Design and Characterization of Sodium Alginate and Poly(vinyl) Alcohol Hydrogels for Enhanced Skin Delivery of Quercetin
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrumentation
2.2. Methods
2.2.1. SA-PVA Hydrogel Preparation
2.2.2. Physicochemical Characterization of the SA-PVA Hydrogels
2.2.3. Determination of Quercetin Loading in the SA-PVA Hydrogels
2.2.4. Swelling Assay
2.2.5. Rheology Studies
2.2.6. Evaluation of the Antioxidant Activity
2.2.7. Permeation Assays
Quercetin Permeation Assay through Isolated SC Model from Pig Ear Skin
Quercetin Permeation Assay through PVPASC
2.2.8. Statistical Analysis
3. Results and Discussion
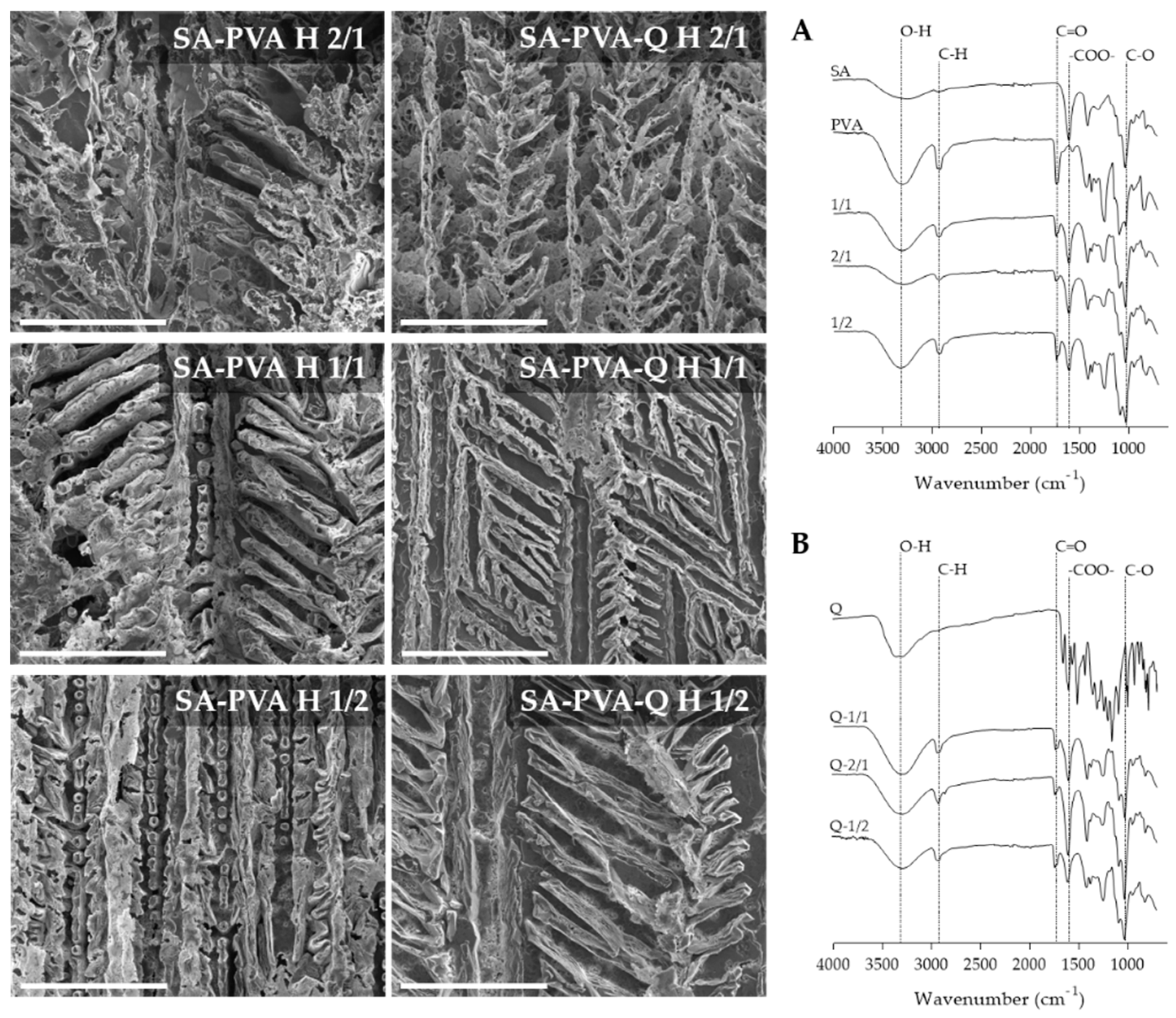
3.1. Preparation and Characterization of SA-PVA Hybrid Hydrogels
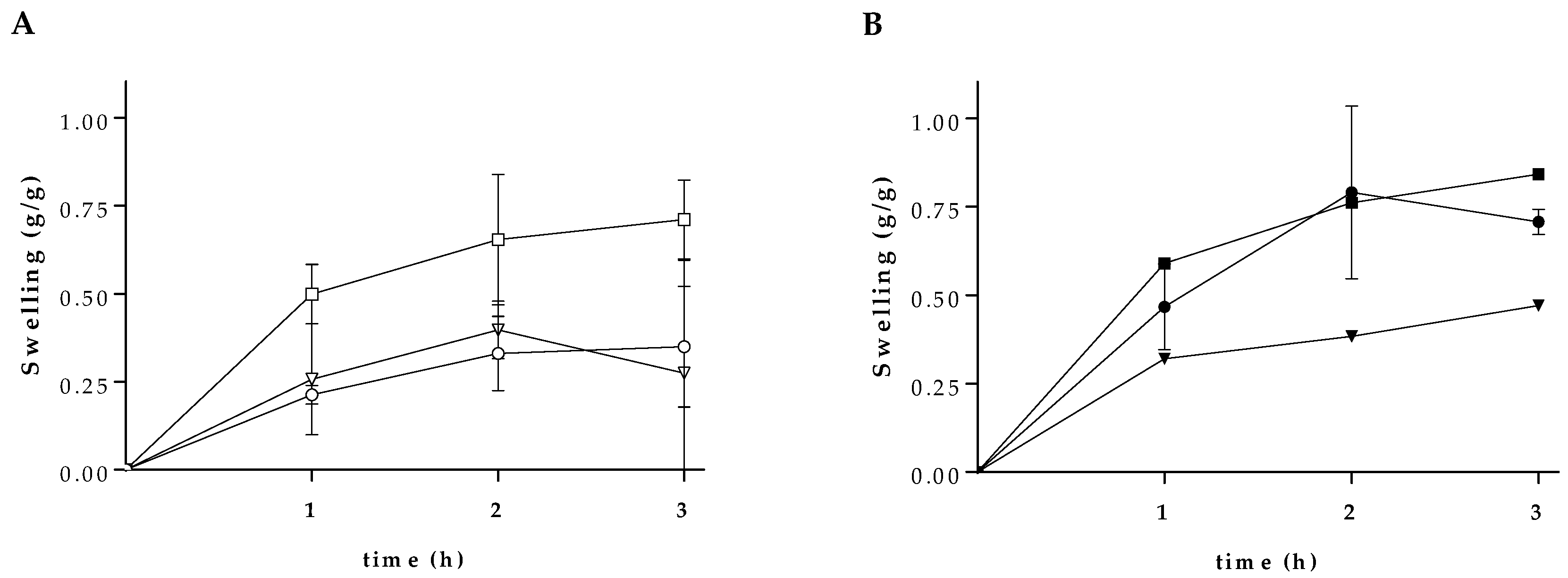
3.2. Swelling Characteristics
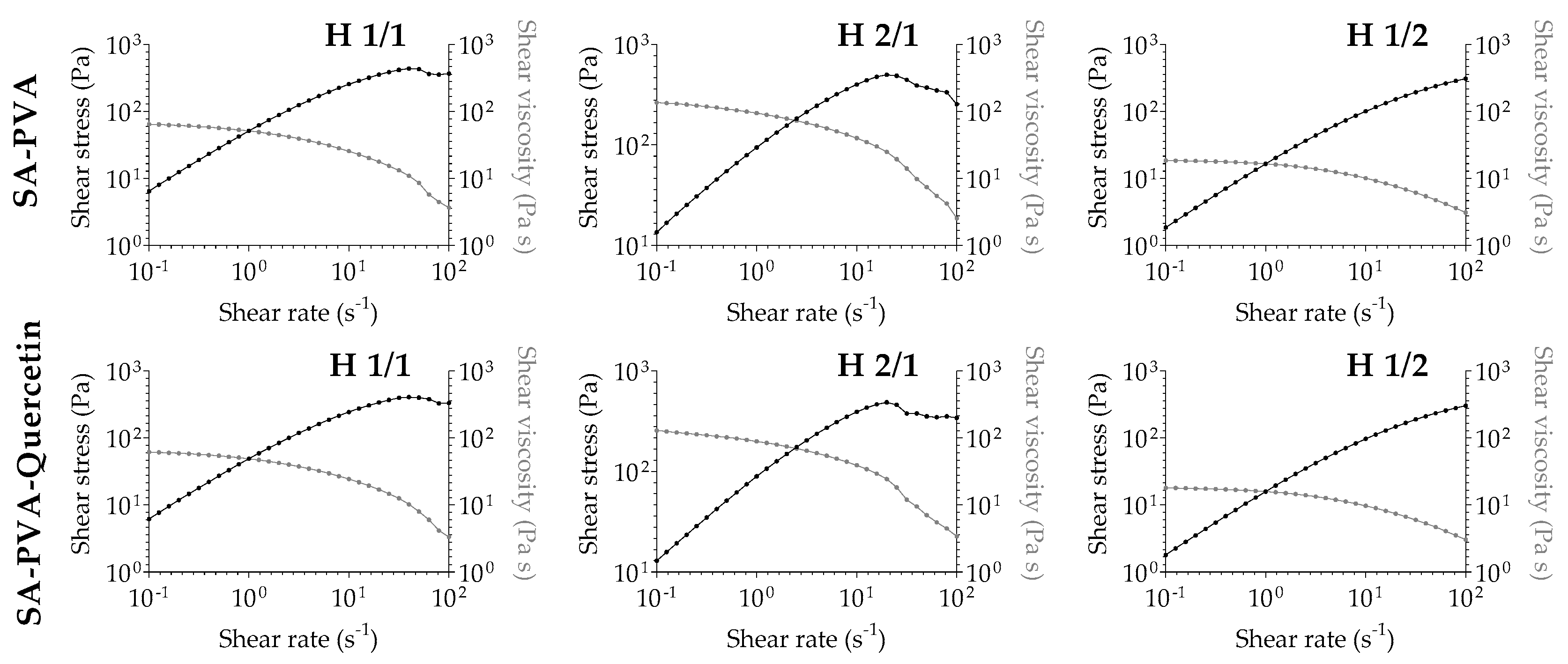
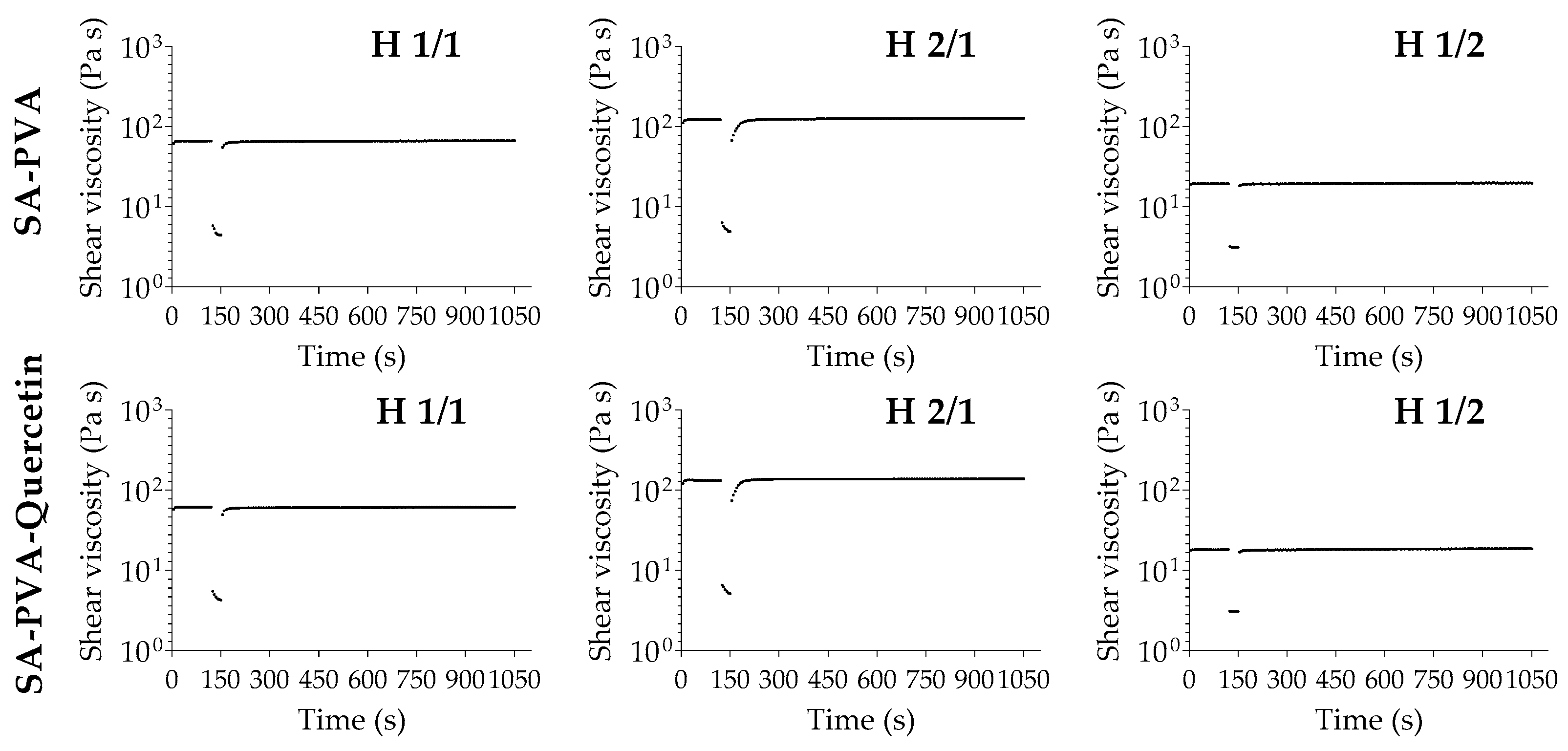
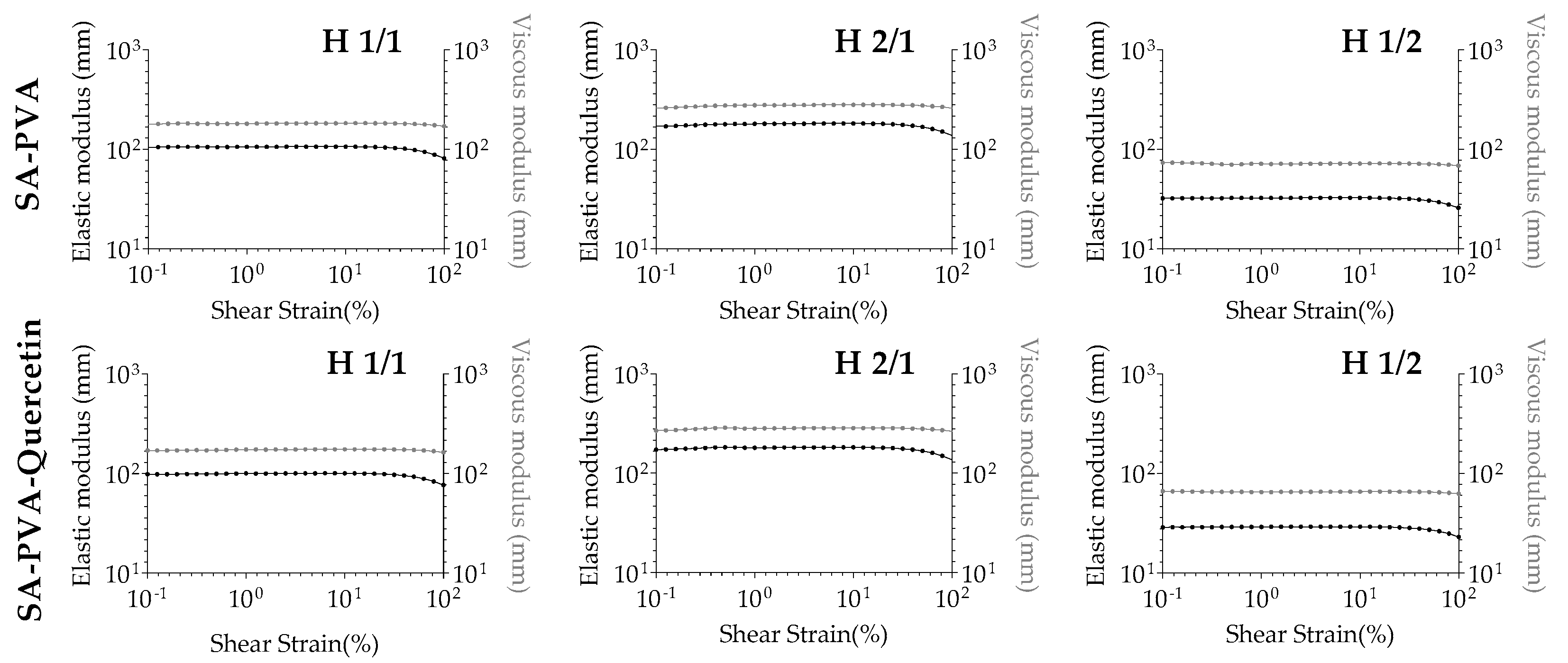
3.3. Rheological Analysis of SA-PVA Hydrogels
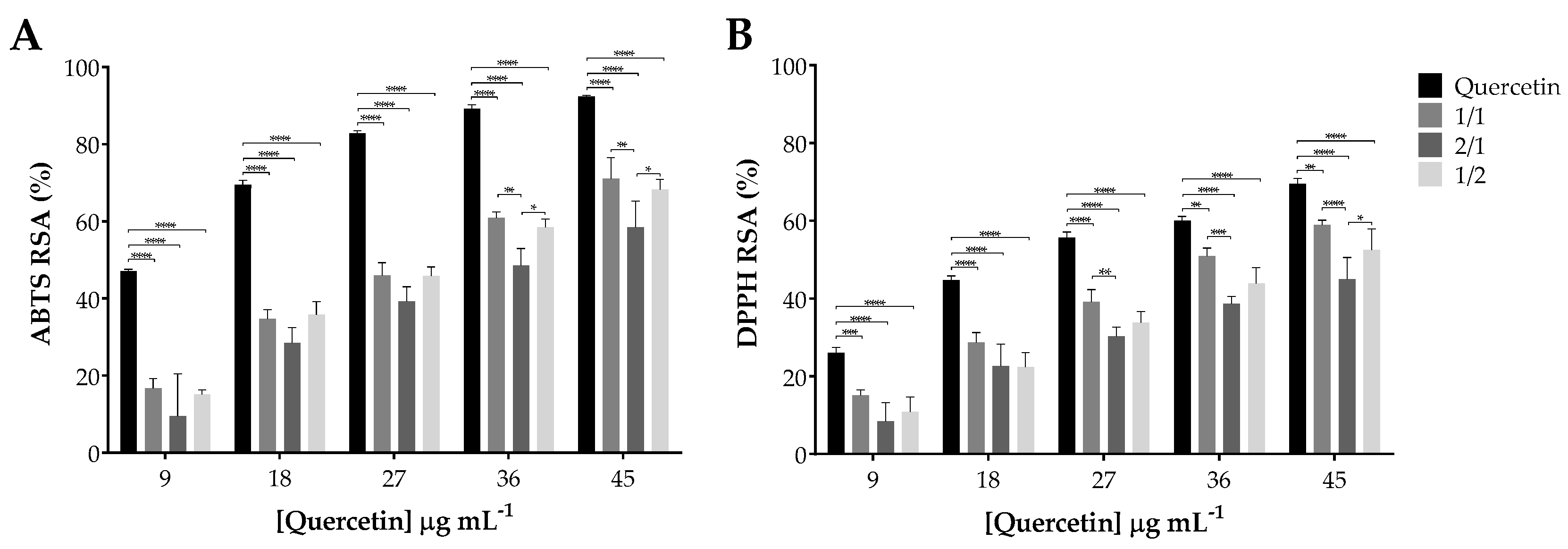
3.4. Antioxidant Activity of Quercetin Incorporated within SA-PVA Hydrogels
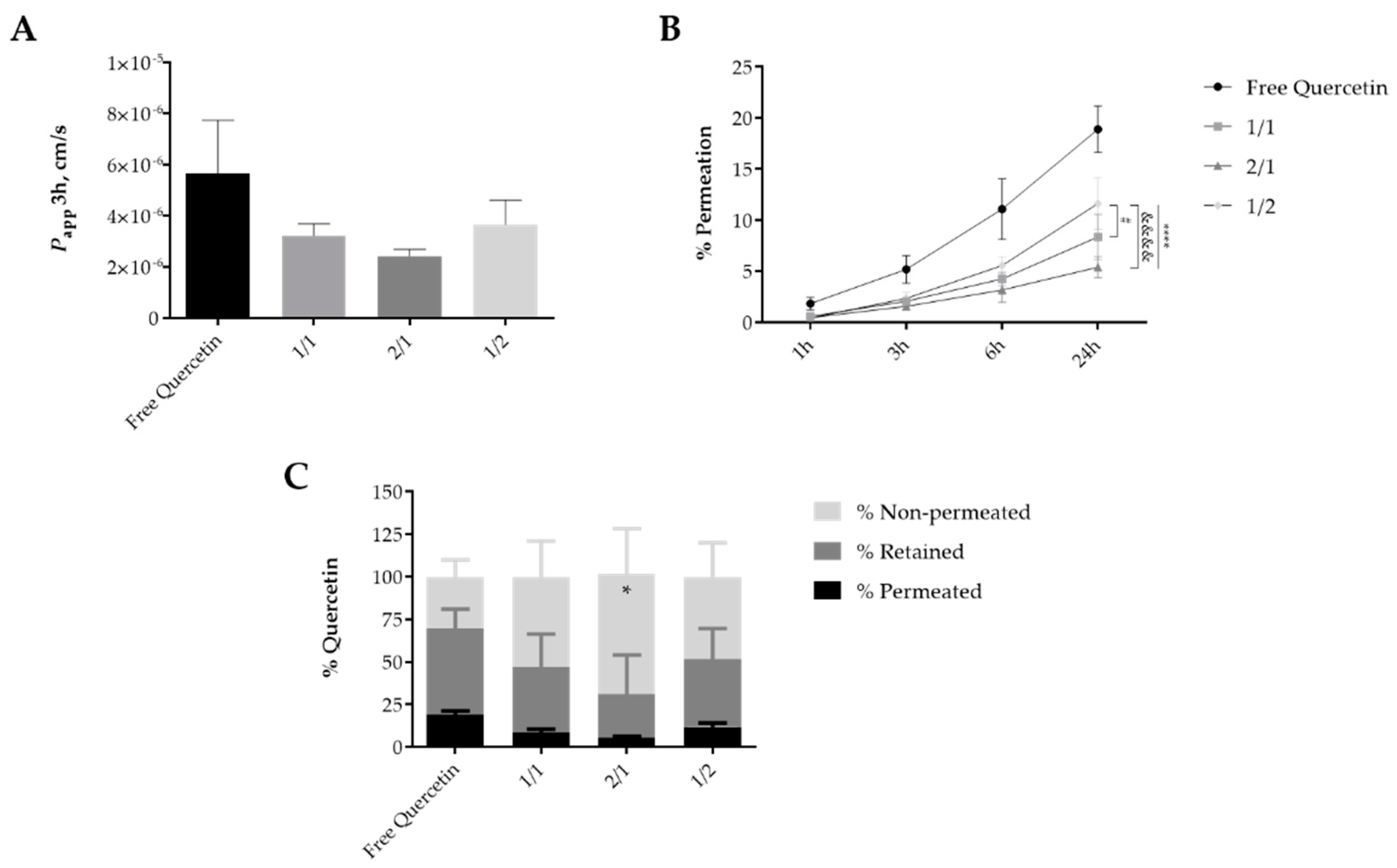
3.5. Permeation of Quercetin-Loaded Hydrogels in SC Models
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jaganathan, S.K.; Mani, M.P.; Khudzari, A.Z.M.; Ismail, A.F.; Ayyar, M.; Rathanasamy, R. Enriched physicochemical and blood-compatible properties of nanofibrous polyurethane patch engrafted with juniper oil and titanium dioxide for cardiac tissue engineering. Int. J. Polym. Anal. Character 2019, 24, 696–708. [Google Scholar] [CrossRef]
