Selected Medicines Used in Iontophoresis
Abstract
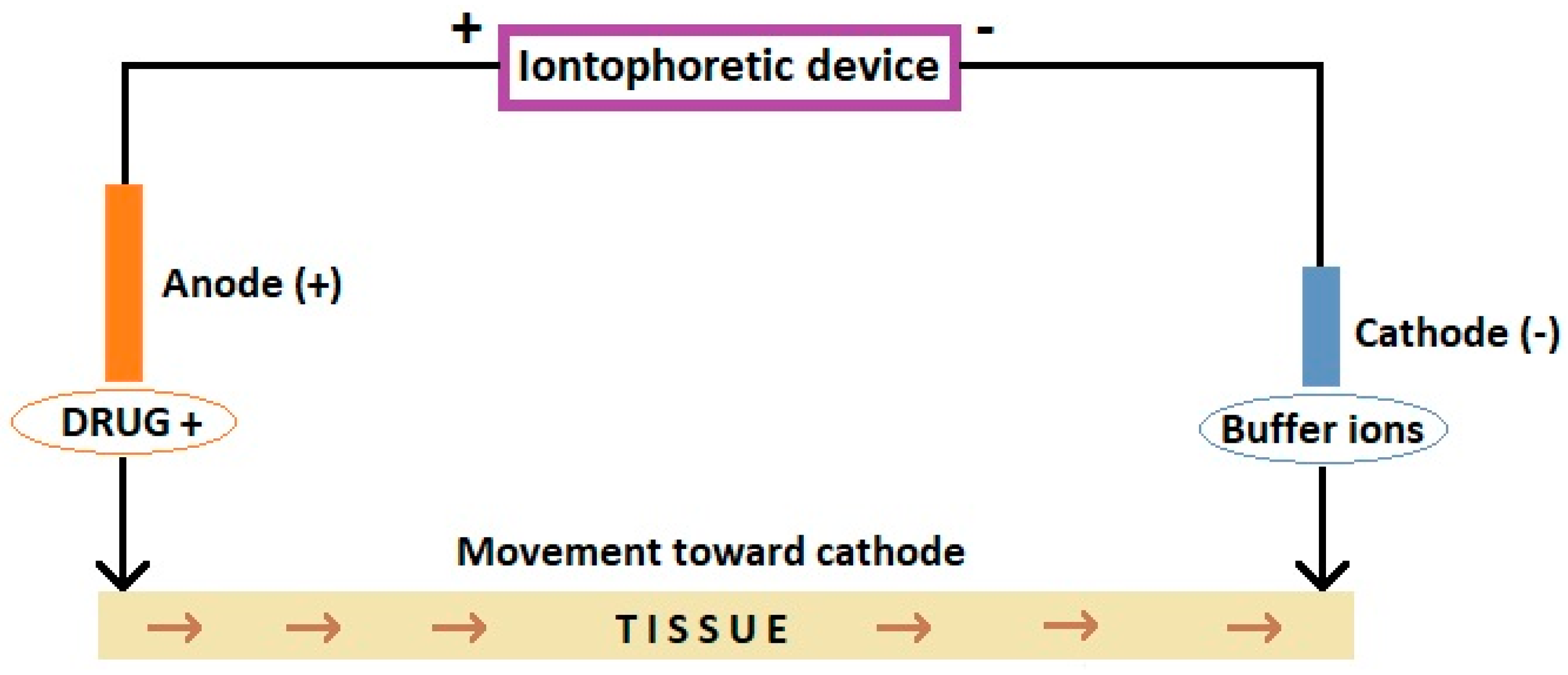
1. Introduction
- Superficial and deep sensory disorders;
- Should not be used in pregnancy;
- Should not be used during lactation;
2. Local Anesthetics
3. Opioids
4. Steroids
5. Non-Steroidal Anti-Inflammatory Drugs
6. Antibacterial Drugs
7. Antifungal Drugs
8. Antiviral Drugs
9. Anti-Cancer Drugs
10. Fluorides
11. Vitamins
12. Summary
Conflicts of Interest
References
- Ebisawa, T.; Nakajima, A.; Haida, H.; Wakita, R.; Ando, S.; Yoshioka, T.; Ikoma, T.; Tanaka, J.; Fukayama, H. Evaluation of calcium alginate gel as electrode material for alternating current iontophoresis of lidocaine using excised rat skin. J. Med. Dent. Sci. 2014, 61, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Dhote, V.; Bhatnagar, P.; Mishra, P.K.; Mahajan, S.C.; Mishra, D.K. Iontophoresis: A potential emergence of a transdermal drug delivery system. Sci. Pharm. 2012, 80, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, M. Zastosowanie ketoprofenu w jonoforezie. Ketoprofen in iontophoresis. Pediatr. Med. Rodz. 2011, 7, 124–128. [Google Scholar]
- Straburzyńska-Lupa, A.; Straburzyński, G. Fizjoterapia; PZWL: Warszawa, Poland, 2004; pp. 481–492. [Google Scholar]
- Zhu, H.; Li, S.K.; Peck, K.D.; Miller, D.J.; Higuchi, W.I. Improvement on conventional constant current DC iontophoresis: Study using constant conductance AC iontophoresis. J. Control. Release 2002, 82, 249–261. [Google Scholar] [CrossRef]
- Rai, R.; Srinivas, C.R. Iontophoresis in dermatology. Indian J. Dermatol. Venereol. Leprol. 2005, 71, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Wanasathop, A.; Li, S.K. Iontophoretic drug delivery in the oral cavity. Pharmaceutics 2018, 10, 121. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Sugiyama, T.; Ikoma, T.; Shimazu, H.; Wakita, R.; Fukayama, H. Drug delivery and transmission of lidocaine using iontophoresis in combination with direct and alternating currents. J. Med. Dent. Sci. 2016, 63, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Chen, Y.S.; Rupenthal, I.D. Overcoming ocular drug delivery barriers through the use of physical forces. Adv. Drug Deliv. Rev. 2018, 126, 96–112. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.D.; Yeh, J.J.; DeSimone, J.M. Use of iontophoresis for the treatment of cancer. J. Control. Release 2018, 284, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Zuo, J.; Du, L.; Li, M.; Liu, B.; Zhu, W.; Jin, Y. Transdermal enhancement effect and mechanism of iontophoresis for non-steroidal anti-inflammatory drugs. Int. J. Pharm. 2014, 466, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Ita, K. Transdermal delivery of vaccines–Recent progress and critical issues. Biomed. Pharmacother. 2016, 83, 1080–1088. [Google Scholar] [CrossRef] [PubMed]
- Ledger, P.W. Skin biological issues in electrically enhanced transdermal delivery. Adv. Drug Deliv. Rev. 1992, 9, 289–307. [Google Scholar] [CrossRef]
- Ita, K. Transdermal iontophoretic drug delivery: Advances and challenges. J. Drug Target. 2016, 24, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Spodaryk, K.; Bromboszcz, J. Fizykoterapia—Potrzeba badań naukowych. Rehabil. Med. 2004, 8, 8–14. [Google Scholar]
- Samborski, W.; Ponikowska, I. Choroby reumatyczne—Standardy leczenia w medycynie uzdrowiskowej. Balneol. Pol. 2005, 1–2, 14–23. [Google Scholar]
- Kralinger, M.T.; Voigt, M.; Kieselbach, G.; Hamasaki, D.; Hayden, B.; Parel, J.M. Ocular delivery of acetylsalicylic acid by repetitive coulomb-controlled iontophoresis. Ophthalmic Res. 2002, 35, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.E.; Kwon, H.K.; Kim, B.I. Application of fluoride iontophoresis to improve remineralization. J. Oral Rehabil. 2009, 36, 770–775. [Google Scholar] [CrossRef] [PubMed]
