Evaluation of a Methylcellulose and Hyaluronic Acid Hydrogel as a Vehicle for Rectal Delivery of Biologics
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Hydrogel
2.3. Rheological Characterization
2.4. Drug Release from Hydrogels
2.5. Cytotoxicity Assays
2.6. Permeability Assay
2.7. Statistical Analysis
3. Results
3.1. Hydrogel Preparation
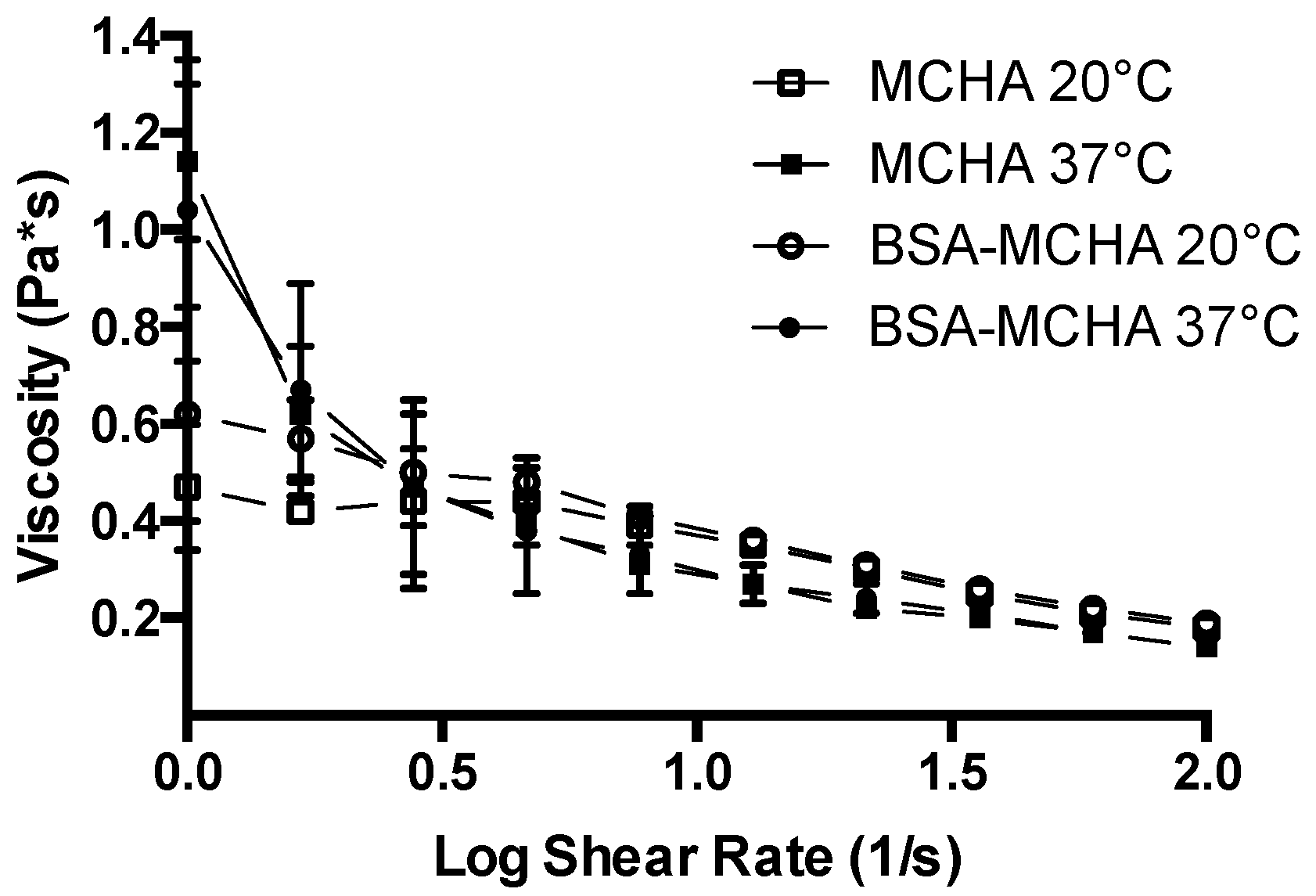
3.2. Rheological Characterization
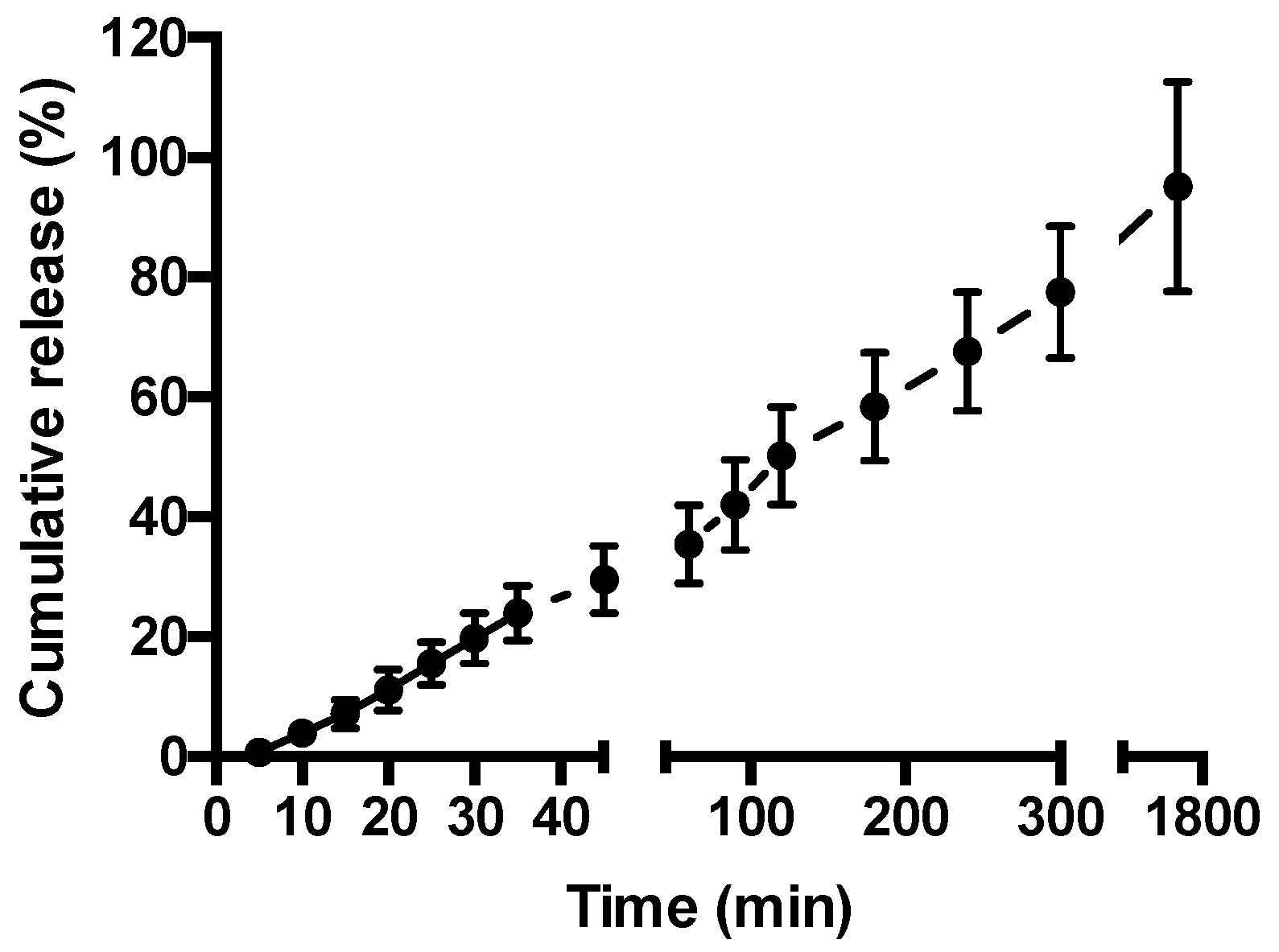
3.3. Drug Release from Hydrogels
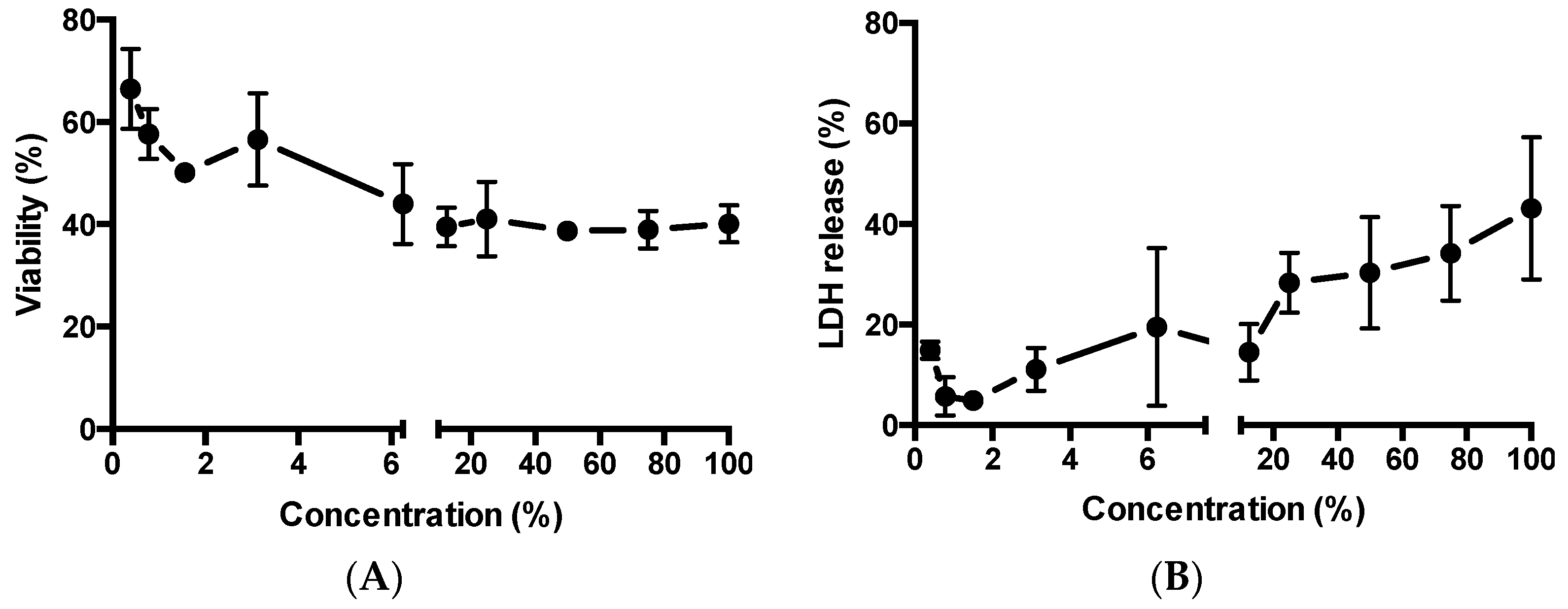
3.4. Cytotoxicity Assay
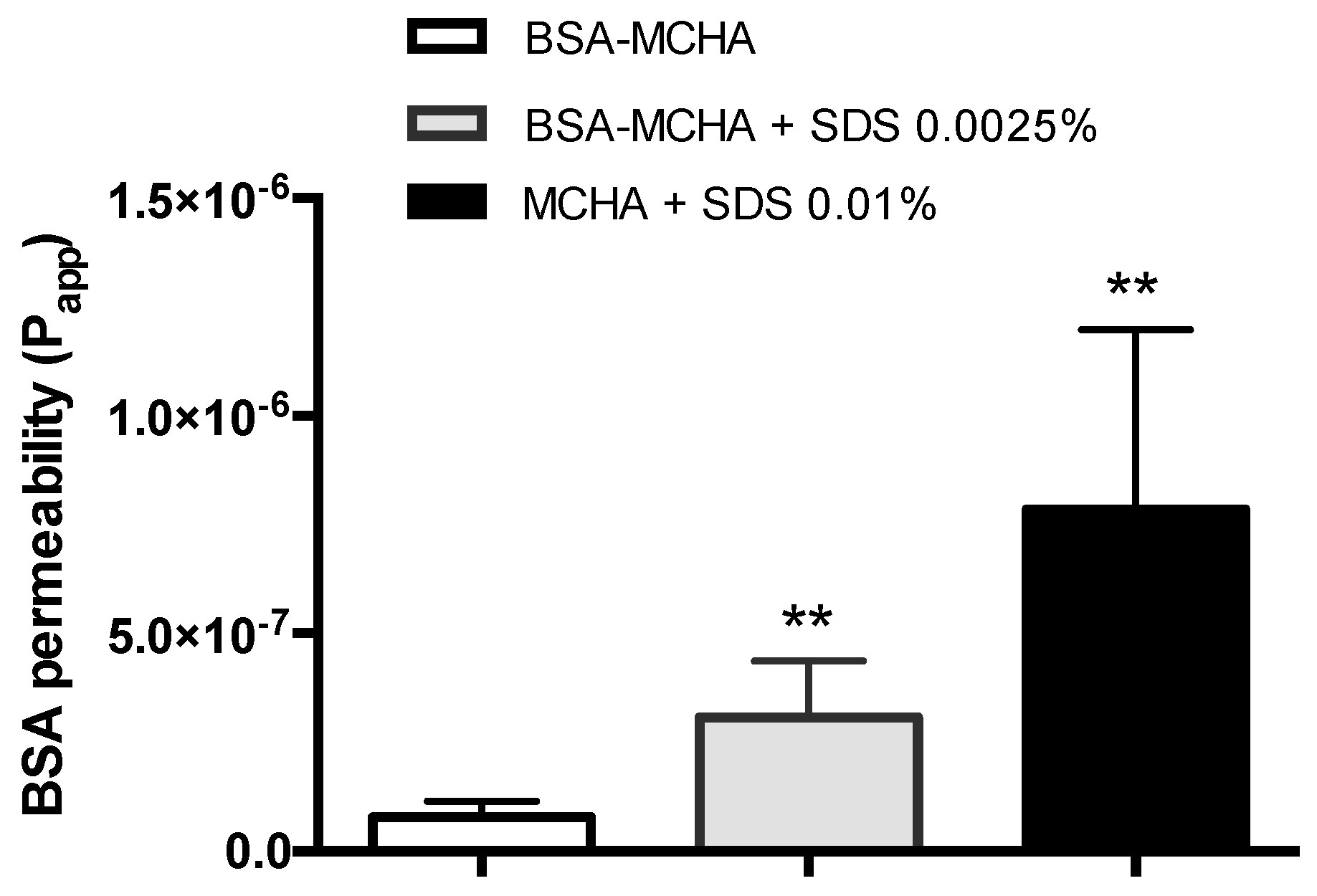
3.5. Permeability Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- D’Haens, G. Risks and benefits of biologic therapy for inflammatory bowel diseases. Gut 2007, 56, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Ben-Horin, S.; Mao, R.; Chen, M. Optimizing biologic treatment in ibd: Objective measures, but when, how and how often? BMC Gastroenterol. 2015, 15, 178. [Google Scholar] [CrossRef] [PubMed]
- Prantera, C.; Rizzi, M. 5-asa in ulcerative colitis: Improving treatment compliance. World J. Gastroenterol. 2009, 15, 4353–4355. [Google Scholar] [CrossRef] [PubMed]
- Kornbluth, A.; Sachar, D.B.; Practice Parameters Committee of the American College of Gastroenterology. Ulcerative colitis practice guidelines in adults: American college of gastroenterology, practice parameters committee. Am. J. Gastroenterol. 2010, 105, 501–523. