The Incredible Journey of Begomoviruses in Their Whitefly Vector
Abstract
1. Introduction to Begomoviruses, Whiteflies and Circulative Transmission
2. The CP: A Key to Begomovirus Translocation in B. tabaci
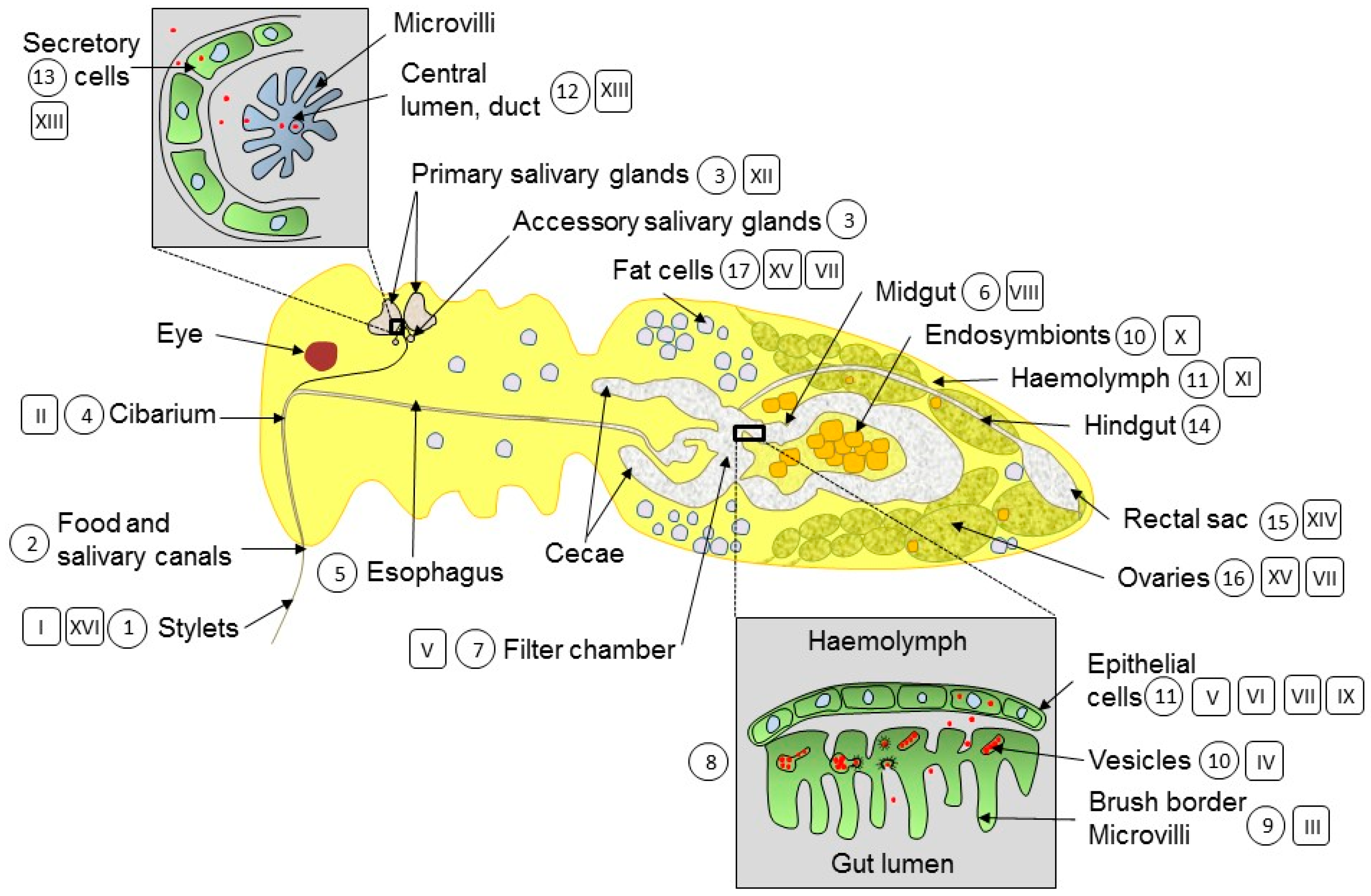
3. Organs, Tissues and Cells Visited by Begomoviruses Circulating in B. tabaci
3.1. Penetration of the Stylet into the Phloem of Infected Plants
3.1.1. The Plant Virus Source
3.1.2. Virus Ingurgitation
3.2. The Cibarium (Foregut) Discriminates between Non-Circulative and Circulative Viruses
3.3. Esophagus and Entry into the Guts
3.4. Guts, Midgut and Filter Chamber
3.4.1. Accumulation of Begomovirus in the Midgut and Filter Chamber
3.4.2. Transcription and Replication
3.4.3. Autophagy
3.4.4. Whitefly Proteins Interacting with Begomovirus Proteins in the Guts
3.4.5. Storage in the Digestive Tract
3.5. Gut-Haemolymph Barrier
3.6. Endosymbionts
3.7. Haemolymph
3.8. Primary Salivary Glands
3.9. Hindgut and Rectal Sac
3.10. Other Tissues: Fat Cells, Reproductive System
3.11. Egestion into the Plant Vascular System
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Czosnek, H. Tomato Yellow Leaf Curl Virus Disease: Management, Molecular Biology, Breeding for Resistance; Springer: Dordrecht, The Netherlands, 2007; pp. 1–420. [Google Scholar]
- Navot, N.; Pichersky, E.; Zeidan, M.; Zamir, D.; Czosnek, H. Tomato yellow leaf curl virus: A whitefly-transmitted geminivirus with a single genomic molecule. Virology 1991, 185, 151–161. [Google Scholar] [CrossRef]
- Díaz-Pendón, J.A.; Cañizares, M.C.; Moriones, E.; Bejarano, E.R.; Czosnek, H.; Navas-Castillo, J. Tomato yellow leaf curl viruses: Ménage à trois between the virus complex, the plant, and the whitefly vector. Mol. Plant Pathol. 2010, 11, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Scholthof, K.-B.G.; Adkins, S.; Czosnek, H.; Palukaitis, P.; Jacquot, E.; Hohn, T.; Hohn, B.; Saunders, K.; Candresse, T.; Ahlquist, P.; et al. Top 10 plant viruses in molecular plant pathology. Mol. Plant Pathol. 2011, 12, 938–954. [Google Scholar] [CrossRef] [PubMed]
- Attathom, S.; Chiemsomba, P.; Kositratana, W.; Sae-Ung, N. Complete nucleotide sequence and genome analysis of bipartite tomato yellow leaf curl virus in Thailand. Kasetsart J. Nat. Sci. 1994, 28, 632–639. [Google Scholar]
- Czosnek, H. Tomato Yellow Leaf Curl Virus. In Encyclopedia of Virology; Mahy, B.W.J., van Regenmortel, M., Eds.; Elsevier: Oxford, UK, 2008; Volume 5, pp. 138–145. [Google Scholar]
- Cui, X.; Tao, X.; Xie, Y.; Fauquet, C.M.; Zhou, X. A DNAβ associated with Tomato Yellow Leaf Curl China Virus is required for symptom induction. J. Virol. 2004, 78, 13966–13974. [Google Scholar] [CrossRef] [PubMed]
- Anfoka, G.; Haj Ahmad, F.; Altaleb, M.; Al Shhab, M. Detection of satellite DNA β in tomato plants with tomato yellow leaf curl disease in Jordan. Plant Dis. 2014, 98, 1017. [Google Scholar] [CrossRef]