- Girenes, G.; Ulusu, T. An in vitro evaluation of the efficacy of a novel iontophoresis fluoride tray on remineralization. J. Clin. Exp. Dent. 2014, 6, e327–e334. [Google Scholar] [CrossRef] [PubMed]
- Siddoju, S.; Sachdeva, V.; Friden, P.M.; Yu, Y.Y.; Banga, A.K. Acyclovir skin depot characterization following in vivo iontophoretic delivery. Skin Res. Technol. 2011, 17, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Kalia, Y.N. Short-duration ocular iontophoresis of ionizable aciclovir prodrugs: A new approach to treat herpes simplex infections in the anterior and posterior segments of the eye. Int. J. Pharm. 2018, 536, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, D.L.; Szlek, M.A.; Kolb, K.; Lloyd, L.B.; Parkinson, T.M. In vivo transscleral iontophoresis of amikacin to rabbit eyes. J. Ocul. Pharmacol. Ther. 2002, 18, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Nicoli, S.; Santi, P. Transdermal delivery of aminoglycosides: Amikacin transport and iontophoretic non-invasive monitoring. J. Control. Release 2006, 111, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, K.; Watanabe, D.; Akita, Y.; Akimoto, M.; Tamada, Y.; Matsumoto, Y. Photodynamic therapy using direct-current pulsed iontophoresis for 5-aminolevulinic acid application. Photodermatol. Photoimmunol. Photomed. 2009, 25, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Mannem, V.; Nanjarapalle, C.; Stagni, G. Iontophoresis of amoxicillin and cefuroxime: Rapid therapeutic concentrations in skin. Drug Dev. Ind. Pharm. 2014, 40, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, N.; Minaiyan, M.; Heshmatipour, M.; Musavinasab, R. Transdermal iontophoretic delivery of celecoxib from gel formulation. Res. Pharm. Sci. 2015, 10, 419–428. [Google Scholar] [PubMed]
- Hobden, J.A.; Reidy, J.J.; O’Callaghan, R.J.; Insler, M.S.; Hill, J.M. Ciprofloxacin iontophoresis for aminoglycoside-resistant pseudomonal keratitis. Investig. Ophthalmol. Vis. Sci. 1990, 31, 1940–1944. [Google Scholar]
- Vaka, S.R.; Sammeta, S.M.; Day, L.B.; Murthy, S.N. Transcorneal iontophoresis for delivery of ciprofloxacin hydrochloride. Curr. Eye Res. 2008, 33, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Gergova, R.T.; Gueorgieva, T.; Dencheva-Garova, M.S.; Krasteva-Panova, A.Z.; Kalchinov, V.; Mitov, I.; Kamenoff, J. Antimicrobial activity of different disinfection methods against biofilms in root canals. J. Investig. Clin. Dent. 2016, 7, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Jose, A.; Labala, S.; Venuganti, V.V. Co-delivery of curcumin and STAT3 siRNA using deformable cationic liposomes to treat skin cancer. J. Drug Target. 2017, 25, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Smutok, M.A.; Mayo, M.F.; Gabaree, C.L.; Ferslew, K.E.; Panus, P.C. Failure to detect dexamethasone phosphate in the local venous blood postcathodic lontophoresis in humans. J. Orthop. Sports Phys. Ther. 2002, 32, 461–468. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mina, R.; Melson, P.; Powell, S.; Rao, M.; Hinze, C.; Passo, M.; Graham, T.B.; Brunner, H.I. Effectiveness of dexamethasone iontophoresis for temporomandibular joint involvement in juvenile idiopathic arthritis. Arthritis Care Res. 2011, 63, 1511–1516. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.G.; Dias, K.; Silva, S.A.; de Rezende, L.C.; Rocha, E.M.; Emery, F.S.; Lopez, R.F. Transcorneal iontophoresis of dendrimers: PAMAM corneal penetration and dexamethasone delivery. J. Control. Release 2015, 200, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Baig, A.; White, D.J.; Li, S.K. Characterization of cornified oral mucosa for iontophoretically enhanced delivery of chlorhexidine. Eur. J. Pharm. Biopharm. 2016, 99, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Crevenna, R.; Burian, A.; Oesterreicher, Z.; Lackner, E.; Jäger, W.; Rezcicek, G.; Keilani, M.; Zeitlinger, M. Iontophoresis driven concentrations of topically administered diclofenac in skeletal muscle and blood of healthy subjects. Eur. J. Clin. Pharmacol. 2015, 71, 1359–1364. [Google Scholar] [CrossRef] [PubMed]
