Solubility and Stability Enhanced Oral Formulations for the Anti-Infective Corallopyronin A
Abstract
1. Introduction
2. Materials and Methods
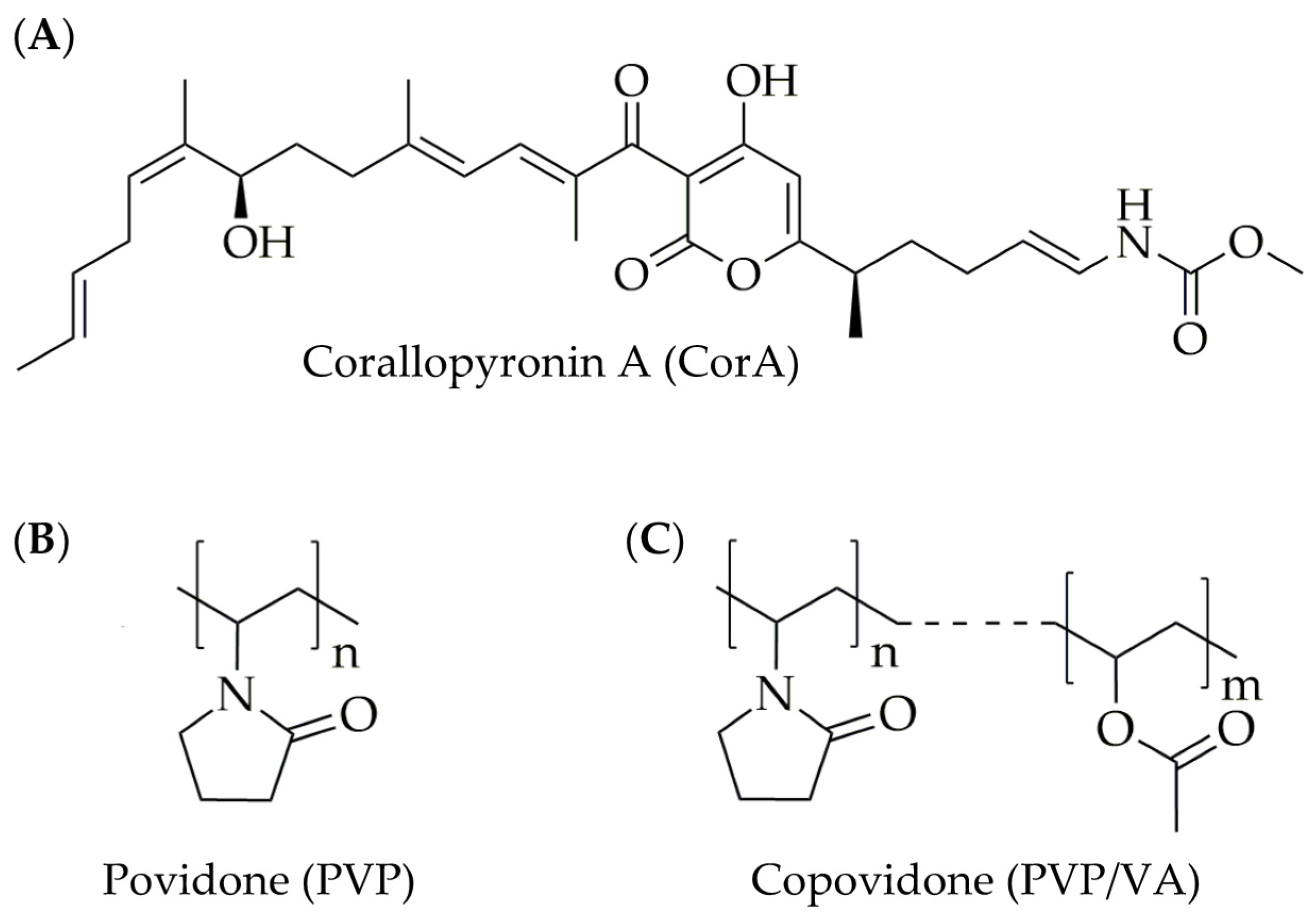
2.1. Materials
2.2. Production of CorA
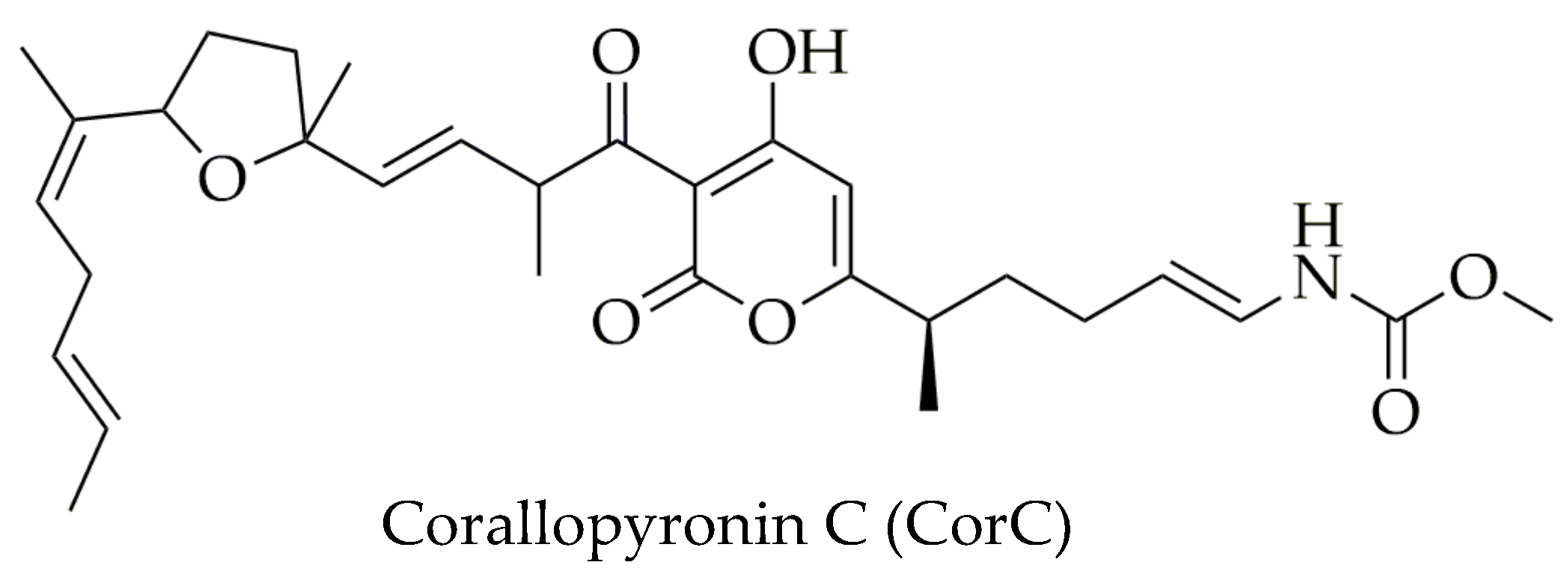
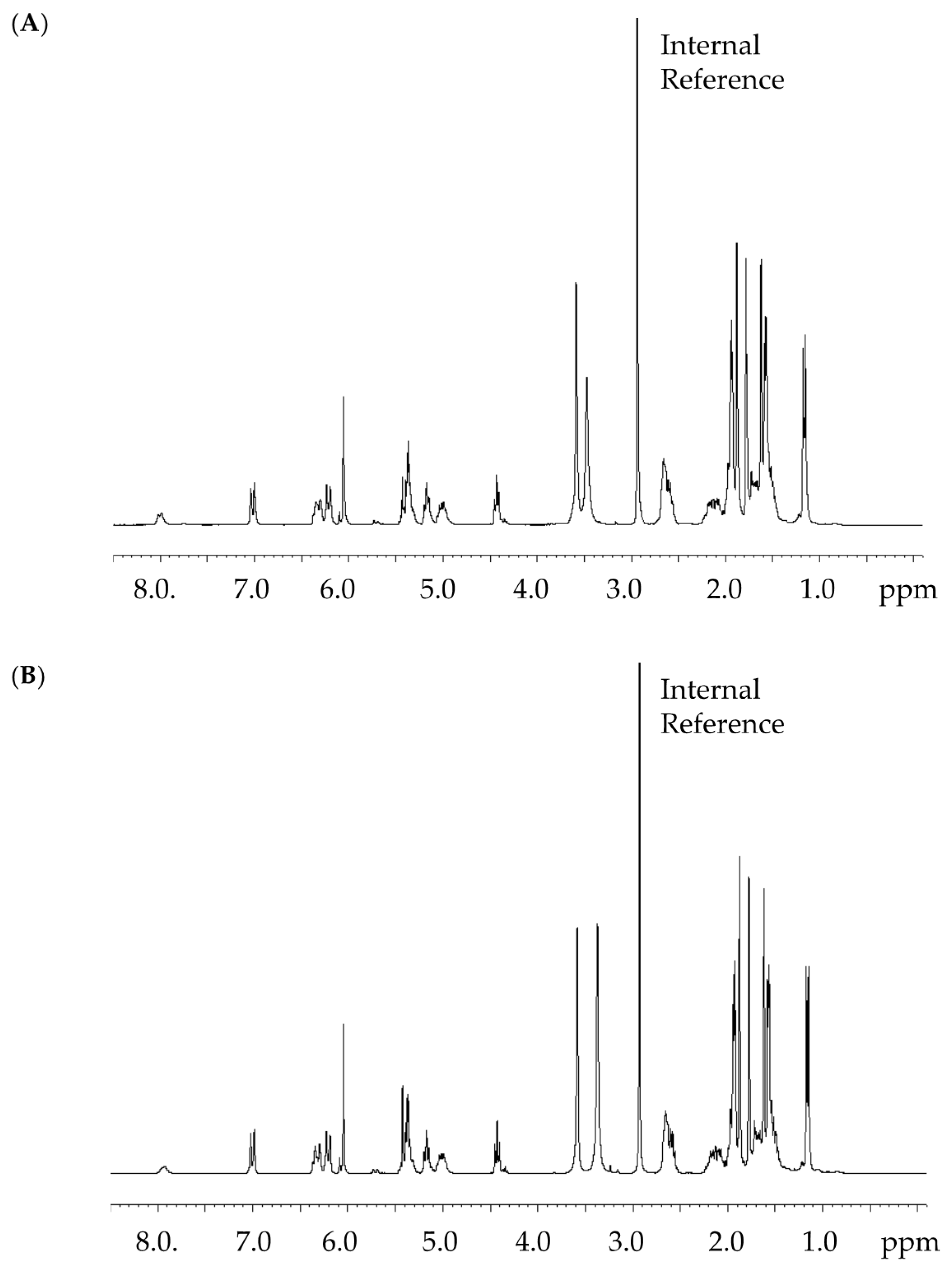
2.3. Content Assay of CorA by 1H NMR
2.4. Physicochemical Characterization of CorA via Pion’s T3 Apparatus
2.5. Preparation of the Spray-Dried CorA-ASD Formulations
2.6. Stability Analysis of CorA by HPLC-DAD
2.7. DSC Analysis
2.8. FT-IR Analysis
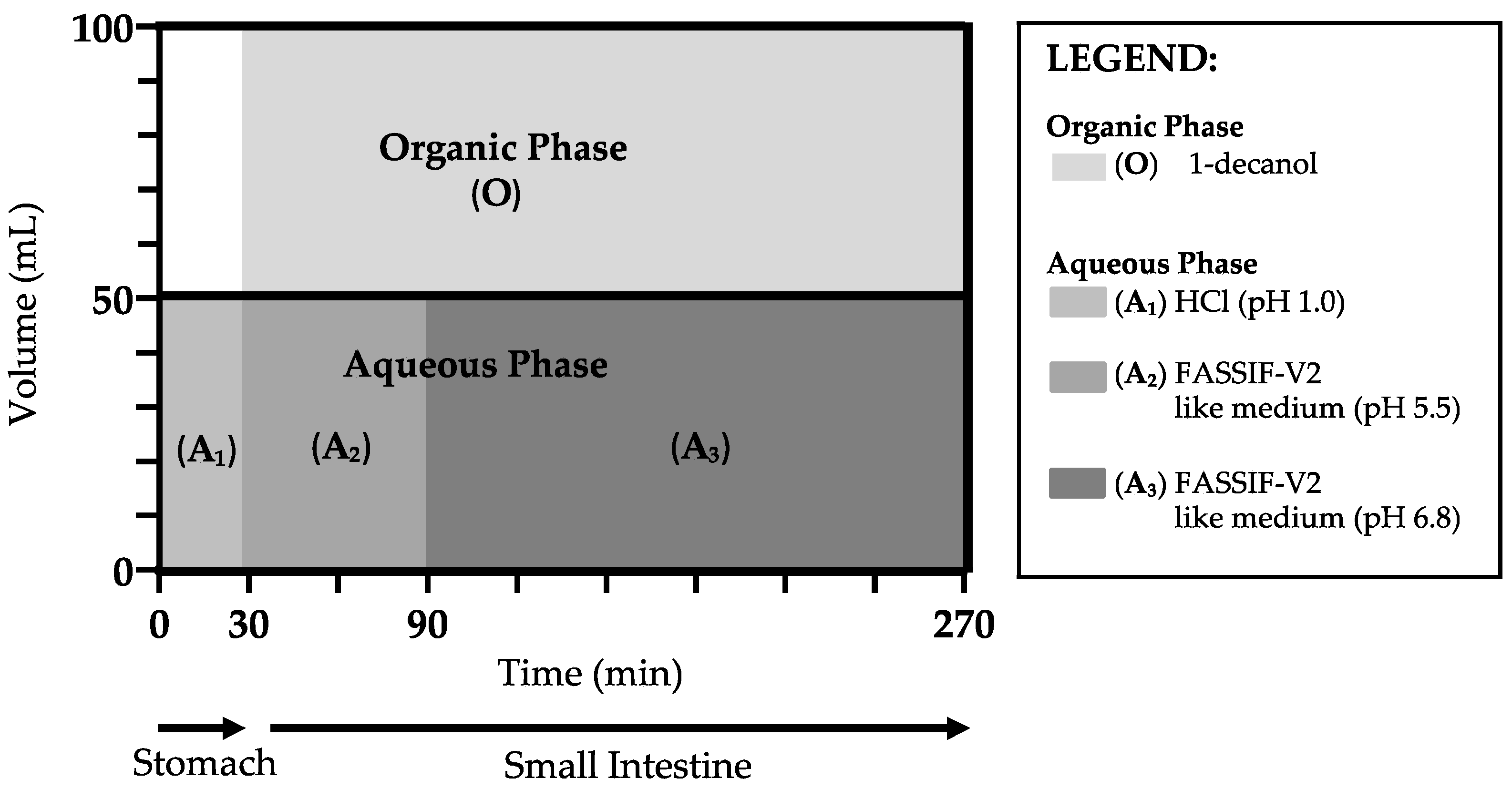
2.9. Biphasic Dissolution Tests via BiPHa+
2.10. Pharmacokinetic Study-Setup and Plasma Sample Analysis
3. Results
3.1. Content Assay of CorA by 1H NMR
3.2. Physicochemical Characterization of CorA via Pions’s T3 Apparatus
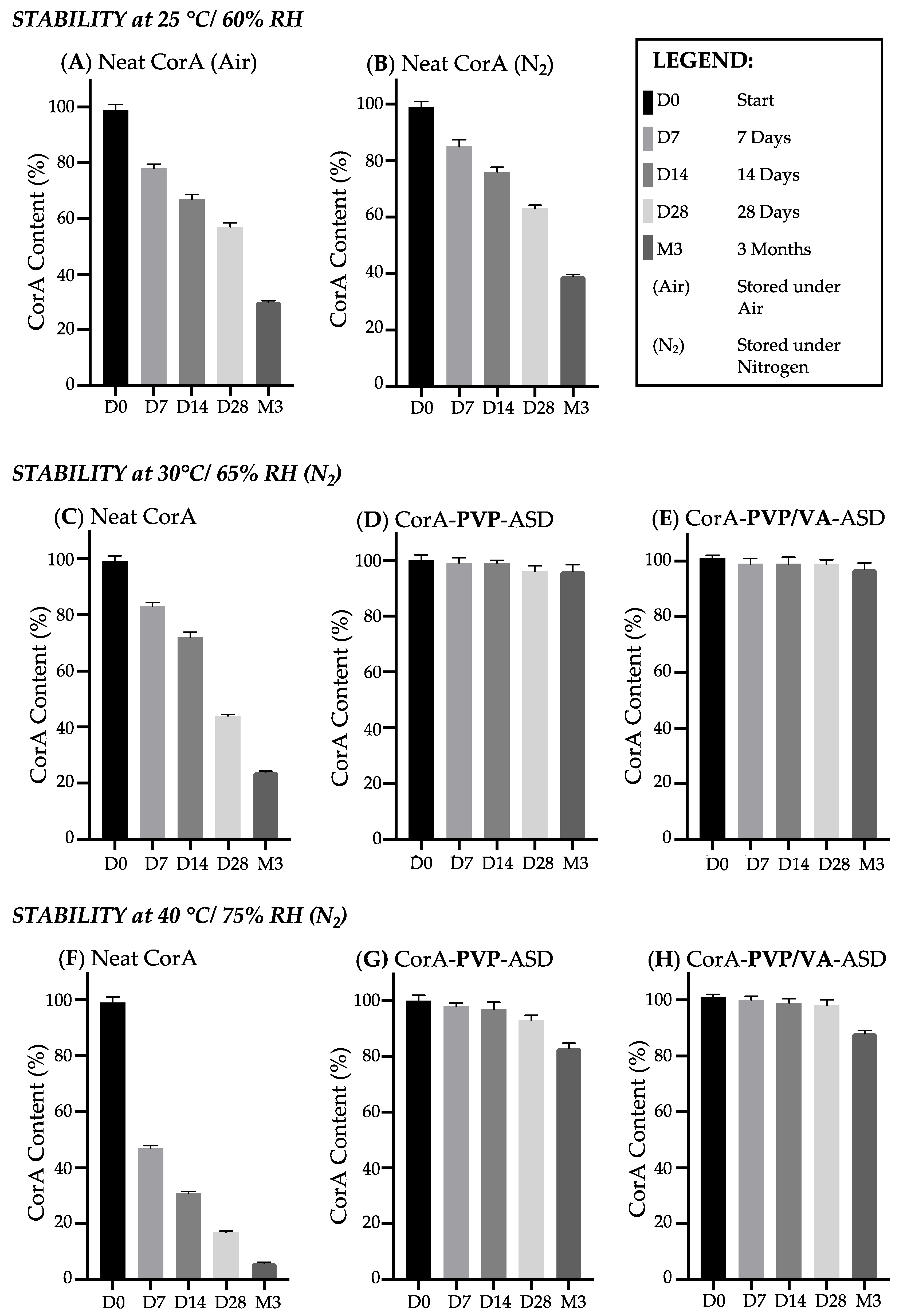
3.3. Stability Analysis by HPLC-DAD
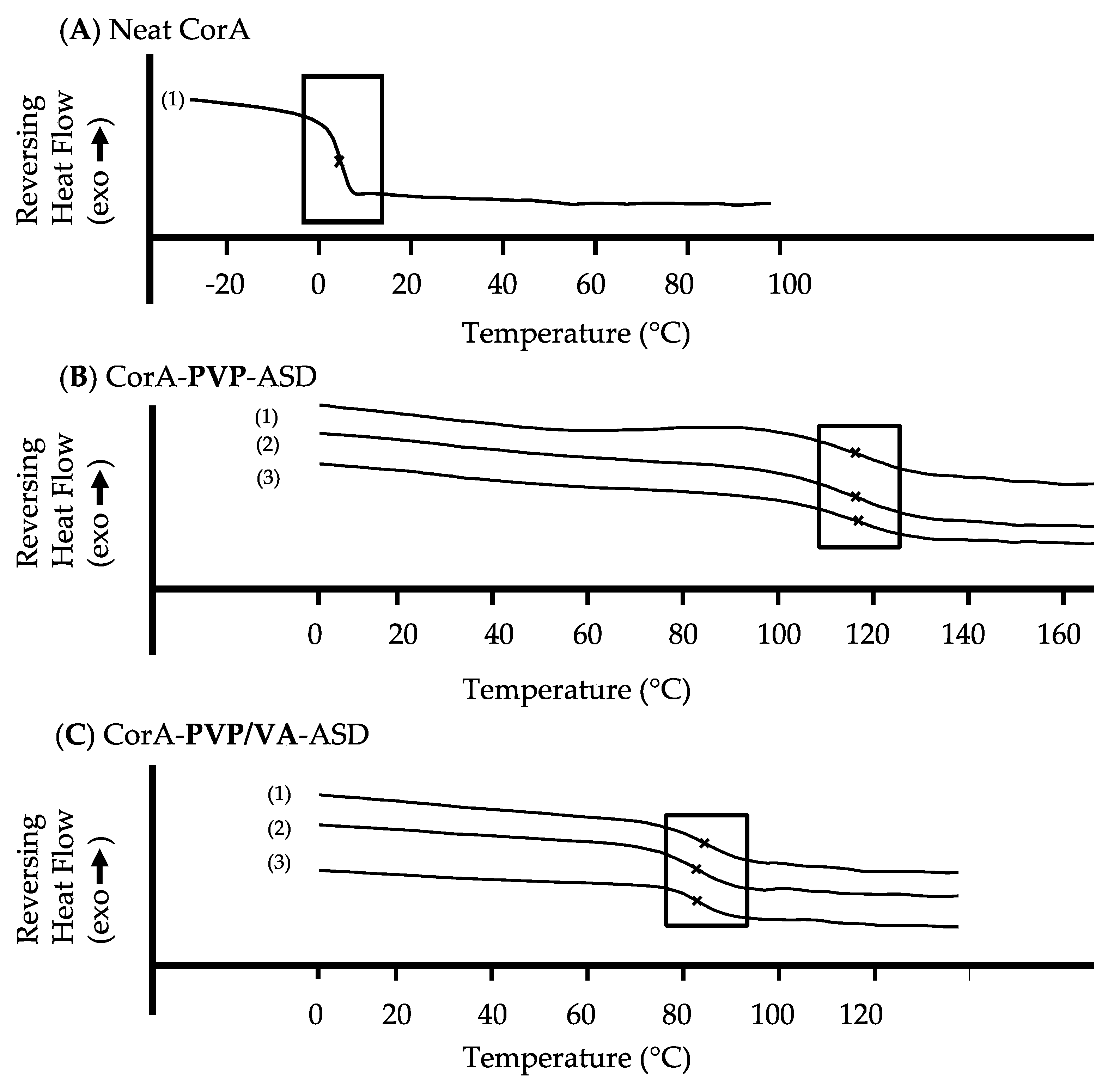
3.4. DSC Analysis of Neat CorA and CorA-ASD Formulations
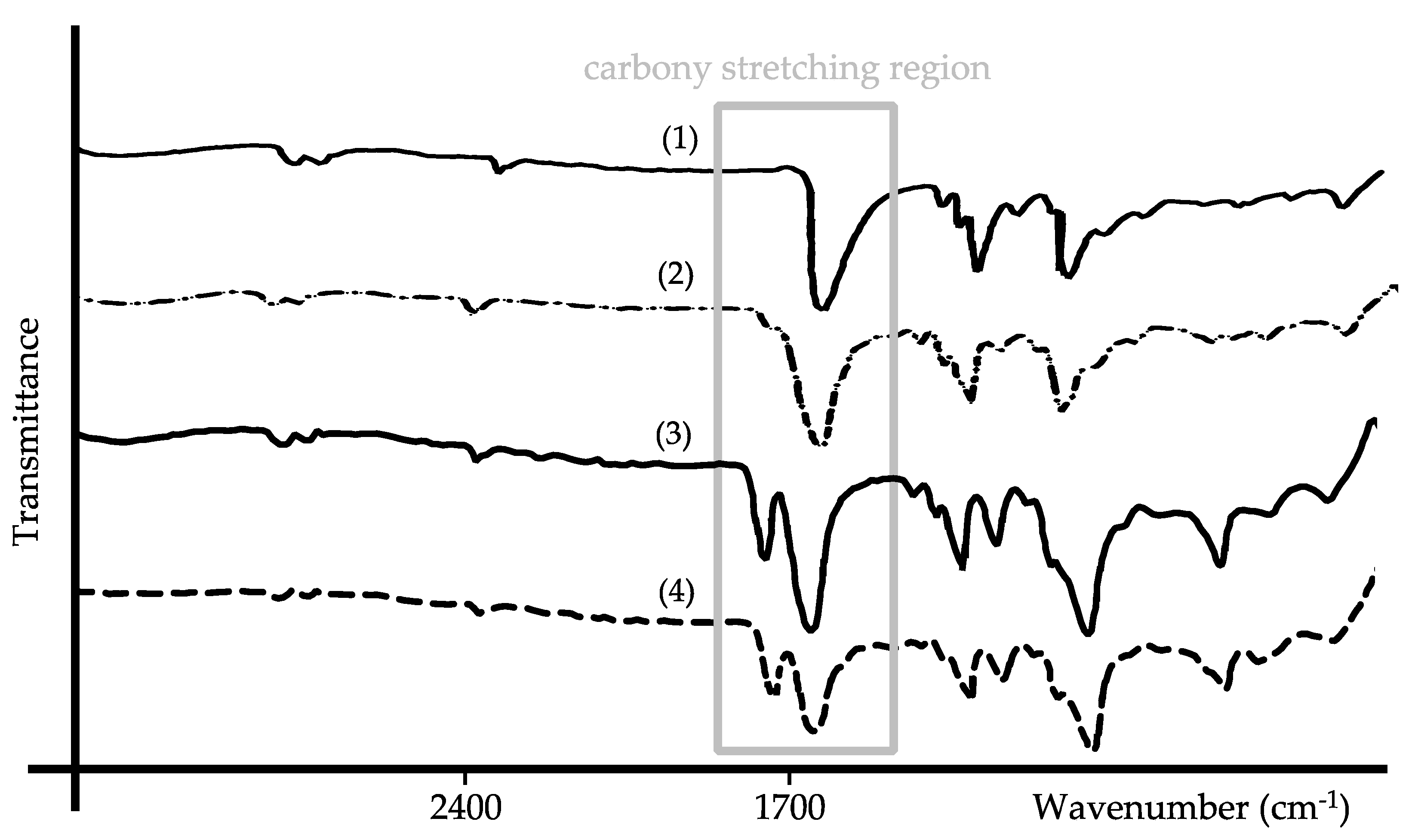
3.5. FT-IR Spectral Comparison of the Neat Polymers and CorA-ASD Formulations
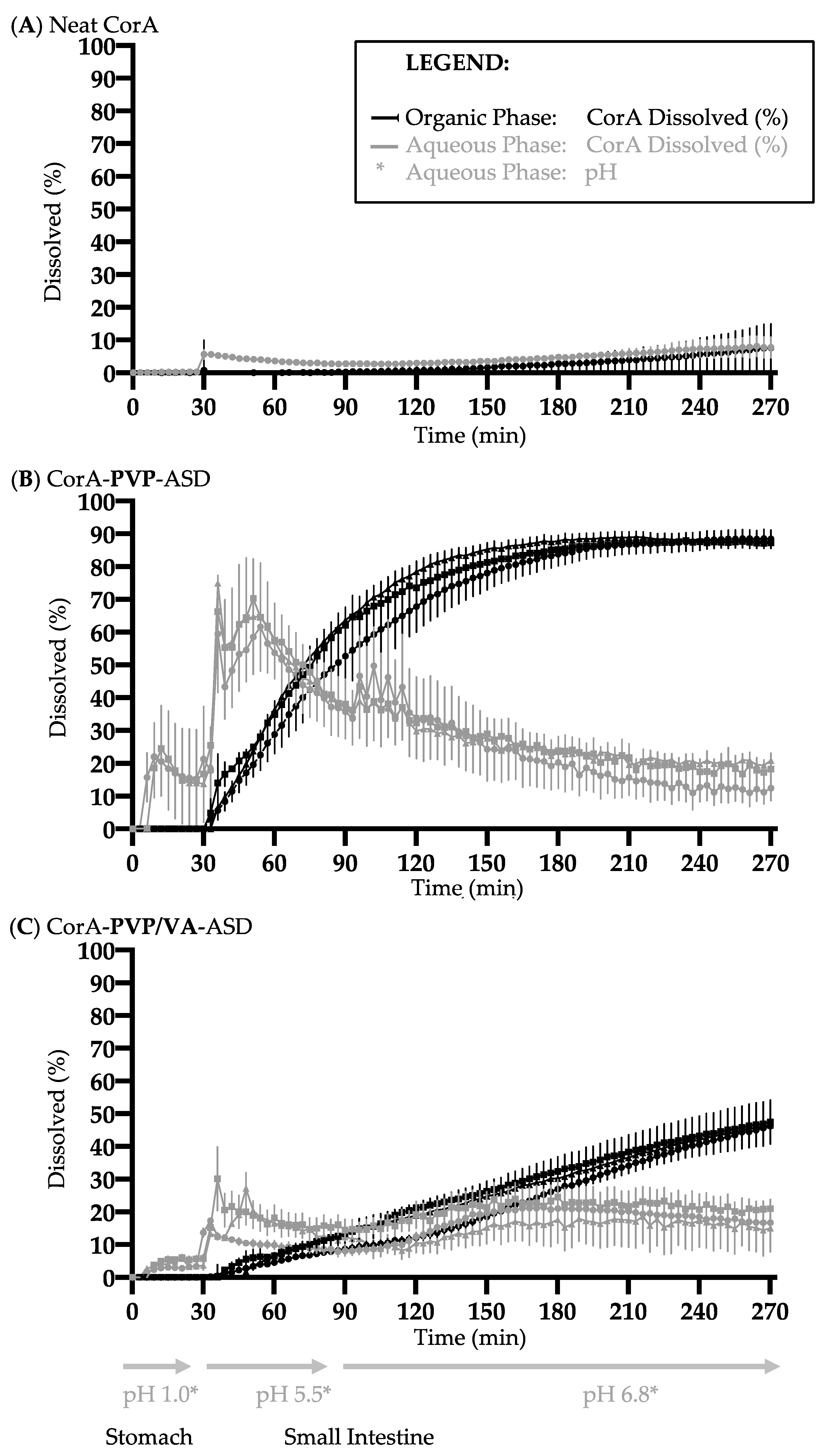
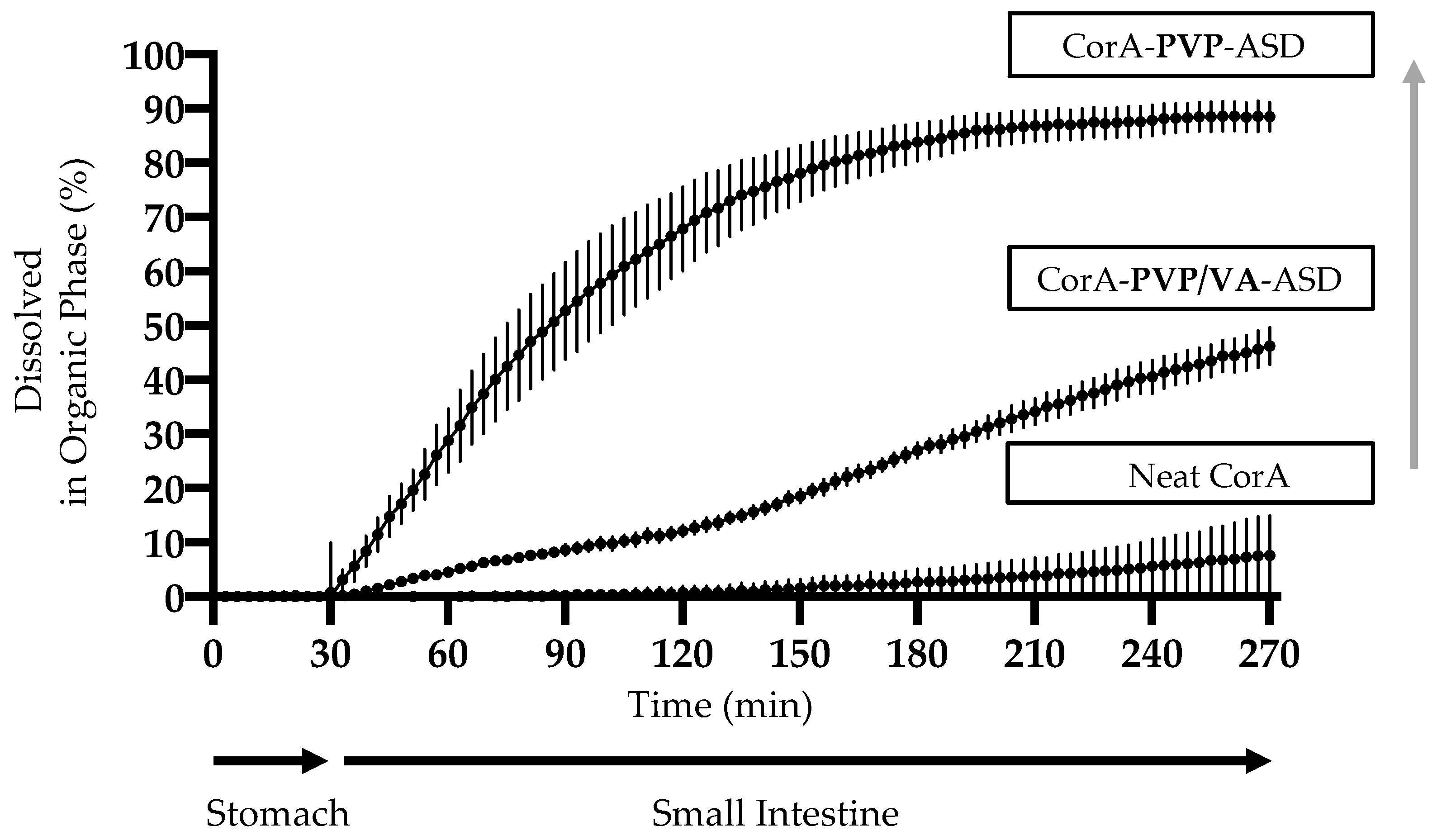
3.6. Biphasic Dissolution Results of Neat CorA and CorA-ASD Formulations via BiPHa+
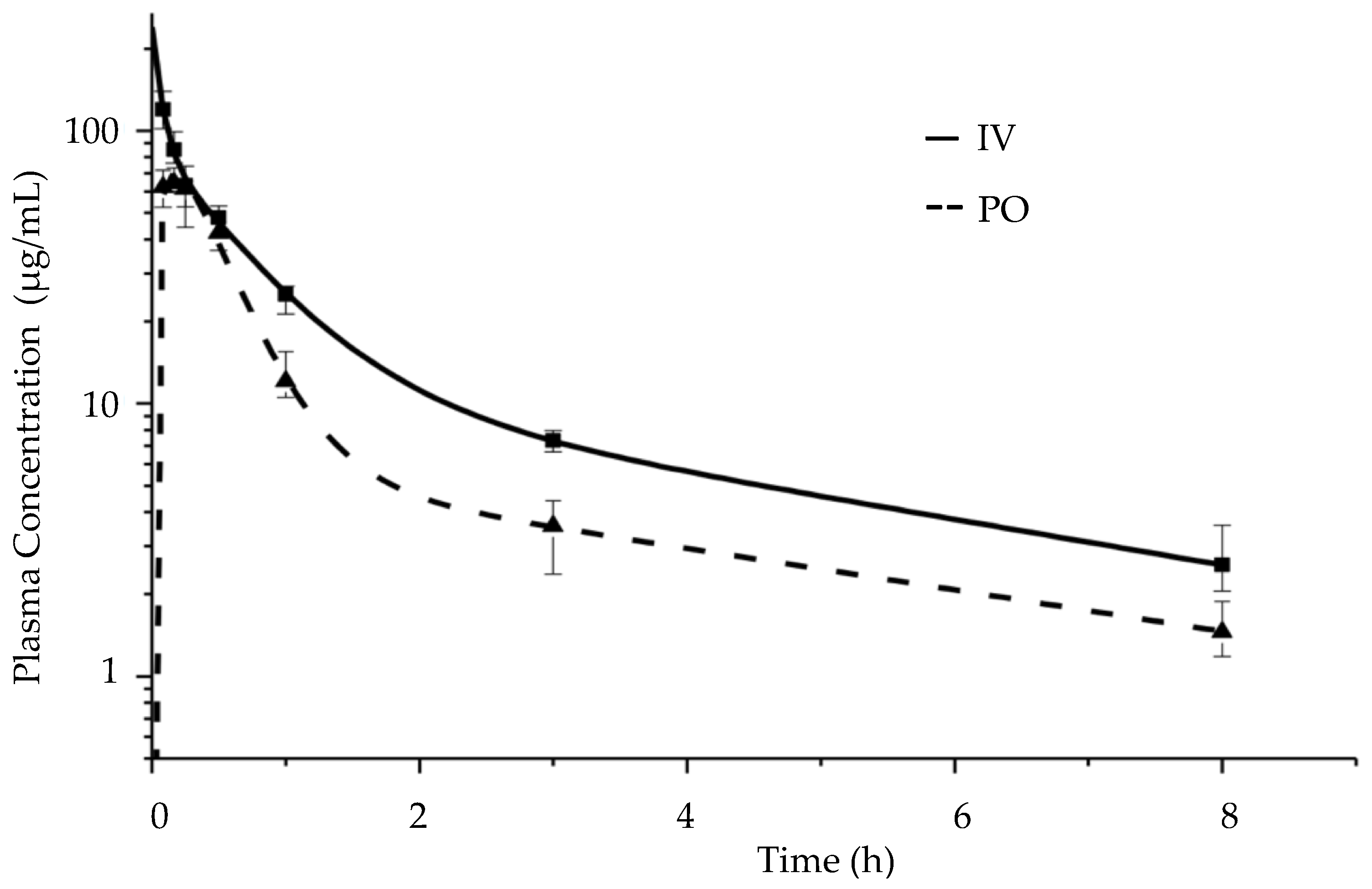
3.7. Pharmacokinetic Study Results in BALB/c Mice
4. Discussion
4.1. Increasing the Stability and Solubility of CorA: Choice of a Suitable Solid Formulation Principle and Technique for the API
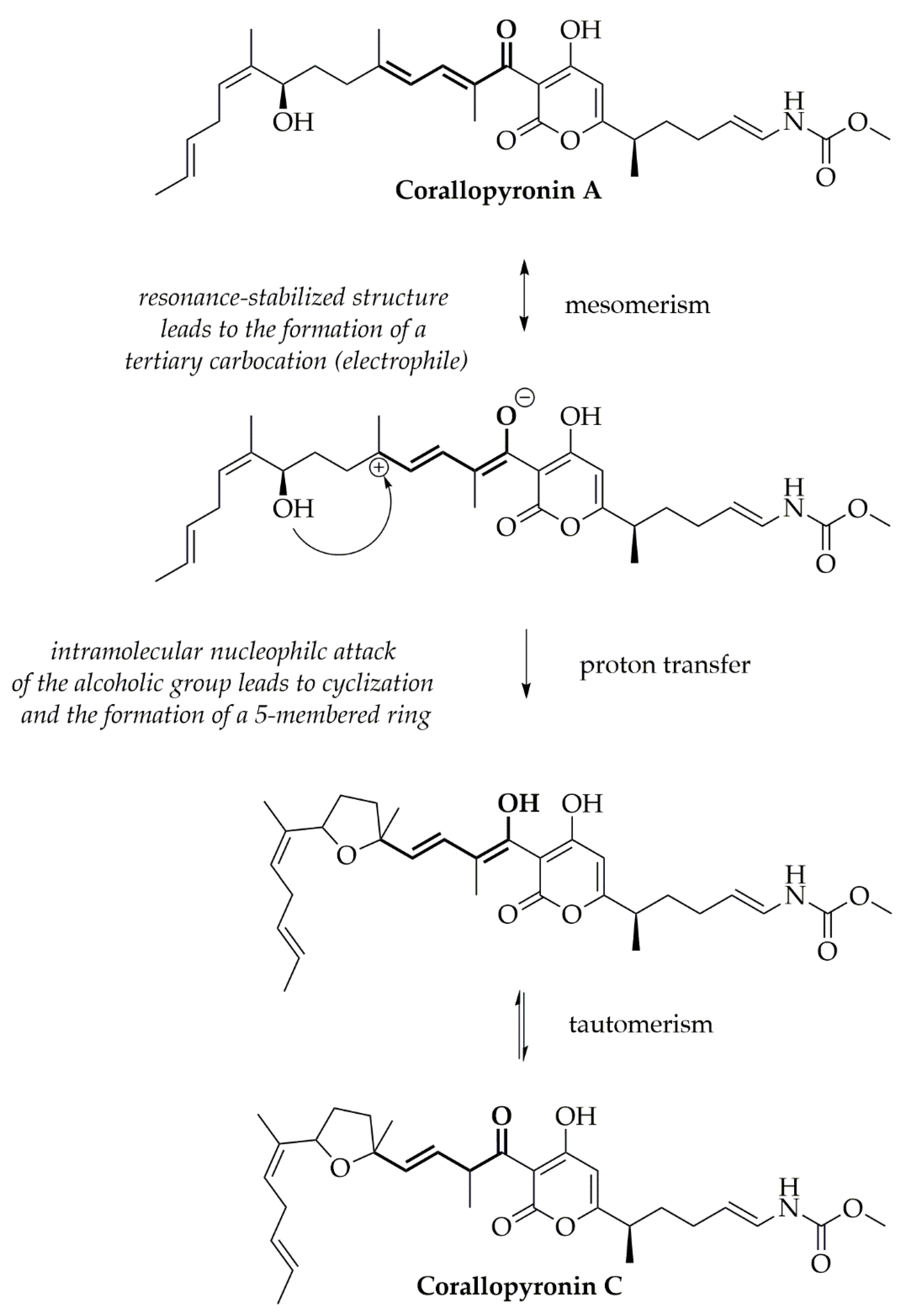
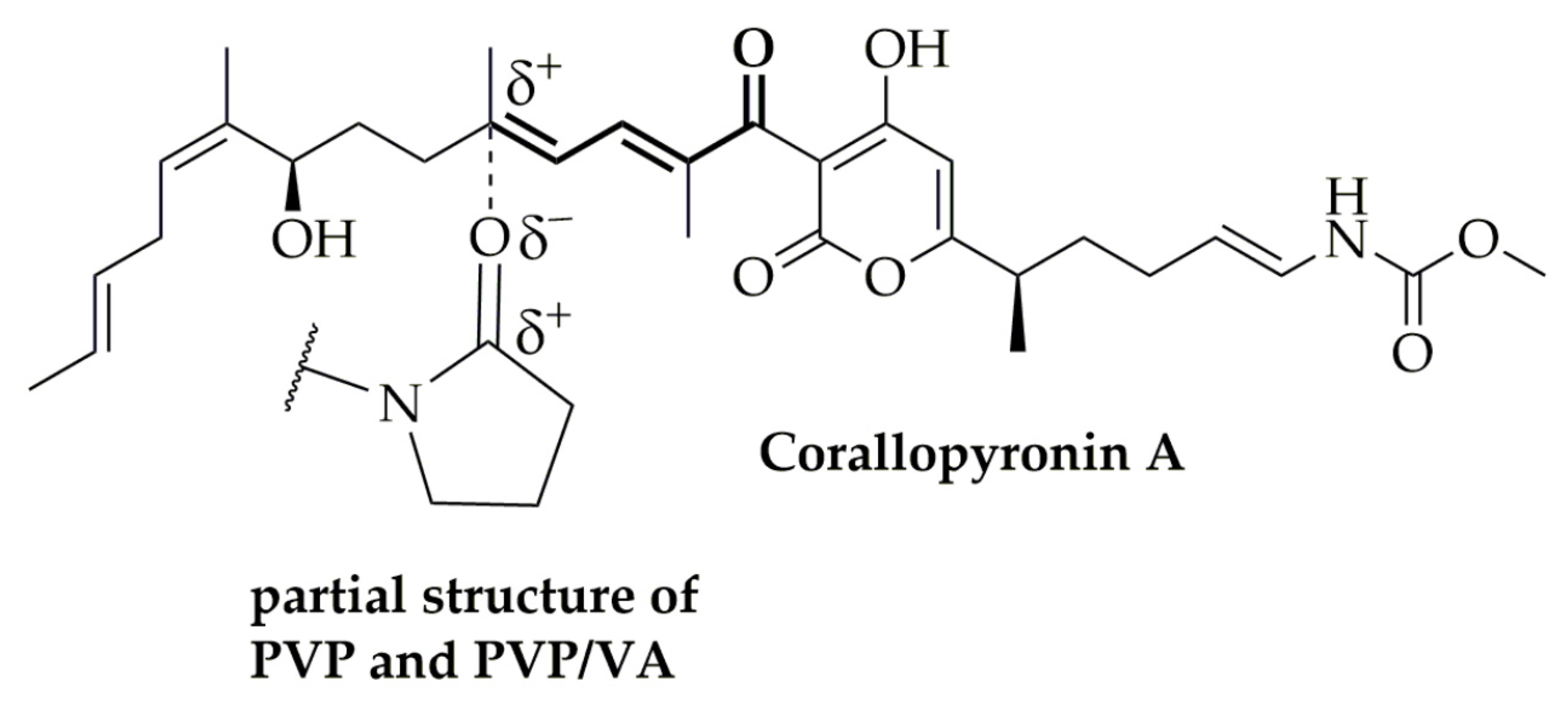
4.2. Stability Analysis and Mechanism of the CorA-ASD Formulations: Via HPLC-DAD, DSC, FT-IR, and Biphasic Dissolution
4.3. Dissolution and Solubility Analysis and Mechanism of the CorA-ASD Formulations: Biphasic Dissolution Apparatus BiPHa+ and Pion’s T3 Apparatus
4.4. Pharmacokinetic Analysis of the CorA-ASD Formulation: Investigations in BALB/c Mice
5. Conclusions
6. Patents
