Encapsulating TGF-β1 Inhibitory Peptides P17 and P144 as a Promising Strategy to Facilitate Their Dissolution and to Improve Their Functionalization
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Fabrication of Protein–Peptide Mucoadhesive Carriers
2.3. Characterization of the Protein-Peptides Mucoadhesive Formulation
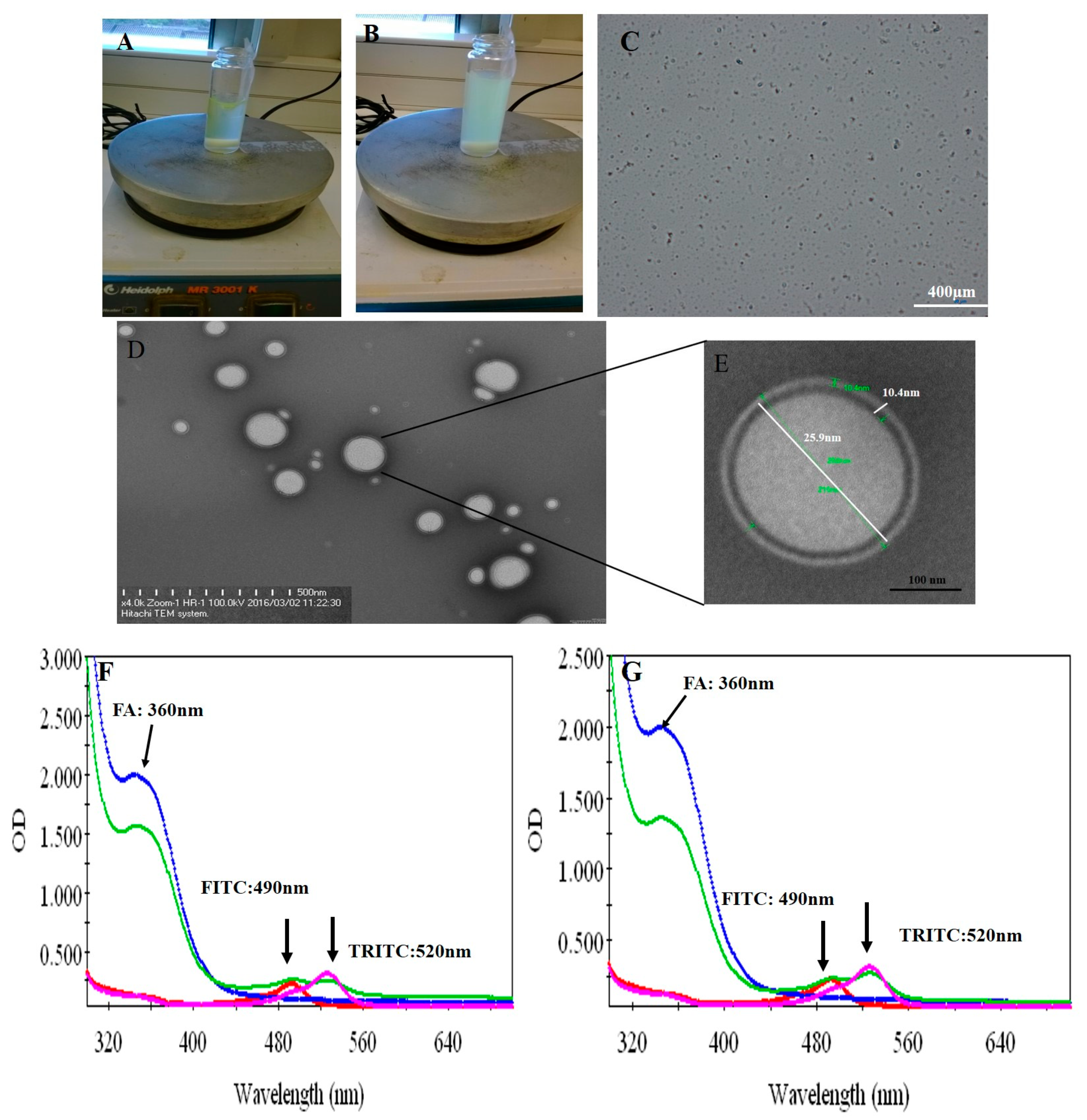
2.3.1. Transmission Electron Microscopy (TEM)
2.3.2. UV–Vis Spectroscopy
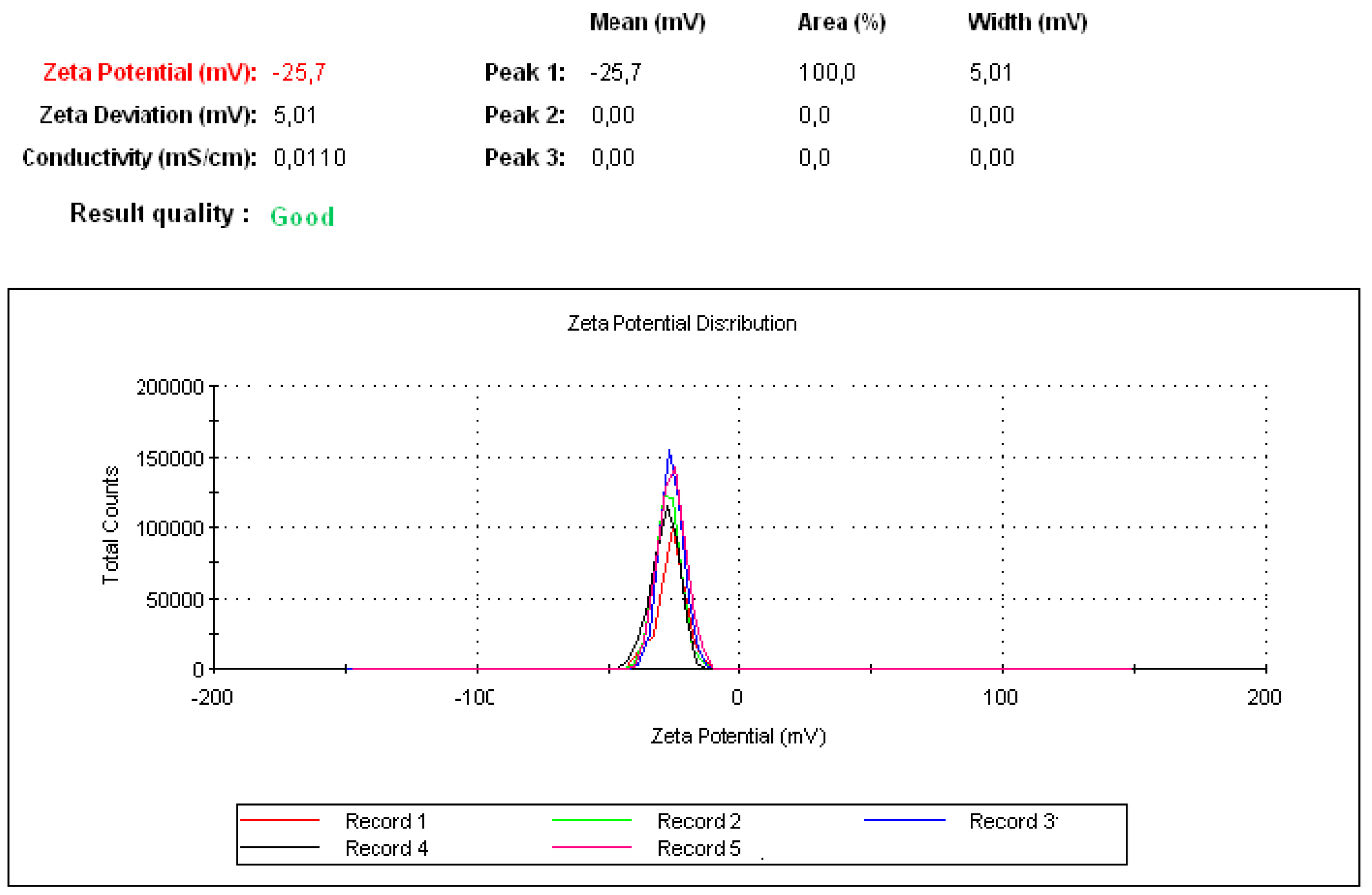
2.3.3. Zeta Potential
2.3.4. ImageJ Analysis and Calculation of the Surface Roughness
2.4. Cell Culture
2.4.1. Cellular Uptake
2.4.2. Proliferation Assay
2.4.3. Western Blot Analysis
2.5. Ethical Approval
3. Results
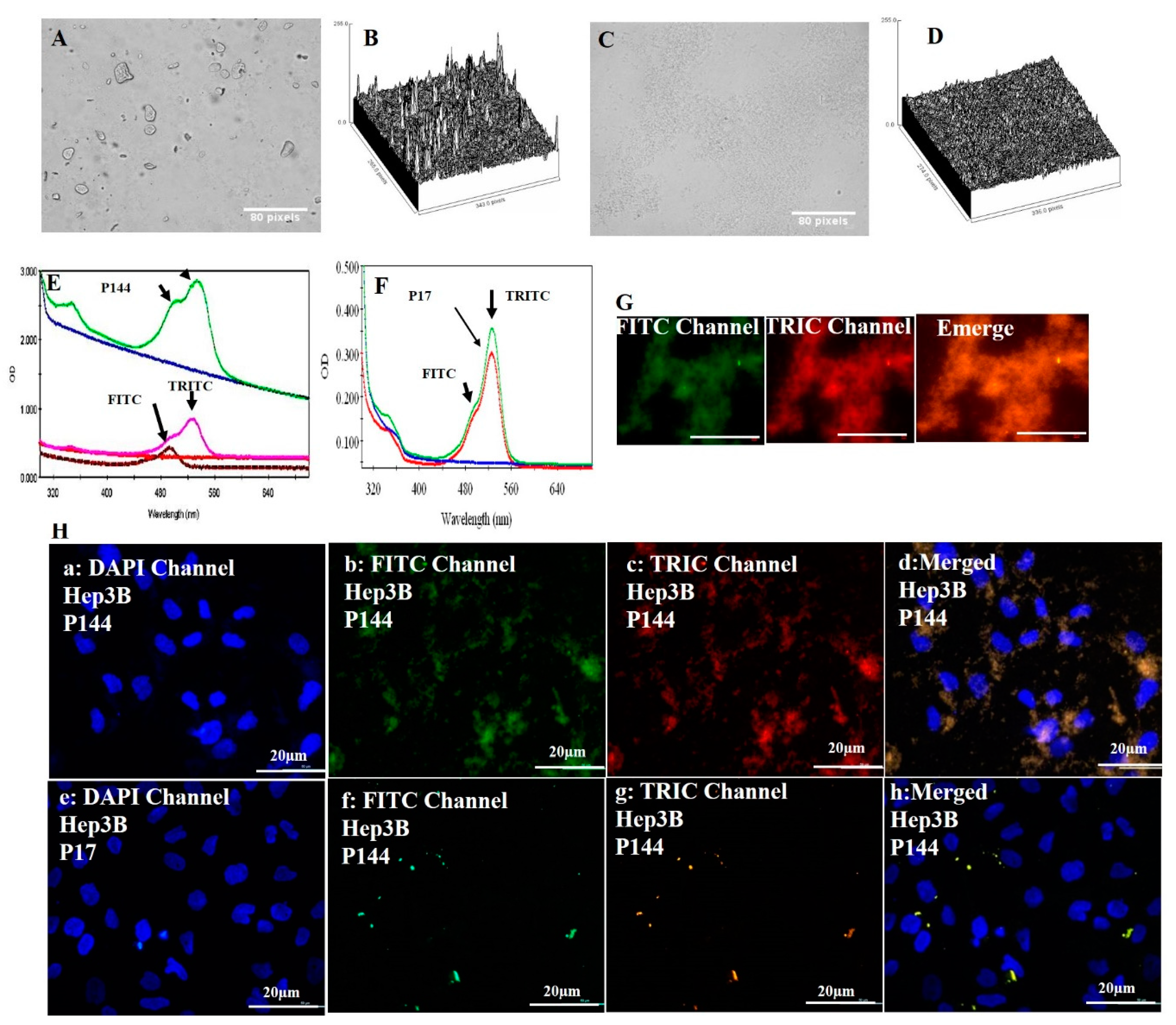
3.1. Physical Properties of Peptide P17 and P144
3.2. Fabrication of Protein–Peptide Mucoadhesive Complex
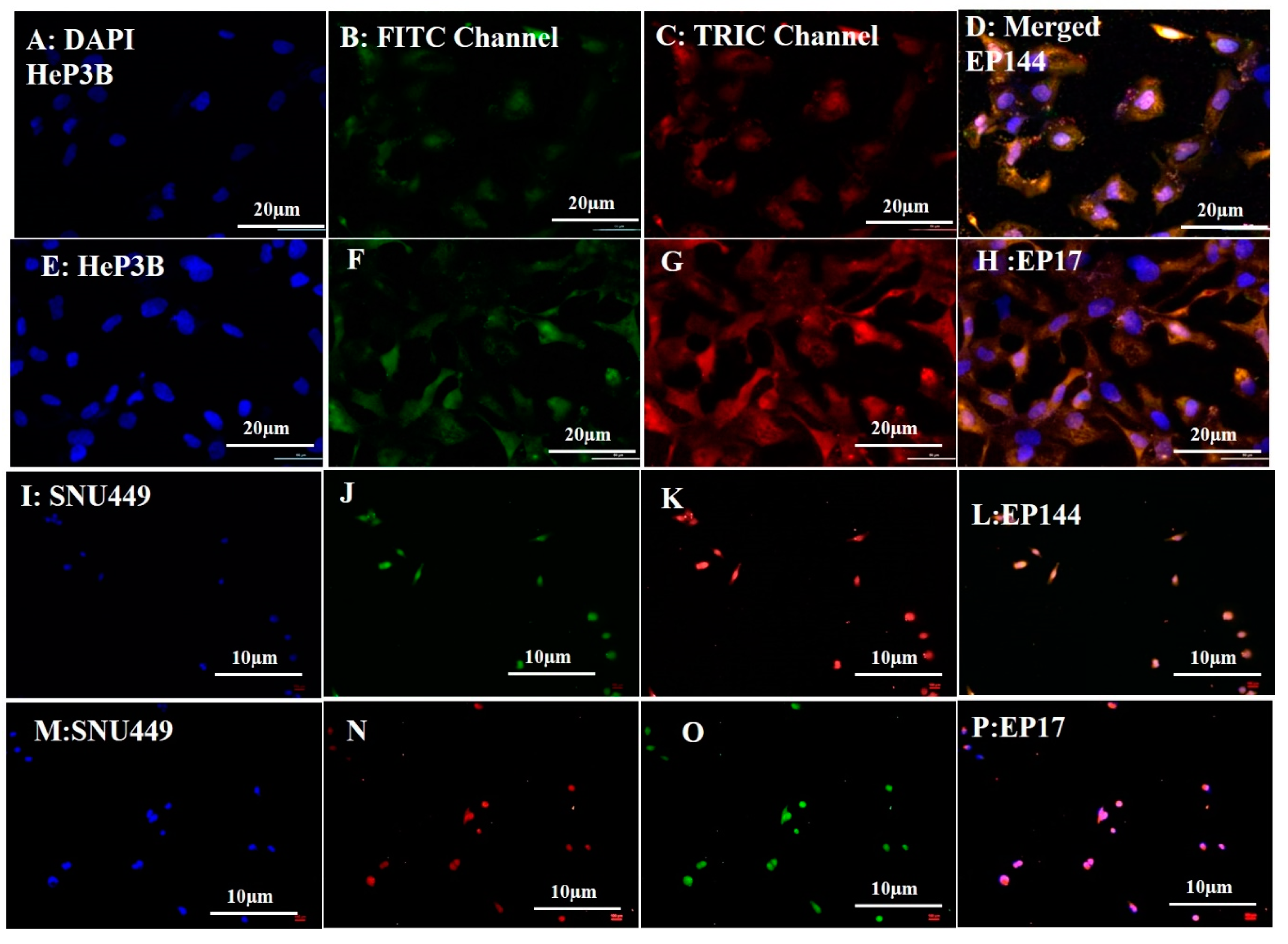
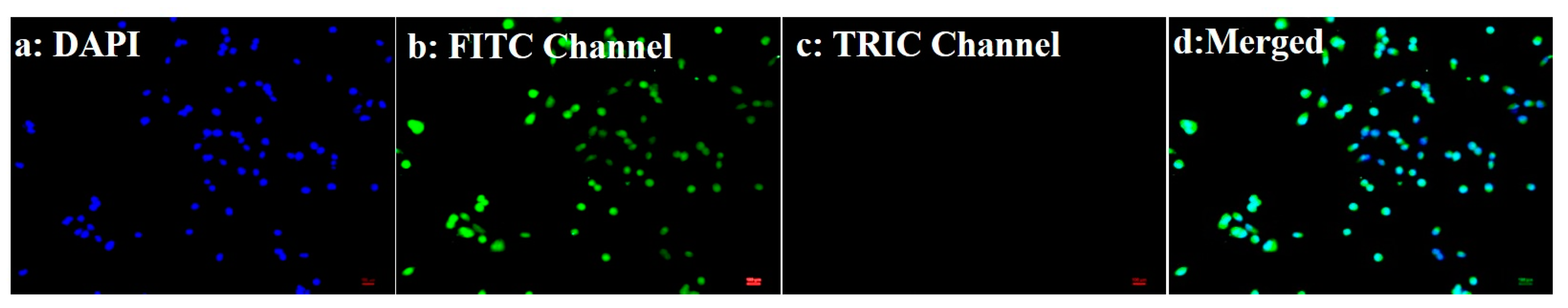
3.3. Biological Experiments: Cellular Uptake
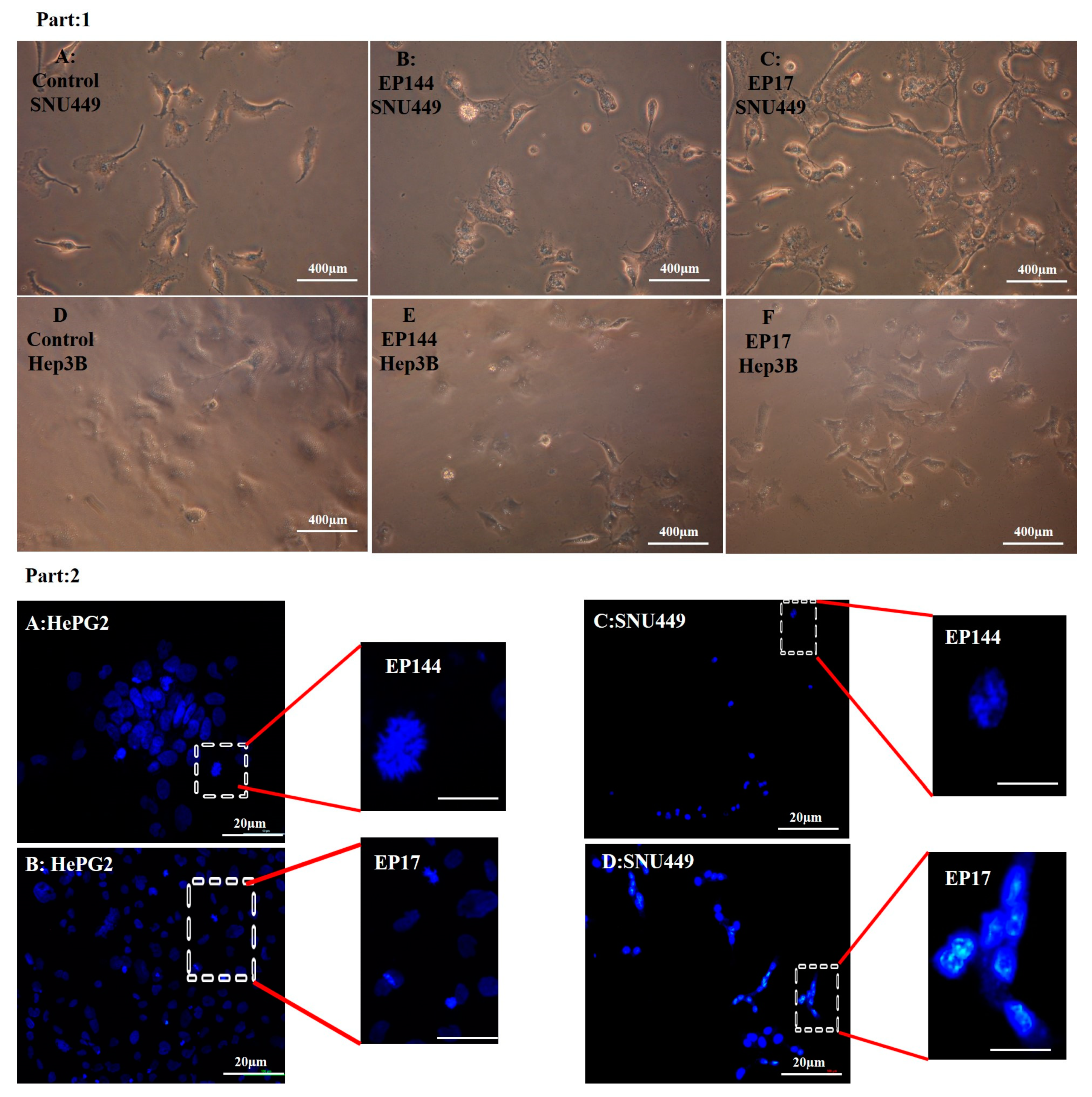
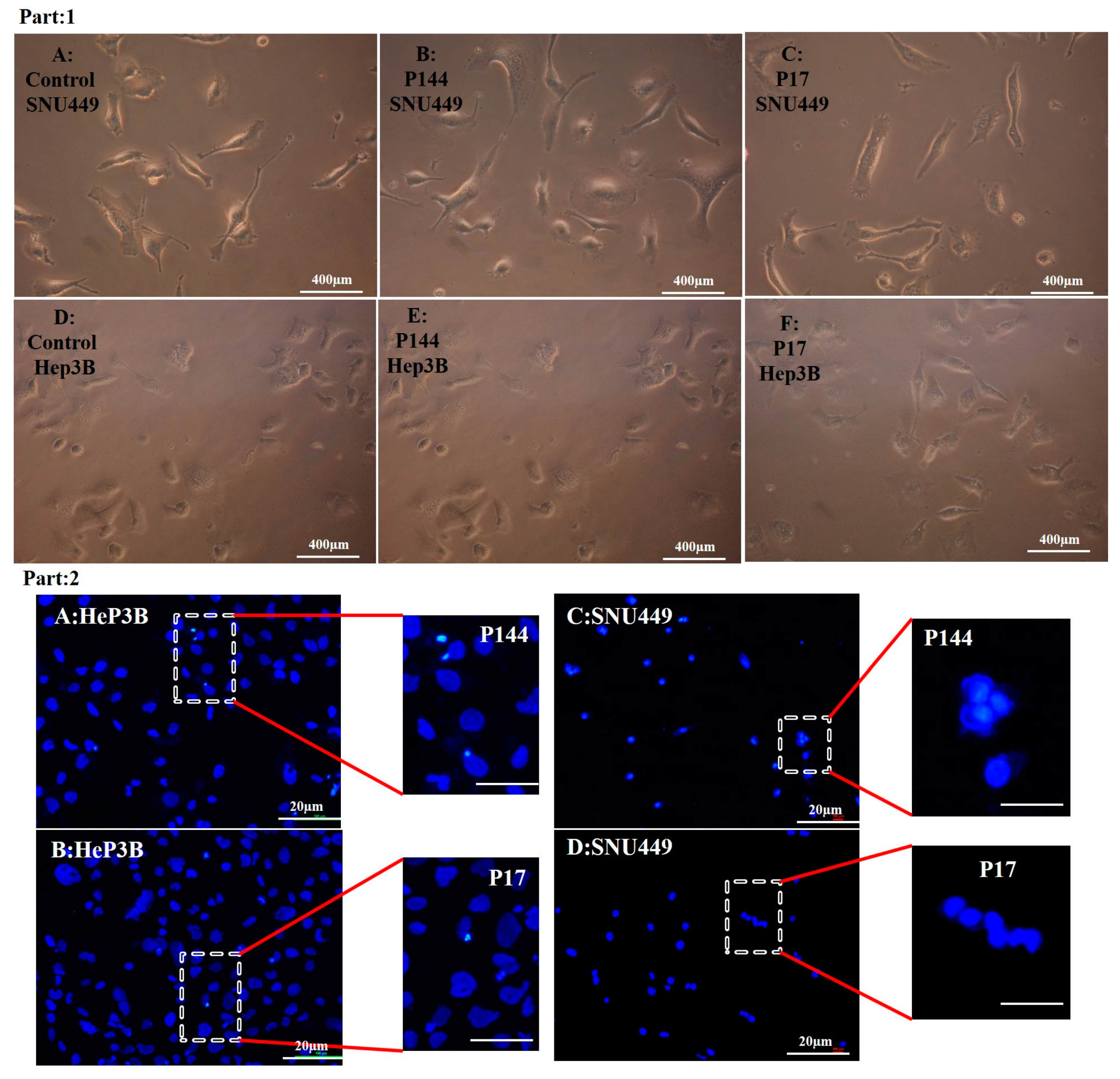
