Molecular Disorder of Bicalutamide—Amorphous Solid Dispersions Obtained by Solvent Methods
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solvent Evaporation
2.3. Spray Drying
2.4. Scanning Electron Microscopy (SEM)
2.5. Differential Scanning Calorimetry (DSC)
2.6. Powder X-ray Diffraction (PXRD)
2.7. Laser Diffraction Measurements
2.8. Infrared Spectroscopy (FTIR)
2.9. Dissolution Study
2.10. Intrinsic Dissolution Rate Study
2.11. Solubility Study
2.12. Statistical Analysis
3. Results and Discussion
3.1. Molecular Arrangement
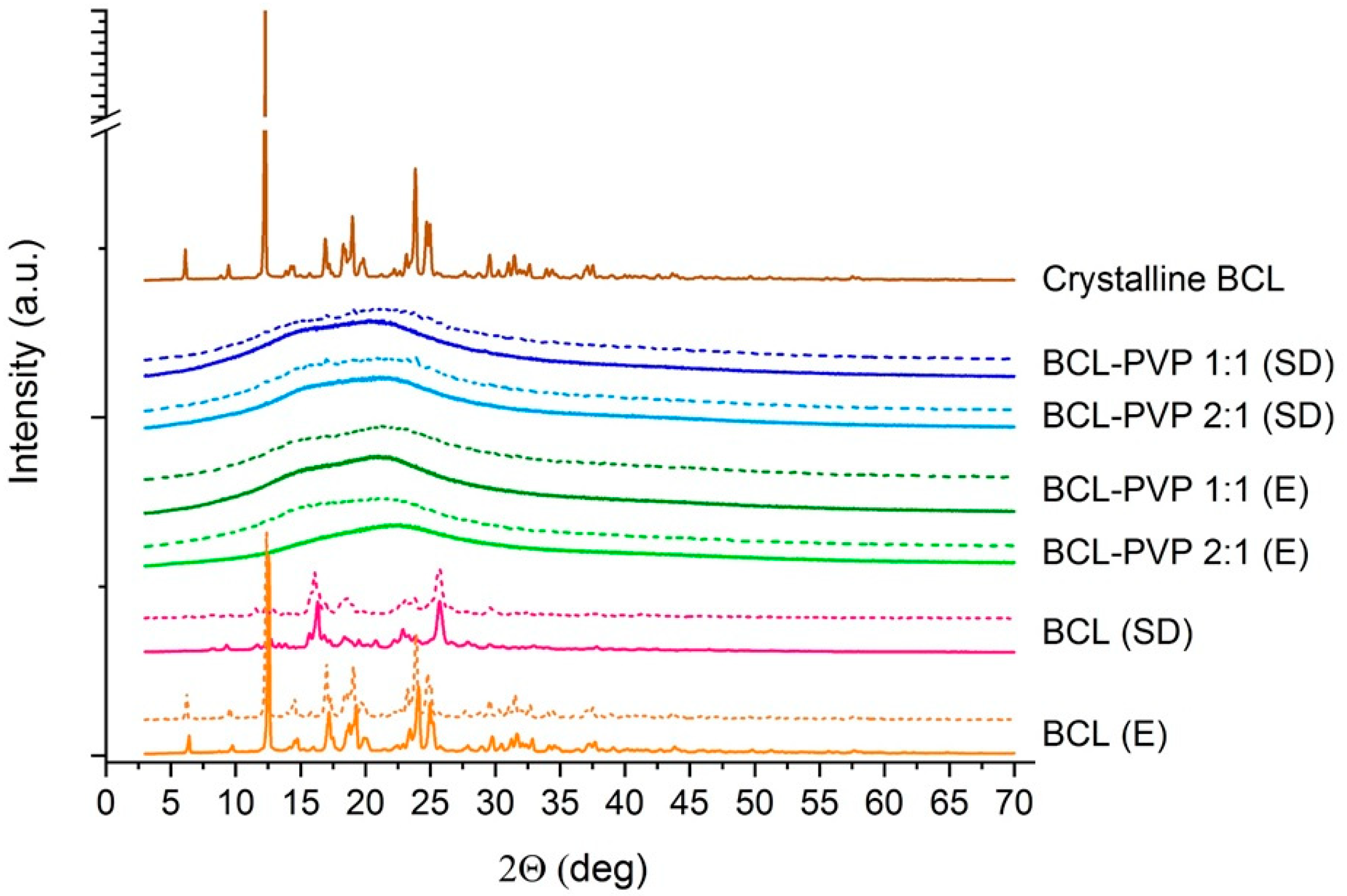
3.1.1. X-ray Powder Diffraction Studies
3.1.2. DSC Studies
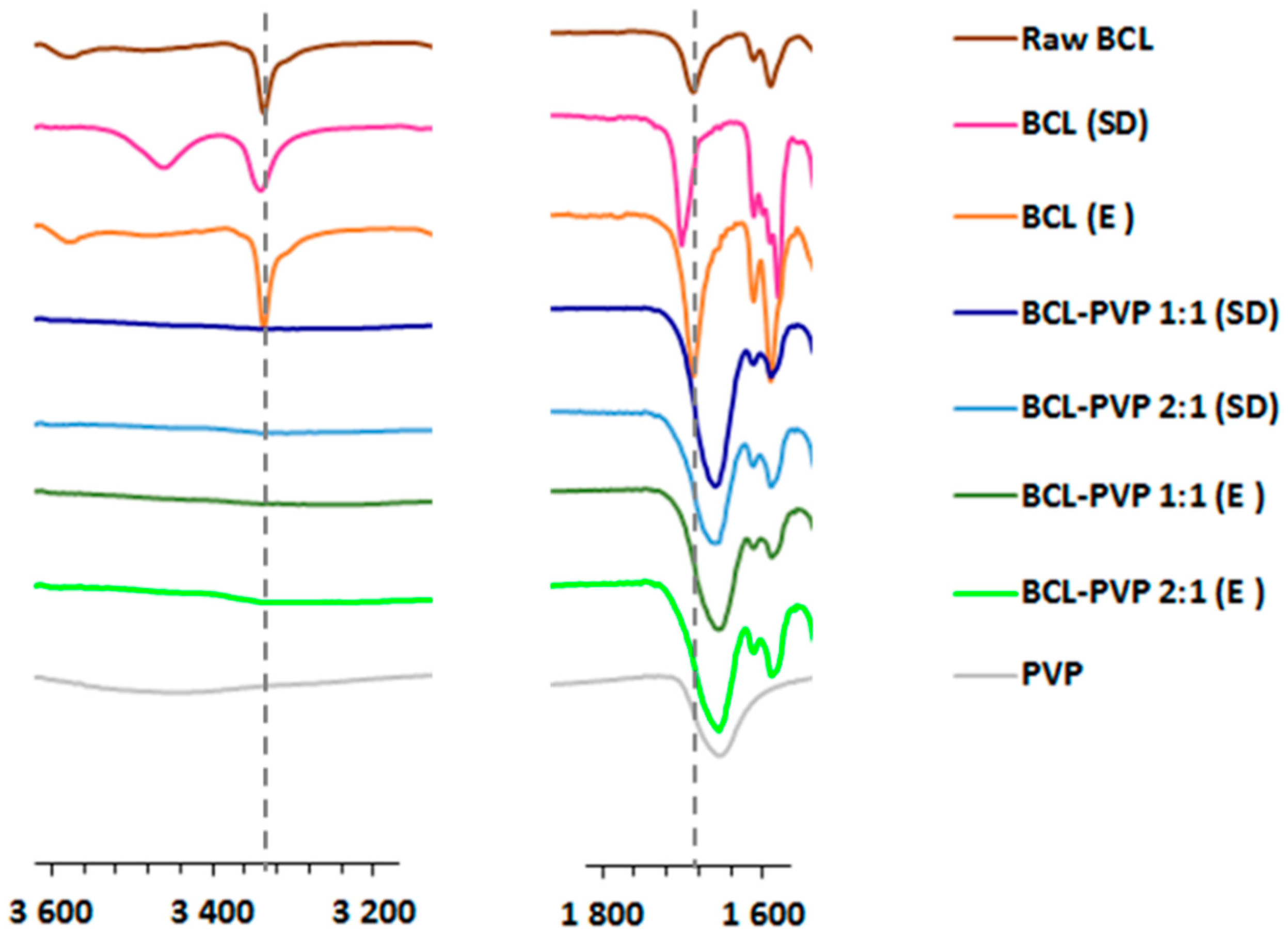
3.1.3. Infrared Spectroscopy
3.2. Particle Size and Morphology
3.2.1. Morphology of Particles from Scanning Electron Microscopy
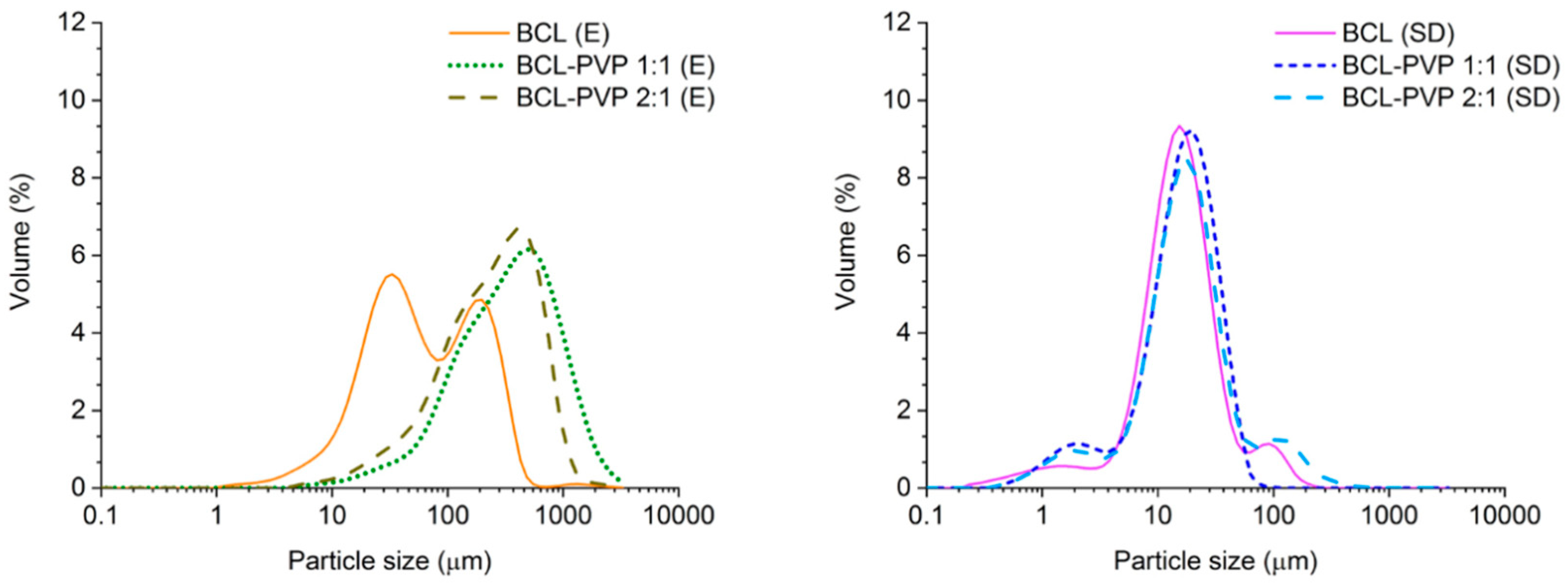
3.2.2. Particle Size Distribution from Laser Diffraction Measurements
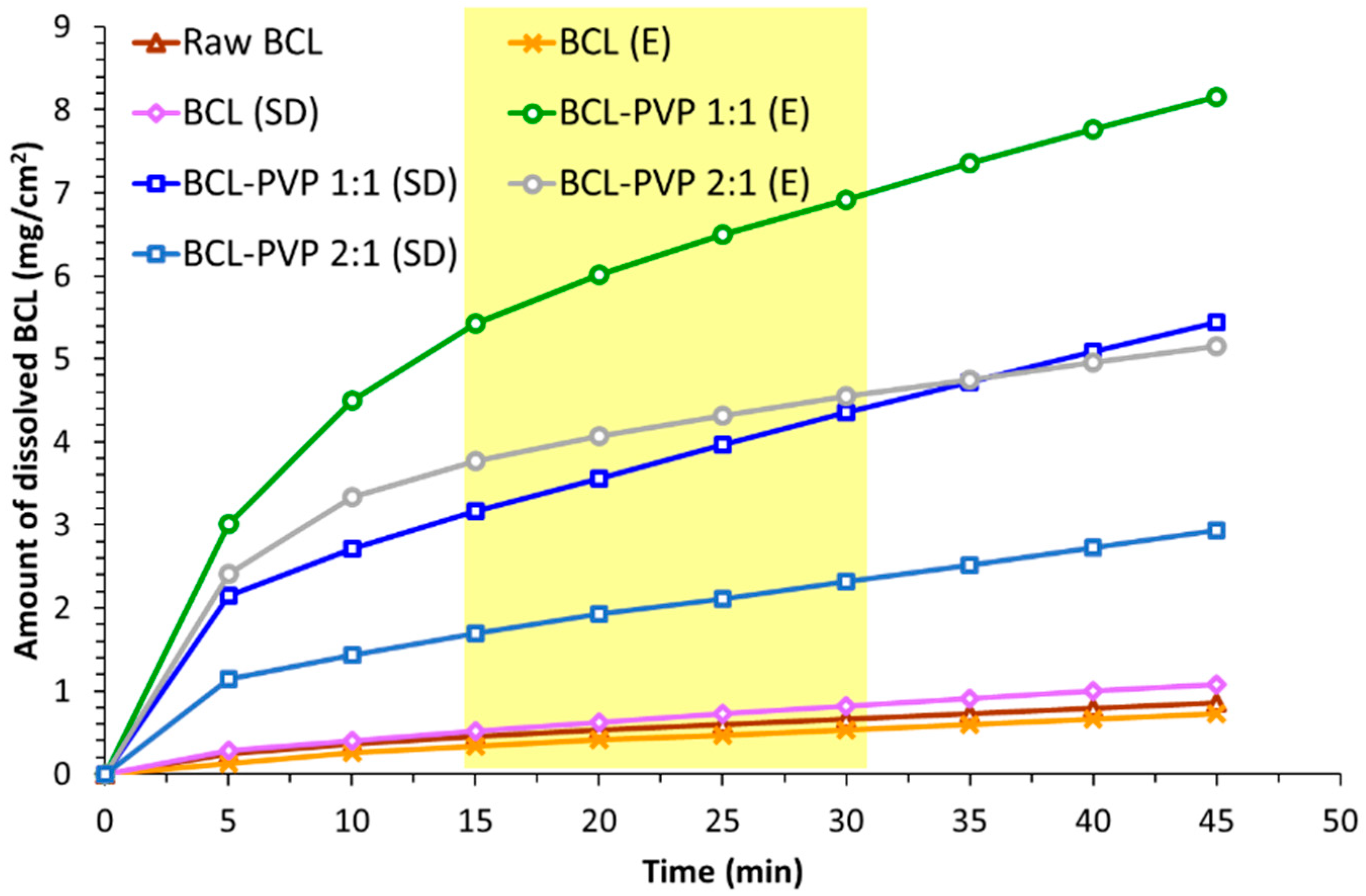
3.3. Dissolution Study
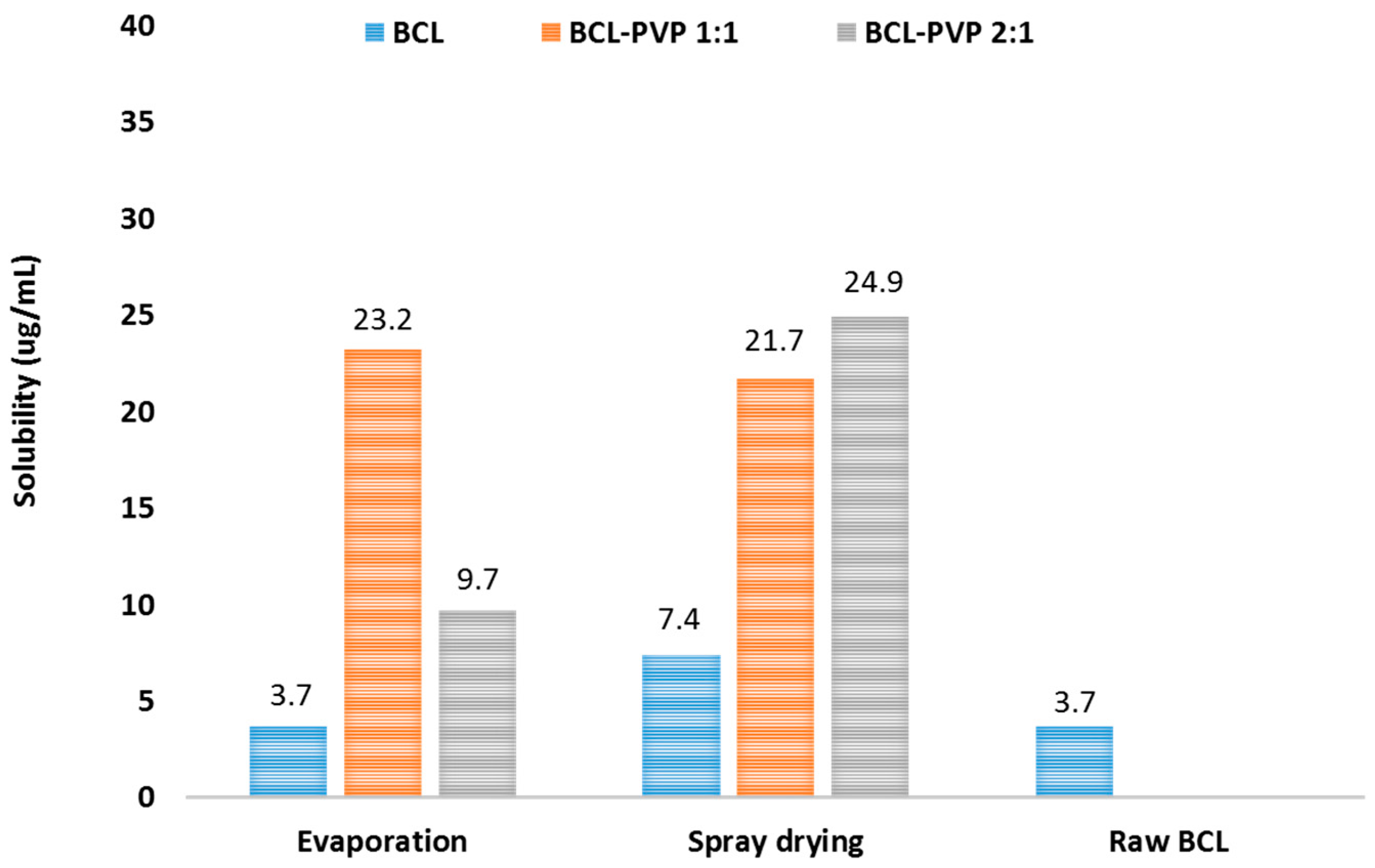
3.4. Saturation Solubility
3.5. Intrinsic Dissolution Study
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baghel, S.; Cathcart, H.; O’Reilly, N.J. Polymeric Amorphous Solid Dispersions: A Review of Amorphization, Crystallization, Stabilization, Solid-State Characterization, and Aqueous Solubilization of Biopharmaceutical Classification System Class II Drugs. Int. J. Pharm. 2016, 105, 2527–2544. [Google Scholar] [CrossRef] [PubMed]
- Rams-Baron, M.; Jachowicz, R.; Boldyreva, E.; Zhou, D.; Jamróz, W.; Paluch, M. Why Amorphous Drugs? In Amorphous Drugs. Benefits and Challenges; Springer: Cham, Switzerland, 2018; pp. 1–7. ISBN 978-3-319-72001-2. [Google Scholar]
