Simulation of Airway Deposition of an Aerosol Drug in COPD Patients
Abstract
1. Introduction
2. Materials and Methods
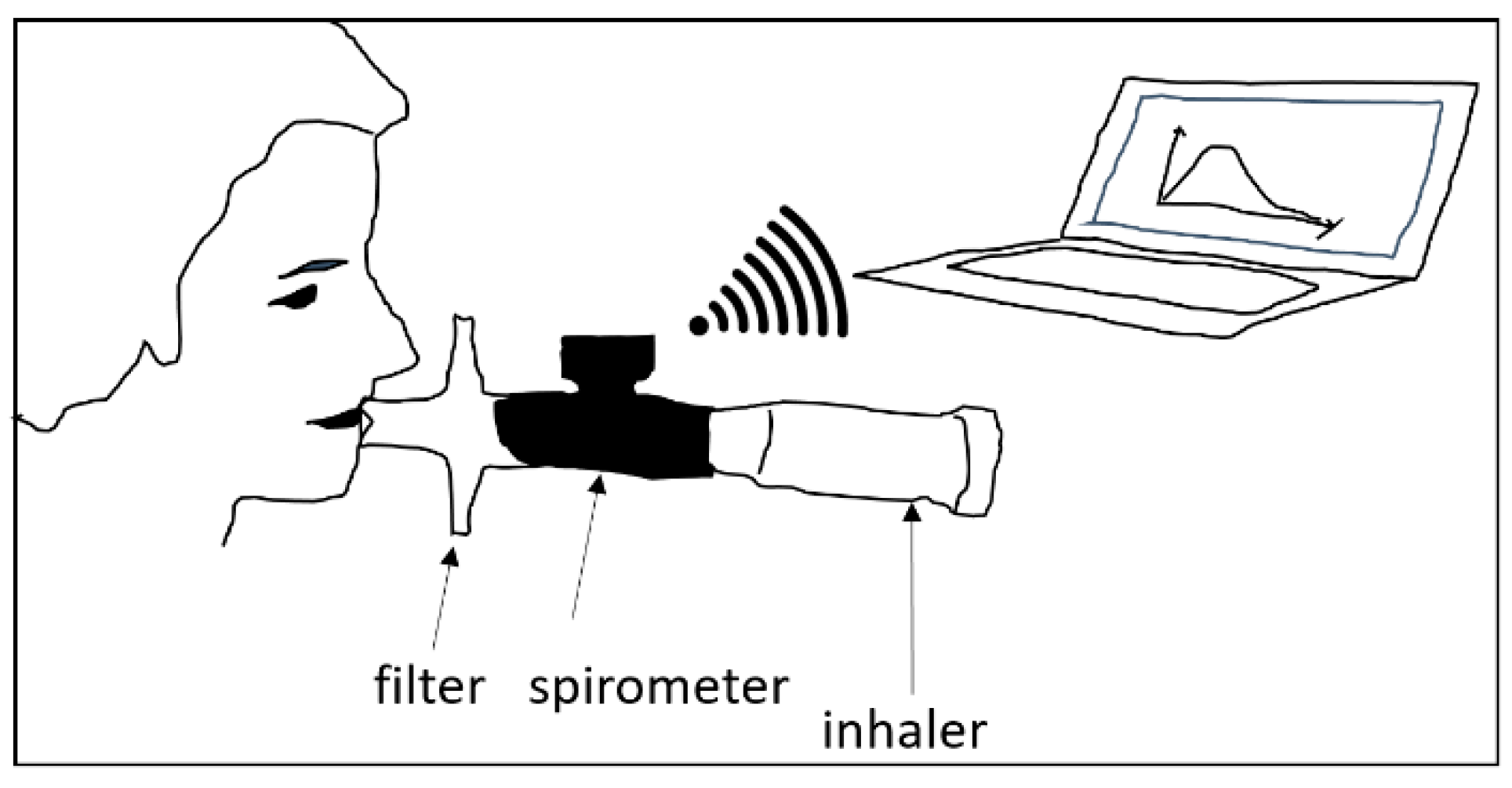
2.1. Measurement of Breathing Profiles
2.2. Determination of Aerosol Aerodynamic Size Distributions
2.3. Simulation of Airway Deposition
3. Results and Discussion
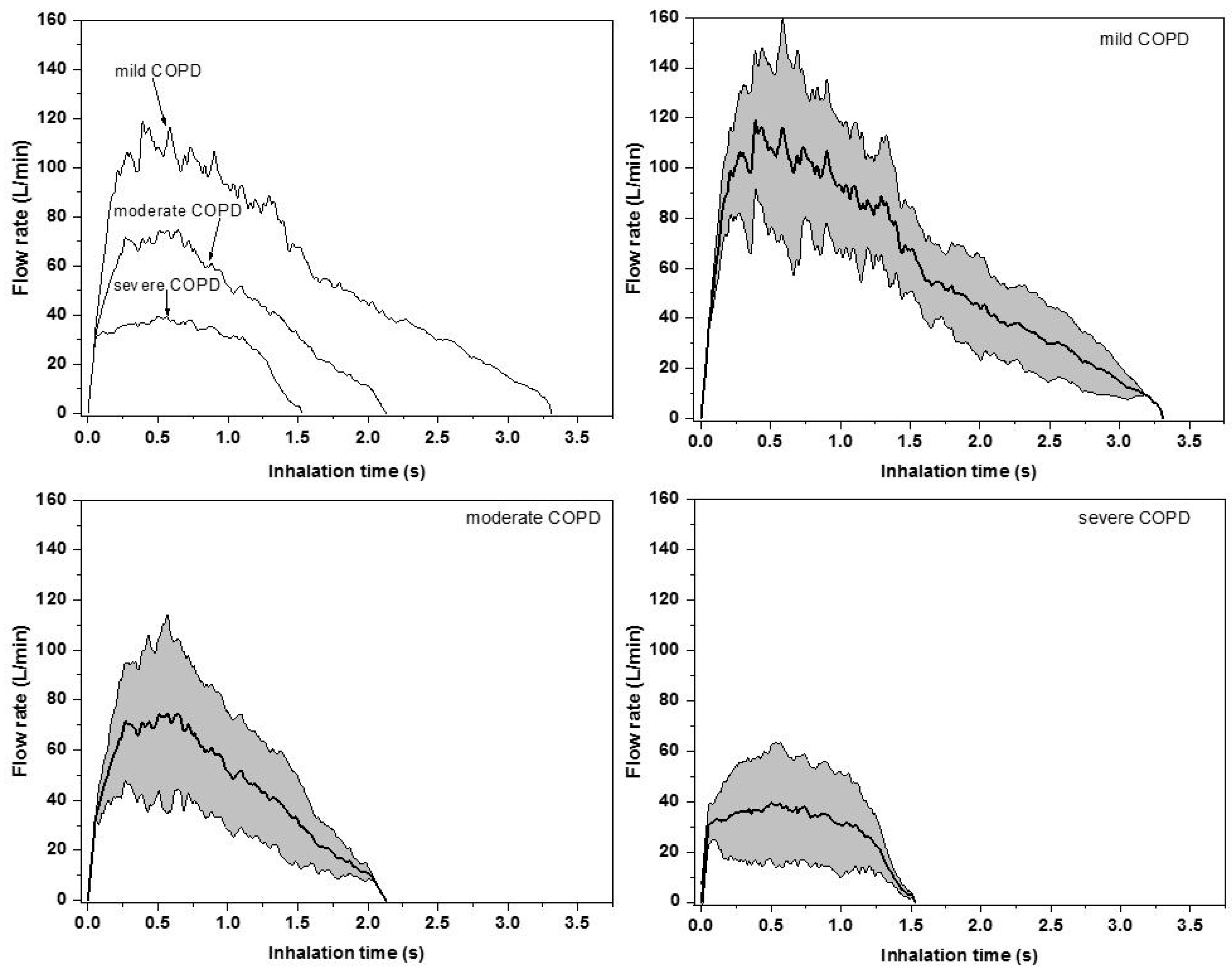
3.1. Measured Breathing Parameters and Profiles
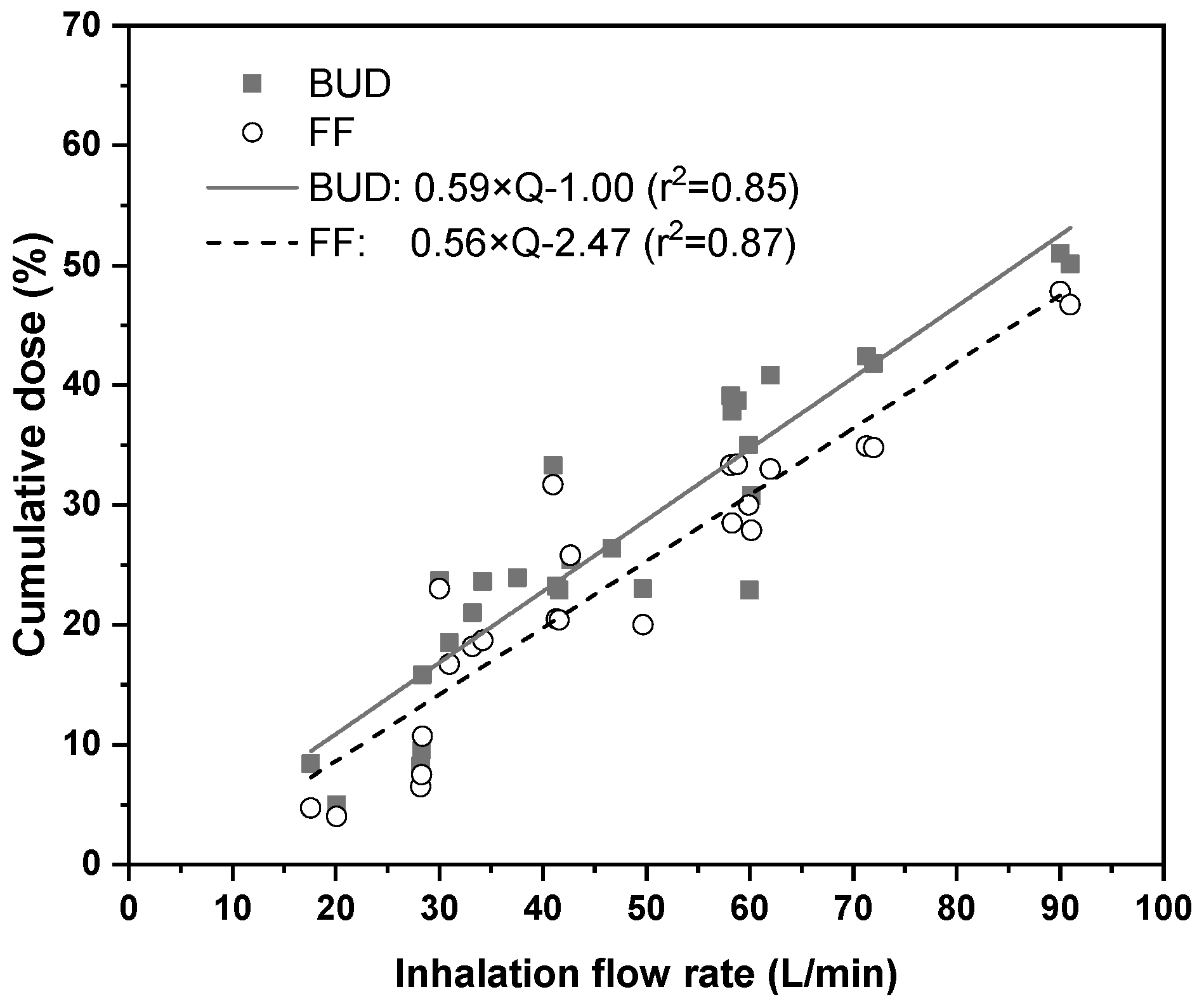
3.2. Aersosol Aerodynamic Size Distributions
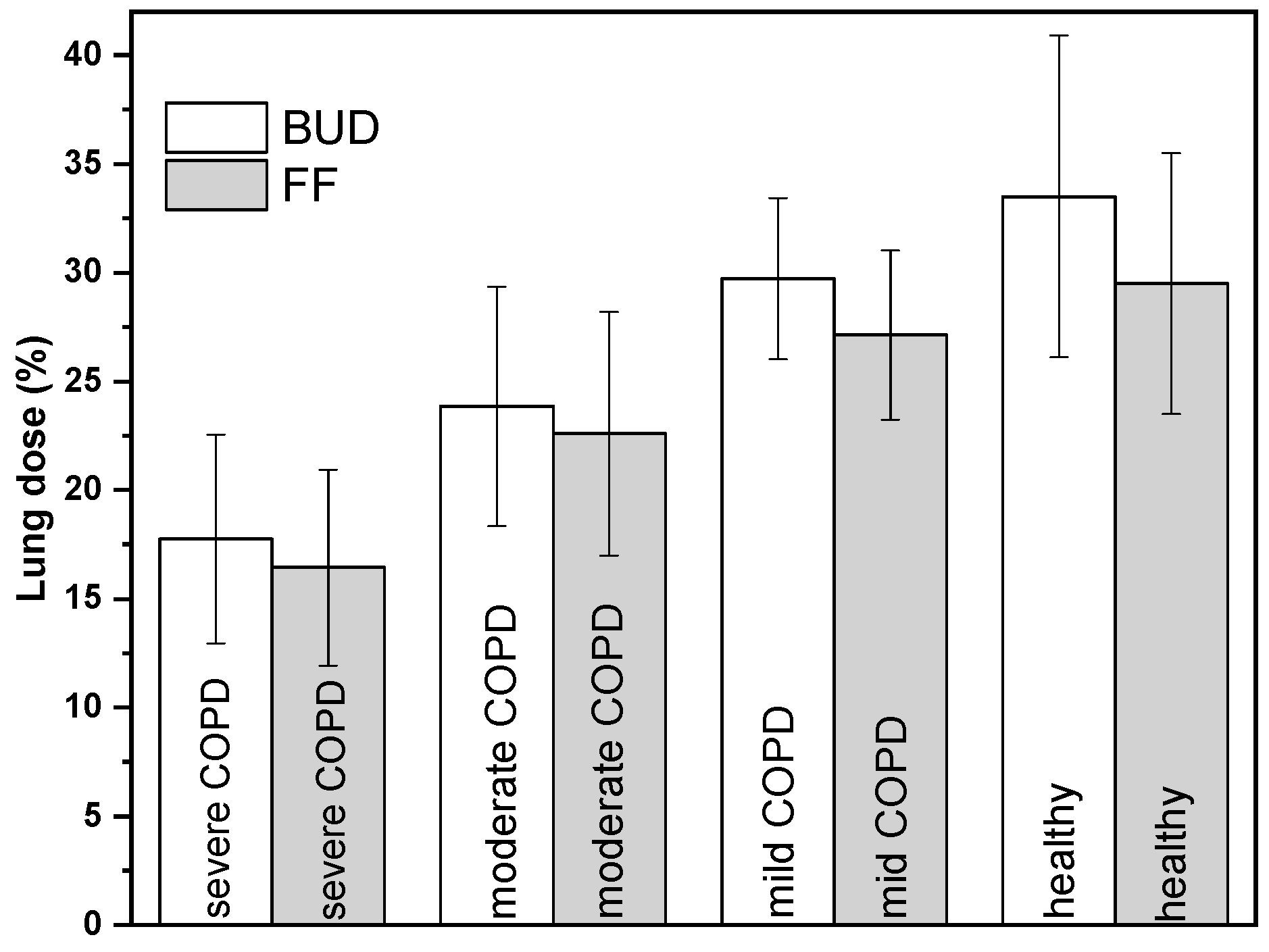