- De Barro, P.J.; Liu, S.S.; Boykin, L.M.; Dinsdale, A.B. Bemisia tabaci: A statement of species status. Annu. Rev. Entomol. 2011, 56, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Park, J.; Le, G.-S.; Lee, S.; Akimoto, S. Taxonomic status of the Bemisia tabaci complex (Hemiptera: Aleyrodidae) and reassessment of the number of its constituent species. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Navas-Castillo, J.; Fiallo-Olive, E.; Sanchez-Campos, S. Emerging virus diseases transmitted by whiteflies. Annu. Rev. Phytopathol. 2011, 49, 219–248. [Google Scholar] [CrossRef] [PubMed]
- Rosell, R.C.; Torres-Jerez, I.; Brown, J.K. Tracing the geminivirus-whitefly transmission pathway by polymerase chain reaction in whitefly extracts, saliva, haemolymph, and honeydew. Phytopathology 1999, 89, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, M.; Morin, S.; Czosnek, H. Rate of Tomato yellow leaf curl virus (TYLCV) translocation in the circulative transmission pathway of its vector, the whitefly Bemisia tabaci. Phytopathology 2001, 91, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Czosnek, H.; Ghanim, M.; Ghanim, M. The circulative pathway of begomoviruses in the whitefly vector Bemisia tabaci-insights from studies with Tomato yellow leaf curl virus. Ann. Appl. Biol. 2002, 140, 215–231. [Google Scholar] [CrossRef]
- Luan, J.-B.; Li, J.-M.; Varela, N.; Wang, Y.-L.; Li, F.-F.; Bao, Y.-Y.; Zhang, C.-X.; Liu, S.-S.; Wang, X.-W. Global analysis of the transcriptional response of whitefly to Tomato yellow leaf curl China virus reveals the relationship of coevolved adaptations. J. Virol. 2011, 85, 3330–3340. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, M.; Czosnek, H. Acquisition of tomato yellow leaf curl virus by the whitefly Bemisia tabaci. J. Gen. Virol. 1991, 72, 2607–2614. [Google Scholar] [CrossRef] [PubMed]
- Caciagli, P.; Bosco, D. Quantitation over time of tomato yellow leaf curl geminivirus DNA in its whitefly vector. Phytopathology 1997, 87, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Becker, N.; Rimbaud, L.; Chiroleu, F.; Reynaud, B.; Thébaud, G.; Lett, J.M. Rapid accumulation and low degradation: Key parameters of Tomato yellow leaf curl virus persistence in its insect vector Bemisia tabaci. Sci. Rep. 2015, 5, 7696. [Google Scholar] [CrossRef] [PubMed]
- Ning, X.; Shi, X.; Liu, B.; Pan, H.; Wei, W.; Zeng, Y.; Sun, X.; Xie, W.; Wang, S.; Wu, Q.; et al. Transmission of Tomato yellow leaf curl virus by Bemisia tabaci as affected by whitefly sex and biotype. Sci. Rep. 2015, 5, 10744. [Google Scholar] [CrossRef] [PubMed]
- Polston, J.E.; Al-Musa, A.; Perring, T.M.; Dodds, J.A. Association of the nucleic acid of squash leaf curl geminivirus with the whitefly Bemisia tabaci. Phytopathology 1990, 80, 850–856. [Google Scholar] [CrossRef]
- Kollenberg, M.; Winter, S.; Götz, M. Quantification and localization of Watermelon chlorotic stunt virus and Tomato yellow leaf curl virus (Geminiviridae) in populations of Bemisia tabaci (Hemiptera, Aleyrodidae) with differential virus transmission characteristics. PLoS ONE 2014, 9, e111968. [Google Scholar] [CrossRef] [PubMed]