- Stillinger, F. Supercooled liquids, glass transition, and the Kauzmann paradox. J. Chem. Phys. 1988, 88, 7818–7825. [Google Scholar] [CrossRef]
- Kanaujia, P.; Poovizhi, P.; Ng, W.K.; Tan, R.B.H. Amorphous formulations for dissolution and bioavailability enhancement of poorly soluble APIs. Powder Technol. 2015, 285, 2–15. [Google Scholar] [CrossRef]
- Zhao, M.; Barker, S.A.; Belton, P.S.; McGregor, C.; Craig, D.Q. Development of fully amorphous dispersions of a low Tg drug via co-spray drying with hydrophilic polymers. Eur. J. Pharm. Biopharm. 2012, 82, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.; Taylor, J.S. Ideal copolymers and the 2nd-order transitions of synthetic rubbers 1. Non-crystalline copolymers. J. Appl. Chem. 1952, 2, 493–500. [Google Scholar] [CrossRef]
- Le-Ngoc Vo, C.; Park, C.; Lee, B.-J. Current trends and future perspectives of solid dispersions containing poorly water-soluble drugs. Eur. J. Pharm. Biopharm. 2013, 85, 799–813. [Google Scholar] [CrossRef]
- Vasconcelos, T.; Marques, S.; das Neves, J.; Sarmento, B. Amorphous solid dispersions: Rational selection of a manufacturing process. Adv. Drug Deliv. Rev. 2016, 100, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Gawai, S.K.; Deshmane, S.V.; Purohit, R.N.; Biyani, K.R. In vivo-in vitro evaluation of solid dispersion containing ibuprofen. AJADD 2013, 1, 66–72. [Google Scholar]
- Tabbakhian, M.; Hasanzadeh, F.; Tavakoli, N.; Jamshidian, Z. Dissolution enhancement of glibenclamide by solid dispersion: Solvent evaporation versus a supercritical fluid-based solvent -antisolvent technique. Res. Pharm. Sci. 2014, 9, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Taylor, L. Dissolution