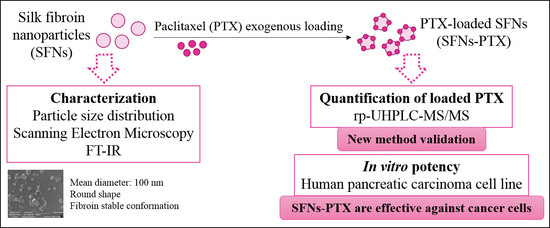
Paclitaxel-Loaded Silk Fibroin Nanoparticles: Method Validation by UHPLC-MS/MS to Assess an Exogenous Approach to Load Cytotoxic Drugs
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Silk Fibroin Extraction and Nanoparticles Preparation
2.3. SFNs Characterization
2.3.1. Particle Size Distribution
2.3.2. Scanning Electron Microscopy (SEM)
2.3.3. Fourier Transform Infrared (FT-IR) Spectroscopy
2.4. Nanoparticles Loading with Paclitaxel
2.5. Paclitaxel Analytical Determination
2.5.1. Chromatography
2.5.2. Mass Spectrometry
2.5.3. Stock, Working Standard Solutions and Quality Control Samples
2.5.4. Validation Study
Calibration Standards and Lower Limit of Quantification
Precision, Accuracy and Recovery
Stability
2.6. Drug Loading and Loading Efficiency Evaluation
2.7. In Vitro Potency Test of PTX, SFNs-PTX and SFNs on Cancer Cells
3. Results
3.1. SFNs Characterization
3.2. Paclitaxel Analytical Determination
3.2.1. Linearity
3.2.2. Precision, Accuracy and LOQ
3.2.3. Recovery
3.2.4. Stability
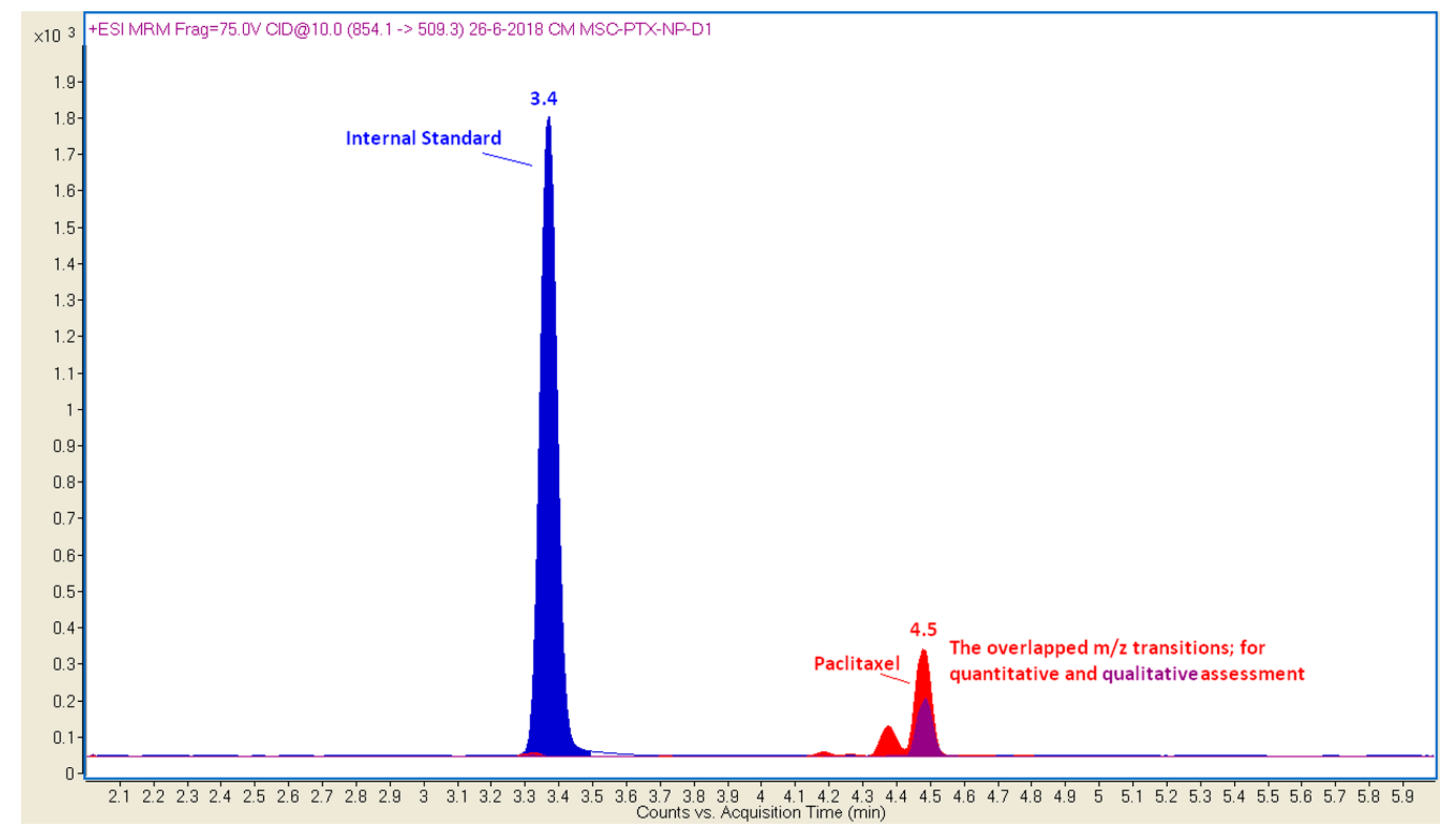
3.2.5. Chromatography Mass Spectrometry
3.3. Drug Loading and Loading Efficiency in SFNs-PTX
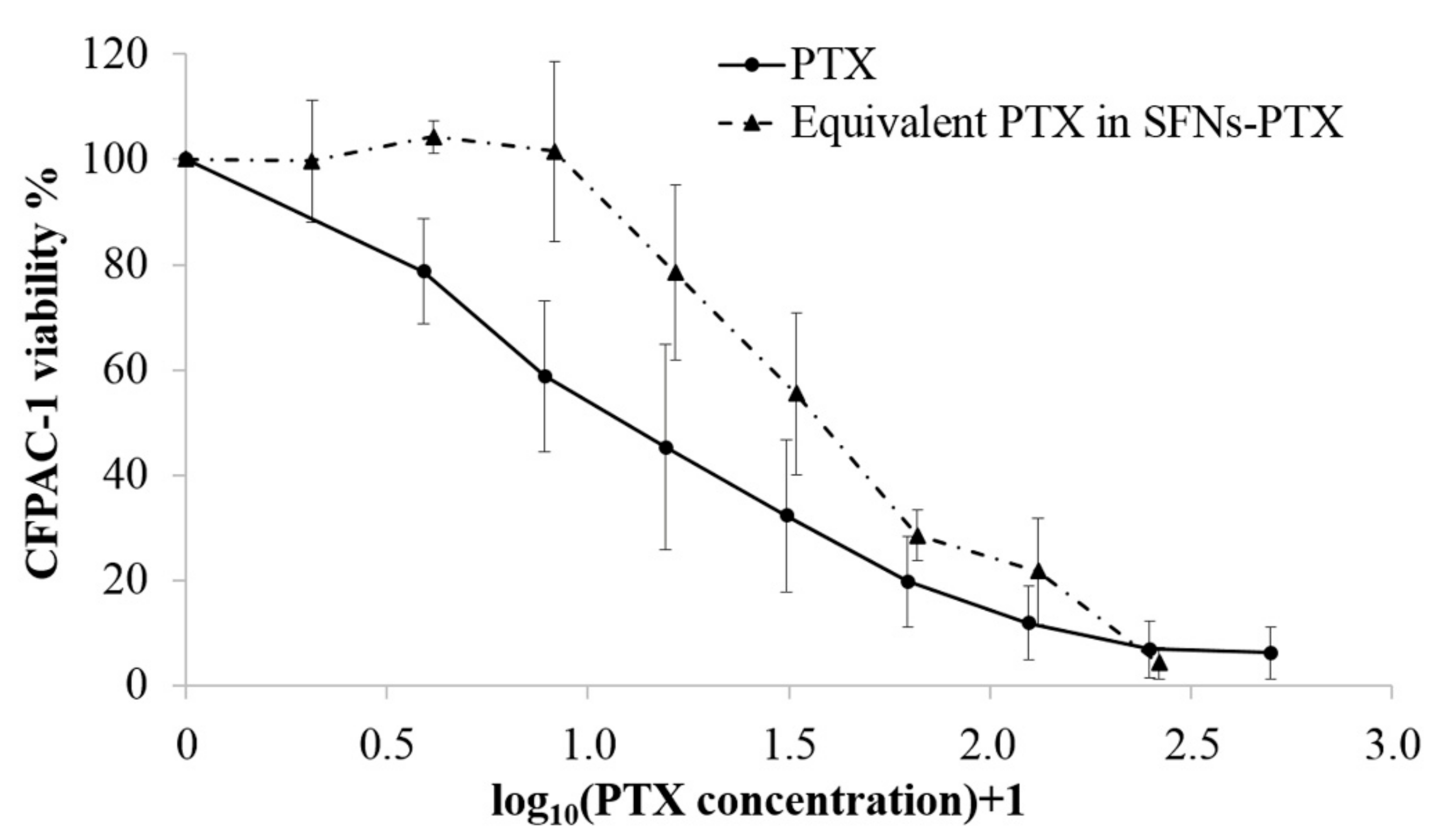
3.4. Potency Test on Tumor Cell Lines
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SF | Silk Fibroin |
| SFNs | Silk Fibroin Nanoparticles |
| PTX | Paclitaxel |
| SFNs-PTX | Silk Fibroin Nanoparticles loaded with Paclitaxel |
| rp-UHPLC-MS/MS | reversed phase liquid chromatography coupled to tandem mass spectrometry |
| LOQ | limit of quantification |
| LOD | limit of detection |
| EVs | Extracellular Vesicles |
| MSC-EVs | Mesenchymal Stem Cell-derived Extracellular Vesicles |
| API | Active Pharmaceutical Ingredient |
| PBS | phosphate buffered saline |
| SEM | Scanning Electron Microscopy |
| NTA | Nano Tracking Analysis |
| DLS | Dynamic Light Scattering |
| PDI | Polydispersity Index |
| FT-IR | Fourier Transform Infrared |
| QCs | Quality control samples |
| HiP | High-Performance Autosampler |
| SRM | Selected Reaction Monitoring |
| LLOQ | Lowest Level of Quantification |
| CV | Coefficient of Variation |
| RE | Recovery Extraction |
| EE | Encapsulation (Loading) Efficiency |
| CFPAC-1 | Human pancreatic carcinoma cell line |
| SD | Standard Deviation |
| RGD | arginine-glycine-aspartic acid sequence |
References