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Time (min) | A (%) | B (%) | Time (min) | A (%) | B (%) |
|---|---|---|---|---|---|
| 0 | 70 | 30 | 17.00 | 40 | 60 |
| 1.50 | 70 | 30 | 19.50 | 40 | 60 |
| 5.00 | 60 | 40 | 23.00 | 30 | 70 |
| 7.50 | 60 | 40 | 25.50 | 30 | 70 |
| 11.00 | 50 | 50 | 29.00 | 20 | 80 |
| 13.50 | 50 | 50 | 30.00 | 20 | 80 |
Appendix B
References
- Hesterkamp, T. Antibiotics Clinical Development and Pipeline. Curr. Top. Microbiol. 2016, 398, 447–474. [Google Scholar] [CrossRef]
- Abadi, A.T.B.; Rizvanov, A.A.; Haertlé, T.; Blatt, N.L. World Health Organization Report: Current Crisis of Antibiotic Resistance. BioNanoScience 2019, 9, 778–788. [Google Scholar] [CrossRef]
- Nielsen, T.B.; Brass, E.P.; Gilbert, D.N.; Bartlett, J.G.; Spellberg, B. Sustainable Discovery and Development of Antibiotics—Is a Nonprofit Approach the Future? N. Engl. J. Med. 2019, 381, 503–505. [Google Scholar] [CrossRef]
- Schäberle, T.F.; Hack, I.M. Overcoming the Current Deadlock in Antibiotic Research. Trends Microbiol. 2014, 22, 165–167. [Google Scholar] [CrossRef]
- Beyer, P.; Paulin, S. The Antibacterial Research and Development Pipeline Needs Urgent Solutions. ACS Infect. Dis. 2020, 6, 1289–1291. [Google Scholar] [CrossRef]
- Paulin, S.; Beyer, P. 2019 Antibacterial Agents in Clinical Development: An Analysis of the Antibacterial Clinical Development Pipeline; WHO: Geneva, Switzerland, 2019; ISBN 978-92-4-000019-3. [Google Scholar]
- Jansen, R.; Höfle, G.; Irschik, H.; Reichenbach, H. Antibiotika aus Gleitenden Bakterien, XXIV. Corallopyronin A, B und C—Drei neue Antibiotika aus Corallococcus coralloides Cc c127 (Myxobacterales). Liebigs Ann. 1985, 4, 822–836. [Google Scholar] [CrossRef]
- Schiefer, A.; Schmitz, A.; Schäberle, T.F.; Specht, S.; Lämmer, C.; Johnston, K.L.; Vassylyev, D.G.; König, G.M.; Hoerauf, A.; Pfarr, K. Corallopyronin A Specifically Targets and Depletes Essential Obligate Wolbachia Endobacteria From Filarial Nematodes In Vivo. J. Infect. Dis. 2012, 206, 249–257. [Google Scholar] [CrossRef]