3.3. Drug Deposition Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anecchino, C.; Rossi, E.; Fanizza, C.; De Rosa, M.; Tognoni, G.; Romero, M. Prevalence of chronic obstructive pulmonary disease and pattern of comorbidities in a general population. Int. J. Chron. Obstruct. Pulmon. Dis. 2007, 2, 567–574. [Google Scholar] [PubMed]
- Farkas, Á.; Jókay, Á.; Füri, P.; Balásházy, I.; Müller, V.; Balázs, O.; Horváth, A. Computer modelling as a tool in characterization and optimization of aerosol drug delivery. Aerosol Air Qual. Res. 2015, 15, 2466–2474. [Google Scholar] [CrossRef]
- Newman, S.P.; Busse, W.W. Evolution of dry powder inhaler design, formulation and performance. Respir. Med. 2002, 96, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Ciciliani, A.-M.; Langguth, P.; Wachtel, H. In vitro dose comparison of Respimat® inhaler with dry powder inhalers for COPD maintenance therapy. Int. J. COPD 2017, 12, 1565–1577. [Google Scholar] [CrossRef] [PubMed]
- Buttini, F.; Brambilla, G.; Copelli, D.; Sisti, V.; Balducci, A.G.; Bettini, R.; Pasquali, I. Effect of flow rate on in vitro aerodynamic performance of NEXThaler® in comparison with Diskus® and Turbohaler® dry powder inhalers. J. Aerosol Med. Pulm. Drug Deliv. 2016, 29, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Azouz, W.; Chetcuti, P.; Hosker, H.; Saralaya, D.; Chrystyn, H. Inhalation characteristics of asthma patients, COPD patients and healthy volunteers with the Spiromax® and Turbuhaler® devices: A randomised, cross-over study. BMC Pulm. Med. 2015, 15, 47. [Google Scholar] [CrossRef] [PubMed]
- Wetterlin, K. Turbuhaler: A new powder inhaler for administration of drugs to the airways. Pharma Res. 1988, 5, 506–508. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Available online: https://goldcopd.org/wp-content/uploads/2017/11/GOLD-2018-v6.0-FINAL-revised-20-Nov_WMS.pdf (accessed on 22 January 2019).