- Crivelli, B.; Perteghella, S.; Bari, E.; Sorrenti, M.; Tripodo, G.; Chlapanidas, T.; Torre, M.L. Silk nanoparticles: From inert supports to bioactive natural carriers for drug delivery. Soft Matter 2018, 14, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Mandracchia, D.; Rosato, A.; Trapani, A.; Chlapanidas, T.; Montagner, I.M.; Perteghella, S.; Di Franco, C.; Torre, M.L.; Trapani, G.; Tripodo, G. Design, synthesis and evaluation of biotin decorated inulin-based polymeric micelles as long-circulating nanocarriers for targeted drug delivery. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1245–1254. [Google Scholar] [CrossRef]
- Tripodo, G.; Chlapanidas, T.; Perteghella, S.; Vigani, B.; Mandracchia, D.; Trapani, A.; Galuzzi, M.; Tosca, M.C.; Antonioli, B.; Gaetani, P.; et al. Mesenchymal stromal cells loading curcumin-INVITE-micelles: A drug delivery system for neurodegenerative diseases. Coll. Surf. B Biointerf. 2015, 125, 300–308. [Google Scholar] [CrossRef]
- Mandracchia, D.; Trapani, A.; Perteghella, S.; Sorrenti, M.; Catenacci, L.; Torre, M.L.; Trapani, G.; Tripodo, G. pH-sensitive inulin-based nanomicelles for intestinal site-specific and controlled release of celecoxib. Carbohydr. Polym. 2018, 181, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef]
- He, X.; Chen, X.L.; Liu, L.S.; Zhang, Y.; Lu, Y.F.; Zhang, Y.J.; Chen, Q.J.; Ruan, C.H.; Guo, Q.; Li, C.; et al. Sequentially Triggered Nanoparticles with Tumor Penetration and Intelligent Drug Release for Pancreatic Cancer Therapy. Adv. Sci. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tian, J.; Wu, A.Q.; Wang, J.M.; Ge, C.C.; Sun, Z.L. Self-assembled silk fibroin nanoparticles loaded with binary drugs in the treatment of breast carcinoma. Int. J. Nanomed. 2016, 11, 4373–4380. [Google Scholar] [CrossRef]
- Lu, Y.; Li, Y.; Wu, W. Injected nanocrystals for targeted drug delivery. Acta Pharm. Sin. B 2016, 6, 106–113. [Google Scholar] [CrossRef]
- Crivelli, B.; Chlapanidas, T.; Perteghella, S.; Lucarelli, E.; Pascucci, L.; Brini, A.T.; Ferrero, I.; Marazzi, M.; Pessina, A.; Torre, M.L.; et al. Mesenchymal stem/stromal cell extracellular vesicles: From active principle to next generation drug delivery system. J. Control. Release 2017, 262, 104–117. [Google Scholar] [CrossRef]
