Evaluations of Quality by Design (QbD) Elements Impact for Developing Niosomes as a Promising Topical Drug Delivery Platform
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Niosome Vesicle Preparation
2.3. HPLC Methods
2.4. Optimization of Drug Loaded Niosomes Using a Systematic Experimental Design
2.5. Niosome Vesicle Characterization
2.5.1. Organoleptic Properties
2.5.2. Drug Content (Assay Determination)
2.5.3. Drug Entrapment Efficiency of Niosome Vesicles
2.5.4. Niosomes Vesicle Size and Zeta Potential
3. Results and Discussion
3.1. Organoleptic Properties
3.2. Optimization of Niosomes by Design of Experiments (DoE)
3.3. Optimization of Critical Material Attributes
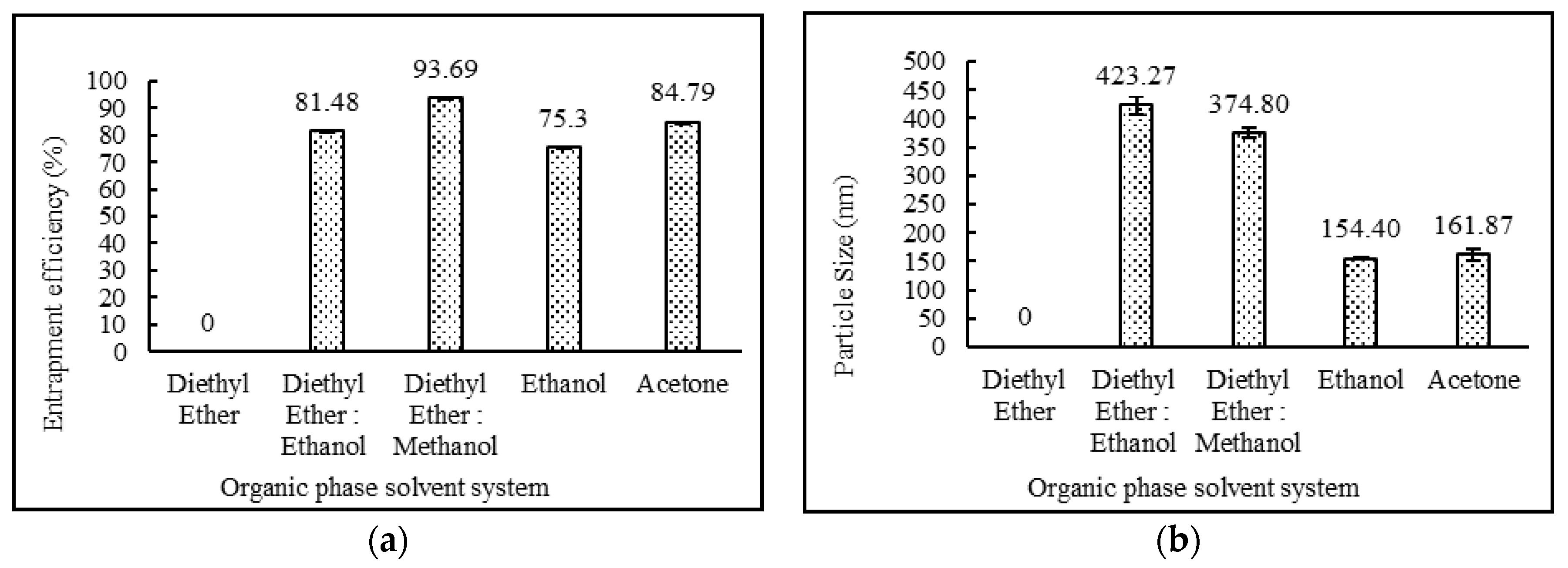
3.3.1. Effect of Organic Phase System
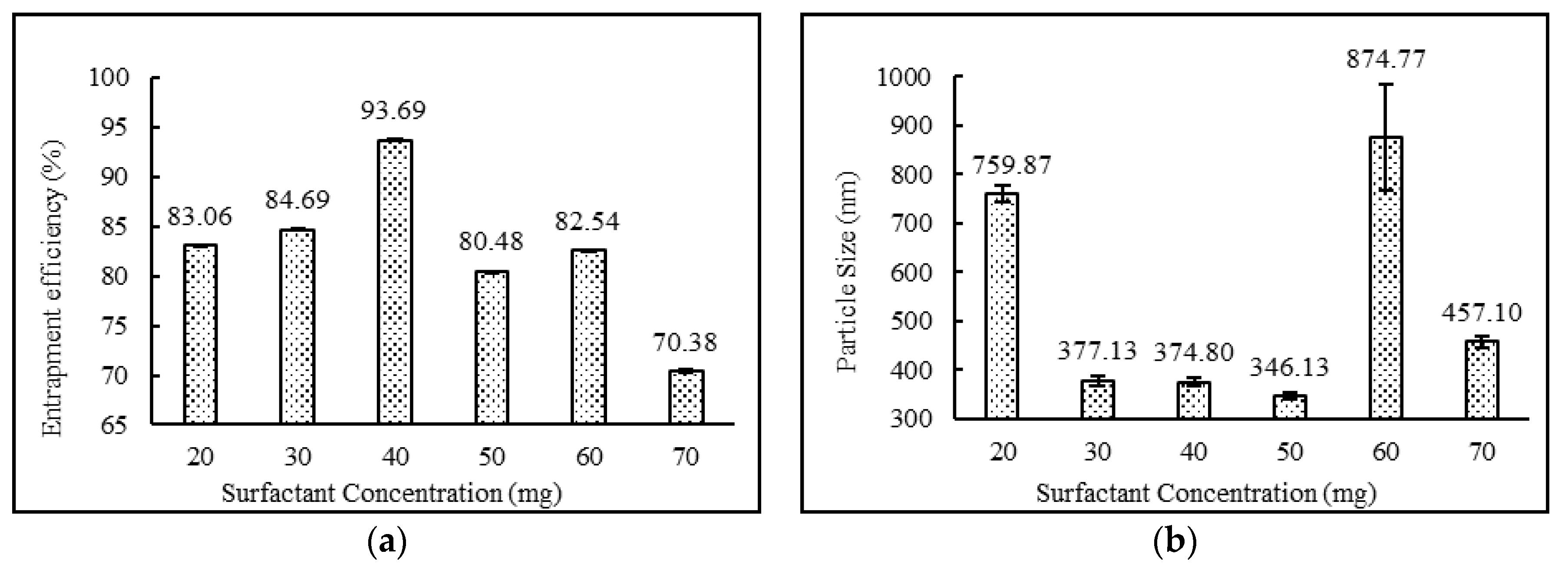
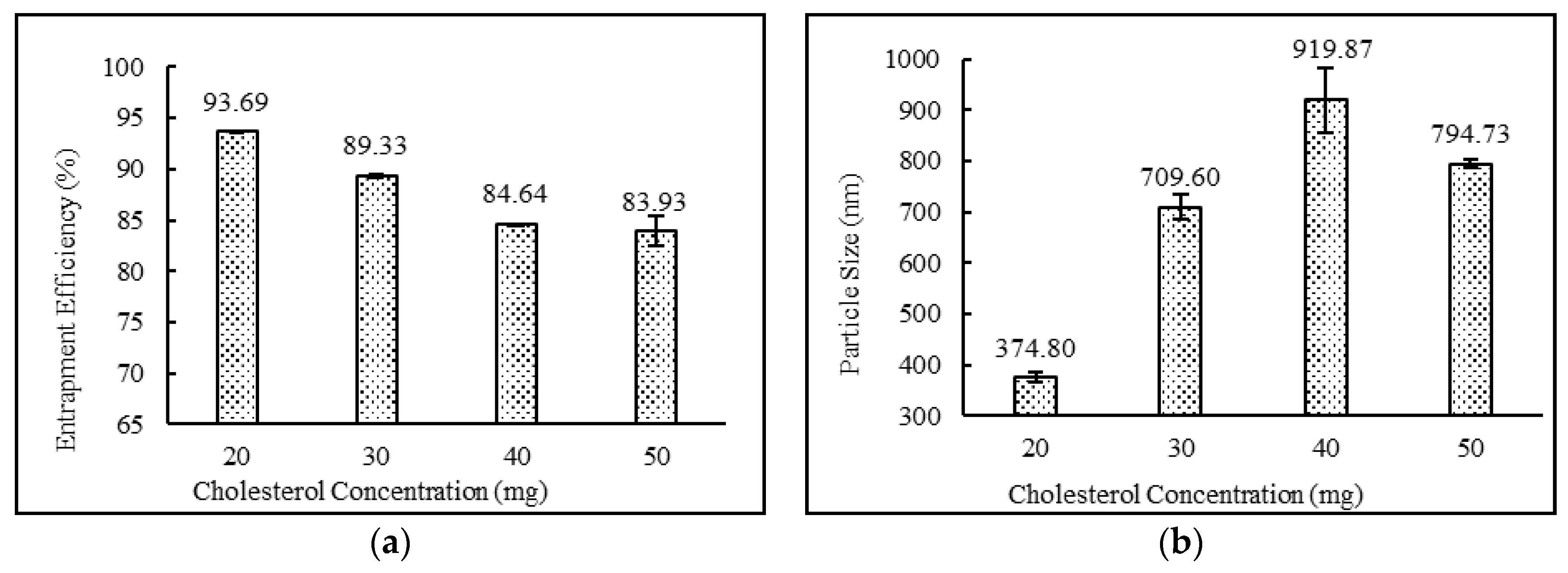
3.3.2. Effect of Drug, Surfactant and Cholesterol Concentrations
3.3.3. Influence of Electrostatic Charge
3.4. Optimization of Critical Process Parameters
3.4.1. Effect of Volume and Temperature
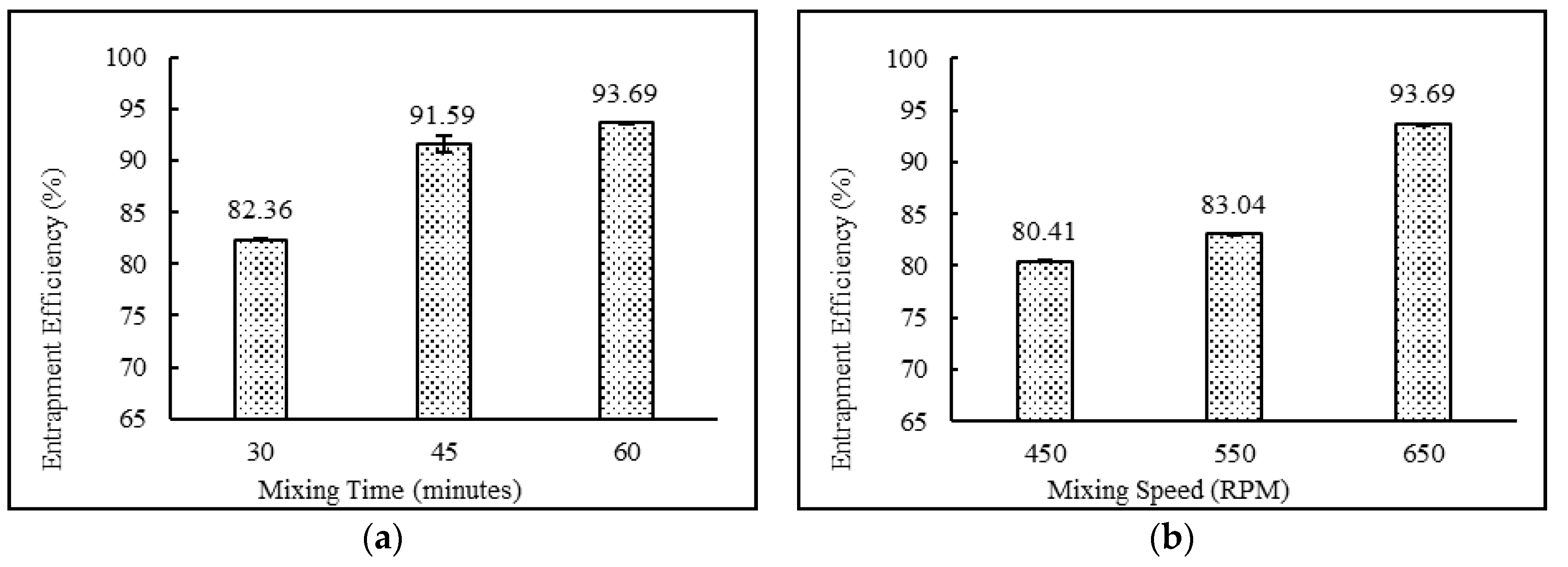
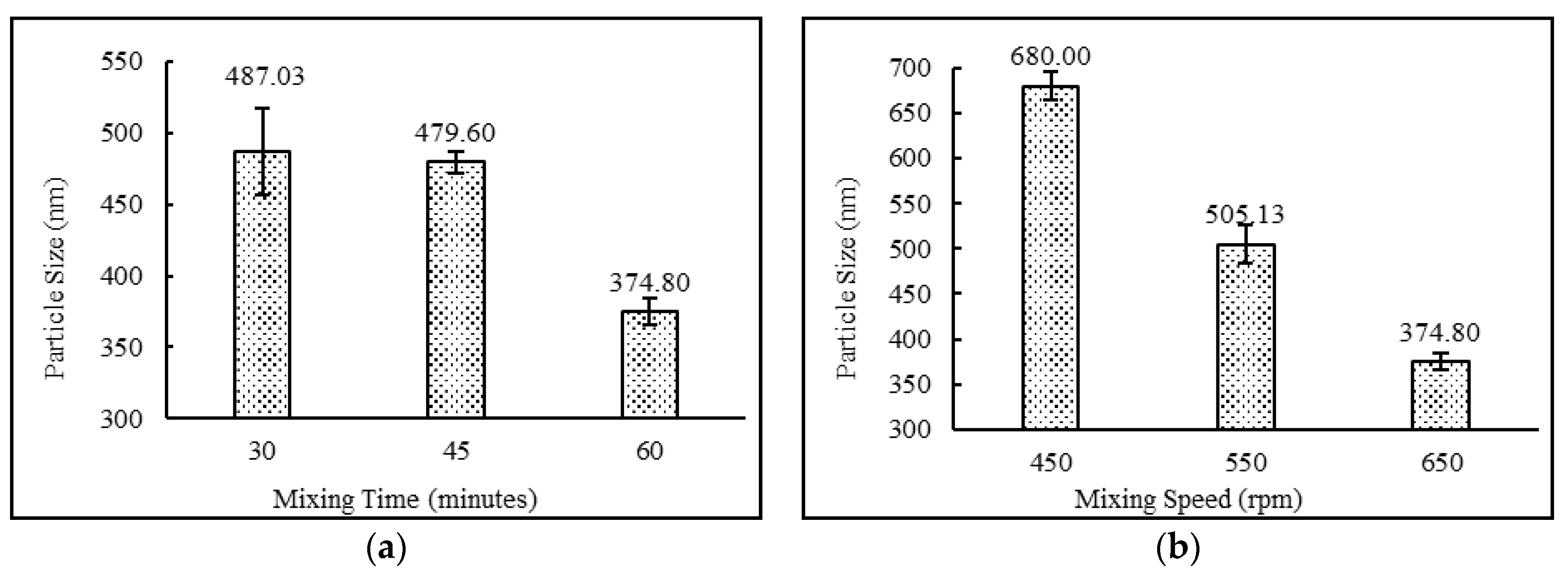
3.4.2. Effect of Mixing Time (minutes) and Speed (rpm)
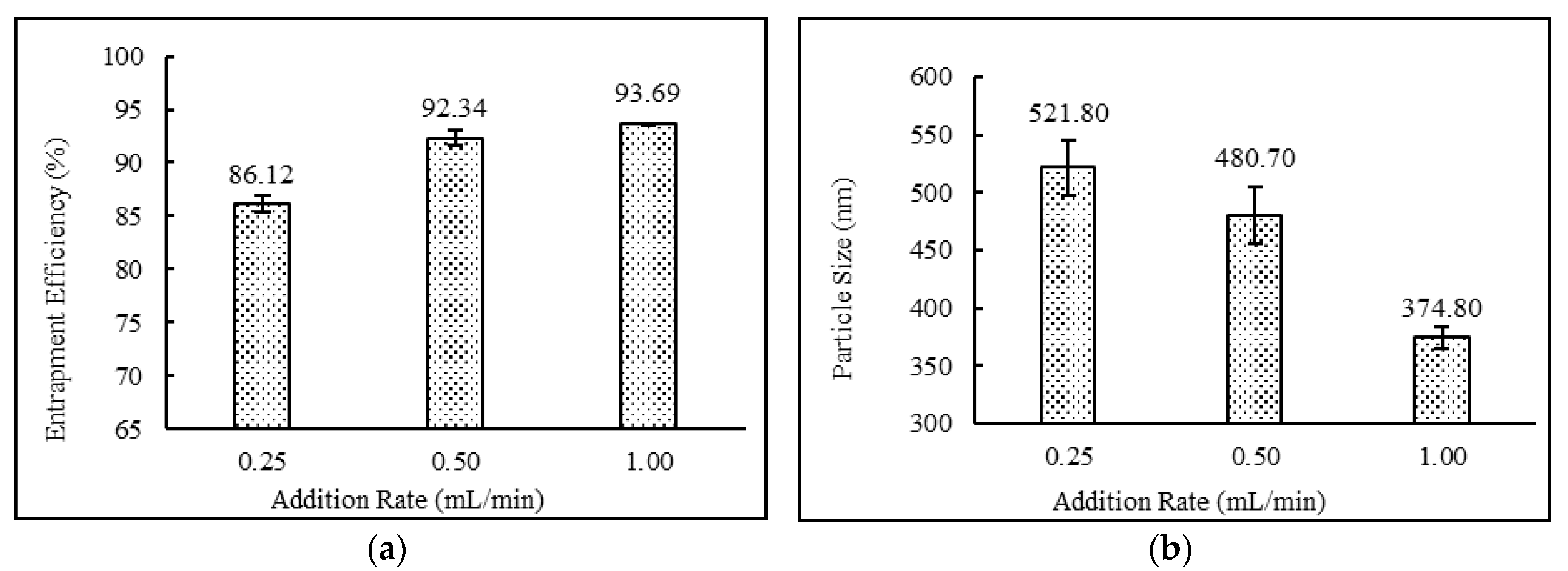
3.4.3. Effect of Addition Rate
3.5. Entrapment Efficiency (%)
3.6. Vesicle Sizes and Polydispersity Index
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moghassemi, S.; Hadjizadeh, A. Nano-Niosomes as nanoscale drug delivery systems: An illustrated review. J. Control. Release 2014, 185, 22–36. [Google Scholar]
- Mujoriya, R.; Bodla, R. Niosomes-Challenge in preparation for pharmaceutical scientist. Int. J. Appl. Pharm. 2011, 3, 11–15. [Google Scholar]
- Sezgin-Bayindir, Z.; Yuksel, N. Investigation of formulation variables and excipient interaction on the production of niosomes. AAPS PharmSciTech 2012, 13, 826–835. [Google Scholar]
- Shatalebi, M.A.; Mostafavi, S.A.; Moghaddas, A. Niosome as a drug carrier for topical delivery of N-acetyl glucosamine. Res. Pharm. Sci. 2010, 5, 107–117. [Google Scholar]
- Hamishehkar, H.; Rahimpour, Y.; Kouhsoltani, M. Niosomes as a propitious carrier for topical drug delivery. Expert Opin. Drug Deliv. 2013, 10, 261–272. [Google Scholar] [CrossRef]
- Manconi, M.; Valenti, D.; Sinico, C.; Lai, F.; Loy, G.; Fadda, A.M. Niosomes as carriers for tretinoin II. Influence of vesicular incorporation on tretinoin photostability. Int. J. Pharm. 2003, 260, 261–272. [Google Scholar] [CrossRef]