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tam, M.; Samaei, S.; Lerouge, S.; Barralet, J.; Stevenson, M.M.; Cerruti, M. Mucoadhesive chitosan hydrogels as rectal drug delivery vessels to treat ulcerative colitis. Acta Biomater. 2017, 48, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.R.; Nguyen, L.P.; Inayathullah, M.; Malkovskiy, A.; Habte, F.; Rajadas, J.; Habtezion, A. A thermo-sensitive delivery platform for topical administration of inflammatory bowel disease therapies. Gastroenterology 2015, 149, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Vllasaliu, D.; Thanou, M.; Stolnik, S.; Fowler, R. Recent advances in oral delivery of biologics: Nanomedicine and physical modes of delivery. Expert Opin. Drug Deliv. 2018, 15, 756–770. [Google Scholar] [CrossRef] [PubMed]
- Cole, H.; Bryan, D.; Lancaster, L.; Mawas, F.; Vllasaliu, D. Chitosan nanoparticle antigen uptake in epithelial monolayers can predict mucosal but not systemic in vivo immune response by oral delivery. Carbohydr. Polym. 2018, 190, 248–254. [Google Scholar] [CrossRef]
- Perinelli, D.R.; Lucarini, S.; Fagioli, L.; Campana, R.; Vllasaliu, D.; Duranti, A.; Casettari, L. Lactose oleate as new biocompatible surfactant for pharmaceutical applications. Eur. J. Pharm. Biopharm. 2018, 124, 55–62. [Google Scholar] [CrossRef]
- Lucarini, S.; Fagioli, L.; Campana, R.; Cole, H.; Duranti, A.; Baffone, W.; Vllasaliu, D.; Casettari, L. Unsaturated fatty acids lactose esters: Cytotoxicity, permeability enhancement and antimicrobial activity. Eur. J. Pharm. Biopharm. 2016, 107, 88–96. [Google Scholar] [CrossRef]
