In Vitro and Ex Vivo Evaluation of Tablets Containing Piroxicam-Cyclodextrin Complexes for Buccal Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Solid Complexes
2.2.2. Tablet Preparation
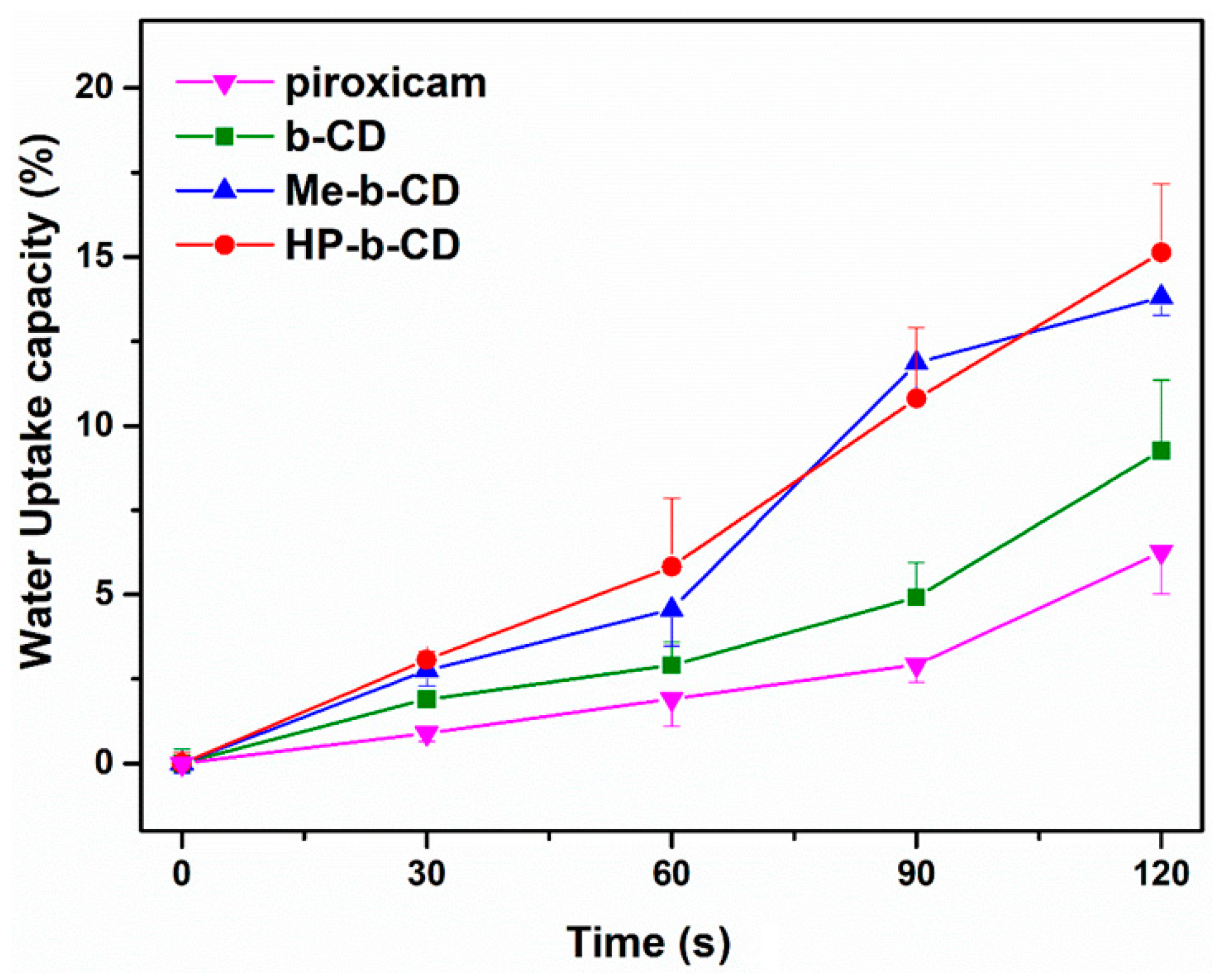
2.2.3. Drug Loading and Water Uptake Studies
2.2.4. Physicochemical Characterization
2.2.5. In Vitro Release Studies
Dissolution Studies
Permeation Studies
2.2.6. Histological Evaluation
2.2.7. Statistical Analysis
3. Results and Discussion
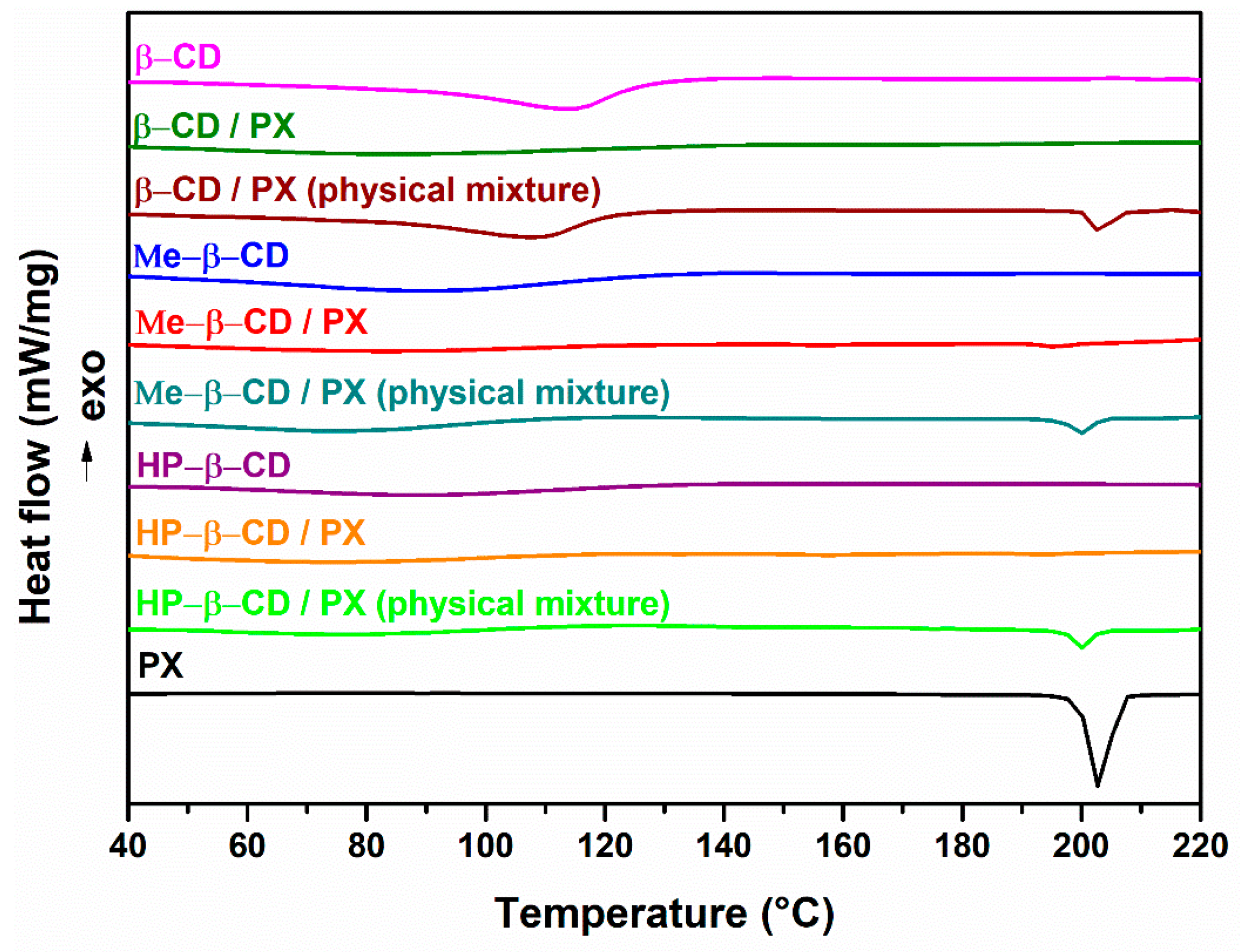
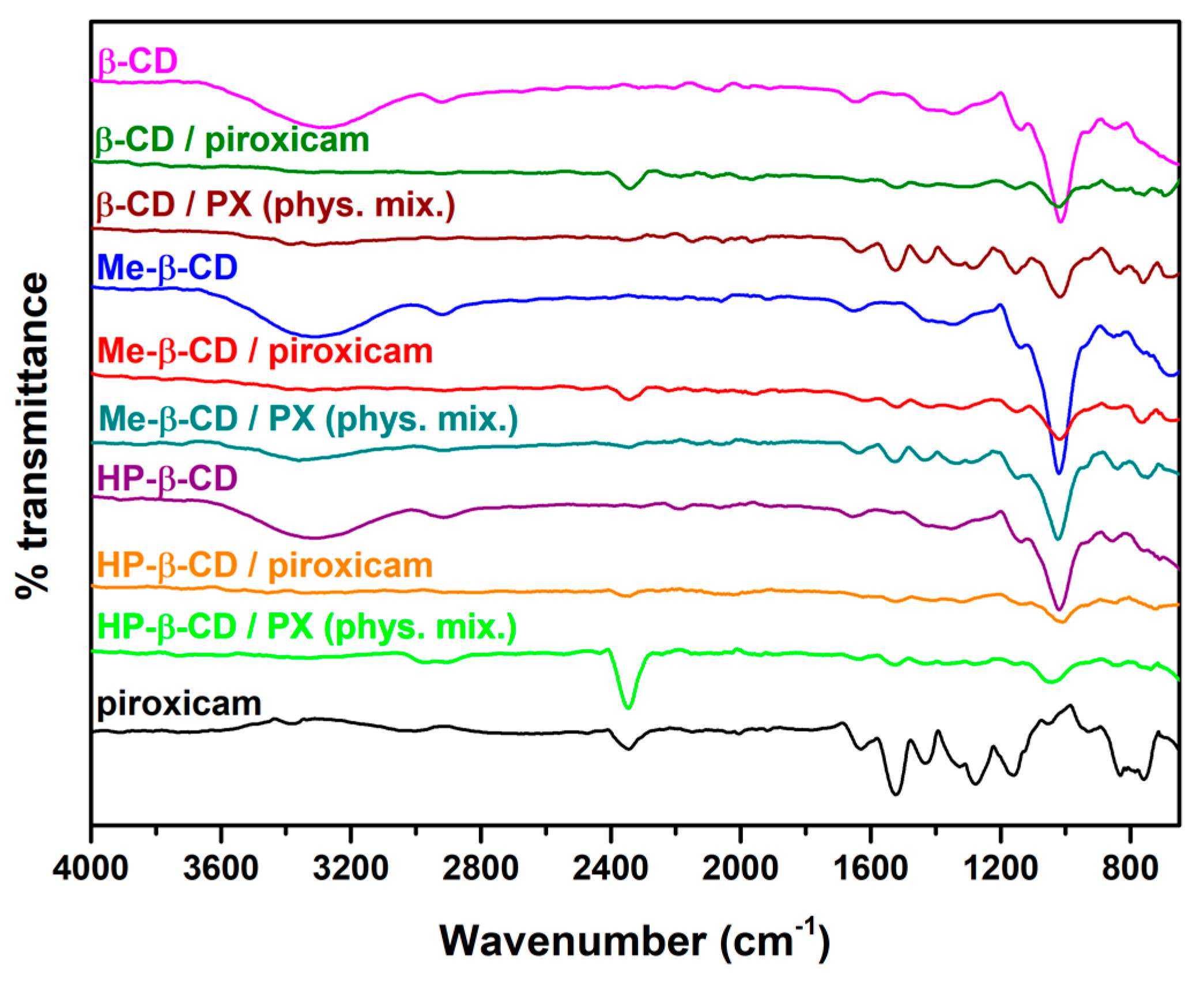
3.1. Drug Content Quantification and Characterization Studies
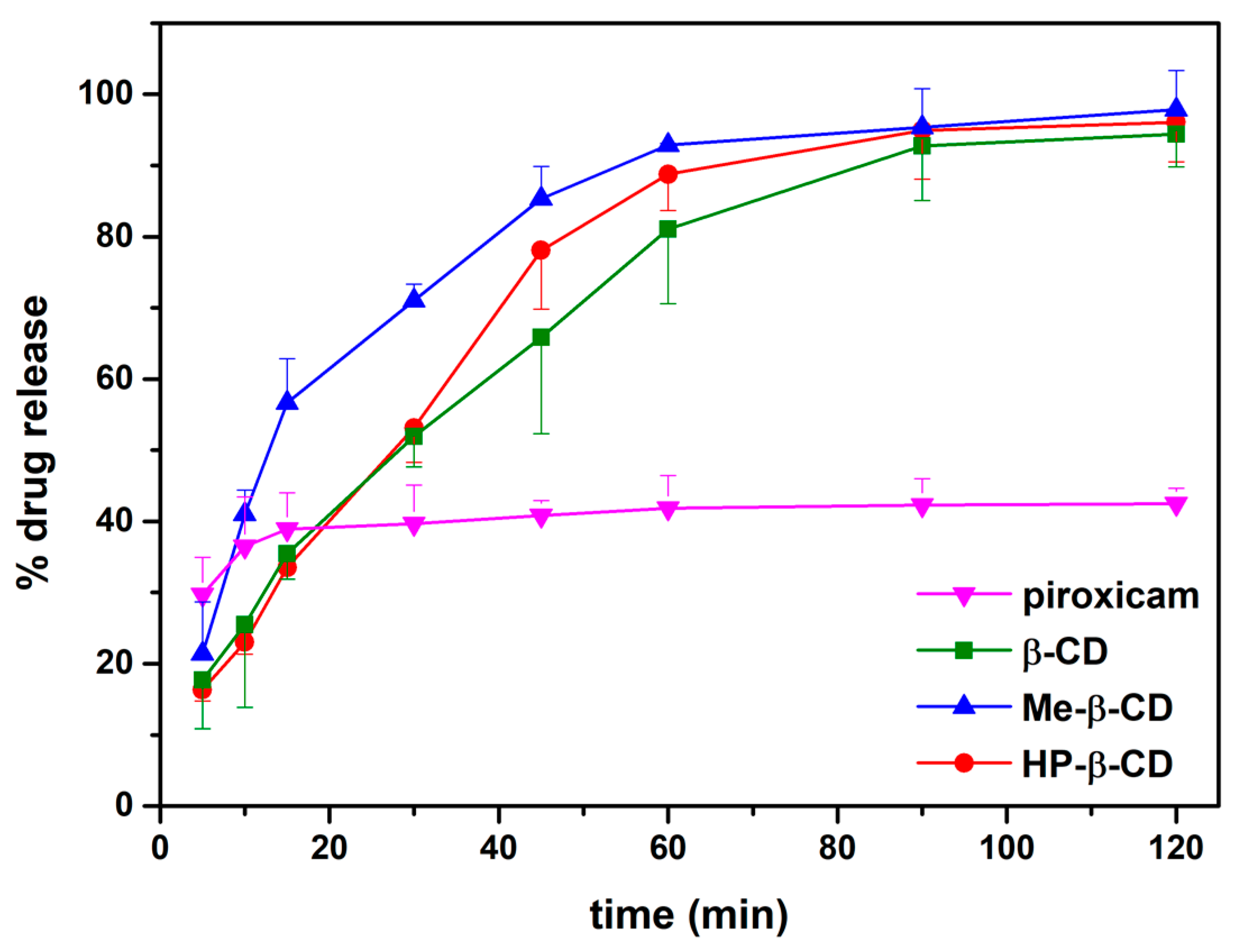
3.2. In Vitro Release
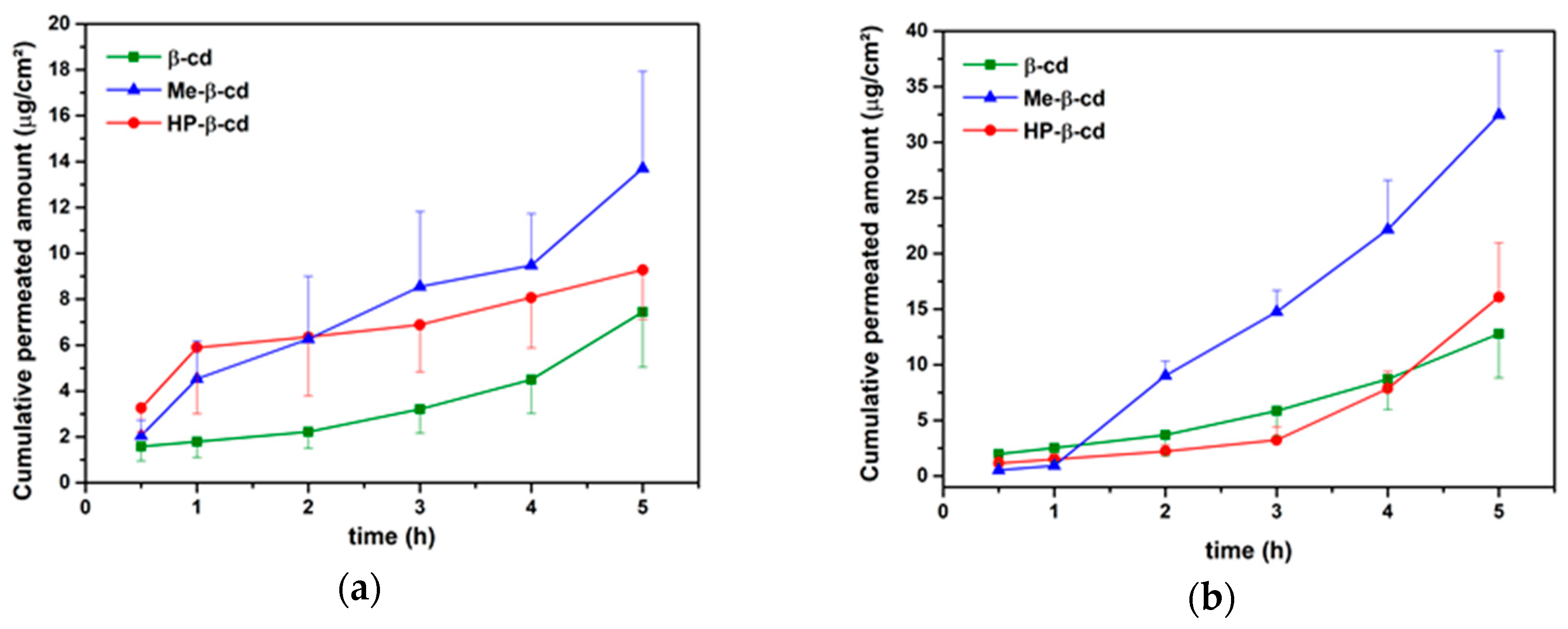
3.3. Drug Permeation
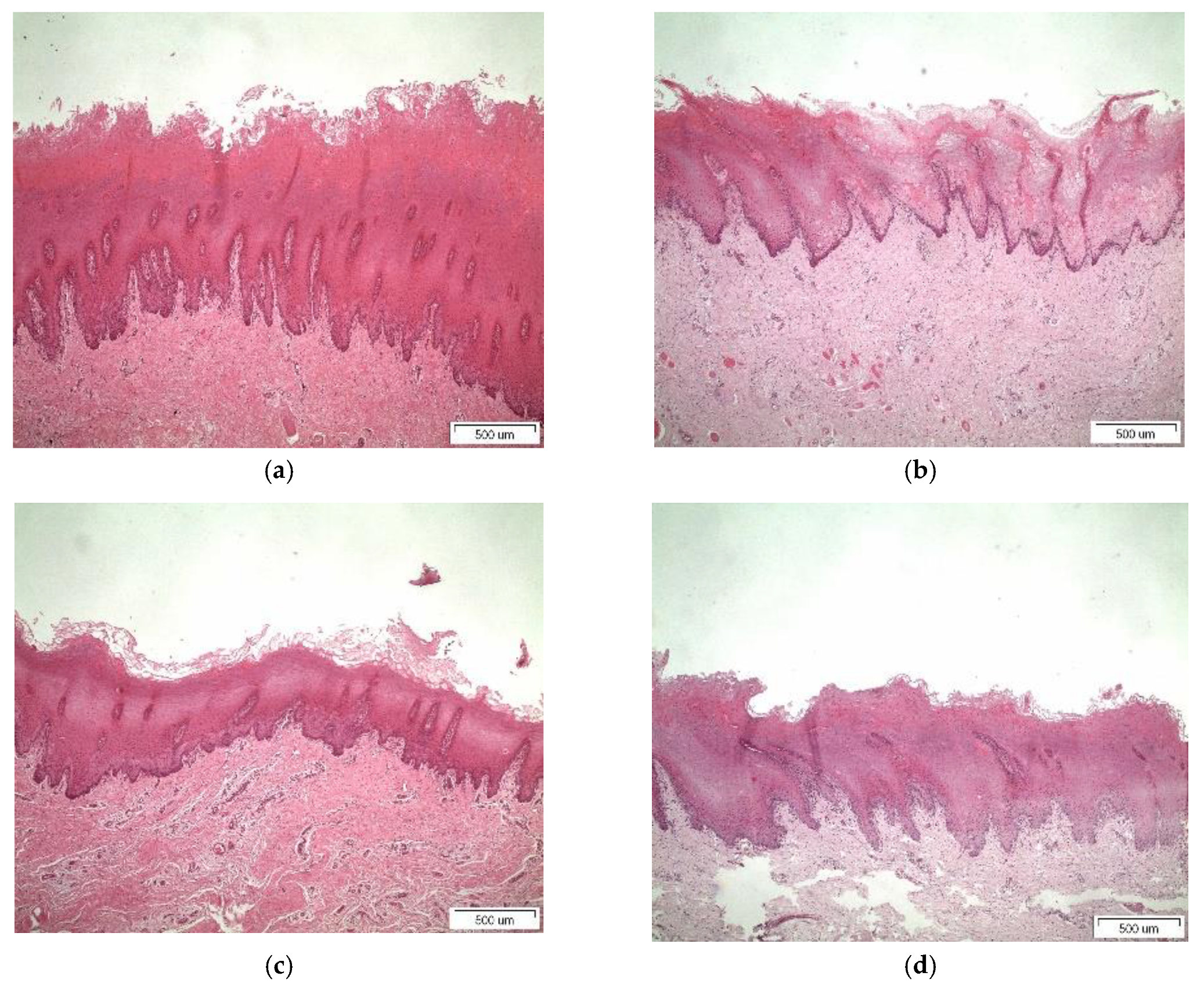
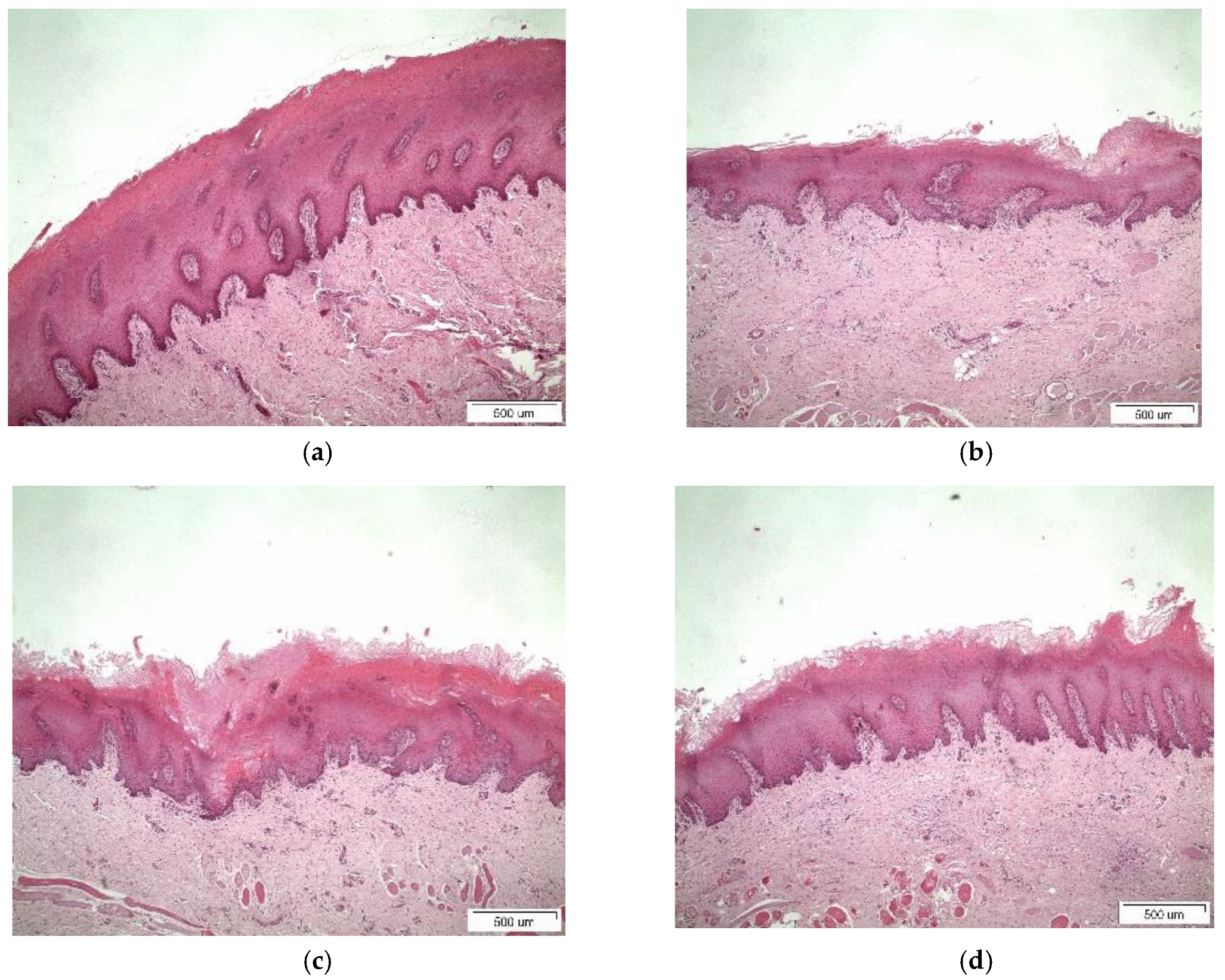
3.4. Histological Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nicolazzo, J.A.; Reed, B.L.; Finnin, B.C. Buccal penetration enhancers—How do they really work? J. Control. Release 2005, 105, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Marxen, E.; Jacobsen, J.; Hyrup, B.; Janfelt, C. Permeability Barriers for Nicotine and Mannitol in Porcine Buccal Mucosa Studied by High-Resolution MALDI Mass Spectrometry Imaging. Mol. Pharm. 