Investigation of the Quantity of Exhaled Aerosols Released into the Environment during Nebulisation
Abstract
:1. Introduction
2. Materials and Methods
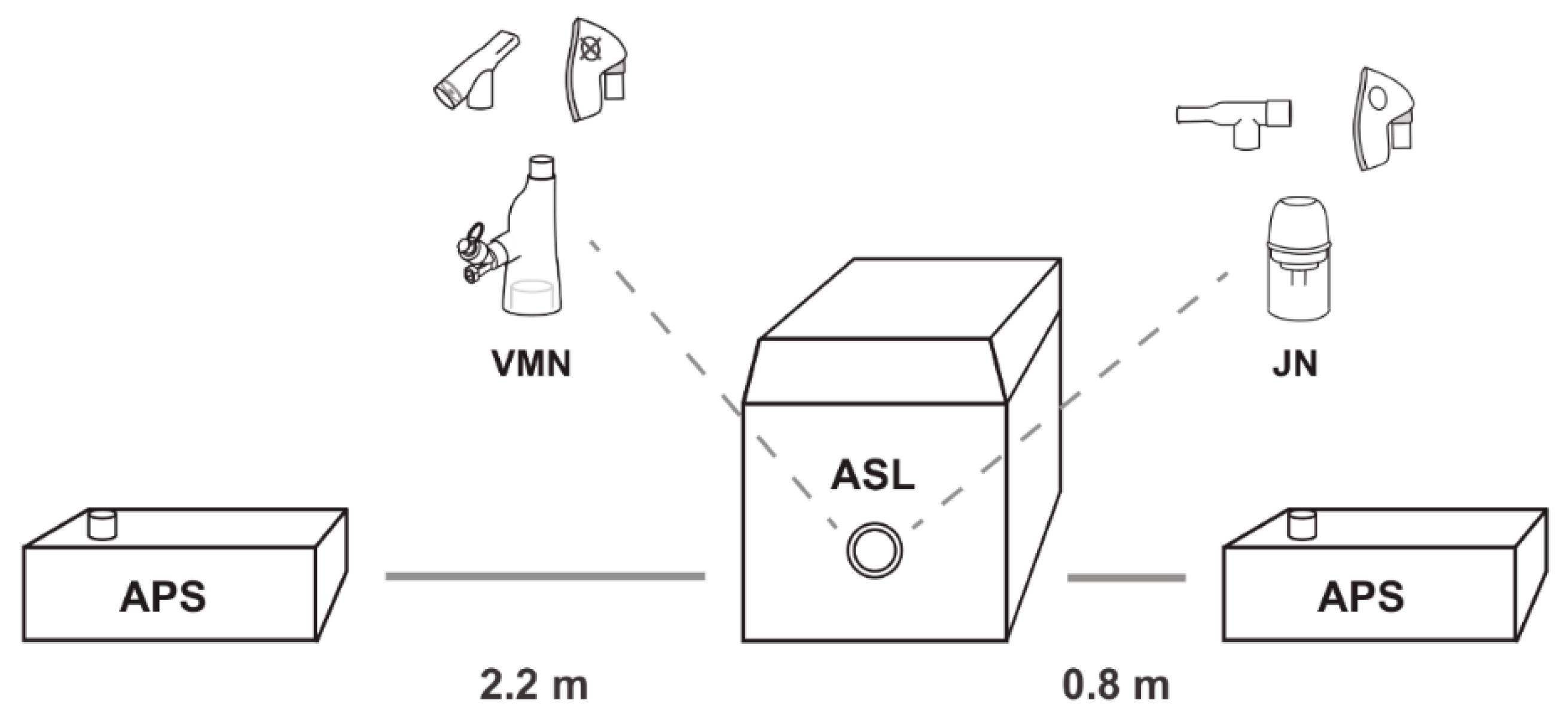
2.1. Nebulisers (VMN & JN) & Non-Invasive Ventilation Interfaces
2.2. Simulated Patient Breathing
2.3. Characterisation of Aerosol Dose Distribution
2.4. Characterisation of Fugitive Emissions
3. Results
3.1. Aerosol Distribution
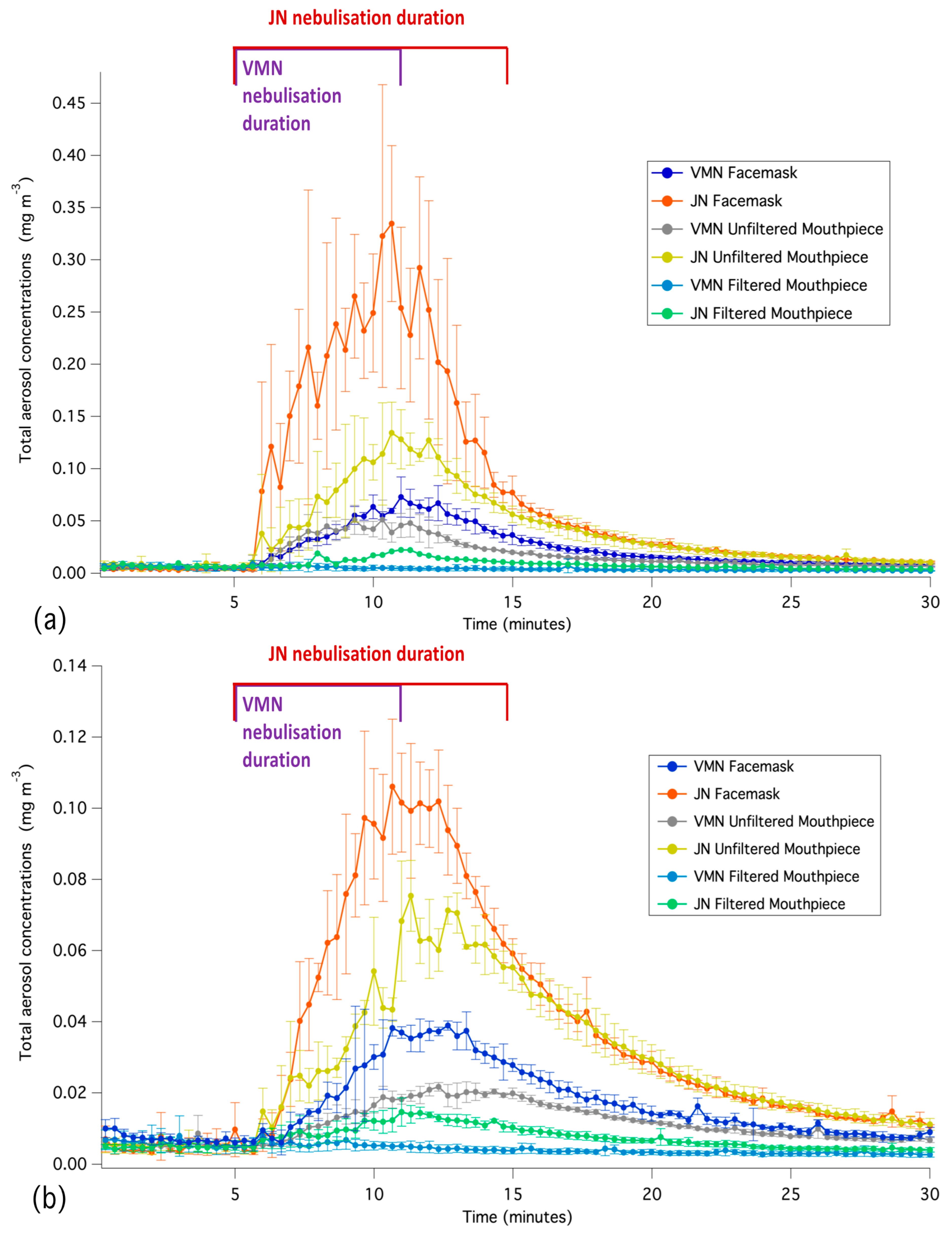
3.2. Time-Averaged Aerosol Concentrations
3.3. Aerosol Droplet Sizing
3.4. Inhalation Exposure
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- ICRP. Human Respiratory Tract Model for Radiological Protection. Ann. ICRP 1994, 24, 1–3. [Google Scholar] [CrossRef]
- Nazaroff, W.W. Indoor particle dynamics. Indoor Air 2004, 14, 175–183. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.A.; Byrne, M.A.; Ashmore, M.R.; Terry, A.C.; Dimitroulopoulou, C. Development of a probabilistic multi-zone multi-source computational model and demonstration of its applications in predicting PM concentrations indoors. Sci. Total Environ. 2014, 490, 798–806. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Morawska, L.; Gilbert, D. Particle deposition rates in residential houses. Atmos. Environ. 2005, 39, 3891–3899. [Google Scholar] [CrossRef]
