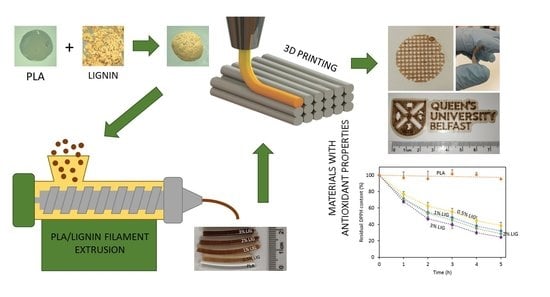
Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. LIG-PLA Pellets Production
2.3. Extrusion of Filaments
2.4. Fused Filament Fabrication
2.5. Material Characterization
2.5.1. Microscopy
2.5.2. Contact Angle Measurement
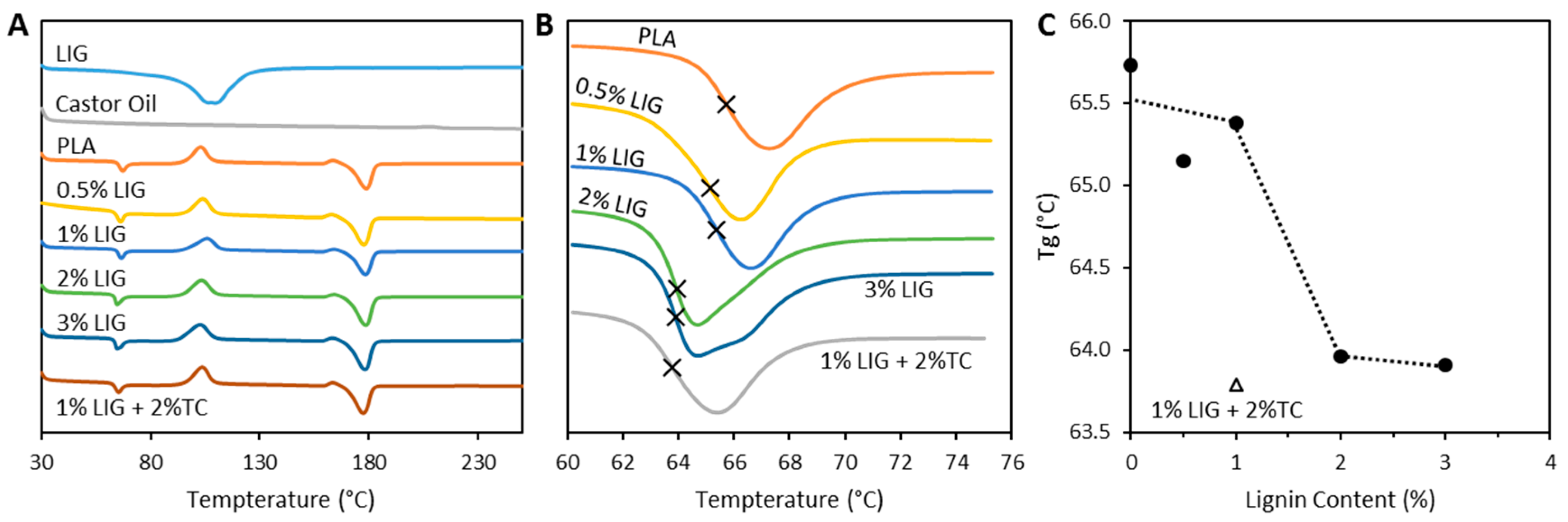
2.5.3. Thermal Properties
2.5.4. Stability Study
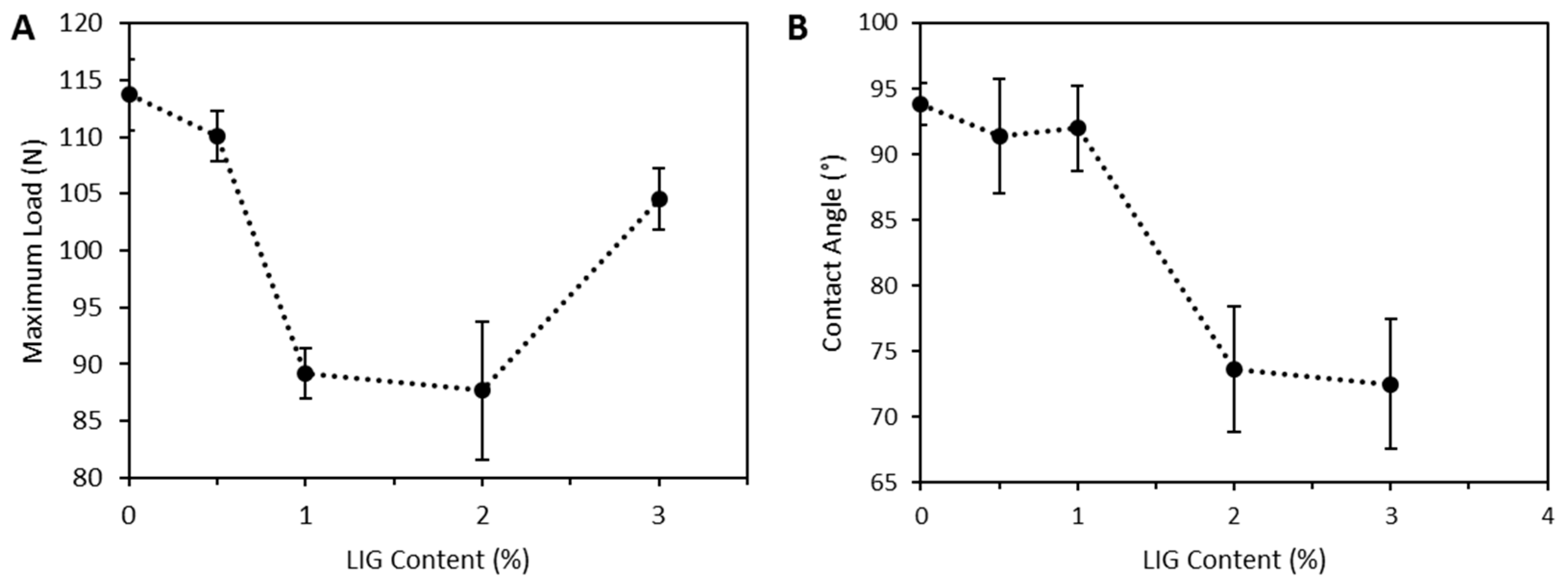
2.5.5. Mechanical Properties
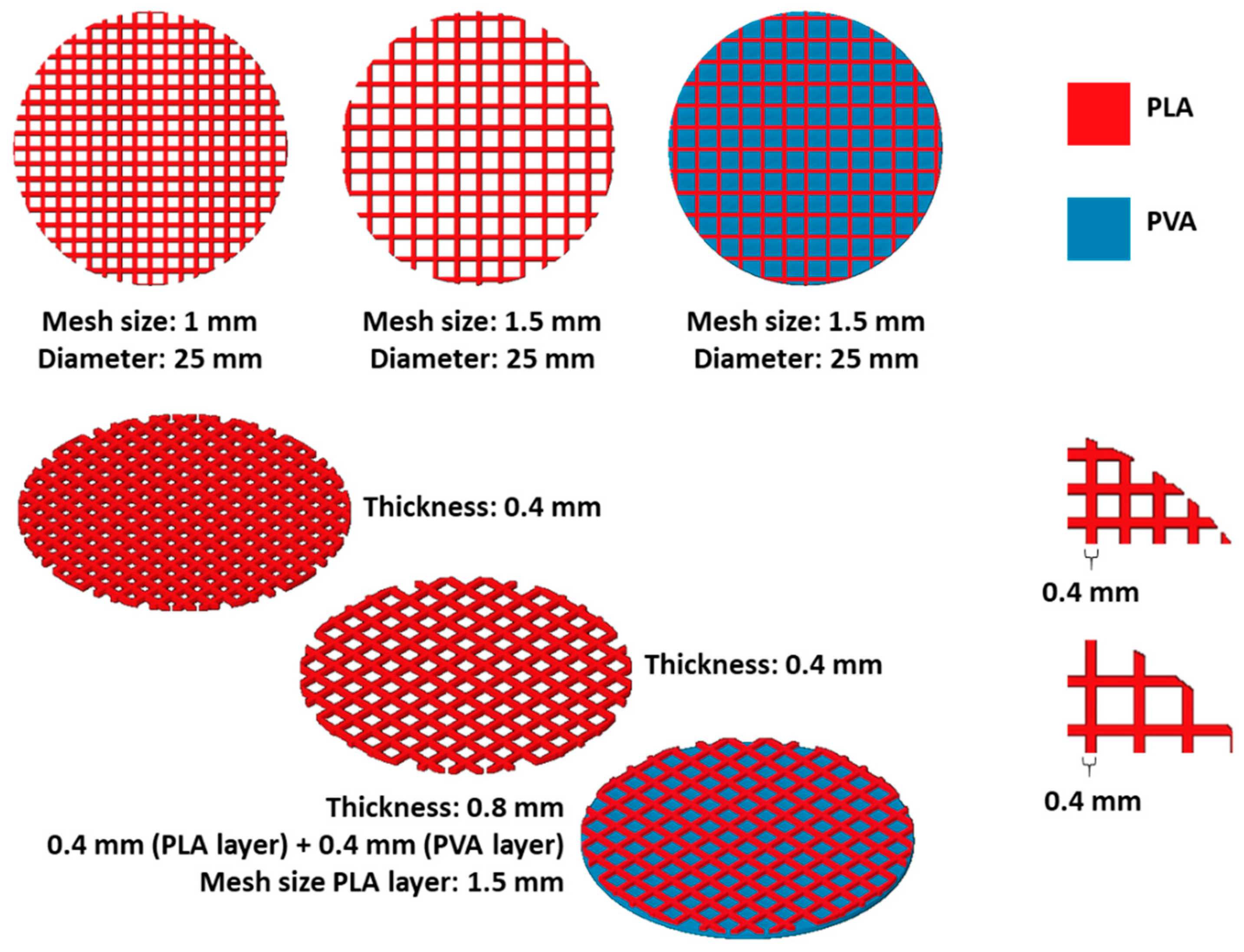
2.6. PLA-LIG Mesh Production and CUR Release for Potential Wound Healing Applications
2.7. Antioxidant Activity
2.8. Antimicrobial Properties
2.9. Statistical Analysis
3. Results
3.1. PLA and LIG Composite Material Preparation and Characterization
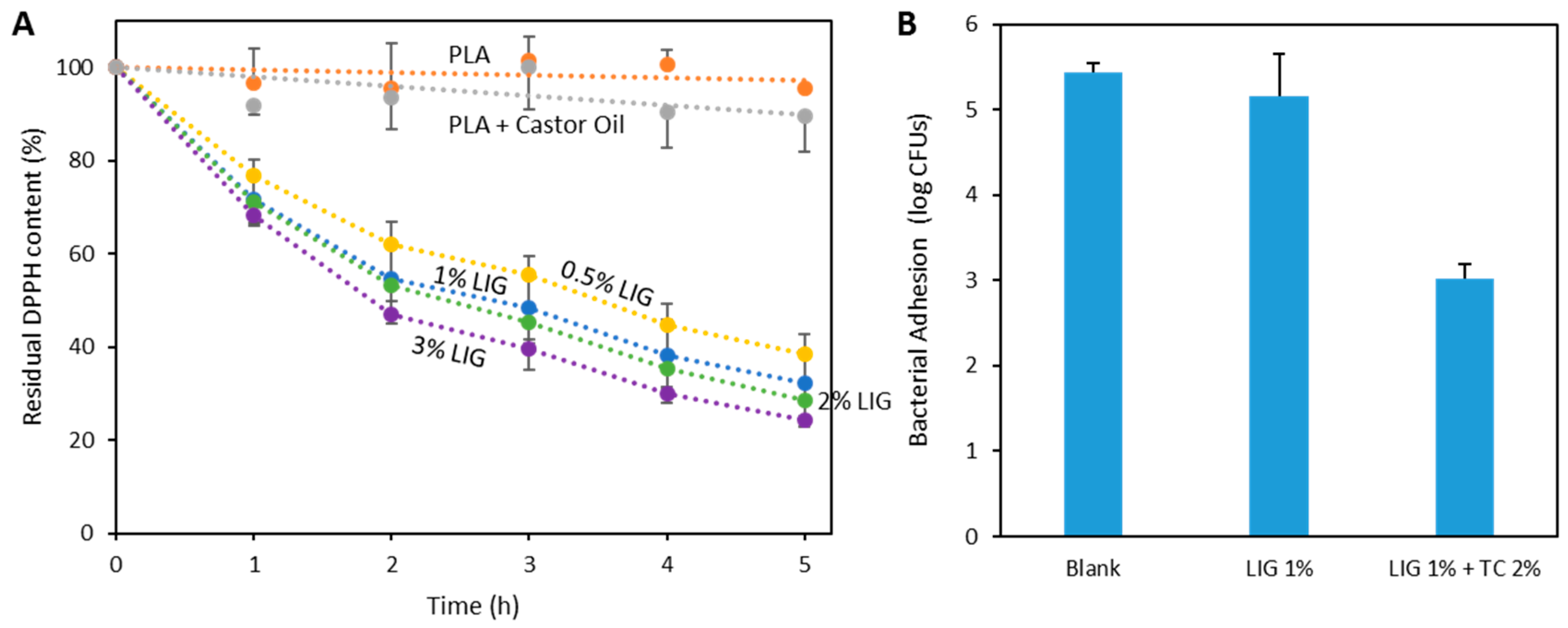
3.2. PLA and LIG Composite Antioxidant and Antimicrobial Properties
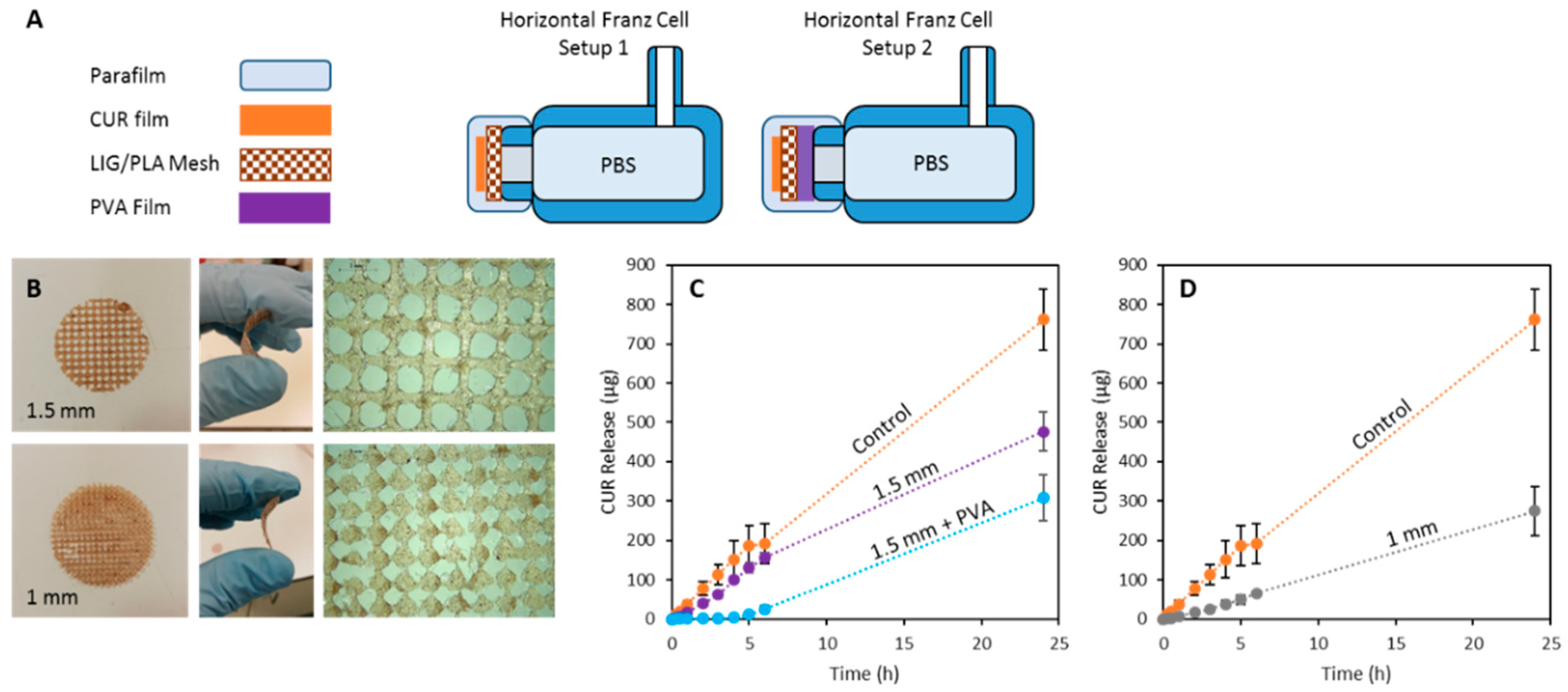
3.3. 3D Printed PLA and LIG Composite Meshes for Potential Wound Healing Applications
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Capel, A.J.; Rimington, R.P.; Lewis, M.P.; Christie, S.D.R. 3D Printing for Chemical, Pharmaceutical and Biological Applications. Nat. Rev. Chem. 2018, 2, 422–436. [Google Scholar] [CrossRef]
- McIlroy, C.; Olmsted, P.D. Disentanglement Effects on Welding Behaviour of Polymer Melts during the Fused-Filament-Fabrication Method for Additive Manufacturing. Polymer 2017, 123, 376–391. [Google Scholar] [CrossRef]
- Goyanes, A.; Buanz, A.B.; Basit, A.W.; Gaisford, S. Fused-Filament 3D Printing (3DP) for Fabrication of Tablets. Int. J. Pharm. 2014, 476, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Baumann, F.; Bugdayci, H.; Grunert, J.; Keller, F.; Roller, D. Influence of Slicing Tools on Quality of 3D Printed Parts. Comput. Aided Des. Appl. 2016, 13, 14–31. [Google Scholar] [CrossRef]
