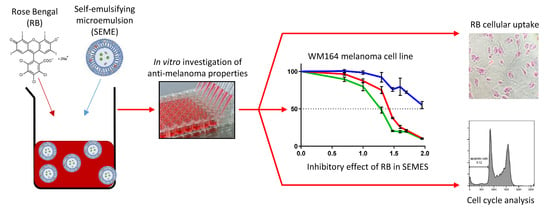
Development of an Oil-in-Water Self-Emulsifying Microemulsion for Cutaneous Delivery of Rose Bengal: Investigation of Anti-Melanoma Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical and Reagents
2.2. Surfactants and Other Excipients of the Self-Emulsifying Microemulsions (SEMEs)
2.3. Preparation of SEMEs
2.4. Cell Culture
2.5. In Vitro Cell Viability Assays: MTT Assay
2.6. Inhibitory Effect of Active Agent in Different Delivery Systems
2.7. Intracellular Uptake of the Active Agent
2.8. Cell Cycle Analysis by Flow Cytometry
2.9. Evaluation of Cell Viability
3. Results
3.1. In Vitro Cell Viability Assay of SEMEs and Its Components
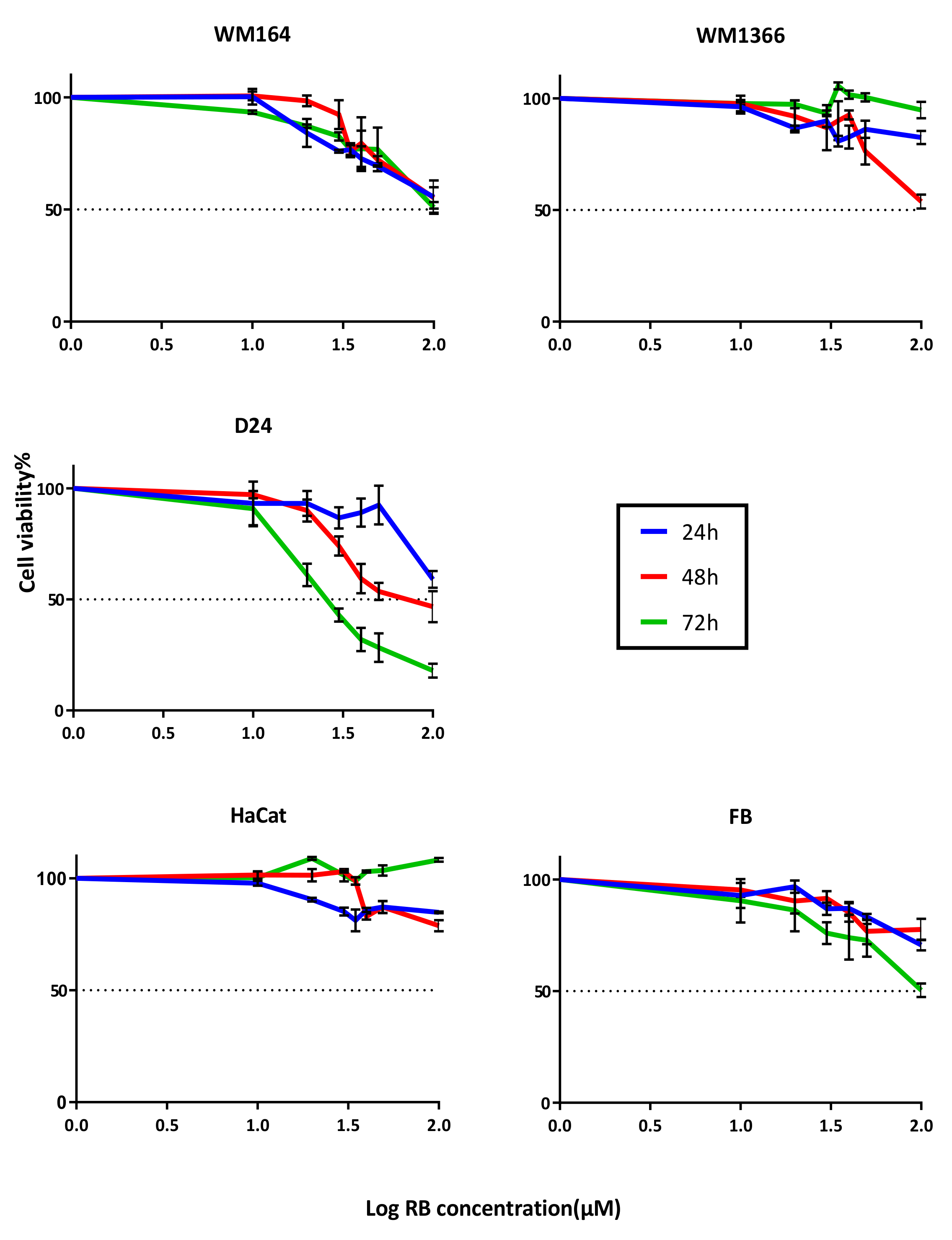
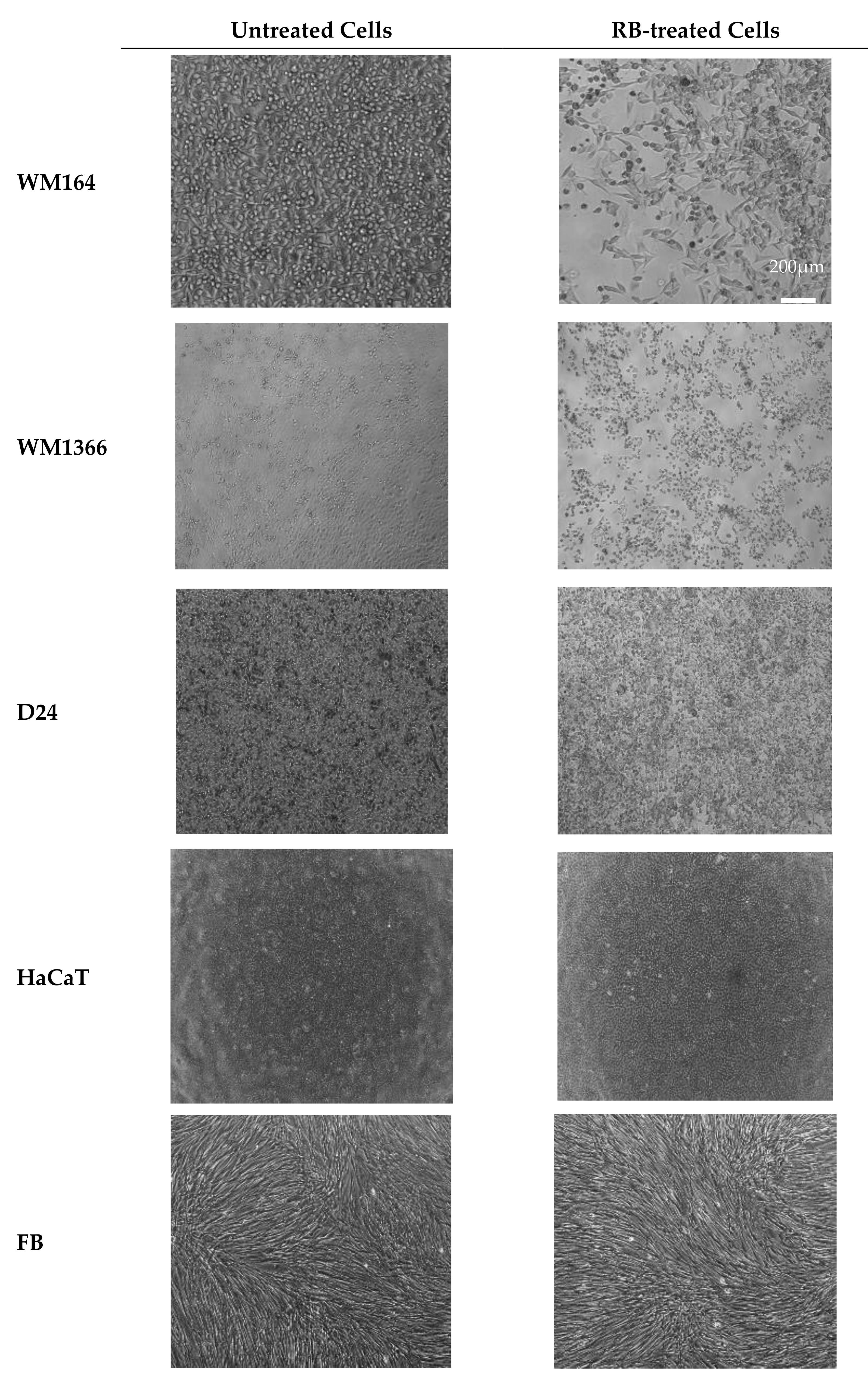
3.2. Treatment with Rose Bengal Aqueous Solution Induced Melanoma Cancer Cell Death In Vitro
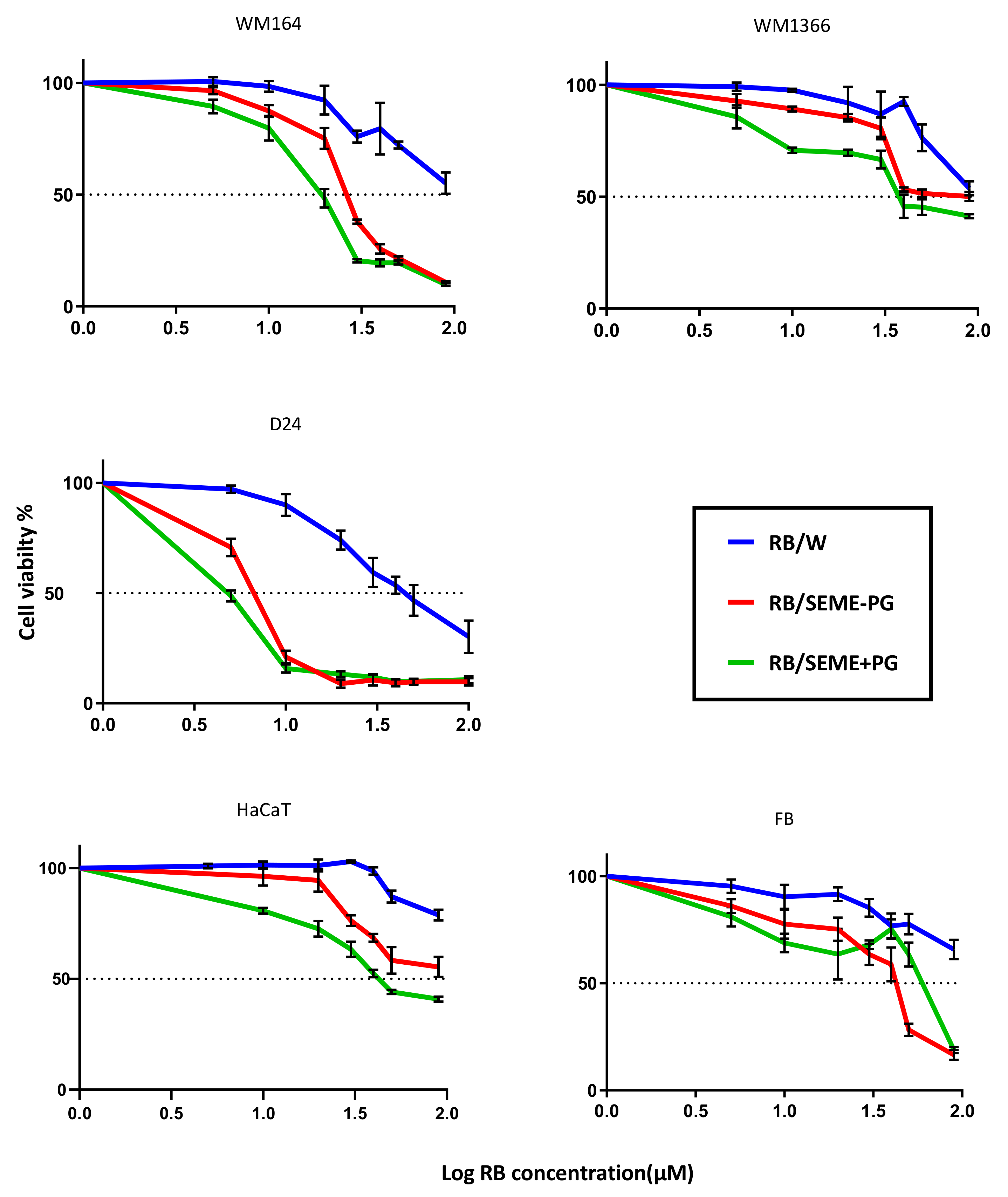
3.3. Inhibitory Effect of Rose Bengal in Different Delivery Systems
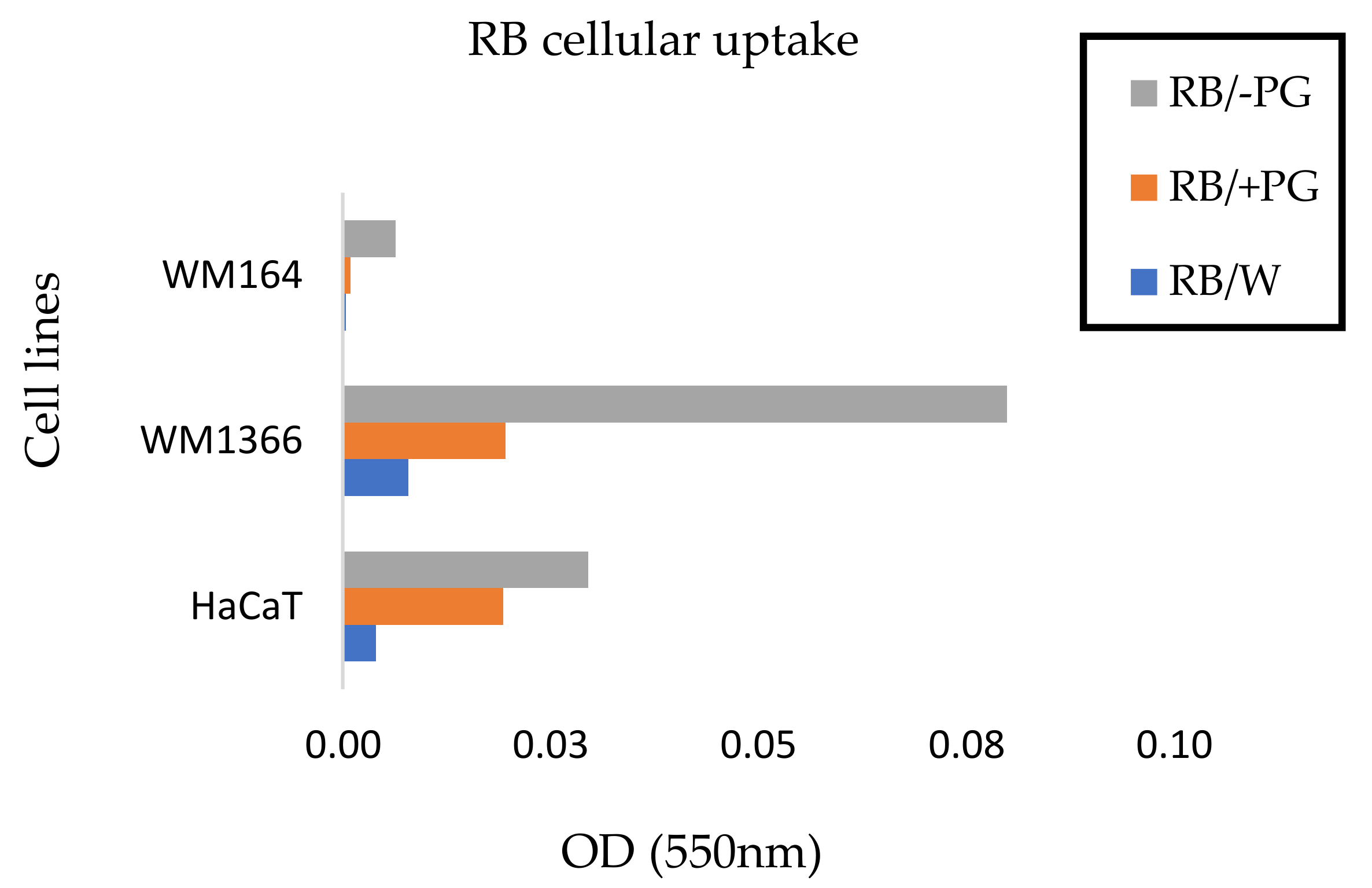
3.4. Effect of SEME on Cellular Uptake Of The Active Agent
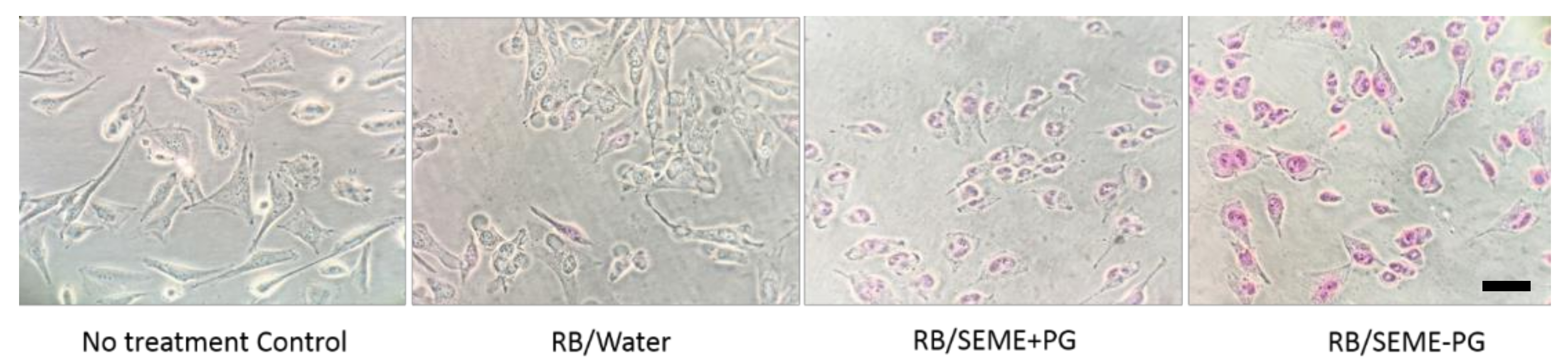
3.5. Cell Cycle Analysis by Flow Cytometry
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cancer in Australia. Available online: https://www.aihw.gov.au/reports/cancer/cancer-in-australia-2017 (accessed on 28 July 2020).
- Cancer Council Queensland. Available online: https://cancerqld.org.au/research/queensland-cancer-statistics/queensland-cancer-statistics-online-qcsol/ (accessed on 28 July 2020).
- Litwin, M.S.; Ryan, R.F.; Reed, R.J.; Krementz, E.T. Topical chemotherapy of advanced cutaneous malignancy with 5-fluorouracil creme. J. Surg. Oncol. 1971, 3, 351–365. [Google Scholar] [CrossRef] [PubMed]
- Agarwala, S. The Role of Intralesional Therapies in Melanoma. Oncolpgy (Williston Park) 2016, 30, 436–441. [Google Scholar]
- Salasche, S. Imiquimod 5% cream: A new treatment option for basal cell carcinoma. Int. J. Dermatol. 2002, 41, 16. [Google Scholar] [CrossRef]
