Silicon Nanotubes as Potential Therapeutic Platforms
Abstract
1. Introduction
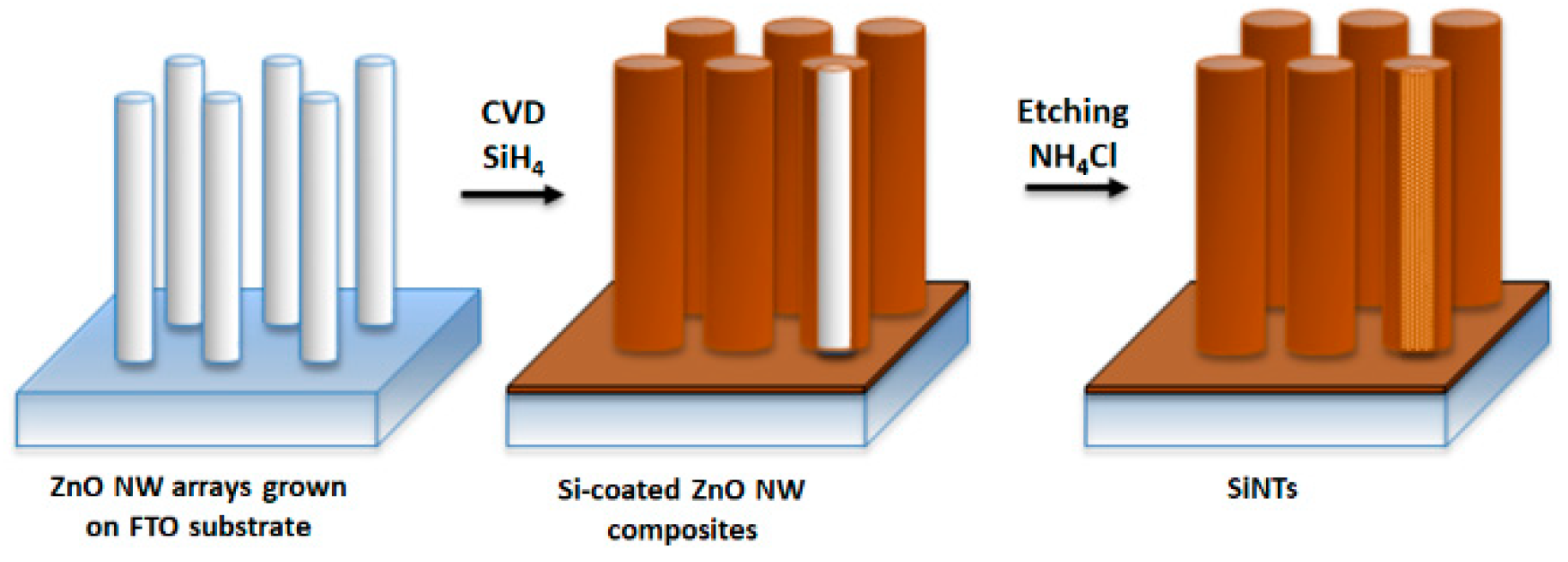
2. Fabrication of Silicon Nanotubes
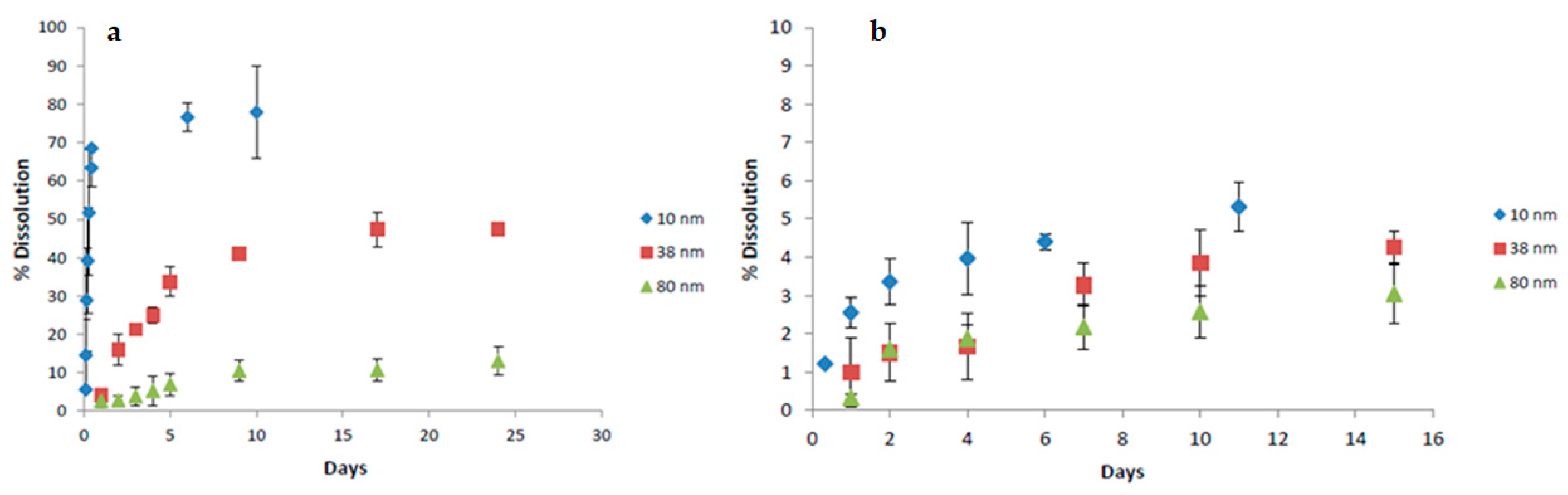
3. Dissolution Properties of Silicon Nanotubes
4. Nanotube Surface Modification Strategies Relevant to Therapeutic Applications
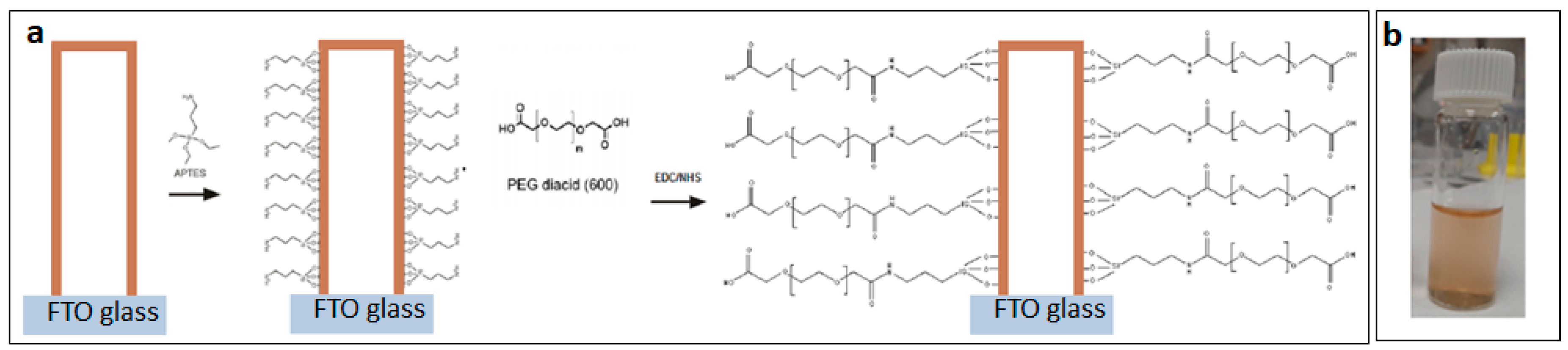
4.1. PEGylation of SiNTs
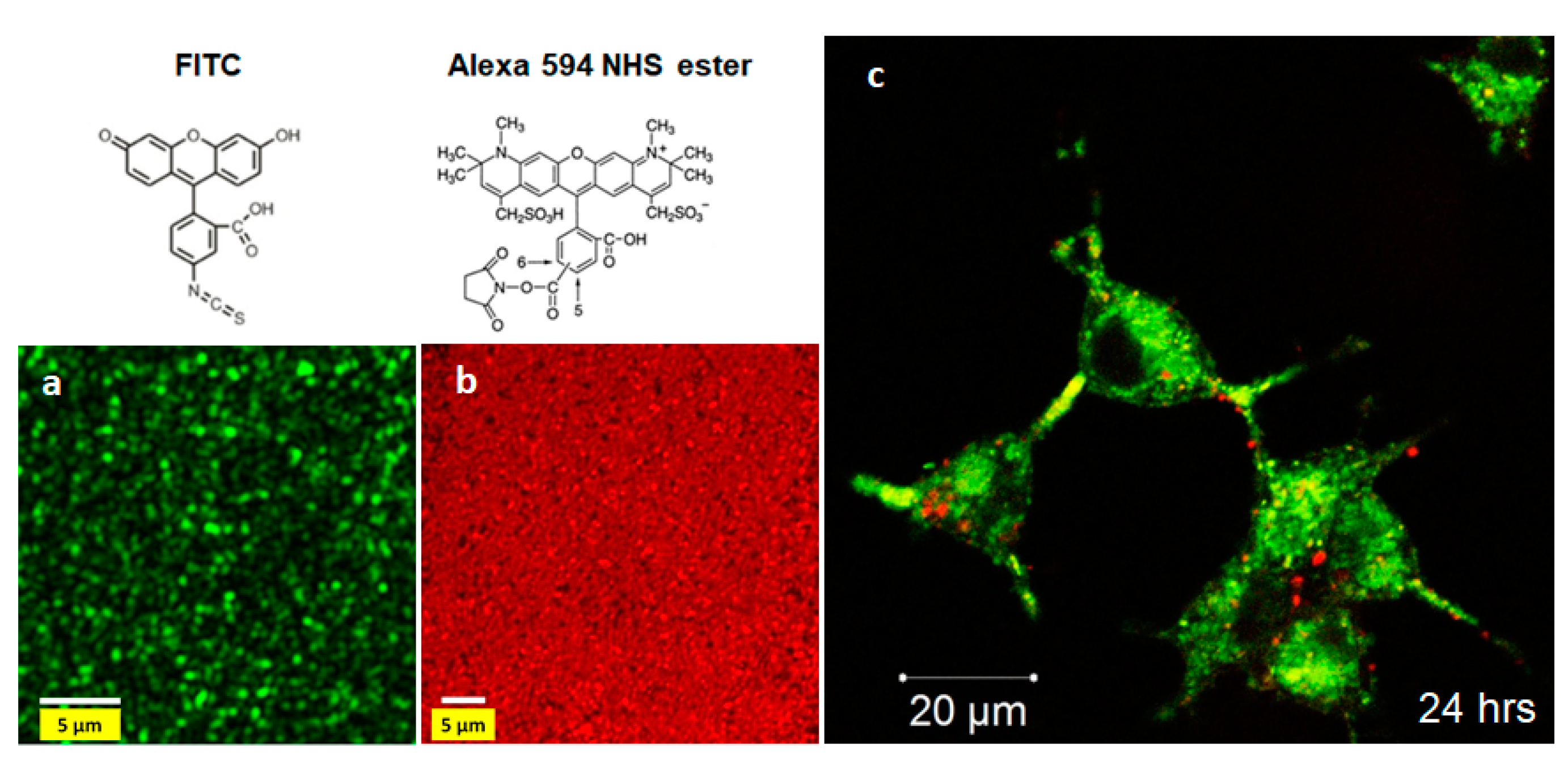
4.2. Fluorescently-Tagged SiNTs
4.3. DNA Immobilization on SiNT Surface through Electrostatic Interaction
4.4. Functionalized SiNTs as a Template for Formation of Platinum Nanocrystals
5. Loading of the Nanotube Interior
6. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Canham, L.T. Bioactive silicon structure fabrication through nanoetching techniques. Adv. Mater. 1995, 7, 1033–1037. [Google Scholar] [CrossRef]
- Anglin, E.J.; Cheng, L.; Freeman, W.R.; Sailor, M.J. Porous silicon in drug delivery devices and materials. Adv. Drug Deliv. Rev. 2008, 60, 1266–1277. [Google Scholar] [CrossRef] [PubMed]
- Low, S.P.; Voelcker, N.H. Biocompatibility of Porous Silicon. In Handbook of Porous Silicon; Canham, L., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 533–545. [Google Scholar] [CrossRef]
- Starkov, V.V.; Gosteva, E.A.; Sedlovets, D.M.; Kah, M.O. Silicon Structures with Variable Morphology of Pores Methods of Obtaining Physical and Optical Properties. J. Electrochem. Soc. 2018, 165, E534–E539. [Google Scholar] [CrossRef]
- Canham, L. Tunable Properties of Porous Silicon. In Handbook of Porous Silicon; Canham, L., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 283–290. [Google Scholar] [CrossRef]
- Li, W.; Liu, Z.; Fontana, F.; Ding, Y.; Liu, D.; Hirvonen, J.T.; Santos, H.A. Tailoring Porous Silicon for Biomedical Applications: From Drug Delivery to Cancer Immunotherapy. Adv. Mater. 2018, 30, 1703740. [Google Scholar] [CrossRef]
- Salonen, J.; Kaukonen, A.M.; Hirvonen, J.; Lehto, V.-P. Mesoporous silicon in drug delivery applications. J. Pharm. Sci. 2008, 97, 632–653. [Google Scholar] [CrossRef]