- Jamroz, W.; Szafraniec, J.; Kurek, M.; Jachowicz, R. 3D Printing in Pharmaceutical and Medical Applications—Recent Achievements and Challenges. Pharm. Res. 2018, 35, 176. [Google Scholar] [CrossRef] [PubMed]
- Trenfield, S.J.; Awad, A.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D Printing Pharmaceuticals: Drug Development to Frontline Care. Trends Pharmacol. Sci. 2018, 39, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Stewart, A.S.; DomÃnguez-Robles, J.; Donnelly, F.R.; Larrañeta, E. Implantable Polymeric Drug Delivery Devices: Classification, Manufacture, Materials, and Clinical Applications. Polymers 2018, 10, 1379. [Google Scholar] [CrossRef]
- Chiulan, I.; Frone, A.N.; Brandabur, C.; Panaitescu, D.M. Recent Advances in 3D Printing of Aliphatic Polyesters. Bioengineering 2017, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Farah, S.; Anderson, D.G.; Langer, R. Physical and Mechanical Properties of PLA, and their Functions in Widespread Applications—A Comprehensive Review. Adv. Drug Deliv. Rev. 2016, 107, 367–392. [Google Scholar] [CrossRef]
- Weisman, J.A.; Nicholson, J.C.; Tappa, K.; Jammalamadaka, U.; Wilson, C.G.; Mills, D.K. Antibiotic and Chemotherapeutic Enhanced Three-Dimensional Printer Filaments and Constructs for Biomedical Applications. Int. J. Nanomed. 2015, 10, 357–370. [Google Scholar] [PubMed]
- Barapatre, A.; Aadil, K.R.; Tiwary, B.N.; Jha, H. In Vitro Antioxidant and Antidiabetic Activities of Biomodified Lignin from Acacia Nilotica Wood. Int. J. Biol. Macromol. 2015, 75, 81–89. [Google Scholar] [CrossRef] [PubMed]
- van Lith, R.; Gregory, E.K.; Yang, J.; Kibbe, M.R.; Ameer, G.A. Engineering Biodegradable Polyester Elastomers with Antioxidant Properties to Attenuate Oxidative Stress in Tissues. Biomaterials 2014, 35, 8113–8122. [Google Scholar] [CrossRef] [PubMed]