- Konstantakou, E.G.; Velentzas, A.D.; Anagnostopoulos, A.K.; Litou, Z.I.; Konstandi, O.A.; Giannopoulou, A.F.; Anastasiadou, E.; Voutsinas, G.E.; Tsangaris, G.T.; Stravopodis, D.J. Deep-proteome mapping of WM-266-4 human metastatic melanoma cells: From oncogenic addiction to druggable targets. PLoS ONE 2017, 12, e0171512. [Google Scholar] [CrossRef] [PubMed]
- Borgheti-Cardoso, L.N.; Viegas, J.S.R.; Silvestrini, A.V.P.; Caron, A.L.; Praça, F.G.; Kravicz, M.; Bentley, M.V.L.B. Nanotechnology approaches in the current therapy of skin cancer. Adv. Drug Deliv. Rev. 2020, 153, 109–136. [Google Scholar] [CrossRef]
- Carter, P.; Narasimhan, B.; Wang, Q. Biocompatible nanoparticles and vesicular systems in transdermal drug delivery for various skin diseases. Int. J. Pharm. 2019, 555, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, Q.; Zhang, Z.; Yuan, L.; Liu, X.; Zhou, L. Preparation of curcumin-loaded liposomes and evaluation of their skin permeation and pharmacodynamics. Molecules (Basel Switz.) 2012, 17, 5972. [Google Scholar] [CrossRef]
- Dwivedi, A.; Mazumder, A.; Fox, L.T.; Brümmer, A.; Gerber, M.; Du Preez, J.L.; Haynes, R.K.; Du Plessis, J. In vitro skin permeation of artemisone and its nano-vesicular formulations. Int. J. Pharm. 2016, 503, 1–7. [Google Scholar] [CrossRef]
- Yu, X.; Du, L.; Li, Y.; Fu, G.; Jin, Y. Improved anti-melanoma effect of a transdermal mitoxantrone ethosome gel. Biomed. Pharmacother. 2015, 73, 6–11. [Google Scholar] [CrossRef]
- Benson, H.A.E.; Grice, J.E.; Mohammed, Y.; Namjoshi, S.; Roberts, M.S. Topical and Transdermal Drug Delivery: From Simple Potions to Smart Technologies. Curr. Drug Deliv. 2019, 16, 444–460. [Google Scholar] [CrossRef]
- Wachter, E.; Dees, C.; Harkins, J.; Scott, T.; Petersen, M.; Rush, R.E.; Cada, A. Topical rose Bengal: Pre-clinical evaluation of pharmacokinetics and safety. Lasers Surg. Med. 2003, 32, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, H.; Zhang, X.; Gillespie, S.; Wachter, E.; Hersey, P. Rose Bengal induces dual modes of cell death in melanoma cells and has clinical activity against melanoma. Melanoma Res. 2006, 16, S8. [Google Scholar] [CrossRef]