- Maher, S.; Mrsny, R.J.; Brayden, D.J. Intestinal permeation enhancers for oral peptide delivery. Adv. Drug Deliv. Rev. 2016, 106, 277–319. [Google Scholar] [CrossRef]
- Hashem, L.; Swedrowska, M.; Vllasaliu, D. Intestinal uptake and transport of albumin nanoparticles: Potential for oral delivery. Nanomedicine 2018, 13, 1255–1265. [Google Scholar] [CrossRef]
- Fowler, R.; Vllasaliu, D.; Falcone, F.H.; Garnett, M.; Smith, B.; Horsley, H.; Alexander, C.; Stolnik, S. Uptake and transport of b12-conjugated nanoparticles in airway epithelium. J. Control. Release 2013, 172, 374–381. [Google Scholar] [CrossRef]
- Hollander, D. The intestinal permeability barrier. A hypothesis as to its regulation and involvement in crohn’s disease. Scand. J. Gastroenterol. 1992, 27, 721–726. [Google Scholar] [CrossRef]
- Soderholm, J.D.; Peterson, K.H.; Olaison, G.; Franzen, L.E.; Westrom, B.; Magnusson, K.E.; Sjodahl, R. Epithelial permeability to proteins in the noninflamed ileum of crohn’s disease? Gastroenterology 1999, 117, 65–72. [Google Scholar] [CrossRef]
- Zeissig, S.; Burgel, N.; Gunzel, D.; Richter, J.; Mankertz, J.; Wahnschaffe, U.; Kroesen, A.J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active crohn’s disease. Gut 2007, 56, 61–72. [Google Scholar] [CrossRef]