- Farkas, Á.; Jókay, Á.; Balásházy, I.; Füri, P.; Müller, V.; Tomisa, G.; Horváth, A. Numerical simulation of particle characteristics and airway deposition distribution of Symbicort® Turbuhaler® dry powder fixed combination aerosol drug. Eur. J. Pharma Sci. 2016, 93, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Tarsin, W.; Assi, K.H.; Chrystyn, H. In-vitro intra- and inter-inhaler flow rate-dependent dosage emission from a combination of budesonide and eformoterol in a dry powder inhaler. J. Aerosol Med. 2004, 17, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Corradi, M.; Chrystyn, H.; Cosio, B.G.; Pirozynski, M.; Loukides, S.; Louis, R.; Spinola, M.; Usmani, O.S. NEXThaler, an innovative dry powder inhaler delivering an extrafine fixed combination of beclomethasone and formoterol to treat large and small airways in asthma. Expert Opin. Drug Deliv. 2014, 14, 1497–1506. [Google Scholar] [CrossRef] [PubMed]
- de Boer, A.H.; Gjaltema, D.; Hagedoorn, P.; Frijlink, H.W. Can ‘extrafine’ dry powder aerosols improve lung deposition? Eur. J. Pharm. Biopharm. 2015, 96, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Bagherisadeghi, G.; Larhrib, E.H.; Chrystyn, H. Real life dose emission characterization using COPD patient inhalation profiles when they inhaled using a fixed dose combination (FDC) of the medium strength Symbicort® Turbuhaler®. Int. J. Pharma 2017, 522, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Hoppentocht, M.; Hagedoorn, P.; Frijlink, H.W.; de Boer, A.H. Technological and practical challenges of dry powder inhalers and formulations. Adv. Drug Deliv. Rev. 2014, 75, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Johal, B.; Howald, M.; Fischer, M.; Marshall, J.; Venthoye, G. Fine particle profile of fluticasone propionate/formoterol fumarate versus other combination products: The DIFFUSE study. Comb. Prod. Ther. 2013, 3, 39–51. [Google Scholar] [CrossRef]
- Assi, K.H.; Tarsin, W.; Chrystyn, H. High performance liquid chromatography assay method for simultaneous quantitation of formoterol and budesonide in Symbicort Turbuhaler. J. Pharm. Biomed. Anal. 2006, 41, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Koblinger, L.; Hofmann, W. Monte Carlo modeling of aerosol deposition in human lungs. Part I: Simulation of particle transport in a stochastic lung structure. J. Aerosol Sci. 1990, 21, 661–674. [Google Scholar] [CrossRef]
- Cheng, Y.S. Aerosol deposition in the extrathoracic region. Aerosol Sci. Technol. 2003, 37, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Raabe, O.G.; Yeh, H.C.; Schum, G.M.; Phalen, R.F. Tracheobronchial Geometry: Human, Dog, Rat, Hamster. Available online: http://mae.engr.ucdavis.edu/wexler/lungs/LF53-Raabe/ (accessed on 22 January 2019).