- Toomey, P.; Kodumudi, K.; Weber, A.; Kuhn, L.; Moore, E.; Sarnaik, A.A.; Pilon-Thomas, S. Intralesional Injection of Rose Bengal Induces a Systemic Tumor-Specific Immune Response in Murine Models of Melanoma and Breast Cancer. PLoS ONE 2013, 8, e68561. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Innamarato, P.P.; Kodumudi, K.; Weber, A.; Nemoto, S.; Robinson, J.L.; Crago, G.; McCardle, T.; Royster, E.; Sarnaik, A.A.; et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 2016, 7, 37893. [Google Scholar] [CrossRef]
- Chiricozzi, A.; Pitocco, R.; Saraceno, R.; Nistico, S.P.; Giunta, A.; Chimenti, S. New topical treatments for psoriasis. Expert Opin. Pharmacother. 2014, 15, 461–470. [Google Scholar] [CrossRef]
- Provectus Pharmaceuticals. An Efficacy and Safety Study of PH-10 Aqueous Hydrogel for the Treatment of Plaque Psoriasis. Available online: https://clinicaltrials.gov/ct2/show/NCT00941278?term=PH-10+and+psoriasis&rank=2 (accessed on 28 July 2020).
- Abd, E.; Namjoshi, S.; Mohammed, Y.H.; Roberts, M.S.; Grice, J.E. Synergistic Skin Penetration Enhancer and Nanoemulsion Formulations Promote the Human Epidermal Permeation of Caffeine and Naproxen. J. Pharm. Sci. 2016, 105, 212–220. [Google Scholar] [CrossRef]
- He, C.-X.; He, Z.-G.; Gao, J.-Q. Microemulsions as drug delivery systems to improve the solubility and the bioavailability of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2010, 7, 445–460. [Google Scholar] [CrossRef]
- Cilurzo, F.; Cristiano, M.C.; Di Marzio, L.; Cosco, D.; Carafa, M.; Ventura, C.A.; Fresta, M.; Paolino, D. Influence of the Supramolecular Micro-Assembly of Multiple Emulsions on their Biopharmaceutical Features and In vivo Therapeutic Response. Curr. Drug Targets 2015, 16, 1612–1622. [Google Scholar] [CrossRef]
- Panapisal, V.; Charoensri, S.; Tantituvanont, A. Formulation of Microemulsion Systems for Dermal Delivery of Silymarin. Off. J. Am. Assoc. Pharm. Sci. 2012, 13, 389–399. [Google Scholar] [CrossRef]
- Patel, M.R.; Patel, R.B.; Parikh, J.R.; Patel, B.G. Novel microemulsion-based gel formulation of tazarotene for therapy of acne. Pharm. Dev. Technol. 2016, 21, 921–932. [Google Scholar] [CrossRef]