- Mahale, N.; Thakkar, P.; Mali, R.; Walunj, D.; Chaudhari, S. Niosomes: Novel sustained release nonionic stable vesicular systems—An overview. Adv. Colloid Interface Sci. 2012, 183, 46–54. [Google Scholar]
- Manosroi, A.; Wongtrakul, P.; Manosroi, J.; Sakai, H.; Sugawara, F.; Yuasa, M.; Abe, M. Characterization of vesicles prepared with various non-ionic surfactants mixed with cholesterol. Colloids Surf. B Biointerfaces 2003, 30, 129–138. [Google Scholar]
- Pardakhty, A.; Varshosaz, J.; Rouholamini, A. In Vitro study of polyoxyethylene alkyl ether niosomes for delivery of insulin. Int. J. Pharm. 2007, 328, 130–141. [Google Scholar]
- Varshosaz, J.; Pardakhty, A.; Hajhashemi, V.I.; Najafabadi, A.R. Development and physical characterization of sorbitan monoester niosomes for insulin oral delivery. Drug Deliv. 2003, 10, 251–262. [Google Scholar]
- Girigoswami, A.; Das, S.; De, S. Fluorescence and dynamic light scattering studies of niosomes-membrane mimetic systems. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2006, 64, 859–866. [Google Scholar] [CrossRef]
- Abdelkader, H.; Wu, Z.; Al-Kassas, R.; Alany, R.G. Niosomes and discomes for ocular delivery of naltrexone hydrochloride: Morphological, rheological, spreading properties and photo-protective effects. Int. J. Pharm. 2012, 433, 142–148. [Google Scholar]
- Agarwal, R.; Katare, O.; Vyas, S. Preparation and In Vitro evaluation of liposomal/niosomal delivery systems for antipsoriatic drug dithranol. Int. J. Pharm. 2001, 228, 43–52. [Google Scholar]
- Desai, T.R.; Finlay, W.H. Nebulization of niosomal all-trans-retinoic acid: An inexpensive alternative to conventional liposomes. Int. J. Pharm. 2002, 241, 311–317. [Google Scholar]
- Bayindir, Z.S.; Yuksel, N. Characterization of niosomes prepared with various nonionic surfactants for paclitaxel oral delivery. J. Pharm. Sci. 2010, 99, 2049–2060. [Google Scholar]
- Manosroi, A.; Jantrawut, P.; Manosroi, J. Anti-Inflammatory activity of gel containing novel elastic niosomes entrapped with diclofenac diethylammonium. Int. J. Pharm. 2008, 360, 156–163. [Google Scholar]
- Muzzalupo, R.; Tavano, L.; Cassano, R.; Trombino, S.; Ferrarelli, T.; Picci, N. A new approach for the evaluation of niosomes as effective transdermal drug delivery systems. Eur. J. Pharm. Biopharm. 2011, 79, 28–35. [Google Scholar]
- Rungphanichkul, N.; Nimmannit, U.; Muangsiri, W.; Rojsitthisak, P. Preparation of curcuminoid niosomes for enhancement of skin permeation. Pharm. Int. J. Pharm. Sci. 2011, 66, 570–575. [Google Scholar]