- Zhai, J.; Mantaj, J.; Vllasaliu, D. Ascorbyl palmitate hydrogel for local, intestinal delivery of macromolecules. Pharmaceutics 2018, 10, 188. [Google Scholar] [CrossRef]
- Zhang, S.; Ermann, J.; Succi, M.D.; Zhou, A.; Hamilton, M.J.; Cao, B.; Korzenik, J.R.; Glickman, J.N.; Vemula, P.K.; Glimcher, L.H.; et al. An inflammation-targeting hydrogel for local drug delivery in inflammatory bowel disease. Sci. Transl. Med. 2015, 7, 300ra128. [Google Scholar] [CrossRef]
- Sohail, M.; Mudassir; Minhas, M.U.; Khan, S.; Hussain, Z.; de Matas, M.; Shah, S.A.; Khan, S.; Kousar, M.; Ullah, K. Natural and synthetic polymer-based smart biomaterials for management of ulcerative colitis: A review of recent developments and future prospects. Drug Deliv. Transl. Res. 2018, 1–20. [Google Scholar] [CrossRef]
- Tirosh, B.; Khatib, N.; Barenholz, Y.; Nissan, A.; Rubinstein, A. Transferrin as a luminal target for negatively charged liposomes in the inflamed colonic mucosa. Mol. Pharm. 2009, 6, 1083–1091. [Google Scholar] [CrossRef]
- Mayol, L.; De Stefano, D.; De Falco, F.; Carnuccio, R.; Maiuri, M.C.; De Rosa, G. Effect of hyaluronic acid on the thermogelation and biocompatibility of its blends with methyl cellulose. Carbohydr. Polym. 2014, 112, 480–485. [Google Scholar] [CrossRef]
- Mike, N.; Udeshi, U.; Asquith, P.; Ferrando, J. Small bowel enema in non-responsive coeliac disease. Gut 1990, 31, 883–885. [Google Scholar] [CrossRef]
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J. Tissue Eng. 2017, 8. [Google Scholar] [CrossRef]