2018, 15, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Kontogiannidou, E.; Andreadis, D.A.; Zografos, A.L.; Nazar, H.; Klepetsanis, P.; van der Merwe, S.M.; Fatouros, D.G. Ex vivo buccal drug delivery of ropinirole hydrochloride in the presence of permeation enhancers: The effect of charge. Pharm. Dev. Technol. 2017, 22, 1017–1021. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nazari, K.; Kontogiannidou, E.; Ahmad, R.H.; Gratsani, A.; Rasekh, M.; Arshad, M.S.; Sunar, B.S.; Armitage, D.; Bouropoulos, N.; Chang, M.W.; et al. Development and characterisation of cellulose based electrospun mats for buccal delivery of non-steroidal anti-inflammatory drug (NSAID). Eur. J. Pharm. Sci. 2017, 102, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.F.; Liu, F.; Brown, M.B. Modeling the oral cavity: In vitro and in vivo evaluations of buccal drug delivery systems. J. Control. Release 2012, 161, 746–756. [Google Scholar] [CrossRef]
- Consuelo, I.; Pizzolato, G.P.; Falson, F.; Guy, R.H.; Jacques, Y. Evaluation of pig esophageal mucosa as a permeability barrier model for buccal tissue. J. Pharm. Sci. 2005, 94, 2777–2788. [Google Scholar] [CrossRef] [PubMed]
- Kurkov, S.V.; Loftsson, T. Cyclodextrins. Int. J. Pharm. 2013, 453, 167–180. [Google Scholar] [CrossRef]
