Vacuum Compression Molding as a Screening Tool to Investigate Carrier Suitability for Hot-Melt Extrusion Formulations
Abstract
1. Introduction
1.1. Hot-Melt Extrusion
1.2. Differential Scanning Calorimetry
1.3. Solvent Casting
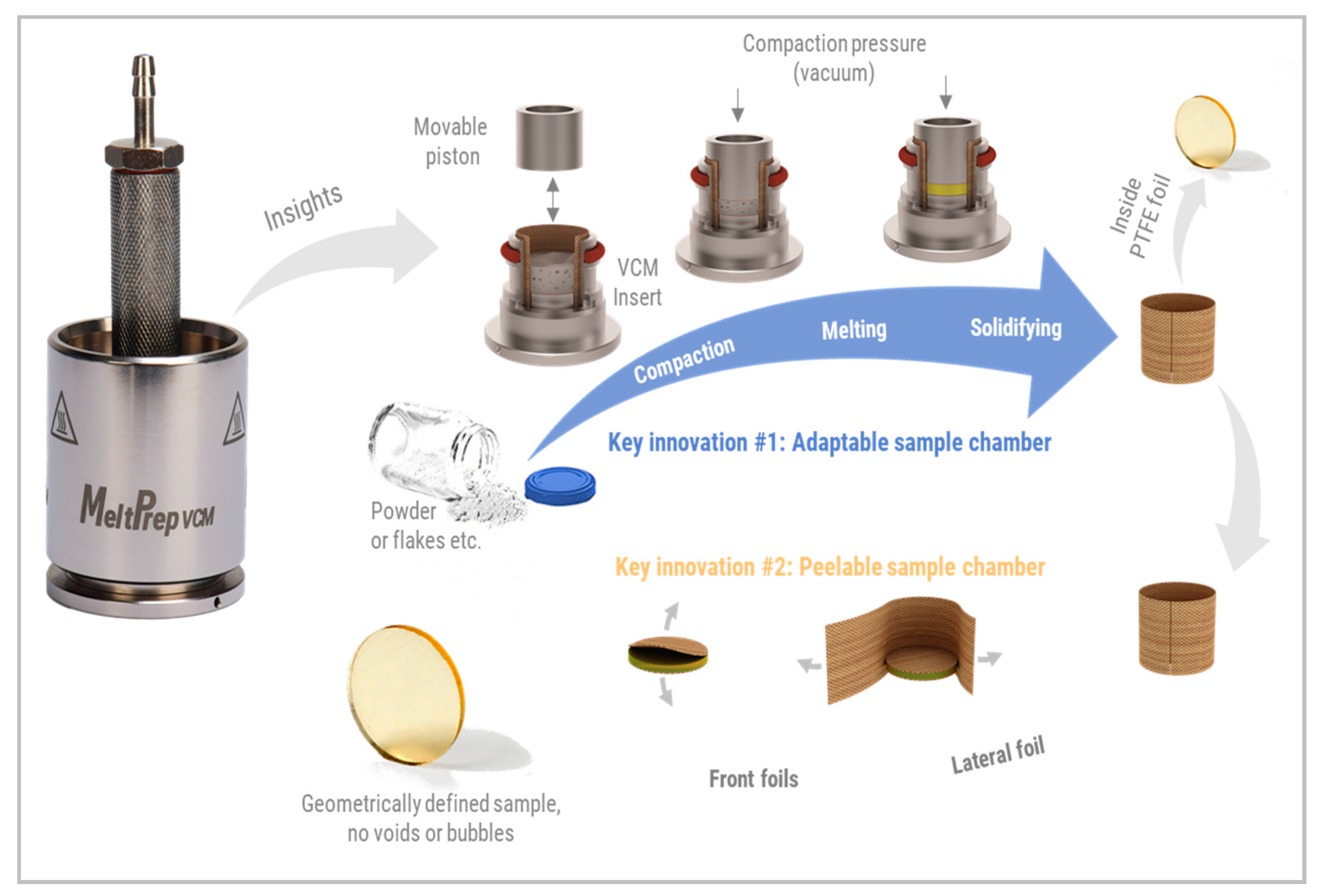
1.4. Vacuum Compression Molding
2. Materials and Methods
2.1. Materials
2.1.1. Active Pharmaceutical Ingredient (API)
2.1.2. Carriers
2.1.3. Marketed Drug Product for Reference
2.2. Processing Methods
2.2.1. Preparation of Cryo-Milled Mixtures for VCM Preparation-Preconditioning of the Samples
2.2.2. Vacuum Compression Molding (VCM)
2.2.3. Hot-Melt Extrusion (HME)
2.3. Characterization Methods
2.3.1. Physical Characterization of the VCM Samples
2.3.2. Differential Scanning Calorimetry (DSC)
2.3.3. PXRD Analysis
2.3.4. Dissolution
3. Results
3.1. Vacuum Compression Molding
3.2. Hot-Melt Extrusion
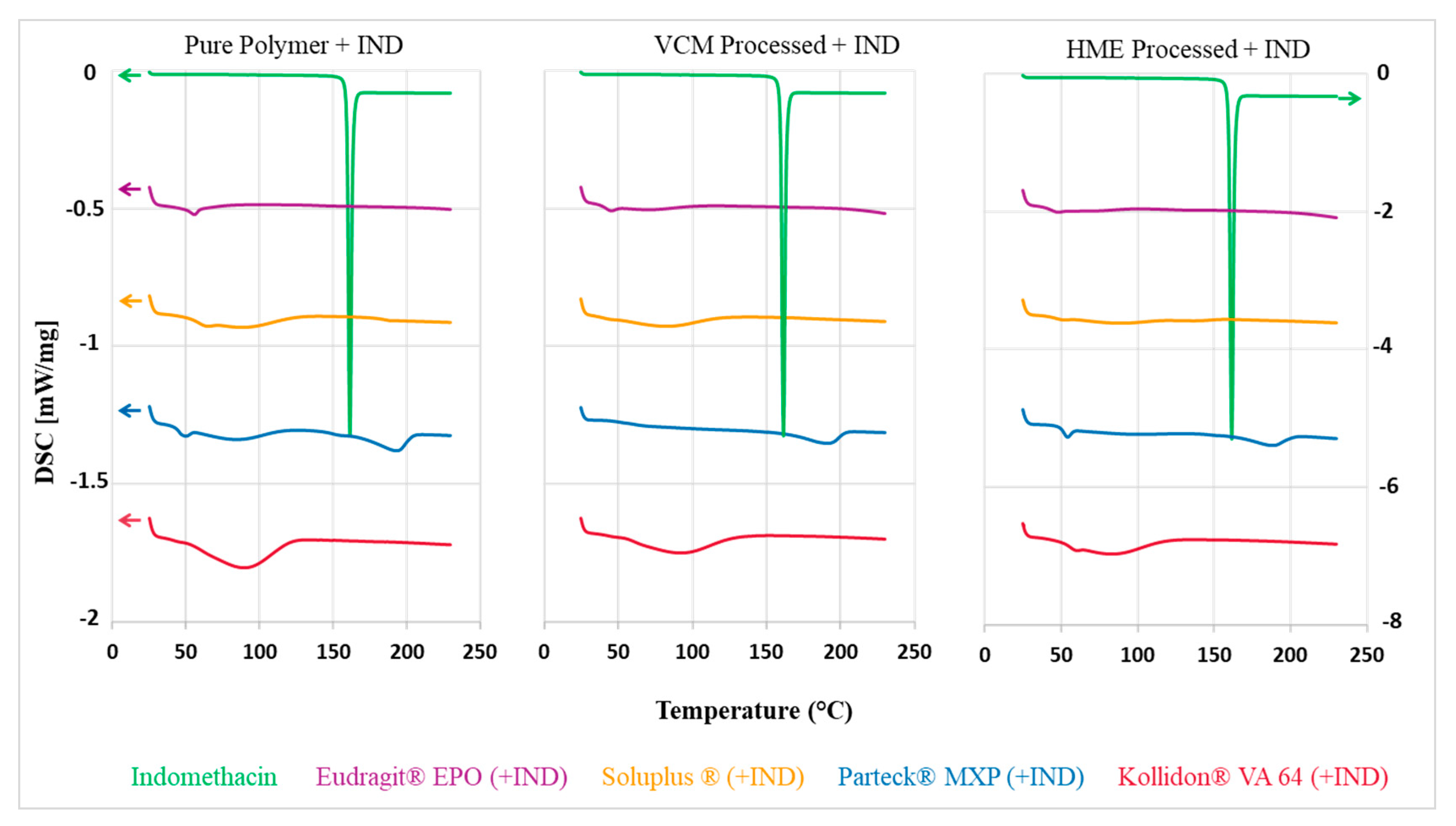
3.3. Differential Scanning Calorimetry
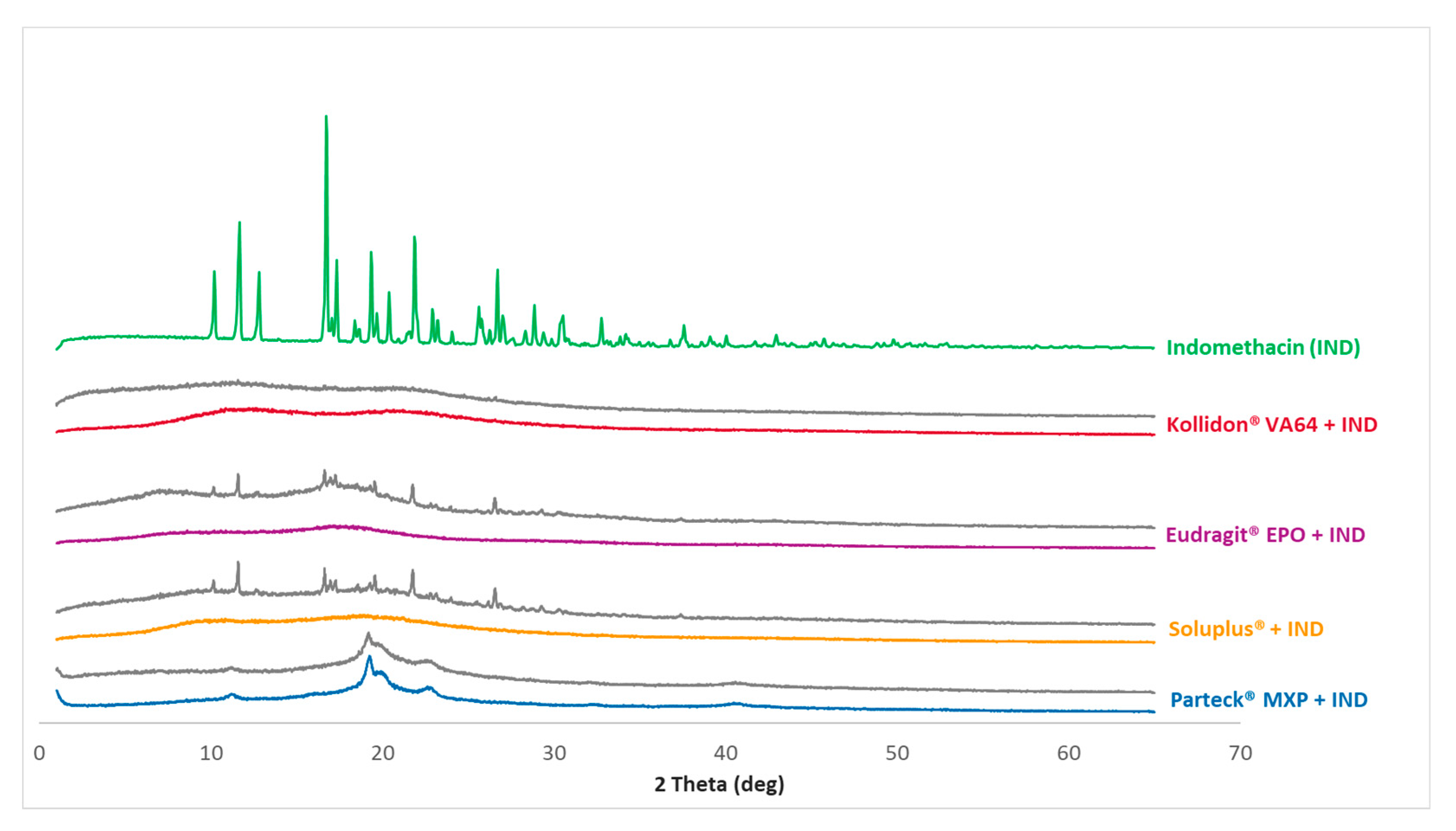
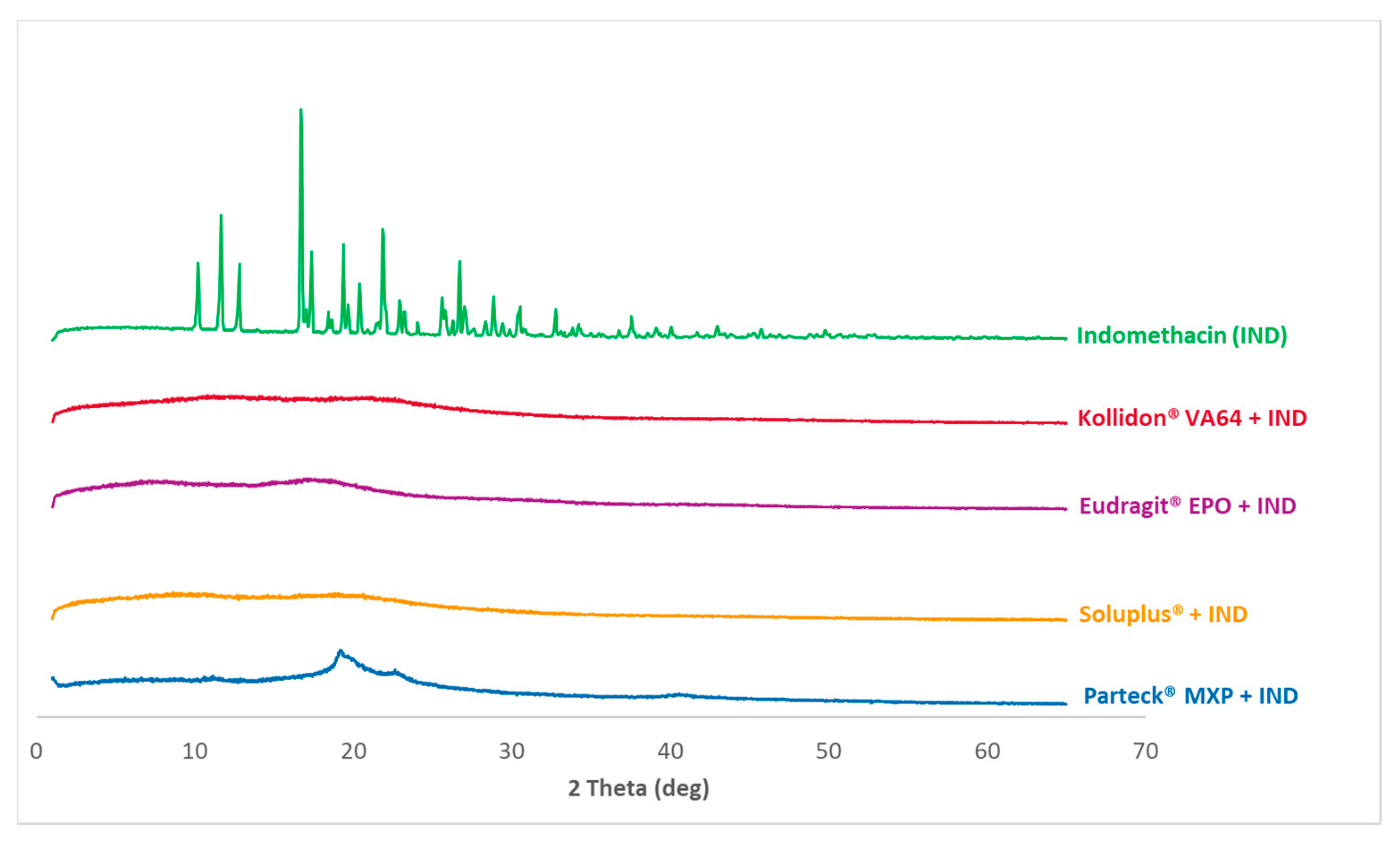
3.4. PXRD Analysis
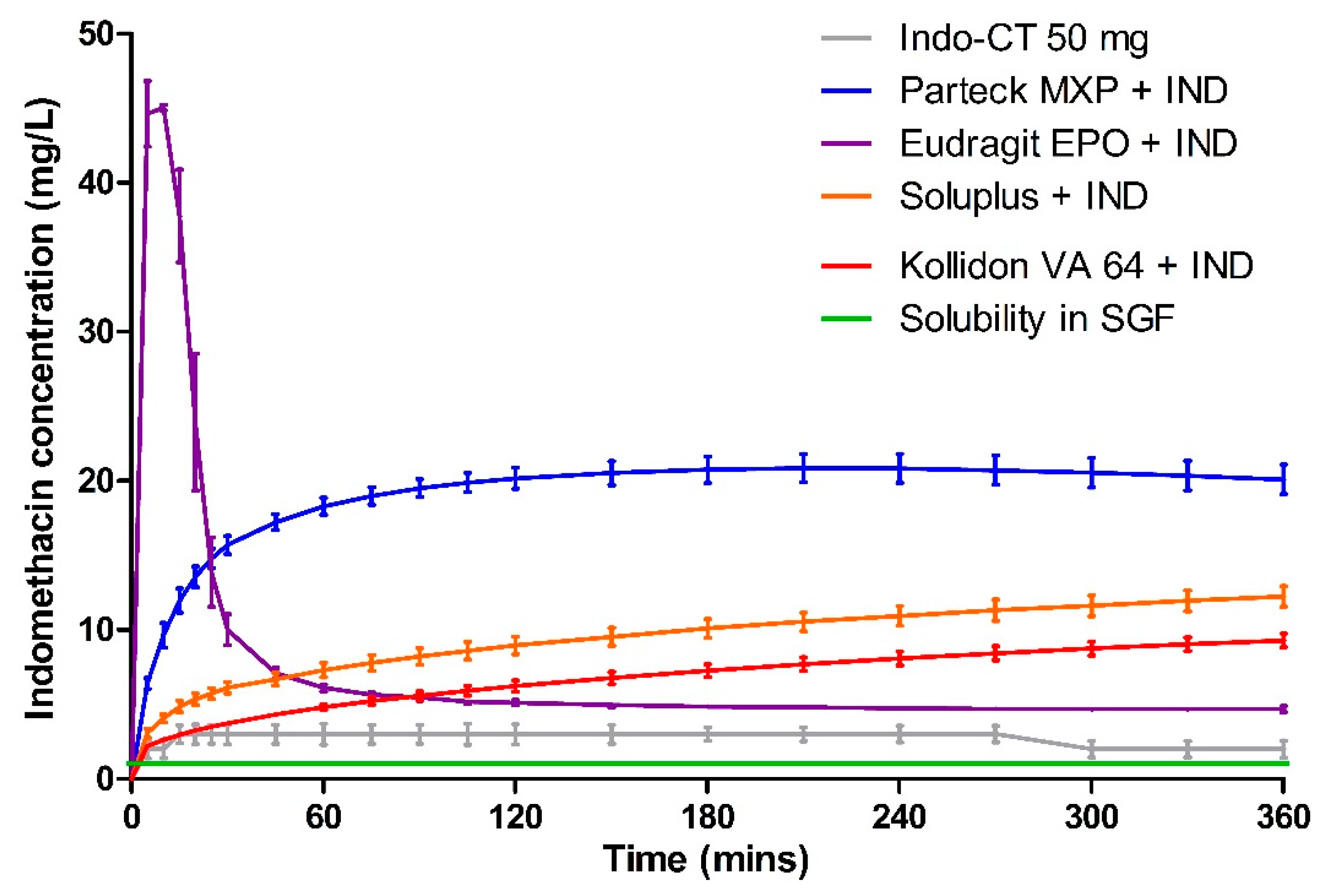
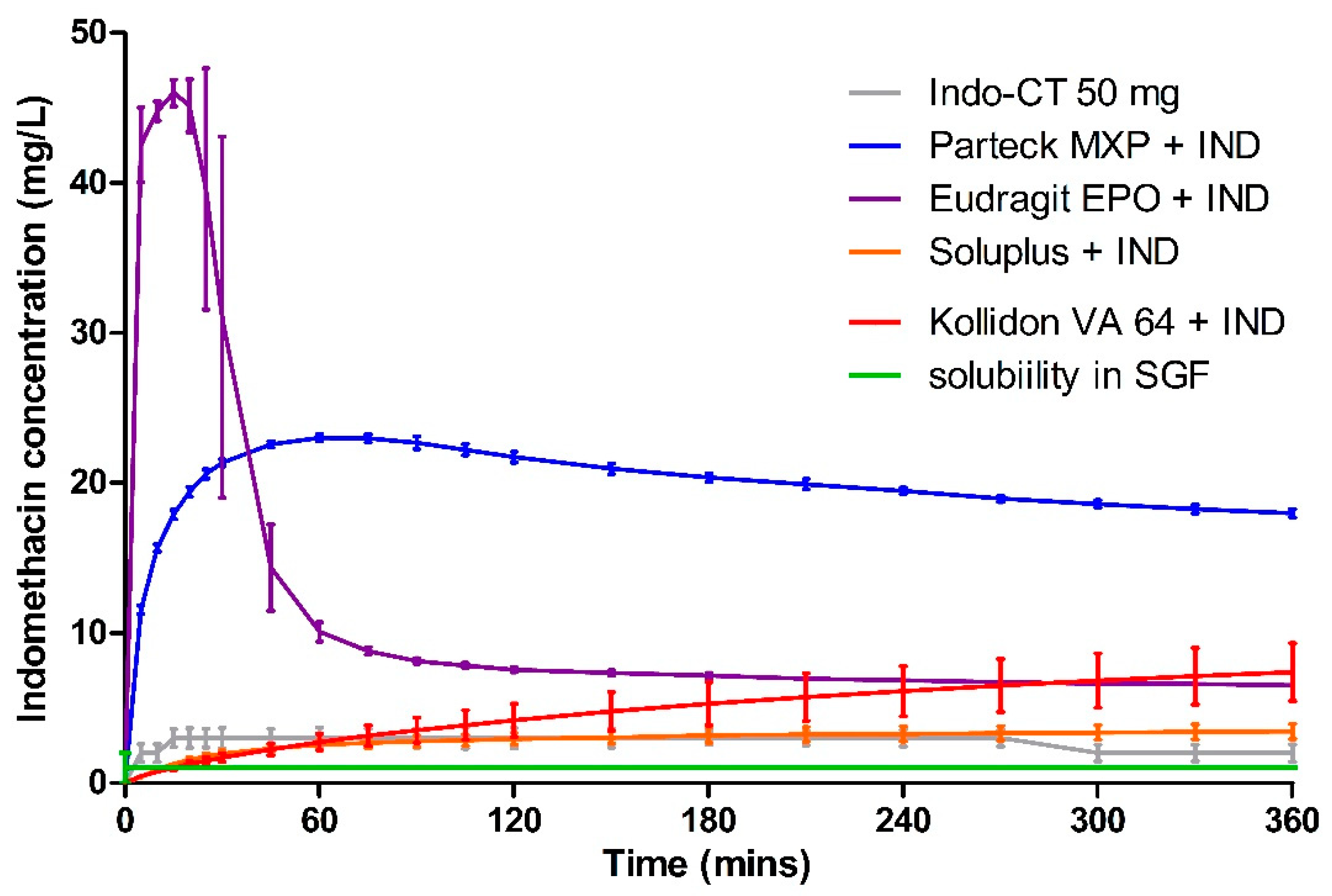
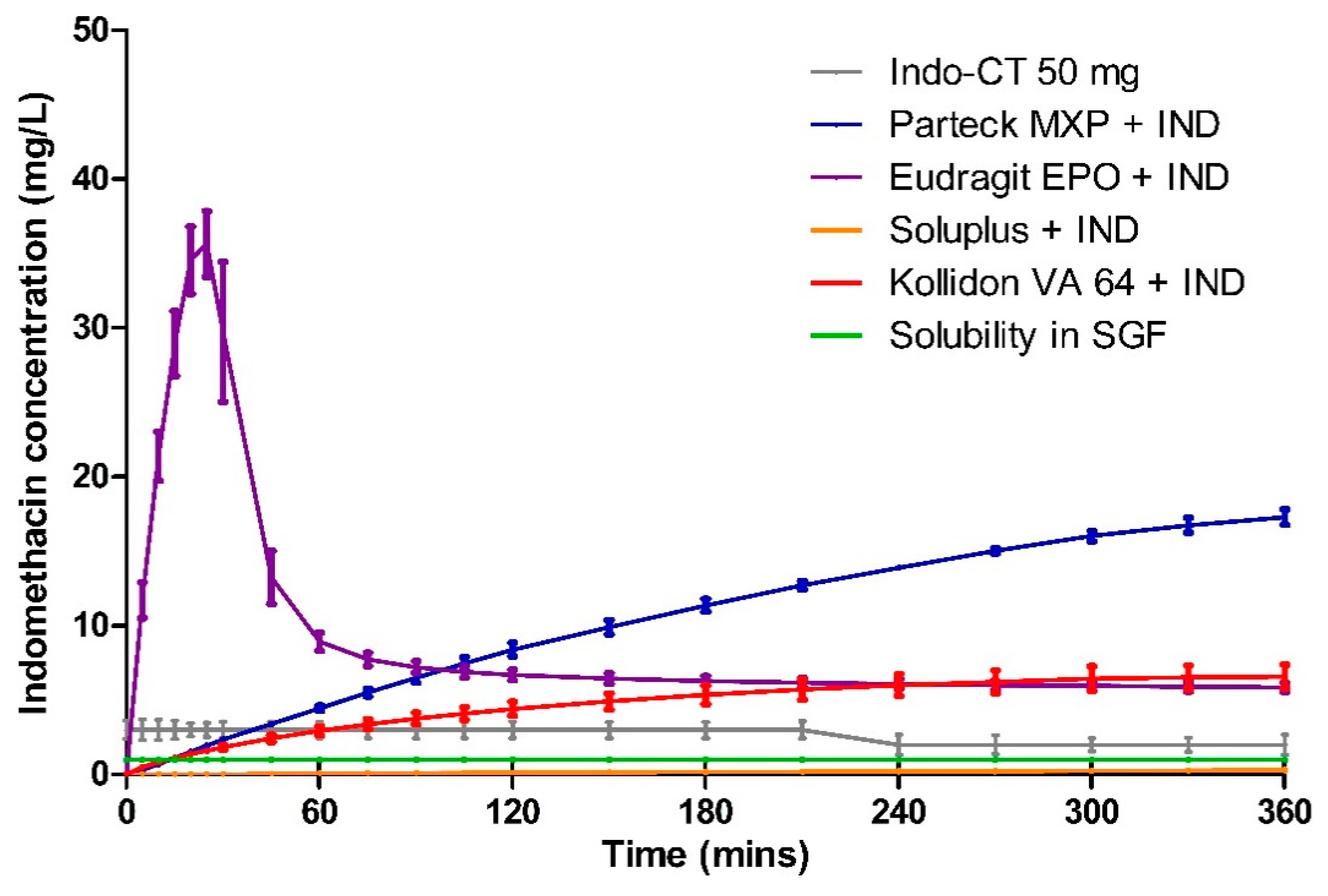
3.5. Dissolution
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- el-Egakey, M.A.; Soliva, M.; Speiser, P. Hot extruded dosage forms. I. Technology and dissolution kinetics of polymeric matrices. Pharm. Acta Helv. 1971, 46, 31–52. [Google Scholar] [PubMed]
- Repka, M.A.; Bandari, S.; Kallakunta, V.R.; Vo, A.Q.; McFall, H.; Pimparade, M.B.; Bhagurkar, A.M. Melt extrusion with poorly soluble drugs—An integrated review. Int. J. Pharm. 2018, 535, 68–85. [Google Scholar] [CrossRef] [PubMed]