- Guo, T.; Guo, Q.; Cui, X.-Y.; Liu, Y.-Q.; Hu, J.; Liu, S.-H. Comparison of transmission of Papaya leaf curl China virus among four cryptic species of the whitefly Bemisia tabaci complex. Sci. Rep. 2015, 5, 15432. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Pan, H.; Liu, B.; Chu, D.; Xie, W.; Wu, Q.; Wang, S.; Xu, B.; Zhang, Y. Insect symbiont facilitates vector acquisition, retention, and transmission of plant virus. Sci. Rep. 2013, 3, 1367. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, G.; Czosnek, H. Long-term association of tomato yellow leaf curl virus with its whitefly vector Bemisia tabaci: Effect on the insect transmission capacity, longevity and fecundity. J. Gen. Virol. 1997, 78, 2683–2689. [Google Scholar] [CrossRef] [PubMed]
- Czosnek, H.; Ghanim, M.; Rubinstein, G.; Morin, S.; Fridman, V.; Zeidan, M. Whiteflies: Vectors– or victims? –of geminiviruses. Adv. Virus Res. 2001, 57, 291–322. [Google Scholar] [PubMed]
- Li, M.; Hu, J.; Xu, F.-C.; Liu, S.-S. Transmission of Tomato Yellow Leaf Curl Virus by two invasive biotypes and a Chinese indigenous biotype of the whitefly Bemisia tabaci. Int. J. Pest Manag. 2010, 56, 275–280. [Google Scholar] [CrossRef]
- Noris, E.; Vaira, A.M.; Caciagli, P.; Masenga, V.; Gronemborn, B.; Accotto, G.P. Amino acids in the capsid protein of Tomato yellow leaf curl virus that are crucial for systemic infection, particle formation, and insect transmission. J. Virololy 1998, 72, 10050–10057. [Google Scholar]
- Caciagli, P.; Medina, V.; Marian, D.; Vecchiati, M.; Masenga, V.; Falcioni, T.; Noris, E. Virion stability is important for the circulative transmission of Tomato yellow leaf curl Sardinia virus by Bemisia tabaci, but virion access to salivary glands does not guarantee transmissibility. J. Virol. 2009, 83, 5784–5795. [Google Scholar] [CrossRef] [PubMed]
- Höhnle, M.; Höfer, P.; Bedford, I.D.; Briddon, R.W.; Markham, P.G.; Frischmuth, T. Exchange of three amino acids in the coat protein results in efficient whitefly transmission of a nontransmissible Abutilon mosaic virus isolate. Virology 2001, 290, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhao, J.J.; Zhang, T.; Li, F.F.; Ghanim, M.; Zhou, X.P.; Ye, G.Y.; Liu, S.S.; Wang, X.W. Specific cells in the primary salivary glands of the whitefly Bemisia tabaci control retention and transmission of Begomoviruses. J. Virol. 2014, 88, 13460–13468. [Google Scholar] [CrossRef] [PubMed]
- Hofer, P.; Bedford, I.D.; Markham, P.G.; Jeske, H.; Frischmuth, T. Coat protein gene replacement results in whitefly transmission of an insect nontransmissible geminivirus isolate. Virology 1997, 236, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, M.; Morin, S.; Zeidan, M.; Czosnek, H. Evidence for transovarial transmission of Tomato yellow leaf curl virus by its vector the whitefly Bemisia tabaci. Virology 1998, 240, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Bosco, D.; Mason, G.; Accotto, G.P. TYLCSV DNA, but not infectivity, can be transovarially inherited by the progeny of the whitefly vector Bemisia tabaci (Gennadius). Virology 2004, 323, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; He, Y.-Z.; Guo, Q.; Guo, T.; Liu, Y.-Q.; Zhou, X.-P.; Liu, S.-S.; Wang, X.-W. Vector development and vitellogenin determine the transovarial transmission of begomoviruses. Proc. Natl. Acad. Sci. USA 2017, 114, 6746–6751. [Google Scholar] [CrossRef] [PubMed]