performance of high drug loading celecoxib amorphous solid dispersions formulated with polymer combinations. Pharm. Res. 2015, 33, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Keratichewanun, S.; Yoshihashi, Y.; Sutanthavibul, N.; Terada, K.; Chatchawalsaisin, J. An investigation of nifedipine miscibility in solid dispersions using Raman spectroscopy. Pharm. Res. 2015, 32, 2458–2473. [Google Scholar] [CrossRef] [PubMed]
- Purohit, H.S.; Taylor, L.S. Miscibility of itraconazole–hydroxypropyl methylcellulose blends: Insights with high resolution analytical methodologies. Mol. Pharm. 2015, 12, 4542–4553. [Google Scholar] [CrossRef] [PubMed]
- Afifi, S. Solid Dispersion Approach Improving Dissolution Rate of Stiripentol: A Novel Antiepileptic Drug. Iran. J. Pharm. Res. 2015, 14, 1001–1014. [Google Scholar] [PubMed]
- Paudel, A.; Worku, A.; Meeus, J.; Guns, S.; Mooter, G. Manufacturing of solid dispersions of poorly water soluble drugs by spray drying: Formulation and process considerations. Int. J. Pharm. 2013, 453, 253–284. [Google Scholar] [CrossRef] [PubMed]
- Fong, S.Y.; Ibisogly, A.; Bauer-Brandl, A. Solubility enhancement of BCS class II drug by solid phospholipid dispersions: Spray drying versus freeze-drying. Int. J. Pharm. 2015, 496, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Wlodarski, K.; Sawicki, W.; Kozyra, A.; Tajber, L. Physical stability of solid dispersions with respect to thermodynamic solubility of tadalafil in PVP-VA. Eur. J. Pharm. Biopharm. 