- Saeed, H.; Mohsen, M.; Fink, J.B.; Dailey, P.; Salah Eldin, A.; Abdelrahman, M.M.; Elberry, A.A.; Rabea, H.; Hussein, R.R.S.; Abdelrahim, M.E.A. Fill volume, humidification and heat effects on aerosol delivery and fugitive emissions during noninvasive ventilation. J. Drug Delivery Sci. Technol. 2017, 39, 372–378. [Google Scholar] [CrossRef]
- Chughtai, A.A.; Seale, H.; MacIntyre, C.R. Availability, consistency and evidence-base of policies and guidelines on the use of mask and respirator to protect hospital health care workers: A global analysis. BMC Res. Notes 2013, 6, 216. [Google Scholar] [CrossRef] [PubMed]
- Croteau, G.A.; Martin, D.B.; Camp, J.; Yost, M.; Conrad, C.; Zeitlin, P.L.; Heald, A.E. Evaluation of Exposure and Health Care Worker Response to Nebulized Administration of tgAAVCF to Patients with Cystic Fibrosis. Ann. Occup. Hyg. 2004, 48, 673–681. [Google Scholar]
- Wittgen, B.P.H.; Kunst, P.W.A.; van der Born, K.; van Wijk, A.W.; Perkins, W.; Pilkiewicz, F.G.; Perez-Soler, R.; Nicholson, S.; Peters, G.J.; Postmus, P.E. Phase I Study of Aerosolized SLIT Cisplatin in the Treatment of Patients with Carcinoma of the Lung. Clin. Cancer Res. 2007, 13, 2414–2421. [Google Scholar] [CrossRef]