- Kalepu, S.; Nekkanti, V. Insoluble drug delivery strategies: Review of recent advances and business prospects. Acta Pharm. Sin. B 2015, 5, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Charalabidis, A.; Sfouni, M.; Bergström, C.; Macheras, P. The Biopharmaceutics Classification System (BCS) and the Biopharmaceutics Drug Disposition Classification System (BDDCS): Beyond guidelines. Int. J. Pharm. 2019, 566, 264–281. [Google Scholar] [CrossRef]
- Kerns, E.H. High throughput physicochemical profiling for drug discovery. J. Pharm. Sci. 2001, 90, 1838–1858. [Google Scholar] [CrossRef]
- Zhang, P.; Shadambikar, G.; Almutairi, M.; Bandari, S.; Repka, M.A. Approaches for developing acyclovir gastro-retentive formulations using hot melt extrusion technology. J. Drug Deliv. Sci. Technol. 2020, 60, 102002. [Google Scholar] [CrossRef]
- Patil, H.; Tiwari, R.V.; Upadhye, S.B.; Vladyka, R.S.; Repka, M.A. Formulation and development of pH-independent/dependent sustained release matrix tablets of ondansetron HCl by a continuous twin-screw melt granulation process. Int. J. Pharm. 2015, 496, 33–41. [Google Scholar] [CrossRef]
- Vasconcelos, T.; Sarmento, B.; Costa, P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov. Today 2007, 12, 1068–1075. [Google Scholar] [CrossRef]
- Crowley, M.M.; Zhang, F.; Repka, M.A.; Thumma, S.; Upadhye, S.B.; Kumar Battu, S.; McGinity, J.W.; Martin, C. Pharmaceutical applications of hot-melt extrusion: Part I. Drug Dev. Ind. Pharm. 2007, 33, 909–926. [Google Scholar] [CrossRef]
- Muehlenfeld, C.; Thommes, M. Miniaturization in Pharmaceutical Extrusion Technology: Feeding as a Challenge of Downscaling. AAPS PharmSciTech 2012, 13, 94–100. [Google Scholar] [CrossRef][Green Version]