- Figueiras, A.; Hombach, J.; Veiga, F.; Bernkop-Schnürch, A. In vitro evaluation of natural and methylated cyclodextrins as buccal permeation enhancing system for omeprazole delivery. Eur. J. Pharm. Biopharm. 2009, 71, 339–345. [Google Scholar] [CrossRef]
- Mura, P. Analytical techniques for characterization of cyclodextrin complexes in the solid state: A review. J. Pharm. Biomed. Anal. 2015, 113, 226–238. [Google Scholar] [CrossRef]
- Loftsson, T.; Jarho, P.; Másson, M.; Järvinen, T. Cyclodextrins in drug delivery. Expert Opin. Drug Deliv. 2005, 2, 335–351. [Google Scholar] [CrossRef]
- Mura, P.; Cirri, M.; Mennini, N.; Casella, G.; Maestrelli, F. Polymeric mucoadhesive tablets for topical or systemic buccal delivery of clonazepam: Effect of cyclodextrin complexation. Carbohydr. Polym. 2016, 152, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, A. Cyclodextrins as drug carrier molecule: A review. Sci. Pharm. 2008, 76, 567–598. [Google Scholar] [CrossRef]
- McEwen, J. Clinical Pharmacology of Piroxicam-β-Cyclodextrin. Clin. Drug Investig. 2000, 19, 27–31. [Google Scholar] [CrossRef]