2015, 96, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Sóti, P.L.; Bocz, K.; Pataki, H.; Eke, Z.; Farkas, A.; Verreck, G.; Kiss, É.; Fekete, P.; Vigh, T.; Wagner, I.; et al. Comparison of spray drying, electroblowing and electrospinning for preparation of eudragit E and itraconazole solid dispersions. Int. J. Pharm. 2015, 494, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Higashi, K.; Hayashi, H.; Yamamoto, K.; Moribe, K. The effect of drug and EUDRAGIT®S 100 miscibility in solid dispersions on the drug and polymer dissolution rate. Int. J. Pharm. 2015, 494, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Smithey, P.G.D.; Taylor, L. Amorphous solid dispersions: An enabling formulation technology for oral delivery of poorly water soluble drugs. AAPS Newsmag. 2013, 16, 11–14. [Google Scholar]
- Singh, A.; Van den Mooter, A.G. Spray drying formulation of amorphous solid dispersions. Adv. Drug Deliv. Rev. 2016, 100, 27–50. [Google Scholar] [CrossRef] [PubMed]
- Masiello, D.; Cheng, S.; Bubley, G.J.; Lu, M.L.; Balk, S.P. Bicalutamide Functions as an Androgen Receptor Antagonist by Assembly of a Transcriptionally Inactive Receptor. J. Biol. Chem. 2002, 227, 26321–26326. [Google Scholar] [CrossRef] [PubMed]
- Szafraniec, J.; Antosik, A.; Knapik-Kowalczuk, J.; Kurek, M.; Syrek, K.; Chmiel, K.; Paluch, M.; Jachowicz, R. Planetary ball milling and supercritical fluid technology as a way to enhance dissolution of bicalutamide. Int. J. Pharm. 2017, 533, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Vega, D.R.; Polla, G.; Martinez, A.; Mendioroz, E.; Reinoso, M. Conformational polymorphism in bicalutamide. Int. J. Pharm. 2007, 328, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.R.; Gu, J.M. N-[4-Cyano-3-trifluoromethyl)phenyl]-3-(4-fluorophenylsulfonyl)-2-hydroxy-2-methylpropionamide. Acta Cryst. 2005, 61, 3897–3898. [Google Scholar] [CrossRef]