- Malinovskaja-Gomez, K.; Espuelas, S.; Garrido, M.J.; Hirvonen, J.; Laaksonen, T. Comparison of liposomal drug formulations for transdermal iontophoretic drug delivery. Eur. J. Pharm. Sci. 2017, 106, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Viscusi, E.R.; Reynolds, L.; Chung, F.; Atkinson, L.E.; Khanna, S. Patient-controlled transdermal fentanyl hydrochloride vs intravenous morphine pump for postoperative pain: A randomized controlled trial. JAMA 2004, 291, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Gratieri, T.; Kalia, Y.N. Targeted local simultaneous iontophoresis of chemotherapeutics for topical therapy of head and neck cancers. Int. J. Pharm. 2014, 460, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Petrilli, R.; Eloy, J.O.; Saggioro, F.P.; Chesca, D.L.; de Souza, M.C.; Dias, M.V.; da Silva, L.L.; Lee, R.J.; Lopez, R.F. Skin cancer treatment effectiveness is improved by iontophoresis of EGFR-targeted liposomes containing 5-FU compared with subcutaneous injection. J. Control. Release 2018, 283, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Eljarrat-Binstock, E.; Raiskup, F.; Frucht-Pery, J.; Domb, A.J. Hydrogel probe for iontophoresis drug delivery to the eye. J. Biomater. Sci. Polym. Ed. 2004, 15, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Eljarrat-Binstock, E.; Raiskup, F.; Stepensky, D.; Domb, A.J.; Frucht-Pery, J. Delivery of gentamicin to the rabbit eye by drug-loaded hydrogel iontophoresis. Investig. Ophthalmol. Vis. Sci. 2004, 45, 2543–2548. [Google Scholar] [CrossRef] [PubMed]
- Frucht-Pery, J.; Mechoulam, H.; Siganos, C.S.; Ever-Hadani, P.; Shapiro, M.; Domb, A. Iontophoresis-gentamicin delivery into the rabbit cornea using a hydrogel delivery probe. Exp. Eye Res. 2004, 78, 745–749. [Google Scholar] [CrossRef]
- Day, R.E.; Megson, S.; Wood, D. Iontophoresis as a means of delivering antibiotics into allograft bone. J. Bone Jt. Surg. Br. 2005, 87, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Frucht-Pery, J.; Raiskup, F.; Mechoulam, H.; Shapiro, M.; Eljarrat-Binstock, E.; Domb, A. Iontophoretic treatment of experimental Pseudomonas keratitis in rabbit eyes using gentamicin-loaded hydrogels. Cornea 2006, 25, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, M.; Hama, S.; Ikeda, Y.; Nagasaki, Y.; Kogure, K. Anti-cancer vaccination by transdermal delivery of antigen peptide-loaded nanogels via iontophoresis. Int. J. Pharm. 2015, 483, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Grossman, R.; Lee, D.A. Transscleral and transcorneal iontophoresis of ketoconazole in the rabbit eye. Ophthalmology 1989, 96, 724–729. [Google Scholar] [CrossRef]
- Lobo, S.; Yan, G. Improving the direct penetration into tissues underneath the skin with iontophoresis delivery of a ketoprofen cationic prodrug. Int. J. Pharm. 2018, 535, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Wakita, R.; Oono, Y.; Oogami, S.; Hayashi, S.; Umino, M. The relation between epinephrine concentration and the anesthetic effect of lidocaine iontophoresis. Pain Pract. 2009, 9, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Silva, S.M.; Damaj, B.B.; Martin, R.; Michniak-Kohn, B.B. Transdermal and transbuccal drug delivery systems: Enhancement using iontophoretic and chemical approaches. Int. J. Pharm. 