- Levy, A.; Czosnek, H. The DNA-B of the non-phloem limited Bean dwarf mosaic virus (BDMV) is able to move the phloem-limited Abutilon mosaic virus (AbMV) out of the phloem, but DNA-B of AbMV is unable to confine BDMV to the phloem. Plant Mol. Biol. 2003, 53, 789–803. [Google Scholar] [CrossRef] [PubMed]
- Ber, R.; Navot, N.; Zamir, D.; Antignus, Y.; Cohen, S.; Czosnek, H. Infection of tomato by the Tomato yellow leaf curl virus: Susceptibility to infection, symptom development and accumulation of viral DNA. Arch. Virol. 1990, 112, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Pollard, D.G. Feeding habits of the cotton whitefly. Ann. Appl. Biol. 1955, 43, 664–671. [Google Scholar] [CrossRef]
- Rosell, R.C.; Lichty, J.E.; Brown, J.K. Ultrastructure of the mouthparts of adult sweetpotato whitefly, Bemisia tabaci Gennadius (Homoptera: Aleyrodidae). Int. J. Insect Morphol. Embryol. 1995, 24, 297–306. [Google Scholar] [CrossRef]
- Morgan, J.K.; Luzio, G.A.; Ammar, E.-D.; Hunter, W.B.; Hall, D.G.; Shatters, R.G., Jr. Formation of stylet sheaths in āere (in air) from eight species of phytophagous hemipterans from six families (suborders: Auchenorrhyncha and Sternorrhyncha). PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Will, T.; Furch, A.C.U.; Zimmermann, M.R. How phloem-feeding insects face the challenge of phloem-located defenses. Front. Plant Sci. 2013, 4, 336. [Google Scholar] [CrossRef] [PubMed]
- Atzmon, G.; van Hoss, H.; Czosnek, H. PCR-amplification of tomato yellow leaf curl virus (TYLCV) from squashes of plants and insect vectors: Application to the study of TYLCV acquisition and transmission. Eur. J. Plant Pathol. 1998, 104, 189–194. [Google Scholar] [CrossRef]
- Jiang, Y.X.; de Blas, C.; Barrios, L.; Fereres, A. Correlation between whitefly (Homoptera: Aleyrodidae) feeding behavior and transmission of Tomato yellow leaf curl virus. Ann. Entomol. Soc. Am. 2000, 93, 573–579. [Google Scholar] [CrossRef]
- Hunter, W.B.; Hiebert, E.; Webb, S.E.; Polston, J.E.; Tsai, J.H. Precibarial and cibarial chemosensilla in the whitefly, Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae). Int. J. Insect Morphol. Embryol. 1996, 25, 295–304. [Google Scholar] [CrossRef]
- Ghanim, M.; Rosell, R.C.; Campbell, L.R.; Czosnek, H.; Brown, J.K.; Ullman, D.E. Digestive, salivary, and reproductive organs of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) B type. J. Morphol. 2001, 248, 22–40. [Google Scholar] [CrossRef] [PubMed]
- Orfanidou, C.G.; Pappi, P.G.; Efthimiou, K.E.; Katis, N.I.; Maliogka, V.I. Transmission of Tomato chlorosis virus (ToCV) by Bemisia tabaci Biotype Q and evaluation of four weed species as viral sources. Plant Dis. 2016, 100, 2043–2049. [Google Scholar] [CrossRef]
- Wisler, G.C.; Duffus, J.E.; Liu, H.Y.; Li, R.H. Ecology and epidemiology of whitefly-transmitted closteroviruses. Plant Dis. 1998, 82, 270–280. [Google Scholar] [CrossRef]
