Brief Effect of a Small Hydrophobic Drug (Cinnarizine) on the Physicochemical Characterisation of Niosomes Produced by Thin-Film Hydration and Microfluidic Methods
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Niosomes
2.3. Morphological Analysis of Niosomes Using Optical Microscope
2.4. Fourier-Transform Infra-Red (FTIR) Spectroscopy
2.5. Thermal Characteristics of Niosomes
2.6. Particle Size, Polydispersity, and Charge of Niosomes
2.7. Measurement
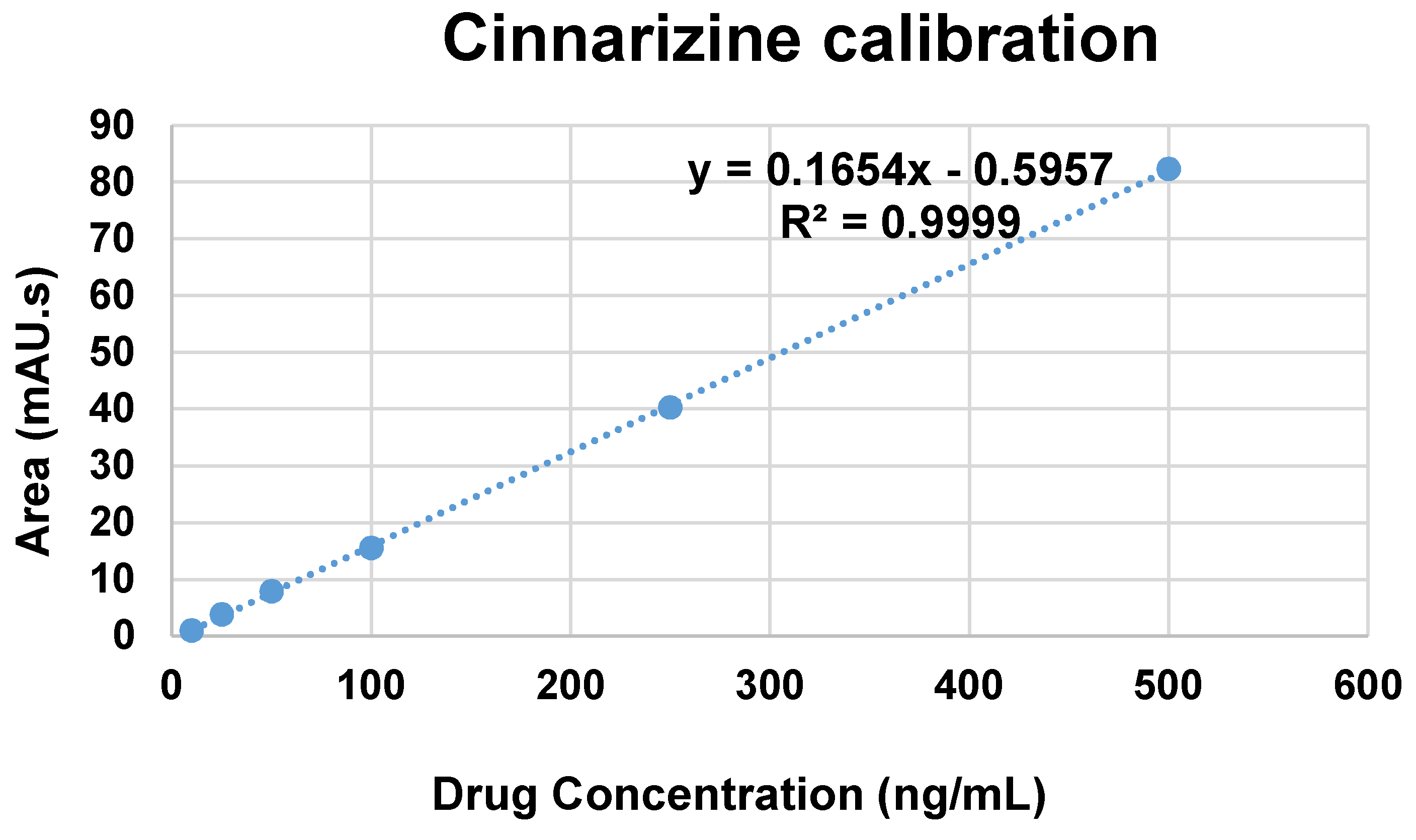
2.7.1. Calibration Curve
2.7.2. Drug Encapsulation Efficiency and Drug Release Study
3. Results and Discussion
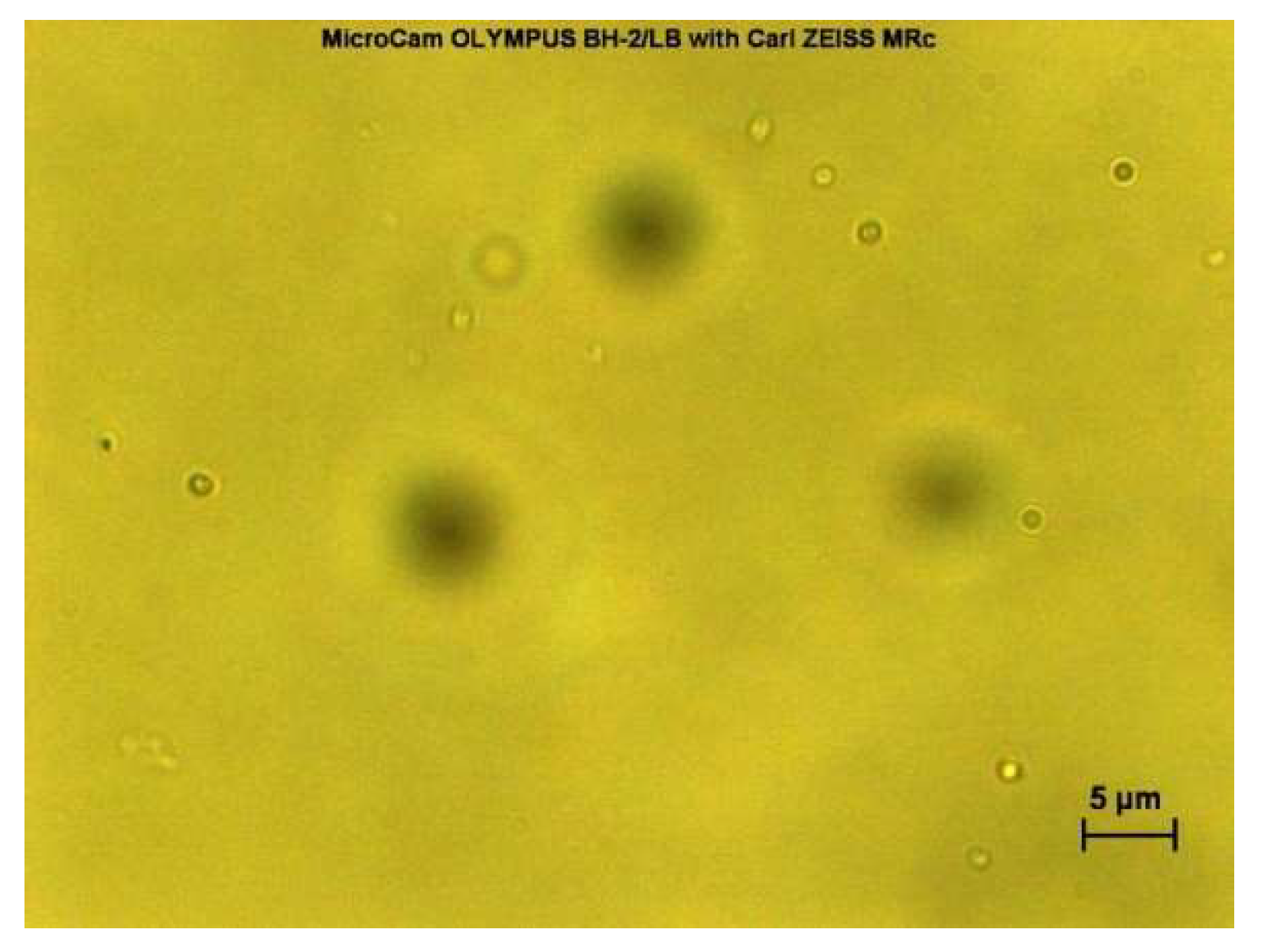
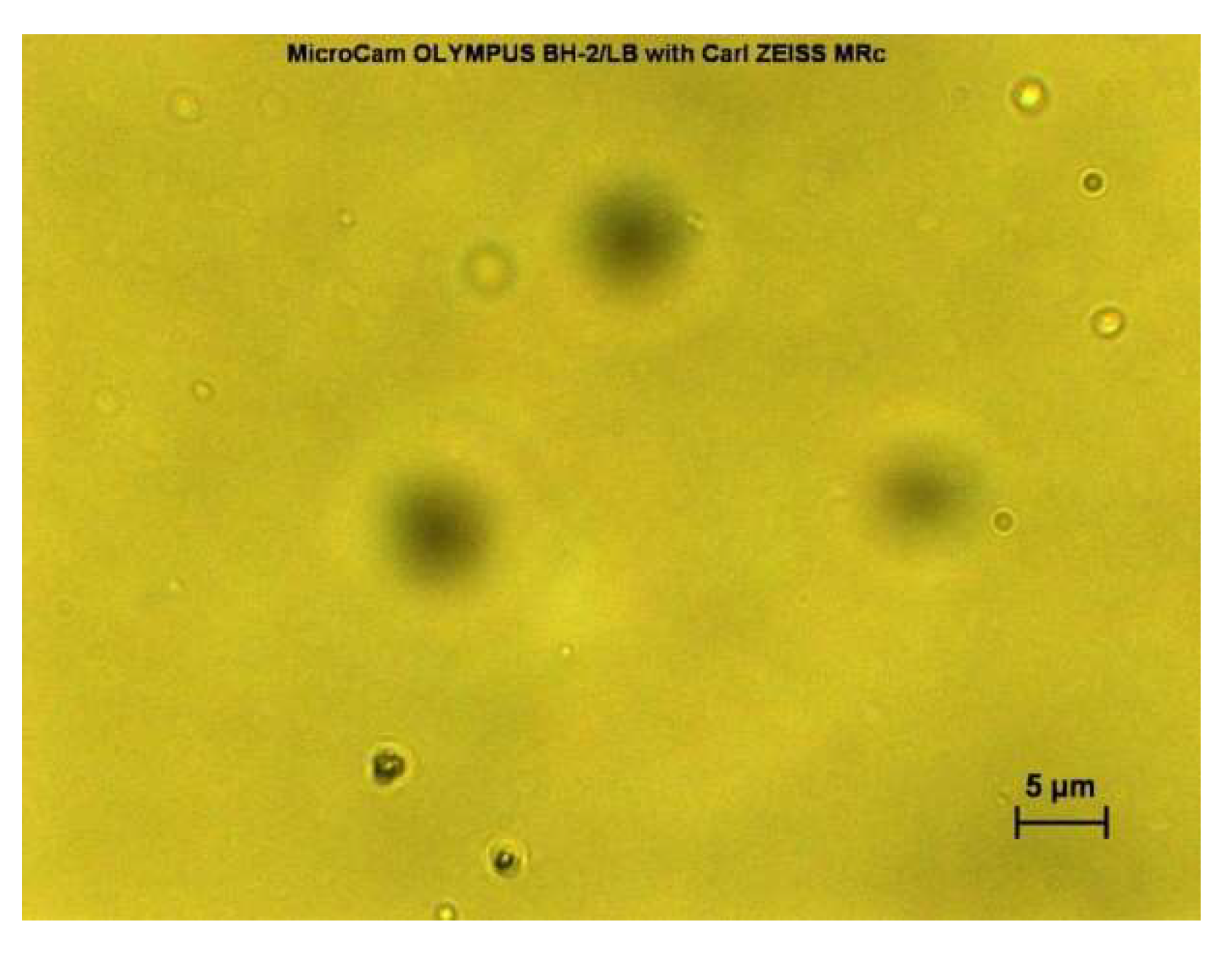
3.1. Morphological Analysis of Niosome Suspensions
3.2. Particle Size, Polydispersity, and Charge of Niosomes
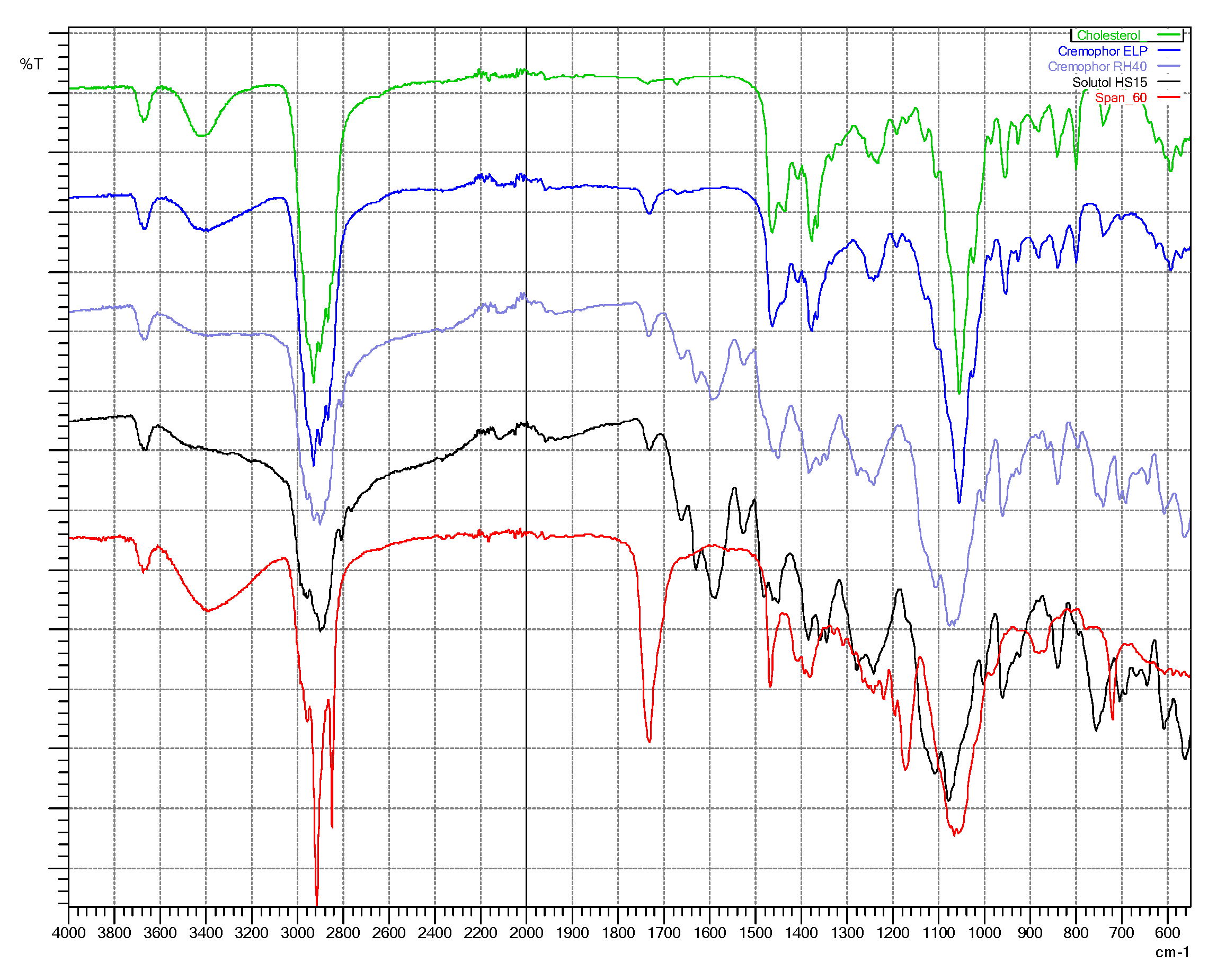
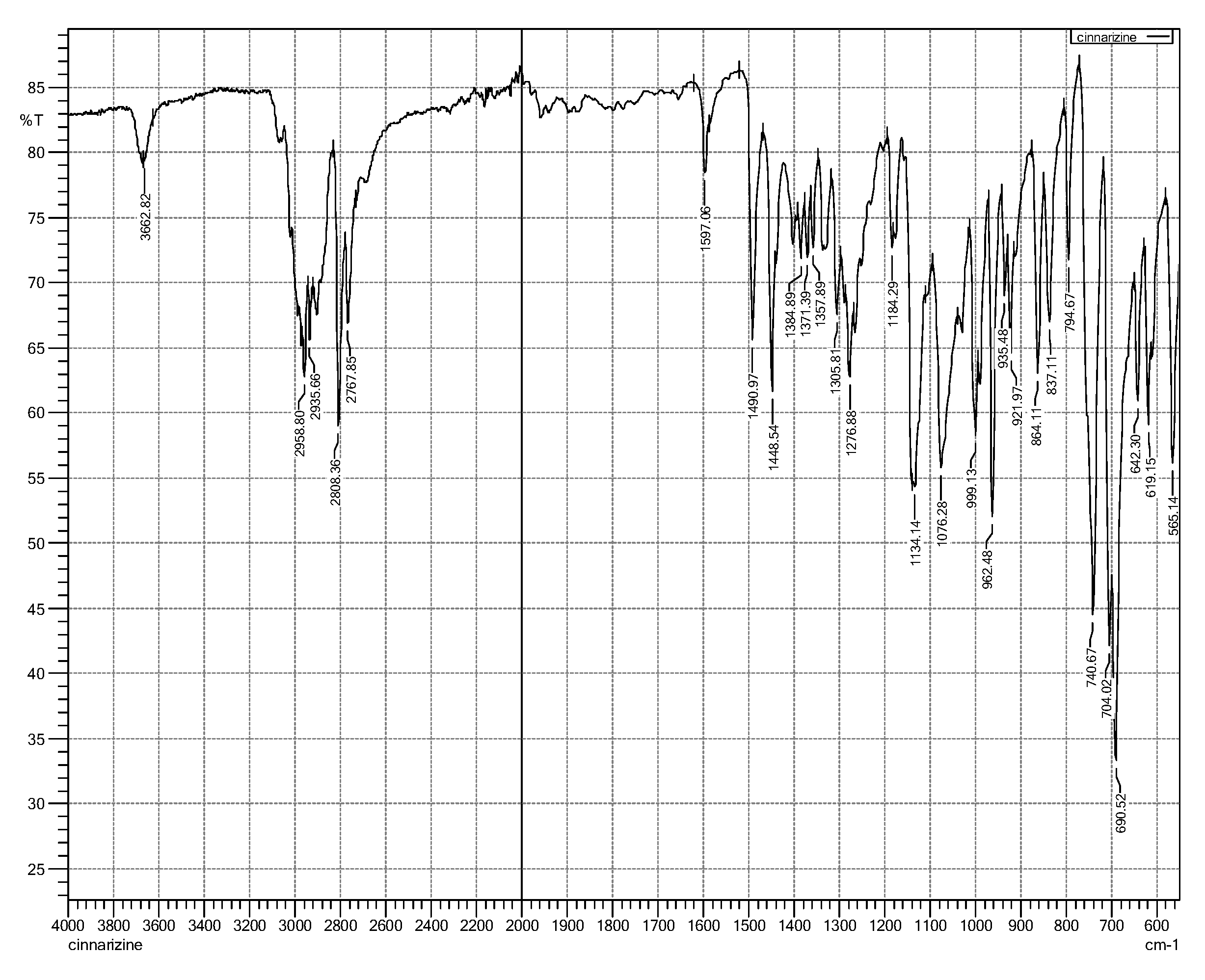
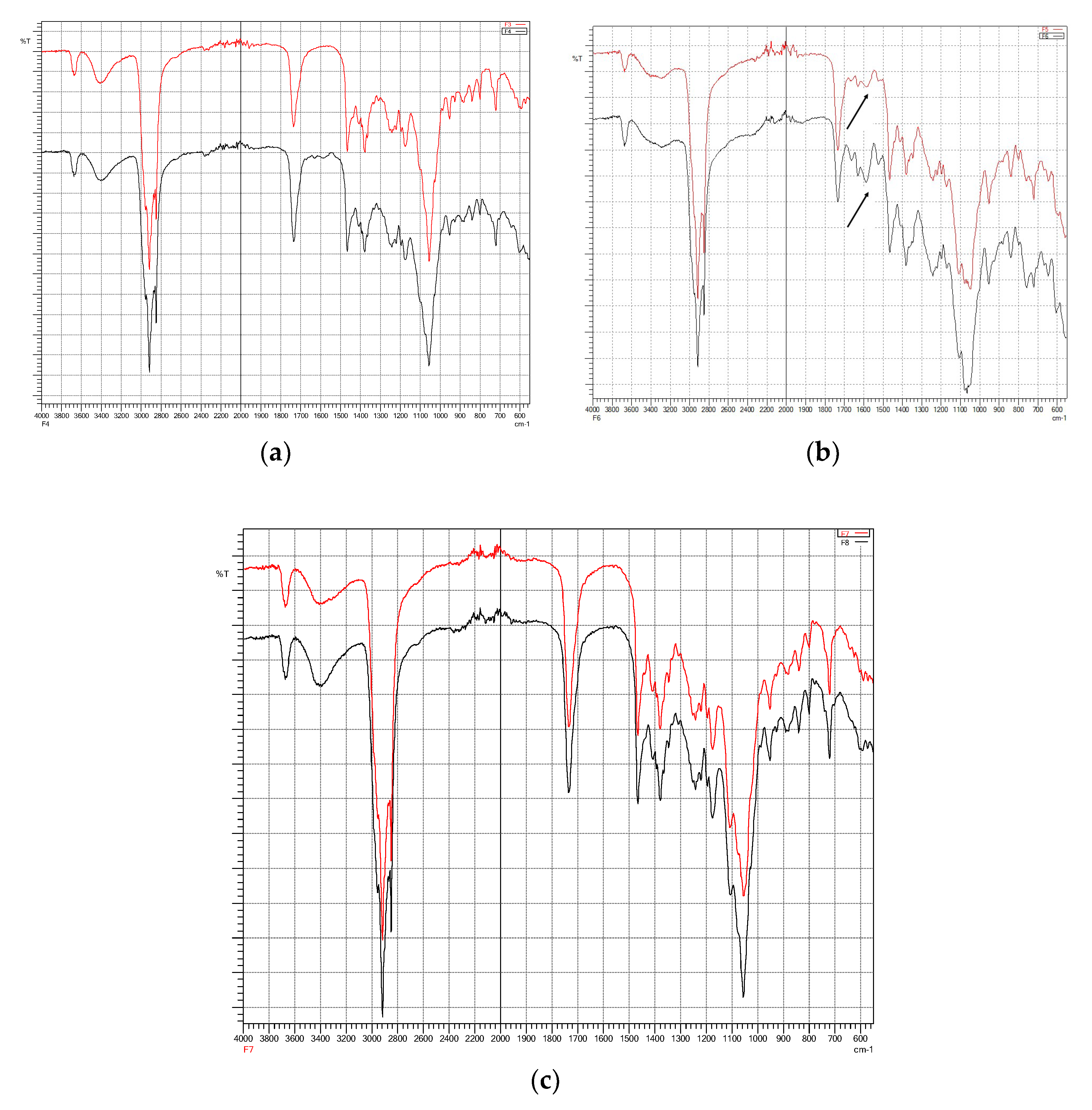
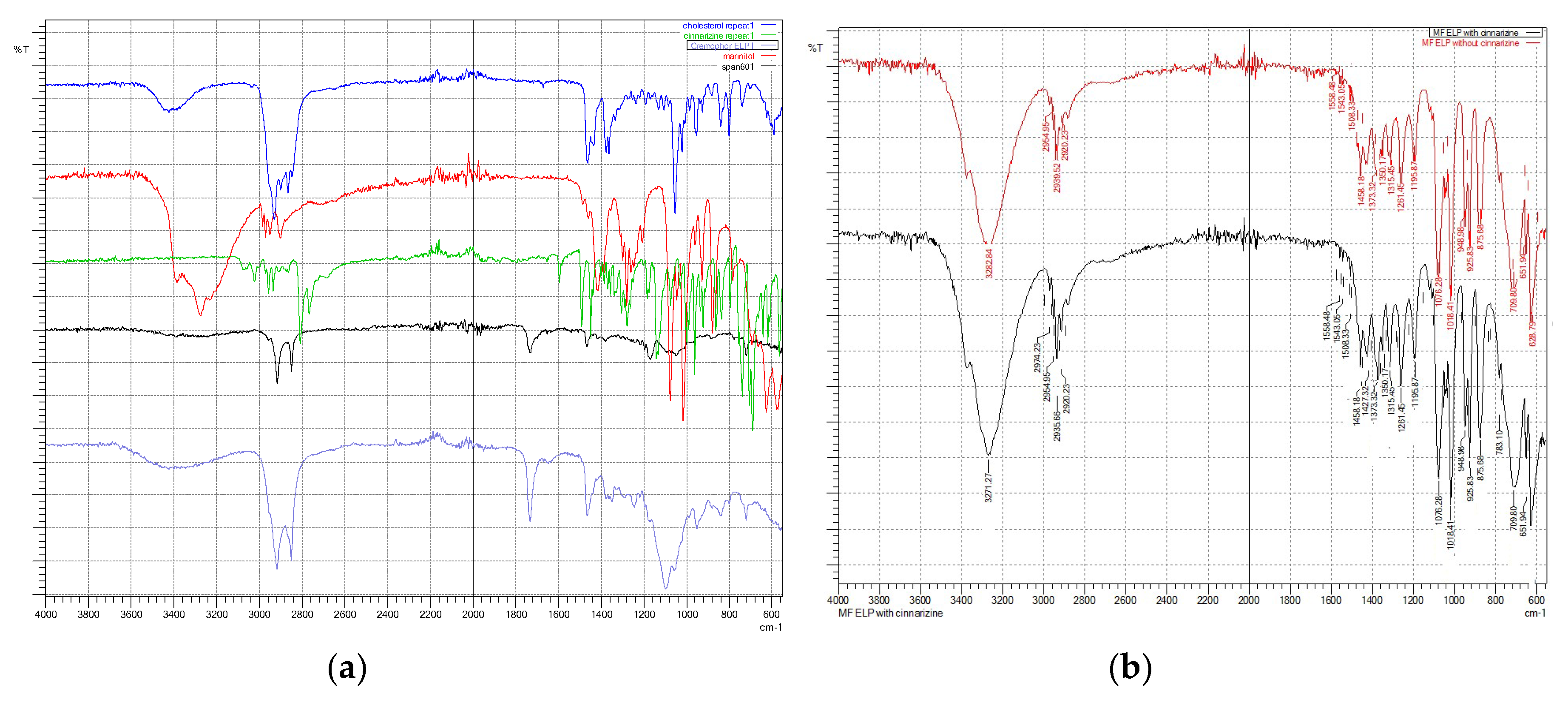
3.3. Fourier-Transform Infra-Red (FTIR) Spectroscopy