- Jug, M.; Bećirević-Laćan, M. Influence of hydroxypropyl-β-cyclodextrin complexation on piroxicam release from buccoadhesive tablets. Eur. J. Pharm. Sci. 2004, 21, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Bouchal, F.; Skiba, M.; Chaffai, N.; Hallouard, F.; Fatmi, S.; Lahiani-Skiba, M. Fast dissolving cyclodextrin complex of piroxicam in solid dispersion Part I: Influence of β-CD and HPβ-CD on the dissolution rate of piroxicam. Int. J. Pharm. 2015, 478, 625–632. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, D.; Lai, J.; Lu, Y.; Yin, Z.; Wu, W. Piroxicam/2-Hydroxypropyl-β-Cyclodextrin Inclusion Complex Prepared by a New Fluid-Bed Coating Technique. J. Pharm. Sci. 2009, 98, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Van Hees, T.; Piel, G.; de Hassonville, S.H.; Evrard, B.; Delattre, L. Determination of the free/included piroxicam ratio in cyclodextrin complexes: Comparison between UV spectrophotometry and differential scanning calorimetry. Eur. J. Pharm. Sci. 2002, 15, 347–353. [Google Scholar] [CrossRef]
- Vrecer, F.; Vrbinc, M.; Meden, A. Characterization of piroxicam crystal modifications. Int. J. Pharm. 2003, 256, 3–15. [Google Scholar] [CrossRef]
- Basan, H.; Göğer, N.G.; Ertaş, N.; Orbey, M.T. Quantitative determination of piroxicam in a new formulation (piroxicam-β-cyclodextrin) by derivative UV spectrophotometric method and HPLC. J. Pharm. Biomed. Anal. 2001, 26, 171–178. [Google Scholar] [CrossRef]