- Zecevic, D.E.; Wagner, K.G. Rational development of solid dispersions via hot-melt extrusion using screening, material characterization, and numeric simulation tools. J. Pharm. Sci. 2013, 102, 2297–2310. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.V.; Patil, H.; Repka, M.A. Contribution of hot-melt extrusion technology to advance drug delivery in the 21st century. Expert Opin. Drug Deliv. 2016, 13, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.M.; Dudhedia, M.S.; Zimny, E. Hot Melt Extrusion: Development of an Amorphous Solid Dispersion for an Insoluble Drug from Mini-scale to Clinical Scale. AAPS PharmSciTech 2016, 17, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Cossé, A.; König, C.; Lamprecht, A.; Wagner, K.G. Hot Melt Extrusion for Sustained Protein Release: Matrix Erosion and In Vitro Release of PLGA-Based Implants. AAPS PharmSciTech 2017, 18, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Sakai, T.; Thommes, M. Investigation into mixing capability and solid dispersion preparation using the DSM Xplore Pharma Micro Extruder. J. Pharm. Pharmacol. 2014, 66, 218–231. [Google Scholar] [CrossRef]
- Rask, M.B.; Knopp, M.M.; Olesen, N.E.; Holm, R.; Rades, T. Comparison of two DSC-based methods to predict drug-polymer solubility. Int. J. Pharm. 2018, 540, 98–105. [Google Scholar] [CrossRef]
- Lin, D.; Huang, Y. A thermal analysis method to predict the complete phase diagram of drug–polymer solid dispersions. Int. J. Pharm. 2010, 399, 109–115. [Google Scholar] [CrossRef]