- Sandri, G.; Rossi, S.; Ferrari, F.; Bonferoni, M.C.; Zerrouk, N.; Caramella, C. Mucoadhesive and penetration enhancement properties of three grades of hyaluronic acid using porcine buccal and vaginal tissue, caco-2 cell lines, and rat jejunum. J. Pharm. Pharmacol. 2004, 56, 1083–1090. [Google Scholar] [CrossRef]
- Purohit, T.J.; Hanning, S.M.; Wu, Z. Advances in rectal drug delivery systems. Pharm. Dev. Technol. 2018, 23, 942–952. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Raj, J.P. Role of biologics and biosimilars in inflammatory bowel disease: Current trends and future perspectives. J. Inflamm. Res. 2018, 11, 215–226. [Google Scholar] [CrossRef]
- Ramadass, S.K.; Perumal, S.; Jabaris, S.L.; Madhan, B. Preparation and evaluation of mesalamine collagen in situ rectal gel: A novel therapeutic approach for treating ulcerative colitis. Eur. J. Pharm. Sci. 2013, 48, 104–110. [Google Scholar] [CrossRef]
- Lopetuso, L.R.; Petito, V.; Cufino, V.; Arena, V.; Stigliano, E.; Gerardi, V.; Gaetani, E.; Poscia, A.; Amato, A.; Cammarota, G.; et al. Locally injected infliximab ameliorates murine dss colitis: Differences in serum and intestinal levels of drug between healthy and colitic mice. Dig. Liver Dis. 2013, 45, 1017–1021. [Google Scholar] [CrossRef]
- Li, L.; Shan, H.; Yue, C.Y.; Lam, Y.C.; Tam, K.C.; Hu, X. Thermally induced association and dissociation of methylcellulose in aqueous solutions. Langmuir 2002, 18, 7291–7298. [Google Scholar] [CrossRef]