- Vignesh, S.; Gopalakrishnan, A.; Poorna, M.R.; Shantikumar, V.N.; Jayakumar, R.; Mony, U. Fabrication of micropatterned alginate-gelatin and k-carrageenan hydrogels of defined shapes using simple wax mould method as a platform for stem cell/induced Pluripotent Stem Cells (iPSC) culture. Int. J. Biol. Macromol. 2018, 112, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, S.B.; Kordestani, S.S.; Mirzadeh, H.; Mansouri, P. Poly (vinyl alcohol)-chitosan blends: Preparation, mechanical and physical properties. Iran. Polym. J. 2003, 12, 139–146. [Google Scholar]
- Iqbal, D.N.; Tariq, M.; Khan, S.M.; Gull, N.; Iqbal, S.S.; Aziz, A.; Nazir, A.; Iqbal, M. Synthesis and characterization of chitosan and guar gum based ternary blends with polyvinyl alcohol. Int. J. Biol. Macromol. 2020, 143, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Harrison, I.P.; Spada, F. Hydrogels for Atopic Dermatitis and Wound Management: A Superior Drug Delivery Vehicle. Pharmaceutics 2018, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Hydrogels as Drug Delivery Systems; Pros and Cons. TiPS 2019, 5, 7–24. [Google Scholar] [CrossRef]
- Ng, C.Y.; Yen, H.; Hsiao, H.Y.; Su, S.C. Phytochemicals in skin cancer prevention and treatment: An updated review. Int. J. Mol. Sci. 2018, 19, 941. [Google Scholar] [CrossRef]
- Vedakumari, W.S.; Ayaz, N.; Karthick, A.S.; Senthil, R.; Sastry, T.P. Quercetin impregnated chitosan-fibrin composite scaffolds as potential wound dressing materials—Fabrication, characterization and in vivo analysis. Eur. J. Pharm. Sci. 2017, 97, 106–112. [Google Scholar] [CrossRef]