- Chen, A.Y.; Walker, G.P.; Carter, D.; Ng, J.C. A virus capsid component mediates virion retention and transmission by its insect vector. Proc. Natl. Acad. Sci. USA 2011, 108, 16777–16782. [Google Scholar] [CrossRef] [PubMed]
- Kiss, Z.A.; Medina, V.; Falk, B.W. Crinivirus replication and host interactions. Front. Microbiol. 2013, 4, 99. [Google Scholar] [CrossRef] [PubMed]
- Wintermantel, W.M.; Wisler, G.C.; Anchieta, A.G.; Liu, H.-Y.; Karasev, A.V.; Tzanetakis, I.E. The complete nucleotide sequence and genome organization of Tomato chlorosis virus. Arch. Virol. 2005, 150, 2287–2298. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Rubio, L.; Yeh, H.H.; Crawford, B.; Falk, B.W. Lettuce infectious yellows virus: In vitro acquisition analysis using partially purified virions and the whitefly Bemisia tabaci. J. Gen. Virol. 1999, 80, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Stewart, L.R.; Medina, V.; Tian, T.; Turina, M.; Falk, B.W.; Ng, J.C.K. A mutation in the Lettuce infectious yellows virus minor coat protein disrupts whitefly transmission but not in planta systemic movement. J. Virol. 2010, 84, 12165–12173. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Hasegawa, D.K.; Ling, K.-S.; Wintermantel, W.M. Application of genomics for understanding plant virus-insect vector interactions and insect vector control. Phytopathology 2016, 106, 1213–1222. [Google Scholar] [CrossRef] [PubMed]
- Cicero, J.M.; Brown, J.K. Functional anatomy of whitefly organs associated with Squash leaf curl virus (Geminiviridae: Begomovirus) transmission by the B biotype of Bemisia tabaci (Hemiptera: Aleyrodidae). Ann. Entomol. Soc. Am. 2011, 104, 261–279. [Google Scholar] [CrossRef]
- Wang, L.L.; Wei, X.M.; Ye, X.D.; Xu, H.X.; Zhou, X.P.; Lu, S.S.; Wang, X.W. Expression and functional characterisation of a soluble form of Tomato yellow leaf curl virus coat protein. Pest Manag. Sci. 2014, 70, 1624–1631. [Google Scholar] [CrossRef] [PubMed]
- Uchibori, M.; Hirata, A.; Suzuki, M.; Ugaki, M. Tomato yellow leaf curl virus accumulates in vesicle-like structures in descending and ascending midgut epithelial cells of the vector whitefly, Bemisia tabaci, but not in those of nonvector whitefly Trialeurodes vaporariorum. J. Gen. Plant Pathol. 2013, 79, 115–122. [Google Scholar] [CrossRef]
- Medina, V.; Pinner, M.S.; Bedford, I.D.; Achon, M.A.; Gemeno, C.; Markham, P.G. Immunolocalization of Tomato yellow leaf curl Sardinia virus in natural host plants and its vector Bemisia tabaci. J. Plant Pathol. 2006, 88, 299–308. [Google Scholar]
- Götz, M.; Popovski, S.; Kollenberg, M.; Gorovits, R.; Brown, J.K.; Cicero, J.; Czosnek, H.; Winter, S.; Ghanim, M. Implication of Bemisia tabaci heat shock protein in Begomovirus-whitefly interactions. J. Virol. 2012, 84, 13241–13252. [Google Scholar] [CrossRef] [PubMed]
- Kanakala, S.; Ghanim, M. Implication of the whitefly Bemisia tabaci cyclophilin B protein in the transmission of Tomato yellow leaf curl virus. Front. Plant Sci. 2016, 7, 1702. [Google Scholar] [CrossRef] [PubMed]