- Perlovich, G.L.; Blokhina, S.V.; Manin, N.G.; Volkova, T.V.; Tkachev, V.V. Polymorphism and solvatomorphism of bicalutamide. Thermophysical study and solubility. J. Therm. Anal. Calorim. 2013, 11, 655–662. [Google Scholar] [CrossRef]
- Westheim, R.J.H. Bicalutamide Forms. U.S. Patent Application US 2004/0063782, 1 April 2004. [Google Scholar]
- Német, Z.; Sztatisz, J.; Demeter, Á. Polymorph transitions of bicalutamide: A remarkable example of mechanical activation. J. Pharm. Sci. 2008, 97, 3222–3232. [Google Scholar] [CrossRef] [PubMed]
- Srikanth, M.V.; Murali Mohan Babu, G.V.; Sunil, S.A.; Sreenivasa Rao, N.; Ramana Murthy, K.V. In-vitro dissolution rate enhancement of poorly water soluble non-steroidal antiandrogen agent, bicalutamide, with hydrophilic carrier. J. Sci. Ind. Res. 2010, 69, 629–634. [Google Scholar]
- Ren, F.; Jing, Q.; Tang, Y.; Shen, Y.; Chen, J.; Gao, F.; Cui, J. Characteristics of bicalutamide solid dispersions and improvement of the dissolution. Drug Dev. Ind. Pharm. 2006, 32, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Szczurek, J.; Rams-Baron, M.; Knapik-Kowalczuk, J.; Antosik, A.; Szafraniec, J.; Jamróz, W.; Dulski, M.; Jachowicz, R.; Paluch, M. Molecular Dynamics, Recrystallization Behavior, and Water Solubility of the Amorphous Anticancer Agent Bicalutamide and Its Polyvinylpyrrolidone Mixtures. Mol. Pharmaceutics 2017, 14, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