- Rothwell, J.A.; Day, A.J.; Morgan, M.R. Experimental determination of octanol-water partition coefficients of quercetin and related flavonoids. J. Agric. Food Chem. 2005, 53, 4355–4360. [Google Scholar] [CrossRef]
- Imran, M.; Iqubal, M.K.; Imtiyaz, K.; Saleem, S.; Mittal, S.; Rizvi, M.M.A.; Ali, J.; Baboota, S. Topical nanostructured lipid carrier gel of quercetin and resveratrol: Formulation, optimization, in vitro and ex vivo study for the treatment of skin cancer. Int. J. Pharm. 2020, 587, 17. [Google Scholar] [CrossRef]
- Hatahet, T.; Morille, M.; Hommoss, A.; Devoisselle, J.M.; Muller, R.H.; Begu, S. Liposomes, lipid nanocapsules and smartCrystals (R): A comparative study for an effective quercetin delivery to the skin. Int. J. Pharm. 2018, 542, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Sunoqrot, S.; Al-Shalabi, E.; Ibrahim, L.H.; Zalloum, H. Nature-Inspired Polymerization of Quercetin to Produce Antioxidant Nanoparticles with Controlled Size and Skin Tone-Matching Colors. Molecules 2019, 24, 3815. [Google Scholar] [CrossRef]
- Verma, N.; Saraf, S. Development and characterization of mannosylated quercetin loaded liposomes for skin carcinoma. Int. J. Pharm. Sci. Res. 2019, 10, 4753–4759. [Google Scholar] [CrossRef]
- Bagde, A.; Patel, K.; Mondal, A.; Kutlehria, S.; Chowdhury, N.; Gebeyehu, A.; Patel, N.; Kumar, N.; Singh, M. Combination of UVB Absorbing Titanium Dioxide and Quercetin Nanogel for Skin Cancer Chemoprevention. AAPS Pharm. Sci. Tech. 2019, 20, 12. [Google Scholar] [CrossRef]