- Lohr, F.; Jenniches, I.; Frizler, M.; Meehan, M.J.; Sylvester, M.; Schmitz, A.; Gütschow, M.; Dorrestein, P.C.; König, G.M.; Schäberle, T.F. α,β → β,γ Double Bond Migration in Corallopyronin A Biosynthesis. Chem. Sci. 2013, 4, 4175–4180. [Google Scholar] [CrossRef]
- Schmitz, A.; Kehraus, S.; Schäberle, T.F.; Neu, E.; Almeida, C.; Roth, M.; König, G.M. Corallorazines from the Myxobacterium Corallococcus coralloides. J. Nat. Prod. 2014, 77, 159–163. [Google Scholar] [CrossRef]
- Schäberle, T.F.; Schiefer, A.; Schmitz, A.; König, G.M.; Hoerauf, A.; Pfarr, K. Corallopyronin A—A Promising Antibiotic for Treatment of Filariasis. Int. J. Med. Microbiol. 2014, 304, 72–78. [Google Scholar] [CrossRef]
- Schäberle, T.F.; Schmitz, A.; Zocher, G.; Schiefer, A.; Kehraus, S.; Neu, E.; Roth, M.; Vassylyev, D.G.; Stehle, T.; Bierbaum, G.; et al. Insights into Structure—Activity Relationships of Bacterial RNA Polymerase Inhibiting Corallopyronin Derivatives. J. Nat. Prod. 2015, 78, 2505–2509. [Google Scholar] [CrossRef] [PubMed]
- Pfarr, K.M.; Hoerauf, A.; Koenig, G.M.; Specht, S.; Schiefer, A.; Schaeberle, T.F.; Schmitz, A.; Kehraus, S. Compounds for Use in the Treatment of Filariasis. U.S. Patent 9,687,470, 27 June 2017. [Google Scholar]
- Kock, F.; Hauptmann, M.; Osterloh, A.; Schäberle, T.F.; Poppert, S.; Frickmann, H.; Menzel, K.-D.; Peschel, G.; Pfarr, K.; Schiefer, A.; et al. Orientia tsutsugamushi Is Highly Susceptible to the RNA Polymerase Switch Region Inhibitor Corallopyronin A In Vitro and In Vivo. Antimicrob. Agents Chemother. 2018, 62, e01732-17. [Google Scholar] [CrossRef] [PubMed]
- Loeper, N.; Graspeuntner, S.; Ledig, S.; Kaufhold, I.; Hoellen, F.; Schiefer, A.; Henrichfreise, B.; Pfarr, K.; Hoerauf, A.; Shima, K.; et al. Elaborations on Corallopyronin A as a Novel Treatment Strategy Against Genital Chlamydial Infections. Front. Microbiol. 2019, 10, 943. [Google Scholar] [CrossRef] [PubMed]