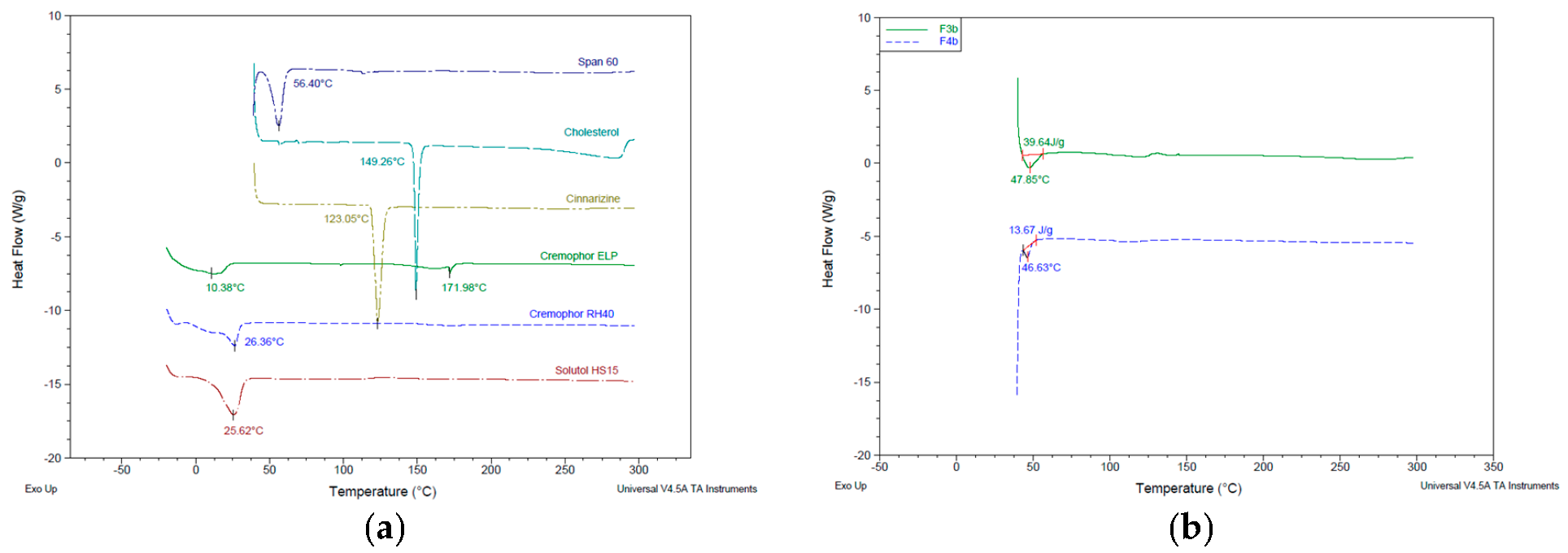
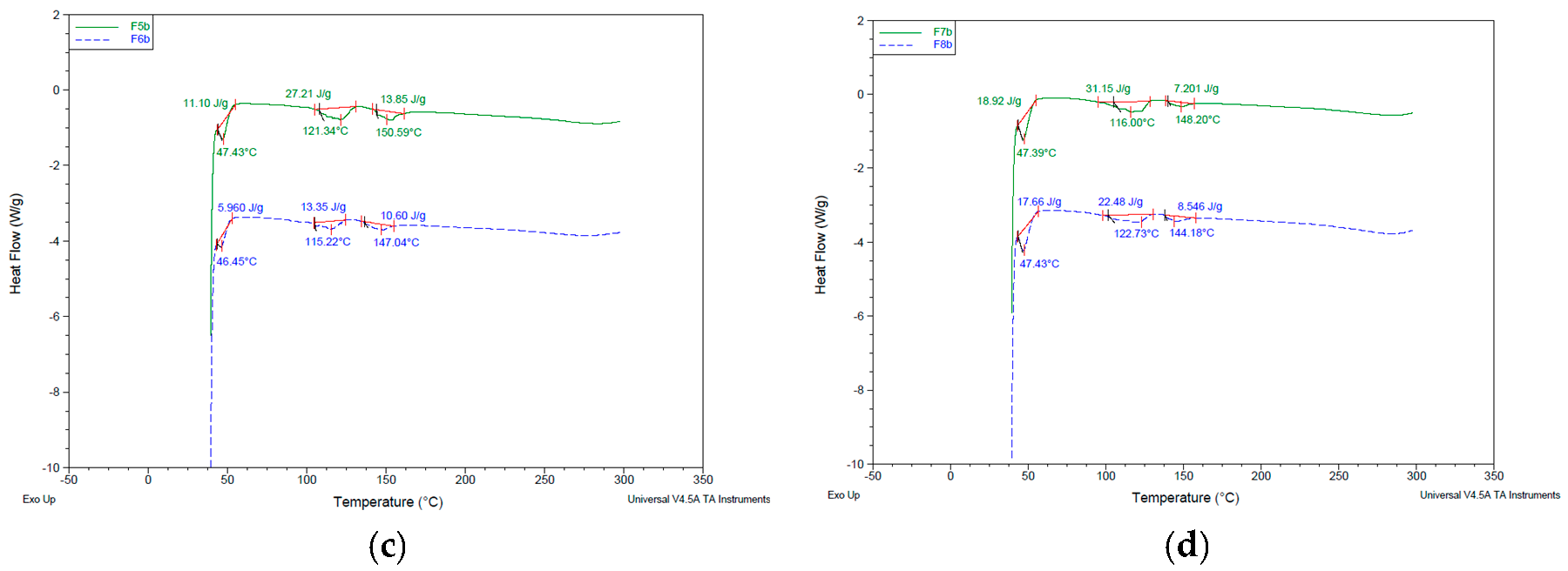
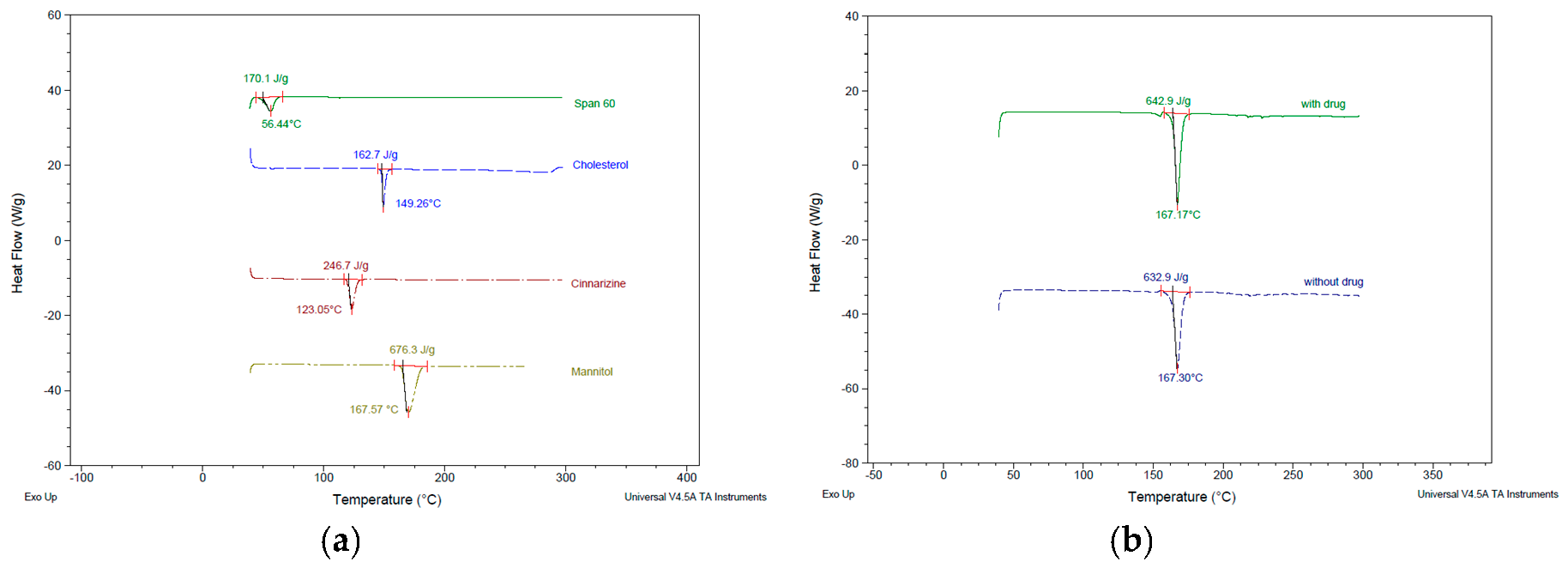
3.4. Thermal Characteristics of Niosomes
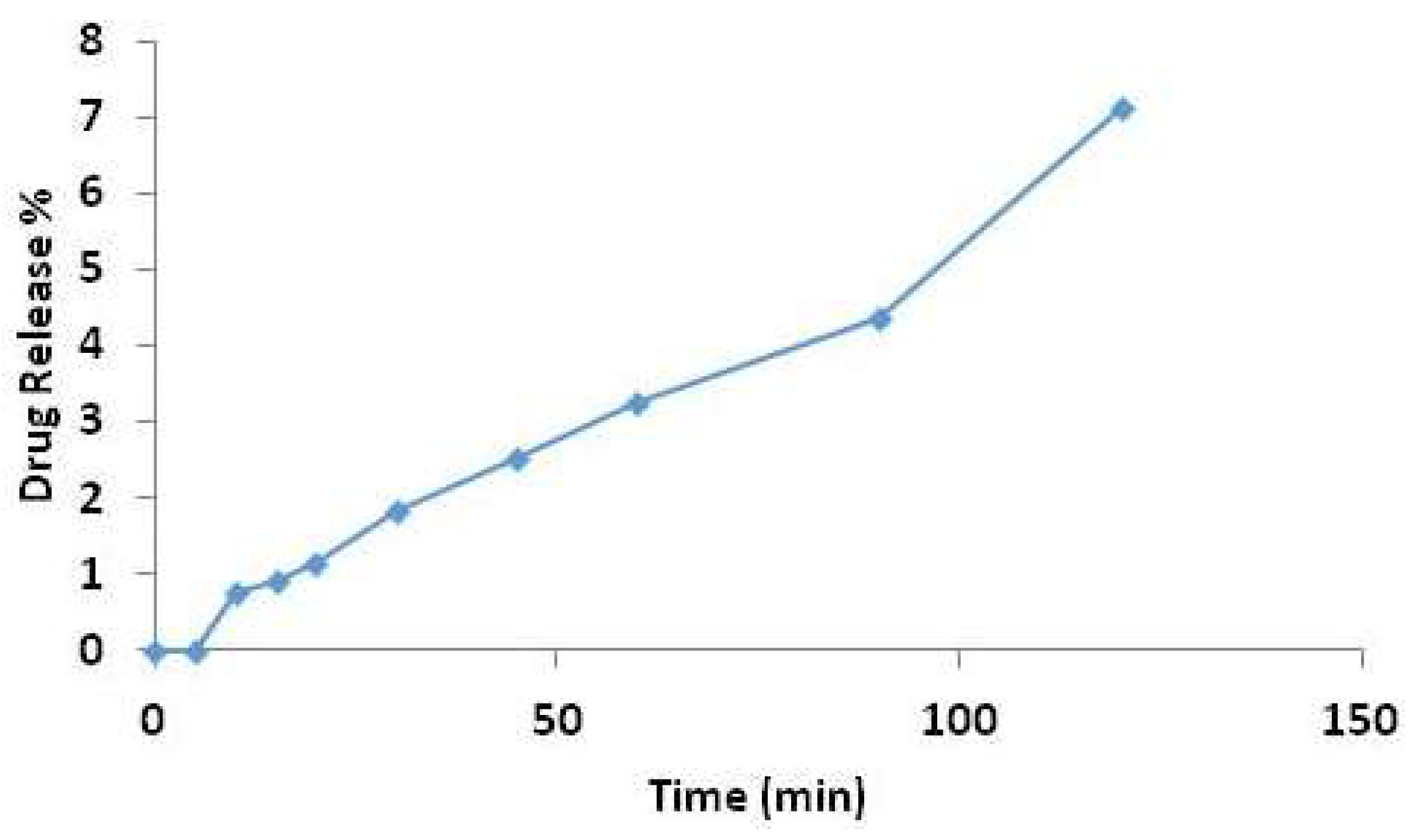
3.5. Drug Loading and Drug Release Studies
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Mahale, N.B.; Thakkar, P.D.; Mali, R.G.; Walunj, D.R.; Chaudhari, S.R. Niosomes: Novel sustained release nonionic stable vesicular systems—An overview. Adv. Colloid. Interface Sci. 2012, 183–184, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Bayindir, Z.S.; Yuksel, N. Characterization of niosomes prepared with various nonionic surfactants for paclitaxel oral delivery. J. Pharm. Sci. 2010, 99, 2049–2060. [Google Scholar] [CrossRef] [PubMed]
- Di Marzio, L.; Esposito, S.; Rinaldi, F.; Marianecci, C.; Carafa, M. Polysorbate 20 vesicles as oral delivery system: In vitro characterization. Colloid. Surface. B-Biointerfaces 2013, 104, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Wilkhu, J.S.; McNeil, S.E.; Anderson, D.E.; Kirchmeier, M.; Perrie, Y. Development