- Chellappan, D.K.; Yee, N.J.; Singh, B.; Panneerselvam, J.; Madheswaran, T.; Chellian, J.; Satija, S.; Mehta, M.; Gulati, M.; Gupta, G.; et al. Formulation and characterization of glibenclamide and quercetin-loaded chitosan nanogels targeting skin permeation. Ther. Deliv. 2019, 10, 281–293. [Google Scholar] [CrossRef]
- Hong, I.K.; Ha, J.H.; Han, S.; Kang, H.; Park, S.N. The Effect of Alkyl Chain Number in Sucrose Surfactant on the Physical Properties of Quercetin-Loaded Deformable Nanoliposome and Its Effect on In Vitro Human Skin Penetration. Nanomaterials 2018, 8, 622. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.Y.; Huang, Y.T.; Chen, Z.Y.; Ye, J.Y.; Xu, H.Y.; Chen, W.R.; Long, X.Y. Niosomal Nanocarriers for Enhanced Skin Delivery of Quercetin with Functions of Anti-Tyrosinase and Antioxidant. Molecules 2019, 24, 2322. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Liu, T.T.; Ma, H.P.; Tian, Y.; Li, L.; Li, Z.; Gao, M.; Zhang, J.B.; Tang, Z.Y. Preparation of Essential Oil-Based Microemulsions for Improving the Solubility, pH Stability, Photostability, and Skin Permeation of Quercetin. AAPS Pharm. Sci. Tech. 2017, 18, 3097–3104. [Google Scholar] [CrossRef]
- Ugazio, E.; Gastaldi, L.; Brunella, V.; Scalarone, D.; Jadhav, S.A.; Oliaro-Bosso, S.; Zonari, D.; Berlier, G.; Miletto, I.; Sapino, S. Thermoresponsive mesoporous silica nanoparticles as a carrier for skin delivery of quercetin. Int. J. Pharm. 2016, 511, 446–454. [Google Scholar] [CrossRef]
- Zhu, X.B.; Zeng, X.W.; Zhang, X.D.; Cao, W.; Wang, Y.L.; Chen, H.J.; Wang, T.; Tsai, H.I.; Zhang, R.; Chang, D.F.; et al. The effects of quercetin-loaded PLGA-TPGS nanoparticles on ultraviolet B-induced skin damages in vivo. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 623–632. [Google Scholar] [CrossRef]