- Knopp, M.M.; Tajber, L.; Tian, Y.; Olesen, N.E.; Jones, D.S.; Kozyra, A.; Löbmann, K.; Paluch, K.; Brennan, C.M.; Holm, R.; et al. Comparative Study of Different Methods for the Prediction of Drug–Polymer Solubility. Mol. Pharm. 2015, 12, 3408–3419. [Google Scholar] [CrossRef]
- Kyeremateng, S.O.; Pudlas, M.; Woehrle, G.H. A Fast and Reliable Empirical Approach for Estimating Solubility of Crystalline Drugs in Polymers for Hot Melt Extrusion Formulations. J. Pharm. Sci. 2014, 103, 2847–2858. [Google Scholar] [CrossRef]
- Mohan, R.; Lorenz, H.; Myerson, A.S. Solubility Measurement Using Differential Scanning Calorimetry. Ind. Eng. Chem. Res. 2002, 41, 4854–4862. [Google Scholar] [CrossRef]
- Lauer, M.E.; Maurer, R.; De Paepe, A.T.; Stillhart, C.; Jacob, L.; James, R.; Kojima, Y.; Rietmann, R.; Kissling, T.; Van den Ende, J.A.; et al. A Miniaturized Extruder to Prototype Amorphous Solid Dispersions: Selection of Plasticizers for Hot Melt Extrusion. Pharmaceutics 2018, 10, 58. [Google Scholar] [CrossRef]
- Auch, C.; Harms, M.; Mäder, K. Melt-based screening method with improved predictability regarding polymer selection for amorphous solid dispersions. Eur. J. Pharm. Sci. 2018, 124, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Chiang, P.-C.; Ran, Y.; Chou, K.-J.; Cui, Y.; Sambrone, A.; Chan, C.; Hart, R. Evaluation of Drug Load and Polymer by Using a 96-Well Plate Vacuum Dry System for Amorphous Solid Dispersion Drug Delivery. AAPS PharmSciTech 2012, 13, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Treffer, D.; Wahl, P.R.; Hörmann, T.R.; Markl, D.; Schrank, S.; Jones, I.; Cruise, P.; Mürb, R.