- Budden, R.; Kühl, U.G.; Bahlsen, J. Experiments on the toxic, sedative and muscle relaxant potency of various drug solvents in mice. Pharmacol. Ther. 1979, 5, 467–474. [Google Scholar] [CrossRef]
- Bárány, E.; Lindberg, M.; Lodén, M. Biophysical characterization of skin damage and recovery after exposure to different surfactants. Contact Dermat. 1999, 40, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Barbaud, A.; Waton, J.; Pinault, A.-L.; Bursztejn, A.-C.; Schmutz, J.-L.; Trechot, P. Cutaneous adverse drug reactions caused by delayed sensitization to carboxymethylcellulose. Contact Dermat. 2011, 64, 294. [Google Scholar] [CrossRef] [PubMed]
- Patrick, E.; Maibach, H.I.; Burkhalter, A. Mechanisms of chemically induced skin irritation: I. Studies of time course, dose response, and components of inflammation in the laboratory mouse. Toxicol. Appl. Pharm. 1985, 81, 476–490. [Google Scholar] [CrossRef]
- Fischer, D.; Li, Y.; Ahlemeyer, B.; Krieglstein, J.; Kissel, T. In vitro cytotoxicity testing of polycations: Influence of polymer structure on cell viability and hemolysis. Biomaterials 2003, 24, 1121–1131. [Google Scholar] [CrossRef]
- Nogueira, D.R.; Mitjans, M.; Infante, M.R.; Vinardell, M.P. Comparative sensitivity of tumor and non-tumor cell lines as a reliable approach for in vitro cytotoxicity screening of lysine-based surfactants with potential pharmaceutical applications. Int. J. Pharm. 2011, 420, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Martinez, V.; Corsini, E.; Mitjans, M.; Pinazo, A.; Vinardell, M.P. Evaluation of eye and skin irritation of arginine-derivative surfactants using different in vitro endpoints as alternatives to the in vivo assays. Toxicol. Lett. 2006, 164, 259–267. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Zhang, Y. Cell toxicity mechanism and biomarker. Clin. Transl. Med. 2018, 7, 1–6. [Google Scholar] [CrossRef]