- Ag Seleci, D.; Seleci, M.; Walter, J.G.; Stahl, F.; Scheper, T. Niosomes as nanoparticular drug carriers: Fundamentals and recent applications. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
- Junyaprasert, V.B.; Singhsa, P.; Suksiriworapong, J.; Chantasart, D. Physicochemical properties and skin permeation of Span 60/Tween 60 niosomes of ellagic acid. Int. J. Pharm. 2012, 423, 303–311. [Google Scholar]
- Manca, M.L.; Manconi, M.; Nacher, A.; Carbone, C.; Valenti, D.; Maccioni, A.M.; Sinico, C.; Fadda, A.M. Development of novel diolein-niosomes for cutaneous delivery of tretinoin: Influence of formulation and In Vitro assessment. Int. J. Pharm. 2014, 477, 176–186. [Google Scholar]
- El-Menshawe, S.F.; Hussein, A.K. Formulation and evaluation of meloxicam niosomes as vesicular carriers for enhanced skin delivery. Pharm. Dev. Technol. 2013, 18, 779–786. [Google Scholar]
- Manconi, M.; Sinico, C.; Valenti, D.; Lai, F.; Fadda, A.M. Niosomes as carriers for tretinoin: III. A study into the In Vitro cutaneous delivery of vesicle-incorporated tretinoin. Int. J. Pharm. 2006, 311, 11–19. [Google Scholar]
- Mali, N.; Darandale, S.; Vavia, P. Niosomes as a vesicular carrier for topical administration of minoxidil: Formulation and in vitro assessment. Drug Deliv. Transl. Res. 2013, 3, 587–592. [Google Scholar]
- Goyal, G.; Garg, T.; Malik, B.; Chauhan, G.; Rath, G.; Goyal, A.K. Development and characterization of niosomal gel for topical delivery of benzoyl peroxide. Drug Deliv. 2015, 22, 1027–1042. [Google Scholar]
- Gupta, A.; Singh, S.; Kotla, N.G.; Webster, T.J. Formulation and evaluation of a topical niosomal gel containing a combination of benzoyl peroxide and tretinoin for antiacne activity. Int. J. Nanomed. 2015, 10, 171. [Google Scholar]
- Benipal, G. Design, development and evaluation of proniosomal gel of an antifungal drug-Ketoconazole. Int. J. Pharm. Sci. Rev. Res. 2015, 31, 265–272. [Google Scholar]
- Tamizharasi, S.; Dubey, A.; Rathi, V.; Rathi, J. Development and characterization of niosomal drug delivery of gliclazide. J. Young Pharm. 2009, 1. [Google Scholar] [CrossRef]
- Hebert, A.A.; Koo, J.; Fowler, J.; Berman, B.; Rosenberg, C.; Levitt, J. Desoximetasone 0.25% and tacrolimus 0.1% ointments versus tacrolimus alone in the treatment of atopic dermatitis. Cutis 2006, 78, 357–363. [Google Scholar]