-K.; Koscher, G.; Roblegg, E. In-line implementation of an image-based particle size measurement tool to monitor hot-melt extruded pellets. Int. J. Pharm. 2014, 466, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Macfhionnghaile, P.; Hu, Y.; Gniado, K.; Curran, S.; Mcardle, P.; Erxleben, A. Effects of Ball-Milling and Cryomilling on Sulfamerazine Polymorphs: A Quantitative Study. J. Pharm. Sci. 2014, 103, 1766–1778. [Google Scholar] [CrossRef]
- Eder, S.; Beretta, M.; Witschnigg, A.; Koutsamanis, I.; Eggenreich, K.; Khinast, J.G.; Koscher, G.; Paudel, A.; Nickisch, K.; Friedrich, M.; et al. Establishment of a Molding Procedure to Facilitate Formulation Development for Co-extrudates. AAPS PharmSciTech 2017, 18, 2971–2976. [Google Scholar] [CrossRef]
- Isabell, I.; GmbH, G. Lean and Efficent Development of a Pseudoephedrine Formulation Resistant to Conversion to Meth. In Proceedings of the 3rd European Conference on Pharmaceutics, Bologna, Italy, 25–26 March 2019. [Google Scholar]
- Evans, R.C.; Bochmann, E.S.; Kyeremateng, S.O.; Gryczke, A.; Wagner, K.G. Holistic QbD approach for hot-melt extrusion process design space evaluation: Linking materials science, experimentation and process modeling. Eur. J. Pharm. Biopharm. 2019, 141, 149–160. [Google Scholar] [CrossRef]
- Narasimhan, B. Mathematical models describing polymer dissolution: Consequences for drug delivery. Adv. Drug Deliv. Rev. 2001, 48, 195–210. [Google Scholar] [CrossRef]
- Beth, A.; Miller-Chou, J.L.K. A review of polymer dissolution. Prog. Polym. Sci. 2003, 28, 1223. [Google Scholar] [CrossRef]
- Herman, M.F.; Edwards, S.F. A reptation model for polymer dissolution. Macromolecules 1990, 23, 3662–3671. [Google Scholar] [CrossRef]