- Shtenberg, G.; Segal, E. Porous Silicon Optical Biosensors. In Handbook of Porous Silicon; Canham, L., Ed.; Springer International Publishing: Cham, Switzerland, 2014; pp. 857–868. [Google Scholar] [CrossRef]
- Fagan, S.B.; Baierle, R.J.; Mota, R.; da Silva, A.J.R.; Fazzio, A. Ab initio calculations for a hypothetical material: Silicon nanotubes. Phys. Rev. B 2000, 61, 9994–9996. [Google Scholar] [CrossRef]
- Coffer, J. Mesoporous nanotubes as biomaterials. Mesoporous Biomater. 2016, 2. [Google Scholar] [CrossRef]
- Huang, X.; Gonzalez-Rodriguez, R.; Rich, R.; Gryczynski, Z.; Coffer, J.L. Fabrication and size dependent properties of porous silicon nanotube arrays. Chem. Commun. 2013, 49, 5760–5762. [Google Scholar] [CrossRef][Green Version]
- Tesfaye, A.T.; Gonzalez, R.; Coffer, J.L.; Djenizian, T. Porous Silicon Nanotube Arrays as Anode Material for Li-Ion Batteries. ACS Appl. Mater. Interfaces 2015, 7, 20495–20498. [Google Scholar] [CrossRef]
- Gonzalez-Rodriguez, R.; Arad-Vosk, N.; Rozenfeld, N.; Sa’ar, A.; Coffer, J.L. Control of CH3NH3PbI3 Perovskite Nanostructure Formation through the Use of Silicon Nanotube Templates. Small 2016, 12, 4477–4480. [Google Scholar] [CrossRef]
- Arad-Vosk, N.; Rozenfeld, N.; Gonzalez-Rodriguez, R.; Coffer, J.L.; Sa’ar, A. Inhibition of a structural phase transition in one-dimensional organometal halide perovskite nanorods grown inside porous silicon nanotube templates. Phys. Rev. B 2017, 95, 085433. [Google Scholar] [CrossRef]
- Shabir, Q. Biodegradability of Porous Silicon. In Handbook of Porous Silicon; Canham, L., Ed.; Springer International Publishing: Cham, Switzerland, 2014; pp. 395–401. [Google Scholar] [CrossRef]
- Le, N.T.; Coffer, J.L. Silicon Nanomaterials Sourcebook; Sattler, K.D., Ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar] [CrossRef]
- Sailor, M.J. Porous Silicon in Practice: Preparation, Characterization and Applications; Wiley-VCH: Weinheim, Germany, 2012. [Google Scholar]
- Tian, Y. Silicon- and Germanium- Based Nanomaterial Cytocompatibility and Gene Delivery; Texas Christian University: Fort Worth, TX, USA, 2014. [Google Scholar]