- Pakkianathan, B.C.; Kontsedalov, S.; Lebedev, G.; Mahadav, A.; Zeidan, M.; Czosnek, H.; Ghanim, M. Replication of Tomato yellow leaf curl virus in its whitefly vector, Bemisia tabaci. J. Virol. 2015, 89, 9791–9803. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Wang, X.R.; Wei, X.M.; Huang, H.; Wu, J.X.; Chen, X.X.; Liu, S.S.; Wang, X.W. The autophagy pathway participates in resistance to Tomato yellow leaf curl virus infection in whiteflies. Autophagy 2016, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, J.; Kitamura, T.; Terami, F.; Honda, K. A selective barrier in the midgut epithelial cell membrane of the nonvector whitefly trialeurodes vaporariorum to Tomato yellow leaf curl virus uptake. J. Gen. Plant Pathol. 2009, 75, 131–139. [Google Scholar] [CrossRef]
- Sánchez-Campos, S.; Rodríguez-Negrete, E.A.; Cruzado, L.; Grande-Pérez, A.; Bejarano, E.R.; Navas-Castillo, J.; Moriones, E. Tomato yellow leaf curl virus: No evidence for replication in the insect vector Bemisia tabaci. Sci. Rep. 2016, 6, 30942. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.K.; Iwasaki, A. Autophagy and antiviral immunity. Curr. Opin. Immunol. 2009, 20, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Haxim, Y.; Ismayil, A.; Jia, Q.; Wang, Y.; Zheng, X.; Chen, T.; Qian, L.; Liu, N.; Wang, Y.; Han, S.; et al. Autophagy functions as an antiviral mechanism against geminiviruses in plants. eLife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Miozzi, L.; Napoli, C.; Sardo, L.; Accotto, G.P. Transcriptomics of the interaction between the monopartite phloem-limited geminivirus Tomato yellow leaf curl Sardinia virus and Solanum lycopersicum highlights a role for plant hormones, autophagy and plant immune system fine tuning during infection. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Gorovits, R.; Fridman, L.; Kolot, M.; Rotem, O.; Ghanim, M.; Shriki, O.; Czosnek, H. Tomato yellow leaf curl virus confronts host degradation by sheltering in small/midsized protein aggregates. Virus Res. 2016, 213, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Gorovits, R.; Moshe, A.; Kolot, M.; Sobol, I.; Czosnek, H. Progressive aggregation of Tomato yellow leaf curl virus coat protein in systemically infected tomato plants, susceptible and resistant to the virus. Virus Res. 2013, 171, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Rana, V.P.; Popli, S.; Saurav, G.K.; Raina, H.S.; Chaubey, R.; Ramamurthy, V.V.; Rajagopal, R.A. Bemisia tabaci midgut protein interacts with begomoviruses and plays a role in virus transmission. Cell. Microbiol. 2016, 18, 663–678. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-Z.; Shi, M.; Huang, Y.-C.; Wang, X.-W.; Stanley, D.; Chen, X.-X. A peptidoglycan recognition protein acts in whitefly (Bemisia tabaci) immunity and involves in Begomovirus acquisition. Sci. Rep. 2016, 6, 37806. [Google Scholar] [CrossRef] [PubMed]
- Hafrén, A.; Macia, J.-L.; Love, A.J.; Milner, J.J.; Drucker, M.; Hofius, D. Selective autophagy limits cauliflower mosaic virus infection by NBR1-mediated targeting of viral capsid protein and particles. Proc. Natl. Acad. Sci. USA 2017, 114, 2026–2035. [Google Scholar] [CrossRef] [PubMed]