- Dhall, S.; Do, D.; Garcia, M.; Wijesinghe, D.S.; Brandon, A.; Kim, J.; Sanchez, A.; Lyubovitsky, J.; Gallagher, S.; Nothnagel, E.A.; et al. A Novel Model of Chronic Wounds: Importance of Redox Imbalance and Biofilm-Forming Bacteria for Establishment of Chronicity. PLoS ONE 2014, 9, e109848. [Google Scholar] [CrossRef] [PubMed]
- Dhall, S.; Do, D.C.; Garcia, M.; Kim, J.; Mirebrahim, S.H.; Lyubovitsky, J.; Lonardi, S.; Nothnagel, E.A.; Schiller, N.; Martins-Green, M. Generating and Reversing Chronic Wounds in Diabetic Mice by Manipulating Wound Redox Parameters. J. Diabetes Res. 2014, 2014, 562625. [Google Scholar] [CrossRef] [PubMed]
- van Lith, R.; Baker, E.; Ware, H.; Yang, J.; Farsheed, A.C.; Sun, C.; Ameer, G. 3D-Printing Strong High-Resolution Antioxidant Bioresorbable Vascular Stents. Adv. Mater. Technol. 2016, 1, 1600138. [Google Scholar] [CrossRef]
- Lücking, T.H.; Sambale, F.; Schnaars, B.; Bulnes-Abundis, D.; Beutel, S.; Scheper, T. 3D-Printed Individual Labware in Biosciences by Rapid Prototyping: In Vitro Biocompatibility and Applications for Eukaryotic Cell Cultures. Eng. Life Sci. 2015, 15, 57–64. [Google Scholar] [CrossRef]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chua, Y.K.; Yap, Y.L.; Loh, X.J. Towards Lignin-Based Functional Materials in a Sustainable World. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent Advances in Green Hydrogels from Lignin: A Review. Int. J. Biol. Macromol. 2015, 72, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, R.; Espinosa, E.; Domínguez-Robles, J.; Loaiza, J.M.; Rodríguez, A. Isolation and Characterization of Lignocellulose Nanofibers from Different Wheat Straw Pulps. Int. J. Biol. Macromol. 2016, 92, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Li, Y.; Qian, Y.; Xiao, Y.; Du, S.; Qiu, X. Synergistic Antioxidant Performance of Lignin and Quercetin Mixtures. ACS Sustain. Chem. Eng. 2017, 5, 8424–8428. [Google Scholar] [CrossRef]