- Li, L.; Yi, T.; Lam, C.W.K. Inhibition of human efflux transporter ABCC2 (MRP2) by self-emulsifying drug delivery system: Influences of concentration and combination of excipients. J. Pharm. Pharm. Sci. 2014, 17, 447–460. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Koevary, S.B. Selective toxicity of rose bengal to ovarian cancer cells in vitro. Int. J. Physiol. Pathophysiol. Pharmacol. 2012, 4, 99–107. [Google Scholar] [PubMed]
- Qin, J.; Kunda, N.; Qiao, G.; Calata, J.F.; Pardiwala, K.; Prabhakar, B.S.; Maker, A.V. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death Dis. 2017, 8, e2584. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.F.; Agarwala, S.S.; Smithers, B.M.; Ross, M.I.; Scoggins, C.R.; Coventry, B.J.; Neuhaus, S.J.; Minor, D.R.; Singer, J.M.; Wachter, E.A. Phase 2 Study of Intralesional PV-10 in Refractory Metastatic Melanoma. Ann. Surg. Oncol. 2015, 22, 2135–2142. [Google Scholar] [CrossRef] [PubMed]
- Bačkorová, M.; Bačkor, M.; Mikeš, J.; Jendželovský, R.; Fedoročko, P. Variable responses of different human cancer cells to the lichen compounds parietin, atranorin, usnic acid and gyrophoric acid. Toxicol. In Vitro 2011, 25, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Duncan, R.; Ferruti, P.; Sgouras, D.; Tuboku-Metzger, A.; Ranucci, E.; Bignotti, F. A Polymer-Triton X-100 Conjugate Capable of PH-Dependent Red Blood Cell Lysis: A Model System Illustrating the Possibility of Drug Delivery Within Acidic Intracellular Compartments. J. Drug Target. 1994, 2, 341–347. [Google Scholar] [CrossRef]
- Rafael, C.D.; Frederico, P.; Dalton, D.; Rodrigo, M.; Daniel, S.P.; Miriam, T.P.L.; Nádia, R.B.R. Chemical composition and cytotoxicity activity of the essential oil of Pterodon emarginatus. Rev. Bras. Farmacogn. 2012, 22, 971–978. [Google Scholar]
- Schröterová, L.; Králová, V.; Voráčová, A.; Hašková, P.; Rudolf, E.; Červinka, M. Antiproliferative effects of selenium compounds in colon cancer cells: Comparison of different cytotoxicity assays. Toxicol. In Vitro 2009, 23, 1406–1411. [Google Scholar] [CrossRef]
- Tan, F.; Wang, M.; Wang, W.; Lu, Y. Comparative evaluation of the cytotoxicity sensitivity of six fish cell lines to four heavy metals in vitro. Toxicol. In Vitro 2008, 22, 164–170. [Google Scholar] [CrossRef]