- Bari, E.; Perteghella, S.; Di Silvestre, D.; Sorlini, M.; Catenacci, L.; Sorrenti, M.; Marrubini, G.; Rossi, R.; Tripodo, G.; Mauri, P.; et al. Pilot Production of Mesenchymal Stem/Stromal Freeze-Dried Secretome for Cell-Free Regenerative Nanomedicine: A Validated GMP-Compliant Process. Cells 2018, 7, 190. [Google Scholar]
- Bari, E.; Perteghella, S.; Catenacci, L.; Sorlini, M.; Croce, S.; Mantelli, M.; Avanzini, M.A.; Sorrenti, M.; Torre, M.L. Freeze-dried and GMP-compliant pharmaceuticals containing mesenchymal exosomes for acellular MSC immunomodulant therapy. Nanomedicine 2019. [Google Scholar] [CrossRef] [PubMed]
- Pascucci, L.; Cocce, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Vigano, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: A new approach for drug delivery. J. Control. Release 2014, 192, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Perteghella, S.; Crivelli, B.; Catenacci, L.; Sorrenti, M.; Bruni, G.; Necchi, V.; Vigani, B.; Sorlini, M.; Torre, M.L.; Chlapanidas, T. Stem cell-extracellular vesicles as drug delivery systems: New frontiers for silk/curcumin nanoparticles. Int. J. Pharm. 2017, 520, 86–97. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.; Jeong, M.; Lee, H.; Goh, U.; Kim, H.; Kim, B.; Park, J.H. Liposome-Based Engineering of Cells to Package Hydrophobic Compounds in Membrane Vesicles for Tumor Penetration. Nano Lett. 2015, 15, 2938–2944. [Google Scholar] [CrossRef]
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular vesicles for drug delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.S.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, L.K.; Chen, J.L.; Wang, L.S.; Gui, X.X.; Ran, J.S.; Xu, G.W.; Zhao, H.S.; Zeng, M.F.; Ji, J.F.; et al. Silk Fibroin Biomaterial Shows Safe and Effective Wound Healing in Animal Models and a Randomized Controlled Clinical Trial. Adv. Healthc. Mater. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.S.; Han, G.C.; Yan, S.Q.; Zhang, Q. 3D Printing of Silk Fibroin for Biomedical Applications. Materials 2019, 12, 504. [Google Scholar]
- Holland, C.; Numata, K.; Rnjak-Kovacina, J.; Seib, F.P. The Biomedical Use of Silk: Past, Present, Future. Adv. Healthc. Mater. 2019, 8. [Google Scholar] [CrossRef]
- Mwangi, T.K.; Bowles, R.D.; Tainter, D.M.; Bell, R.D.; Kaplan, D.L.; Setton, L.A. Synthesis and characterization of silk fibroin microparticles for intra-articular drug delivery. Int. J. Pharm. 2015, 485, 7–14. [Google Scholar] [CrossRef]