- Larrañeta, E.; Imízcoz, M.; Toh, J.X.; Irwin, N.J.; Ripolin, A.; Perminova, A.; Domíguez-Robles, J.; Rodríguez, A.; Donnelly, R.F. Synthesis and Characterization of Lignin Hydrogels for Potential Applications as Drug Eluting Antimicrobial Coatings for Medical Materials. ACS Sustain. Chem. Eng. 2018, 6, 9037–9046. [Google Scholar] [CrossRef] [PubMed]
- Pucciariello, R.; Bonini, C.; D’Auria, M.; Villani, V.; Giammarino, G.; Gorrasi, G. Polymer Blends of Steam-Explosion Lignin and Poly(Ε-Caprolactone) by High-Energy Ball Milling. J. Appl. Polym. Sci. 2008, 109, 309–313. [Google Scholar] [CrossRef]
- Dong, X.; Dong, M.; Lu, Y.; Turley, A.; Jin, T.; Wu, C. Antimicrobial and Antioxidant Activities of Lignin from Residue of Corn Stover to Ethanol Production. Ind. Crops Prod. 2011, 34, 1629–1634. [Google Scholar] [CrossRef]
- Kai, D.; Low, Z.W.; Liow, S.; Abdul Karim, A.; Ye, H.; Jin, G.; Li, K.; Loh, X.J. Development of Lignin Supramolecular Hydrogels with Mechanically Responsive and Self-Healing Properties. ACS Sustain. Chem. Eng. 2015, 3, 2160–2169. [Google Scholar] [CrossRef]
- Domínguez-Robles, J.; Sánchez, R.; Díaz-Carrasco, P.; Espinosa, E.; García-Domínguez, M.T.; Rodríguez, A. Isolation and Characterization of Lignins from Wheat Straw: Application as Binder in Lithium Batteries. Int. J. Biol. Macromol. 2017, 104, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Azadfar, M.; Gao, A.H.; Bule, M.V.; Chen, S. Structural Characterization of Lignin: A Potential Source of Antioxidants Guaiacol and 4-Vinylguaiacol. Int. J. Biol. Macromol. 