What Drives Innovation: The Canadian Touch on Liposomal Therapeutics
Abstract
1. Perspective
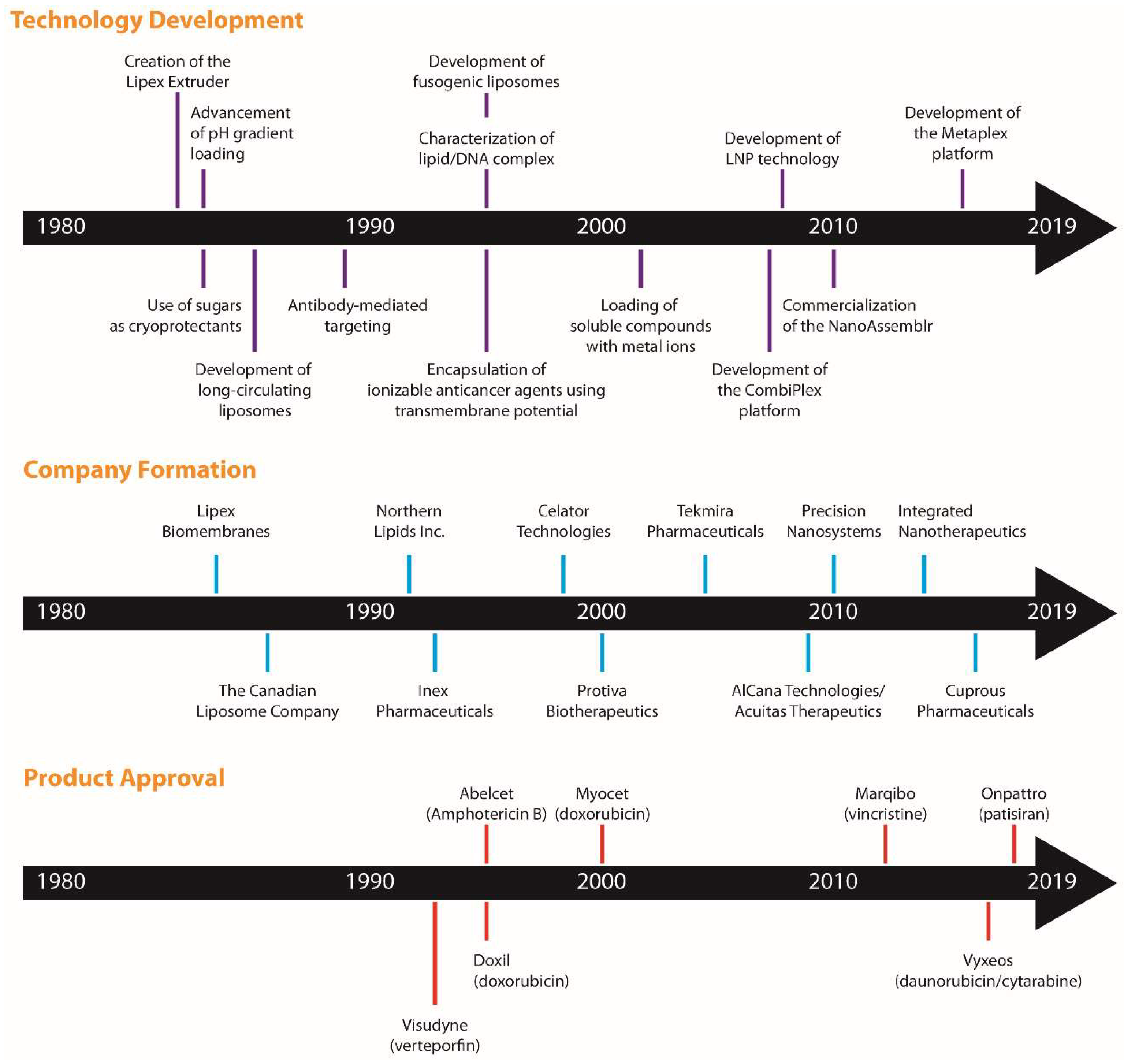
2. A Brief History
3. Technologies for the Production of Liposomes
4. Technologies for the Storage of Liposomes
5. Optimization of Liposomes for Pharmaceutical Use
5.1. Improvement of Encapsulation Efficiency of Passive Loading
5.2. Development of Remote Loading Methods
5.3. Development of Liposomes for Encapsulation of Nucleic Acids
6. Other Key Canadian Discoveries that Impacted the Development of Therapeutically Interesting Drugs
6.1. Selective Drug Delivery with Liposomes
6.2. The “PEGylation” Technology
6.3. Strategies to Encapsulate Multiple Agents
7. The Canadian Impact on Regulatory Approved and Investigational Liposomal Formulations
7.1. Liposomal Formulations of Amphotericin B: Abelcet® and iCo-019
7.2. Liposomal Formulations of Doxorubicin: Myocet® and Doxil®
7.3. Visudyne®
7.4. Marqibo®
7.5. Vyxeos®
7.6. Onpattro®
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shade, C.W. Liposomes as Advanced Delivery Systems for Nutraceuticals. Integr. Med. 2016, 15, 33–36. [Google Scholar]
- Weissig, V. Liposomes Came First: The Early History of Liposomology. In Liposomes: Methods and Protocols; D’Souza, G.G.M., Ed.; Springer: New York, NY, USA, 2017; pp. 1–15. [Google Scholar]
- Cullis, P.T.; Kruijff, B.D. Lipid polymorphism and the functional roles of lipids in biological membranes. Biochim. Biophys. Acta (BBA)-Rev. Biomembr. 1979, 559, 399–420. [Google Scholar] [CrossRef]
- Cullis, P.; Kruijff, B.D.; Hope, M.; Nayar, R.; Schmid, S.J.C. Phospholipids and membrane transport. Can. J. Biochem. 1980, 58, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Tam, Y.K.; Madden, T.D.; Hope, M.J. Pieter Cullis’ quest for a lipid-based, fusogenic delivery system for nucleic acid therapeutics: Success with siRNA so what about mRNA? J. Drug Target. 2016, 24, 774–779. [Google Scholar] [CrossRef] [PubMed]
- Sessa, G.; Weissmann, G. Incorporation of lysozyme into liposomes. A model for structure-linked latency. J. Biol. Chem. 1970, 245, 3295–3301. [Google Scholar]
- Trosper, T.; Raveed, D.; Ke, B. Chlorophyll a-containing liposomes. Biochim. Biophys. Acta 1970, 223, 463–465. [Google Scholar] [CrossRef]
- Gregoriadis, G.; Ryman, B.E. Lysosomal localization of -fructofuranosidase-containing liposomes injected into rats. Biochem. J. 1972, 129, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G. Drug entrapment in liposomes. FEBS Lett. 1973, 36, 292–296. [Google Scholar] [CrossRef]
- Gregoriadis, G.; Buckland, R.A. Enzyme-containing liposomes alleviate a model for storage disease. Nature 1973, 244, 170–172. [Google Scholar] [CrossRef]
- Blomhoff, H.K.; Blomhoff, R.; Christensen, T.B. Enhanced stability of β-galactosidase in parenchymal and nonparenchymal liver cells by conjugation with dextran. Biochim. Biophys. Acta (BBA) Gener. Subj. 1983, 757, 202–208. [Google Scholar] [CrossRef]
- Allison, A.G.; Gregoriadis, G. Liposomes as immunological adjuvants. Nature 1974, 252, 252. [Google Scholar] [CrossRef] [PubMed]
- Gregoriadis, G.; Allison, A.C. Entrapment of proteins in liposomes prevents allergic reactions in pre-immunised mice. FEBS Lett. 1974, 45, 71–74. [Google Scholar] [CrossRef]
- Arakawa, E.; Imai, Y.; Kobayashi, H.; Okumura, K.; Sezaki, H. Application of drug-containing liposomes to the duration of the intramuscular absorption of water-soluble drugs in rats. Chem. Pharm. Bull. 1975, 23, 2218–2222. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Taneda, K.; Kobayashi, H.; Okumura, K.; Muranishi, S.; Sezaki, H. Application of liposomes to the pharmaceutical modification of the distribution characteristics of drugs in the rat. Chem. Pharm. Bull. 1975, 23, 3069–3074. [Google Scholar] [CrossRef] [PubMed]
- Juliano, R.L.; Stamp, D. The effect of particle size and charge on the clearance rates of liposomes and liposome encapsulated drugs. Biochem. Biophys. Res. Commun. 1975, 63, 651–658. [Google Scholar] [CrossRef]
- Steerenberg, P.A.; Storm, G.; de Groot, G.; Claessen, A.; Bergers, J.J.; Franken, M.A.; van Hoesel, Q.G.; Wubs, K.L.; de Jong, W.H. Liposomes as drug carrier system for cis-diamminedichloroplatinum (II). II. Antitumor activity in vivo, induction of drug resistance, nephrotoxicity and Pt distribution. Cancer Chemother. Pharmacol. 1988, 21, 299–307. [Google Scholar] [CrossRef]
- Sharma, A.; Straubinger, N.L.; Straubinger, R.M. Modulation of human ovarian tumor cell sensitivity to N-(phosphonacetyl)-l-aspartate (PALA) by liposome drug carriers. Pharm. Res. 1993, 10, 1434–1441. [Google Scholar] [CrossRef] [PubMed]
- Desmukh, D.S.; Bear, W.D.; Wisniewski, H.M.; Brockerhoff, H. Long-living liposomes as potential drug carriers. Biochem. Biophys. Res. Commun. 1978, 82, 328–334. [Google Scholar] [CrossRef]
- Mezei, M.; Gulasekharam, V. Liposomes—A selective drug delivery system for the topical route of administration. Lotion dosage form. Life Sci. 1980, 26, 1473–1477. [Google Scholar] [CrossRef]
- Harsanyi, B.B.; Hilchie, J.C.; Mezei, M. Liposomes as drug carriers for oral ulcers. J. Dent. Res. 1986, 65, 1133–1141. [Google Scholar] [CrossRef]
- Foldvari, M.; Gesztes, A.; Mezei, M. Dermal drug delivery by liposome encapsulation: Clinical and electron microscopic studies. J. Microencapsul. 1990, 7, 479–489. [Google Scholar] [CrossRef]
- Caddeo, C.; Manconi, M.; Sinico, C.; Valenti, D.; Celia, C.; Monduzzi, M.; Fadda, A.M. Penetration Enhancer-Containing Vesicles: Does the Penetration Enhancer Structure Affect Topical Drug Delivery? Curr. Drug Targets 2015, 16, 1438–1447. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Valenti, D.; Nacher, A.; Manconi, M.; Fadda, A.M. Exploring the co-loading of lidocaine chemical forms in surfactant/phospholipid vesicles for improved skin delivery. J. Pharm. Pharmacol. 2015, 67, 909–917. [Google Scholar] [CrossRef]
- Peyman, G.A.; Charles, H.C.; Liu, K.R.; Khoobehi, B.; Niesman, M. Intravitreal liposome-encapsulated drugs: A preliminary human report. Int. Ophthalmol. 1988, 12, 175–182. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal formulations in clinical use: An updated review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Kalra, J.; Bally, M.B. Liposomes. In Fundamentals of Pharmaceutical Nanoscience; Springer: Berlin/Heidelberg, Germany, 2013; pp. 27–63. [Google Scholar]
- Leung, A.W.; Kalra, J.; Santos, N.D.; Bally, M.B.; Anglesio, M.S. Harnessing the potential of lipid-based nanomedicines for type-specific ovarian cancer treatments. Nanomedicine 2014, 9, 501–522. [Google Scholar] [CrossRef]