- Nosenko, M.A.; Moysenovich, A.M.; Zvartsev, R.V.; Arkhipova, A.Y.; Zhdanova, A.S.; Agapov, I.I.; Vasilieva, T.V.; Bogush, V.G.; Debabov, V.G.; Nedospasov, S.A.; et al. Novel Biodegradable Polymeric Microparticles Facilitate Scarless Wound Healing by Promoting Re-epithelialization and Inhibiting Fibrosis. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Farago, S.; Lucconi, G.; Perteghella, S.; Vigani, B.; Tripodo, G.; Sorrenti, M.; Catenacci, L.; Boschi, A.; Faustini, M.; Vigo, D.; et al. A dry powder formulation from silk fibroin microspheres as a topical auto-gelling device. Pharm. Dev. Technol. 2016, 21, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Lv, B.H.; Tan, W.; Zhu, C.C.; Shang, X.J.; Zhang, L. Properties of a Stable and Sustained-Release Formulation of Recombinant Human Parathyroid Hormone (rhPTH) with Chitosan and Silk Fibroin Microparticles. Med. Sci. Monit. 2018, 24, 7532–7540. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Li, Y.; Xie, M.B. Silk Fibroin-Based Nanoparticles for Drug Delivery. Int. J. Mol. Sci. 2015, 16, 4880–4903. [Google Scholar] [PubMed]
- Wu, P.Y.; Liu, Q.; Li, R.T.; Wang, J.; Zhen, X.; Yue, G.F.; Wang, H.Y.; Cui, F.B.; Wu, F.L.; Yang, M.; et al. Facile Preparation of Paclitaxel Loaded Silk Fibroin Nanoparticles for Enhanced Antitumor Efficacy by Locoregional Drug Delivery. ACS Appl. Mater. Interf. 2013, 5, 12638–12645. [Google Scholar] [CrossRef] [PubMed]
- Subia, B.; Chandra, S.; Talukdar, S.; Kundu, S.C. Folate conjugated silk fibroin nanocarriers for targeted drug delivery. Integr. Biol. 2014, 6, 203–214. [Google Scholar] [CrossRef]
- Crivelli, B.; Bari, E.; Perteghella, S.; Catenacci, L.; Sorrenti, M.; Mocchi, M.; Farago, S.; Tripodo, G.; Prina-Mello, A.; Torre, M.L. Silk fibroin nanoparticles for celecoxib and curcumin delivery: ROS-scavenging and anti-inflammatory activities in an in vitro model of osteoarthritis. Eur. J. Pharm. Biopharm. 2019, 137, 37–45. [Google Scholar] [CrossRef]
- Kim, D.W.; Hwang, H.S.; Kim, D.S.; Sheen, S.H.; Heo, D.H.; Hwang, G.; Kang, S.H.; Kweon, H.; Jo, Y.Y.; Kang, S.W.; et al. Effect of silk fibroin peptide derived from silkworm Bombyx mori on the anti-inflammatory effect of Tat-SOD in a mice edema model. BMB Rep. 2011, 44, 787–792. [Google Scholar] [CrossRef]