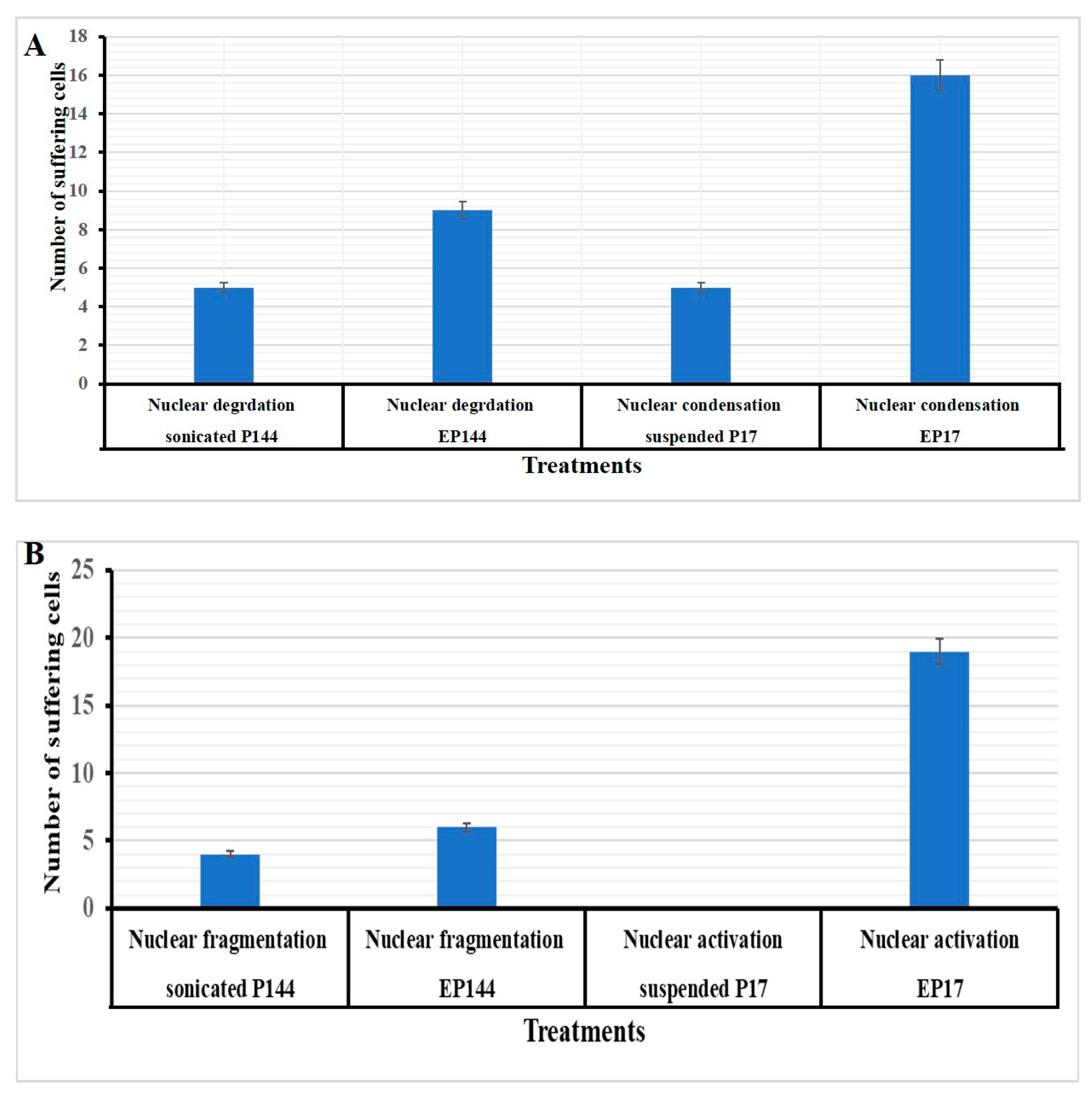
3.4. Determination of Nuclear Morphology by Using DAPI Staining
3.5. Antiproliferative Effect of Encapsulated Peptides
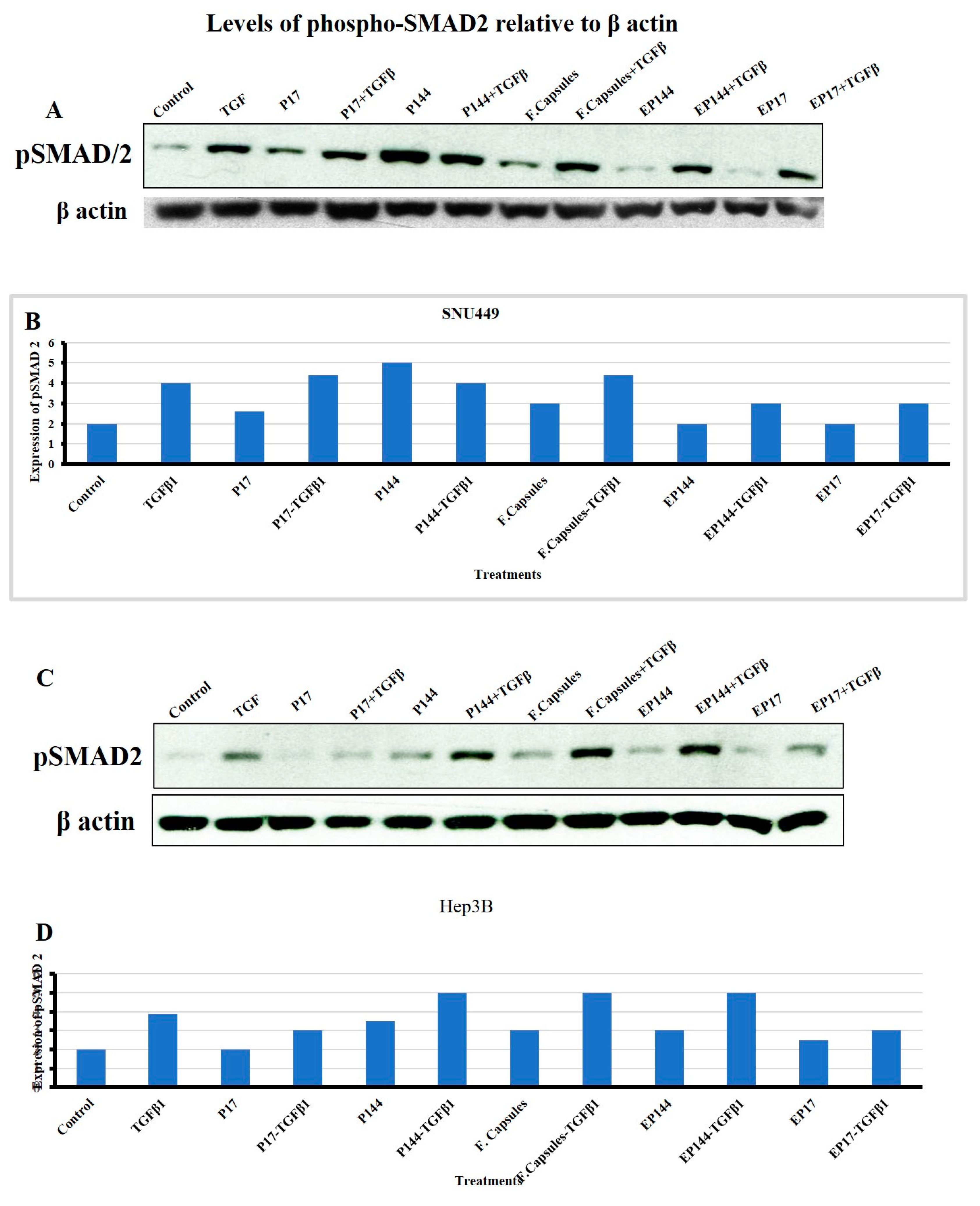
3.6. Effects of P144 and P17 on SMAD2 Phosphorylation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- David, C.J.; Massague, J. Contextual determinants of TGF-ß action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 2018, 19, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K.; Miyazono, K. Regulation of TGF- ß family signaling by inhibitory SMADs. Cold Spring Harb. Perspect. Biol. 2017, 9, a022095. [Google Scholar]
- Hanafy, N.A.; El Kemary, M. TGFβ1 as a Good and Bad Biological Molecule: Structure and Function. Biomed. J. Sci. Tech. Res. BJSTR 2019. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar]
- Zhang, Y.; Alexander, P.B.; Wang, X.F. TGF- ß family signaling in the control of cell proliferation and survival. Cold Spring Harb. Perspect. Biol. 2017, 9, a022145. [Google Scholar] [CrossRef]
