Light-Activatable Transfection System Using Hybrid Vectors Composed of Thermosensitive Dendron Lipids and Gold Nanorods
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of MUA-AuNR
2.3. Preparation of Hybrid Vector and Complex
2.4. General Characterization
2.5. Agarose Gel Electrophoresis
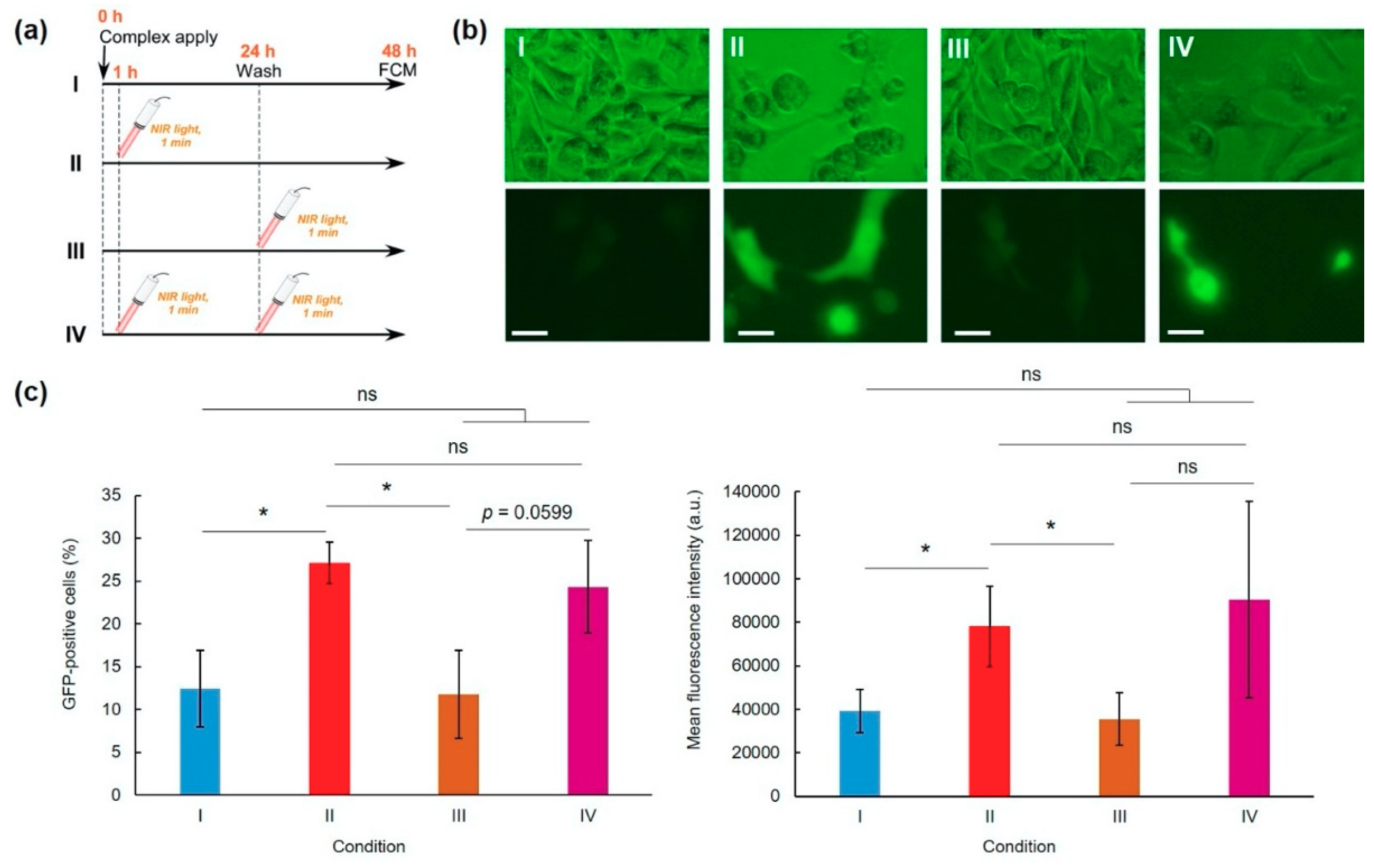
2.6. Transfection
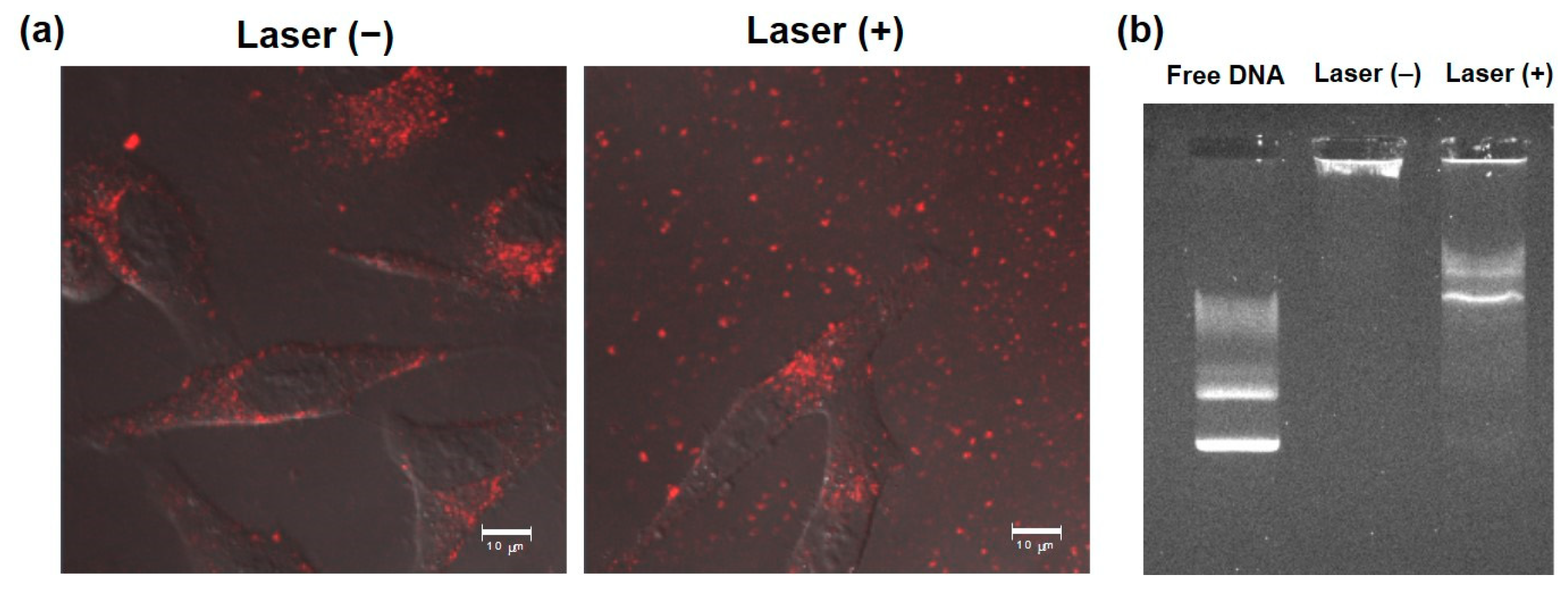
2.7. Cellular Association of Complexes
2.8. Intracellular Distribution Analysis
2.9. Statistical Analysis
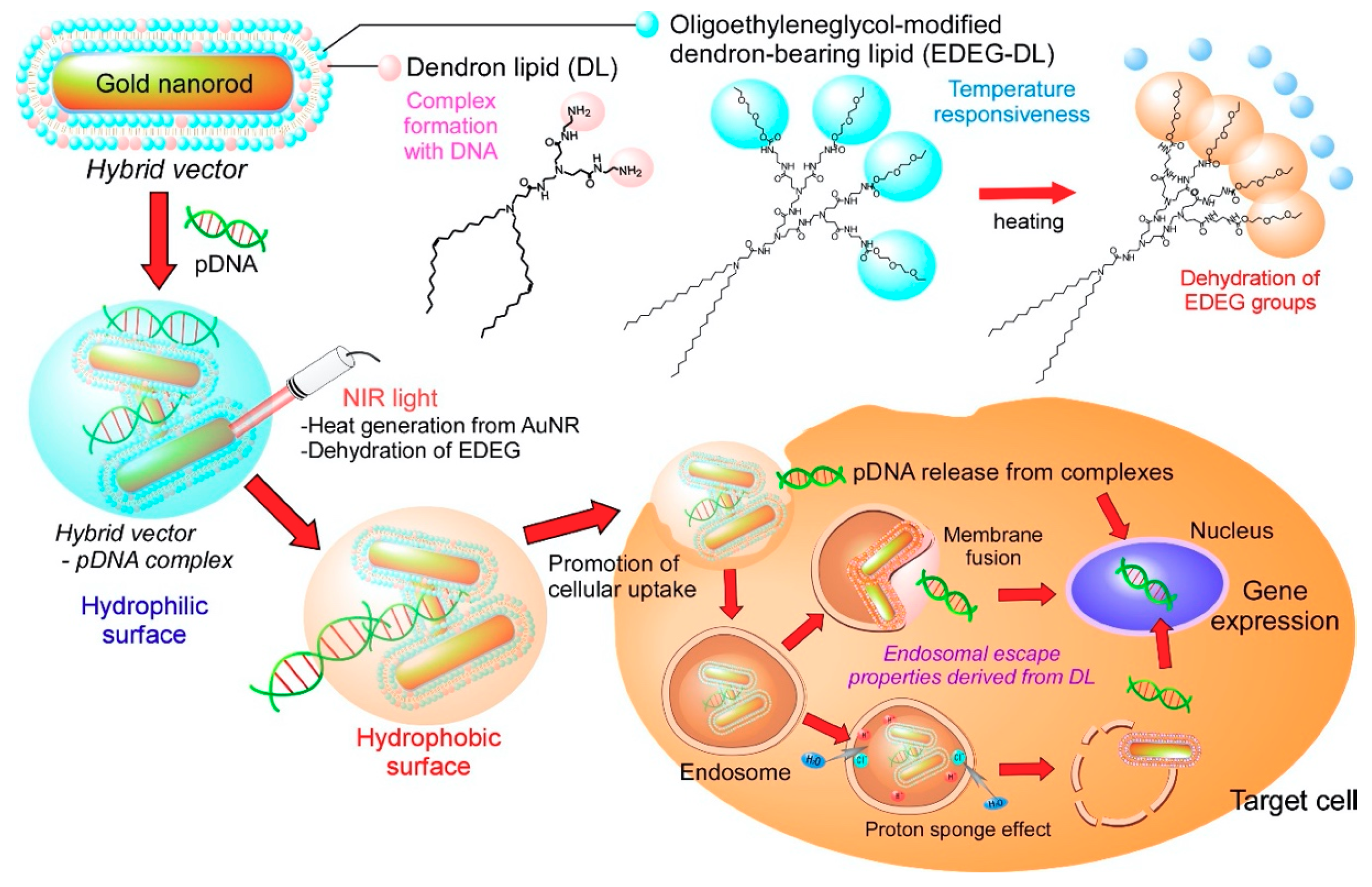
3. Results
3.1. Characterization of Hybrid Vectors and Complexes
3.2. Transfection
3.3. Intracellular Behavior
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, G.; Li, L.; Panwar, N.; Wang, J.; Tjin, S.C.; Wang, X.; Yong, K.-T. Non-viral gene therapy using multifunctional nanoparticles: Status, challenges, and opportunities. Coord. Chem. Rev. 2018, 374, 133–152. [Google Scholar] [CrossRef]
- Lundstrom, K. Viral vectors in gene therapy. Diseases 2018, 6, 42. [Google Scholar] [CrossRef]
- Zylberberg, C.; Gaskill, K.; Pasley, S.; Matosevic, S. Engineering liposomal nanoparticles for targeted gene therapy. Gene Ther. 2017, 24, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Aied, A.; Greiser, U.; Pandit, A.; Wang, W. Polymer gene delivery: Overcoming the obstacles. Drug Discov. Today 2013, 18, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Jiang, Y.; Peng, H.; Chen, Y.; Zhu, P.; Huang, Y. Recent progress in microRNA delivery for cancer therapy by non-viral synthetic vectors. Adv. Drug Deliv. Rev. 2015, 81, 142–160. [Google Scholar] [CrossRef] [PubMed]
