Clinical Relevance of Total HIV DNA in Peripheral Blood Mononuclear Cell Compartments as a Biomarker of HIV-Associated Neurocognitive Disorders (HAND)
Abstract
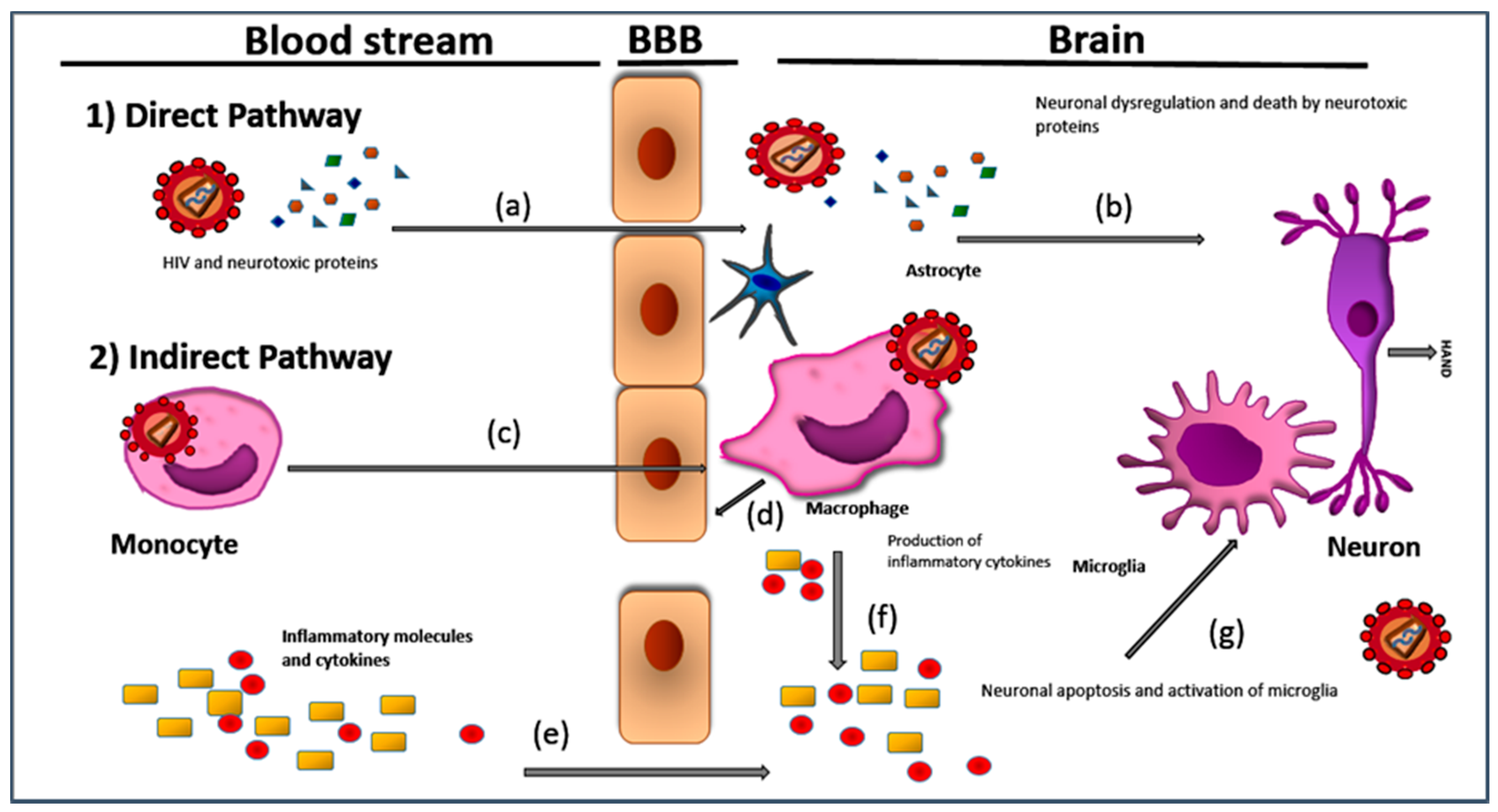
1. Introduction and Brief Pathology of HAND
2. Latency and Different Forms of HIV DNA in the Periphery
3. What Samples Can Be Used to Measure HIV DNA as a Biomarker for HAND?
3.1. Unfractionated PBMC
3.2. Lymphocytes
3.3. Monocytes
4. Quantitative PCR Assays to Detect Total HIV-1 DNA
5. Relevance of Total HIV DNA qPCR Assays to HAND
6. HIV-1 DNA qPCR Used in HAND Studies
7. Possible Use of Digital PCR to Detect HIV DNA in HAND
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Elbirt, D.; Mahlab-Guri, K.; Bezel-Rosenberg, S.; Gill, H.; Attali, M.; Asher, L. HIV-associated neurocognitive disorders. IMAJI 2015, 17, 54–58. [Google Scholar]
- Sanmarti, M.; Ibáñez, L.; Huertas, S.; Badenes, D.; Dalmau, D.; Slevin, M.; Krupinski, J.; Popa-Wagner, A.; Jaen, A. HIV-associated neurocognitive disorders. JMP 2014, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Simioni, S.; Cavassini, M.; Annoni, J.M.; Rimbault Abraham, A.; Bourquin, I.; Schiffer, V.; Calmy, A.; Chave, J.P.; Giacobini, E.; Hirschel, B.; et al. Cognitive dysfunction in HIV patients despite long-standing suppression of viremia. AIDS 2010, 24, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Rao, V.R.; Ruiz, A.P.; Prasad, V.R. Viral and cellular factors underlying neuropathogenesis in HIV associated neurocognitive disorders (HAND). AIDS Res. Ther. 2014, 19, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Antinori, A.; Arendt, G.; Becker, J.T.; Byrd, D.A.; Cherner, M.; Clifford, D.B.; Cinque, P.; Epstein, L.G.; Goodkin, K.; Gisslen, M.; et al. Updated research nosology for HIV-associated neurocognitive disorders. Neurology 2007, 69, 1789–1799. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Banks, W.A. Role of the immune system in HIV-associated neuroinflammation and neurocognitive implications. Brain Behav. Immun. 2015, 45, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Saksena, N.K. HIV Associated Neurocognitive Disorders. Infect. Dis. Rep. 2013, 5, s1e8. [Google Scholar] [CrossRef] [PubMed]
- Kovalevich, J.; Langford, D. Neuronal toxicity in HIV CNS disease. Future Virol. 2012, 7, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Appay, V.; Sauce, D. Immune activation and inflammation in HIV-1 infection: Causes and consequences. J. Pathol. 2008, 214, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kamat, A.; Lyons, J.L.; Misra, V.; Uno, H.; Morgello, S.; Singer, E.J.; Gabuzda, D. Monocyte activation markers in cerebrospinal fluid associated with impaired neurocognitive testing in advanced HIV infection. J. Acquir. Immune Defic. Syndr. 2012, 60, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Valcour, V.G.; Ananworanich, J.; Agsalda, M.; Sailasuta, N.; Chalermchai, T.; Schuetz, A.; Shikuma, C.; Liang, C.Y.; Jirajariyavej, S.; Sithinamsuwan, P.; et al. HIV DNA Reservoir Increases Risk for Cognitive Disorders in cART-Naïve Patients. PLoS ONE 2013, 8, e70164. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J.; Reynolds, S.; Cox, C.; Miller, E.N.; Sinsheimer, J.S.; Becker, J.T.; Martin, E.; Sacktor, N. Neuropsychology Working Group of the Multicenter AIDS Cohort Study. The longitudinal and interactive effects of HIV status, stimulant use, and host genotype upon neurocognitive functioning. J. Neurovirol. 2014, 3, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Colon, K.; Perez-Laspiur, J.; Quiles, R.; Rodriguez, Y.; Wajna, V.; Shaffer, S.A.; Leszyk, J.; Slolasky, R.L., Jr.; Melendel, L.M. Macrophage secretome from women with HIV-associated neurocognitive disorders. Proteom. Clin. 2016, 10, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Carroll, A.; Brew, B. HIV-associated neurocognitive disorders: Recent advances in pathogenesis, biomarkers, and treatment. F1000Research 2017, 6, 312. [Google Scholar] [CrossRef] [PubMed]