- Jee, J.P.; Pangeni, R.; Jha, S.K.; Byun, Y.; Park, J.W. Preparation and in vivo evaluation of a topical hydrogel system incorporating highly skin-permeable growth factors, quercetin, and oxygen carriers for enhanced diabetic wound-healing therapy. Int. J. Nanomed. 2019, 14, 5449–5475. [Google Scholar] [CrossRef]
- Jiang, Q.; Wu, Y.; Zhang, H.; Liu, P.; Yao, J.; Yao, P.; Chen, J.; Duan, J. Development of essential oils as skin permeation enhancers: Penetration enhancement effect and mechanism of action. Pharm. Biol. 2017, 55, 1592–1600. [Google Scholar] [CrossRef] [PubMed]
- Swartzendruber, D.C.; Kitko, D.J.; Wertz, P.W.; Madison, K.C.; Downing, D.T. Isolation of corneocyte envelopes from porcine epidermis. Arch. Dermat. Res. 1988, 280, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Bowser, P.A.; White, R.J. Isolation, barrier properties and lipid analysis of stratum compactum, a discrete region of the stratum corneum. Br. J. Dermat. 1985, 112, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Groen, D.; Gooris, G.S.; Ponec, M.; Bouwstra, J.A. Two new methods for preparing a unique stratum corneum substitute. Biochim. Biophys. Acta Biomembr. 2008, 1778, 2421–2429. [Google Scholar] [CrossRef]
- Moniz, T.; Lima, S.A.C.; Reis, S. Application of the Human stratum corneum lipid-based mimetic model in assessment of drug-loaded nanoparticles for skin administration. Int. J. Pharm. 2020, 591, 119960. [Google Scholar] [CrossRef]
- Simon, A.; Amaro, M.I.; Healy, A.M.; Cabral, L.M.; de Sousa, V.P. Comparative evaluation of rivastigmine permeation from a transdermal system in the Franz cell using synthetic membranes and pig ear skin with in vivo-in vitro correlation. Int. J. Pharm. 2016, 512, 234–241. [Google Scholar] [CrossRef]