2011, 421, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Simon, L.; Hu, L.; Michniak-Kohn, B. Effects of iontophoresis and chemical enhancers on the transport of lidocaine and nicotine across the oral mucosa. Pharm. Res. 2012, 29, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Suda, H. Facilitatory effect of AC-iontophoresis of lidocaine hydrochloride on the permeability of human enamel and dentine in extracted teeth. Arch. Oral Biol. 2013, 58, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Cubayachi, C.; Couto, R.O.; de Gaitani, C.M.; Pedrazzi, V.; Freitas, O.; Lopez, R.F. Needle-free buccal anesthesia using iontophoresis and amino amide salts combined in a mucoadhesive formulation. Colloids Surf. B Biointerfaces 2015, 136, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Telo, I.; Tratta, E.; Guasconi, B.; Nicoli, S.; Pescina, S.; Govoni, P.; Santi, P.; Padula, C. In-vitro characterization of buccal iontophoresis: The case of sumatriptan succinate. Int. J. Pharm. 2016, 506, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Sekijima, H.; Ehara, J.; Hanabata, Y.; Suzuki, T.; Kimura, S.; Lee, V.H.; Morimoto, Y.; Ueda, H. Characterization of ocular iontophoretic drug transport of ionic and nonionic compounds in isolated rabbit cornea and conjunctiva. Biol. Pharm. Bull. 2016, 39, 959–968. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Draper, D.O.; Coglianese, M.; Castel, C. Absorption of iontophoresis-driven 2% lidocaine with epinephrine in the tissues at 5 mm below the surface of the skin. J. Athl. Train. 2011, 46, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Smitayothin, T.L.; Vongsavan, K.; Rirattanapong, P.; Kraivaphan, P.; Vongsavan, N.; Matthews, B. The iontophoresis of lignocaine with epinephrine into carious dentine for pain control during cavity preparation in human molars. Arch. Oral Biol. 2015, 60, 1104–1108. [Google Scholar] [CrossRef] [PubMed]
- Thongkukiatkun, W.; Vongsavan, K.; Kraivaphan, P.; Rirattanapong, P.; Vongsavan, N.; Matthews, B. Effects of the iontophoresis of lignocaine with epinephrine into exposed dentine on the sensitivity of the dentine in man. Arch. Oral Biol. 2015, 60, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Puapichartdumrong, P.; Ikeda, H.; Suda, H. Facilitation of iontophoretic drug delivery through intact and caries-affected dentine. Int. Endod. J. 2003, 36, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Mencucci, R.; Ambrosini, S.; Paladini, I.; Favuzza, E.; Boccalini, C.; Raugei, G.; Vannelli, G.B.; Marini, M. Early effects of corneal collagen cross-linking by iontophoresis in ex vivo human corneas. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Rong, S.; Wang, C.; Han, B.; Feng, P.; Lan, W.; Gao, Z.; Li, X.; Chen, W. Iontophoresis-assisted accelerated riboflavin/ultraviolet A scleral cross-linking: A potential treatment for pathologic myopia. Exp. Eye Res. 2017, 162, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.Z.; Pang, X.; Fan, Z.J.; Li, N.; Li, G.; Peng, X.J. Iontophoresis-assisted corneal crosslinking using 0.1% riboflavin for progressive keratoconus. Int. J. Ophthalmol. 2017, 10, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, M.; Giannini, D.; Lombardo, G.; Serrao, S. Randomized controlled trial comparing transepithelial corneal cross-linking using iontophoresis with the Dresden Protocol in progressive keratoconus. Ophthalmology 2017, 124, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Lemos, C.N.; Cubayachi, C.; Dias, K.; Mendonça, J.N.; Lopes, N.P.; Furtado, N.A.J.C.; Lopez, R.F. Iontophoresis-stimulated silk fibroin films as a peptide delivery system for wound healing. Eur. J. Pharm. Biopharm. 2018, 128, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Shirwaiker, R.A.; Wysk, R.A.; Kariyawasam, S.; Voigt, R.C.; Carrion, H.; Nembhard, H.B. Interdigitated silver-polymer-based antibacterial surface system activated by oligodynamic iontophoresis—An empirical characterization study. Biomed. Microdevices 2014, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.J.; Weil, T.M. Reduction of enamel acid solubility with electrophoretic fluoride applications. J. Dent. Res. 1966, 45, 1563. [Google Scholar] [CrossRef] [PubMed]