- Rodriguez, R.G. Silicon Nanotubes and Selected Investigations in Energy and Biomaterial Applications; Texas Christian University: Fort Worth, TX, USA, 2017. [Google Scholar]
- Nijdam, A.J.; Ming-Cheng, M.; Geho, D.H.; Fedele, R.; Herrmann, P.; Killian, K.; Espina, V.; Petricoin, E.F.; Liotta, L.A.; Ferrari, M. Physicochemically modified silicon as a substrate for protein microarrays. Biomaterials 2007, 28, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Majoul, N.; Aouida, S.; Bessaïs, B. Progress of porous silicon APTES-functionalization by FTIR investigations. Appl. Surf. Sci. 2015, 331, 388–391. [Google Scholar] [CrossRef]
- Lee, H.S.; Kang, S.J.; Kim, D. A Mini Review: Recent Advances in Surface Modification of Porous Silicon. Materials 2018, 11, 2557. [Google Scholar] [CrossRef] [PubMed]
- De Stefano, L.; Oliviero, G.; Amato, J.; Borbone, N.; Piccialli, G.; Mayol, L.; Rendina, I.; Terracciano, M.; Rea, I. Aminosilane functionalizations of mesoporous oxidized silicon for oligonucleotide synthesis and detection. J. R. Soc. Interface 2013, 10, 20130160. [Google Scholar] [CrossRef]
- Cardellini, A.; Alberghini, M.; Govind Rajan, A.; Misra, R.P.; Blankschtein, D.; Asinari, P. Multi-scale approach for modeling stability, aggregation, and network formation of nanoparticles suspended in aqueous solutions. Nanoscale 2019, 11, 3979–3992. [Google Scholar] [CrossRef]
- Lee, S.B. Nanotoxicology: Toxicity and biological effects of nanoparticles for new evaluation standards. Nanomedicine 2011, 6, 759–761. [Google Scholar] [CrossRef]
- Lazzari, S.; Moscatelli, D.; Codari, F.; Salmona, M.; Morbidelli, M.; Diomede, L. Colloidal stability of polymeric nanoparticles in biological fluids. J. Nanoparticle Res. 2012, 14, 920. [Google Scholar] [CrossRef]
- Izak-Nau, E.; Huk, A.; Reidy, B.; Uggerud, H.; Vadset, M.; Eiden, S.; Voetz, M.; Himly, M.; Duschl, A.; Dusinska, M.; et al. Impact of storage conditions and storage time on silver nanoparticles’ physicochemical properties and implications for their biological effects. Rsc. Adv. 2015, 5, 84172–84185. [Google Scholar] [CrossRef]