- Twaites, B.R.; de las Heras Alarcón, C.; Lavigne, M.; Saulnier, A.; Pennadam, S.S.; Cunliffe, D.; Górecki, D.C.; Alexander, C. Thermoresponsive polymers as gene delivery vectors: Cell viability, DNA transport and transfection studies. J. Control. Release 2005, 108, 472–483. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Ma, L.; Yan, J.; Yan, M.; Gao, C.; Shen, J. The gene transfection efficiency of thermoresponsive N,N,N-trimethyl chitosan chloride-g-poly(N-isopropylacrylamide) copolymer. Biomaterials 2007, 28, 4488–4500. [Google Scholar] [CrossRef] [PubMed]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, 5007–5008. [Google Scholar] [CrossRef]
- Weissleder, R. A clearer vision for in vivo imaging: Progress continues in the development of smaller, more penetrable probes for biological imaging. Nat. Biotechnol. 2001, 19, 316–317. [Google Scholar] [CrossRef]
- Ntziachristos, V.; Ripoll, J.; Wang, L.V.; Weissleder, R. Looking and listening to light: The evolution of whole-body photonic imaging. Nat. Biotechnol. 2005, 23, 313–320. [Google Scholar] [CrossRef]
- Kang, H.; Trondoli, A.C.; Zhu, G.; Chen, Y.; Chang, Y.J.; Liu, H.; Huang, Y.F.; Zhang, X.; Tan, W. Near-infrared light-responsive core–shell nanogels for targeted drug delivery. ACS Nano 2011, 5, 5094–5099. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Jain, P.K.; El-Sayed, I.H.; El-Sayed, M.A. Gold nanoparticles: Interesting optical properties and recent applications in cancer diagnostics and therapy. Nanomedicine 2007, 2, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Harada, A.; Emi, N.; Kono, K. Preparation of efficient gene carriers using a polyamidoamine dendron-bearing lipid: Improvement of serum resistance. Bioconjug. Chem. 2005, 16, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; El-Sayed, M.A. Gold nanoparticles: Optical properties and implementations in cancer diagnosis and photothermal therapy. J. Adv. Res. 2010, 1, 13–28. [Google Scholar] [CrossRef]
- Jain, P.K.; Lee, K.S.; El-Sayed, I.H.; El-Sayed, M.A. Calculated absorption and scattering properties of gold nanoparticles of different size, shape, and composition: Applications in biological imaging and biomedicine. J. Phys. Chem. B 2006, 110, 7238–7248. [Google Scholar] [CrossRef]
- Lee, K.S.; El-Sayed, M.A. Dependence of the enhanced optical scattering efficiency relative to that of absorption for gold metal nanorods on aspect ratio, size, end-cap shape, and medium refractive index. J. Phys. Chem. B 2005, 109, 20331–20338. [Google Scholar] [CrossRef]
- Chen, C.C.; Lin, Y.P.; Wang, C.W.; Tzeng, H.C.; Wu, C.H.; Chen, Y.C.; Chen, C.P.; Chen, L.C.; Wu, Y.C. DNA−gold nanorod conjugates for remote control of localized gene expression by near infrared irradiation. J. Am. Chem. Soc. 2006, 128, 3709–3715. [Google Scholar] [CrossRef]
- Yamashita, S.; Fukushima, H.; Akiyama, Y.; Niidome, Y.; Mori, T.; Katayama, Y.; Niidome, T. Controlled-release system of single-stranded DNA triggered by the photothermal effect of gold nanorods and its in vivo application. Bioorg. Med. Chem. 2011, 19, 2130–2135. [Google Scholar] [CrossRef]