- Rams-Baron, M.; Wlodarczyk, P.; Dulski, M.; Wlodarczyk, A.; Kruk, D.; Rachocki, A.; Jachowicz, R.; Paluch, M. The indications of tautomeric conversion in amorphous bicalutamide drug. Eur. J. Pharm. Sci. 2017, 110, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Kaptay, G. On the size and shape dependence of the solubility of nanoparticles in solutions. Int. J. Pharm. 2012, 430, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.D.; Trevaskis, N.L.; Charman, S.A.; Shanker, R.M.; Charman, W.N.; Pouton, C.W.; Porter, C.J. Strategies to address low drug solubility in discovery and development. Pharmacol. Rev. 2013, 65, 315–499. [Google Scholar] [CrossRef] [PubMed]
- Issa, M.G.; Ferraz, H.G. Intrinsic Dissolution as a Tool for Evaluating Drug Solubility in Accordance with the Biopharmaceutics Classification System. Dissolut. Technol. 2011, 18, 6–13. [Google Scholar] [CrossRef]
- Shekunov, B.; Montgomery, E.R. Theoretical analysis of drug dissolution: I. solubility and intrinsic dissolution rate. J. Pharm. Sci. 2016, 105, 2685–2697. [Google Scholar] [CrossRef] [PubMed]
- Mendyk, A.; Jachowicz, R. Unified methodology of neural analysis in decision support systems built for pharmaceutical technology. Expert Syst. Appl. 2007, 32, 1124–1131. [Google Scholar] [CrossRef]
- Mendyk, A.; Jachowicz, R.; Fijorek, K.; Dorożyński, P.; Kulinowski, P.; Polak, S. KinetDS: An open source software for dissolution test data analysis. Dissolut. Technol. 2012, 19, 6–11. [Google Scholar] [CrossRef]




| Process | Sample | Tg (K) | Tc (K) | Tm (K) |
|---|---|---|---|---|
| Spray drying | BCL–PVP 1:1 (SD) | 375 | - | - |
| BCL–PVP 2:1 (SD) | 357 | 416 | 444; 446 | |
| Evaporation | BCL–PVP 1:1 (E) | 376 | - | - |
| BCL–PVP 2:1 (E) | 357 | 396 | 449 |
| Process | Sample | Dv 10 (µm) | Dv 50 (µm) | Dv 90 (µm) |
|---|---|---|---|---|
| Spray drying | BCL–PVP 1:1 (SD) | 3.6 | 16.4 | 34.9 |