- ten Dijke, P.; Arthur, H.M. Extracellular control of TGF-ß beta signalling in vascular development and disease. Nat. Rev. Mol. Cell Biol. 2007, 8, 857–869. [Google Scholar] [CrossRef]
- Heldin, C.H.; Moustakas, A. Signaling receptors for TGF-ß family members. Cold Spring Harb. Perspect. Biol. 2016, 8, a022053. [Google Scholar] [CrossRef]
- Zarranz-Ventura, J.; Fernández-Robredo, P.; Recalde, S.; Salinas-Alamán, A.; Borrás-Cuesta, F.; Dotor, J.; García-Layana, A. Transforming growth factor-beta inhibition reduces progression of early choroidal neovascularization lesions in rats: P17 and P144 peptides. PLoS ONE 2013, 31, e65434. [Google Scholar] [CrossRef]
- Murillo-Cuesta, S.; Rodríguez-de la Rosa, L.; Contreras, J.; Celaya, A.M.; Camarero, G.; Rivera, T.; Varela-Nieto, I. Transforming growth factor β1 inhibition protects from noise-induced hearing loss. Front. Aging Neurosci. 2015, 7, 32. [Google Scholar]
- Ezquerro, I.J.; Lasarte, J.J.; Dotor, J. A synthetic peptide from transforming growth factor β type III receptor inhibits liver fibrogenesis in rats with carbon tetrachloride liver injury. Cytokine 2003, 22, 12–20. [Google Scholar] [CrossRef]