- Barbakow, F.H.; van der Merwe, E.H.M.; Scanes, S.G.; Retief, D.H.; Fatti, L.P. An in vivo evaluation of an iontophoretic technique for increasing the surface fluoride content in enamel. J. Dent. Assoc. S. Afr. 1973, 28, 624–629. [Google Scholar]
- Singal, P.; Gupta, R.; Pandit, N. 2% sodium fluoride-iontophoresis compared to a commercially available desensitizing agent. J. Periodontol. 2005, 76, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.J.; Meng, Q.F.; Chen, Y.M.; Smales, R.J.; Yip, K.H. Effect of fluoride iontophoresis on the microtensile bond strength between dentin and two adhesive systems. J. Dent. 2008, 36, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.E.; Baek, H.J.; Choi, Y.H.; Jeong, S.H.; Park, Y.D.; Song, K.B. Comparison of remineralization effect of three topical fluoride regimens on enamel initial carious lesions. J. Dent. 2010, 38, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Brahmbhatt, N.; Bhavsar, N.; Sahayata, V.; Acharya, A.; Kshatriya, P. A double blind controlled trial comparing three treatment modalities for dentin hypersensitivity. Med. Oral Patol. Oral Cir. Bucal 2012, 17, e483–e490. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Baig, A.; Li, S.K. Passive and iontophoretic transport of fluorides across enamel in vitro. J. Pharm. Sci. 2014, 103, 1692–1700. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.E.; Kim, B.I. Can the application of fluoride iontophoresis improve remineralisation of early caries lesions? Oral Health Prev. Dent. 2016, 14, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.R.; Varma, S.; Suragimath, G.; Abbayya, K.; Zope, S.A.; Kale, V. Comparative evaluation of efficacy of iontophoresis with 0.33% sodium fluoride gel and diode laser alone on occlusion of dentinal tubules. J. Clin. Diagn. Res. 2017, 11, ZC123–ZC126. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Saito, A.; Watanabe, K.; Saeki, K.; Nakashima, H.; Maki, K. Preventive effects of iontophoresis on bovine enamel decalcification through enhancing uptake and transportation of fluoride—In vitro study. Pediatr. Dent. J. 2018, 28, 103–109. [Google Scholar] [CrossRef]
- Labala, S.; Jose, A.; Chawla, S.R.; Khan, M.S.; Bhatnagar, S.; Kulkarni, O.P.; Venuganti, V.V.K. Effective melanoma cancer suppression by iontophoretic co-delivery of STAT3 siRNA and imatinib using gold nanoparticles. Int. J. Pharm. 2017, 525, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, V.; Kim, H.D.; Friden, P.M.; Banga, A.K. Iontophoresis mediated in vivo intradermal delivery of terbinafine hydrochloride. Int. J. Pharm. 2010, 393, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Choi, T.B.; Lee, D.A. Transscleral and transcorneal iontophoresis of vancomycin in rabbit eyes. J. Ocul. Pharmacol. 1988, 4, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Güngör, S.; Delgado-Charro, M.B.; Ruiz-Perez, B.; Schubert, W.; Isom, P.; Moslemy, P.; Patane, M.A.; Guy, R.H. Trans-scleral iontophoretic delivery of low molecular weight therapeutics. J. Control. Release 2010, 147, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Karpiński, T.M. Wpływ anestetyków lokalnych na odczyn zapalny. Influence of local anesthetics on inflammatoy reaction. Nowiny Lek. 2005, 74, 732–736. [Google Scholar]