- Schiefer, A.; Hübner, M.P.; Krome, A.K.; Lämmer, C.; Neufeld, H.; Charverra-Munoz, L.; Jansen, R.; Kehraus, S.; König, G.M.; Pogorevc, D.; et al. Corallopyronin A for Short-Course Anti-Wolbachial, Macrofilaricidal Treatment of Filarial Infections. PLoS Negl. Trop. Dis. 2020, in press. [Google Scholar]
- Nieto, K.; Mallery, S.R.; Schwendeman, S.P. Microencapsulation of Amorphous Solid Dispersions of Fenretinide Enhances Drug Solubility and Release from PLGA in vitro and in vivo. Int. J. Pharm. 2020, 119475. [Google Scholar] [CrossRef]
- Ting, J.M.; Porter, W.W.; Mecca, J.M.; Bates, F.S.; Reineke, T.M. Advances in Polymer Design for Enhancing Oral Drug Solubility and Delivery. Bioconjug. Chem. 2018, 29, 939–952. [Google Scholar] [CrossRef]
- Shah, N.; Sandhu, H.; Choi, D.S.; Chokshi, H.; Malick, A.W. Advances in delivery science and technology. Controlled Release Society Amorphous Solid Dispersions: Theory and Practice; Springer: New York, NY, USA, 2014; ISBN 978-1-4939-1597-2. [Google Scholar]
- Van den Mooter, G. The Use of Amorphous Solid Dispersions: A Formulation Strategy to Overcome Poor Solubility and Dissolution Rate. Drug Discov. Today Technol. 2012, 9, e79–e85. [Google Scholar] [CrossRef]
- Hancock, B.C.; Shamblin, S.L.; Zografi, G. Molecular Mobility of Amorphous Pharmaceutical Solids Below their Glass Transition Temperatures. Pharm. Res. 1995, 12, 799–806. [Google Scholar] [CrossRef]
- Li, B.; Konecke, S.; Harich, K.; Wegiel, L.; Taylor, L.S.; Edgar, K.J. Solid Dispersion of Quercetin in Cellulose Derivative Matrices Influences Both Solubility and Stability. Carbohydr. Polym. 2013, 92, 2033–2040. [Google Scholar] [CrossRef]
- Simões, M.F.; Nogueira, B.A.; Tabanez, A.M.; Fausto, R.; Pinto, R.M.A.; Simões, S. Enhanced Solid-State Stability of Amorphous Ibrutinib Formulations Prepared by Hot-Melt Extrusion. Int. J. Pharm. 2020, 579, 119156. [Google Scholar] [CrossRef]