- Dotor, J.; Lopez-Vazquez, A.B.; Lasarte, J.J.; Sarobe, P.; Garcia-Granero, M.; Riezu-Boj, J.I.; Martinez, A.; Feijoo, E.; Lopez-Sagaseta, J.; Hermida, J.; et al. Identification of peptide inhibitors of transforming growth factor beta 1 using a phage displayed peptide library. Cytokine 2007, 39, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Gil-Guerrero, L.; Dotor, J.; Huibregtse, I.J.; Casares, N.; Lopez-Vazquez, A.B.; Rudilla, F.; Riezu-Boj, J.I.; Lopez-Sagaseta, J.; Hermida, J.; Van Deventer, S.; et al. In vitro and in vivo down-regulation of regulatory T- cell activity with a peptide inhibitor of TGF-beta1. J. Immunol. 2008, 181, 126–135. [Google Scholar]
- Santiago, B.; Gutierrez-Canas, I.; Dotor, J.; Palao, G.; Lasarte, J.J.; Ruiz, J.; Prieto, J.; Borras-Cuesta, F.; Pablos, J.L. Topical application of a peptide inhibitor of transforming growth factor-beta1 ameliorates bleomycin-induced skin fibrosis. J. Investig. Dermatol. 2005, 125, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Kratz, F.; Müller-Driver, R.; Hofmann, I.; Drevs, J.; Unger, C. A novel macromolecular prodrug concept exploiting endogenous serum albumin as a drug carrier for cancer chemotherapy. J. Med. Chem. 2000, 43, 1253–1256. [Google Scholar] [CrossRef]
- Llopiz, D.; Dotor, J.; Zabaleta, A.; Lasarte, J.J.; Prieto, J.; Borras-Cuesta, F.; Sarobe, P. Combined immunization with adjuvant molecules poly(I:C) and anti-CD40 plus a tumour antigen has potent prophylactic and therapeutic antitumor effects. Cancer Immunol. Immunother. 2008, 57, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Recalde, S.; Zarranz-Ventura, J.; Fernández-Robredo, P.; García-Gómez, P.J.; Salinas-Alamán, A.; Borrás-Cuesta, F.; Dotor, J.; García-Layana, A. Transforming growth factor-β inhibition decreases diode laser-induced choroidal neovascularization development in rats: P17 and P144 peptides. Investig. Ophthalmol. Vis. Sci. 2011, 52, 7090–7097. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.S.; Dotor, J.; Hontanilla, B. Effect of P144® (Anti-TGF-β) in an "In Vivo" Human Hypertrophic Scar Model in Nude Mice. PLoS ONE 2015, 10, e0144489. [Google Scholar] [CrossRef]
- Da Violante, G.; Zerrouk, N.; Richard, I.; Provot, G.; Chaumeil, J.C.; Arnaud, P. Evaluation of the cytotoxicity effect of dimethyl sulfoxide (DMSO) on Caco2/TC7 colon tumor cell cultures. Biol. Pharm. Bull. 2002, 25, 1600–1603. [Google Scholar] [CrossRef]
- Hanafy, N.A.N. Encapsulation of cancer signalling pathway inhibitors as a protective way for healthy cells. Commentary. Med. Res. Innov. 2018. [Google Scholar] [CrossRef]
- Soukupova, J.; Malfettone, A.; Hyroššová, P.; Hernández-Alvarez, M.I.; Peñuelas-Haro, I.; Bertran, E.; Junza, A.; Capellades, J.; Giannelli, G.; Yanes, O.; et al. Role of the Transforming Growth Factor-β in regulating hepatocellular carcinoma oxidative metabolism. Sci. Rep. 2017, 7, 12486. [Google Scholar] [CrossRef]
- American Type Culture Collection. Available online: www.lgcstandards-atcc.org (accessed on 5 November 2018).