- Haefeli-Bleuer, B.; Weibel, E.R. Morphometry of the human pulmonary acinus. Anat. Rec. 1988, 220, 401–414. [Google Scholar] [CrossRef] [PubMed]
| Breathing Parameter | Severe COPD | Moderate COPD | Mild COPD |
|---|---|---|---|
| PIF (L/min) | 39 ± 22.6 | 75 ± 40.2 | 119 ± 30.9 |
| IV (L) | 0.7 ± 0.2 | 1.6 ± 0.8 | 3.3 ± 0.6 |
| tin (s) | 1.5 ± 0.5 | 2.1 ± 1.1 | 3.3 ± 0.5 |
| tb-h (s) | 6.4 ± 4.2 | 7.2 ± 3.5 | 8.8 ± 2.9 |
| Q (L/min) | 28 ± 9.5 | 45.7 ± 18.1 | 60 ± 16.3 |
| Aerosol Parameter | Severe COPD | Moderate COPD | Mild COPD | |||
|---|---|---|---|---|---|---|
| BUD | FF | BUD | FF | BUD | FF | |
| ED (%) | 49.74 | 37.13 | 60 | 51.29 | 68.30 | 62.73 |
| f1 (%) | 1.87 | 1.83 | 3.64 | 3.25 | 5.07 | 4.39 |
| f3 (%) | 8.33 | 6.09 | 18.95 | 16.53 | 27.53 | 24.97 |
| f5 (%) | 16.46 | 15.77 | 27.56 | 25.56 | 36.53 | 33.47 |
| f7 (%) | 21.73 | 22.55 | 34.11 | 32.93 | 42.44 | 38.82 |
| f10 (%) | 23.05 | 25.23 | 36.09 | 35.68 | 44.85 | 40.49 |
| flarge (%) | 26.69 | 11.9 | 23.91 | 15.61 | 23.45 | 22.24 |
| MMAD (μm) | 3.03 | 3.44 | 2.67 | 2.87 | 2.45 | 2.43 |
| GSD (–) | 2.02 | 2.23 | 1.87 | 1.91 | 1.81 | 1.80 |
| Dose Fractions | Severe COPD | Moderate COPD | Mild COPD | |||
|---|---|---|---|---|---|---|
| BUD | FF | BUD | FF | BUD | FF | |
| DEV (%) | 50.3 ± 8.1 | 62.9 ± 7.3 | 40.0 ± 4.3 | 48.7 ± 4.4 | 31.7 ± 5.1 | 37.3 ± 4.8 |
| ET (%) | 27.3 ± 4.6 | 16.3 ± 4.5 | 31.7 ± 3.8 | 24.7 ± 3.7 | 34.8 ± 4.8 | 32.2 ± 5.0 |
| LUNG (%) | 17.7 ± 4.8 | 16.4 ± 4.5 | 23.8 ± 5.5 | 22.6 ± 5.6 | 29.7 ± 3.7 | 27.1 ± 3.9 |
| EXH (%) | 4.6 ± 1.1 | 4.4 ± 1.8 | 4.5 ± 1.6 | 4.1 ± 1.9 | 3.8 ± 1.6 | 3.4 ± 1.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farkas, Á.; Lizal, F.; Jedelsky, J.; Elcner, J.; Horváth, A.; Jicha, M. Simulation of Airway Deposition of an Aerosol Drug in COPD Patients. Pharmaceutics 2019, 11, 153. https://doi.org/10.3390/pharmaceutics11040153
Farkas Á, Lizal F, Jedelsky J, Elcner J, Horváth A, Jicha M. Simulation of Airway Deposition of an Aerosol Drug in COPD Patients. Pharmaceutics. 2019; 11(4):153. https://doi.org/10.3390/pharmaceutics11040153
Chicago/Turabian StyleFarkas, Árpád, Frantisek Lizal, Jan Jedelsky, Jakub Elcner, Alpár Horváth, and Miroslav Jicha. 2019. "Simulation of Airway Deposition of an Aerosol Drug in COPD Patients" Pharmaceutics 11, no. 4: 153. https://doi.org/10.3390/pharmaceutics11040153
APA StyleFarkas, Á., Lizal, F., Jedelsky, J., Elcner, J., Horváth, A., & Jicha, M. (2019). Simulation of Airway Deposition of an Aerosol Drug in COPD Patients. Pharmaceutics, 11(4), 153. https://doi.org/10.3390/pharmaceutics11040153