- Rirattanapong, P.; Vongsavan, K.; Kraivaphan, P.; Vongsavan, N.; Matthews, B. Effect of the topical application of 50% lignocaine hydrochloride on the sensitivity of dentine in man. Arch. Oral Biol. 2013, 58, 1549–1555. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Ikeda, H.; Suda, H. The effect of pulpward pressure on the response to 50% lidocaine (lignocaine) applied to exposed dentine in cats. Arch. Oral Biol. 2002, 47, 333–336. [Google Scholar] [CrossRef]
- Markowitz, K. Pretty painful: Why does tooth bleaching hurt? Med. Hypotheses 2010, 74, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Saliba, S.A.; Teeter-Heyl, C.L.; McKeon, P.; Ingeroll, C.D.; Saliba, E.N. Effect of duration and amplitude of direct current when lidocaine is delivered by iontophoresis. Pharmaceutics 2011, 3, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Power, I. Fentanyl HCl iontophoretic transdermal system (ITS): Clinical application of iontophoretic technology in the management of acute postoperative pain. Br. J. Anaesth. 2007, 98, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, F.; Eberhart, L.; Wennberg, E.; Dodds, S.J.; Van Bellinghen, L.; Annemans, L.; Kavanagh, S.; Choe, Y. Fentanyl HCl iontophoretic transdermal system versus morphine IV-PCA for postoperative pain management: Survey of healthcare provider opinion. Curr. Med. Res. Opin. 2009, 25, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Patane, M.A.; Cohen, A.; From, S.; Torkildsen, G.; Welch, D.; Ousler, G.W., 3rd. Ocular iontophoresis of EGP-437 (dexamethasone phosphate) in dry eye patients: Results of a randomized clinical trial. Clin. Ophthalmol. 2011, 5, 633–643. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cohen, A.E.; Assang, C.; Patane, M.A.; From, S.; Korenfeld, M.; Avion Study Investigators. Evaluation of dexamethasone phosphate delivered by ocular iontophoresis for treating noninfectious anterior uveitis. Ophthalmology 2012, 119, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Patane, M.A.; Schubert, W.; Sanford, T.; Gee, R.; Burgos, M.; Isom, W.P.; Ruiz-Perez, B. Evaluation of ocular and general safety following repeated dosing of dexamethasone phosphate delivered by transscleral iontophoresis in rabbits. J. Ocul. Pharmacol. Ther. 2013, 29, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.R.; Morris, R.L.; Boeh, S.D.; Panus, P.C.; Sembrowich, W.L. Effects of iontophoresis current magnitude and duration on dexamethasone deposition and localized drug retention. Phys. Ther. 2003, 83, 161–170. [Google Scholar] [PubMed]
- Wieckiewicz, M.; Boening, K.; Wiland, P.; Shiau, Y.Y.; Paradowska-Stolarz, A. Reported concepts for the treatment modalities and pain management of temporomandibular disorders. J. Headache Pain 2015, 16, 106. [Google Scholar] [CrossRef] [PubMed]
- Kogut, G.; Kwolek, A. Zaburzenia czynnościowe układu ruchowego narządu żucia-diagnostyka i leczenie. Rehabil. Med. 2006, 10, 41–48. [Google Scholar]
- Buescher, J.J. Temporomandibular joint disorders. Am. Fam. Phys. 2007, 76, 1477–1482. [Google Scholar]
- Wałach, A.; Pihut, M.; Loster, J. Charakterystyka zabiegów fizjoterapeutycznych stosowanych w leczeniu pacjentów z zaburzeniami czynnościowymi narządu żucia. Protet. Stomatol. 2006, 56, 274–281. [Google Scholar]
- Zimmermann-Górska, I.; Puszczewicz, M.; Białkowska-Puszczewicz, G. Ocena płynu stawowego jako podstawa stosowania preparatu Mobilat-żel w chorobach stawów. Reumatologia 1999, 37, 258–263. [Google Scholar]