- Kono, K.; Ikeda, R.; Tsukamoto, K.; Yuba, E.; Kojima, C.; Harada, A. Polyamidoamine dendron-bearing lipids as a nonviral vector: Influence of dendron generation. Bioconjug. Chem. 2012, 23, 871–879. [Google Scholar] [CrossRef]
- Takahashi, T.; Kono, K.; Itoh, T.; Emi, N.; Takagishi, T. Synthesis of novel cationic lipids having polyamidoamine dendrons and their transfection activity. Bioconjug. Chem. 2003, 14, 764–773. [Google Scholar] [CrossRef]
- Takahashi, T.; Yuba, E.; Kojima, C.; Harada, A.; Kono, K. Synthesis of a polyamidoamine dendron-bearing lipid having sugar moieties and its use for preparation of nonviral gene vectors. Res. Chem. Intermed. 2009, 35, 1005–1014. [Google Scholar] [CrossRef]
- Takahashi, T.; Hirose, J.; Kojima, C.; Harada, A.; Kono, K. Synthesis of poly(amidoamine) dendron-bearing lipids with poly(ethylene glycol) grafts and their use for stabilization of nonviral gene vectors. Bioconjug. Chem. 2007, 18, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Kono, K.; Murakami, E.; Hiranaka, Y.; Yuba, E.; Kojima, C.; Harada, A.; Sakurai, K. Thermosensitive molecular assemblies from poly(amidoamine) dendron-based lipids. Angew. Chem. Int. Ed. 2011, 50, 6332–6336. [Google Scholar] [CrossRef] [PubMed]
- Kono, K. Dendrimer-based bionanomaterials produced by surface modification, assembly and hybrid formation. Polym. J. 2012, 44, 531–540. [Google Scholar] [CrossRef]
- Hashimoto, T.; Hirai, Y.; Yuba, E.; Harada, A.; Kono, K. Temperature-responsive molecular assemblies using oligo(ethylene glycol)-attached polyamidoamine dendron lipids and their functions as drug carriers. J. Funct. Biomater. 2020. under revision. [Google Scholar]