- Shakel, Z.; Nunes, C.; Costa Lima, S.A.; Reis, S. Development of a novel human stratum corneum model, as a tool in the optimization of drug formulations. Int. J. Pharm. 2019, 569, 118571. [Google Scholar] [CrossRef]
- Chen, G.; He, L.; Zhang, P.; Zhang, J.; Mei, X.; Wang, D.; Zhang, Y.; Ren, X.; Chen, Z. Encapsulation of green tea polyphenol nanospheres in PVA/alginate hydrogel for promoting wound healing of diabetic rats by regulating PI3K/AKT pathway. Mat. Sci. Eng. C Mater. Biol. Appl. 2020, 110, 110686. [Google Scholar] [CrossRef]
- Miya, M.; Iwamoto, R.; Mima, S. FT-IR study of intermolecular interactions in polymer blends. J. Polym. Sci. 1984, 22, 1149–1151. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Tang, K.; Hu, X.; Zou, G. Physicochemical characterization and antioxidant activity of quercetin-loaded chitosan nanoparticles. J. Appl. Polym. Sci. 2008, 107, 891–897. [Google Scholar] [CrossRef]
- Souza, M.P.; Vaz, A.F.M.; Correia, M.T.S.; Cerqueira, M.A.; Vicente, A.A.; Carneiro-da-Cunha, M.G. Quercetin-loaded lecithin/chitosan nanoparticles for functional food applications. Food Bioproc. Tech. 2014, 7, 1149–1159. [Google Scholar] [CrossRef]
- Bialik-Wąs, K.; Pluta, K.; Malina, D.; Majka, T.M. Alginate/PVA-based hydrogel matrices with Echinacea purpurea extract as a new approach to dermal wound healing. Int. J. Polym. Mater. 2019. [Google Scholar] [CrossRef]