- Yoshioka, M.; Hancock, B.C.; Zografi, G. Inhibition of indomethacin crystallization in poly (vinylpyrrolidone) coprecipitates. J. Pharm. Sci. 1995, 84, 983–986. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-Y.; Lin, H.-L.; Chi, Y.-T.; Huang, Y.-T.; Kao, C.-Y.; Hsieh, W.-H. Thermoanalytical and Fourier transform infrared spectral curve-fitting techniques used to investigate the amorphous indomethacin formation and its physical stability in Indomethacin-Soluplus® solid dispersions. Int. J. Pharm. 2015, 496, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Tres, F.; Treacher, K.; Booth, J.; Hughes, L.P.; Wren, S.A.C.; Aylott, J.W.; Burley, J.C. Indomethacin-Kollidon VA64 Extrudates: A Mechanistic Study of pH-Dependent Controlled Release. Mol. Pharm. 2016, 13, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Semjonov, K.; Kogermann, K.; Laidmäe, I.; Antikainen, O.; Strachan, C.J.; Ehlers, H.; Yliruusi, J.; Heinämäki, J. The formation and physical stability of two-phase solid dispersion systems of indomethacin in supercooled molten mixtures with different matrix formers. Eur. J. Pharm. Sci. 2017, 97, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Jelić, D.; Liavitskaya, T.; Vyazovkin, S. Thermal stability of indomethacin increases with the amount of polyvinylpyrrolidone in solid dispersion. Thermochim. Acta 2019, 676, 172–176. [Google Scholar] [CrossRef]
- Nokhodchi, A. The effect of type and concentration of vehicles on the dissolution rate of a poorly soluble drug (indomethacin) from liquisolid compacts. J. Pharm. Pharm. Sci. 2005, 8, 18–25. [Google Scholar]