- McGuire, J.L.; Gill, A.L.; Douglas, S.D.; Kolson, D.; CHARTER Group. Central and peripheral markers of peripheral neurodegradation and monocyte activation in HIV-associated neurocognitive disorders. J. Neurovirol. 2015, 21, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Brew, B.J.; Bhalla, R.B.; Paul, M.; Gallardo, H.; McArthur, J.C.; Schwartz, M.K.; Price, R.W. Cerebrospinal fluid neopterin in human immunodeficiency virus type 1 infection. Ann. Neurol. 1990, 28, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Burdo, T.H.; Soulas, C.; Orzechowski, K.; Button, J.; Krishnan, A.; Sugimoto, C.; Alvarez, X.; Kuroda, M.J.; Williams, K.C. Increased monocyte turnover from bone marrow correlates with severity of SIV encephalitis and CD163 levels in plasma. PLoS Pathog. 2010, 6, e1000842. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Meyerhoff, D.J.; Price, R.W.; Peterson, J.; Lee, E.; Young, A.C.; Walter, R.; Fuchs, D.; Brew, B.J.; Cinque, P.; et al. Cerebrospinal fluid and neuroimaging biomarker abnormalities suggest early neurological injury in a subset of individuals during primary HIV infection. J. Infect. Dis. 2013, 207, 1703–1712. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.T.; Kingsley, L.; Mullen, J.; Cohen, B.; Martin, E.; Miller, E.N.; Ragin, A.; Sacktor, N.; Selnes, O.A.; Visscher, B.R. Multicenter AIDS Cohort Study. Vascular risk factors, HIV serostatus, and cognitive dysfunction in gay and bisexual men. Neurology 2009, 73, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.; McArthur, J.; Siliciano, R.F. Reservoirs for HIV-1: Mechanisms for viral persistence in the presence of antiviral immune responses and antiretroviral therapy. Annu. Rev. Immunol. 2000, 18, 665–708. [Google Scholar] [CrossRef] [PubMed]
- Olivares, O.; Pernas, M.; Casado, C.; Lopez-Galindez, C. Human Immunodeficiency Virus Type 1 two-long terminal repeat circles: A subject of debate. AIDS Rev. 2016, 18, 23–31. [Google Scholar] [PubMed]
- Koelsch, K.K.; Liu, L.; Haubrich, R.; May, S.; Havlir, D.; Gunthard, H.F.; Ignacio, C.C.; Campos-Soto, P.; Little, S.J.; Shafer, R.; Robbins, G.K.; D’Aquila, R.T.; et al. Dynamics of total, linear nonintegrated, and integrated HIV-1 DNA in vivo and in vitro. J. Infect. Dis. 2008, 197, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Van Lint, C.; Bouchat, S.; Marcello, A. HIV-1 transcription and latency: An update. Retrovirology 2013, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Valcour, V.G.; Shiramizu, B.T.; Cecilia, M.; Shikuma, C.M. HIV DNA in circulating monocytes as a mechanism to dementia and other HIV complications. J. Leukoc. Biol. 2010, 87, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Cockerham, L.R.; Deeks, S.G. Biomarker reveals HIV’s hidden reservoir. eLife 2014, 3, e04742. [Google Scholar] [CrossRef] [PubMed]
- Re, M.C.; Vitone, F.; Biagetti, C.; Schiavone, P.; Alessandrini, F.; Bon, I.; de Crignis, E.; Gibellini, D. HIV-1 DNA proviral load in treated and untreated HIV-1 seropositive patients. Clin. Microbiol. Infect. 2010, 6, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Gibellini, D.; Borderi, M.; De Crignis, E.; Cicola, R.; Cimatti, L.; Vitone, F.; Chiodo, F.; Re, M.C. HIV-1 DNA in peripheral blood monocytes and lymphocytes from naïve and HAART-treated indviduals. J. Infect. 2008, 56, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Yukl, S.A.; Shergill, A.K.; Ho, T.; Killian, M.; Girling, V.; Epling, L.; Li, P.; Wong, L.K.; Crouch, P.; Deeks, S.G.; et al. Distribution of HIV DNA and RNA in cell subsets differs in gut and blood of HIV-positive patients on ART: Implications for viral persistence. J. Infect. Dis. 2013, 208, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Chun, T.W.; Nickle, D.C.; Justement, J.S.; Meyers, J.H.; Roby, G.; Hallahan, C.W.; Kottilil, S.; Moir, S.; Mican, J.M.; Mullins, J.I.; et al. Persistence of HIV in gut associated lymphoid tissue despite long-term antiviral therapy. J. Infect. Dis. 2008, 197, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Rouzioux, C.; Richman, D. How to best measure HIV reservoirs. Curr. Opin. HIV AIDS 2013, 8, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Cysique, L.A.; Hey-Cunningham, W.J.; Dermody, N.; Chan, P.; Brew, B.J.; Koelsch, K.K. Peripheral Blood Mononuclear Cells HIV DNA Levels Impact Intermittently on Neurocognition. PLoS ONE 2015, 10, e0120488. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Conceicao, V.; Gupta, P.; Saksena, N.K. Why are the neurodegenerative disease-related pathways overrepresented in primary HIV-infected peripheral blood mononuclear cells: A genome-wide perspective. Virol. J. 2012, 9, 308. [Google Scholar] [CrossRef] [PubMed]