- Shao, M.; Hussain, Z.; Thu, H.E.; Khan, S.; Katas, H.; Ahmed, T.A.; Tripathy, M.; Leng, J.; Qin, H.L.; Bukhari, S.N.A. Drug nanocarrier, the future of atopic diseases: Advanced drug delivery systems and smart management of disease. Colloids Surf. B Biointerfaces 2016, 147, 475–491. [Google Scholar]
- Nayak, A.; Laha, B.; Sen, K. Development of hydroxyapatite-ciprofloxacin bone-implants using “Quality by design”. Acta Pharm. 2011, 61, 25–36. [Google Scholar]
- Grangeia, H.B.; Silva, C.; Simoes, S.P.; Reis, M.S. Quality by design in pharmaceutical manufacturing: A systematic review of current status, challenges and future perspectives. Eur. J. Pharm. Biopharm. 2020, 147, 19–37. [Google Scholar] [CrossRef]
- Garg, N.K.; Sharma, G.; Singh, B.; Nirbhavane, P.; Tyagi, R.K.; Shukla, R.; Katare, O.P. Quality by design (QbD)-enabled development of aceclofenac loaded-nano structured lipid carriers (NLCs): An improved dermatokinetic profile for inflammatory disorder(s). Int. J. Pharm. 2017, 517, 413–431. [Google Scholar] [CrossRef]
- Maltesen, M.J.; Bjerregaard, S.; Hovgaard, L.; Havelund, S.; Van De Weert, M. Quality by design-spray drying of insulin intended for inhalation. Eur. J. Pharm. Biopharm. 2008, 70, 828–838. [Google Scholar]
- Mishra, V.; Thakur, S.; Patil, A.; Shukla, A. Quality by design (QbD) approaches in current pharmaceutical set-up. Expert Opin. Drug Deliv. 2018, 15, 737–758. [Google Scholar]
- Katona, G.; Balogh, G.T.; Dargo, G.; Gaspar, R.; Marki, A.; Ducza, E.; Sztojkov-Ivanov, A.; Tomosi, F.; Kecskemeti, G.; Janaky, T.; et al. Development of meloxicam-human serum albumin nanoparticles for nose-to-brain delivery via application of a quality by design approach. Pharmaceutics 2020, 12, 97. [Google Scholar] [CrossRef]
- Sharma, Y.; Kumar, K.; Padhy, S.K. Formulation and evaluation of Atorvastatin calcium Niosomes. Int. J. Life Sci. Sci. Res. 2016, 2, 1–4. [Google Scholar]
- Desai Sanjeevani, R.D.S.J.I. Development and evaluation of the antinflammatory triamcinolone acetonide loaded niosome. World J. Pharm. Res. 2012, 1, 1464–1474. [Google Scholar]
- Yekta Özer, A.; Atilla Hincal, A.; Bouwstra, J. A novel drug delivery system: Non-Ionic surfactant vesicles. Eur. J. Pharm. Biopharm. 1991, 37, 75–79. [Google Scholar]
- Jivrani, S.D.; Patel, V.K. Formulation, development and evaluation of niosomal drug delivery system for clindamycin phosphate. Pharma Sci. Monit. 2014, 5, 256–274. [Google Scholar]