- Shults, R.A.; Baron, S.; Decker, J.; Deitchman, S.D.; Connor, J.D. Health care worker exposure to aerosolized ribavirin: Biological and air monitoring. J. Occup. Environ. Med. 1996, 38, 257–263. [Google Scholar] [CrossRef]
- O’Riordan, T.G.; Smaldone, G.C. Exposure of health care workers to aerosolized pentamidine. Chest 1992, 101, 1494–1499. [Google Scholar] [CrossRef]
- Carnathan, B.; Martin, B.; Colice, G. Second hand (S)-albuterol: RT exposure risk following racemic albuterol. Respir Care 2001, 46, 1084. [Google Scholar]
- Simonds, A.K.; Hanak, A.; Chatwin, M.; Morrell, M.; Hall, A.; Parker, K.H.; Siggers, J.H.; Dickinson, R.J. Evaluation of droplet dispersion during non-invasive ventilation, oxygen therapy, nebuliser treatment and chest physiotherapy in clinical practice: Implications for management of pandemic influenza and other airborne infections. Health Technol. Assess. 2010, 14, 131–172. [Google Scholar] [CrossRef] [PubMed]
- WHO. Implementation of the Global Action on Antimicrobial Resistance. Available online: http://www.who.int/antimicrobial-resistance/news/WHO-GAP-AMR-Newsletter-No-32-Nov-2017.pdf?ua=1 (accessed on 20 September 2018).
- CDC. Infection Control Precautions for Aerosol-Generating Procedures on Patients Who have Suspected Severe Acute Respiratory Syndrome (SARS). Available online: https://stacks.cdc.gov/view/cdc/25034 (accessed on 18 August 2018).