- Salamatmiller, N.; Chittchang, M.; Johnston, T. The use of mucoadhesive polymers in buccal drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1666–1691. [Google Scholar] [CrossRef]
- Senel, S.; Hincal, A.A. Drug permeation enhancement via buccal route: Possibilities and limitations. J. Control. Release 2001, 72, 133–144. [Google Scholar] [CrossRef]
- Jug, M.; Bećirević-Laćan, M. Multicomponent Complexes of Piroxicam with Cyclodextrins and Hydroxypropyl Methylcellulose. Drug Dev. Ind. Pharm. 2004, 30, 1051–1060. [Google Scholar] [CrossRef] [PubMed]
- Shan-Yang, L.; Yuh-Horng, K. Solid particulates of drug-β-cyclodextrin inclusion complexes directly prepared by a spray-drying technique. Int. J. Pharm. 1989, 56, 249–259. [Google Scholar] [CrossRef]
- Pires, F.Q.; Pinho, L.A.; Freire, D.O.; Silva, I.C.R.; Sa-Barreto, L.L.; Cardozo-Filho, L.; Gratieri, T.; Gelfuso, G.M.; Cunha-Filho, M. Thermal analysis used to guide the production of thymol and Lippia origanoides essential oil inclusion complexes with cyclodextrin. J. Therm. Anal. Calorim. 2019, 137, 543–553. [Google Scholar] [CrossRef]