- Remington, J.P. Remington: The Science and Practice of Pharmacy; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; Volume 1. [Google Scholar]
- Shilakari Asthana, G.; Sharma, P.K.; Asthana, A. In Vitro and In Vivo evaluation of niosomal formulation for controlled delivery of clarithromycin. Scientifica 2016, 2016, 6492953. [Google Scholar] [CrossRef]
- Lingan, M.A.; Sathali, A.A.H.; Kumar, M.V.; Gokila, A. Formulation and evaluation of topical drug delivery system containing clobetasol propionate niosomes. Sci. Rev. Chem. Commun. 2011, 1, 7–17. [Google Scholar]
- Kumbhar, D.; Wavikar, P.; Vavia, P. Niosomal gel of lornoxicam for topical delivery: In Vitro assessment and pharmacodynamic activity. AAPS PharmSciTech 2013, 14, 1072–1082. [Google Scholar]
- Ranga, P.; Natarajan, R.; Rajendran, N. Formulation and evaluation of zidovudine loaded niosomes. J. Pharm. Nanotechnol. 2013, 1, 12–18. [Google Scholar]
- Yoshioka, T.; Sternberg, B.; Florence, A.T. Preparation and properties of vesicles (niosomes) of sorbitan monoesters (Span 20, 40, 60 and 80) and a sorbitan triester (Span 85). Int. J. Pharm. 1994, 105, 1–6. [Google Scholar]
- Okore, V.; Attama, A.; Ofokansi, K.; Esimone, C.; Onuigbo, E. Formulation and evaluation of niosomes. Indian J. Pharm. Sci. 2011, 73, 323. [Google Scholar]
| No | Variables: CMAs and CPPs | Parameters |
|---|---|---|
| 1 | Organic phase preparation (Solvent) | a. Diethyl Ether |
| b. Diethyl Ether: Ethanol (80:20) | ||
| c. Diethyl Ether: Methanol (75:25) | ||
| d. Ethanol | ||
| e. Acetone | ||
| 2 | Drug concentration (mg) | a. 20 mg (0.2%) |
| b. 30 mg (0.3%) | ||
| c. 40 mg (0.4%) | ||
| 3 | Surfactant concentration (mg) | a. 20 mg (0.2%)) |
| b. 30 mg (0.3%) | ||
| c. 40 mg (0.4% | ||
| d. 50 mg (0.5%) | ||
| e. 60 mg (0.6%) | ||
| f. 70 mg (0.7%) | ||
| 4 | Cholesterol concentration (mg) | a. 20 mg (0.2%) |
| b. 30 mg (0.3%) | ||
| c. 40 mg (0.4%) | ||
| d. 50 mg (0.5%) | ||
| 5 | Selection of lipid | a. Stearic acid |
| b. Stearylamines | ||
| c. No lipids | ||
| 6 | External phase temperature (°C) | a. 55 °C |
| b. 65 °C | ||
| c. 75 °C | ||
| 7 | External phase volume (mL) | a. 10 mL |
| b. 20 mL | ||
| c. 30 mL | ||
| 8 | Internal phase volume (mL) | a. 10 mL |
| b. 15 mL | ||
| c. 20 mL | ||
| 9 | Mixing speed (rpm) | a. 450 rpm |
| b. 550 rpm | ||
| c. 650 rpm | ||
| 10 | Mixing time (minutes) | a. 30 min |
| b. 45 min | ||
| c. 60 min | ||