- Morais, M.C.C.; Stuhl, I.; Sabino, A.U.; Lautenschlager, W.; Queiroga, A.; Tortelli, T.C.; Chammas, R.; Suhov, Y.; Ramos, A. Stochastic model of contact inhibition and the proliferation of melanoma in situ. Clin. Cancer Res. 2018, 24, 99. [Google Scholar] [CrossRef]
- Sha, X.; Yan, G.; Wu, Y.; Li, J.; Fang, X. Effect of self-microemulsifying drug delivery systems containing Labrasol on tight junctions in Caco-2 cells. Eur. J. Pharm. Sci. 2005, 24, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Ujhelyi, Z.; Fenyvesi, F.; Váradi, J.; Fehér, P.; Kiss, T.; Veszelka, S.; Deli, M.; Vecsernyés, M.; Bácskay, I. Evaluation of cytotoxicity of surfactants used in self-micro emulsifying drug delivery systems and their effects on paracellular transport in Caco-2 cell monolayer. Eur. J. Pharm. Sci. 2012, 47, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Ujhelyi, Z.; Kalantari, A.; Vecsernyés, M.; Róka, E.; Fenyvesi, F.; Póka, R.; Kozma, B.; Bácskay, I. The enhanced inhibitory effect of different antitumor agents in self-microemulsifying drug delivery systems on human cervical cancer HeLa cells. Molecules 2015, 20, 13226–13239. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chang, X.; Weng, T.; Zhao, X.; Gao, Z.; Yang, Y.; Xu, H.; Yang, X. A study of microemulsion systems for transdermal delivery of triptolide. J. Control. Release 2004, 98, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.-J.; Fu, Y.-S.; Lin, Y.-H.; Huang, Y.-B.; Wu, P.-C. The Effect of Nanoemulsion as a Carrier of Hydrophilic Compound for Transdermal Delivery. PLoS ONE 2014, 9, e102850. [Google Scholar] [CrossRef]
- Wu, H.; Ramachandran, C.; Weiner, N.D.; Roessler, B.J. Topical transport of hydrophilic compounds using water-in-oil nanoemulsions. Int. J. Pharm. 2001, 220, 63–75. [Google Scholar] [CrossRef]
- Lin, Y.; Shen, Q.; Katsumi, H.; Okada, N.; Fujita, T.; Jiang, X.; Yamamoto, A. Effects of Labrasol and other pharmaceutical excipients on the intestinal transport and absorption of rhodamine123, a P-glycoprotein substrate, in rats. Biol. Pharm. Bull. 2007, 30, 1301–1307. [Google Scholar] [CrossRef]