- Casabianca, A.; Gori, C.; Orlandi, C.; Forbici, F.; Federico Perno, C.; Magnani, M. Fast and sensitive quantitative detection of HIV DNA in whole blood leucocytes by SYBR green I real-time PCR assay. Mol. Cell. Probes 2007, 21, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Avettand-Fenoel, V.; Chaix, M.L.; Blanche, S.; Burgard, M.; Floch, C.; Toure, K.; Allemon, M.C.; Warszawski, J.; Rouzioux, C. LTR real-time PCR for HIV-1 DNA quantification in blood cells for early diagnosis in infants born to seropositive mothers treated in HAART area (ANRS CO 01). J. Med. Virol. 2009, 81, 217–223. [Google Scholar] [PubMed]
- Ganter, P.; Mélard, A.; Damond, F.; Delaugerre, C.; Dina, J.; Gueudin, M.; Maillard, A.; Sauné, K.; Rodallec, A.; Tuaillon, E.; et al. Nterlaboratory quality control of total HIV-1 DNA load measurement for multicenter reservoir studies. J. Med. Virol. 2017, 89, 2047–2050. [Google Scholar] [CrossRef] [PubMed]
- Casabianca, A.; Orlandi, C.; Canovari, B.; Scotti, M.; Acetoso, M.; Valentini, M.; Petrelli, E.; Magnani, M. A real time PCR platform for the simultaneous quantification of total and extrachromosomal HIV DNA Forms in Blood of HIV-1 Infected patients. PLoS ONE 2012, 9, e111919. [Google Scholar] [CrossRef] [PubMed]
- Mexas, A.M.; Graf, E.H.; Pace, M.J.; Yu, J.J.; Papasavvas, E.; Azzoni, L.; Busch, M.P.; Di Mascio, M.; Foulkes, A.S.; Migueles, S.A. Concurrent measures of total and integrated HIV DNA monitor reservoirs and ongoing replication in eradication trials. AIDS 2012, 26, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.; Dong, H.; Katzenstein, D.A.; Merigan, T.C. Quantification and comparison of proviral load in peripheral monuclear cells and isolated CD4+ T cells. J. Acquir. Immune Defic. Syndr. 1993, 6, 237–240. [Google Scholar] [PubMed]
- Katzenstein, T.L.; Oliveri, R.S.; Benfield, T.; Eugen-Olsen, J.; Nielsen, C.; Gerstoft, J.; Copenhagen AIDS Cohort Study Group. Cell-associated HIV DNA measured early during infection has prognostic value independent of serum HIV RNA measured concomitantly. Scand. J. Infect. Dis. 2002, 34, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Li, T. Factors associated with the size of HIV DNA reservoir. Chin. Med. J. 2017, 130, 224–230. [Google Scholar] [PubMed]
- Alidjinou, E.K.; Bocket, L.; Hober, D. Quantification of viral DNA during HIV-1infection: A review of relevant clinical uses and laboratory methods. Pathol. Biol. 2015, 63, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Jumare, J.; Sunshine, S.; Ahmed, H.; El-Kamary, S.S.; Magder, L.; Hungerford, L.; Burdo, T.; Eyzaguirre, L.M.; Umlauf, A.; Cherner, M.; et al. Peripheral blood lymphocyte HIV DNA levels correlate with HIV associated neurocognitive disorders in Nigeria. J. Neurovirol. 2017, 23, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Ellery, P.J.; Tippett, E.; Chiu, Y.L.; Paukovics, G.; Cameron, P.U.; Solomon, A.; Lewin, S.R.; Gorry, P.R.; Jaworowski, A.; Greene, W.C.; et al. The CD16+ monocyte subset is more permissive to infection and preferentially harbors HIV-1 in vivo. J. Immunol. 2007, 178, 6581–6589. [Google Scholar] [CrossRef] [PubMed]
- Pang, S.; Koyanagi, Y.; Miles, S.; Wiley, C.; Vinters, H.V.; Chen, I.S. High levels of unintegrated HIV-1 DNA in brain tissue of AIDS dementia patients. Nature 1990, 343, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Koncarevic, S.; Lößner, C.; Kuhn, K.; Prinz, T.; Pike, I.; Zucht, H. In-Depth Profiling of the Peripheral Blood Mononuclear Cells Proteome for Clinical Blood Proteomics. Int. J. Proteom. 2014. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H. Cellular and Molecular Immunology; Elsevier Science: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Mesko, B.; Poliska, S.; Nagy, L. Gene expression profiles in peripheral blood for the diagnosis of autoimmune diseases. Trends Mol. Med. 2011, 17, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Anderson, N.L.; Anderson, A.G. The human plasma proteome: History, character, and diagnostic prospects. Mol. Cell. Proteom. 2002, 11, 845–867. [Google Scholar] [CrossRef]