of a solid dosage platform for the oral delivery of bilayer vesicles. Eur. J. Pharm. Sci. 2017, 108, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Gurrapu, A.; Jukanti, R.; Bobbala, S.R.; Kanuganti, S.; Jeevana, J.B. Improved oral delivery of valsartan from maltodextrin based proniosome powders. Adv. Powder Technol. 2012, 23, 583–590. [Google Scholar] [CrossRef]
- Bayindir, Z.S.; Antep, M.N.; Yuksel, N. Development and characterization of mixed niosomes for oral delivery using candesartan cilexetil as a model poorly water-soluble drug. AAPS PharmSciTech 2015, 16, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Bini, K.B.; Akhilesh, D.; Prabhakara, P.; Kamath, J.V. Development and characterization of non-ionic surfactant vesicles (niosomes) for oral delivery of lornoxicam. Int. J. Drug Devel Res. 2012, 4, 147–154. [Google Scholar]
- Khan, M.I.; Madni, A.; Peltonen, L. Development and in-vitro characterization of sorbitan monolaurate and poloxamer 184 based niosomes for oral delivery of diacerein. Eur. J. Pharm. Sci. 2016, 95, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Jadon, P.S.; Gajbhiye, R.S.; Jadon, R.S.; Gajbhiye, K.R.; Ganesh, N. Enhanced Oral Bioavailability of Griseofulvin via Niosomes. AAPS PharmSciTech 2009, 10, 1186–1192. [Google Scholar] [CrossRef] [PubMed]
- Mokhtar, M.; Sammour, O.A.; Hammad, M.A.; Megrab, N.A. Effect of formulation parameters on flurbiprofen encapsulation and release rates of niosomes prepared from proniosomes. Int. J. Pharm. 2008, 361, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.; Sammour, O.; El-Ghamry, H.; Abu-Selem, M. Preparation and in-vitro evaluation of diclofenac sodium niosomal formulations. Int. J. Pharm. Sci. Res. 2013, 4, 1757–1765. [Google Scholar]
- Kumar, G.P.; Rajeshwarrao, P. Nonionic surfactant vesicular systems for effective drug delivery—An overview. Acta Pharm. Sin. B 2011, 1, 208–219. [Google Scholar]