- Guerrini, L.; Alvarez-Puebla, A.R.; Pazos-Perez, N. Surface Modifications of Nanoparticles for Stability in Biological Fluids. Materials 2018, 11, 1154. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef] [PubMed]
- Daley, K.R.; Kubarych, K.J. An “Iceberg” Coating Preserves Bulk Hydration Dynamics in Aqueous PEG Solutions. J. Phys. Chem. B 2017, 121, 10574–10582. [Google Scholar] [CrossRef] [PubMed]
- Gabizon, A.; Catane, R.; Uziely, B.; Kaufman, B.; Safra, T.; Cohen, R.; Martin, F.; Huang, A.; Barenholz, Y. Prolonged Circulation Time and Enhanced Accumulation in Malignant Exudates of Doxorubicin Encapsulated in Polyethylene-glycol Coated Liposomes. Cancer Res. 1994, 54, 987. [Google Scholar] [PubMed]
- Jokerst, J.V.; Lobovkina, T.; Zare, R.N.; Gambhir, S.S. Nanoparticle PEGylation for imaging and therapy. Nanomedicine 2011, 6, 715–728. [Google Scholar] [CrossRef]
- Gonzalez-Rodriguez, R.; Granitzer, P.; Rumpf, K.; Coffer, J.L. New MRI contrast agents based on silicon nanotubes loaded with superparamagnetic iron oxide nanoparticles. R. Soc. Open Sci. 2018, 5, 180697. [Google Scholar] [CrossRef]
- Specht, E.A.; Braselmann, E.; Palmer, A.E. A Critical and Comparative Review of Fluorescent Tools for Live-Cell Imaging. Annu. Rev. Physiol. 2017, 79, 93–117. [Google Scholar] [CrossRef]
- Shang, L.; Nienhaus, G.U. Small fluorescent nanoparticles at the nano–bio interface. Mater. Today 2013, 16, 58–66. [Google Scholar] [CrossRef]
- Natte, K.; Behnke, T.; Orts-Gil, G.; Würth, C.; Friedrich, J.F.; Österle, W.; Resch-Genger, U. Synthesis and characterisation of highly fluorescent core–shell nanoparticles based on Alexa dyes. J. Nanoparticle Res. 2012, 14, 680. [Google Scholar] [CrossRef]
- Wang, W.; Nallathamby, P.D.; Foster, C.M.; Morrell-Falvey, J.L.; Mortensen, N.P.; Doktycz, M.J.; Gu, B.; Retterer, S.T. Volume labeling with Alexa Fluor dyes and surface functionalization of highly sensitive fluorescent silica (SiO2) nanoparticles. Nanoscale 2013, 5, 10369–10375. [Google Scholar] [CrossRef]