- Whitney, A.R.; Diehn, A.; Popper, S.J.; Alizadeh, A.A.; Boldrick, J.C.; Relman, D.A.; Brown, P.O. Individuality and variation in gene expression patterns in human blood. Proc. Natl. Acad. Sci. USA 2003, 100, 1896. [Google Scholar] [CrossRef] [PubMed]
- Mcdermott, J.; Giri, A.A.; Martini, I.; Bono, M.; Giacomini, M.; Campelli, A.; Tagliaferro, L.; Cara, A.; Varnier, O.E. Level of Human Immunodeficiency Virus DNA in peripheral Blood Mononuclear cells correlates with efficacy of Antiretroviral Therapy. J. Clin. Microbiol. 1999, 37, 2361–2365. [Google Scholar] [PubMed]
- De Oliveira, M.F.; Gianella, S.; Letendre, S.; Scheffler, K.; Pond, S.L.K.; Smith, D.M.; Strain, M.; Ellis, R.J. Comparative analysis of cell-associated HIV DNA levels in cerebrospinal fluid and peripheral blood by droplet digital PCR. PLoS ONE 2015. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, S.; Graf, E.H.; Dahl, C.; Strain, M.C.; Yukl, S.A.; Lysenko, E.S.; Bosch, R.J.; Lai, J.; Chioma, S.; Emad, F.; et al. Comparative analysis of measures of viral reservoirs in HIV eradication studies. PloS Pathog. 2013, 9, e1003174. [Google Scholar] [CrossRef] [PubMed]
- Valcour, V.; Sithinamsuwan, P.; Letendre, S.; Ances, B. Pathogenesis of HIV in the Central Nervous System. Curr. HIV/AIDS Rep. 2011, 8, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Kallianpur, K.J.; Kirk, G.R.; Sailasuta, N.; Valcour, V.; Shiramizu, B.; Nakamoto, B.K.; Shikuma, C. Regional cortical thinning associated with detectable levels of HIV DNA. Cereb. Cortex 2012, 22, 2065–2075. [Google Scholar] [CrossRef] [PubMed]
- Schnittman, S.M.; Psallidopoulos, M.C.; Lane, H.C.; Thompson, L.; Baseler, M.; Massari, F.; Fox, CH.; Salzman, N.P.; Fauci, A.S. The reservoir for HIV-1 in human peripheral blood is a T cell that maintains expression of CD4. Science 1989, 245, 305–308. [Google Scholar] [CrossRef] [PubMed]
- McBreen, S.; Imlach, S.; Shirafuji, T.; Scott, G.R.; Leen, C.; Bell, J.E.; Simmonds, P. Infection of the CD45RA1 (naive) Subset of peripheral CD81 lymphocytes by Human immunodeficiency virus type 1 in vivo. J. Virol. 2001, 75, 4091–4102. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.M.; Zaunders, J.J.; McBride, K.L.; Xu, Y.; Bailey, M.; Suzuki, K.; Cooper, D.A.; Emery, S.; Kelleher, A.D.; Koelsch, K.K.; et al. HIV DNA subspecies persist in both activated and resting memory CD4− T Cells during antiretroviral therapy. J. Virol. 2014, 88, 3516–3526. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Hill, B.J.; Ambrozak, D.R.; Price, D.A.; Guenaga, F.J.; Casazza, J.P.; Kuruppu, J.; Yazdani, J.; Migueles, S.A.; Connors, M.; et al. T-Cell Subsets that harbor Human Immunodeficiency Virus (HIV) In Vivo: Implications for HIV Pathogenesis. J. Virol. 2004, 78, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Douek, D.C. Disrupting T-cell homeostasis: How HIV-1 infection causes disease. AIDS Rev. 2003, 5, 172–177. [Google Scholar] [PubMed]
- Gabuzda, D.H.; Hess, J.L.; Small, J.A.; Clements, J.E. Regulation of the visna virus long terminal repeat in macrophages involves cellular factors that bind sequences containing AP-1 sites. Mol. Cell. Biol. 1989, 9, 2728–2733. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.H.; Sasseville, V.G.; Smith, M.O.; Vogel, P.; Pauley, D.R.; Heyes, M.P.; Lackner, A.A. Neuroinvasion by simian immunodeficiency virus coincides with increased numbers of perivascular macrophages/microglia and intrathecal immune activation. J. Neurovirol. 1996, 2, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Sasse, T.; Wu, J.; Zhou, L.; Saksena, K. Monocytes and their role in Human Immunodeficiency virus pathogenesis. Am. J. Infect. Dise. 2012, 8, 92–105. [Google Scholar]