- Pollard, J.; Rajabi-Siahboomi, A.; Badhan, R.K.S.; Mohammed, A.R.; Perrie, Y. High-throughput screening of excipients with a biological effect: A kinetic study on the effects of surfactants on efflux-mediated transport. J. Pharm. Pharmacol. 2019, 71, 889–897. [Google Scholar] [CrossRef]
- Sydykov, B.; Oldenhof, H.; de Oliveira Barros, L.; Sieme, H.; Wolkers, W.F. Membrane permeabilization of phosphatidylcholine liposomes induced by cryopreservation and vitrification solutions. Biochim. Biophys. Acta (Bba)-Biomembr. 2018, 1860, 467–474. [Google Scholar] [CrossRef]
- Malajczuk, C.J.; Hughes, Z.E.; Mancera, R.L. Molecular dynamics simulations of the interactions of DMSO, mono- and polyhydroxylated cryosolvents with a hydrated phospholipid bilayer. Biochim. Biophys. Acta-Biomembr. 2013, 1828, 2041–2055. [Google Scholar] [CrossRef]
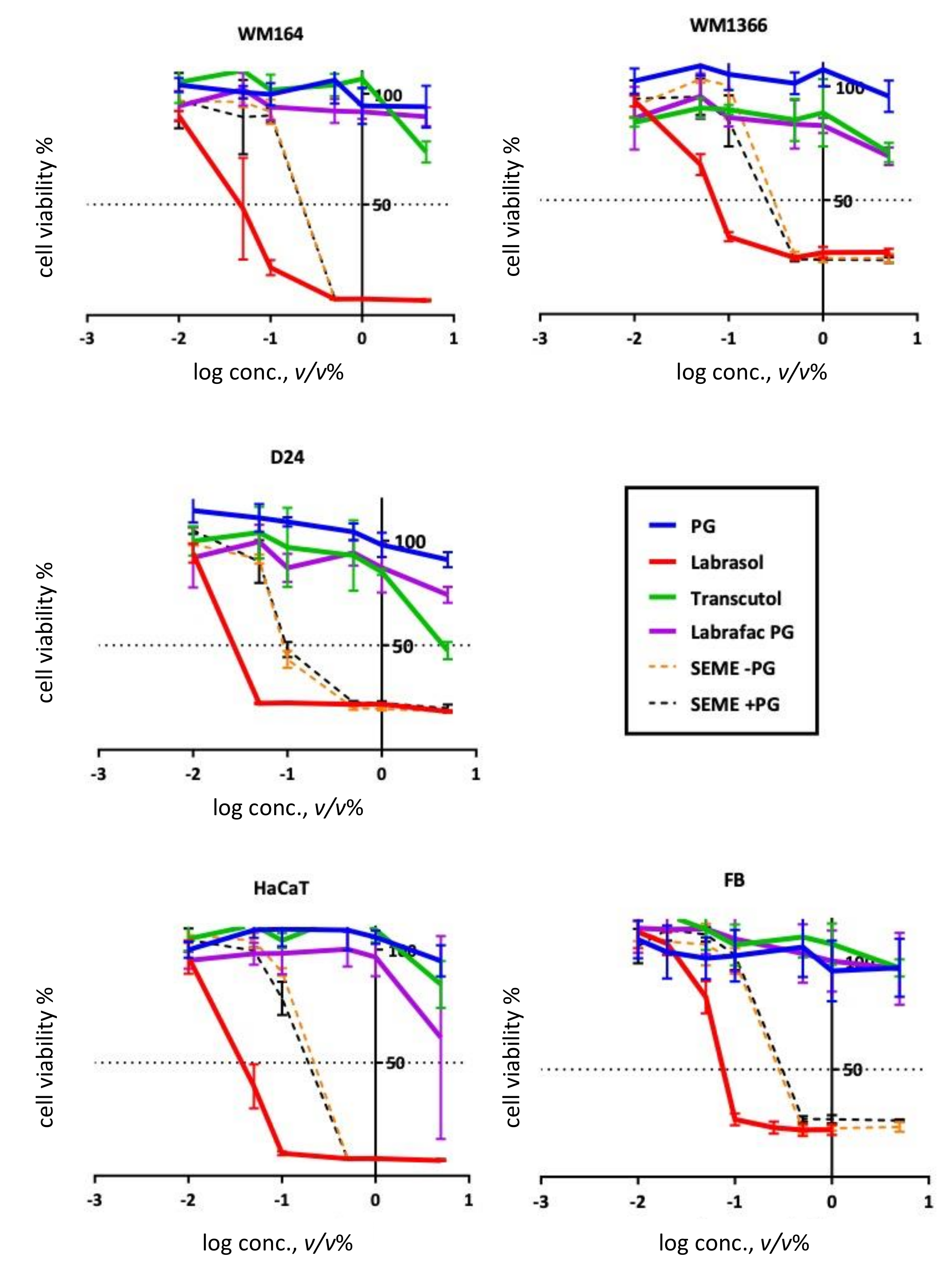
| Excipients | IC50 (v/v %) | ||||
|---|---|---|---|---|---|
| WM164 | WM1366 | D24 | HaCaT | Fibroblast | |
| PG | >Max Conc. | >Max Conc. | 14.74 (very wide) | >Max Conc. | 52.13 (very wide) |
| Labrasol® | 0.04 (0.034–0.058) | 0.11 (0.06–0.21) | 0.03 (0.02–0.06 | 0.04 (0.03–0.04) | 0.08 (0.06–0.13) |
| Transcutol® | ~5.6 (very wide) | >Max Conc. | 4.55 (3.1-8.2) | 5.75 (very wide) | >Max Conc. |
| Labrafac® | >Max Conc. | >Max Conc. | 92.16 (very wide) | 6.35 (4.03-8.02) | >Max Conc. |
| SEME–PG | 0.22 (0.18–0.27) | 0.37 (0.25–0.52) | 0.15 (0.09–0.23) | 0.22 (0.17–0.27) | 0.34 (0.25–0.45) |
| SEME + PG | 0.21 (0.13–0.29) | 0.30 (0.21–0.45) | 0.18 (0.11–0.28) | 0.18 (0.14–0.22) | 0.39 (0.28–0.53) |
| RB Aqueous Solution | IC50 (µM) | ||||
|---|---|---|---|---|---|
| WM164 | WM1366 | D24 | HaCaT | Fibroblast | |
| 24 h | 112.2 (92.5–147.2) | >Max Conc. | 130.9 (106.6–197) | 2254 (633.2–36213) | 238 (181.7–356.1) |
| 48 h | 106.6 (88.9–138.8) | 109.8 (93.74–139.2) | 69.97 (60.2–84.67) | 221.3 (155.7–465.4) | 458.7 (252.1–1405) |
| 72 h | 109.1 (93–136) | ~113.7 (Very wide) | 27.33 (25.2–29.6) | >Max Conc. | 108.2 (87.1–150.5) |