- Hao, Y.; Zhao, F.; Li, N.; Yang, Y.; Li, K. Studies on a high encapsulation of colchicine by a niosome system. Int. J. Pharm. 2002, 244, 73–80. [Google Scholar] [CrossRef]
- Nasseri, B. Effect of cholesterol and temperature on the elastic properties of niosomal membranes. Int. J. Pharm. 2005, 300, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Moghassemi, S.; Hadjizadeh, A. Nano-niosomes as nanoscale drug delivery systems: An illustrated review. J. Control. Release 2014, 185, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.H.; Moghaddam, B.; Kirby, D.J.; Mohammed, A.R.; Perrie, Y. The role of lipid geometry in designing liposomes for the solubilisation of poorly water soluble drugs. Int. J. Pharm. 2013, 453, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Alani, A.W.G.; Rao, D.A.; Seidel, R.; Wang, J.; Jiao, J.; Kwon, G.S. The effect of novel surfactants and Solutol HS15 on paclitaxel aqueous solubility an permeability across a Caco-2 monolayer. J. Pharm. Sci. 2010, 99, 3473–3485. [Google Scholar] [CrossRef] [PubMed]
- Marianecci, C.; Di Marzio, L.; Rinaldi, F.; Celia, C.; Paolino, D.; Alhaique, F.; Esposito, S.; Carafa, M. Niosomes from 80s to present: The state of the art. Adv. Colloid. Interface Sci. 2014, 205, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Correia, G.M.; Briuglia, M.L.; Niosi, F.; Lamprou, D.A. Microfluidic manufacturing of phospholipid nanoparticles: Stability, encapsulation efficacy, and drug release. Int. J. Pharm. 2017, 516, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Haress, N.G. Cinnarizine: Comprehensive profile. Profiles Drug Subst. Excipients Relat. Method. 2015, 40, 1–41. [Google Scholar]
- Berlin, M.; Przyklenk, K.H.; Richtberg, A.; Baumann, W.; Dressman, J.B. Prediction of oral absorption of cinnarizine—A highly supersaturating poorly soluble weak base with borderline permeability. Eur. J. Pharm. Biopharm. 2014, 88, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Nowacka-Krukowska, H.; Rakowska, M.; Neubart, K.; Kobylinska, M. High-performance liquid chromatographic assay for cinnarizine in human plasma. Acta Pol Pharm. 2007, 64, 407–411. [Google Scholar] [PubMed]
- Sanson, C.; Schatz, C.; Meins, J.F.L.; Soum, A.; Thevenot, J.; Garanger, E.; Lecommandoux, S. A simple method to achieve high doxorubicin loading in biodegradable polymersomes. J. Control. Release 2010, 147, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Kazi, K.M.; Mandal, A.S.; Biswas, N.; Guha, A.; Chatterjee, S.; Behera, M.; Kuotsu, K. Niosome: A future of targeted drug delivery systems. J. Adv. Pharm. Technol. Res. 2010, 1, 374–380. [Google Scholar]
- Yoshioka, T.; Sternberg, B.; Florence, A.T. Preparation and properties of vesicles (niosomes) of sorbitan monoesters (Span 20, 40, 60, and 80) and a sorbitan trimester (Span 85). Int. J. Pharm. 1994, 105, 1–6. [Google Scholar] [CrossRef]
- Inform white paper. Malvern Instruments Limited. Dynamic Light Scattering Common terms defined. 2011. Available online: http://www.biophysics.bioc.cam.ac.uk/wp-content/uploads/2011/02/DLS_Terms_defined_Malvern.pdf (accessed on 10 October 2018).