- Maximiano, F.P.; Costa, G.H.Y.; de Sá Barreto, L.C.L.; Bahia, M.T.; Cunha-Filho, M.S.S. Development of effervescent tablets containing benznidazole complexed with cyclodextrin. J. Pharm. Pharmacol. 2011, 63, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, V.; Ilic-Stojanovic, S.; Nikolic, L.; Cakic, M.; Zdravkovic, A.; Kapor, A.; Popsavin, M. Photostability of piroxicam in the inclusion complex with 2-hydroxypropyl-β-cyclodextrin. Hem. Ind. 2014, 68, 107–116. [Google Scholar] [CrossRef]
- Cannavà, C.; Crupi, V.; Ficarra, P.; Guardo, M.; Majolino, D.; Stancanelli, R.; Venuti, V. Physicochemical characterization of coumestrol/β-cyclodextrins inclusion complexes by UV–vis and FTIR-ATR spectroscopies. Vib. Spectrosc. 2008, 48, 172–178. [Google Scholar] [CrossRef]
- Irie, T.; Wakamatsu, K.; Arima, H.; Aritomi, H.; Uekama, K. Enhancing effects of cyclodextrins on nasal absorption of insulin in rats. Int. J. Pharm. 1992, 84, 129–139. [Google Scholar] [CrossRef]
- Nazari, K.; Kontogiannidou, E.; Haj Ahmad, R.; Andreadis, D.; Rasekh, M.; Bouropoulos, N.; van Der Merwe, S.M.; Chang, M.W.; Fatouros, D.G.; Ahmad, Z. Fibrous polymeric buccal film formulation, engineering and bio-interface assessment. Eur. Polym. J. 2017, 97, 147–157. [Google Scholar] [CrossRef]