- Jiang, X.; Zhang, L.; Liu, H.; Yuan, N.; Hou, P.; Zhang, R.; Wu, T. Expansion of CD14+CD16+ monocytes is related to acute leukemia. Int. J. Clin. Exp. Med. 2015, 8, 12297–12306. [Google Scholar] [PubMed]
- Fischer-Smith, T.; Bell, C.; Croul, S.; Lewis, M.; Rappaport, J. Monocyte/macrophage trafficking in acquired immunodeficiency syndrome encephalitis: Lessons from human and nonhuman primate studies. J. Neurovirol. 2008, 14, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Fischer-Smith, T.; Croul, S.; Sverstiuk, A.E.; Capini, C.; L’Heureux, D.; Regulier, E.G.; Richardson, M.W.; Amini, S.; Morgello, S.; Khalili, K.; et al. CNS invasion by CD14+CD16+ peripheral blood-derived monocytes in HIV dementia: Perivascular accumulation and reservoir of HIV infection. J. Virol. 2001, 7, 528–541. [Google Scholar]
- Innocenti, P.; Ottmann, M.; Morand, P.; Leclercq, P.; Seigneurin, J.M. HIV-1 in blood monocytes: Frequency of detection of proviral DNA using PCR and comparison with the total CD4 count. AIDS Res. Hum. Retrovir. 1992, 8, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.W.; Veenstra, M.; Gaskill, P.J.; Morgello, S.; Calderon, T.M.; Berman, J.W. Monocyte mediate HIV neuropathogenesis: Mechanisms that contribute to HIV associated neurocognitive disorders. Curr. HIV Res. 2014, 12, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Van Furth, R.; Cohn, Z.A. The origin and kinetics of mononuclear phagocytes. J. Exp. Med. 1968, 128, 415–435. [Google Scholar] [CrossRef] [PubMed]
- Sonza, S.; Maerz, A.; Deacon, N.; Meanger, J.; Millis, J.; Crowe, S. Human immunodeficiency virus type 1 is blocked prior to reverse transcription and integration in freshly isolated blood monocytes. J. Virol. 1996, 70, 3863–3869. [Google Scholar] [PubMed]
- McElrath, M.J.; Pruett, J.E.; Cohn, Z.A. Mononuclear phagocytes of blood and bone marrow: Comparative roles as viral reservoirs in human immunodeficiency virus type 1 infections. Proc. Natl. Acad. Sci. USA 1989, 86, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Muthui, D.; Holte, S.; Nickle, D.; Feng, F. Evidence for human immunodeficiency virus type 1 replication in vivo in CD14+ monocytes and its potential role as a source of virus in patients on highly active antiretroviral therapy. J. Virol. 2002, 76, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Lindl, K.A.; Marks, D.R.; Kolson, D.L.; Jordan-Sciutto, K.L. HIV—Associated neurocognitive disorder: Pathogenesis and therapeutic opportunities. J. Neuroimmune Pharmacol. 2010, 5, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Shiramizu, B.; Gartnerb, S.; Williamsa, A.; Shikumaa, C.; Ratto-Kima, S.; Wattersa, M.; Aguona, J.; Valcoura, V. Circulating proviral HIV DNA and HIV-associated dementia. AIDS 2005, 19, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Pulliam, L.; Gascon, R.; Stubblebine, M.; McGuire, D.; McGrath, M.S. Unique monocyte subset in patients with AIDS dementia. Lancet 1997, 349, 692–695. [Google Scholar] [CrossRef]
- Ryan, L.A.; Zheng, J.; Brester, M.; Bohac, D.; Hahn, F.; Anderson, J.; Ratanasuwan, W.; Gendelman, H.E.; Swindells, S. Plasma levels of soluble CD14 and tumor necrosis factor-alpha type II receptor correlate with cognitive dysfunction during human immunodeficiency virus type 1 infection. J. Infect. Dis. 2001, 184, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Rappaport, J.; Volsky, D.J. Role of the Macrophage in HIV-Associated Neurocognitive Disorders and Other Comorbidities in Patients on Effective Antiretroviral Treatment. J. Neurovirol. 2015, 21, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Scutari, R.; Alteri, C.; Perno, C.F.; Svicher, V.; Aquaro, S. The Role of HIV Infection in Neurologic Injury. Brain Sci. 2017, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Shiramizu, B.; Ratto-Kim, S.; Sithinamsuwan, P.; Nidhinandana, S.; Thitivichianlert, S.; Watt, G.; de Souza, M.; Chuenchitra, T.; Sukwit, S.; Chitpatima, S.; et al. HIV DNA and dementia in treatment-naive HIV-1-infected individuals in Bangkok, Thailand. Int. J. Med. Sci. 2006, 4, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Valcour, V.G.; Shiramizu, B.T.; Sithinamsuwan, P.; Nidhinandana, S.; Ratto-Kim, S.; Ananworanich, J.; Siangphoe, U.; Kim, J.H.; de Souza, M.; Degruttola, V.; et al. Age, apolipoprotein E4, and the risk of HIV dementia: The Hawaii Aging with HIV Cohort. J. Neuroimmunol. 