- Takahashi, T.; Kojima, C.; Harada, A.; Kono, K. Alkyl chain moieties of polyamidoamine dendron-bearing lipids influence their function as a nonviral gene vector. Bioconjug. Chem. 2007, 18, 1349–1354. [Google Scholar] [CrossRef]
- Kono, K.; Takeda, K.; Li, X.; Yuba, E.; Harada, A.; Ozaki, T.; Mori, S. Dually functionalized dendrimers by temperature-sensitive surface modification and gold nanoparticle loading for biomedical application. RSC Adv. 2014, 4, 27811–27819. [Google Scholar] [CrossRef]
- Jana, N.R. Gram-scale synthesis of soluble, near-monodisperse gold nanorods and other anisotropic nanoparticles. Small 2005, 1, 875–882. [Google Scholar] [CrossRef]
- Cao, J.; Galbraith, E.K.; Sun, T.; Grattan, K.T.V. Effective surface modification of gold nanorods for localized surface plasmon resonance-based biosensors. Sens. Actuators B Chem. 2012, 169, 360–367. [Google Scholar] [CrossRef]
- Zhou, J.; Cao, Z.; Panwar, N.; Hu, R.; Wang, X.; Qu, J.; Tjin, S.C.; Xu, G.; Yong, K.T. Functionalized gold nanorods for nanomedicine: Past, present and future. Coord. Chem. Rev. 2017, 352, 15–66. [Google Scholar] [CrossRef]
- Chen, H.; Shao, L.; Li, Q.; Wang, J. Gold nanorods and their plasmonic properties. Chem. Soc. Rev. 2013, 42, 2679–2724. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Thomas, R.; Swathi, R.S.; Thomas, K.G. Au nanorod quartets and Raman signal enhancement: Towards the design of plasmonic platforms. Nanoscale 2014, 6, 10454–10459. [Google Scholar] [CrossRef]