2015, 75, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Zhang, K.; Jiang, L.; Wong, H.Z.; Li, Z.; Zhang, Z.; Loh, X.J. Sustainable and Antioxidant Lignin-Polyester Copolymers and Nanofibers for Potential Healthcare Applications. ACS Sustain. Chem. Eng. 2017, 5, 6016–6025. [Google Scholar] [CrossRef]
- Stewart, D. Lignin as a Base Material for Materials Applications: Chemistry, Application and Economics. Ind. Crops Prod. 2008, 27, 202–207. [Google Scholar] [CrossRef]
- Jönsson, A.S.; Wallberg, O. Cost Estimates of Kraft Lignin Recovery by Ultrafiltration. Desalination 2009, 237, 254–267. [Google Scholar] [CrossRef]
- Domínguez-Robles, J.; Peresin, M.S.; Tamminen, T.; Rodríguez, A.; Larrañeta, E.; Jääskeläinen, A.S. Lignin-Based Hydrogels with “super-Swelling” Capacities for Dye Removal. Int. J. Biol. Macromol. 2018, 115, 1249–1259. [Google Scholar] [CrossRef]
- Figueiredo, P.; Lintinen, K.; Kiriazis, A.; Hynninen, V.; Liu, Z.; Bauleth-Ramos, T.; Rahikkala, A.; Correia, A.; Kohout, T.; Sarmento, B.; et al. In Vitro Evaluation of Biodegradable Lignin-Based Nanoparticles for Drug Delivery and Enhanced Antiproliferation Effect in Cancer Cells. Biomaterials 2017, 121, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Erakovic, S.; Jankovic, A.; Tsui, G.C.; Tang, C.Y.; Miskovic-Stankovic, V.; Stevanovic, T. Novel Bioactive Antimicrobial Lignin Containing Coatings on Titanium obtained by Electrophoretic Deposition. Int. J. Mol. Sci. 2014, 15, 12294–12322. [Google Scholar] [CrossRef] [PubMed]
- Qazi, S.S.; Li, D.; Briens, C.; Berruti, F.; Abou-Zaid, M.M. Antioxidant Activity of the Lignins Derived from Fluidized-Bed Fast Pyrolysis. Molecules 2017, 22, 372. [Google Scholar] [CrossRef] [PubMed]