| 11 | Addition Rate (mL/min) | a. 0.25 mL/min |
| b. 0.50 mL/min | ||
| c. 1.00 mL/min |
| Batch ID # | Critical Material Attributes | Critical Processing Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Drug (mg) | Organic Phase Composition | Span 60 (mg) | Cholesterol (mg) | Stearic Acid (mg) | External Phase Temperature (°C) | External Phase Volume (mL) | Internal Phase Volume (mL) | Mixing Speed (RPM) | Mixing Time (Minutes) | Add. Rate (mL/Min) | |
| DND-1 | 20 | diethyl ether | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-2 | 20 | diethyl ether : ethanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-3 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-4 | 20 | ethanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-5 | 20 | acetone | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-6 | 30 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-7 | 40 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-8 | 20 | diethyl ether : methanol | 20 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-9 | 20 | diethyl ether : methanol | 30 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-10 | 20 | diethyl ether : methanol | 50 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-11 | 20 | diethyl ether : methanol | 60 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-12 | 20 | diethyl ether : methanol | 70 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-13 | 20 | diethyl ether : methanol | 40 | 30 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-14 | 20 | diethyl ether : methanol | 40 | 40 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-15 | 20 | diethyl ether : methanol | 40 | 50 | 5 | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-16 | 20 | diethyl ether : methanol | 40 | 20 | 5 (SA) | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-17 | 20 | diethyl ether : methanol | 40 | 20 | N/A | 65 | 20 | 10 | 650 | 60 | 1.00 |
| DND-18 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 55 | 20 | 10 | 650 | 60 | 1.00 |
| DND-19 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 75 | 20 | 10 | 650 | 60 | 1.00 |
| DND-20 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 10 | 10 | 650 | 60 | 1.00 |
| DND-21 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 30 | 10 | 650 | 60 | 1.00 |
| DND-22 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 15 | 650 | 60 | 1.00 |
| DND-23 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 20 | 650 | 60 | 1.00 |