- Ghasemzadeh, H.; Ghanaat, F. Antimicrobial alginate/PVA silver nanocomposite hydrogel, synthesis and characterization. J. Polym. Res. 2014, 21, 355. [Google Scholar] [CrossRef]
- Carvalho, F.C.; Calixto, G.; Hatakeyama, I.N.; Luz, G.M.; Gremião, M.P.D.; Chorilli, M. Rheological, mechanical, and bioadhesive behavior of hydrogels to optimize skin delivery systems. Drug Dev. Ind. Pharm. 2013, 39, 1750–1757. [Google Scholar] [CrossRef]
- Karpushki, E. Shear rheology of polymer hydrogels. Part I: Proper measurement conditions. In Rheology: Principles, Applications and Environmental Impacts; Nova Science Publishers, Inc.: New York, NY, USA, 2015; pp. 191–204. [Google Scholar]
- Lee, C.H.; Moturi, V.; Lee, Y. Thixotropic property in pharmaceutical formulations. J. Control. Release 2009, 136, 88–98. [Google Scholar] [CrossRef]
- Md, S.; Alhakamy, N.A.; Aldawsari, H.M.; Kotta, S.; Ahmad, J.; Akhter, S.; Alam, M.S.; Khan, M.A.; Awan, Z.; Sivakumar, P.M. Improved analgesic and anti-inflammatory effect of diclofenac sodium by topical nanoemulgel: Formulation development—In vitro and in vivo studies. J. Chem. 2020, 2020. [Google Scholar] [CrossRef]
- Wu, G.; Jin, K.; Liu, L.; Zhang, H. A rapid self-healing hydrogel based on PVA and sodium alginate with conductive and cold-resistant properties. Soft Matter 2020, 16, 3319–3324. [Google Scholar] [CrossRef]
- Deuschle, V.C.K.N.; Deuschle, R.A.N.; Bortoluzzi, M.R.; Athayde, M.L. Physical chemistry evaluation of stability, spreadability, in vitro antioxidant, and photo-protective capacities of topical formulations containing Calendula officinalis L. leaf extract. Braz. J. Pharm. Sci. 2015, 51, 63–75. [Google Scholar] [CrossRef]