- Olson, F.; Hunt, C.A.; Szoka, F.C.; Vail, W.J.; Papahadjopoulos, D. Preparation of liposomes of defined size distribution by extrusion through polycarbonate membranes. Biochim. Biophys. Acta (BBA) Biomembr. 1979, 557, 9–23. [Google Scholar] [CrossRef]
- Mayer, L.D.; Hope, M.J.; Cullis, P.R. Vesicles of variable sizes produced by a rapid extrusion procedure. Biochim. Biophys. Acta (BBA) Biomembr. 1986, 858, 161–168. [Google Scholar] [CrossRef]
- Cullis, P.R.; Hope, M.J.; Bally, M.B. Extrusion Technique for Producing Unilamellar Vesicles. U.S. Patent US 5,008,050, 16 April 1991. [Google Scholar]
- Patil, Y.P.; Jadhav, S. Novel methods for liposome preparation. Chem. Phys. Lipids 2014, 177, 8–18. [Google Scholar] [CrossRef]
- Jahn, A.; Vreeland, W.N.; Gaitan, M.; Locascio, L.E. Controlled Vesicle Self-Assembly in Microfluidic Channels with Hydrodynamic Focusing. J. Am. Chem. Soc. 2004, 126, 2674–2675. [Google Scholar] [CrossRef]
- Evers, M.J.W.; Kulkarni, J.A.; van der Meel, R.; Cullis, P.R.; Vader, P.; Schiffelers, R.M. State-of-the-Art Design and Rapid-Mixing Production Techniques of Lipid Nanoparticles for Nucleic Acid Delivery. Small Methods 2018, 2, 1700375. [Google Scholar] [CrossRef]
- Zhigaltsev, I.V.; Belliveau, N.; Hafez, I.; Leung, A.K.; Huft, J.; Hansen, C.; Cullis, P.R.J.L. Bottom-up design and synthesis of limit size lipid nanoparticle systems with aqueous and triglyceride cores using millisecond microfluidic mixing. Langmuir 2012, 28, 3633–3640. [Google Scholar] [CrossRef]
- Belliveau, N.M.; Huft, J.; Lin, P.J.; Chen, S.; Leung, A.K.; Leaver, T.J.; Wild, A.W.; Lee, J.B.; Taylor, R.J.; Tam, Y.K. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Mol. Ther.-Nucleic Acids 2012, 1, e37. [Google Scholar] [CrossRef] [PubMed]
- Forbes, N.; Hussain, M.T.; Briuglia, M.L.; Edwards, D.P.; Horst, J.H.T.; Szita, N.; Perrie, Y. Rapid and scale-independent microfluidic manufacture of liposomes entrapping protein incorporating in-line purification and at-line size monitoring. Int. J. Pharm. 2018, 556, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Bally, M.B.; Zhang, Y.-P.; Reimer, D.L.; Wheeler, J.J. Lipid-Nucleic Acid Particles Prepared via a hydrophobic Lipid-Nucleic Acid Complex Intermediate and Use for Gene Transfer. U.S. Patent US 5,705,385, 6 January 1998. [Google Scholar]
- Wong, F.M.; Reimer, D.L.; Bally, M.B. Cationic lipid binding to DNA: Characterization of complex formation. Biochemistry 1996, 35, 5756–5763. [Google Scholar] [CrossRef]
- Reimer, D.L.; Zhang, Y.P.; Kong, S.; Wheeler, J.J.; Graham, R.W.; Bally, M.B. Formation of Novel Hydrophobic Complexes between Cationic Lipids and Plasmid DNA. Biochemistry 1995, 34, 12877–12883. [Google Scholar] [CrossRef] [PubMed]
- Crowe, L.M.; Crowe, J.H.; Rudolph, A.; Womersley, C.; Appel, L. Preservation of freeze-dried liposomes by trehalose. Arch. Biochem. Biophys. 1985, 242, 240–247. [Google Scholar] [CrossRef]
- Janoff, A.S.; Cullis, P.R.; Bally, M.B.; Fountain, M.W.; Ginsberg, R.S.; Hope, M.J.; Madden, T.D.; Schieren, H.P.; Jablonski, R.L. Methods of Dehydrating, Storing and Rehydrating Liposomes. U.S. Patent US 5,922,350, 13 July 1999. [Google Scholar]
- Janoff, A.S.; Cullis, P.R.; Bally, M.B.; Fountain, M.W.; Ginsberg, R.S.; Hope, M.J.; Madden, T.D.; Schieren, H.P.; Jablonski, R.L. Dehydrated Liposomes. U.S. Patent US 4,880,635, 14 November 1989. [Google Scholar]
- Janoff, A.S.; Cullis, P.R.; Bally, M.B.; Fountain, M.W.; Ginsberg, R.S.; Hope, M.J.; Madden, T.D.; Schieren, H.P.; Jablonski, R.L. Method of Dehydrating Liposomes Using Protective Sugars. U.S. Patent US 5,578,320, 26 November 1996. [Google Scholar]
- Madden, T.D.; Bally, M.B.; Hope, M.J.; Cullis, P.R.; Schieren, H.P.; Janoff, A.S. Protection of large unilamellar vesicles by trehalose during dehydration: Retention of vesicle contents. Biochim. Biophys. Acta (BBA) Biomembr. 1985, 817, 67–74. [Google Scholar] [CrossRef]
- Dos Santos, N.; Cox, K.A.; McKenzie, C.A.; van Baarda, F.; Gallagher, R.C.; Karlsson, G.; Edwards, K.; Mayer, L.D.; Allen, C.; Bally, M.B. pH gradient loading of anthracyclines into cholesterol-free liposomes: Enhancing drug loading rates through use of ethanol. Biochim. Biophys. Acta (BBA) Biomembr. 2004, 1661, 47–60. [Google Scholar] [CrossRef]
- Cabral-lilly, D.; Mayer, L.; Tardi, P.; Watkins, D.; Zeng, Y. Method of Lyophilizing Liposomes. U.S. Patent 14/352,662, 24 July 2018. [Google Scholar]
- Kirby, C.; Gregoriadis, G. Dehydration-Rehydration Vesicles: A Simple Method for High Yield Drug Entrapment in Liposomes. Bio/Technology 1984, 2, 979. [Google Scholar] [CrossRef]
- Arora, A.; Byrem, T.M.; Nair, M.G.; Strasburg, G.M. Modulation of Liposomal Membrane Fluidity by Flavonoids and Isoflavonoids. Arch. Biochem. Biophys. 2000, 373, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Bally, M.B.; Hope, M.J.; Van Echteld, C.J.A.; Cullis, P.R. Uptake of safranine and other lipophilic cations into model membrane systems in response to a membrane potential. Biochim. Biophys. Acta (BBA) Biomembr. 1985, 812, 66–76. [Google Scholar] [CrossRef]
- Fritze, A.; Hens, F.; Kimpfler, A.; Schubert, R.; Peschka-Süss, R. Remote loading of doxorubicin into liposomes driven by a transmembrane phosphate gradient. Biochim. Biophys. Acta (BBA) Biomembr. 2006, 1758, 1633–1640. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, P.R.; Wong, K.F.; Redelmeier, T.E.; Wheeler, J.J.; Cullis, P.R. Accumulation of doxorubicin and other lipophilic amines into large unilamellar vesicles in response to transmembrane pH gradients. Biochim. Biophys. Acta (BBA) Biomembr. 1993, 1149, 329–338. [Google Scholar] [CrossRef]
- Madden, T.D.; Harrigan, P.R.; Tai, L.C.; Bally, M.B.; Mayer, L.D.; Redelmeier, T.E.; Loughrey, H.C.; Tilcock, C.P.; Reinish, L.W.; Cullis, P.R. The accumulation of drugs within large unilamellar vesicles exhibiting a proton gradient: A survey. Chem. Phys. Lipids 1990, 53, 37–46. [Google Scholar] [CrossRef]
- Fenske, D.B.; Wong, K.F.; Maurer, E.; Maurer, N.; Leenhouts, J.M.; Boman, N.; Amankwa, L.; Cullis, P.R. Ionophore-mediated uptake of ciprofloxacin and vincristine into large unilamellar vesicles exhibiting transmembrane ion gradients. Biochim. Biophys. Acta (BBA) Biomembr. 1998, 1414, 188–204. [Google Scholar] [CrossRef]
- Nii, T.; Ishii, F. Encapsulation efficiency of water-soluble and insoluble drugs in liposomes prepared by the microencapsulation vesicle method. Int. J. Pharm. 2005, 298, 198–205. [Google Scholar] [CrossRef]