- Arunkumar, S.; Shivakumar, H.N.; Narasimha Murthy, S. Effect of terpenes on transdermal iontophoretic delivery of diclofenac potassium under constant voltage. Pharm. Dev. Technol. 2017, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gianni, C. Update on antifungal therapy with terbinafine. G. Ital. Dermatol. Venereol. 2010, 145, 415–424. [Google Scholar] [PubMed]
- Wolinsky, J.B.; Colson, Y.L.; Grinstaff, M.W. Local drug delivery strategies for cancer treatment: Gels, nanoparticles, polymeric films, rods, and wafers. J. Control. Release 2012, 159, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.D.; Jajja, M.R.; O’Neill, A.T.; Bickford, L.R.; Keeler, A.W.; Hyder, N.; Wagner, K.; Deal, A.; Little, R.E.; Moffitt, R.A.; et al. Local iontophoretic administration of cytotoxic therapies to solid tumors. Sci. Transl. Med. 2015, 7, 273ra14. [Google Scholar] [CrossRef] [PubMed]
- Welch, M.L.; Grabski, W.J.; McCollough, M.L.; Skelton, H.G.; Smith, K.J.; Menon, P.A.; Anderson, L.L. 5-fluorouracil iontophoretic therapy for Bowen’s disease. J. Am. Acad. Dermatol. 1997, 36, 956–958. [Google Scholar] [CrossRef]
- Bacro, T.R.; Holladay, E.B.; Stith, M.J.; Maize, J.C.; Smith, C.M. Iontophoresis treatment of basal cell carcinoma with cisplatin: A case report. Cancer Detect. Prev. 2000, 24, 610–619. [Google Scholar] [PubMed]
- Zuckerman, J.E.; Davis, M.E. Clinical experiences with systemically administered siRNA-based therapeutics in cancer. Nat. Rev. Drug Discov. 2015, 14, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Pardoll, D.; Jove, R. STATs in cancer inflammation and immunity: A leading role for STAT3. Nat. Rev. Cancer 2009, 9, 798–809. [Google Scholar] [CrossRef] [PubMed]
| Drug | Iontophoresis Conditions | Tissue | References |
|---|---|---|---|
| Acetylsalicylic acid | 5 mA/cm2, 10 min | In vivo rabbit eye | [17] |
| Acetylsalicylic acid (Aspirin) | 0.06 mA, 10 min | Rat skin | [11] |
| Acidulated phosphate fluoride | 0.4 mA, 4 min | Bovine enamel | [18] |
| Acidulated phosphate fluoride | 0.2 mA, 4 min | Human teeth | [19] |
| Aciclovir | 0.2 mA/cm2, 10 min | In vivo rat skin | [20] |
| Aciclovir | 0.5 mA/cm2, 5 min | Porcine eye | [21] |
| Aciclovir prodrugs | 0.5 mA/cm2, 5 min | Porcine eye | [21] |
| Amikacin | 3.7–7.4 mA/cm2, 20 min | In vivo rabbit eye | [22] |
| Amikacin sulphate | 0.5 mA/cm2, 2 h | Rabbit skin | [23] |
| 5-Aminolevulinic acid | 1–2 mA, 10 min | In vivo human skin | [24] |
| Amoxicillin | 100, 200, 300 µA/cm2 | In vivo rabbit skin | [25] |
| Cefuroxime | 100, 200, 300 µA/cm2 | In vivo rabbit skin | [25] |
| Celecoxib | 0.1–0.5 mA/cm2, 25–300 min | Rat skin | [26] |
| Ciprofloxacin | 0.8 mA, 10 min | In vivo rabbit cornea | [27] |
| Ciprofloxacin hydrochloride | 6.25 mA/cm2, 5 min | Porcine cornea and whole eye | [28] |
| Cupral | 1.5 mA, 10 min | Human teeth root canals | [29] |
| Curcumin with STAT3 siRNA | 0.47 mA/cm2, 4 h | Porcine skin | [30] |
| Dexamethasone | 0.36 mA/cm2, 10 and 20 min | In vivo human skin | [31] |
| Dexamethasone | 4 mA, 15–30 min | In vivo human temporomandibular joint | [32] |
| Dexamethasone | 5.1 mA/cm2, 4 min | In vivo rabbit cornea | [33] |
| Dexamethasone | 1 mA/cm2, 180 min | Porcine cornea | [33] |
| Dexamethasone | 0.1 mA, 8 h | Bovine palate | [34] |
| Diclofenac | 5 and 10 mA, 20 min | In vivo human muscle | [35] |
| Diclofenac sodium | 0.5 mA/cm2, 8 h | Porcine skin | [36] |
| Ibuprofen | 0.06 mA, 10 min | Rat skin | [11] |
| Indemethacin | 0.06 mA, 10 min | Rat skin | [11] |
| Fentanyl hydrochloride | 10 min | In vivo human skin | [37] |
| 5-Fluorouracil | 1 mA/cm2, 10 and 20 min | Bovine buccal mucosa | [38] |
| 5-Fluorouracil | 0.5 mA/cm2, 15 min and 6 h | Porcine skin | [39] |
| Gentamicin sulfate | 2.5–5.1 mA/cm2, 1 and 2 min | In vivo rabbit cornea | [40] |
| Gentamicin sulfate | 5.1 mA/cm2, 1 min | In vivo rabbit cornea | [41] |