- McCormack, B.; Gregoriadis, G. Comparative studies of the fate of free and liposome-entrapped hydroxypropyl-β-cyclodextrin/drug complexes after intravenous injection into rats: Implications in drug delivery. Biochim. Et Biophys. Acta (BBA) Gen. Subj. 1996, 1291, 237–244. [Google Scholar] [CrossRef]

| Samples | Drug Content (%) ± SD | Encapsulation Efficiency (%) |
|---|---|---|
| β-CD | 19.34 ± 0.93 | 85.57% |
| Me-β-CD | 18.49 ± 0.92 | 96.51% |
| HP-β-CD | 20.58 ± 0.95 | 94.66% |
| Samples (Tablets) | Korsmeyer-Peppas Model | First Order Model | |||
|---|---|---|---|---|---|
| k | n | R2 | k | R2 | |
| β-CD | 9.64 ± 0.05 | 0.49 ± 0.04 | 0.97 | 0.03 ± 0.002 | 0.99 |
| Me-β-CD | 20.17 ± 0.03 | 0.34 ± 0.02 | 0.91 | 0.05 ± 0.007 | 0.98 |
| HP-β-CD | 9.88 ± 0.07 | 0.49 ± 0.08 | 0.93 | 0.03 ± 0.003 | 0.99 |
| Samples | Jss (μg/cm2 h) | Papp (× 10−3) (cm/h) |
|---|---|---|
| β-CD | 1.165 ± 0.44 | 0.21 ± 0.08 |
| Me-β-CD | 2.405 ± 0.87 | 0.42 ± 0.12 |
| HP-β-CD | 1.850 ± 0.43 | 0.30 ± 0.07 |
| β-CD (tablet) | 3.019 ± 0.68 | 0.51 ± 0.14 |
| Me-β-CD (tablet) | 7.778 ± 1.69 | 1.26 ± 0.35 |
| HP-β-CD (tablet) | 5.438 ± 1.04 | 0.94 ± 0.34 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kontogiannidou, E.; Ferrari, M.; Deligianni, A.-D.; Bouropoulos, N.; Andreadis, D.A.; Sorrenti, M.; Catenacci, L.; Nazari, K.; Arshad, M.S.; Chang, M.-W.; et al. In Vitro and Ex Vivo Evaluation of Tablets Containing Piroxicam-Cyclodextrin Complexes for Buccal Delivery. Pharmaceutics 2019, 11, 398. https://doi.org/10.3390/pharmaceutics11080398