| BCL–PVP 2:1 (SD) | 4.6 | 17.1 | 60.1 | |
| BCL (SD) | 5.3 | 15.0 | 38.5 | |
| Evaporation | BCL–PVP 1:1 (E) | 80.0 | 352.0 | 1087.0 |
| BCL–PVP 2:1 (E) | 54.1 | 252.0 | 672.0 | |
| BCL (E) | 14.5 | 55.1 | 251.0 |
| Process | Sample | IDR (mg/cm2/min) |
|---|---|---|
| - | Raw BCL | 0.0144 |
| Spray drying | BCL-PVP 1:1 (SD) | 0.0796 |
| BCL-PVP 2:1 (SD) | 0.0411 | |
| BCL (SD) | 0.0204 | |
| Evaporation | BCL-PVP 1:1 (E) | 0.0992 |
| BCL-PVP 2:1 (E) | 0.0523 | |
| BCL (E) | 0.0131 |
| Function | Equation |
|---|---|
| First-order | |
| Korsmeyer–Peppas | |
| Hixson–Crowell |
| Formulation | MDT (min) | DE (%) | Mathematical Model | ||
|---|---|---|---|---|---|
| Zero Order | Korsmeyer–Peppas | Hixson–Crowell | |||
| (Constant Release) | (Diffusion Based Release) | (Erosion Based Release) | |||
| Raw BCL (not processed) | 19.55 | 5.58 | k = 0.1037 Q0 = 2.4571 r2 = 0.9594 p = 0.0035 | k = 1.0895 n = 0.5002 r2 = 0.9981 p < 0.0001 | k = 0.0117 Q0 = 2.66 r2 = 0.9036 p = 0.0114 |
| BCL (E) | 20.65 | 22.39 | k = 0.4455 Q0 = 8.9816 r2 = 0.9686 p = 0.0024 * | k = 3.8641 n = 0.5362 r2 = 0.9991 p < 0.0001 * | k = 0.0199 Q0 = 10.02 r2 = 0.9131 p = 0.0096 * |
| BCL (SD) | 15.99 | 53.05 | k = 0.8489 Q0 = 27.25 r2 = 0.9040 p = 0.0130 | k = 12.2261 n = 0.4501 r2 = 0.9827 p = 0.0011 * | k = 0.0216 Q0 = 28.13 r2 = 0.8440 p = 0.0263 |
| BCL-PVP 1:1 (SD) | 6.28 | 70.30 | k = 0.4905 Q0 = 55.90 r2 = 0.5796 p = 0.1350 | k = 35.4591 n = 0.2164 r2 = 0.8413 p = 0.0358 | k = 0.0104 Q0 = 55.06 r2 = 0.5611 p = 0.1540 |
| BCL-PVP 2:1 (SD) | 3.60 | 83.93 | k = 0.1530 Q0 = 82.11 r2 = 0.5043 p = 0.1790 | k = 73.9170 n = 0.0512 r2 = 0.7916 p = 0.0464 | k = 0.0027 Q0 = 82.02 r2 = 0.5014 p = 0.1840 |