- Ciobanu, M.; Heurtault, B.; Schultz, P.; Ruhlmann, C.; Muller, C.D.; Frisch, B. Layersome: Development and optimization of stable liposomes as drug delivery system. Int. J. Pharm. 2007, 344, 154–157. [Google Scholar] [CrossRef]
- Franze, S.; Marengo, A.; Stella, B.; Minghetti, P.; Arpicco, S.; Cilurzo, F. Hyaluronan-decorated liposomes as drug delivery systems for cutaneous administration. Int. J. Pharm. 2018, 535, 333–339. [Google Scholar] [CrossRef]
- Cheung, B.C.L.; Sun, T.H.T.; Leenhouts, J.M.; Cullis, P.R. Loading of doxorubicin into liposomes by forming Mn2+-drug complexes. Biochim. Biophys. Acta (BBA) Biomembr. 1998, 1414, 205–216. [Google Scholar] [CrossRef]
- Abraham, S.A.; Edwards, K.; Karlsson, G.; MacIntosh, S.; Mayer, L.D.; McKenzie, C.; Bally, M.B. Formation of transition metal–doxorubicin complexes inside liposomes. Biochim. Biophys. Acta (BBA) Biomembr. 2002, 1565, 41–54. [Google Scholar] [CrossRef]
- Wehbe, M.; Anantha, M.; Backstrom, I.; Leung, A.; Chen, K.; Malhotra, A.; Edwards, K.; Bally, M.B. Nanoscale Reaction Vessels Designed for Synthesis of Copper-Drug Complexes Suitable for Preclinical Development. PLoS ONE 2016, 11, e0153416. [Google Scholar] [CrossRef] [PubMed]
- Wehbe, M.; Chernov, L.; Chen, K.; Bally, M.B. PRCosomes: Pretty reactive complexes formed in liposomes. J. Drug Target. 2016, 24, 787–796. [Google Scholar] [CrossRef]
- Hope, M.J.; Madden, T.D.; Cullis, P.R.; Maier, M.; Jayaraman, M.; Rajeev, K.G.; Akinc, A.; Manoharan, M. Methods and Compositions for Delivery of Nucleic Acids. U.S. Patent Application US 15/152,216, 3 November 2016. [Google Scholar]
- Hope, M.J.; Semple, S.C.; Chen, J.; Madden, T.D.; Cullis, P.R.; Ciufolini, M.A.; Mui, B.L.S. Amino Lipids and Methods for the Delivery of Nucleic Acids. U.S. Patent US 9,139,554, 22 September 2015. [Google Scholar]
- Jayaraman, M.; Ansell, S.M.; Mui, B.L.; Tam, Y.K.; Chen, J.; Du, X.; Butler, D.; Eltepu, L.; Matsuda, S.; Narayanannair, J.K.; et al. Maximizing the Potency of siRNA Lipid Nanoparticles for Hepatic Gene Silencing In Vivo. Angew. Chem. Int. Ed. 2012, 51, 8529–8533. [Google Scholar] [CrossRef]
- Fendler, J.H.; Romero, A. Liposomes as drug carriers. Life Sci. 1977, 20, 1109–1120. [Google Scholar] [CrossRef]
- Stamp, D.; Juliano, R.L. Factors affecting the encapsulation of drugs within liposomes. Can. J. Physiol. Pharmacol. 1979, 57, 535–539. [Google Scholar] [CrossRef]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015, 10, 975–999. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.W.; Deamer, D.W. Catecholamine uptake and concentration by liposomes maintaining pH gradients. Biochim. Biophys. Acta (BBA) Biomembr. 1976, 455, 269–271. [Google Scholar] [CrossRef]
- Cullis, P.R.; Hope, M.J.; Bally, M.B.; Madden, T.D.; Mayer, L.D.; Fenske, D.B. Influence of pH gradients on the transbilayer transport of drugs, lipids, peptides and metal ions into large unilamellar vesicles. Biochim. Biophys. Acta (BBA)-Rev. Biomembr. 1997, 1331, 187–211. [Google Scholar] [CrossRef]
- Wheeler, J.J.; Veiro, J.A.; Cullis, P.R. Ionophore-mediated loading of Ca2+ into large unilamellar vesicles in response to transmembrane pH gradients. Mol. Membr. Biol. 1994, 11, 151–157. [Google Scholar] [CrossRef]
- Immordino, M.L.; Dosio, F.; Cattel, L. Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006, 1, 297–315. [Google Scholar]
- Tardi, P.; Johnstone, S.; Webb, M.; Bally, M.; Abraham, S. Liposome Loading with Metal Ions. U.S. Patent US 7,238,367, 3 July 2007. [Google Scholar]
- Taggar, A.S.; Alnajim, J.; Anantha, M.; Thomas, A.; Webb, M.; Ramsay, E.; Bally, M.B. Copper–topotecan complexation mediates drug accumulation into liposomes. J. Control. Release 2006, 114, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Abraham, S.A.; Edwards, K.; Karlsson, G.; Hudon, N.; Mayer, L.D.; Bally, M.B. An evaluation of transmembrane ion gradient-mediated encapsulation of topotecan within liposomes. J. Control. Release 2004, 96, 449–461. [Google Scholar] [CrossRef]
- Ramsay, E.; Alnajim, J.; Anantha, M.; Taggar, A.; Thomas, A.; Edwards, K.; Karlsson, G.; Webb, M.; Bally, M. Transition Metal-Mediated Liposomal Encapsulation of Irinotecan (CPT-11) Stabilizes the Drug in the Therapeutically Active Lactone Conformation. Pharm. Res. 2006, 23, 2799–2808. [Google Scholar] [CrossRef]
- DiGiulio, S. FDA approves onivyde combo regimen for advanced pancreatic cancer. Oncol. Times 2015. [Google Scholar] [CrossRef]
- Tardi, P.; Johnstone, S.; Harasym, N.; Xie, S.; Harasym, T.; Zisman, N.; Harvie, P.; Bermudes, D.; Mayer, L. In vivo maintenance of synergistic cytarabine:daunorubicin ratios greatly enhances therapeutic efficacy. Leukemia Res. 2009, 33, 129–139. [Google Scholar] [CrossRef]
- Banys, P. The clinical use of disulfiram (Antabuse®): A review. J. Psychoact. Drugs 1988, 20, 243–261. [Google Scholar] [CrossRef] [PubMed]
- San Andres, M.P.; Marina, M.L.; Vera, S. Spectrophotometric determination of copper(II), nickel(II) and cobalt(II) as complexes with sodium diethyldithiocarbamate in cationic micellar medium of hexadecyltrimethylammonium salts. Talanta 1994, 41, 179–185. [Google Scholar] [CrossRef]
- Wehbe, M.; Anantha, M.; Shi, M.; Leung, A.W.; Dragowska, W.H.; Sanche, L.; Bally, M.B. Development and optimization of an injectable formulation of copper diethyldithiocarbamate, an active anticancer agent. Int. J. Nanomed. 2017, 12, 4129–4146. [Google Scholar] [CrossRef]
- Chen, D.; Cui, Q.C.; Yang, H.; Dou, Q.P. Disulfiram, a clinically used anti-alcoholism drug and copper-binding agent, induces apoptotic cell death in breast cancer cultures and xenografts via inhibition of the proteasome activity. Cancer Res. 2006, 66, 10425–10433. [Google Scholar] [CrossRef]
- Liu, P.; Brown, S.; Goktug, T.; Channathodiyil, P.; Kannappan, V.; Hugnot, J.; Guichet, P.; Bian, X.; Armesilla, A.; Darling, J.L. Cytotoxic effect of disulfiram/copper on human glioblastoma cell lines and ALDH-positive cancer-stem-like cells. Br. J. Cancer 2012, 107, 1488. [Google Scholar] [CrossRef] [PubMed]
- Duncan, C.; White, A.R. Copper complexes as therapeutic agents. Metallomics 2012, 4, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C.J. Advances in copper complexes as anticancer agents. Chem. Rev. 2013, 114, 815–862. [Google Scholar] [CrossRef] [PubMed]
- Marzano, C.; Pellei, M.; Tisato, F.; Santini, C. Copper complexes as anticancer agents. Anti-Cancer Agents Med. Chem. 2009, 9, 185–211. [Google Scholar] [CrossRef]
- Rafique, S.; Idrees, M.; Nasim, A.; Akbar, H.; Athar, A.J.B.; Reviews, M.B. Transition metal complexes as potential therapeutic agents. Biotechnol. Mol. Biol. Rev. 2010, 5, 38–45. [Google Scholar]