- Hermanson, G.T. Chapter 10—Fluorescent Probes. In Bioconjugate Techniques (Third Edition); Hermanson, G.T., Ed.; Academic Press: Boston, MA, USA, 2013; pp. 395–463. [Google Scholar] [CrossRef]
- Whitson, K.B.; Beechem, J.M.; Beth, A.H.; Staros, J.V. Preparation and characterization of Alexa Fluor 594-labeled epidermal growth factor for fluorescence resonance energy transfer studies: Application to the epidermal growth factor receptor. Anal. Biochem. 2004, 324, 227–236. [Google Scholar] [CrossRef]
- Lostalé-Seijo, I.; Montenegro, J. Synthetic materials at the forefront of gene delivery. Nat. Rev. Chem. 2018, 2, 258–277. [Google Scholar] [CrossRef]
- Wu, P.; Chen, H.; Jin, R.; Weng, T.; Ho, J.K.; You, C.; Zhang, L.; Wang, X.; Han, C. Non-viral gene delivery systems for tissue repair and regeneration. J. Transl. Med. 2018, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Matkar, P.N.; Leong-Poi, H.; Singh, K.K. Cardiac gene therapy: Are we there yet? Gene Ther. 2016, 23, 635. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Tai, P.W.L.; Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 2019, 18, 358–378. [Google Scholar] [CrossRef]
- Nayak, S.; Herzog, R.W. Progress and prospects: Immune responses to viral vectors. Gene Ther. 2009, 17, 295. [Google Scholar] [CrossRef]
- Van Bruggen, C.; Hexum, J.K.; Tan, Z.; Dalal, R.J.; Reineke, T.M. Nonviral Gene Delivery with Cationic Glycopolymers. Acc. Chem. Res. 2019, 52, 1347–1358. [Google Scholar] [CrossRef]
- Yin, H.; Kanasty, R.L.; Eltoukhy, A.A.; Vegas, A.J.; Dorkin, J.R.; Anderson, D.G. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 2014, 15, 541–555. [Google Scholar] [CrossRef]
- Loh, X.J.; Lee, T.-C.; Dou, Q.; Deen, G.R. Utilising inorganic nanocarriers for gene delivery. Biomater. Sci. 2016, 4, 70–86. [Google Scholar] [CrossRef]
- Riley, K.M.; Vermerris, W. Recent Advances in Nanomaterials for Gene Delivery—A Review. Nanomaterials 2017, 7, 94. [Google Scholar] [CrossRef]