| BCL-PVP 1:1 (E) | 7.65 | 73.17 | k = 0.5099 Q0 = 58.37 r2 = 0.4847 p = 0.1920 | k = 35.4333 n = 0.2287 r2 = 0.7469 p = 0.0769 | k = 0.0107 Q0 = 56.933 r2 = 0.4759 p = 0.2100 |
| BCL-PVP 2:1 (E) | 5.19 | 79.11 | k = 0.2097 Q0 = 75.53 r2 = 0.8494 p = 0.0260 | k = 67.22 n = 0.0632 r2 = 0.9917 p = 0.0003 * | k = 0.0037 Q0 = 75.55 r2 = 0.8394 p = 0.0284 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szafraniec, J.; Antosik, A.; Knapik-Kowalczuk, J.; Gawlak, K.; Kurek, M.; Szlęk, J.; Jamróz, W.; Paluch, M.; Jachowicz, R. Molecular Disorder of Bicalutamide—Amorphous Solid Dispersions Obtained by Solvent Methods. Pharmaceutics 2018, 10, 194. https://doi.org/10.3390/pharmaceutics10040194
Szafraniec J, Antosik A, Knapik-Kowalczuk J, Gawlak K, Kurek M, Szlęk J, Jamróz W, Paluch M, Jachowicz R. Molecular Disorder of Bicalutamide—Amorphous Solid Dispersions Obtained by Solvent Methods. Pharmaceutics. 2018; 10(4):194. https://doi.org/10.3390/pharmaceutics10040194
Chicago/Turabian StyleSzafraniec, Joanna, Agata Antosik, Justyna Knapik-Kowalczuk, Karolina Gawlak, Mateusz Kurek, Jakub Szlęk, Witold Jamróz, Marian Paluch, and Renata Jachowicz. 2018. "Molecular Disorder of Bicalutamide—Amorphous Solid Dispersions Obtained by Solvent Methods" Pharmaceutics 10, no. 4: 194. https://doi.org/10.3390/pharmaceutics10040194
APA StyleSzafraniec, J., Antosik, A., Knapik-Kowalczuk, J., Gawlak, K., Kurek, M., Szlęk, J., Jamróz, W., Paluch, M., & Jachowicz, R. (2018). Molecular Disorder of Bicalutamide—Amorphous Solid Dispersions Obtained by Solvent Methods. Pharmaceutics, 10(4), 194. https://doi.org/10.3390/pharmaceutics10040194