- Lozano-Perez, A.A.; Rodriguez-Nogales, A.; Ortiz-Cullera, V.; Algieri, F.; Garrido-Mesa, J.; Zorrilla, P.; Rodriguez-Cabezas, M.E.; Garrido-Mesa, N.; Utrilla, M.P.; De Matteis, L.; et al. Silk fibroin nanoparticles constitute a vector for controlled release of resveratrol in an experimental model of inflammatory bowel disease in rats. Int. J. Nanomed. 2014, 9, 4507–4520. [Google Scholar] [CrossRef]
- Sottani, C.; Minoia, C.; Colombo, A.; Zucchetti, M.; Dincalci, M.; Fanelli, R. Structural characterization of mono- and dihydroxylated metabolites of paclitaxel in rat bile using liquid chromatography ion spray tandem mass spectrometry. Rapid Commun. Mass Spectrom. 1997, 11, 1025–1032. [Google Scholar] [CrossRef]
- Sottani, C.; Minoia, C.; D’Incalci, M.; Paganini, M.; Zucchetti, M. High-performance liquid chromatography tandem mass spectrometry procedure with automated solid phase extraction sample preparation for the quantitative determination of paclitaxel (Taxol (R)) in human plasma. Rapid Commun. Mass Spectrom. 1998, 12, 251–255. [Google Scholar] [CrossRef]
- Zhang, W.; Dutschman, G.E.; Li, X.; Cheng, Y.C. Quantitation of paclitaxel and its two major metabolites using a liquid chromatography-electrospray ionization tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 2018–2022. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, J.R.; Qu, J.; Straubinger, N.L.; Straubinger, R.M. Highly selective and sensitive assay for paclitaxel accumulation by tumor cells based on selective solid phase extraction and micro-flow liquid chromatography coupled to mass spectrometry. Analyst 2008, 133, 1742–1748. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Johnson, J.L.; Khan, S.; Ahmad, A.; Ahmad, I. Paclitaxel quantification in mouse plasma and tissues containing liposome-entrapped paclitaxel by liquid chromatography-tandem mass spectrometry: Application to a pharmacokinetics study. Anal. Biochem. 2005, 336, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Baati, T.; Schembri, T.; Villard, C.; Correard, F.; Braguer, D.; Esteve, M.A. An ultrasensitive LC-MS/MS method with liquid phase extraction to determine paclitaxel in both cell culture medium and lysate promising quantification of drug nanocarriers release in vitro. J. Pharm. Biomed. Anal. 2015, 115, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Braal, C.L.; de Bruijn, P.; Atrafi, F.; van