- Taymouri, S.; Varshosaz, J. Effects of different types of surfactants on the physical properties and stability of carvedilol nano-niosomes. Adv. Biomed. Res. 2016, 5, 48. [Google Scholar] [PubMed]
- Biswal, S.; Murthy, P.N.; Sahu, J.; Sahoo, P.; Amir, F. Vesicles of non-ionic surfactants (niosomes) and drug delivery potential. Int. J. Pharm. Sci. Nanotechnol. 2008, 1, 1–8. [Google Scholar]
- Akhter, S.; Kushwaha, S.; Warsi, M.H.; Anwar, M.; Ahmad, M.Z.; Ahmad, I.; Talegaonkar, S.; Khan, Z.I.; Khar, R.K.; Ahmad, F.J. Development and evaluation of nanosized niosomal dispersion for oral delivery of Ganciclovir. Drug Dev. Ind. Pharm. 2012, 38, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Nikolakakis, I.; Panagopoulou, A.; Salis, A.; Malamataris, S. Relationships between the properties of self-emulsifying pellets and of the emulsions used as massing liquids for their preparation. AAPS PharmSciTech 2015, 16, 129–139. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Obeid, M.A.; Khadra, I.; Mullen, A.B.; Tate, R.J.; Ferro, V.A. The effects of hydration media on the characteristics of non-ionic surfactant vesicles (NISV) prepared by microfluidics. Int. J. Pharm. 2017, 516, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, P.R.; Haseth, J.A.D.; Winefordner, J.D. In Fourier Transform Infrared Spectrometry, 2nd ed.Wiley: Hoboken, NJ, USA, 2017; pp. 1–18. [Google Scholar]
- Smith, E.A.; Dea, P.K. Differential scanning calorimetry studies of phospholipid membranes: The interdigitated gel phase. In Applications of Calorimetry in a Wide Context: Differential Scanning Calorimetry, Isothermal Titration Calorimetry and Microcalorimetry; Elkordy, A.A., Ed.; IntechOpen: London, UK, 2013; pp. 407–444. [Google Scholar]
| Formulation Code | Composition (mg) | % Molar Ratio | Cinnarizine | Total Lipid Content (mg) |
|---|---|---|---|---|
| F1 | Span® 60: cholesterol 105.4:94.6 | 50:50 | − | 200 |
| F2 | Span® 60: cholesterol 105.4:94.6 | 50:50 | + | 200 |
| F3 | Span® 60: cholesterol: Cremophor® ELP 73.0:65.6:61.4 | 45:45:10 | − | 200 |
| F4 | Span® 60: cholesterol: Cremophor® ELP 73.0:65.6:61.4 | 45:45:10 | + | 200 |
| F5 | Span® 60: cholesterol: Cremophor® RH40 61.6:55.2:83.2 | 45:45:10 | − | 200 |
| F6 | Span® 60: cholesterol: Cremophor® RH40 61.6:55.2:83.2 | 45:45:10 | + | 200 |
| F7 | Span® 60: cholesterol: Solutol® HS15 81.8:73.2:45.0 | 45:45:10 | − | 200 |
| F8 | Span® 60: cholesterol: Solutol® HS15 81.8:73.2:45.0 | 45:45:10 | + | 200 |
| Time (min) | %A | %B |
|---|---|---|
| 0 | 70 | 30 |
| 0.5 | 70 | 30 |
| 3 | 10 | 90 |
| 3.5 | 10 | 90 |
| 3.6 | 70 | 30 |
| 5 | 70 | 30 |
| Formulation | z-Average (nm) | Polydispersity Index (PDI) | Zeta Potential (mV) |
|---|---|---|---|
| S60:Cho − | * | * | * |
| S60:Cho + | * | * | * |
| S60:Cho:ELP − | 1256.3 ± 59.9 | 0.759 ± 0.110 | ** |
| S60:Cho:ELP + | 3701.0 ± 414.6 | 0.518 ± 0.074 | −1.82 ± 6.78 |
| S60:Cho:RH40 − | 827.5 ± 147.8 | 0.759 ± 0.128 | −0.063 ± 18.5 |
| S60:Cho:RH40 + | 1244.0 ± 314.9 | 0.714 ± 0.106 | 0.039 ± 18.1 |
| S60:Cho:HS15 − | 5330.3 ± 1348.2 | 0.262 ± 0.223 | −0.199 ± 20.1 |
| S60:Cho:HS15 + | 7320.7 ± 675.2 | 0.933 ± 0.115 | 0.118 ± 16.5 |
| Formulation | z-Average (nm) | PDI | ZP (mV) |
|---|---|---|---|
| S60:Cho − | 155.6 ± 0.4 | 0.059 ± 0.011 | −0.474 ± 9.7 |
| S60:Cho + | 217.9 ± 0.9 | 0.053 ± 0.009 | −1.266 ± 9.6 |
| S60:Cho:ELP − | 350.3 ± 7.1 | 0.208 ± 0.012 | 5.272 ± 3.5 |
| S60:Cho:ELP + | 355.3 ± 1.5 | 0.202 ± 0.021 | −4.486 ± 5.3 |
| S60:Cho:RH40 − | 224.7 ± 2.2 | 0.103 ± 0.019 | −3.921 ± 4.3 |
| S60:Cho:RH40 + | 172.2 ± 0.4 | 0.209 ± 0.007 | −1.803 ± 4.8 |
| S60:Cho:HS15 − | 310.8 ± 0.6 | 0.032 ± 0.051 | −4.801 ± 3.6 |
| S60:Cho:HS15 + | 303.7 ± 1.9 | 0.011 ± 0.010 | −0.764 ± 14.3 |
| Frequency (cm−1) | Assignments |
|---|---|
| 2959 | CH stretching (aromatic, alkene, mono-substituted) |
| 2936 | CH stretch (aliphatic, alkane) |
| 1597 | C=C (aromatic stretch) |
| 1490, 1448 | CH2 (alkane) |
| 1134 | C-N stretching |
| 999, 962 | =C-H out of plane (out-of-plane) (aromatic, alkene) |
| Formulation | %EE a | %EE b |
|---|---|---|
| S60:Cho:ELP + | 8.9 | 19.5 |
| S60:Cho:RH40 + | 0.37 | 10.8 |
| S60:Cho:HS15 + | 0.53 | 8.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeo, L.K.; Olusanya, T.O.B.; Chaw, C.S.; Elkordy, A.A. Brief Effect of a Small Hydrophobic Drug (Cinnarizine) on the Physicochemical Characterisation of Niosomes Produced by Thin-Film Hydration and Microfluidic Methods. Pharmaceutics 2018, 10, 185. https://doi.org/10.3390/pharmaceutics10040185
Yeo LK, Olusanya TOB, Chaw CS, Elkordy AA. Brief Effect of a Small Hydrophobic Drug (Cinnarizine) on the Physicochemical Characterisation of Niosomes Produced by Thin-Film Hydration and Microfluidic Methods. Pharmaceutics. 2018; 10(4):185. https://doi.org/10.3390/pharmaceutics10040185
Chicago/Turabian StyleYeo, Li Key, Temidayo O. B. Olusanya, Cheng Shu Chaw, and Amal Ali Elkordy. 2018. "Brief Effect of a Small Hydrophobic Drug (Cinnarizine) on the Physicochemical Characterisation of Niosomes Produced by Thin-Film Hydration and Microfluidic Methods" Pharmaceutics 10, no. 4: 185. https://doi.org/10.3390/pharmaceutics10040185
APA StyleYeo, L. K., Olusanya, T. O. B., Chaw, C. S., & Elkordy, A. A. (2018). Brief Effect of a Small Hydrophobic Drug (Cinnarizine) on the Physicochemical Characterisation of Niosomes Produced by Thin-Film Hydration and Microfluidic Methods. Pharmaceutics, 10(4), 185. https://doi.org/10.3390/pharmaceutics10040185