- De Jaeghere, W.; De Beer, T.; Van Bocxlaer, J.; Remon, J.P.; Vervaet, C. Hot-melt extrusion of polyvinyl alcohol for oral immediate release applications. Int. J. Pharm. 2015, 492, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kolter, K.; Karl, M.; Gryczke, A. Hot-Melt Extrusion with BASF Pharma Polymers; BASF The Chemical Company: Shakopee, MN, USA, 2012. [Google Scholar]
- Patra, C.N.; Priya, R.; Swain, S.; Jena, G.K.; Panigrahi, K.C.; Ghose, D. Pharmaceutical significance of Eudragit: A review. Future J. Pharm. Sci. 2017, 3, 33–45. [Google Scholar] [CrossRef]
- Kohlgrüber, K. Co-Rotating Twin-Screw Extruders: Fundamentals. In Co-Rotating Twin-Screw Extruders: Fundamentals; Kohlgrüber, K., Ed.; Hanser: New York, NY, USA, 2007; pp. I–XV. ISBN 978-1-56990-747-4. [Google Scholar]
- Otsuka, M.; Matsumoto, T.; Kaneniwa, N. Effect of environmental temperature on polymorphic solid-state transformation of indomethacin during grinding. Chem. Pharm. Bull. (Tokyo) 1986, 34, 1784–1793. [Google Scholar] [CrossRef]
- Trasi, N.S.; Byrn, S.R. Mechanically Induced Amorphization of Drugs: A Study of the Thermal Behavior of Cryomilled Compounds. AAPS PharmSciTech 2012, 13, 772–784. [Google Scholar] [CrossRef]
- Crowley, K.J.; Zografi, G. Cryogenic grinding of indomethacin polymorphs and solvates: Assessment of amorphous phase formation and amorphous phase physical stability. J. Pharm. Sci. 2002, 91, 492–507. [Google Scholar] [CrossRef]
- Center for Drug Evaluation and Research. Dissolution Testing of Immediate Release Solid Oral Dosage Forms. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/dissolution-testing-immediate-release-solid-oral-dosage-forms (accessed on 30 July 2020).