2007, 157, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Shiramizu, B.; Williams, A.E.; Shikuma, C.; Valcour, V. Amount of HIV DNA in peripheral blood mononuclear cells is proportional to the severity of HIV-1-associated neurocognitive disorders. J. Neuropsychiatry Clin. Neurosci. 2009, 21, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Kallianpur, K.J.; Valcour, V.G.; Lerdlum, S.; Busovaca, E.; Agsalda, M.; Sithinamsuwan, P.; Chalermchai, T.; Fletcher, J.L.K.; Tipsuk, S.; Shikuma, C.M.; et al. HIV DNA in CD14+ reservoirs is associated with brain atrophy in cART-Naïve patients. AIDS 2014, 28, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Aga, E.; Cillo, A.R.; Yates, A.L.; Besson, G.; Fyne, E.; Koontz, D.L.; Jennings, C.; Zheng, L.; Mellorsa, J.W. Novel Assays for Measurement of Total Cell-Associated HIV-1 DNA and RNA. Novel assays for measurement of total cell-associated HIV-1 DNA and RNA. J. Clin. Microbiol. 2016, 54, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Bogh, M.; Machuca, R.; Gerstoft, J.; Pedersen, C.; Obel, N.; Kvinesdal, B.; Nielsen, H.; Nielsen, C. Subtype specific problems with the qualitative Amplicor HIV-1 DNA PCR test. J. Clin. Virol. 2001, 20, 149–153. [Google Scholar] [CrossRef]
- Vandesompele, J.; Kubista, M.; Pfaffl, M.W. Reference gene validation software for improved normalization. In Real-Time PCR: Current Technology and Applications; Logan, J., Edwards, K., Saunders, N., Eds.; Caister Academic Press: Poole, UK, 2009; pp. 47–64. [Google Scholar]
- Nixon, D.E.; Landay, A.L. Biomarkers of immune dysfunction in HIV. Curr. Opin. HIV AIDS 2010, 5, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Komninakis, S.V.; Domingos, E.M.; Santos, D.E.M.; Santos, C.; Oliveros, M.P.R.; Sanabani, S.; Diaza, R.S. HIV-1 Proviral DNA Loads (as Determined by Quantitative PCR) in patients subjected to structured treatment interruption after antiretroviral therapy failure. JCM 2012, 50, 2132–2133. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, Y.; Yamaguchi, H.; Einaga, N.; Esumi, M. Pitfalls of DNA Quantification Using DNA-Binding Fluorescent Dyes and Suggested Solutions. PLoS ONE 2016, 11, e0150528. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Livak, J.K.; Schmittgen, T. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Agsalda-Garcia, M.A.; Sithinamsuwan, P.; Valcour, V.G.; Chalermchai, T.; Tipsuk, S.; Kuroda, J.; Nakamura, C.; Ananworanich, J.; Zhang, G.; Schuetz, A.; et al. CD14+ Enriched Peripheral Cells Secrete Cytokines Unique to HIV-associated Neurocognitive Disorders. J. Acquir. Immune Defic. Syndr. 2017, 74, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Vandergeeten, C.; Fromentin, R.; Merlin, E.; Lowani, M.B.; DaFonsesca, S.; Bakeman, W.; McNulty, A.; Ramgopal, M.; Michael, N.; Kim, J.H.; et al. Cross-clade ultrasensitive PCR-based assays to measure HIV persistence in large cohort studies. J. Virol. 2014, 88, 12385–12396. [Google Scholar] [CrossRef] [PubMed]
- Malnati, M.S.; Scarlatti, G.; Gatto, F.; Salvatori, F.; Cassina, G.; Rutigliano, T.; Volpi, R.; Lusso, P. A universal real-time PCR assay for the quantification of group-M HIV-1 proviral load. Nat. Protoc. 2008, 3, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
- Rouzioux, C.; Hubert, J.B.; Burgard, M.; Deveau, C.; Goujard, C.; Bary, M.; Sereni, D.; Viard, J.P.; Delfraissy, J.F.; Meyer, L.; et al. Early levels of HIV-1 DNA in peripheral blood mononuclear cells are predictive of disease progression independently of HIV-1 RNA levels and CD4+ T cell counts. J. Infect. Dis. 2005, 192, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Minga, A.K.; Anglaret, X.; d’Aquin Toni, T.; Chaix, M.L.; Dohoun, L.; Abo, Y.; Coulibaly, A.; Duvignac, J.; Gabillard, D.; Rouet, F.; et al. HIV-1 DNA in peripheral blood mononuclear cells is strongly associated with HIV-1 disease progression in recently infected West African adults. J. Acquir. Immune Defic. Syndr. 2008, 48, 350–354. [Google Scholar] [PubMed]