- Mercer, J.; Schelhaas, M.; Helenius, A. Virus entry by endocytosis. Annu. Rev. Biochem. 2010, 79, 803–833. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.-L.; Chen, Q.-F.; Zhao, J.-J.; Guo, T.; Wang, X.-W.; Hariton-Shalev, A.; Czosnek, H.; Liu, S.-S. Clathrin-mediated endocytosis is involved in Tomato yellow leaf curl virus transport across the midgut barrier of its whitefly vector. Virology 2017, 502, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Baumann, P.; Munson, M.A.; Lai, C.-Y.; Clark, M.A.; Baumann, L.; Moran, N.A.; Campbell, B.C. Origin and properties of bacterial endosymbionts of aphids, whiteflies and mealybugs. ASM News 1993, 59, 21–24. [Google Scholar]
- Chiel, E.; Gottlieb, Y.; Zchori-Fein, E.; Mozes-Daube, N.; Katzir, N.; Inbar, M.; Ghanim, M. Biotype-dependent secondary symbiont communities in sympatric populations of Bemisia tabaci. Bull. Entomol. Res. 2007, 97, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, Y.; Ghanim, M.; Gueguen, G.; Kontsedalov, S.; Vavre, F.; Fleury, F.; Zchori-Fein, E. Inherited intracellular ecosystem: Symbiotic bacteria share bacteriocytes in whiteflies. FASEB J. 2008, 22, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Brumin, M.; Levy, M.; Ghanim, M. Transovarial transmission of Rickettsia spp. and organ-specific infection of the whitefly Bemisia tabaci. Appl. Environ. Microbiol. 2012, 78, 5565–5574. [Google Scholar] [CrossRef] [PubMed]
- Kliot, A.; Cilia, M.; Czosnek, H.; Ghanim, M. Implication of the bacterial endosymbiont Rickettsia spp. in the whitefly Bemisia tabaci interactions with Tomato yellow leaf curl virus. J. Virol. 2014, 88, 5652–5660. [Google Scholar] [CrossRef] [PubMed]
- Nation, J.L., Jr. Circulatory system. In Insect Physiology and Biochemistry, 3nd ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 405–432. [Google Scholar]
- Morin, S.; Ghanim, M.; Zeidan, M.; Czosnek, H.; Verbeek, M.; van den Heuvel, J.F. A GroEL homologue from endosymbiotic bacteria of the whitefly Bemisia tabaci is implicated in the circulative transmission of Tomato yellow leaf curl virus. Virology 1999, 30, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Morin, S.; Ghanim, M.; Sobol, I.; Czosnek, H. The GroEL protein of the whitefly Bemisia tabaci interacts with the coat protein of transmissible and non-transmissible begomoviruses in the yeast two-hybrid system. Virology 2000, 276, 404–416. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, Y.; Zchori-Fein, E.; Mozes-Daube, N.; Kontsedalov, S.; Skaljac, M.; Brumin, N.; Sobol, I.; Czosnek, H.; Vavre, F.; Fleury, F.; et al. The transmission efficiency of Tomato yellow leaf curl virus is correlated with the presence of a specific symbiotic bacterium species. J. Virol. 2010, 84, 9310–9317. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, J.F.J.M.; Verbeek, M.; van der Wilk, F. Endosymbiotic bacteria associated with circulative transmission of potato leafroll virus by Myzus persicae. J. Gen. Virol. 1994, 75, 2559–2565. [Google Scholar] [CrossRef] [PubMed]