- Tran, K.; Cimon, K.; Severn, M.; Pessoa-Silva, C.L.; Conly, J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: A systematic review. PLoS ONE 2012, 7, e35797. [Google Scholar] [CrossRef] [PubMed]
- Ari, A.; Fink, J.B.; Pilbeam, S.P. Secondhand aerosol exposure during mechanical ventilation with and without expiratory filters: An in-vitro study. Ind. J. Resp. Care 2016, 5, 677–682. [Google Scholar]
- Somogyi, R.; Vesely, A.E.; Azami, T.; Preiss, D.; Fisher, J.; Correia, J.; Fowler, R.A. Dispersal of respiratory droplets with open vs closed oxygen delivery masks: Implications for the transmission of severe acute respiratory syndrome. Chest 2004, 125, 1155–1157. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Chow, B.K.; Chu, L.C.Y.; Ng, S.S.; Hall, S.D.; Gin, T.; Chan, M.T.V. Exhaled Air and Aerosolized Droplet Dispersion During Application of a Jet Nebulizer. Chest 2009, 135, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Chow, B.K.; Lo, T.; Ng, S.S.; Ko, F.W.; Gin, T.; Chan, M.T.V. Exhaled Air Dispersion During Noninvasive Ventilation via Helmets and a Total Facemask. Chest 2015, 147, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Chow, B.K.; Ng, S.S.; Chu, L.C.Y.; Hall, S.D.; Gin, T.; Chan, M.T.V. Exhaled Air Dispersion Distances During Noninvasive Ventilation via Different Respironics Face Masks. Chest 2009, 136, 998–1005. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Hall, S.D.; Chan, M.T.V.; Chow, B.K.; Tsou, J.Y.; Joynt, G.M.; Sullivan, C.E.; Sung, J.J.Y. Noninvasive Positive-Pressure Ventilation: An Experimental Model to Assess Air and Particle Dispersion. Chest 2006, 130, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Ip, M.; Tang, J.W.; Hui, D.S.; Wong, A.L.; Chan, M.T.V.; Joynt, G.M.; So, A.T.P.; Hall, S.D.; Chan, P.K.S.; Sung, J.J.Y. Airflow and droplet spreading around oxygen masks: A simulation model for infection control research. Am. J. Infection Control 2007, 35, 684–689. [Google Scholar] [CrossRef] [PubMed]
- ISO. Anaesthetic and Respiratory Equipment—Nebulizing Systems and Components; International Organization for Standardization: Geneva, Switzerland, 2013; Available online: https://www.iso.org/standard/59482.html (accessed on 5 April 2018).
- U.S. EPA. Exposure Factors Handbook, 2011th ed.; National Center for Environmental Assessment: Washington, DC, USA, 2011.
- Dolovich, M.B.; Dhand, R. Aerosol drug delivery: Developments in device design and clinical use. Lancet 2011, 377, 1032–1045. [Google Scholar] [CrossRef]
- Smaldone, G.C. Advances in aerosols: Adult respiratory disease. J. Aerosol Med. 2006, 19, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Bennett, G.; Joyce, M.; Sweeney, L.; MacLoughlin, R. In Vitro Determination of the Main Effects in the Design of High-Flow Nasal Therapy Systems with Respect to Aerosol Performance. Pulmonary Ther. 2018, 4, 73–86. [Google Scholar] [CrossRef]
- Ciuzas, D.; Prasauskas, T.; Krugly, E.; Sidaraviciute, R.; Jurelionis, A.; Seduikyte, L.; Kauneliene, V.; Wierzbicka, A.; Martuzevicius, D. Characterization of indoor aerosol temporal variations for the real-time management of indoor air quality. Atmos. Environ. 2015, 118, 107–117. [Google Scholar] [CrossRef]
- Long, C.M.; Suh, H.H.; Catalano, P.J.; Koutrakis, P. Using time-and size-resolved particulate data to quantify indoor penetration and deposition behavior. Environ. Sci. Technol. 2001, 35, 2089–2099. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.A.; Byrne, M.A.; Ashmore, M.R.; Terry, A.C.; Dimitroulopoulou, C. A simulation study of the changes in PM2.5 concentrations due to interzonal airflow variations caused by internal door opening patterns. Atmos. Environ. 2014, 87, 183–188. [Google Scholar] [CrossRef]