- Wehbe, M.; Leung, A.W.; Abrams, M.J.; Orvig, C.; Bally, M.B. A Perspective–can copper complexes be developed as a novel class of therapeutics? Dalton Trans. 2017, 46, 10758–10773. [Google Scholar] [CrossRef] [PubMed]
- Weder, J.E.; Dillon, C.T.; Hambley, T.W.; Kennedy, B.J.; Lay, P.A.; Biffin, J.R.; Regtop, H.L.; Davies, N.M. Copper complexes of non-steroidal anti-inflammatory drugs: An opportunity yet to be realized. Coord. Chem. Rev. 2002, 232, 95–126. [Google Scholar] [CrossRef]
- Wehbe, M.; Lo, C.; Leung, A.W.Y.; Dragowska, W.H.; Ryan, G.M.; Bally, M.B. Copper (II) complexes of bidentate ligands exhibit potent anti-cancer activity regardless of platinum sensitivity status. Investig. New Drugs 2017, 35, 682–690. [Google Scholar] [CrossRef]
- Wehbe, M.; Malhotra, A.K.; Anantha, M.; Lo, C.; Dragowska, W.H.; Dos Santos, N.; Bally, M.B. Development of a copper-clioquinol formulation suitable for intravenous use. Drug Deliv. Transl. Res. 2018, 8, 239–251. [Google Scholar] [CrossRef]
- Prosser, K.E.; Leung, A.W.; Harrypersad, S.; Lewis, A.R.; Bally, M.B.; Walsby, C.J. Transition Metal Ions Promote the Bioavailability of Hydrophobic Therapeutics: Cu and Zn Interactions with RNA Polymerase I Inhibitor CX5461. Chemistry 2018, 24, 6334–6338. [Google Scholar] [CrossRef]
- Leung, A.; Anantha, M.; Dragowska, W.; Wehbe, M.; Bally, M.J. Copper-CX-5461: A novel liposomal formulation for a small molecule rRNA synthesis inhibitor. J. Control. Release 2018, 286, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kepp, O.; Senovilla, L.; Vitale, I.; Vacchelli, E.; Adjemian, S.; Agostinis, P.; Apetoh, L.; Aranda, F.; Barnaba, V.; Bloy, N. Consensus guidelines for the detection of immunogenic cell death. Oncoimmunology 2014, 3, e955691. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Kepp, O.; Zitvogel, L. Immunogenic cell death in cancer therapy. Ann. Rev. Immunol. 2013, 31, 51–72. [Google Scholar] [CrossRef]
- Obeid, M.; Tesniere, A.; Ghiringhelli, F.; Fimia, G.M.; Apetoh, L.; Perfettini, J.-L.; Castedo, M.; Mignot, G.; Panaretakis, T.; Casares, N. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat. Med. 2007, 13, 54. [Google Scholar] [CrossRef] [PubMed]
- Terenzi, A.; Pirker, C.; Keppler, B.K.; Berger, W. Anticancer metal drugs and immunogenic cell death. J. Inorg. Biochem. 2016, 165, 71–79. [Google Scholar] [CrossRef]
- Mookerjee, A.; Basu, J.M.; Majumder, S.; Chatterjee, S.; Panda, G.S.; Dutta, P.; Pal, S.; Mukherjee, P.; Efferth, T.; Roy, S.J. A novel copper complex induces ROS generation in doxorubicin resistant Ehrlich ascitis carcinoma cells and increases activity of antioxidant enzymes in vital organs in vivo. BMC Cancer 2006, 6, 267. [Google Scholar] [CrossRef]
- Boyle, E.; Freeman, P.; Goudie, A.; Mangan, F.; Thomson, M.J. The role of copper in preventing gastrointestinal damage by acidic anti-inflammatory drugs. J. Pharm. Pharmacol. 1976, 28, 865–868. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Ghani, A.-S.; Abu-Hijleh, A.-L.; Nahas, N.; Amin, R. Hypoglycemic effect of copper(II) acetate imidazole complexes. Biol. Trace Elem. Res. 1996, 54, 143–151. [Google Scholar] [CrossRef]
- Yasumatsu, N.; Yoshikawa, Y.; Adachi, Y.; Sakurai, H. Antidiabetic copper(II)-picolinate: Impact of the first transition metal in the metallopicolinate complexes. Bioorg. Med. Chem. 2007, 15, 4917–4922. [Google Scholar] [CrossRef]
- Djoko, K.Y.; Achard, M.E.S.; Phan, M.-D.; Lo, A.W.; Miraula, M.; Prombhul, S.; Hancock, S.J.; Peters, K.M.; Sidjabat, H.E.; Harris, P.N.; et al. Copper Ions and Coordination Complexes as Novel Carbapenem Adjuvants. J. Antimicrob. Agents Chemother. 2018, 62, e02280-17. [Google Scholar] [CrossRef]
- Haeili, M.; Moore, C.; Davis, C.J.; Cochran, J.B.; Shah, S.; Shrestha, T.B.; Zhang, Y.; Bossmann, S.H.; Benjamin, W.H.; Kutsch, O.J.; et al. Copper complexation screen reveals compounds with potent antibiotic properties against methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2014, 58, 3727–3736. [Google Scholar] [CrossRef] [PubMed]
- Gaggelli, E.; Kozlowski, H.; Valensin, D.; Valensin, G.J. Copper homeostasis and neurodegenerative disorders (Alzheimer’s, prion, and Parkinson’s diseases and amyotrophic lateral sclerosis). Chem. Rev. 2006, 106, 1995–2044. [Google Scholar] [CrossRef]
- Kozlowski, H.; Luczkowski, M.; Remelli, M.; Valensin, D.J. Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases). Coord. Chem. Rev. 2012, 256, 2129–2141. [Google Scholar] [CrossRef]
- Tisato, F.; Marzano, C.; Porchia, M.; Pellei, M.; Santini, C. Copper in diseases and treatments, and copper-based anticancer strategies. Med. Res. Rev. 2010, 30, 708–749. [Google Scholar] [CrossRef] [PubMed]
- Giampietro, R.; Spinelli, F.; Contino, M.; Colabufo, N.A. The Pivotal Role of Copper in Neurodegeneration: A New Strategy for the Therapy of Neurodegenerative Disorders. Mol. Pharm. 2018, 15, 808–820. [Google Scholar] [CrossRef]
- Gershon, H.; Ghirlando, R.; Guttman, S.B.; Minsky, A. Mode of formation and structural features of DNA-cationic liposome complexes used for transfection. Biochemistry 1993, 32, 7143–7151. [Google Scholar] [CrossRef]
- Dimitraidis, G.J. Introduction of ribonucleic acids into cells by means of liposomes. Nucleic Acids Res. 1978, 5, 1381–1386. [Google Scholar] [CrossRef][Green Version]
- Fraley, R.T.; Fornari, C.S.; Kaplan, S. Entrapment of a bacterial plasmid in phospholipid vesicles: Potential for gene transfer. Proc. Natl. Acad. Sci. USA 1979, 76, 3348–3352. [Google Scholar] [CrossRef]
- Fraley, R.; Subramani, S.; Berg, P.; Papahadjopoulos, D. Introduction of liposome-encapsulated SV40 DNA into cells. J. Biol. Chem. 1980, 255, 10431–10435. [Google Scholar]
- Wong, F.M.P.; MacAdam, S.A.; Kim, A.; Oja, C.; Ramsay, E.C.; Bally, M.B. A Lipid-based Delivery System for Antisense Oligonucleotides Derived from a Hydrophobic Complex. J. Drug Target. 2002, 10, 615–623. [Google Scholar] [CrossRef]
- Wheeler, J.; Bally, M.B.; Zhang, Y.-P.; Reimer, D.L.; Hope, M. Method of Preventing Aggregation of a Lipid: Nucleic Acid Complex. U.S. Patent US 6,858,224, 22 Febuary 2005. [Google Scholar]
- Wheeler, J.J.; Palmer, L.; Ossanlou, M.; MacLachlan, I.; Graham, R.W.; Zhang, Y.P.; Hope, M.J.; Scherrer, P.; Cullis, P.R. Stabilized plasmid-lipid particles: Construction and characterization. Gene Ther. 1999, 6, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Semple, S.C.; Akinc, A.; Chen, J.; Sandhu, A.P.; Mui, B.L.; Cho, C.K.; Sah, D.W.Y.; Stebbing, D.; Crosley, E.J.; Yaworski, E.; et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 2010, 28, 172. [Google Scholar] [CrossRef]