- Denninger, A.; Westedt, U.; Rosenberg, J.; Wagner, K.G. A Rational Design of a Biphasic DissolutionSetup—Modelling of Biorelevant Kinetics for a Ritonavir Hot-Melt Extruded Amorphous Solid Dispersion. Pharmaceutics 2020, 12, 237. [Google Scholar] [CrossRef]
- Pogorevc, D.; Panter, F.; Schillinger, C.; Jansen, R.; Wenzel, S.C.; Müller, R. Production Optimization and Biosynthesis Revision of Corallopyronin A, a Potent Anti-Filarial Antibiotic. Metabol. Eng. 2019, 55, 201–211. [Google Scholar] [CrossRef]
- Takács-Novák, K.; Avdeef, A. Interlaboratory Study of Log P Determination by Shake-Slask and Potentiometric Methods. J. Pharm. Biomed. Anal. 1996, 14, 1405–1413. [Google Scholar] [CrossRef]
- Stuart, M.; Box, K. Chasing Equilibrium: Measuring the Intrinsic Solubility of Weak Acids and Bases. Anal. Chem. 2005, 77, 983–990. [Google Scholar] [CrossRef]
- Box, K.; Comer, J. Using Measured pKa, LogP and Solubility to Investigate Supersaturation and Predict BCS Class. Curr. Drug Metab. 2008, 9, 869–878. [Google Scholar] [CrossRef]
- Box, K.; Comer, J.; Gravestock, T.; Stuart, M. New Ideas about the Solubility of Drugs. Chem. Biodivers. 2009, 6, 1767–1788. [Google Scholar] [CrossRef]
- Schönherr, D.; Wollatz, U.; Haznar-Garbacz, D.; Hanke, U.; Box, K.J.; Taylor, R.; Ruiz, R.; Beato, S.; Becker, D.; Weitschies, W. Characterisation of Selected Active Agents Regarding PKa Values, Solubility Concentrations and PH Profiles by SiriusT3. Eur. J. Pharm. Biopharm. 2015, 92, 155–170. [Google Scholar] [CrossRef]
- Rowe, R.C. Handbook of Pharmaceutical Excipients, 6th ed.; APhA, (PhP) Pharmaceutical Press: London, UK, 2009; ISBN 978-0-85369-792-3. [Google Scholar]
- Niazi, S.K. Bioavailability and Bioequivalence Studies Submitted in NDAs or INDs—General Considerations. In Handbook of Pharmaceutical Manufacturing Formulations; CRC Press: Boca Raton, FL, USA, 2019; pp. 201–213. ISBN 978-1-315-10285-6. [Google Scholar]
- Taylor, L.S.; Zografi, G. Spectroscopic Characterization of Interactions Between PVP and Indomethacin in Amorphous Molecular Dispersions. Pharm. Res. 1997, 14, 1691–1698. [Google Scholar] [CrossRef]