- Rana, V.S.; Singh, S.T.; Priya, N.G.; Kumar, J.; Rajagopal, R. Arsenophonus GroEL interacts with CLCuV and is localized in midgut and salivary gland of whitefly B. tabaci. PLoS ONE 2012, 7, 42168. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.-L.; Li, J.-M.; Luan, J.-B.; Ye, X.-D.; Wang, X.-W.; Liu, S.-S. Transcriptome analysis of the salivary glands of an invasive whitefly. PLoS ONE 2012, 7, 239303. [Google Scholar]
- Chen, W.; Hasegawa, D.K.; Kaur, N.; Kliot, A.; Valle Pinheiro, P.; Luan, J.; Stensmyr, M.C.; Zheng, Y.; Liu, W.; Sun, H.; et al. The draft genome of whitefly Bemisia tabaci MEAM1, a global crop pest, provides novel insights into virus transmission, host adaptation, and insecticide resistance. BMC Biol. 2016, 14, 110. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Chen, C.; Yang, Z.; Guo, L.; Yang, X.; Wang, D.; Chen, M.; Huang, J.; Wen, Y.; Zeng, Y.; et al. Genome sequencing of the sweetpotato whitefly Bemisia tabaci MED/Q. Gigascience 2017, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Leshkowitz, D.; Gazit, S.; Reuveni, E.; Ghanim, M.; Czosnek, H.; McKenzie, C.; Shatters, R.G., Jr.; Brown, J.K. (Whitefly (Bemisia tabaci) genome project: Analysis of sequenced clones from egg, instar, and adult (viruliferous and non-viruliferous) cDNA libraries. BMC Genom. 2006, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Seal, S.; Patel, M.V.; Collins, C.; Colvin, J.; Bailey, D. Next generation sequencing and quantitative real-time PCR technologies for characterization of the Bemisia tabaci Asia 1 mtCOI phylogenetic clade. J. Integr. Agric. 2012, 11, 281–292. [Google Scholar] [CrossRef]
- Ye, X.-D.; Su, Y.-L.; Zhao, Q.-Y.; Xia, W.-Q.; Liu, S.-S.; Wang, X.-W. Transcriptomic analyses reveal the adaptive features and biological differences of guts from two invasive whitefly species. BMC Genom. 2014, 15, 370. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.W.; Luan, J.B.; Li, J.M.; Bao, Y.Y.; Zhang, C.X.; Liu, S.S. De novo characterization of a whitefly transcriptome and analysis of its gene expression during development. BMC Genom. 2010, 11, 400. [Google Scholar] [CrossRef] [PubMed]
- Hariton-Shalev, A.; Sobol, I.; Ghanim, M.; Liu, S.S.; Czosnek, H. The Whitefly Bemisia tabaci knottin-1 gene is implicated in regulating the quantity of Tomato yellow leaf curl virus ingested and transmitted by the insect. Viruses 2016, 8, 205. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czosnek, H.; Hariton-Shalev, A.; Sobol, I.; Gorovits, R.; Ghanim, M. The Incredible Journey of Begomoviruses in Their Whitefly Vector. Viruses 2017, 9, 273. https://doi.org/10.3390/v9100273
Czosnek H, Hariton-Shalev A, Sobol I, Gorovits R, Ghanim M. The Incredible Journey of Begomoviruses in Their Whitefly Vector. Viruses. 2017; 9(10):273. https://doi.org/10.3390/v9100273
Chicago/Turabian StyleCzosnek, Henryk, Aliza Hariton-Shalev, Iris Sobol, Rena Gorovits, and Murad Ghanim. 2017. "The Incredible Journey of Begomoviruses in Their Whitefly Vector" Viruses 9, no. 10: 273. https://doi.org/10.3390/v9100273
APA StyleCzosnek, H., Hariton-Shalev, A., Sobol, I., Gorovits, R., & Ghanim, M. (2017). The Incredible Journey of Begomoviruses in Their Whitefly Vector. Viruses, 9(10), 273. https://doi.org/10.3390/v9100273