- Khorashad, L.K.; Besteiro, L.V.; Wang, Z.; Valentine, J.; Govorov, A.O. Localization of excess temperature using plasmonic hot spots in metal nanostructures: Combining nano-optical antennas with the Fano effect. J. Phys. Chem. C 2016, 120, 13215–13226. [Google Scholar] [CrossRef]
- Nakatsuji, H.; Kawabata-Galbraith, K.; Kurisu, J.; Imahori, H.; Murakami, T.; Kengaku, M. Surface chemistry for cytosolic gene delivery and photothermal transgene expression by gold nanorods. Sci. Rep. 2017, 7, 4694. [Google Scholar] [CrossRef] [PubMed]
- Rui, Y.; Pang, B.; Zhang, J.; Liu, Y.; Hu, H.; Liu, Z.; Ama Baidoo, S.; Liu, C.; Zhao, Y.; Li, S. Near-infrared light-activatable siRNA delivery by microcapsules for combined tumour therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Liu, F.; Li, W.; Guo, X.; Wang, Z.; Zhang, H.; Li, Q.; Luo, L.; Du, Y.; Jin, Y.; et al. Smart carbon nanotubes with laser-controlled behavior in gene delivery and therapy through a non-digestive trafficking pathway. Small 2016, 12, 6753–6766. [Google Scholar] [CrossRef] [PubMed]
- Wiraja, C.; Mathiyazhakan, M.; Movahedi, F.; Upputuri, P.K.; Cheng, Y.; Pramanik, M.; Yang, L.; Becker, D.L.; Xu, C. Near-infrared light-sensitive liposomes for enhanced plasmid DNA transfection. Bioeng. Transl. Med. 2016, 1, 357–364. [Google Scholar] [CrossRef]
- Yuba, E.; Nakajima, Y.; Tsukamoto, K.; Iwashita, S.; Kojima, C.; Harada, A.; Kono, K. Effect of unsaturated alkyl chains on transfection activity of poly(amidoamine) dendron-bearing lipids. J. Control. Release 2012, 160, 552–560. [Google Scholar] [CrossRef]