- Hurley, D.; Carter, D.; Foong Ng, L.Y.; Davis, M.; Walker, G.M.; Lyons, J.G.; Higginbotham, C.L. An Investigation of the Inter-Molecular Interaction, Solid-State Properties and Dissolution Properties of Mixed Copovidone Hot-Melt Extruded Solid Dispersions. J. Drug Deliv. Sci. Technol. 2019, 53, 101132. [Google Scholar] [CrossRef]
- Ludden, T.M.; Beal, S.L.; Sheiner, L.B. Comparison of the Akaike Information Criterion, the Schwarz Criterion and the F Test as Guides to Model Selection. J. Pharmacokinet. Biopharm. 1994, 22, 431–445. [Google Scholar] [CrossRef]
- Yamaoka, K.; Nakagawa, T.; Uno, T. Application of Akaike’s Information Criterion (AIC) in the Evaluation of Linear Pharmacokinetic Equations. J. Pharmacokinet. Biopharm. 1978, 6, 165–175. [Google Scholar] [CrossRef]
- Schwarz, G. Estimating the Dimension of a Model. Ann. Statist. 1978, 6, 461–464. [Google Scholar] [CrossRef]
- Chiou, W.L.; Riegelman, S. Pharmaceutical Applications of Solid Dispersion Systems. J. Pharm. Sci. 1971, 60, 1281–1302. [Google Scholar] [CrossRef]
- Vig, B.; Morgen, M. Formulation, Process Development, and Scale-Up. In Developing Solid Oral Dosage Forms; Elsevier: Amsterdam, The Netherlands, 2017; pp. 793–820. ISBN 978-0-12-802447-8. [Google Scholar]
- Qian, F.; Huang, J.; Hussain, M.A. Drug–Polymer Solubility and Miscibility: Stability Consideration and Practical Challenges in Amorphous Solid Dispersion Development. J. Pharm. Sci. 2010, 99, 2941–2947. [Google Scholar] [CrossRef]
- Knopp, M.M.; Wendelboe, J.; Holm, R.; Rades, T. Effect of Amorphous Phase Separation and Crystallization on the In Vitro and In Vivo Performance of an Amorphous Solid Dispersion. Eur. J. Pharm. Biopharm. 2018, 130, 290–295. [Google Scholar] [CrossRef]
- Heigoldt, U.; Sommer, F.; Daniels, R.; Wagner, K.-G. Predicting In Vivo Absorption Behavior of Oral Modified Release Dosage Forms Containing PH-Dependent Poorly Soluble Drugs Using a Novel PH-Adjusted Biphasic In Vitro Dissolution Test. Eur. J. Pharm. Biopharm. 2010, 76, 105–111. [Google Scholar] [CrossRef]