- Zhou, Q.Y.; Tu, C.Y.; Shao, C.X.; Wang, W.K.; Zhu, J.D.; Cai, Y.; Mao, J.Y.; Chen, W. GC7 blocks epithelial-mesenchymal transition and reverses hypoxia-induced chemotherapy resistance in hepatocellular carcinoma cells. Am. J. Transl. Res. 2017, 9, 2608–2617. [Google Scholar] [PubMed]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Reduction diameter of CaCO3 crystals by using poly acrylic acid might improve cellular uptake of encapsulated curcumin in breast cancer. J. Nanomed. Res. 2018, 7, 235–239. [Google Scholar]
- Hanafy, N.A.N.; Ferraro, M.M.; Gaballo, A.; Dini, L.; Tasco, V.; Nobile, C.; De Giorgi, M.L.; Carallo, S.; Rinaldi, R.; Leporatti, S. Fabrication and Characterization of ALK1fc-Loaded Fluoro-Magnetic Nanoparticles Rods for Inhibiting TGF β1 in HCC. RSC Adv. 2016, 6, 48834–48842. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Mucoadhesive curcumin crosslinked carboxy methyl cellulose might increase inhibitory efficiency for liver cancer treatment. Mater. Sci. Eng. C 2020, in press. [Google Scholar]
- Safer, A.M.; Sen, A.; Hanafy, N.A.; Mousa, S.A. Quantification of the healing effect in hepatic fibrosis induced by Chitosan Nano-encapsulated Green Tea in Rat Model Studied—At Ultrastructural level. J. Nanosci. Nanotechnol. 2015, 15, 1–7. [Google Scholar]
- Hanafy, N.A.; Nobile, C.; De Giorgi, M.L.; Ran, B.; Cao, Y.; Giannelli, G.; Leporatti, S. LY2157299-Loaded Carriers Inhibiting Wound Healing in Hepatocellular Carcinoma. J. Biotech. 2014, 185S, S18–S36. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; De Giorgi, M.L.; Nobile, C.; Giannelli, G.; Quarta, A.; Leporatti, S. P0253: Inhibition of glycolysis by using nano lipid bromopyruvic chitosan carrier is a promising tool to prevent HCC invasiveness. J. Hepatol. 2015, 62, S401. [Google Scholar] [CrossRef]
- Chiba, K.; Kawakami, K.; Tohyama, K. Simultaneous evaluation of cell viability by neutral red, MTT and crystal violet staining assays of the same cells. Toxicol. Vitro 1998, 12, 251–258. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; Quarta, A.; Di Corato, R.; Dini, L.; Nobile, C.; De Giorgi, M.L.; Tasco, V.; Carallo, S.; Cascione, M.; Rinaldi, R.; et al. Encapsulation of SHT-DNA, SIRNA and polypeptide -17 inside Hybrid polymeric nano-protein folic acid (HPNP-FA) carriers as targeted TGF beta inhibitor for Hepatocellular carcinoma. J. Hepatol. 2016, 64, S425–S630. [Google Scholar] [CrossRef]
- Jang, C.W.; Chen, C.H.; Chen, C.C.; Chen, J.; Su, Y.H.; Chen, R.H. TGF-β induces apoptosis through Smad-mediated expression of DAP-kinase. Nat. Cell Biology 2002, 4, 51–58. [Google Scholar] [CrossRef]
- Beija, M.; Afonso, C.A.M.; Martinho, J.M.G. Synthesis and applications of Rhodamine derivatives as fluorescent probes. Chem. Soc. Rev. 2009, 38, 2410–2433. [Google Scholar] [CrossRef] [PubMed]
- Parodi, A.; Miao, J.; Soond, S.M.; Rudzińska, M.R.; Zamyatnin, A.A. Albumin Nanovectors in Cancer Therapy and Imaging. Biomolecules 2019, 9, 218. [Google Scholar] [CrossRef] [PubMed]
- Scheicher, B.; Lorenzer, C.; Gegenbauer, K.; Partlic, J.; Andreae, F.; Kirsch, A.H.; Rosenkranz, A.R.; Werzer, O.; Zimmer, A. Manufacturing of a Secretoneurin Drug Delivery System with Self-Assembled Protamine Nanoparticles by Titration. PLoS ONE 2016, 11, e0164149. [Google Scholar] [CrossRef] [PubMed]
- Piston, D.W.; Kremers, G.J. Fluorescent protein FRET: The good, the bad and the ugly. Trends Biochem. Sci. 2007, 32, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Teale, F.W.J.; Weber, G. Ultraviolet fluorescence of the aromatic amino acids. Biochem. J. 1957, 65, 476–482. [Google Scholar] [CrossRef]
- Junghans, M.; Kreuter, J.; Zimmer, A. Antisense delivery using protamine-oligonucleotide particles. Nucleic Acids Res. 2000, 28, e45. [Google Scholar] [CrossRef]
- Kubista, M.; Aakerman, B.; Norden, B. Characterization of interaction between DNA and 4′, 6-diamidino-2-phenylindole by optical spectroscopy. Biochemistry 1987, 26, 4545–4553. [Google Scholar] [CrossRef]
- Barcellona, M.L.; Favilla, R.; Von Berger, J.; Avitabile, M.; Ragusa, N.; Masotti, L. DNA-4′-6-diamidine-2-phenylindole interactions: A comparative study employing fluorescence and ultraviolet spectroscopy. Arch. Biochem. Biophys. 1986, 250, 48–53. [Google Scholar] [CrossRef]
- Kntayya, S.B.; Ibrahim, D.M.; Ain, M.N.; Iori, R.; Ioannides, C.; Abdull Razis, A.F. Induction of Apoptosis and Cytotoxicity by Isothiocyanate Sulforaphene in Human Hepatocarcinoma HepG2 Cells. Nutrients 2018, 10, 718. [Google Scholar] [CrossRef]
- Estandarte, A.K.; Botchway, S.; Lynch, C.; Yusuf, M.; Robinson, I. The use of DAPI fluorescence lifetime imaging for investigating chromatin condensation in human chromosomes. Sci. Rep. 2016, 16, 31417. [Google Scholar] [CrossRef]
- Gallo-Oller, G.; Vollmann-Zwerenz, A.; Melendez, B.; Rey, J.; Hau, P.; Dotor, J.; Castresana, J. P144, a Transforming Growth Factor beta inhibitor peptide, generates antitumoral effects and modifies SMAD7 and SKI levels in human glioblastoma cell lines. Cancer Lett. 2016, 381, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Llopiz, D.; Dotor, J.; Casares, N.; Bezunartea, J.; Díaz-Valdés, N.; Ruiz, M.; Aranda, F.; Berraondo, P.; Prieto, J.; Lasarte, J.J.; et al. Peptide inhibitors of transforming growth factor-beta enhance the efficacy of antitumor immunotherapy. Int. J. Cancer 2009, 125, 2614–2623. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Pan, Y.; Liao, D.; Tang, J.; Yao, D. Peptide 17, an inhibitor of YAP/TEAD4 pathway, mitigates lung cancer malignancy. Trop. J. Pharm. Res. 2018, 17, 1256. [Google Scholar] [CrossRef]