| RB/Vehicles | IC50 (μM) | ||||
|---|---|---|---|---|---|
| WM164 | WM1366 | D24 | HaCaT | Fibroblast | |
| RB/W | 102.8 (86.4–131.9) | 98.93 (86.6–120.8) | 45.6 (42.0–49.8) | 152.8 (127.3–204.1) | 188.4 (142.2–284.1) |
| RB/SEME–PG | 27.03 (25.4–28.7) | 69.55 (57.7–90.2) | 6.73 (6.07–7.4) | 86.95 (71.9–114.9) | 37.2 (32.7–42.2) |
| RB/SEME+PG | 18.58 (17.1–20.1) | 47.06 (39.7–57.5) | 4.86 (4.2–5.6) | 49.27 (44.7–54.9) | 55.95 (41.1–89.4) |
| Apoptotic Cells % (Sub G0G1) | Untreated | RB/W | RB/SEME–PG | RB/SEME + PG |
|---|---|---|---|---|
| D24 | 2.54 | 2.66 | 4.79 | 5.83 |
| WM164 | 3.94 | 6.69 | 9.12 | 9.50 |
| WM1366 | 0.89 | 2.29 | 4.04 | 5.24 |
| HaCaT | 1.07 | 1.15 | 1.36 | 1.03 |
| Fibroblast | 0.00 | 0.99 | 0.93 | 0.69 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forouz, F.; Dabbaghi, M.; Namjoshi, S.; Mohammed, Y.; Roberts, M.S.; Grice, J.E. Development of an Oil-in-Water Self-Emulsifying Microemulsion for Cutaneous Delivery of Rose Bengal: Investigation of Anti-Melanoma Properties. Pharmaceutics 2020, 12, 947. https://doi.org/10.3390/pharmaceutics12100947
Forouz F, Dabbaghi M, Namjoshi S, Mohammed Y, Roberts MS, Grice JE. Development of an Oil-in-Water Self-Emulsifying Microemulsion for Cutaneous Delivery of Rose Bengal: Investigation of Anti-Melanoma Properties. Pharmaceutics. 2020; 12(10):947. https://doi.org/10.3390/pharmaceutics12100947
Chicago/Turabian StyleForouz, Farzaneh, Maryam Dabbaghi, Sarika Namjoshi, Yousuf Mohammed, Michael S. Roberts, and Jeffrey E. Grice. 2020. "Development of an Oil-in-Water Self-Emulsifying Microemulsion for Cutaneous Delivery of Rose Bengal: Investigation of Anti-Melanoma Properties" Pharmaceutics 12, no. 10: 947. https://doi.org/10.3390/pharmaceutics12100947
APA StyleForouz, F., Dabbaghi, M., Namjoshi, S., Mohammed, Y., Roberts, M. S., & Grice, J. E. (2020). Development of an Oil-in-Water Self-Emulsifying Microemulsion for Cutaneous Delivery of Rose Bengal: Investigation of Anti-Melanoma Properties. Pharmaceutics, 12(10), 947. https://doi.org/10.3390/pharmaceutics12100947