- Suspe’ne, R.; Meyerhans, A. Quantification of unintegrated HIV-1 DNA at the single cell level in vivo. PLoS ONE 2012, 7, e36246. [Google Scholar] [CrossRef] [PubMed]
- Avettand-Fenoel, V.; Hocqueloux, L.; Ghosn, J.; Cheret, A.; Frange, P.; Melard, A.; Viard, J.-P.; Rouzioux, C. Total HIV DNA, a marker of viral reservoir dynamics with clinical implications. Clin. Microbiol. Rev. 2016, 29, 859–880. [Google Scholar] [CrossRef] [PubMed]
- Lillo, F.B.; Grasso, M.A.; Lorine, S.; Bellotti, M.G.; Colucci, G. Few modifications of the Cobus Amplicor HIV Monitor 1.5 test allow reliable quantitation of HIV-1 proviral load in peripheral blood mononuclear cells. J. Virol. Methods 2004, 120, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Pavsic, J.; Zel, J.; Milavec, M. Assessment of the realtime PCR and different digital PCR platforms for DNA quantification. Anal. Bioanal. Chem. 2016, 408, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Trypsteen, W.; Kiselinova, M.; Vandekerckhove, L.; De Spiegelaere, W. Diagnostic utility of digital PCR for HIV reservoir quantification. J. Virus Erad. 2016, 2, 162–169. [Google Scholar] [PubMed]
- Strain, M.C.; Lada, S.M.; Luong, T.; Rought, S.E.; Gianella, S.; Terry, V.H.; Spina, C.A. Highly precise measurement of HIV DNA by droplet digital PCR. PLoS ONE 2013, 8, e55943. [Google Scholar] [CrossRef] [PubMed]
- Bosman, K.J.; Nijhuis, M.; van Ham, P.M.; Wensing, A.J.M.; Vervisch, K.; Vandekerckhove, L.; De Spiegelaere, W. Comparison of digital PCR platforms and semi-nested qPCR as a tool to determine the size of the HIV reservoir. Sci. Rep. 2015, 5, 13811. [Google Scholar] [CrossRef] [PubMed]
| Assay | Normalisation | Unique Features and Advantages/Disadvantages | PCR Template Loading Quantity | LOD | Reference |
|---|---|---|---|---|---|
| Whole blood ANRS LTR real time PCR Method | HIV-DNA copies/µg converted to copies/million leucocytes | HIV DNA extracted from re-suspended whole blood cell pellets No separation of cells Its quicker and cost serving Normalisation using cell count/cytometry is not accurate | 1 µg DNA, equivalent to 150,000 cells | 1 Copy | [34] |
| LTR ANRS LTR real time PCR Method | Copies/million/PBMC | First assay for estimating HIV reservoir size to be evaluated for inter-laboratory reproducibility. The assay available as a commercial | 1 µg DNA, equivalent to 150,000 cells | 1 Copy | [34] |
| SYBR Green gag HIV DNA PCR | Copies/hundred thousand cells | Uses SYBR green fluorescence and amplifies a 142-gag fragment The assay is not sensitive especially in suppressed patients | 300 ng, equivalent to 1,000,000 cells | 50 Copies | [27] |
| Cross-Clade Ultrasensitive nested PCR | CD3 gene copy cell equivalents | Assay is performed on cell lysate, hence circumvents laborious nucleic acid extraction Allows quantification of HIV DNA A, B, C, D, CRF01_A/E, and CRF02_A/G HIV-1 subtypes, targeting highly conserved LTR-gag region. Cell input equivalence is accurately quantified by CD3 gene quantitative PCR. It has a disadvantage of 2 rounds of PCR | 100,000 Cells | 3 Copies | [91] |
| universal real-time PCR for group-M HIV-1 DNA | CCR5 gene copy cell equivalence | HIV-1 M-specific quantitative measures the HIV-1 Proviral DNA load in group M with a similar degree of sensitivity and accuracy across subtypes. Uses cell lysate as a template Predilution required if loading quantity is above 150,000 cells | 1 µg per reaction, equivalent to 150,000 cells | 1 Copy | [92] |
| Novel Assay for Total cell associated HIV-1 DNA | CCR5 gene copy cell equivalence | Sensitive Quantitative real time PCR for HIV-1 Cell associated DNA (CAD) targeting 3’ region of the pol gene. Assay involves enhanced nucleic extraction by ensuring adequate cell lysis through ultrasonic cell disruption | 1.7 µg | 3 Copies | [82] |
| Reference | Target | Sample | Clinical Data | Study Outcome | Advantage/Disadvantage |
|---|---|---|---|---|---|