Kontogiannidou E, Ferrari M, Deligianni A-D, Bouropoulos N, Andreadis DA, Sorrenti M, Catenacci L, Nazari K, Arshad MS, Chang M-W, et al. In Vitro and Ex Vivo Evaluation of Tablets Containing Piroxicam-Cyclodextrin Complexes for Buccal Delivery. Pharmaceutics. 2019; 11(8):398. https://doi.org/10.3390/pharmaceutics11080398
Chicago/Turabian StyleKontogiannidou, Eleni, Martina Ferrari, Asteria-Danai Deligianni, Nikolaos Bouropoulos, Dimitrios A. Andreadis, Milena Sorrenti, Laura Catenacci, Kazem Nazari, Muhammad Sohail Arshad, Ming-Wei Chang, and et al. 2019. "In Vitro and Ex Vivo Evaluation of Tablets Containing Piroxicam-Cyclodextrin Complexes for Buccal Delivery" Pharmaceutics 11, no. 8: 398. https://doi.org/10.3390/pharmaceutics11080398
APA StyleKontogiannidou, E., Ferrari, M., Deligianni, A.-D., Bouropoulos, N., Andreadis, D. A., Sorrenti, M., Catenacci, L., Nazari, K., Arshad, M. S., Chang, M.-W., Ahmad, Z., & Fatouros, D. G. (2019). In Vitro and Ex Vivo Evaluation of Tablets Containing Piroxicam-Cyclodextrin Complexes for Buccal Delivery. Pharmaceutics, 11(8), 398. https://doi.org/10.3390/pharmaceutics11080398