| DND-24 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 450 | 60 | 1.00 |
| DND-25 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 550 | 60 | 1.00 |
| DND-26 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 30 | 1.00 |
| DND-27 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 45 | 1.00 |
| DND-28 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 0.25 |
| DND-29 | 20 | diethyl ether : methanol | 40 | 20 | 5 | 65 | 20 | 10 | 650 | 60 | 0.50 |
| Batch ID # | RESULTS (NIOSOMAL DISPERSION) | |||||
|---|---|---|---|---|---|---|
| Dispersion Formed or Not | Organoleptic Properties | Entrapment Efficiency (%) | Particle Size (nm) | Polydispersity Index | Zeta Potential (mV) | |
| DND-1 | No | N/A | N/A | N/A | N/A | N/A |
| DND-2 | Yes | White Milky | 81.48 ± 0.07 | 423.27 ± 16.48 | 0.294 ± 0.04 | −75.63 ± 0.61 |
| DND-3 | Yes | White Milky | 93.69 ± 0.05 | 374.80 ± 9.48 | 0.289 ± 0.01 | −63.83 ± 4.26 |
| DND-4 | Yes | White Milky | 75.30 ± 0.06 | 154.40 ± 1.47 | 0.144 ± 0.02 | −54.13 ± 1.16 |
| DND-5 | Yes | White Milky | 84.79 ± 0.06 | 161.87 ± 10.83 | 0.330 ± 0.03 | −52.10 ± 1.51 |
| DND-6 | Yes | White Milky | 89.38 ± 0.03 | 411.20 ± 22.53 | 0.235 ± 0.03 | −43.03 ± 0.59 |
| DND-7 | Yes | White Milky | 91.43 ± 0.01 | 655.07 ± 46.64 | 0.276 ± 0.02 | −46.30 ± 0.87 |
| DND-8 | Yes | White Milky | 83.06 ± 0.04 | 759.87 ± 16.66 | 0.316 ± 0.01 | −51.03 ± 0.15 |
| DND-9 | Yes | White Milky | 84.69 ± 0.03 | 377.13 ± 10.90 | 0.322 ± 0.02 | −69.53 ± 0.40 |
| DND-10 | Yes | White Milky | 80.48 ± 0.03 | 346.13 ± 6.03 | 0.303 ± 0.01 | −58.30 ± 0.66 |
| DND-11 | Yes | White Milky | 82.54 ± 0.05 | 874.77 ± 109.42 | 0.394 ± 0.01 | −57.53 ± 1.86 |
| DND-12 | Yes | White Milky | 70.38 ± 0.20 | 457.10 ± 12.21 | 0.328 ± 0.03 | −38.50 ± 2.14 |
| DND-13 | Yes | White Milky | 89.33 ± 0.11 | 709.60 ± 24.44 | 0.345 ± 0.01 | −81.67 ± 2.30 |
| DND-14 | Yes | White Milky | 84.64 ± 0.03 | 919.87 ± 64.56 | 0.371 ± 0.01 | −54.67 ± 1.20 |
| DND-15 | Yes | White Milky | 83.93 ± 1.42 | 794.73 ± 9.07 | 0.324 ± 0.01 | −57.60 ± 2.01 |
| DND-16 | Yes | White Milky | 82.71 ± 0.11 | 56237.83 ± N/A | N/A | 11.43 ± 0.76 |
| DND-17 | Yes | White Milky | 63.90 ± 0.12 | 616.33 ± 7.02 | 0.348 ± 0.01 | −41.53 ± 0.72 |
| DND-18 | Yes | White Milky | 80.75 ± 0.02 | 435.77 ± 27.99 | 0.441 ± 0.05 | −40.23 ± 1.30 |
| DND-19 | Yes | White Milky | 70.41 ± 0.05 | 460.95 ± 12.07 | 0.320 ± 0.02 | −60.07 ± 0.78 |