- Litwiniuk, M.; Krejner, A.; Speyrer, M.S.; Gauto, A.R.; Grzela, T. Hyaluronic acid in inflammation and tissue regeneration. Wounds 2016, 28, 78–88. [Google Scholar]
- Chiu, C.T.; Kuo, S.N.; Hung, S.W.; Yang, C.Y. Combined treatment with hyaluronic acid and mesalamine protects rats from inflammatory bowel disease induced by intracolonic administration of trinitrobenzenesulfonic acid. Molecules 2017, 22, 904. [Google Scholar] [CrossRef]
- Mahboubian, A. Development of Thermoresponsive Polymeric Hydrogels as In Vitro 3D Cell Culture Matrices. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2015. [Google Scholar]
- Kang, C.E.; Poon, P.C.; Tator, C.H.; Shoichet, M.S. A New Paradigm for Local and Sustained Release of Therapeutic Molecules to the Injured Spinal Cord for Neuroprotection and Tissue Repair. Tissue Eng. Part A 2008, 15, 595–604. [Google Scholar] [CrossRef]
- Sun, D.; Wang, N.; Li, L. Integrated sds removal and peptide separation by strong-cation exchange liquid chromatography for sds-assisted shotgun proteome analysis. J. Proteome Res. 2012, 11, 818–828. [Google Scholar] [CrossRef]
- Inacio, A.S.; Mesquita, K.A.; Baptista, M.; Ramalho-Santos, J.; Vaz, W.L.; Vieira, O.V. In vitro surfactant structure-toxicity relationships: Implications for surfactant use in sexually transmitted infection prophylaxis and contraception. PLoS ONE 2011, 6, e19850. [Google Scholar] [CrossRef]