- Larrañeta, E.; Henry, M.; Irwin, N.J.; Trotter, J.; Perminova, A.A.; Donnelly, R.F. Synthesis and Characterization of Hyaluronic Acid Hydrogels Crosslinked using a Solvent-Free Process for Potential Biomedical Applications. Carbohydr. Polym. 2018, 181, 1194–1205. [Google Scholar] [CrossRef] [PubMed]
- McCoy, C.P.; Irwin, N.J.; Brady, C.; Jones, D.S.; Carson, L.; Andrews, G.P.; Gorman, S.P. An Infection-Responsive Approach to Reduce Bacterial Adhesion in Urinary Biomaterials. Mol. Pharm. 2016, 13, 2817–2822. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Neoh, K.G.; Shi, Z.; Kang, E.T.; Tambyah, P.A.; Chiong, E. Inhibition of Escherichia Coli and Proteus Mirabilis Adhesion and Biofilm Formation on Medical Grade Silicone Surface. Biotechnol. Bioeng. 2012, 109, 336–345. [Google Scholar] [CrossRef]
- Jones, D.S.; McGovern, J.G.; Woolfson, A.D.; Gorman, S.P. Role of Physiological Conditions in the Oropharynx on the Adherence of Respiratory Bacterial Isolates to Endotracheal Tube Poly(Vinyl Chloride). Biomaterials 1997, 18, 503–510. [Google Scholar] [CrossRef]
- Miles, A.A.; Misra, S.S.; Irwin, J.O. The Estimation of the Bactericidal Power of the Blood. J. Hyg. 1938, 38, 732–749. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Zhang, Y.; Yu, Z. Effect of Desulfonation of Lignosulfonate on the Properties of Poly(Lactic Acid)/Lignin Composites. Bioresources 2017, 12, 4810–4829. [Google Scholar] [CrossRef]
- Thompson, R.C.; Moore, C.J.; vom Saal, F.S.; Swan, S.H. Plastics, the Environment and Human Health: Current Consensus and Future Trends. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2009, 364, 2153–2166. [Google Scholar] [CrossRef] [PubMed]