- Smilek, J.; Jarábková, S.; Velcer, T.; Pekař, M. Compositional and Temperature Effects on the Rheological Properties of Polyelectrolyte-Surfactant Hydrogels. Polymers 2019, 11, 927. [Google Scholar] [CrossRef]
- Moniz, T.; Lima, S.A.C.; Reis, S. Human skin models: From healthy to disease mimetic systems characteristics and applications. Br. J. Pharmacol. 2020, 177, 4314–4329. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, L.; Barbosa, A.I.; Moniz, T.; Costa Lima, S.; Costa, P.; Celia, C.; Reis, S. Design and Characterization of Sodium Alginate and Poly(vinyl) Alcohol Hydrogels for Enhanced Skin Delivery of Quercetin. Pharmaceutics 2020, 12, 1149. https://doi.org/10.3390/pharmaceutics12121149
Esposito L, Barbosa AI, Moniz T, Costa Lima S, Costa P, Celia C, Reis S. Design and Characterization of Sodium Alginate and Poly(vinyl) Alcohol Hydrogels for Enhanced Skin Delivery of Quercetin. Pharmaceutics. 2020; 12(12):1149. https://doi.org/10.3390/pharmaceutics12121149
Chicago/Turabian StyleEsposito, Ludovico, Ana Isabel Barbosa, Tânia Moniz, Sofia Costa Lima, Paulo Costa, Christian Celia, and Salette Reis. 2020. "Design and Characterization of Sodium Alginate and Poly(vinyl) Alcohol Hydrogels for Enhanced Skin Delivery of Quercetin" Pharmaceutics 12, no. 12: 1149. https://doi.org/10.3390/pharmaceutics12121149
APA StyleEsposito, L., Barbosa, A. I., Moniz, T., Costa Lima, S., Costa, P., Celia, C., & Reis, S. (2020). Design and Characterization of Sodium Alginate and Poly(vinyl) Alcohol Hydrogels for Enhanced Skin Delivery of Quercetin. Pharmaceutics, 12(12), 1149. https://doi.org/10.3390/pharmaceutics12121149