- Frank, K.J.; Locher, K.; Zecevic, D.E.; Fleth, J.; Wagner, K.G. In Vivo Predictive Mini-Scale Dissolution for Weak Bases: Advantages of PH-Shift in Combination with an Absorptive Compartment. Eur. J. Pharm. Sci. 2014, 61, 32–39. [Google Scholar] [CrossRef]
- Locher, K.; Borghardt, J.M.; Frank, K.J.; Kloft, C.; Wagner, K.G. Evolution of a Mini-Scale Biphasic Dissolution Model: Impact of Model Parameters on Partitioning of Dissolved API and Modelling of In Vivo-Relevant Kinetics. Eur. J. Pharm. Biopharm. 2016, 105, 166–175. [Google Scholar] [CrossRef]
- Lu, Y.; Tang, N.; Lian, R.; Qi, J.; Wu, W. Understanding the Relationship Between Wettability and Dissolution of Solid Dispersion. Int. J. Pharm. 2014, 465, 25–31. [Google Scholar] [CrossRef]
- Verma, S.; Rudraraju, V.S. Wetting Kinetics: An Alternative Approach Towards Understanding the Enhanced Dissolution Rate for Amorphous Solid Dispersion of a Poorly Soluble Drug. AAPS Pharmscitech 2015, 16, 1079–1090. [Google Scholar] [CrossRef]
- Kuti, J.L. Optimizing Antimicrobial Pharmacodynamics: A Guide for your Stewardship Program. Rev. Méd. Clín. Condes 2016, 27, 615–624. [Google Scholar] [CrossRef]
| Excipients and Parameters | CorA-PVP-ASD | CorA-PVP/VA-ASD |
|---|---|---|
| CorA | 1.6022 g | 1.6008 g |
| Polymer | 6.4237 g | 6.4032 g |
| Organic Solvent (Ethanol 99%) | 52.930 g | 52.931 g |
| Theoretical Active Ingredient Content | 20% | 20% |
| Inlet Temperature | 85 °C | 85 °C |
| Outlet Temperature | 59 °C | 59 °C |
| Flow Rate | 5.6 mL/min | 4.5 mL/min |
| pH | Solubility (µg/mL) | Log D |
|---|---|---|
| 1.0 | 0000.11 | 5.42 → log P (neutral XH) |
| 2.0 | 0000.11 | 5.41 |
| 3.0 | 0000.14 | 5.31 |
| 4.0 | 0000.40 | 4.85 |
| 5.0 | 0002.98 | 3.97 |
| 6.0 | 0028.86 | 3.00 |
| 6.5 | 0091.13 | 2.52 |
| 7.0 | 0288.00 | 2.09 |
| 7.4 | 0723.20 | 1.81 |
| 8.0 | 2874.00 | 1.54 |
| API and Excipients | Tg (°C) |
|---|---|
| Neat CorA | 5 |