Geijn, M.; Rijcken, C.J.F.; Mathijssen, R.H.J.; Koolen, S.L.W. A new method for the determination of total and released docetaxel from docetaxel-entrapped core-crosslinked polymeric micelles (CriPec (R)) by LC-MS/MS and its clinical application in plasma and tissues in patients with various tumours. J. Pharm. Biomed. Anal. 2018, 161, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Furman, C.; Carpentier, R.; Barczyk, A.; Chavatte, P.; Betbeder, D.; Lipka, E. Development and validation of a reversed-phase HPLC method for the quantification of paclitaxel in different PLGA nanocarriers. Electrophoresis 2017, 38, 2536–2541. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Preat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Maguire, C.M.; Sillence, K.; Roesslein, M.; Hannell, C.; Suarez, G.; Sauvain, J.J.; Capracotta, S.; Contal, S.; Cambier, S.; El Yamani, N.; et al. Benchmark of Nanoparticle Tracking Analysis on Measuring Nanoparticle Sizing and Concentration. J. Micro Nano-Manuf. 2017, 5. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services Food and Drug Administration. Bioanalytical Method Validation—Guidance for Industry; U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM): Rockville, MD, USA, 2001. [Google Scholar]
- Pessina, A.; Bonomi, A.; Cocce, V.; Invernici, G.; Navone, S.; Cavicchini, L.; Sisto, F.; Ferrari, M.; Vigano, L.; Locatelli, A.; et al. Mesenchymal Stromal Cells Primed with Paclitaxel Provide a New Approach for Cancer Therapy. PLoS ONE 2011, 6, e28321. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 9. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 5. [Google Scholar] [CrossRef]
- Jimenez-Amezcua, I.; Carmona, F.J.; Romero-Garcia, I.; Quiros, M.; Cenis, J.L.; Lozano-Perez, A.A.; Maldonado, C.R.; Barea, E. Silk fibroin nanoparticles as biocompatible nanocarriers of a novel light-responsive CO-prodrug. Dalton Trans. 2018, 47, 10434–10438. [Google Scholar] [CrossRef]
- Maitz, M.F.; Sperling, C.; Wongpinyochit, T.; Herklotz, M.; Werner, C.; Seib, F.P. Biocompatibility assessment of silk nanoparticles: Hemocompatibility and internalization by human blood cells. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2633–2642. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.J.; Shao, Z.Z.; Chen, X. Paclitaxel-loaded silk fibroin nanospheres. J. Biomed. Mater. Res. Part A 2012, 100A, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.Y.; Wu, P.Y.; Sha, H.Z.; Qian, H.Q.; Wang, Q.; Cheng, L.; Yang, Y.; Yang, M.; Liu, B.R. Anti-EGFR-iRGD recombinant protein conjugated silk fibroin nanoparticles for enhanced tumor targeting and antitumor efficiency. Oncotargets Ther. 2016, 9, 3153–3162. [Google Scholar] [CrossRef]