| [73] | gag | PBMC | HAD patients on HAART 20–39 years and 50 years and above | Quantities of HIV DNA correlated with HAD HIV was higher in activated monocytes than CD14− in two patients | Very sensitive Quantitect Sybr Green PCR assay with detection limit of 1–3 copies and applicable suppressed patients and those with low cell PBMC or cell subset counts |
| [78] | gag | PBMC, CD14+, CD14− | 15 HAART-naïve HAD patients and 15 Non demented patients Average age, 34.1 | HIV DNA in PBMC was significantly higher in HAD non demented patients HIV DNA in five HAD patients was higher in CD14/CD16 than CD4 | Very sensitive Quantitect Sybr Green PCR assay with detection limit of 1–3 copies and can be applied to suppressed patients and samples with low cell count |
| [31] | pol | PBMC | HIV Subtype B chronic patients on cART for six months CD4+ < 350/mL Median Age 56, 74 | HIV DNA was associated with HAD and not Non demented forms of HAND | Very sensitive assay with one copy as LOD, hence suitable for supressed HAND patients PCR has a wide dynamic range from three copies to 300,000 copies High efficiencies for both HIV DNA and β-actin normaliser (93.7% and 86.6% respectively) -Assay restricted to detecting Subtype B |
| [42] | LTR-gag | CD14− and CD14+ | ART naïve participants (median age, 32) with 17 impaired and 19 unimpaired Patients had higher HIV DNA in lymphocytes than monocytes | Strong association between HIV DNA levels in lymphocytes and HAND | The target region for amplification is highly conserved and the Universal PCR assay has been customised for subtype G and CRF02_AG. However, performance of the assays not detailed |
| [79] | gag | CD14+ | 12 HAART treated HAD and 15 non HAD Infected with HIV-1 CRF01_AE Median Age, 32 | Baseline monocyte HIV DNA correlated with HIV DNA and 48 weeks after HAART. Monocyte HIV DNA level was below detection limit in all non-HAD patients after 48 weeks | Detection limit of the assay was 10 copies/106 cells which is relatively high. |
| [11] | gag | CD14+PBMC | Treatment naïve with three clinical categories of HAND (ANI, MND, HAD) CD4+ <350/mL –CRF AE_01Mean Age, 35 | No correlation between PBMC HIV DNA Before CD14+ enrichment, CD14+ was associated with HAND(ANI, MND, HAD) –CD14+ HIV DNA was associated with glial dysfunction, neuronal injury and CSF immune activation (neopterin) | Very sensitive assay with a limit of detection of one copy which is suitable for HAND suppressed patients |
| [51] | pol | PBMC | 22–40 years and 50–71 years ART-supressed HIV subjects | Higher HIV DNA levels were associated with more severe neurocognitive impairment in older patients No association between HIV DNA and HAND in young adults. | Very sensitive droplet digital PCR with Limit of Detection (LOD) of one copy and more precise than qPCR due insensitivity to primer and probe mismatches absolute quantification, no external standards required |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruhanya, V.; Jacobs, G.B.; Glashoff, R.H.; Engelbrecht, S. Clinical Relevance of Total HIV DNA in Peripheral Blood Mononuclear Cell Compartments as a Biomarker of HIV-Associated Neurocognitive Disorders (HAND). Viruses 2017, 9, 324. https://doi.org/10.3390/v9110324
Ruhanya V, Jacobs GB, Glashoff RH, Engelbrecht S. Clinical Relevance of Total HIV DNA in Peripheral Blood Mononuclear Cell Compartments as a Biomarker of HIV-Associated Neurocognitive Disorders (HAND). Viruses. 2017; 9(11):324. https://doi.org/10.3390/v9110324
Chicago/Turabian StyleRuhanya, Vurayai, Graeme B. Jacobs, Richard H. Glashoff, and Susan Engelbrecht. 2017. "Clinical Relevance of Total HIV DNA in Peripheral Blood Mononuclear Cell Compartments as a Biomarker of HIV-Associated Neurocognitive Disorders (HAND)" Viruses 9, no. 11: 324. https://doi.org/10.3390/v9110324
APA StyleRuhanya, V., Jacobs, G. B., Glashoff, R. H., & Engelbrecht, S. (2017). Clinical Relevance of Total HIV DNA in Peripheral Blood Mononuclear Cell Compartments as a Biomarker of HIV-Associated Neurocognitive Disorders (HAND). Viruses, 9(11), 324. https://doi.org/10.3390/v9110324