- Hu, Q.; Shew, C.R.; Bally, M.B.; Madden, T.D. Programmable fusogenic vesicles for intracellular delivery of antisense oligodeoxynucleotides: Enhanced cellular uptake and biological effects. Biochim. Biophys. Acta (BBA) Biomembr. 2001. [Google Scholar] [CrossRef]
- Holland, J.W.; Madden, T.D.; Cullis, P.R. Liposome Having an Exchangeable Component. U.S. Patent US 6,673,364, 6 January 2004. [Google Scholar]
- Tam, Y.Y.; Chen, S.; Cullis, P.R. Advances in Lipid Nanoparticles for siRNA Delivery. Pharmaceutics 2013, 5, 498–507. [Google Scholar] [CrossRef]
- Zhao, Y.; Huang, L. Chapter Two—Lipid Nanoparticles for Gene Delivery. In Advances in Genetics; Huang, L., Liu, D., Wagner, E., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 88, pp. 13–36. [Google Scholar]
- Heyes, J.; Palmer, L.; Bremner, K.; MacLachlan, I. Cationic lipid saturation influences intracellular delivery of encapsulated nucleic acids. J. Control. Release 2005, 107, 276–287. [Google Scholar] [CrossRef]
- Maier, M.A.; Jayaraman, M.; Matsuda, S.; Liu, J.; Barros, S.; Querbes, W.; Tam, Y.K.; Ansell, S.M.; Kumar, V.; Qin, J.; et al. Biodegradable lipids enabling rapidly eliminated lipid nanoparticles for systemic delivery of RNAi therapeutics. Mol. Ther. 2013, 21, 1570–1578. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M. Long-circulating (sterically stabilized) liposomes for targeted drug delivery. Trends Pharmacol. Sci. 1994, 15, 215–220. [Google Scholar] [CrossRef]
- Allen, T.M.; Hansen, C.; Rutledge, J.J. Liposomes with prolonged circulation times: Factors affecting uptake by reticuloendothelial and other tissues. Biochim. Biophys. Acta (BBA)-Biomembr. 1989, 981, 27–35. [Google Scholar] [CrossRef]
- de Menezes, D.E.L.; Pilarski, L.M.; Allen, T.M.J. In vitro and in vivo targeting of immunoliposomal doxorubicin to human B-cell lymphoma. Cancer Res. 1998, 58, 3320–3330. [Google Scholar]
- Dos Santos, N. Characterization of Cholesterol-Free Liposomes for Use in Delivery of Anti-Cancer Drugs; University of British Columbia: Vancouver, BC, Canada, 2004. [Google Scholar]
- Gregoriadis, G.; Neerunjun, E.D. Homing of liposomes to target cells. Biochem. Biophys. Res. Commun. 1975, 65, 537–544. [Google Scholar] [CrossRef]
- Leserman, L.D.; Machy, P.; Barbet, J. Cell-specific drug transfer from liposomes bearing monoclonal antibodies. Nature 1981, 293, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Guru, P.Y.; Agrawal, A.K.; Singha, U.K.; Singhal, A.; Gupta, C.M. Drug targeting in Leishmania donovani infections using tuftsin-bearing liposomes as drug vehicles. FEBS Lett. 1989, 245, 204–208. [Google Scholar] [CrossRef]
- Osborne, M.P.; Richardson, V.J.; Jeyasing, K.; Ryman, B.E. Radionuclide-labelled liposomes—A new lymph node imaging agent. Int. J. Nuclear Med. Biol. 1979, 6, 75–83. [Google Scholar] [CrossRef]
- Morgan, J.R.; Williams, K.E.; Davies, R.L.; Leach, K.; Thomson, M.; Williams, L.A. Localisation of experimental staphylococcal abscesses by 99MTC-technetium-labelled liposomes. J. Med. Microbiol. 1981, 14, 213–217. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A New Concept for Macromolecular Therapeutics in Cancer Chemotherapy: Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Heath, T.D.; Fraley, R.T.; Papahdjopoulos, D. Antibody targeting of liposomes: Cell specificity obtained by conjugation of F(ab’)2 to vesicle surface. Science 1980, 210, 539. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.J.; Hubbell, W.L.; Papahadjopoulos, D. Immunospecific targeting of liposomes to cells: A novel and efficient method for covalent attachment of Fab’ fragments via disulfide bonds. Biochemistry 1981, 20, 4229–4238. [Google Scholar] [CrossRef]
- Ahmad, I.; Allen, T.M. Antibody-mediated specific binding and cytotoxicity of liposome-entrapped doxorubicin to lung cancer cells in vitro. Cancer Res 1992, 52, 4817–4820. [Google Scholar]
- Park, J.W.; Hong, K.; Kirpotin, D.B.; Colbern, G.; Shalaby, R.; Baselga, J.; Shao, Y.; Nielsen, U.B.; Marks, J.D.; Moore, D.; et al. Anti-HER2 immunoliposomes: Enhanced efficacy attributable to targeted delivery. Clin Cancer Res. 2002, 8, 1172–1181. [Google Scholar]
- ElBayoumi, T.A.; Torchilin, V.P. Tumor-targeted nanomedicines: enhanced antitumor efficacy in vivo of doxorubicin-loaded, long-circulating liposomes modified with cancer-specific monoclonal antibody. Clin. Cancer Res. 2009, 15, 1973. [Google Scholar] [CrossRef]
- Kirpotin, D.B.; Drummond, D.C.; Shao, Y.; Shalaby, M.R.; Hong, K.; Nielsen, U.B.; Marks, J.D.; Benz, C.C.; Park, J.W. Antibody Targeting of Long-Circulating Lipidic Nanoparticles Does Not Increase Tumor Localization but Does Increase Internalization in Animal Models. Cancer Res. 2006, 66, 6732. [Google Scholar] [CrossRef]
- Sapra, P.; Allen, T.M. Internalizing antibodies are necessary for improved therapeutic efficacy of antibody-targeted liposomal drugs. Cancer Res. 2002, 62, 7190–7194. [Google Scholar] [PubMed]
- Song, S.; Liu, D.; Peng, J.; Sun, Y.; Li, Z.; Gu, J.-R.; Xu, Y. Peptide ligand-mediated liposome distribution and targeting to EGFR expressing tumor in vivo. Int. J. Pharm. 2008, 363, 155–161. [Google Scholar] [CrossRef]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef]
- Lee, T.-Y.; Lin, C.-T.; Kuo, S.-Y.; Chang, D.-K.; Wu, H.-C. Peptide-Mediated Targeting to Tumor Blood Vessels of Lung Cancer for Drug Delivery. Cancer Res. 2007, 67, 10958. [Google Scholar] [CrossRef]
- Mai, J.; Song, S.; Rui, M.; Liu, D.; Ding, Q.; Peng, J.; Xu, Y. A synthetic peptide mediated active targeting of cisplatin liposomes to Tie2 expressing cells. J. Control. Release 2009, 139, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Lammers, T.; Hennink, W.E.; Storm, G. Tumour-targeted nanomedicines: Principles and practice. Br. J. Cancer 2008, 99, 392. [Google Scholar] [CrossRef]
- Bertrand, N.; Wu, J.; Xu, X.; Kamaly, N.; Farokhzad, O.C. Cancer nanotechnology: The impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Deliv. Rev. 2014, 66, 2–25. [Google Scholar] [CrossRef] [PubMed]
- Andresen, T.L.; Jensen, S.S.; Jørgensen, K. Advanced strategies in liposomal cancer therapy: Problems and prospects of active and tumor specific drug release. Prog. Lipid Res. 2005, 44, 68–97. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Chonn, A. Large Unilamellar Liposomes with Low Uptake into the Reticuloendothelial System. FEBS Lett. 1987, 223, 42–46. [Google Scholar] [CrossRef]
- Bakkerwoudenberg, I.A.J.M.; Lokerse, A.F.; Tenkate, M.T.; Storm, G. Enhanced Localization of Liposomes with Prolonged Blood-Circulation Time in Infected Lung-Tissue. Biochim. Biophys. Acta 1992, 1138, 318–326. [Google Scholar] [CrossRef]