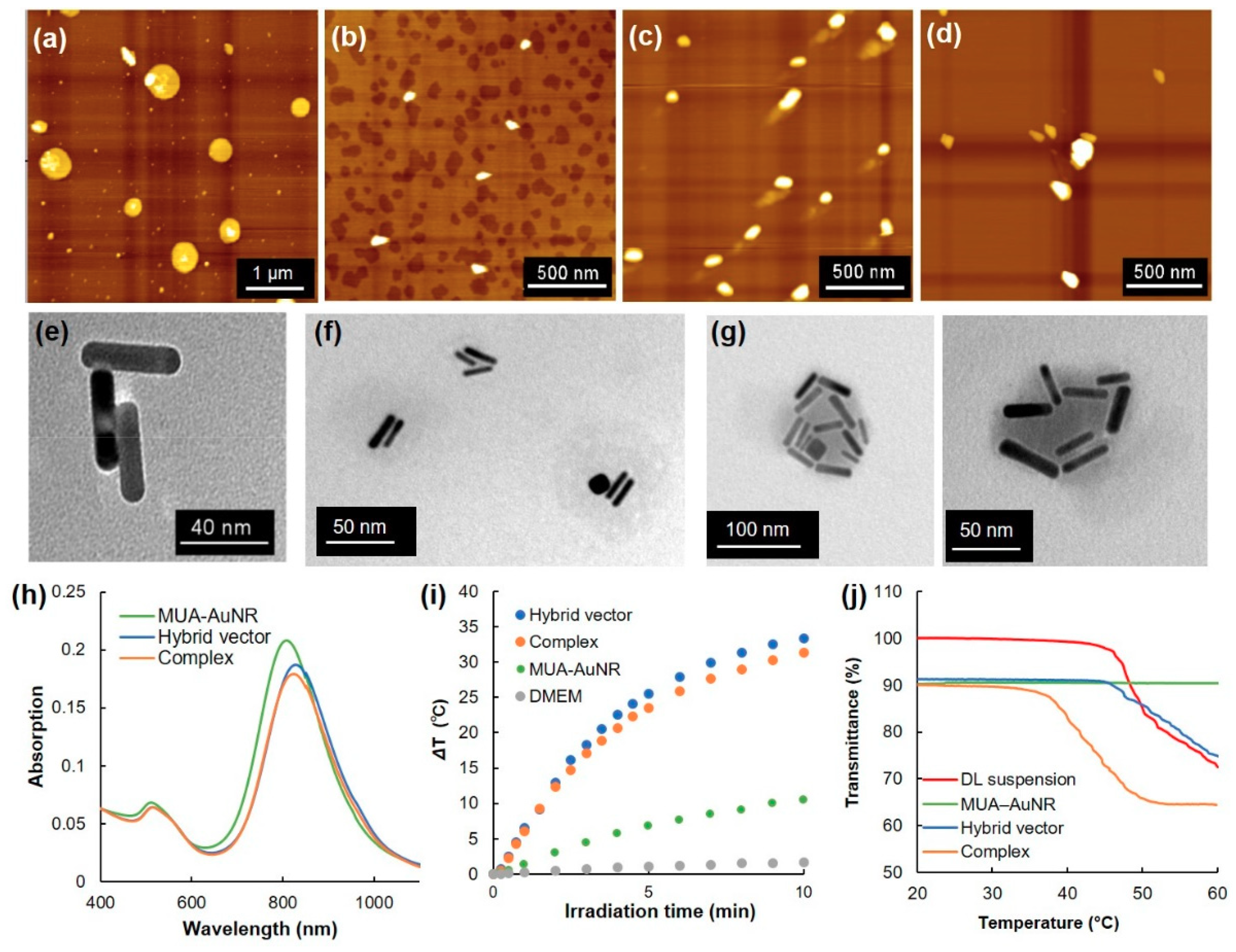


| Sample | Temperature (°C) | Particle Size (nm) | Zeta Potential (mV) |
|---|---|---|---|
| MUA-AuNR | 25 | 36 ± 1 | −23.5 ± 1.7 |
| DL suspension | 25 | 724 ± 73 | 9.19 ± 0.2 |
| 50 | 2565 ± 248 | – | |
| Hybrid vector | 25 | 135 ± 12 | 4.19 ± 0.4 |
| 50 | 297 ± 19 | – | |
| Complex | 25 | 185 ± 50 | 3.66 ± 0.8 |
| 50 | 300 ± 21 | – |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashimoto, T.; Hirata, T.; Yuba, E.; Harada, A.; Kono, K. Light-Activatable Transfection System Using Hybrid Vectors Composed of Thermosensitive Dendron Lipids and Gold Nanorods. Pharmaceutics 2020, 12, 239. https://doi.org/10.3390/pharmaceutics12030239
Hashimoto T, Hirata T, Yuba E, Harada A, Kono K. Light-Activatable Transfection System Using Hybrid Vectors Composed of Thermosensitive Dendron Lipids and Gold Nanorods. Pharmaceutics. 2020; 12(3):239. https://doi.org/10.3390/pharmaceutics12030239
Chicago/Turabian StyleHashimoto, Takuya, Tomoya Hirata, Eiji Yuba, Atsushi Harada, and Kenji Kono. 2020. "Light-Activatable Transfection System Using Hybrid Vectors Composed of Thermosensitive Dendron Lipids and Gold Nanorods" Pharmaceutics 12, no. 3: 239. https://doi.org/10.3390/pharmaceutics12030239
APA StyleHashimoto, T., Hirata, T., Yuba, E., Harada, A., & Kono, K. (2020). Light-Activatable Transfection System Using Hybrid Vectors Composed of Thermosensitive Dendron Lipids and Gold Nanorods. Pharmaceutics, 12(3), 239. https://doi.org/10.3390/pharmaceutics12030239