- Peters, S.G. Continuous bronchodilator therapy. Chest 2007, 131, 286–289. [Google Scholar] [CrossRef]
- Berlinski, A.; Waldrep, J.C. Four hours of continuous albuterol nebulization. Chest 1998, 114, 847–853. [Google Scholar] [CrossRef]
- Kelly, H.; Keim, K.; McWilliams, B. Comparison of two methods of delivering continuously nebulized albuterol. Ann. Pharmacother. 2003, 37, 23–26. [Google Scholar] [CrossRef]
- Hynicka, L.M.; Ensor, C.R. Prophylaxis and Treatment of Respiratory Syncytial Virus in Adult Immunocompromised Patients. Ann. Pharmacother. 2012, 46, 558–566. [Google Scholar] [CrossRef]
- Goodson, W.H., III; Lowe, L.; Carpenter, D.O.; Gilbertson, M.; Manaf, A.A.; de Cerain Salsamendi, A.L.; Lasfar, A.; Carnero, A.; Azqueta, A.; Amedei, A.; et al. Assessing the carcinogenic potential of low-dose exposures to chemical mixtures in the environment: The challenge ahead. Carcinogenesis 2015, 36 (Suppl. 1), S254–S296. [Google Scholar] [CrossRef] [PubMed]
- Hinds, W.C. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles; John Wiley & Sons: New York, NY, USA, 1999. [Google Scholar]
- Wittgen, B.P.; Kunst, P.W.; Perkins, W.R.; Lee, J.K.; Postmus, P.E. Assessing a system to capture stray aerosol during inhalation of nebulized liposomal cisplatin. J. Aerosol Med. 2006, 19, 385–391. [Google Scholar] [CrossRef] [PubMed]
| Nebuliser/Interface | Residual Mass | Inhaled Dose | Exhaled Dose |
|---|---|---|---|
| VMN/valved facemask | 6.87 ± 1.43%, | 46.69 ± 3.06% | N/A |
| JN/open facemask | 39.03 ± 2.85% | 21.95 ± 0.66% | N/A |
| VMN/unfiltered mouthpiece | 6.49 ± 1.11% | 49.46 ± 3.17% | N/A |
| JN/unfiltered mouthpiece | 40.10 ± 5.49% | 29.22 ± 1.71% | N/A |
| VMN/filtered mouthpiece | 5.26 ± 2.04% | 50.32 ± 1.90% | 14.53 ± 7.85% |
| JN/filtered mouthpiece | 38.43 ± 1.57% | 35.94 ± 1.81% | 29.45 ± 1.41% |
| Nebuliser Type | Facemask (mg m−3) | Unfiltered Mouthpiece (mg m−3) | Filtered Mouthpiece (mg m−3) |
|---|---|---|---|
| Jet Nebuliser | 0.072 ± 0.001 | 0.039 ± 0.004 | 0.009 ± 0.001 |
| Vibrating Mesh Nebuliser | 0.022 ± 0.001 | 0.017 ± 0.002 | 0.004 ± 0.001 |
| Nebuliser Type | Facemask (µm) | Unfiltered Mouthpiece (µm) | Filtered Mouthpiece (µm) |
|---|---|---|---|
| Jet Nebuliser | 1.338 ± 0.098 | 1.267 ± 0.07 | 1.155 ± 0.081 |
| Vibrating Mesh Nebuliser | 1.160 ± 0.205 | 0.890 ± 0.044 | 1.432 ± 0.241 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGrath, J.A.; O’Sullivan, A.; Bennett, G.; O’Toole, C.; Joyce, M.; Byrne, M.A.; MacLoughlin, R. Investigation of the Quantity of Exhaled Aerosols Released into the Environment during Nebulisation. Pharmaceutics 2019, 11, 75. https://doi.org/10.3390/pharmaceutics11020075
McGrath JA, O’Sullivan A, Bennett G, O’Toole C, Joyce M, Byrne MA, MacLoughlin R. Investigation of the Quantity of Exhaled Aerosols Released into the Environment during Nebulisation. Pharmaceutics. 2019; 11(2):75. https://doi.org/10.3390/pharmaceutics11020075
Chicago/Turabian StyleMcGrath, James A., Andrew O’Sullivan, Gavin Bennett, Ciarraí O’Toole, Mary Joyce, Miriam A. Byrne, and Ronan MacLoughlin. 2019. "Investigation of the Quantity of Exhaled Aerosols Released into the Environment during Nebulisation" Pharmaceutics 11, no. 2: 75. https://doi.org/10.3390/pharmaceutics11020075
APA StyleMcGrath, J. A., O’Sullivan, A., Bennett, G., O’Toole, C., Joyce, M., Byrne, M. A., & MacLoughlin, R. (2019). Investigation of the Quantity of Exhaled Aerosols Released into the Environment during Nebulisation. Pharmaceutics, 11(2), 75. https://doi.org/10.3390/pharmaceutics11020075