- Shen, Z.; Fisher, A.; Liu, W.K.; Li, Y. 1—PEGylated “stealth” nanoparticles and liposomes. In Engineering of Biomaterials for Drug Delivery Systems; Parambath, A., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 1–26. [Google Scholar]
- Blume, G.; Cevc, G. Liposomes for the sustained drug release in vivo. Biochim. Biophys. Acta 1990, 1029, 91–97. [Google Scholar] [CrossRef]
- Kamps, J.A.; Swart, P.J.; Morselt, H.W.; Pauwels, R.; De Bethune, M.P.; De Clercq, E.; Meijer, D.K.; Scherphof, G.L. Preparation and characterization of conjugates of (modified) human serum albumin and liposomes: Drug carriers with an intrinsic anti-HIV activity. Biochim. Biophys. Acta 1996, 1278, 183–190. [Google Scholar] [CrossRef]
- Allen, T.M.; Papahadjopoulos, D. STERICALLY STABILIZED (“STEALTH”). Liposome Technol. 1992, 3, 59. [Google Scholar]
- Agrawal, V.; Paul, M.K.; Mukhopadhyay, A.K. 6-mercaptopurine and daunorubicin double drug liposomes-preparation, drug-drug interaction and characterization. J. Liposome Res. 2005, 15, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, E.C.; Santos, N.D.; Dragowska, W.H.; Laskin, J.J.; Bally, M.B. The Formulation of Lipid-Based Nanotechnologies for the Delivery of Fixed Dose Anticancer Drug Combinations. Curr. Drug Deliv. 2005, 2, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Mayer, L.D.; Harasym, T.O.; Tardi, P.G.; Harasym, N.L.; Shew, C.R.; Johnstone, S.A.; Ramsay, E.C.; Bally, M.B.; Janoff, A.S. Ratiometric dosing of anticancer drug combinations: Controlling drug ratios after systemic administration regulates therapeutic activity in tumor-bearing mice. Mol. Cancer Ther. 2006, 5, 1854–1863. [Google Scholar] [CrossRef] [PubMed]
- Mayer, L.D.; Janoff, A.S. Optimizing combination chemotherapy by controlling drug ratios. Mol. Int. 2007, 7, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Janoff, A.; Mayer, L.; Redman, J.; Swenson, C. Fixed Ratio Drug Combination Treatments for Solid Tumors. U.S. Patent US 7,842,676, 30 November 2010. [Google Scholar]
- Bovier, P.A. Epaxal®: A virosomal vaccine to prevent hepatitis A infection. Expert Rev. Vaccin. 2008, 7, 1141–1150. [Google Scholar] [CrossRef]
- Usonis, V.; Bakasenas, V.; Valentelis, R.; Katiliene, G.; Vidzeniene, D.; Herzog, C. Antibody titres after primary and booster vaccination of infants and young children with a virosomal hepatitis A vaccine (Epaxal®). Vaccine 2003, 21, 4588–4592. [Google Scholar] [CrossRef]
- Gabizon, A.; Goren, D.; Fuks, Z.; Barenholz, Y.; Dagan, A.; Meshorer, A. Enhancement of adriamycin delivery to liver metastatic cells with increased tumoricidal effect using liposomes as drug carriers. Cancer Res. 1983, 43, 4730–4735. [Google Scholar]
- Gabizon, A.; Barenholz, Y. Adriamycin-Containing Liposomes in Cancer Chemotherapy; Wiley: New York, NY, USA, 1988; pp. 365–379. [Google Scholar]
- James, N.; Coker, R.; Tomlinson, D.; Harris, J.; Gompels, M.; Pinching, A.; Stewart, J. Liposomal doxorubicin (Doxil): An effective new treatment for Kaposi’s sarcoma in AIDS. Clin. Oncol. 1994, 6, 294–296. [Google Scholar] [CrossRef]
- Clark, J.M.; Whitney, R.R.; Olsen, S.J.; George, R.J.; Swerdel, M.R.; Kunselman, L.; Bonner, D.P. Amphotericin B lipid complex therapy of experimental fungal infections in mice. Antimicrob. Agents Chemother. 1991, 35, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Janoff, A.; Perkins, W.; Saletan, S.; Swenson, C. Amphotericin B lipid complex (ABLC™): A molecular rationale for the attenuation of amphotericin B related toxicities. J. Liposome Res. 1993, 3, 451–471. [Google Scholar] [CrossRef]
- Adedoyin, A.; Bernardo, J.F.; Swenson, C.E.; Bolsack, L.E.; Horwith, G.; DeWit, S.; Kelly, E.; Klasterksy, J.; Sculier, J.-P.; DeValeriola, D.; et al. Pharmacokinetic profile of ABELCET (amphotericin B lipid complex injection): Combined experience from phase I and phase II studies. Antimicrob. Agents Chemother. 1997, 41, 2201–2208. [Google Scholar] [CrossRef] [PubMed]
- Gill, P.S.; Wernz, J.; Scadden, D.T.; Cohen, P.; Mukwaya, G.M.; von Roenn, J.H.; Jacobs, M.; Kempin, S.; Silverberg, I.; Gonzales, G.J. Randomized phase III trial of liposomal daunorubicin versus doxorubicin, bleomycin, and vincristine in AIDS-related Kaposi’s sarcoma. J. Clin. Oncol. 1996, 14, 2353–2364. [Google Scholar] [CrossRef]
- Forssen, E.A.; Ross, M.E. Daunoxome® Treatment of Solid Tumors: Preclinical and Clinical Investigations. J. Liposome Res. 1994, 4, 481–512. [Google Scholar] [CrossRef]
- Noskin, G.; Pietrelli, L.; Gurwith, M.; Bowden, R.J.B.m.t. Treatment of invasive fungal infections with amphotericin B colloidal dispersion in bone marrow transplant recipients. Bone Marrow Transplant. 1999, 23, 697. [Google Scholar] [CrossRef]
- Adler-Moore, J. AmBisome targeting to fungal infections. Bone Marrow Transplant. 1994, 14, S3–S7. [Google Scholar]
- Adler-Moore, J.; Proffitt, R.T. Ambisome: Lipsomal formulation, structure, mechanism of action and pre-clinical experience. J. Antimicrob. Chemother. 2002, 49, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Proffitt, R.T.; Satorius, A.; Chiang, S.-M.; Sullivan, L.; Adler-Moore, J.P. Pharmacology and toxicology of a liposomal formulation of amphotericin B (AmBisome) in rodents. J. Antimicrob. Chemother. 1991, 28, 49–61. [Google Scholar] [CrossRef]
- Herzog, C.; Hartmann, K.; Künzi, V.; Kürsteiner, O.; Mischler, R.; Lazar, H.; Glück, R. Eleven years of Inflexal® V—A virosomal adjuvanted influenza vaccine. Vaccine 2009, 27, 4381–4387. [Google Scholar] [CrossRef]
- Glantz, M.J.; Jaeckle, K.A.; Chamberlain, M.C.; Phuphanich, S.; Recht, L.; Swinnen, L.J.; Maria, B.; LaFollette, S.; Schumann, G.B.; Cole, B.F. A randomized controlled trial comparing intrathecal sustained-release cytarabine (DepoCyt) to intrathecal methotrexate in patients with neoplastic meningitis from solid tumors. Clin. Cancer Res. 1999, 5, 3394–3402. [Google Scholar] [PubMed]
- Murry, D.J.; Blaney, S.M. Clinical pharmacology of encapsulated sustained-release cytarabine. Ann. Pharmacother. 2000, 34, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Balazsovits, J.A.E.; Mayer, L.D.; Bally, M.B.; Cullis, P.R.; McDonell, M.; Ginsberg, R.S.; Falk, R.E. Analysis of the effect of liposome encapsulation on the vesicant properties, acute and cardiac toxicities, and antitumor efficacy of doxorubicin. Cancer Chemother. Pharmacol. 1989, 23, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Swenson, C.; Perkins, W.; Roberts, P.; Janoff, A.S. Liposome technology and the development of Myocet™(liposomal doxorubicin citrate). Breast 2001, 10, 1–7. [Google Scholar] [CrossRef]