- Wu, P.Y.; Liu, Q.; Wang, Q.; Qian, H.Q.; Yu, L.X.; Liu, B.R.; Li, R.T. Novel silk fibroin nanoparticles incorporated silk fibroin hydrogel for inhibition of cancer stem cells and tumor growth. Int. J. Nanomed. 2018, 13, 5405–5418. [Google Scholar] [CrossRef]
- Schnittert, J.; Bansal, R.; Storm, G.; Prakash, J. Integrins in wound healing, fibrosis and tumor stroma: High potential targets for therapeutics and drug delivery. Adv. Drug Deliv. Rev. 2018, 129, 37–53. [Google Scholar] [CrossRef]
- Rodriguez-Nogales, A.; Algieri, F.; De Matteis, L.; Lozano-Perez, A.A.; Garrido-Mesa, J.; Vezza, T.; de la Fuente, J.M.; Cenis, J.L.; Galvez, J.; Rodriguez-Cabezas, M.E. Intestinal anti-inflammatory effects of RGD-functionalized silk fibroin nanoparticles in trinitrobenzenesulfonic acid-induced experimental colitis in rats. Int. J. Nanomed. 2016, 11, 5945–5958. [Google Scholar] [CrossRef]

| Matrix | Conc. Range (ng/mL) | Equation | Weighting Factor |
|---|---|---|---|
| Mobile phase | 5–250 | Ymean = 0.00128 ± (0.002)x − 0.0008 ± (0.001) | 1/x |
| SFN matrix | 5–250 | Ymean = 0.00098 ± (0.0002)x − 0.0022 ± (0.002) | 1/x |
| LLOQ | QC1 | QC2 | QC3 | ||
|---|---|---|---|---|---|
| Actual concentrations (ng/mL) | 5.00 | 10.00 | 50.00 | 100.00 | |
| Intraday | |||||
| Measured concentrations | |||||
| Day1 | 4.66 | 9.15 | 51.66 | 100.32 | |
| 4.87 | 9.55 | 48.53 | 102.78 | ||
| 5.17 | 9.76 | 52.72 | 102.86 | ||
| 5.07 | 9.29 | 52.59 | 99.55 | ||
| Mean ± SD (QCs; n = 4) | 4.34 ± 0.022 | 9.44 ± 0.27 | 51.37 ± 1.95 | 101.37 ± 1.69 | |
| Precision (%) | 4.52 | 2.90 | 3.79 | 1.67 | |
| Accuracy (%) | 98.85 | 99.57 | 101.48 | 100.99 | |
| Day2 | 4.77 | 11.07 | 55.85 | 89.64 | |
| 4.67 | 11.82 | 57.57 | 89.78 | ||
| 5.02 | 11.72 | 55.85 | 89.86 | ||
| 4.90 | 11.96 | 54.48 | 99.47 | ||
| Mean ± SD (QCs; n = 4) | 4.84 ± 0.16 | 11.64 ± 0.39 | 55.94 ± 0.99 | 92.18 ± 4.85 | |
| Precision (%) | 3.21 | 3.38 | 1.77 | 5.27 | |
| Accuracy (%) | 96.75 | 116.43 | 111.87 | 93.66 | |
| Day3 | 4.49 | 9.50 | 57.77 | 114.05 | |
| 4.69 | 9.98 | 52.35 | 100.41 | ||