- Franco, P.; De Marco, I. Eudragit: A Novel Carrier for Controlled Drug Delivery in Supercritical Antisolvent Coprecipitation. Polymers 2020, 12, 234. [Google Scholar] [CrossRef] [PubMed]
- Wlodarski, K.; Zhang, F.; Liu, T.; Sawicki, W.; Kipping, T. Synergistic effect of polyvinyl alcohol and copovidone in itraconazole amorphous solid dispersions. Pharm. Res. 2018, 35, 16. [Google Scholar] [CrossRef] [PubMed]
- Terife, G.; Wang, P.; Faridi, N.; Gogos, C.G. Hot melt mixing and foaming of soluplus® and indomethacin. Polym. Eng. Sci. 2012, 52, 1629–1639. [Google Scholar] [CrossRef]



| API | Mw (g/mol) | logP | Tm (°C) | Tg (°C) | pKa | SGF Solubility [37] |
|---|---|---|---|---|---|---|
| Indomethacin | 357.8 | 4.27 | 155 ± 0.1 | 49 ± 0.1 | 4.5 | 0.004 g/1000 g |
| Polymer | Classification | Average Molecular Weight (g/mol) | Glass Transition (°C) | Degradation Temperature (°C) | Water Solubility (pH-7) |
|---|---|---|---|---|---|
| Parteck®MXP [38] | Non-ionic | 32,000 | 54 | 250 | Soluble |
| Soluplus® [39] | Non-ionic | 118,000 | 65–70 | 250 | Soluble |
| Kollidon®VA-64 [39] | Non-ionic | 45,000 | 100 | 230 | Soluble |
| Eudragit® EPO [40] | Ionic | 47,000 | 48 | 200 | Insoluble |
| Polymers | 20 mm VCM Disc Insert | 8 mm VCM Disc Insert | ||
|---|---|---|---|---|
| Heating Temperature (°C) | Heating Time (min) | Heating Temperature (°C) | Heating Time (min) | |
| Parteck® MXP | 230 | 5 | 230 | 5 |
| Soluplus® | 170 | 5 | 170 | 4 |
| Kollidon® VA-64 | 160 | 5 | 160 | 4 |
| Eudragit® EPO | 190 | 5 | 190 | 4 |
| Polymer | Pressure (bar) | Melt Temperature (°C) | Barrel Temperature for All The Zones (°C) | Torque % of Max. | Torque (Nm) |
|---|---|---|---|---|---|
| Parteck® MXP | 8–10 | 181 | 190 | 20 | 1–2 |
| Soluplus® | 1 | 151 | 160 | 24 | 1–4 |
| Kollidon® VA 64 | 0–1 | 152 | 160 | 40 | 2–4 |
| Eudragit® EPO | 0 | 152 | 160 | 43 | 2–6 |
| Excipients | Before VCM Process Weight (mg) | After VCM Process Weight (mg) | ||
|---|---|---|---|---|
| 8 mm | 20 mm | 8 mm | 20 mm | |
| Parteck® MXP | 167.51 ± 0.29 | 498.35 ± 0.84 | 166.46 ± 0.39 | 495.75 ± 1.77 |
| Soluplus® | 168.67 ± 1.21 | 502.43 ± 1.27 | 166.90 ± 0.56 | 499.20 ± 0.31 |
| Kollidon® VA 64 | 167.23 ± 2.05 | 500.85 ± 1.57 | 165.47 ± 2.56 | 498.98 ± 1.68 |
| Eudragit® EPO | 167.24 ± 1.78 | 500.12 ± 0.45 | 165.37 ± 1.08 | 497.39 ± 1.32 |
| Polymer | f2 Value |
|---|---|
| Parteck® MXP | 70.33 |
| Soluplus® | 61.19 |
| Kollidon® VA 64 | 84.59 |
| Eudragit® EPO | 51.78 |
| Polymer | Milled HME (mg·L−1·min) | Milled VCM (mg·L−1·min) | 8 mm VCM (mg·L−1·min) |
|---|---|---|---|
| Parteck® MXP | 7196.02 ± 1.09 | 6940.29 ± 3.96 | 3778.45 ± 1.25 |
| Soluplus® | 1053.25 ± 14.22 | 3421.46 ± 7.52 | 57.31 ± 22.96 |
| Kollidon® VA 64 | 1752.93 ± 25.91 | 2459.68 ± 6.43 | 1725.85 ± 14.17 |
| Eudragit® EPO | 3855.90 ± 5.74 | 2557.37 ± 2.38 | 3140.94 ± 7.94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shadambikar, G.; Kipping, T.; Di-Gallo, N.; Elia, A.-G.; Knüttel, A.-N.; Treffer, D.; Repka, M.A. Vacuum Compression Molding as a Screening Tool to Investigate Carrier Suitability for Hot-Melt Extrusion Formulations. Pharmaceutics 2020, 12, 1019. https://doi.org/10.3390/pharmaceutics12111019
Shadambikar G, Kipping T, Di-Gallo N, Elia A-G, Knüttel A-N, Treffer D, Repka MA. Vacuum Compression Molding as a Screening Tool to Investigate Carrier Suitability for Hot-Melt Extrusion Formulations. Pharmaceutics. 2020; 12(11):1019. https://doi.org/10.3390/pharmaceutics12111019
Chicago/Turabian StyleShadambikar, Gauri, Thomas Kipping, Nicole Di-Gallo, Alessandro-Giuseppe Elia, Anja-Nadine Knüttel, Daniel Treffer, and Michael. A Repka. 2020. "Vacuum Compression Molding as a Screening Tool to Investigate Carrier Suitability for Hot-Melt Extrusion Formulations" Pharmaceutics 12, no. 11: 1019. https://doi.org/10.3390/pharmaceutics12111019
APA StyleShadambikar, G., Kipping, T., Di-Gallo, N., Elia, A.-G., Knüttel, A.-N., Treffer, D., & Repka, M. A. (2020). Vacuum Compression Molding as a Screening Tool to Investigate Carrier Suitability for Hot-Melt Extrusion Formulations. Pharmaceutics, 12(11), 1019. https://doi.org/10.3390/pharmaceutics12111019