- Keam, S.J.; Scott, L.J.; Curran, M.P. Verteporfin. Drugs 2003, 63, 2521–2554. [Google Scholar] [CrossRef] [PubMed]
- Frennesson, C.I.; Nilsson, S.E. Encouraging results of photodynamic therapy with Visudyne in a clinical patient material of age-related macular degeneration. Acta Ophthalmol. Scand. 2004, 82, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Kim, J.; Kim, S. Extended-release formulation of morphine for subcutaneous administration. Cancer Chemother. Pharmacol. 1993, 33, 187–190. [Google Scholar] [CrossRef]
- Biteau, K.; Guiho, R.; Chatelais, M.; Taurelle, J.; Chesneau, J.; Corradini, N.; Heymann, D.; Redini, F. L-MTP-PE and zoledronic acid combination in osteosarcoma: Preclinical evidence of positive therapeutic combination for clinical transfer. Am. J. Cancer Res. 2016, 6, 677. [Google Scholar]
- Richard, B.M.; Newton, P.; Ott, L.R.; Haan, D.; Brubaker, A.N.; Cole, P.I.; Ross, P.E.; Rebelatto, M.C.; Nelson, K.G. The safety of EXPAREL®(bupivacaine liposome injectable suspension) administered by peripheral nerve block in rabbits and dogs. J. Drug Deliv. 2012, 2012, 962101. [Google Scholar] [CrossRef] [PubMed]
- Mantripragada, S. A lipid based depot (DepoFoam® technology) for sustained release drug delivery. Prog. Lipid Res. 2002, 41, 392–406. [Google Scholar] [CrossRef]
- Boman, N.L.; Masin, D.; Mayer, L.D.; Cullis, P.R.; Bally, M.B. Liposomal vincristine which exhibits increased drug retention and increased circulation longevity cures mice bearing P388 tumors. Cancer Res. 1994, 54, 2830–2833. [Google Scholar] [PubMed]
- Webb, M.S.; Logan, P.; Kanter, P.M.; Onge, G.S.; Gelmon, K.; Harasym, T.; Mayer, L.D.; Bally, M.B. Preclinical pharmacology, toxicology and efficacy of sphingomyelin/cholesterol liposomal vincristine for therapeutic treatment of cancer. Cancer Chemother. Pharmacol. 1998, 42, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Kalra, A.V.; Kim, J.; Klinz, S.G.; Paz, N.; Cain, J.; Drummond, D.C.; Nielsen, U.B.; Fitzgerald, J.B. Preclinical activity of nanoliposomal irinotecan is governed by tumor deposition and intratumor prodrug conversion. Cancer Res. 2014, 74, 7003–7013. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.-S.; Tardi, P.G.; Dos Santos, N.; Xie, X.; Fan, M.; Liboiron, B.D.; Huang, X.; Harasym, T.O.; Bermudes, D.; Mayer, L.D. Leukemia-selective uptake and cytotoxicity of CPX-351, a synergistic fixed-ratio cytarabine: Daunorubicin formulation, in bone marrow xenografts. Leuk. Res. 2010, 34, 1214–1223. [Google Scholar] [CrossRef] [PubMed]
- Bayne, W.F.; Mayer, L.D.; Swenson, C.E. Pharmacokinetics of CPX-351 (cytarabine/daunorubicin HCl) liposome injection in the mouse. J. Pharm. Sci. 2009, 98, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Janoff, A.S.; Boni, L.T.; Popescu, M.C.; Minchey, S.R.; Cullis, P.R.; Madden, T.D.; Taraschi, T.; Gruner, S.M.; Shyamsunder, E.; Tate, M.W. Unusual lipid structures selectively reduce the toxicity of amphotericin B. Proc. Natl. Acad. Sci. USA 1988, 85, 6122. [Google Scholar] [CrossRef] [PubMed]
- Wasan, E.K.; Bartlett, K.; Gershkovich, P.; Sivak, O.; Banno, B.; Wong, Z.; Gagnon, J.; Gates, B.; Leon, C.G.; Wasan, K.M. Development and characterization of oral lipid-based Amphotericin B formulations with enhanced drug solubility, stability and antifungal activity in rats infected with Aspergillus fumigatus or Candida albicans. Int. J. Pharm. 2009, 372, 76–84. [Google Scholar] [CrossRef]
- Waterhouse, D.N.; Tardi, P.G.; Mayer, L.D.; Bally, M.B. A comparison of liposomal formulations of doxorubicin with drug administered in free form: Changing toxicity profiles. Drug Saf. 2001, 24, 903–920. [Google Scholar] [CrossRef]
- Abraham, S.A.; Waterhouse, D.N.; Mayer, L.D.; Cullis, P.R.; Madden, T.D.; Bally, M.B. The liposomal formulation of doxorubicin. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2005; Volume 391, pp. 71–97. [Google Scholar]
- Charrois, G.J.R.; Allen, T.M. Drug release rate influences the pharmacokinetics, biodistribution, therapeutic activity, and toxicity of pegylated liposomal doxorubicin formulations in murine breast cancer. Biochim. Biophys. Acta (BBA) Biomembr. 2004, 1663, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Bally, M.B.; Nayar, R.; Masin, D.; Hope, M.J.; Cullis, P.R.; Mayer, L.D. Liposomes with entrapped doxorubicin exhibit extended blood residence times. Biochim. Biophys. Acta (BBA) Biomembr. 1990, 1023, 133–139. [Google Scholar] [CrossRef]
- Leonard, R.C.F.; Williams, S.; Tulpule, A.; Levine, A.M.; Oliveros, S. Improving the therapeutic index of anthracycline chemotherapy: Focus on liposomal doxorubicin (Myocet™). Breast 2009, 18, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.; Dos Santos, N.; Gallagher, R.; Chiu, G.N.C.; Shu, Y.; Li, W.M.; Johnstone, S.A.; Janoff, A.S.; Mayer, L.D.; Webb, M.S.; et al. Controlling the Physical Behavior and Biological Performance of Liposome Formulations Through Use of Surface Grafted Poly(ethylene Glycol). Biosci. Rep. 2002, 22, 225. [Google Scholar] [CrossRef]
- Parr, M.J.; Masin, D.; Cullis, P.R.; Bally, M.B. Accumulation of liposomal lipid and encapsulated doxorubicin in murine Lewis lung carcinoma: The lack of beneficial effects by coating liposomes with poly (ethylene glycol). J. Pharmacol. Exp. Ther. 1997, 280, 1319–1327. [Google Scholar] [PubMed]
- Mayer, L.D.; Bally, M.B.; Cullis, P.R. Uptake of adriamycin into large unilamellar vesicles in response to a pH gradient. Biochim. Biophys. Acta 1986, 857, 123–126. [Google Scholar] [CrossRef]
- Chowdhary, R.K.; Shariff, I.; Dolphin, D. Drug release characteristics of lipid based benzoporphyrin derivative. J. Pharm. Pharm. Sci. 2003, 6, 13–19. [Google Scholar]
- Schmidt-Erfurth, U.; Hasan, T. Mechanisms of Action of Photodynamic Therapy with Verteporfin for the Treatment of Age-Related Macular Degeneration. Surv. Ophthalmol. 2000, 45, 195–214. [Google Scholar] [CrossRef]
- Semple, S.C.; Leone, R.; Wang, J.; Leng, E.C.; Klimuk, S.K.; Eisenhardt, M.L.; Yuan, Z.-N.; Edwards, K.; Maurer, N.; Hope, M.J.; et al. Optimization and Characterization of a Sphingomyelin/Cholesterol Liposome Formulation of Vinorelbine with Promising Antitumor Activity. J. Pharm. Sci. 2005, 94, 1024–1038. [Google Scholar] [CrossRef]
- Webb, M.S.; Harasym, T.O.; Masin, D.; Bally, M.B.; Mayer, L.D. Sphingomyelin-cholesterol liposomes significantly enhance the pharmacokinetic and therapeutic properties of vincristine in murine and human tumour models. Br. J. Cancer 1995, 72, 896. [Google Scholar] [CrossRef]