| Gentamicin sulfate | 0.51–3.1 mA/cm2, 1 min | In vivo rabbit cornea | [42] |
| Gentamicin sulfate | 40 mA, 1, 5 and 10 min | Sheep and human bone | [43] |
| Gentamicin sulfate | 0.8 and 2 mA/cm2, 1 min | In vivo rabbit cornea | [44] |
| Gp100 peptide | 0.4 mA/cm2, 1 h | In vivo mouse melanoma | [45] |
| Ketoconazole | 4–6 mA, 15 min | Rabbit eye | [46] |
| Ketoprofen cationic prodrug | 0.2 mA, 6 h | Rat skin | [47] |
| Leucovorin | 1 mA/cm2, 10 and 20 min | Bovine buccal mucosa | [38] |
| Lidocaine | 1 mA, 10 min | In vivo human skin | [48] |
| Lidocaine | 0.3 mA, 8 h | Porcine buccal mucosa | [49] |
| Lidocaine | 0.3 mA/cm2, 8 h | Porcine buccal mucosa | [50] |
| Lidocaine | 3 V, 1 kHz, 2–20 min | Human enamel/dentine | [51] |
| Lidocaine | 1 mA/cm2, 1 h | Porcine buccal mucosa | [52] |
| Lidocaine | 0.5 mA/cm2, 6 h | Porcine buccal mucosa | [53] |
| Lidocaine | 0.5–5 mA/cm2 or 0.5–20 mA/cm2, 30 min | Rabbit cornea or rabbit conjunctiva | [54] |
| Lignocaine with epinephrine | 1 mA, 10 min | In vivo human skin | [48] |
| Lignocaine with epinephrine | 40 mA, 10.5 min | In vivo human skin | [55] |
| Lignocaine with epinephrine | 0.2 mA, 2–14 min | Human dentine | [56] |
| Lignocaine with epinephrine | 0.12 mA, 90 s | Human dentine | [57] |
| Metronidazole | 0.05 mA, 10 min | Human dentine | [58] |
| Naproxen sodium | 0.05 mA, 10 min | Human dentine | [58] |
| Potassium iodide | 1.5 mA, 10 min | Human teeth root canals | [29] |
| Riboflavin | 1 mA, 5 min + UVA irradiance | Human cornea | [59] |
| Riboflavin | 10 min | In vivo rabbit eye | [60] |
| Riboflavin | 1 mA, 5 min | In vivo human eye | [61] |
| Riboflavin-5-phosphate | 1 mA, 5 min | In vivo human cornea | [62] |
| Silk fibroin with neurotensin | 0.2 mA/cm2, 30 min | Macrophages | [63] |
| Silver-polymer-based surface | 1.5–15 µA, 30 min | Bacterial strains | [64] |
| Sodium fluoride | 0.5–0.6 mA, 30 s | Rat enamel | [65] |
| Sodium fluoride | 10 mA, 2–3.3 min | Human enamel | [66] |
| Sodium fluoride | 0.5 mA, 2 min | In vivo human teeth | [67] |
| Sodium fluoride | 0.5 mA, 5 min | Human dentine | [68] |
| Sodium fluoride | 0.4 mA, 4 min | Bovine enamel | [18] |
| Sodium fluoride | 0.2 mA, 4 min | Bovine enamel | [69] |
| Sodium fluoride | 0.5 mA, 2 min | In vivo human teeth | [70] |
| Sodium fluoride | 0.2 mA, 4 min | Human teeth | [19] |
| Sodium fluoride | 0.1 mA, 8 h | Bovine enamel | [71] |
| Sodium fluoride | 0.1 mA, 8 h | Bovine palate | [34] |
| Sodium fluoride | 0.3 mA, 4 min, for 5 days | Bovine enamel | [72] |
| Sodium fluoride | 1.5 mA, 3 min | Human dentine | [73] |
| Sodium fluoride | 0.5 mA, 3 min 0.5 mA, 5 min 0.5 mA, 10 min | Bovine enamel | [74] |
| Sodium salicylate | 0.05 mA, 10 min | Human dentine | [58] |
| Sodium salicylate | 0.1 mA, 8 h | Bovine palate | [34] |
| STAT3 siRNA with imatinib mesylate | 0.5 mA/cm2, 2 h | In vivo mouse melanoma | [75] |
| Terbinafine hydrochloride | 0.25 mA, 10, 15 and 20 min | In vivo rat skin | [76] |
| Vancomycin | 0.5–3.5 mA, 10 min | Rabbit eye | [77] |
| Vancomycin | 2.55–10.2 mA/cm2, 120 min | Rabbit sclera | [78] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karpiński, T.M. Selected Medicines Used in Iontophoresis. Pharmaceutics 2018, 10, 204. https://doi.org/10.3390/pharmaceutics10040204
Karpiński TM. Selected Medicines Used in Iontophoresis. Pharmaceutics. 2018; 10(4):204. https://doi.org/10.3390/pharmaceutics10040204
Chicago/Turabian StyleKarpiński, Tomasz M. 2018. "Selected Medicines Used in Iontophoresis" Pharmaceutics 10, no. 4: 204. https://doi.org/10.3390/pharmaceutics10040204
APA StyleKarpiński, T. M. (2018). Selected Medicines Used in Iontophoresis. Pharmaceutics, 10(4), 204. https://doi.org/10.3390/pharmaceutics10040204