| DND-20 | Yes | White Milky | 95.58 ± 0.03 | 355.30 ± 8.11 | 0.282 ± 0.01 | −58.53 ± 1.33 |
| DND-21 | Yes | White Milky | 74.56 ± 0.06 | 823.77 ± 19.59 | 0.334 ± 0.01 | −53.77 ± 0.95 |
| DND-22 | Yes | White Milky | 77.68 ± 0.06 | 444.37 ± 0.11 | 0.318 ± 0.01 | −51.17 ± 0.81 |
| DND-23 | Yes | White Milky | 76.48 ± 0.10 | 439.70 ± 56.28 | 0.273 ± 0.03 | −47.23 ± 1.75 |
| DND-24 | Yes | White Milky | 80.41 ± 0.09 | 680.00 ± 15.57 | 0.292 ± 0.01 | −34.13 ± 1.17 |
| DND-25 | Yes | White Milky | 83.04 ± 0.06 | 505.13 ± 20.78 | 0.261 ± 0.03 | −48.73 ± 1.10 |
| DND-26 | Yes | White Milky | 82.36 ± 0.03 | 487.03 ± 30.69 | 0.304 ± 0.01 | −48.73 ± 0.81 |
| DND-27 | Yes | White Milky | 91.59 ± 0.84 | 479.60 ± 7.79 | 0.310 ± 0.02 | −49.53 ± 2.48 |
| DND-28 | Yes | White Milky | 86.12 ± 0.78 | 521.80 ± 23.80 | 0.310 ± 0.00 | −54.17 ± 1.25 |
| DND-29 | Yes | White Milky | 92.34 ± 0.68 | 480.70 ± 24.61 | 0.302 ± 0.03 | −58.60 ± 0.44 |
| Fixed Parameters | Impacting Parameters |
|---|---|
| Method of preparation (Diethyl ether : methanol :: 75 : 25) | Surfactant concentration |
| Drug concentration (20 mg) | Cholesterol concentration |
| Lipid (Stearic Acid—5 mg) | Mixing speed |
| External phase temperature (65 °C) | Mixing time |
| External phase volume (20 mL) | Addition rate |
| Internal phase volume (10 mL) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, P.; Goodyear, B.; Haq, A.; Puri, V.; Michniak-Kohn, B. Evaluations of Quality by Design (QbD) Elements Impact for Developing Niosomes as a Promising Topical Drug Delivery Platform. Pharmaceutics 2020, 12, 246. https://doi.org/10.3390/pharmaceutics12030246
Shah P, Goodyear B, Haq A, Puri V, Michniak-Kohn B. Evaluations of Quality by Design (QbD) Elements Impact for Developing Niosomes as a Promising Topical Drug Delivery Platform. Pharmaceutics. 2020; 12(3):246. https://doi.org/10.3390/pharmaceutics12030246
Chicago/Turabian StyleShah, Parinbhai, Benjamin Goodyear, Anika Haq, Vinam Puri, and Bozena Michniak-Kohn. 2020. "Evaluations of Quality by Design (QbD) Elements Impact for Developing Niosomes as a Promising Topical Drug Delivery Platform" Pharmaceutics 12, no. 3: 246. https://doi.org/10.3390/pharmaceutics12030246
APA StyleShah, P., Goodyear, B., Haq, A., Puri, V., & Michniak-Kohn, B. (2020). Evaluations of Quality by Design (QbD) Elements Impact for Developing Niosomes as a Promising Topical Drug Delivery Platform. Pharmaceutics, 12(3), 246. https://doi.org/10.3390/pharmaceutics12030246