- Dicko, A.; Kwak, S.; Frazier, A.A.; Mayer, L.D.; Liboiron, B.D. Biophysical characterization of a liposomal formulation of cytarabine and daunorubicin. Int. J. Pharm. 2010, 391, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Tardi, P.G.; Dos Santos, N.; Harasym, T.O.; Johnstone, S.A.; Zisman, N.; Tsang, A.W.; Bermudes, D.G.; Mayer, L.D. Drug ratio–dependent antitumor activity of irinotecan and cisplatin combinations in vitro and in vivo. Mol. Cancer Ther. 2009. [Google Scholar] [CrossRef] [PubMed]
- Tolcher, A.W.; Mayer, L.D. Improving combination cancer therapy: The CombiPlex® development platform. Future Oncol. 2018, 14, 1317–1332. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, S.; Harvie, P.; Shew, C.; Kadhim, S.; Harasym, T.; Tardi, P.; Harasym, N.; Bally, M.; Mayer, L.D. Synergistic antitumor activity observed for a fixed ratio liposome formulation of Cytarabine (Cyt):Daunorubicin (Daun) against preclinical leukemia models. Cancer Res. 2005, 65, 329. [Google Scholar]
- Hoy, S.M. Patisiran: First Global Approval. Drugs 2018, 78, 1625–1631. [Google Scholar] [CrossRef] [PubMed]
| Method Developed | Utility | References |
|---|---|---|
| Dehydration-rehydration method | Improve passive encapsulation efficiency | [48] |
| Modulation of lipid fluidity | Improve passive encapsulation efficiency | [49] |
| pH gradient loading | Remote loading | [50,51,52,53] |
| Use of ionophore to load small molecules | Improve remote loading efficiency | [54] |
| Use of ethanol to load small molecules | Improve remote loading efficiency | [46] |
| Microencapsulation method | Improve loading efficiency of water soluble and insoluble compound | [55] |
| Layersomes | Improve liposome stability and oral delivery | [56] |
| Hyaluronan coating of liposomes | Enable topical applications | [57] |
| Use of metal ion gradient | Stabilize water-soluble compounds | [58,59] |
| Metaplex technology | Enable development of poorly soluble metal-binding compounds | [60,61] |
| Use of cationic lipids | Deliver nucleic acids | [39,40] |
| Lipid nanoparticle (LNP) technology | Optimize delivery of nucleic acids for clinical use | [62,63,64] |
| Canadian Discoveries | References |
|---|---|
| Use of antibodies to mediate targeting with liposomes | [121,122] |
| Selective targeting of liposomes to the blood compartment | [123] |
| Use of GM1 ganglioside in liposomes, leading to the development of “PEGylation” | [122] |
| Role of PEG in preventing liposome aggregation | [124] |
| Development of low-cholesterol liposomes with lipids that prevent aggregation | [124] |
| Maintenance of the drug-drug ratio for two drugs encapsulated in one liposome | [77] |
| Approval Year | Trade Name | Active Agent | Lipid Composition | Approved Indication(s) | Current Ownership | References |
|---|---|---|---|---|---|---|
| 1993 | Epaxal (discontinued) | Inactivated hepatitis A virus (strain RGSB) | DOPC:DOPE (75:25 molar ratio) | Hepatitis A | Janssen Pharmaceuticals | [156,157] |
| 1995 | Doxil | Doxorubicin | HSPC:Cholesterol:PEG 2000-DSPE (56:39:5 molar ratio) | Ovarian, breast cancer, Kaposi’s sarcoma | Janssen Pharmaceuticals | [158,159,160] |
| 1995 | Abelcet | Amphotericin B | DMPC:DMPG (7:3 molar ratio) | Invasive severe fungal infections | Leadiant Biosciences | [161,162,163] |
| 1996 | DaunoXome | Daunorubicin | DSPC:Cholesterol (2:1 molar ratio) | AIDS-related Kaposi’s sarcoma | Galen Pharmaceuticals | [164,165] |
| 1996 | Amphotec | Amphotericin B | Cholesteryl sulphate:Amphotericin B (1:1 molar ratio) | Severe fungal infections | Kadmon Pharmaceuticals | [166] |
| 1997 | Ambisome | Amphotericin B | HSPC:DSPG:Cholesterol:Amphotericin B (2:0.8:1:0.4 molar ratio) | Presumed fungal infections | Astellas Pharma & Gilead Sciences | [167,168,169] |
| 1997 | Inflexal V (recalled) | Inactivated hemaglutinine of Influenza virus strains A and B | DOPC:DOPE (75:25 molar ratio) | Influenza | Crucell, Berna Biotech | [170] |
| 1999 | Depocyt (discontinued) | Cytarabine/Ara-C | Cholesterol:Triolein:DOPC:DPPG (11:1:7:1 molar ratio) | Neoplastic meningitis | Pacira Pharmaceuticals | [171,172] |
| 2000 | Myocet | Doxorubicin | EPC:Cholesterol (55:45 molar ratio) | Combination therapy with cyclophosphamide in metastatic breast cancer | Teva Pharmaceutical Industries | [173,174] |
| 2000 | Visudyne | Verteporfin | EPG:DMPC (3:5 molar ratio) | Choroidal neovascularisation | Cheplapharm Arzneimittel GmbH | [175,176] |
| 2004 | DepoDur (discontinued) | Morphine sulfate | Cholesterol:Triolein:DOPC:DPPG (11:1:7:1 molar ratio) | Pain management | Flynn Pharmaceuticals | [177] |
| 2009 | Mepact | Mifamurtide | DOPS:POPC (3:7 molar ratio) | High-grade, resectable, non-metastatic osteosarcoma | Takeda Pharmaceutical Ltd. | [178] |
| 2011 | Exparel | Bupivacaine | DEPC, DPPG, Cholesterol and Tricaprylin | Pain management | Pacira Pharmaceuticals, Inc. | [179,180] |
| 2012 | Marqibo | Vincristine | SM:Cholesterol (55:45 molar ratio) | Acute lymphoblastic leukemia | Spectrum Pharmaceuticals | [181,182] |
| 2015 | Onivyde | Irinotecan | DSPC:MPEG-2000:DSPE (3:2:0.015 molar ratio) | Combination therapy with fluorouracil and leucovorin in metastatic adenocarcinoma of the pancreas | Ipsen Biopharmaceuticals | [183] |
| 2017 | Vyxeos | Daunorubicin/Cytarabine | DSPC:DSPG:CHOL (7:2:1 molar ratio) | Therapy related acute myeloid leukemia (t-AML) or AML with myelodysplasia-related changes (AML-MRC) | Jazz Pharmaceuticals | [184,185] |
| 2018 | Onpattro | Patisiran | Dlin-MC3-DMA, PEG2000-C-DMG | Hereditary transthyretin-mediated amyloidosis | Alnylam Pharmaceuticals, Inc. | [185] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leung, A.W.Y.; Amador, C.; Wang, L.C.; Mody, U.V.; Bally, M.B. What Drives Innovation: The Canadian Touch on Liposomal Therapeutics. Pharmaceutics 2019, 11, 124. https://doi.org/10.3390/pharmaceutics11030124
Leung AWY, Amador C, Wang LC, Mody UV, Bally MB. What Drives Innovation: The Canadian Touch on Liposomal Therapeutics. Pharmaceutics. 2019; 11(3):124. https://doi.org/10.3390/pharmaceutics11030124
Chicago/Turabian StyleLeung, Ada W. Y., Carolyn Amador, Lin Chuan Wang, Urmi V. Mody, and Marcel B. Bally. 2019. "What Drives Innovation: The Canadian Touch on Liposomal Therapeutics" Pharmaceutics 11, no. 3: 124. https://doi.org/10.3390/pharmaceutics11030124
APA StyleLeung, A. W. Y., Amador, C., Wang, L. C., Mody, U. V., & Bally, M. B. (2019). What Drives Innovation: The Canadian Touch on Liposomal Therapeutics. Pharmaceutics, 11(3), 124. https://doi.org/10.3390/pharmaceutics11030124