- Pick, C.; Argento, C.; Drazer, G.; Frechette, J. Micropatterned Charge Heterogeneities via Vapor Deposition of Aminosilanes. Langmuir 2015, 31, 10725–10733. [Google Scholar] [CrossRef]
- Ma, P.a.; Xiao, H.; Li, C.; Dai, Y.; Cheng, Z.; Hou, Z.; Lin, J. Inorganic nanocarriers for platinum drug delivery. Mater. Today 2015, 18, 554–564. [Google Scholar] [CrossRef]
- Tsang, R.Y.; Al-Fayea, T.; Au, H.-J. Cisplatin Overdose. Drug Saf. 2009, 32, 1109–1122. [Google Scholar] [CrossRef] [PubMed]
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573. [Google Scholar] [CrossRef] [PubMed]
- Khiati, S.; Luvino, D.; Oumzil, K.; Chauffert, B.; Camplo, M.; Barthélémy, P. Nucleoside–Lipid-Based Nanoparticles for Cisplatin Delivery. ACS Nano 2011, 5, 8649–8655. [Google Scholar] [CrossRef] [PubMed]
- Mochida, Y.; Cabral, H.; Miura, Y.; Albertini, F.; Fukushima, S.; Osada, K.; Nishiyama, N.; Kataoka, K. Bundled Assembly of Helical Nanostructures in Polymeric Micelles Loaded with Platinum Drugs Enhancing Therapeutic Efficiency against Pancreatic Tumor. ACS Nano 2014, 8, 6724–6738. [Google Scholar] [CrossRef]
- Mandriota, G.; Di Corato, R.; Benedetti, M.; De Castro, F.; Fanizzi, F.P.; Rinaldi, R. Design and Application of Cisplatin-Loaded Magnetic Nanoparticle Clusters for Smart Chemotherapy. ACS Appl. Mater. Interfaces 2019, 11, 1864–1875. [Google Scholar] [CrossRef]
- Reishus, J.W.; Martin, D.S. cis-Dichlorodiammineplatinum(II). Acid Hydrolysis and Isotopic Exchange of the Chloride Ligands1. J. Am. Chem. Soc. 1961, 83, 2457–2462. [Google Scholar] [CrossRef]
- Dhara, S. A rapid method for the synthesis of cis- [Pt (NH3) 2Cl2]. Indian J. Chem. 1970, 8, 193–194. [Google Scholar]
- Le, N.T.; Coffer, J.L. Formation of Platinum Nanocrystals on Porous Silicon Nanotubes. In Proceedings of the Porous Semiconductors—Science and Technology Conference 2018, La Grande Motte, France, 11–16 March 2018. [Google Scholar]
- Chien, C.-T.; Yan, J.-Y.; Chiu, W.-C.; Wu, T.-H.; Liu, C.-Y.; Lin, S.-Y. Caged Pt Nanoclusters Exhibiting Corrodibility to Exert Tumor-Inside Activation for Anticancer Chemotherapeutics. Adv. Mater. 2013, 25, 5067–5073. [Google Scholar] [CrossRef]