- Moseley, R.; Walker, M.; Waddington, R.J.; Chen, W.Y. Comparison of the Antioxidant Properties of Wound Dressing Materials—Carboxymethylcellulose, Hyaluronan Benzyl Ester and Hyaluronan, Towards Polymorphonuclear Leukocyte-Derived Reactive Oxygen Species. Biomaterials 2003, 24, 1549–1557. [Google Scholar] [CrossRef]
- Reesi, F.; Minaiyan, M.; Taheri, A. A Novel Lignin-Based Nanofibrous Dressing Containing Arginine for Wound-Healing Applications. Drug Deliv. Transl. Res. 2018, 8, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jia, Y.; Liu, Z.; Miao, J. Influence of the Lignin Content on the Properties of Poly(Lactic Acid)/Lignin-Containing Cellulose Nanofibrils Composite Films. Polymers 2018, 10, 1013. [Google Scholar] [CrossRef]
- Li, H.; Legros, N.; Ton-That, M.; Rakotovelo, A. PLA-Thermoplastic Lignin Blends. Plast. Eng. 2013, 69, 60–64. [Google Scholar] [CrossRef]
- Kai, D.; Ren, W.; Tian, L.; Chee, P.L.; Liu, Y.; Ramakrishna, S.; Loh, X.J. Engineering Poly(Lactide)-Lignin Nanofibers with Antioxidant Activity for Biomedical Application. ACS Sustain. Chem. Eng. 2016, 4, 5268–5276. [Google Scholar] [CrossRef]
- Kariz, M.; Sernek, M.; Obućina, M.; Kuzman, M.K. Effect of Wood Content in FDM Filament on Properties of 3D Printed Parts. Mater. Today Comm. 2018, 14, 135–140. [Google Scholar] [CrossRef]
- Meullemiestre, A.; Kamal, I.; Maache-Rezzoug, Z.; Chemat, F.; Rezzoug, S.A. Antioxidant Activity and Total Phenolic Content of Oils Extracted from Pinus Pinaster Sawdust Waste. Screening of Different Innovative Isolation Techniques. Waste Biomass Valorization 2014, 5, 283–292. [Google Scholar] [CrossRef]
- Tejada, S.; Manayi, A.; Daglia, M.; Nabavi, S.F.; Sureda, A.; Hajheydari, Z.; Gortzi, O.; Pazoki-Toroudi, H.; Nabavi, S.M. Wound Healing Effects of Curcumin: A Short Review. Curr. Pharm. Biotechnol. 2016, 17, 1002–1007. [Google Scholar] [CrossRef]
