |
| LIPO4 | 24 | 6 | 5 | 80%DPPC:20%HSPC:PEG |
| LIPO5 | 27 | 3 | 5 | 90%DPPC:10%HSPC:PEG |
| LIPO6 | 30 | 0 | 5 | 100%DPPC:PEG |
| Liposome Formulation | IC50 (µM) (RSD < 20%) | ||
|---|---|---|---|
| 37 °C | 38 °C | 39 °C | |
| LIPO1 HEAT RES LIPO | >40 | >40 | ~40 |
| LIPO2 | >40 | >40 | ~40 |
| LIPO3 HEAT SENS LIPO | >40 | >40 | 36.8 |
| LIPO4 | >40 | 30.9 | 15.5 |
| LIPO5 | >40 | 9.9 | 5.2 |
| LIPO6 | ~40 | 5.9 | 4.6 |
| Treatment Parameters | IC50 Values (µM) on HT-29 and C26 Cell Lines (RSD < 20%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| HEAT SENS LIPO | HEAT RES LIPO | Compared to other Treatments | |||||||
| Cell Line | Time | Without Pre-Heating 37 °C | Heating to 42 °C | without Pre-Heating 37 °C | Heating to 42 °C | Neocuproine | 2 µM Cu(II) + Neocuproine | Cu(II)-Neocuproine Preformed Complex 1:1 | Cu(II)-Neocuproine Preformed Complex 1:2 |
| HT-29 | 4 h | >40 | 19.4 | >40 | >40 | >500 | 1.6 | 1.15 | n.a. |
| 24 h | 5 | 2.8 | 9.8 | 10.1 | ~300 | 0.16 | 0.6 | >25 | |
| 72 h | 0.2 | 0.2 | 0.2 | 0.3 | 0.083 | 0.025 | 0.05 | 0.053 | |
| C26 | 4 h | >40 | 18.6 | >40 | >40 | >500 | 1.8 | 1.05 | n.a. |
| 24 h | 3.6 | 3 | 4.2 | 4.1 | ~500 | 0.21 | 0.49 | >25 | |
| 72 h | 0.2 | 0.15 | 0.3 | 0.2 | 0.15 | 0.037 | 0.13 | 0.12 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaál, A.; Garay, T.M.; Horváth, I.; Máthé, D.; Szöllősi, D.; Veres, D.S.; Mbuotidem, J.; Kovács, T.; Tóvári, J.; Bergmann, R.; et al. Development and In Vivo Application of a Water-Soluble Anticancer Copper Ionophore System Using a Temperature-Sensitive Liposome Formulation. Pharmaceutics 2020, 12, 466. https://doi.org/10.3390/pharmaceutics12050466
Gaál A, Garay TM, Horváth I, Máthé D, Szöllősi D, Veres DS, Mbuotidem J, Kovács T, Tóvári J, Bergmann R, et al. Development and In Vivo Application of a Water-Soluble Anticancer Copper Ionophore System Using a Temperature-Sensitive Liposome Formulation. Pharmaceutics. 2020; 12(5):466. https://doi.org/10.3390/pharmaceutics12050466
Chicago/Turabian StyleGaál, Anikó, Tamás M. Garay, Ildikó Horváth, Domokos Máthé, Dávid Szöllősi, Dániel S. Veres, Jeremiah Mbuotidem, Tibor Kovács, József Tóvári, Ralf Bergmann, and et al. 2020. "Development and In Vivo Application of a Water-Soluble Anticancer Copper Ionophore System Using a Temperature-Sensitive Liposome Formulation" Pharmaceutics 12, no. 5: 466. https://doi.org/10.3390/pharmaceutics12050466
APA StyleGaál, A., Garay, T. M., Horváth, I., Máthé, D., Szöllősi, D., Veres, D. S., Mbuotidem, J., Kovács, T., Tóvári, J., Bergmann, R., Streli, C., Szakács, G., Mihály, J., Varga, Z., & Szoboszlai, N. (2020). Development and In Vivo Application of a Water-Soluble Anticancer Copper Ionophore System Using a Temperature-Sensitive Liposome Formulation. Pharmaceutics, 12(5), 466. https://doi.org/10.3390/pharmaceutics12050466