- Upadhyay, A.; Moss-Taylor, L.; Kim, M.J.; Ghosh, A.C.; O’Connor, M.B. TGF-β family signaling in drosophila. Cold Spring Harb. Perspect. Biol. 2017, 9, a022152. [Google Scholar] [CrossRef]
- Savage-Dunn, C.; Padgett, R.W. The TGFβ family in caenorhabditis elegans. Cold Spring Harb. Perspect. Biol. 2017, 9, a022178. [Google Scholar] [CrossRef]
- Hao, Y.; Baker, D.; ten Dijke, P. TGFβ-Mediated Epithelial-Mesenchymal Transition and Cancer Metastasis. Int. J. Mol. Sci. 2019, 20, 2767. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; Quarta, A.; Di Corato, R.; Dini, L.; Nobile, C.; Tasco, V.; Carallo, S.; Cascione, M.; Malfettone, A.; Soukupova, J.; et al. Hybrid polymeric-protein nano-carriers (HPPNC) for targeted delivery of TGFβ inhibitors to hepatocellular carcinoma cells. J. Mater. Sci. Mater. Med. 2017, 28, 120. [Google Scholar] [CrossRef]
- Hanafy, N.A.; De Giorgi, M.L.; Nobile, C.; Rinaldi, R.; Leporatti, S. Control of Colloidal CaCO3 suspension by using biodegradable polymers during fabrication. Beni-Suef Univ. J. Basic Appl. Sci. 2015, 4, 60–70. [Google Scholar]
- Chaganti, L.K.; Venkatakrishnan, N.; Bose, K. An efficient method for FITC labelling of proteins using tandem affinity purification. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef]
- Takikawa, M.; Nakamura, S.; Ishihara, M.; Takabayashi, Y.; Fujita, M.; Hattori, H. Improved angiogenesis and healing in crush syndrome by fibroblast growth factor-2 containing low-molecular-weight heparin (Fragmin)/protamine nanoparticles. J. Surg. Res. 2015, 196, 247–257. [Google Scholar] [CrossRef]
- Mayer, G.; Vogel, V.; Weyermann, J.; Lochmann, D.; van den Broek, J.A.; Tziatzios, C. Oligonucleotideprotamine- albumin nanoparticles: Protamine sulfate causes drastic size reduction. J. Control. Release 2005, 106, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Malfettone, A.; Soukupova, J.; Bertran, E.; Crosas-Molist, E.; Lastra, R.; Fernando, J.; Koudelkova, P.; Rani, B.; Fabra, Á; Serrano, T.; et al. Transforming growth factor-β-induced plasticity causes a migratory stemness phenotype in hepatocellular carcinoma. Cancer Lett. 2017, 392, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Bertran, E.; Crosas-Molist, E.; Sancho, P.; Caja, L.; Lopez-Luque, J.; Navarro, E.; Egea, G.; Lastra, R.; Serrano, T.; Ramos, E.; et al. Overactivation of the TGF-β pathway confers a mesenchymal-like phenotype and CXCR4-dependent migratory properties to liver tumor cells. Hepatology 2013, 58, 2032–2044. [Google Scholar] [CrossRef] [PubMed]
- Hata, A.; Chen, Y.G. TGF-β signaling from receptors to smads. Cold Spring Harb. Perspect. Biol. 2016, 8, a022061. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Long, J.; Liu, Z.; Tao, W.; Wang, D. Identification and Evolution of TGF-β Signaling Pathway Members in Twenty-Four Animal Species and Expression in Tilapia. Int. J. Mol. Sci. 2018, 19, 1154. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, N.A.; Dini, L.; Citti, C.; Cannazza, G.; Leporatti, S. Inhibition of Glycolysis by Using a Micro/Nano-Lipid Bromopyruvic Chitosan Carrier as a Promising Tool to Improve Treatment of Hepatocellular Carcinoma. Nanomaterials 2018, 8, 34. [Google Scholar] [CrossRef]
- Hanafy, N.A.; De Giorgi, M.L.; Nobile, C.; Cascione, M.; Rinaldi, R.; Leporatti, S. CaCO3 rods as chitosan polygalacturonic acid carriers for brompyruvic acid delivery. Sci. Adv. Mater. 2016, 8, 514–523. [Google Scholar] [CrossRef]
- Hanafy, N.A.N. The growth of hepatocellular carcinoma can be inhibited by encapsulation of TGF ß1 antagonists. SL Pharmacol. Toxicol. 2018, 1, 112. [Google Scholar]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Understanding TGF β1 signalling pathway is well strategy to use its encapsulated antagonist as nano therapeutic molecules. Transl. Sci. 2018, 2018. [Google Scholar]
- Hanafy, N.A.N. Glycolysis is a promising target for encapsulation nano-therapeutic molecules against cancer cells. Commentary. Integr. Cancer Sci. Therap. 2017, 2017. [Google Scholar]
- Hanafy, N.A.N. Development and Production of Multifunctional Bio-Nano-Engineered Drug Delivery Systems Loaded by TGF Inhibitors for Delivering into Hepatocellular Carcinoma Cells. Ph.D. Thesis, Salento University Italy, Lecce, Italy, 2017. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanafy, N.A.N.; Fabregat, I.; Leporatti, S.; El Kemary, M. Encapsulating TGF-β1 Inhibitory Peptides P17 and P144 as a Promising Strategy to Facilitate Their Dissolution and to Improve Their Functionalization. Pharmaceutics 2020, 12, 421. https://doi.org/10.3390/pharmaceutics12050421
Hanafy NAN, Fabregat I, Leporatti S, El Kemary M. Encapsulating TGF-β1 Inhibitory Peptides P17 and P144 as a Promising Strategy to Facilitate Their Dissolution and to Improve Their Functionalization. Pharmaceutics. 2020; 12(5):421. https://doi.org/10.3390/pharmaceutics12050421
Chicago/Turabian StyleHanafy, Nemany A. N., Isabel Fabregat, Stefano Leporatti, and Maged El Kemary. 2020. "Encapsulating TGF-β1 Inhibitory Peptides P17 and P144 as a Promising Strategy to Facilitate Their Dissolution and to Improve Their Functionalization" Pharmaceutics 12, no. 5: 421. https://doi.org/10.3390/pharmaceutics12050421
APA StyleHanafy, N. A. N., Fabregat, I., Leporatti, S., & El Kemary, M. (2020). Encapsulating TGF-β1 Inhibitory Peptides P17 and P144 as a Promising Strategy to Facilitate Their Dissolution and to Improve Their Functionalization. Pharmaceutics, 12(5), 421. https://doi.org/10.3390/pharmaceutics12050421