- Ajalloueian, F.; Tavanai, H.; Hilborn, J.; Donzel-Gargand, O.; Leifer, K.; Wickham, A.; Arpanaei, A. Emulsion Electrospinning as an Approach to Fabricate PLGA/Chitosan Nanofibers for Biomedical Applications. Biomed. Res. Int. 2014, 2014, 475280. [Google Scholar] [CrossRef]
- Cherif Lahimer, M.; Ayed, N.; Horriche, J.; Belgaied, S. Characterization of Plastic Packaging Additives: Food Contact, Stability and Toxicity. Arab. J. Chem. 2017, 10, S1938–S1954. [Google Scholar] [CrossRef]
- Addivant’s Weston 705 Antioxidant Gains Food Contact Approval in Mercosur Region. Addit. Polym. 2018, 2018, 3–4. [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Robles, J.; Martin, N.K.; Fong, M.L.; Stewart, S.A.; Irwin, N.J.; Rial-Hermida, M.I.; Donnelly, R.F.; Larrañeta, E. Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications. Pharmaceutics 2019, 11, 165. https://doi.org/10.3390/pharmaceutics11040165
Domínguez-Robles J, Martin NK, Fong ML, Stewart SA, Irwin NJ, Rial-Hermida MI, Donnelly RF, Larrañeta E. Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications. Pharmaceutics. 2019; 11(4):165. https://doi.org/10.3390/pharmaceutics11040165
Chicago/Turabian StyleDomínguez-Robles, Juan, Niamh K. Martin, Mun Leon Fong, Sarah A. Stewart, Nicola J. Irwin, María Isabel Rial-Hermida, Ryan F. Donnelly, and Eneko Larrañeta. 2019. "Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications" Pharmaceutics 11, no. 4: 165. https://doi.org/10.3390/pharmaceutics11040165
APA StyleDomínguez-Robles, J., Martin, N. K., Fong, M. L., Stewart, S. A., Irwin, N. J., Rial-Hermida, M. I., Donnelly, R. F., & Larrañeta, E. (2019). Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications. Pharmaceutics, 11(4), 165. https://doi.org/10.3390/pharmaceutics11040165