| CorA-PVP-ASD formulation (20% drug load) | 116 |
| Neat PVP | 160 |
| CorA-PVP/VA-ASD formulation (20% drug load) | 84 |
| Neat PVP/VA | 110 |
| Pharmacokinetic Parameters | IV Median (IQR) | PO Median (IQR) |
|---|---|---|
| AUC(0–8h) (µg·h/mL) | 115.5 (102.4–127.1) | 67.8 (60.8–71.6) |
| AUC(0-inf) (µg·h/mL) | 127.7 (110.2–149.0) | 75.9 (70.4–76.9) |
| Cmax (µg/mL) | 119.6 (103.7–136.7) | 64.3 (61.8–70.7) |
| Tmax (min) | 5 * | 10 (10–11.3) |
| Fabs (%) | 100 ** | 59 (55.1–60.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krome, A.K.; Becker, T.; Kehraus, S.; Schiefer, A.; Steinebach, C.; Aden, T.; Frohberger, S.J.; López Mármol, Á.; Kapote, D.; Jansen, R.; et al. Solubility and Stability Enhanced Oral Formulations for the Anti-Infective Corallopyronin A. Pharmaceutics 2020, 12, 1105. https://doi.org/10.3390/pharmaceutics12111105
Krome AK, Becker T, Kehraus S, Schiefer A, Steinebach C, Aden T, Frohberger SJ, López Mármol Á, Kapote D, Jansen R, et al. Solubility and Stability Enhanced Oral Formulations for the Anti-Infective Corallopyronin A. Pharmaceutics. 2020; 12(11):1105. https://doi.org/10.3390/pharmaceutics12111105
Chicago/Turabian StyleKrome, Anna K., Tim Becker, Stefan Kehraus, Andrea Schiefer, Christian Steinebach, Tilman Aden, Stefan J. Frohberger, Álvaro López Mármol, Dnyaneshwar Kapote, Rolf Jansen, and et al. 2020. "Solubility and Stability Enhanced Oral Formulations for the Anti-Infective Corallopyronin A" Pharmaceutics 12, no. 11: 1105. https://doi.org/10.3390/pharmaceutics12111105
APA StyleKrome, A. K., Becker, T., Kehraus, S., Schiefer, A., Steinebach, C., Aden, T., Frohberger, S. J., López Mármol, Á., Kapote, D., Jansen, R., Chaverra-Muñoz, L., Hübner, M. P., Pfarr, K., Hesterkamp, T., Stadler, M., Gütschow, M., König, G. M., Hoerauf, A., & Wagner, K. G. (2020). Solubility and Stability Enhanced Oral Formulations for the Anti-Infective Corallopyronin A. Pharmaceutics, 12(11), 1105. https://doi.org/10.3390/pharmaceutics12111105