- Anderberg, E.K.; Artursson, P. Epithelial transport of drugs in cell culture. Viii: Effects of sodium dodecyl sulfate on cell membrane and tight junction permeability in human intestinal epithelial (caco-2) cells. J. Pharm. Sci. 1993, 82, 392–398. [Google Scholar] [CrossRef]
- Yu, Q.; Wang, Z.; Pencheng, L.; Yang, Q. The effect of various absorption enhancers on tight junction in the human intestinal caco-2 cell line. Drug Dev. Ind. Pharm. 2012, 39, 587–592. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aprodu, A.; Mantaj, J.; Raimi-Abraham, B.; Vllasaliu, D. Evaluation of a Methylcellulose and Hyaluronic Acid Hydrogel as a Vehicle for Rectal Delivery of Biologics. Pharmaceutics 2019, 11, 127. https://doi.org/10.3390/pharmaceutics11030127
Aprodu A, Mantaj J, Raimi-Abraham B, Vllasaliu D. Evaluation of a Methylcellulose and Hyaluronic Acid Hydrogel as a Vehicle for Rectal Delivery of Biologics. Pharmaceutics. 2019; 11(3):127. https://doi.org/10.3390/pharmaceutics11030127
Chicago/Turabian StyleAprodu, Andreea, Julia Mantaj, Bahijja Raimi-Abraham, and Driton Vllasaliu. 2019. "Evaluation of a Methylcellulose and Hyaluronic Acid Hydrogel as a Vehicle for Rectal Delivery of Biologics" Pharmaceutics 11, no. 3: 127. https://doi.org/10.3390/pharmaceutics11030127
APA StyleAprodu, A., Mantaj, J., Raimi-Abraham, B., & Vllasaliu, D. (2019). Evaluation of a Methylcellulose and Hyaluronic Acid Hydrogel as a Vehicle for Rectal Delivery of Biologics. Pharmaceutics, 11(3), 127. https://doi.org/10.3390/pharmaceutics11030127