- Xia, H.; Li, F.; Hu, X.; Park, W.; Wang, S.; Jang, Y.; Du, Y.; Baik, S.; Cho, S.; Kang, T.; et al. pH-Sensitive Pt Nanocluster Assembly Overcomes Cisplatin Resistance and Heterogeneous Stemness of Hepatocellular Carcinoma. ACS Cent. Sci. 2016, 2, 802–811. [Google Scholar] [CrossRef]
- Shoshan, M.S.; Vonderach, T.; Hattendorf, B.; Wennemers, H. Peptide-Coated Platinum Nanoparticles with Selective Toxicity against Liver Cancer Cells. Angew. Chem. Int. Ed. 2019, 58, 4901–4905. [Google Scholar] [CrossRef] [PubMed]
- Granitzer, P.; Rumpf, K.; Gonzalez, R.; Coffer, J.; Reissner, M. Magnetic properties of superparamagnetic nanoparticles loaded into silicon nanotubes. Nanoscale Res. Lett. 2014, 9, 413. [Google Scholar] [CrossRef] [PubMed]
- Granitzer, P.; Rumpf, K.; Gonzalez-Rodriguez, R.; Coffer, J.L.; Reissner, M. The effect of nanocrystalline silicon host on magnetic properties of encapsulated iron oxide nanoparticles. Nanoscale 2015, 7, 20220–20226. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rodriguez, R. 10 nm Magnetite Nanoparticles Inside Si NTs. Available online: https://www.youtube.com/watch?v=1xDFAQSS2T0 (accessed on 1 September 2019).





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, N.T.; Tian, Y.; Gonzalez-Rodriguez, R.; Coffer, J.L. Silicon Nanotubes as Potential Therapeutic Platforms. Pharmaceutics 2019, 11, 571. https://doi.org/10.3390/pharmaceutics11110571
Le NT, Tian Y, Gonzalez-Rodriguez R, Coffer JL. Silicon Nanotubes as Potential Therapeutic Platforms. Pharmaceutics. 2019; 11(11):571. https://doi.org/10.3390/pharmaceutics11110571
Chicago/Turabian StyleLe, Nguyen T., Yuan Tian, Roberto Gonzalez-Rodriguez, and Jeffery L. Coffer. 2019. "Silicon Nanotubes as Potential Therapeutic Platforms" Pharmaceutics 11, no. 11: 571. https://doi.org/10.3390/pharmaceutics11110571
APA StyleLe, N. T., Tian, Y., Gonzalez-Rodriguez, R., & Coffer, J. L. (2019). Silicon Nanotubes as Potential Therapeutic Platforms. Pharmaceutics, 11(11), 571. https://doi.org/10.3390/pharmaceutics11110571