| 5.49 | 9.44 | 53.81 | 104.46 | ||
| 4.99 | 10.43 | 52.63 | 112.98 | ||
| Mean ± SD (QCs; n = 4) | 4.92 ± 0.43 | 9.84 ± 0.46 | 54.14 ± 2.80 | 107.98 ± 6.62 | |
| Precision (%) | 8.84 | 4.72 | 5.18 | 6.13 | |
| Accuracy (%) | 98.36 | 98.38 | 108.28 | 107.98 | |
| Inter-day | |||||
| Mean ± SD (n = 12) | 4.90 ± 0.27 | 10.31 ± 1.06 | 53.82 ± 2.65 | 100.5 ± 7.60 | |
| Precision (%) | 5.55 | 10.29 | 4.92 | 7.84 | |
| Accuracy (%) | 98.00 | 104.80 | 107.60 | 100.50 | |
| Levels | Mean Integration Ratio ± Standard Deviation | ||||
|---|---|---|---|---|---|
| Mobile Phase | CV% | Matrix | CV% | RE% | |
| LLOQ | 0.0046 ± 0.0005 | 10.3 | 0.0037 ± 0.0007 | 19.6 | 81.20 |
| QC1 | 0.0093 ± 0.0011 | 11.4 | 0.0069 ± 0.0008 | 11.7 | 74.50 |
| QC2 | 0.054 ± 0.0065 | 12.2 | 0.038 ± 0.0016 | 4.2 | 70.50 |
| QC3 | 0.12 ± 0.11 | 9.8 | 0.099 ± 0.014 | 13.6 | 87.60 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perteghella, S.; Sottani, C.; Coccè, V.; Negri, S.; Cavicchini, L.; Alessandri, G.; Cottica, D.; Torre, M.L.; Grignani, E.; Pessina, A. Paclitaxel-Loaded Silk Fibroin Nanoparticles: Method Validation by UHPLC-MS/MS to Assess an Exogenous Approach to Load Cytotoxic Drugs. Pharmaceutics 2019, 11, 285. https://doi.org/10.3390/pharmaceutics11060285
Perteghella S, Sottani C, Coccè V, Negri S, Cavicchini L, Alessandri G, Cottica D, Torre ML, Grignani E, Pessina A. Paclitaxel-Loaded Silk Fibroin Nanoparticles: Method Validation by UHPLC-MS/MS to Assess an Exogenous Approach to Load Cytotoxic Drugs. Pharmaceutics. 2019; 11(6):285. https://doi.org/10.3390/pharmaceutics11060285
Chicago/Turabian StylePerteghella, Sara, Cristina Sottani, Valentina Coccè, Sara Negri, Loredana Cavicchini, Giulio Alessandri, Danilo Cottica, Maria Luisa Torre, Elena Grignani, and Augusto Pessina. 2019. "Paclitaxel-Loaded Silk Fibroin Nanoparticles: Method Validation by UHPLC-MS/MS to Assess an Exogenous Approach to Load Cytotoxic Drugs" Pharmaceutics 11, no. 6: 285. https://doi.org/10.3390/pharmaceutics11060285
APA StylePerteghella, S., Sottani, C., Coccè, V., Negri, S., Cavicchini, L., Alessandri, G., Cottica, D., Torre, M. L., Grignani, E., & Pessina, A. (2019). Paclitaxel-Loaded Silk Fibroin Nanoparticles: Method Validation by UHPLC-MS/MS to Assess an Exogenous Approach to Load Cytotoxic Drugs. Pharmaceutics, 11(6), 285. https://doi.org/10.3390/pharmaceutics11060285